Antioxidant Activities of Total Phenols of Prunella vulgaris L. in Vitro and in Tumor-bearing Mice
Abstract
:Abbreviations
| (PV) | Prunella vulgaris L. |
| (ABTS˙+) | 2,2’-azino-di(3-ethylbenzthiazoline- 6-sulfonic acid |
| (DPPH) | 2,2-diphenyl-1-picrylhydrazyl |
| (FRAP) | ferric reducing antioxidant power |
| (SOD) | superoxide dismutase |
| (MDA) | malondialdehyde |
| (ROS) | reactive oxygen species |
| (CTX) | cyclophosphamide |
| (GSH) | glutathione |
| (LPO) | lipid peroxidation |
| (CAT) | catalase |
| (HPLC) | High performance liquid chromatography |
| (DAD) | diode array detector |
| (SD) | standard deviation |
1. Introduction
2. Results and Discussion
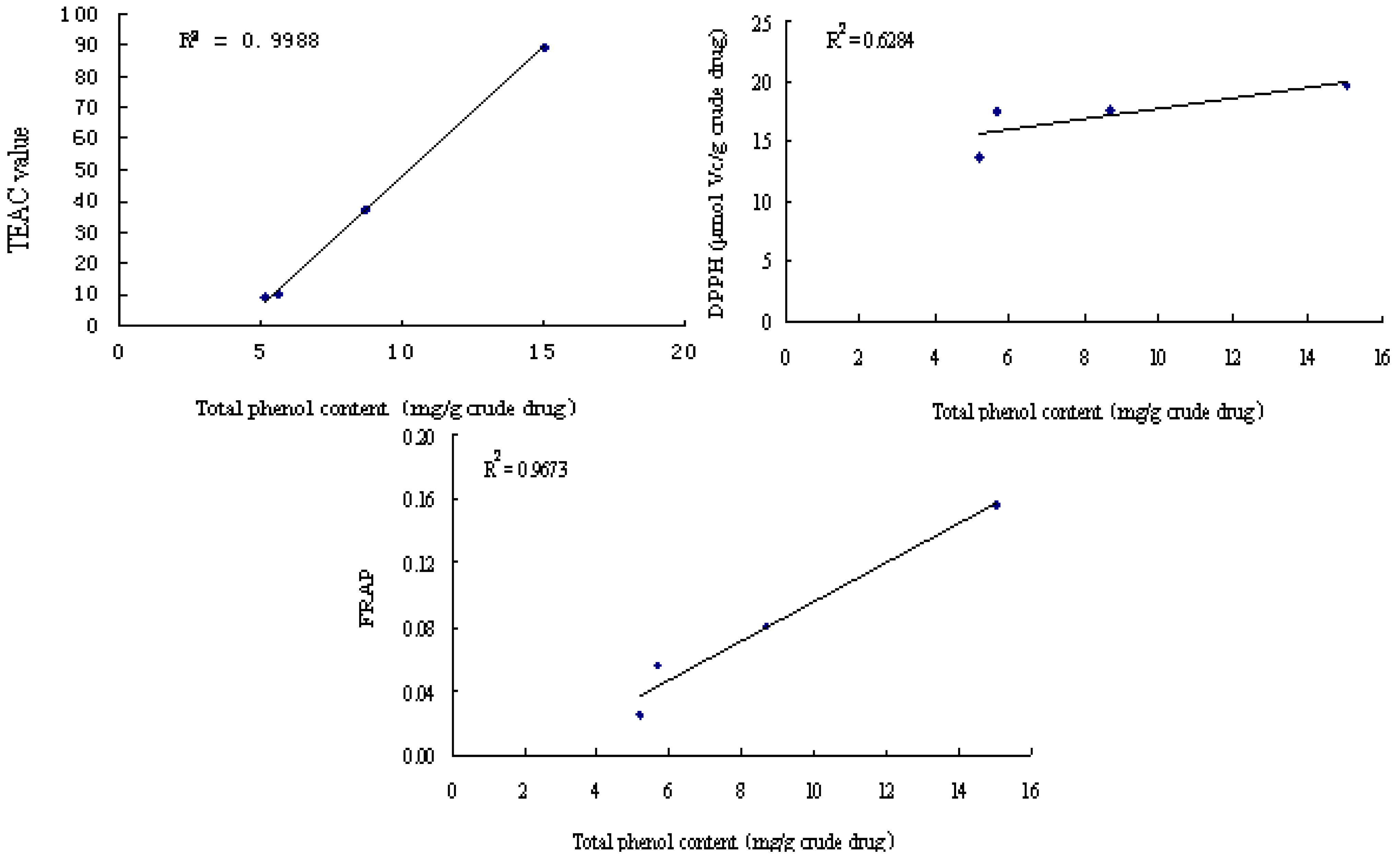
2.1. Comparison on antioxidant activity of different PV extract fractions
| Extracts | TEAC | DPPH value | FRAP value |
|---|---|---|---|
| (μmol Trolox/g crude drug) | (μmol Vc/g crude drug) | (μmol Fe(II)/g crude drug) | |
| P-95 | 8.926 | 13.710 | 0.0252 |
| P-60 | 89.307 | 19.662 | 0.1567 |
| P-30 | 37.335 | 17.645 | 0.0802 |
| P-w | 9.779 | 17.536 | 0.0558 |
2.2. HPLC analysis for antioxidant compounds
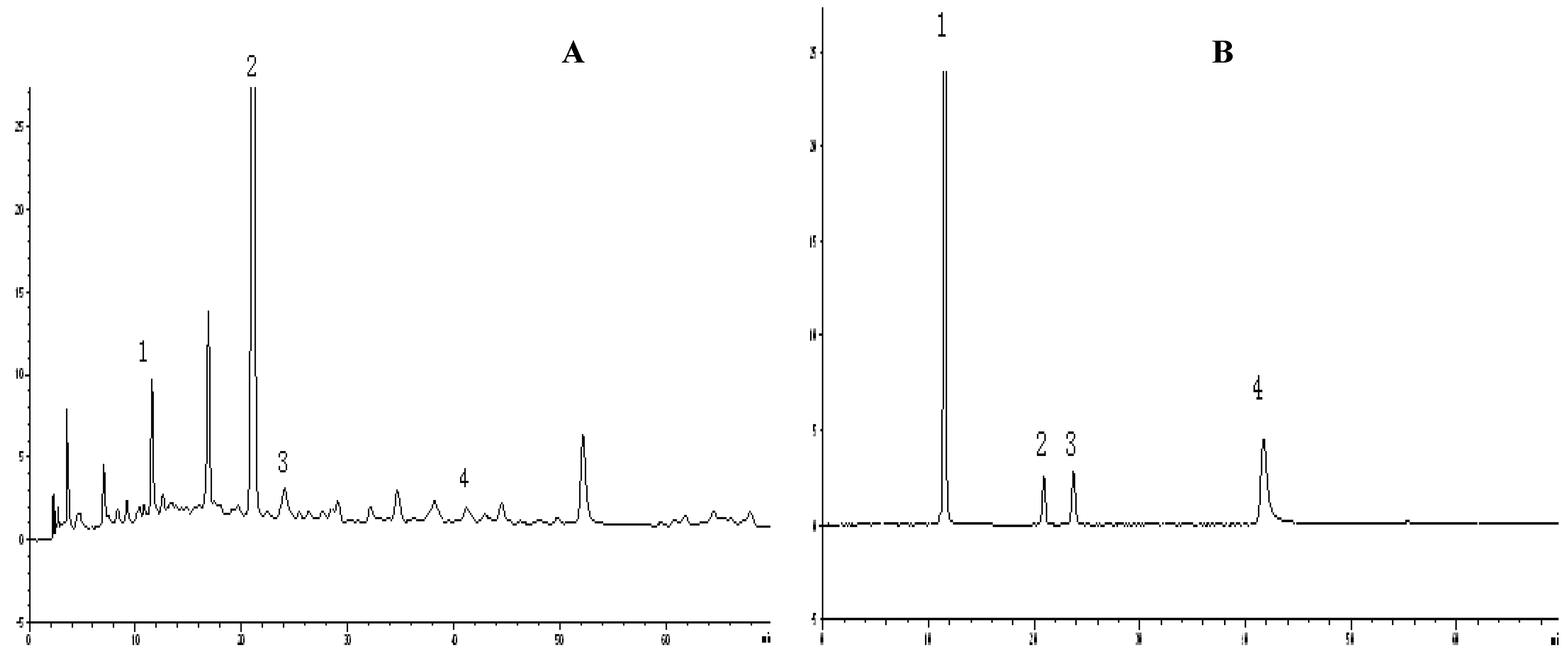

2.3. The correlation between total phenols content and antioxidant activity
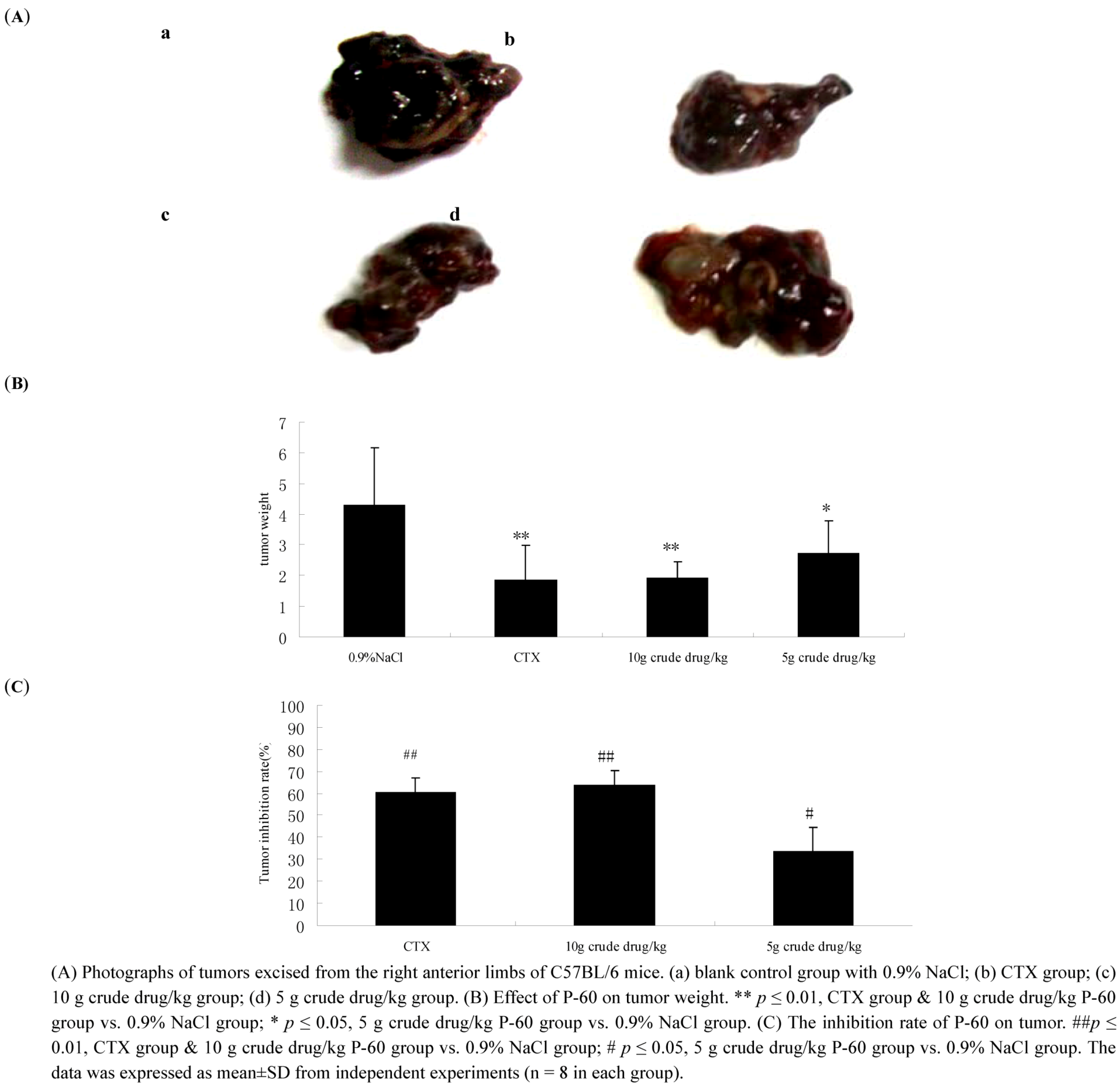
2.4. Effect of P-60 on tumor growth in C57BL/6 mice
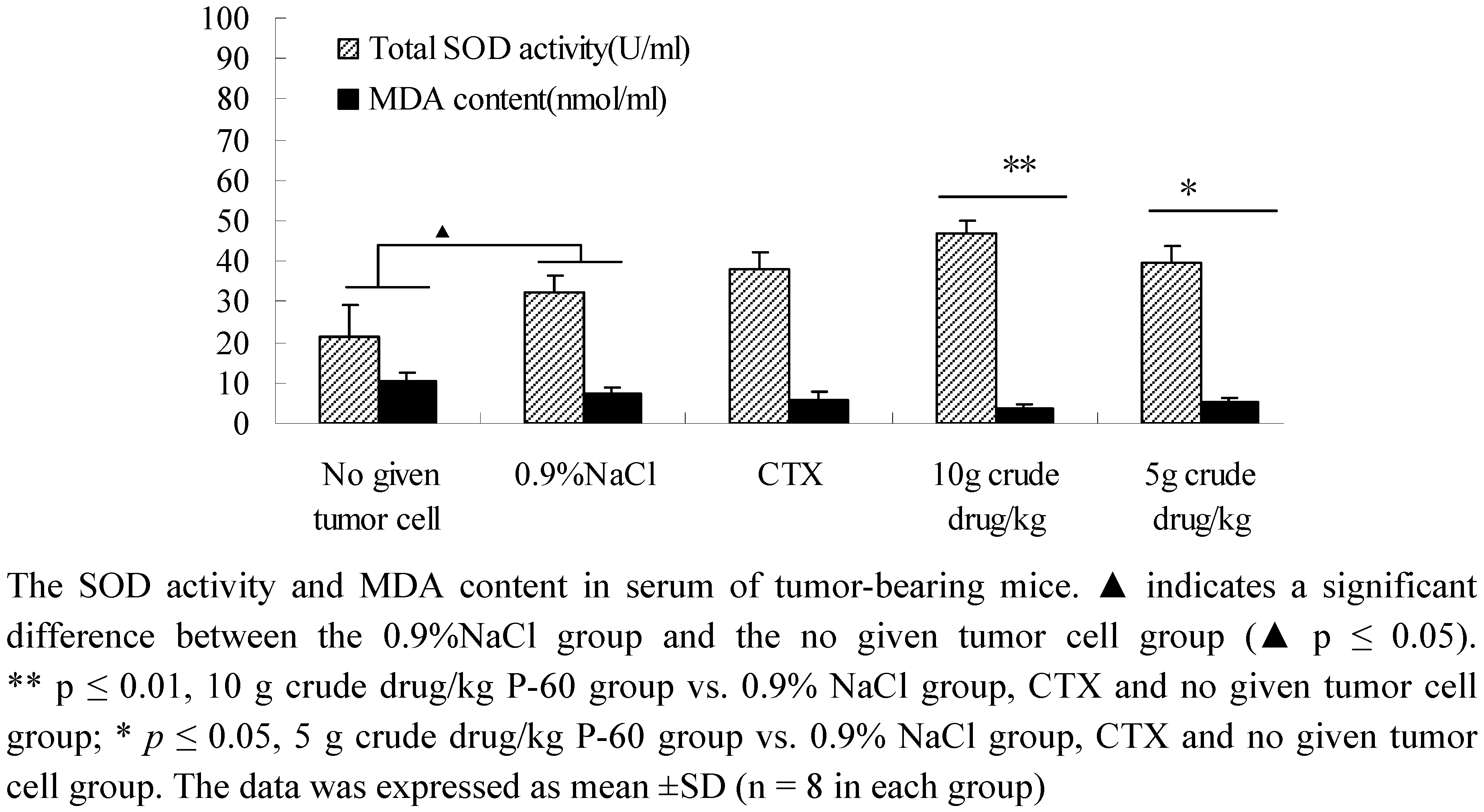
2.5. Determination of total SOD activity and MDA content in tumor-bearing mice
3. Experimental
3.1. General
3.2. Preparation of PV extract
