Acanmontanoside, a New Phenylethanoid Diglycoside from Acanthus montanus
Abstract
:1. Introduction
2. Results and Discussion
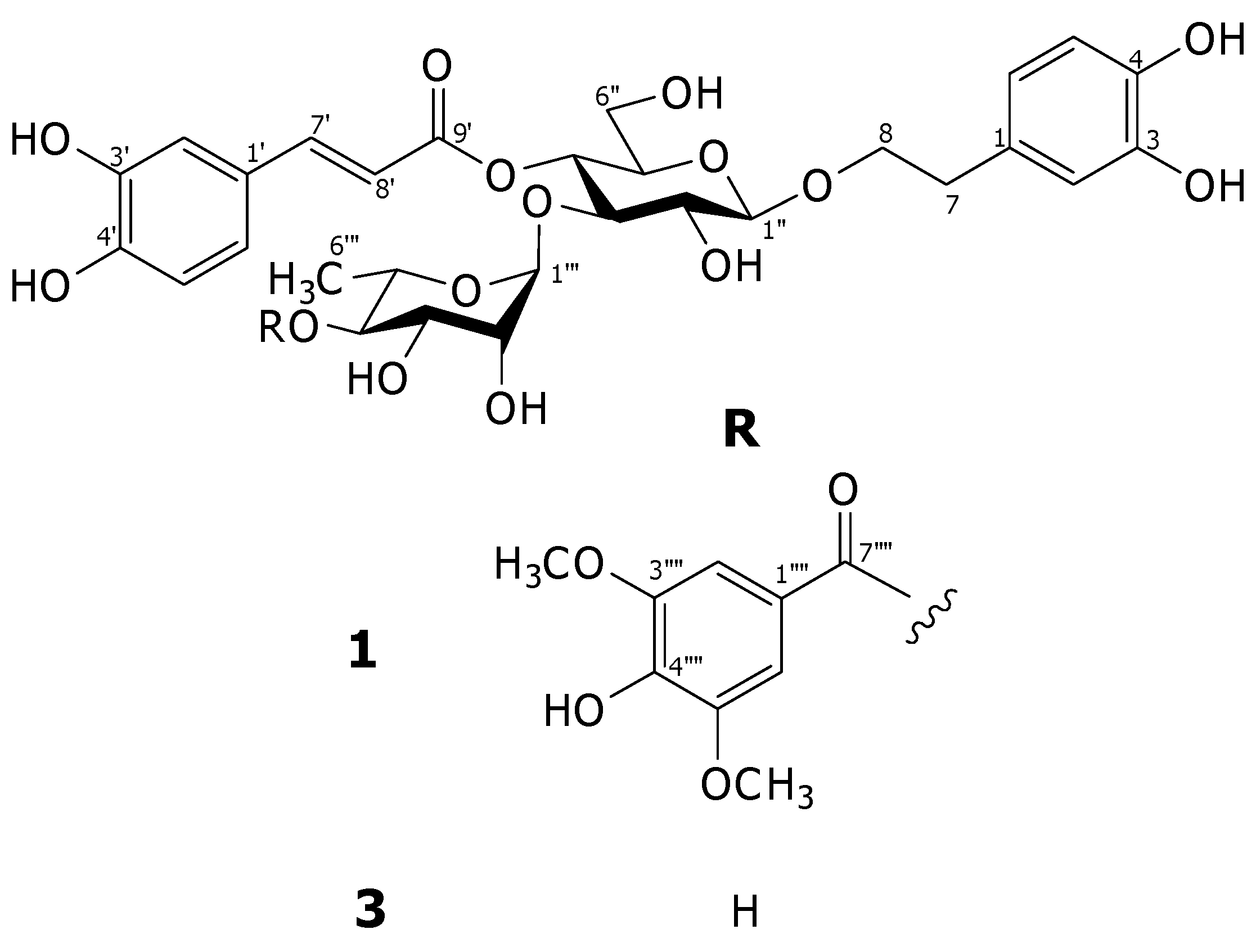
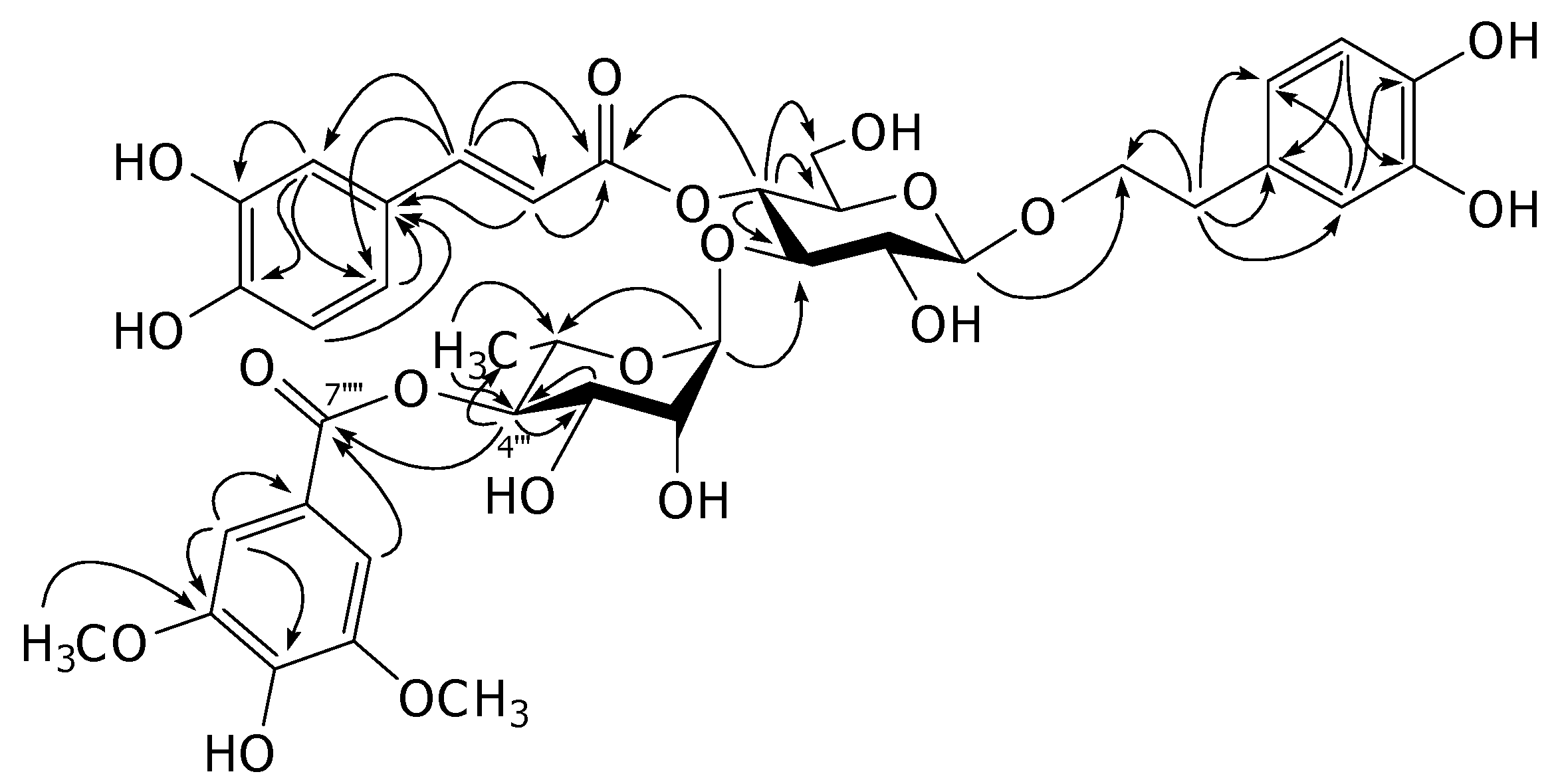
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.3. Acanmontanoside (1)
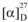 −157.5 (MeOH, c = 0.70); 1H- and 13C-NMR (CD3OD): see Table 1; HRESIMS, m/z: 803.2408 [M-H]− (calcd for C38H43O19:803.2404).