3.3. HPLC analysis
3.4. Antioxidant activity by ABTS˙+, DPPH, FRAP in vitro
3.5. Effect of P-60 on tumor growth
3.6. SOD activity and MDA content in tumor-bearing C57BL/6 mice
3.7. Statistical analysis
4. Conclusions
Acknowledgements
References and Notes
- Lai, S.C.; Ho, Y.L.; Huang, S.C.; Huang, T.H.; Lai, Z.R.; Wu, C.R.; Lian, K.Y.; Chang, Y.S. Antioxidant and antiproliferative activities of Desmodium triflorum (L.) DC. Am. J. Chin. Med. 2010, 38, 329–333. [Google Scholar] [CrossRef]
- Tripathi, U.N.; Chandra, D. The plant extracts of Momordica charantia and Trigonella foenum-graecum have anti-oxidant and anti-hyperglycemic properties for cardiac tissue during diabetes mellitus. Oxid. Med. Cell. Longev. 2009, 2, 290–296. [Google Scholar] [CrossRef]
- Shimoni, N.; Kaplan, M.; Keidar, S. Cardiovascular diseases in patients with high levels of plasma high density lipoprotein: Association with increased plasma oxidative state. Isr. Med. Assoc. J. 2003, 5, 702–705. [Google Scholar]
- Sasidharan, S.; Aravindran, S.; Latha, L.Y.; Vijenthi, R.; Saravanan, D.; Amutha, S. In vitro antioxidant activity and hepatoprotective effects of Lentinula edodes against paracetamol-induced hepatotoxicity. Molecules 2010, 15, 4478–4489. [Google Scholar] [CrossRef]
- Mostafa, G.A.; El-Hadidi, E.S.; Hewedi, D.H.; Abdou, M.M. Oxidative stress in Egyptian children with autism: Relation to autoimmunity. J. Neuroimmunol. 2010, 219, 114–118. [Google Scholar] [CrossRef]
- Butterfield, D.A.; Bader Lange, M.L.; Sultana, R. Involvements of the lipid peroxidation product, HNE, in the pathogenesis and progression of Alzheimer's disease. Biochim. Biophys. Acta 2010, 1801, 924–929. [Google Scholar] [CrossRef]
- Widodo, N.; Priyandoko, D.; Shah, N.; Wadhwa, R.; Kaul, S.C. Selective killing of cancer cells by Ashwagandha leaf extract and its component Withanone involves ROS signaling. PLoS One 2010, 5, e13536. [Google Scholar]
- Simić, A.; Manojlović, D.; Segan, D.; Todorović, M. Electrochemical behavior and antioxidant and prooxidant activity of natural phenolics. Molecules 2007, 12, 2327–2340. [Google Scholar] [CrossRef]
- Liu, F.; Ng, T.B. Antioxidative and free radical scavenging activities of selected medicinal herbs. Life Sci. 2000, 66, 725–735. [Google Scholar] [CrossRef]
- Lamaison, J.L.; Petitjean-Freytet, C.; Carnat, A. Medicinal Lamiaceae with antioxidant properties, a potential source of rosmarinic acid. Pharm. Acta Helv. 1991, 66, 185–188. [Google Scholar]
- Matés, J.M.; Segura, J.A.; Alonso, F.J.; Márquez, J. Natural Antioxidants: Therapeutic Prospects for Cancer and Neurological Diseases. Mini Rev. Med. Chem. 2009, 9, 1202–1214. [Google Scholar] [CrossRef]
- Gupta, A.; Srivastava, S.; Prasad, R.; Natu, S.M.; Mittal, B.; Negi, M.P.; Srivastava, A.N. Oxidative stress in non-small cell lung cancer patients after chemotherapy: Association with treatment response. Respirology 2010, 15, 349–356. [Google Scholar] [CrossRef]