−157.5 (MeOH, c = 0.70); 1H- and 13C-NMR (CD3OD): see Table 1; HRESIMS, m/z: 803.2408 [M-H]− (calcd for C38H43O19:803.2404).| No. | 13C | 1H | ||
|---|---|---|---|---|
| 3 | 1 | 1 | ||
| Aglycone | ||||
| 1 | 131.5 | 131.6 | ||
| 2 | 117.1 | 117.2 | 6.71 (1H, br s) | |
| 3 | 146.1 | 146.2 | ||
| 4 | 144.6 | 144.7 | ||
| 5 | 116.3 | 116.4 | 6.69 (1H, d, J = 7.2 Hz) | |
| 6 | 121.3 | 121.4 | 6.57 (1H, br d, J = 7.2 Hz) | |
| 7 | 36.5 | 36.6 | 2.80 (2H, t, J = 7.3 Hz) | |
| 8 | 72.2 | 72.2 | 3.74 (1H)a | |
| 4.05 (1H)a | ||||
| Caffeoyl moiety | ||||
| 1' | 127.7 | 127.3 | ||
| 2' | 115.2 | 114.9 | 6.86 (1H, br s) | |
| 3' | 149.7 | 149.8 | ||
| 4' | 146.8 | 146.7 | ||
| 5' | 116.5 | 116.4 | 6.59 (1H, d, J = 8.3 Hz) | |
| 6' | 123.2 | 123.2 | 6.43 (1H, br d, J = 8.3 Hz) | |
| 7' | 148.0 | 148.1 | 7.52 (1H, d, J = 16.0 Hz) | |
| 8' | 114.7 | 114.5 | 6.21 (1H, d, J = 16.0 Hz) | |
| 9' | 168.3 | 168.3 | ||
| Glc | ||||
| 1'' | 104.2 | 104.2 | 4.39 (1H, d, J = 7.9 Hz) | |
| 2'' | 76.2 | 76.5 | 3.45 (1H, dd, J = 7.9, 8.4 Hz) | |
| 3'' | 81.6 | 80.7 | 3.92 (1H, dd, J = 8.4, 9.3 Hz) | |
| 4'' | 70.4 | 70.2 | 4.99 (1H, dd, J = 9.3, 9.4 Hz) | |
| 5'' | 76.0 | 76.0 | 3.56 (1H, m) | |
| 6'' | 62.4 | 62.4 | 3.54 (1H)a | |
| 3.64 (1H)a | ||||
| Rha | ||||
| 1''' | 103.0 | 102.3 | 5.37 (1H, br s) | |
| 2''' | 72.3 | 72.3 | 3.72 (1H)a | |
| 3''' | 72.0 | 70.7 | 3.94 (1H)a | |
| 4''' | 73.8 | 76.3 | 5.02 (1H, dd, J = 9.7, 9.7 Hz) | |
| 5''' | 70.6 | 68.2 | 3.86 (1H)a | |
| 6''' | 18.4 | 18.7 | 1.05 (3H, d, J = 6.2 Hz) | |
| Syringyl moiety | ||||
| 1'''' | 121.5 | |||
| 2'''', 6'''' | 108.5 | 7.22 (2H, s) | ||
| 3'''', 5'''' | 148.8 | |||
| 4'''' | 142.1 | |||
| 7'''' | 168.0 | |||
| MeO-3'''', 5'''' | 57.0 | 3.86 (6H, s) | ||
4. Conclusions
Acknowledgements
- Sample Availability: Samples of the compounds are available from the authors.