- Tutin, T.G.; Burges, N.A.; Chater, A.O.; Edmondson, J.R.; Heywood, V.H.; Moore, D.M.; Valentine, D.H.; Walters, S.M.; Webb, D.A. Flora Europaea; Cambridge Univ. Press: Cambridge, UK, 1993; Volume 1-5. [Google Scholar]
- Jitka, P.; Milan, K.; Jaromír, S.; Zden.; Jaroslav, V.; Jitka, U. Biological Activities of Prunella vulgaris Extract. Phytother. Res. 2003, 17, 1082–1087. [Google Scholar]
- Harput, U.S.; Saracoglu1, I.; Ogihara, Y. Effects of two Prunella species on lymphocyte pliferation and nitric oxide production. Phytother. Res. 2006, 20, 157–159. [Google Scholar]
- Fang, X.; Yu, M.M.; Yuen, W.H.; Zee, S.Y.; Chang, R.C. Immune modulatory effects of Prunella vulgaris L. on monocytes/macrophages. Int. J. Mol. Med. 2005, 16, 1109–1116. [Google Scholar]
- Feng, L.; Jia, X.B.; Shi, F.; Chen, Y. Identification of Two Poly- saccharides from Prunella vulgaris L. and Evaluation on Their Anti-Lung Adenocarcinoma Activity. Molecules 2010, 15, 5093–5103. [Google Scholar]
- Collins, N.H.; Lessey, E.C.; DuSell, C.D.; McDonnell, D.P.; Fowler, L.; Palomino, W.A.; Illera, M.J.; Yu, X.; Mo, B.; Houwing, A.M.; Lessey, B.A. Characterization of antiestrogenic activity of the Chinese herb, prunella vulgaris, using in vitro and in vivo (Mouse Xenograft) models. Biol. Reprod. 2009, 80, 375–383. [Google Scholar]
- Zhang, K.J.; Zhang, M.Z.; Wang, Q.D.; Liu, W.L. The experimental research about the effect of Prunella vulgaris L. on Raji cells growth and expression of apoptosis related protein. Zhong Yao Cai. 2006, 29, 1207–1210. [Google Scholar]
- Tanaka, T.; Kojima, T.; Kawamori, T.; Wang, A.; Suzui, M.; Okamoto, K.; Mori, H. Inhibition of 4-nitroquinoline -1-oxide-induced rat tongue carcinogenesis by the naturally occurring plant phenolics caffeic, ellagic, chlorogenic and ferulic acids. Carcinogenesis 1993, 14, 1321–1325. [Google Scholar]
- Moon, D.O.; Kim, M.O.; Lee, J.D.; Choi, Y.H.; Kim, G.Y. Rosmarinic acid sensitizes cell death through suppression of TNF-alpha-induced NF-kappaB activation and ROS generation in human leukemia U937 cells. Cancer Lett. 2010, 288, 183–191. [Google Scholar]
- Lin, C.W.; Hou, W.C.; Shen, S.C.; Juan, S.H.; Ko, C.H.; Wang, L.M.; Chen, Y.C. Quercetin inhibition of tumor invasion via suppressing PKC delta/ERK/AP-1- dependent matrix metalloproteinase-9 activation in breast carcinoma cells. Carcinogenesis 2008, 29, 1807–1815. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. Food Sci. Technol.-Lebensm.-Wiss. Technol. 1995, 28, 25–30. [Google Scholar]
- Kim, A.J.; Han, M.R.; Kim, M.H.; Lee, M.; Yoon, T.J.; Ha, S.D. The antioxidant and chemopreventive potentialities of Mosidae (Adenophora remotiflora) leaves. Nutr. Res. Pract. 2010, 4, 30–35. [Google Scholar]
- Zdarilová, A.; Svobodová, A.; Simánek, V.; Ulrichová, J. Prunella vulgaris extract and rosmarinic acid suppress lipopolysaccharide-induced alteration in human gingival fibroblasts. Toxicol. In Vitro 2009, 23, 386–392. [Google Scholar]
- Ozcan, E. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clinical Biochem. 2004, 37, 277. [Google Scholar]