References
- Kanchanapoom, T.; Kamel, M.S.; Kasai, R.; Yamasaki, K.; Picheansoonthon, C.; Hiraga, Y. Lignan glucosides from Acanthus ilicifolius. Phytochemistry 2001, 56, 369–372. [Google Scholar]
- Kanchanapoom, T.; Kamel, M.S.; Kasai, R.; Picheansoonthon, C.; Hiraga, Y.; Yamasaki, K. Benzoxazinoid glucosides from Acanthus ilicifolius. Phytochemistry 2001, 58, 637–640. [Google Scholar]
- Kanchanapoom, T.; Kasai, R.; Yamasaki, K. Flavonoid glycosides from Acanthus ilicifolius L. Nat. Med. 2001, 56, 122. [Google Scholar]
- Kanchanapoom, T.; Kasai, R.; Picheansoonthon, C.; Yamasaki, K. Megastigmane, aliphatic alcohol and benzoxazinoid glycosides from Acanthus ebracteatus. Phytochemistry 2001, 58, 811–817. [Google Scholar]
- Kanchanapoom, T.; Noiarsa, P.; Otsuka, H.; Ruchirawat, S. Chemical constituents of Acanthus volubilis Wall. Biochem.Syst. Ecol. 2006, 34, 442–445. [Google Scholar] [CrossRef]
- Adeyemi, O.O.; Okpo, S.O.; Young-Nwafor, C.C. The relaxant activity of the methanolic extract of Acanthus montanus on intestinal smooth muscles. J. Ethnopharmacol. 1999, 68, 169–173. [Google Scholar] [CrossRef]
- Adeyemi, O.O.; Okpo, S.O.; Okpaka, O. The analgesic effect of the methanolic extract of Acanthus montanus. J. Ethnopharmacol. 2004, 90, 45–48. [Google Scholar] [CrossRef]
- Asongalem, E.A.; Foyet, H.S.; Ekobo, S.; Dimo, T.; Kamtchouing, P. Anti-inflammatory, lack of central analgesia and antipyretic properties of Acanthus montanus (Ness) T. Anderson. J. Ethnopharmacol. 2004, 95, 63–68. [Google Scholar] [CrossRef]
- Okoli, C.O.; Akah, P.A.; Onuoha, N.J.; Okoye, T.C.; Nwoye, A.C.; Nworu, C.S. Acanthus montanus: An experimental evaluation of the antimicrobial, anti-inflammatory and immunological properties of a traditional remedy for furuncles. BMC Compl. Alternative Med. 2008, 8, 27. [Google Scholar] [CrossRef]
- Anam, E.M. Pentacyclic triterpenoids from Acanthus montanus (Acanthaceae). Indian J. Chem. B 1997, 36, 110–113. [Google Scholar]
- Anam, E.M. Novel triterpenoid glycoside and triterpenoid acid from the root extract of Acanthus montanus (Acanthaceae). Indian J. Chem. B 1997, 36, 901–904. [Google Scholar]
- Yamamura, S.; Ozawa, K.; Ohtani, K.; Kasai, R.; Yamasaki, K. Antihistaminic flavones and aliphatic glycosides from Mentha spicata. Phytochemistry 1998, 48, 131–136. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Noiarsa, P.; Ruchirawat, S.; Kanchanapoom, T. Acanmontanoside, a New Phenylethanoid Diglycoside from Acanthus montanus. Molecules 2010, 15, 8967-8972. https://doi.org/10.3390/molecules15128967
Noiarsa P, Ruchirawat S, Kanchanapoom T. Acanmontanoside, a New Phenylethanoid Diglycoside from Acanthus montanus. Molecules. 2010; 15(12):8967-8972. https://doi.org/10.3390/molecules15128967
Chicago/Turabian StyleNoiarsa, Pawadee, Somsak Ruchirawat, and Tripetch Kanchanapoom. 2010. "Acanmontanoside, a New Phenylethanoid Diglycoside from Acanthus montanus" Molecules 15, no. 12: 8967-8972. https://doi.org/10.3390/molecules15128967
APA StyleNoiarsa, P., Ruchirawat, S., & Kanchanapoom, T. (2010). Acanmontanoside, a New Phenylethanoid Diglycoside from Acanthus montanus. Molecules, 15(12), 8967-8972. https://doi.org/10.3390/molecules15128967