- Meng, L.H.; Lozano, Y.F.; Gaydou, E.M.; Li, B. Antioxidant Activities of Polyphenols Extracted from Perilla frutescens Varieties. Molecules 2009, 14, 133–140. [Google Scholar]
- Benzie, I. FF.; Wai, Y.; Strain, J.J. Antioxidant (reducing) efficiency of ascorbate in plasma is not affected by concentration. J. Nutr. Biochem. 1999, 10, 146. [Google Scholar]
- kottová, N.; Kazdová, L.; Oliyarnyk, O.; Vecera Rv Sobolová, L.; Ulrichová, J. Phenolics-rich extracts from Silybum marianum and Prunella vulgaris reduce a high-sucrose diet induced oxidative stress in hereditary hypertriglyceridemic rats. Pharmacol. Res. 2004, 50, 123–130. [Google Scholar]
- Von, G.A.; Joubert, E.; Hansmann, C.F. Comparison of antioxidant activity of aspalathin with that of other plant phenols of Rooibosed tea (Aspalathon linearis), α-tocopherol, BHT, and BHA. J. Agric. Food. Chem. 1997, 45, 632. [Google Scholar]
- Reichling, J.; Nolkemper, S.; Stintzing, F.C.; Schnitzler, P. Impact of ethanolic lamiaceae extracts on herpes virus infectivity in cell culture. Forsch Komplementmed. 2008, 15, 313–320. [Google Scholar]
- Niwa, Y. Oxidative injury and its defense system in vivo. Rinsho Byori. 1999, 47, 189–209. [Google Scholar]
- Feng, R.; Ni, HM.; Wang, SY.; Tourkova, I.L.; Shurin, M.R.; Harada, H.; Yin, X.M. Cyanidin-3-rutinoside, a natural polyphenol antioxidant, selectively kills leukemic cells by induction of oxidative stress. J. Biol. Chem. 2007, 282, 13468–13476. [Google Scholar]
- Jirovsky, D.; Kosina, P.; Myslinova, M.; Styskala, J.; Ulrichova, J.; Šimanek, V. HPLC analysis of rosmarinic acid in feed enriched with aerial parts of Prunella vulgaris and its metabolites in pig plasma using dual-channel coulometric detection. J. Agric. Food Chem. 2007, 55, 7631–7637. [Google Scholar]
- Psotová, J.; Kolár, M.; Sousek, J.; Svagera, Z.; Vicar, J.; Ulrichová, J. Biological activities of Prunella vulgaris extract. Phytother. Res. 2003, 17, 1082–1087. [Google Scholar]
- Sample Availability: Samples of Prunella vulgaris L. are available from the authors.
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Feng, L.; Jia, X.; Zhu, M.-M.; Chen, Y.; Shi, F. Antioxidant Activities of Total Phenols of Prunella vulgaris L. in Vitro and in Tumor-bearing Mice. Molecules 2010, 15, 9145-9156. https://doi.org/10.3390/molecules15129145
Feng L, Jia X, Zhu M-M, Chen Y, Shi F. Antioxidant Activities of Total Phenols of Prunella vulgaris L. in Vitro and in Tumor-bearing Mice. Molecules. 2010; 15(12):9145-9156. https://doi.org/10.3390/molecules15129145
Chicago/Turabian StyleFeng, Liang, Xiaobin Jia, Mao-Mao Zhu, Yan Chen, and Feng Shi. 2010. "Antioxidant Activities of Total Phenols of Prunella vulgaris L. in Vitro and in Tumor-bearing Mice" Molecules 15, no. 12: 9145-9156. https://doi.org/10.3390/molecules15129145
APA StyleFeng, L., Jia, X., Zhu, M.-M., Chen, Y., & Shi, F. (2010). Antioxidant Activities of Total Phenols of Prunella vulgaris L. in Vitro and in Tumor-bearing Mice. Molecules, 15(12), 9145-9156. https://doi.org/10.3390/molecules15129145




