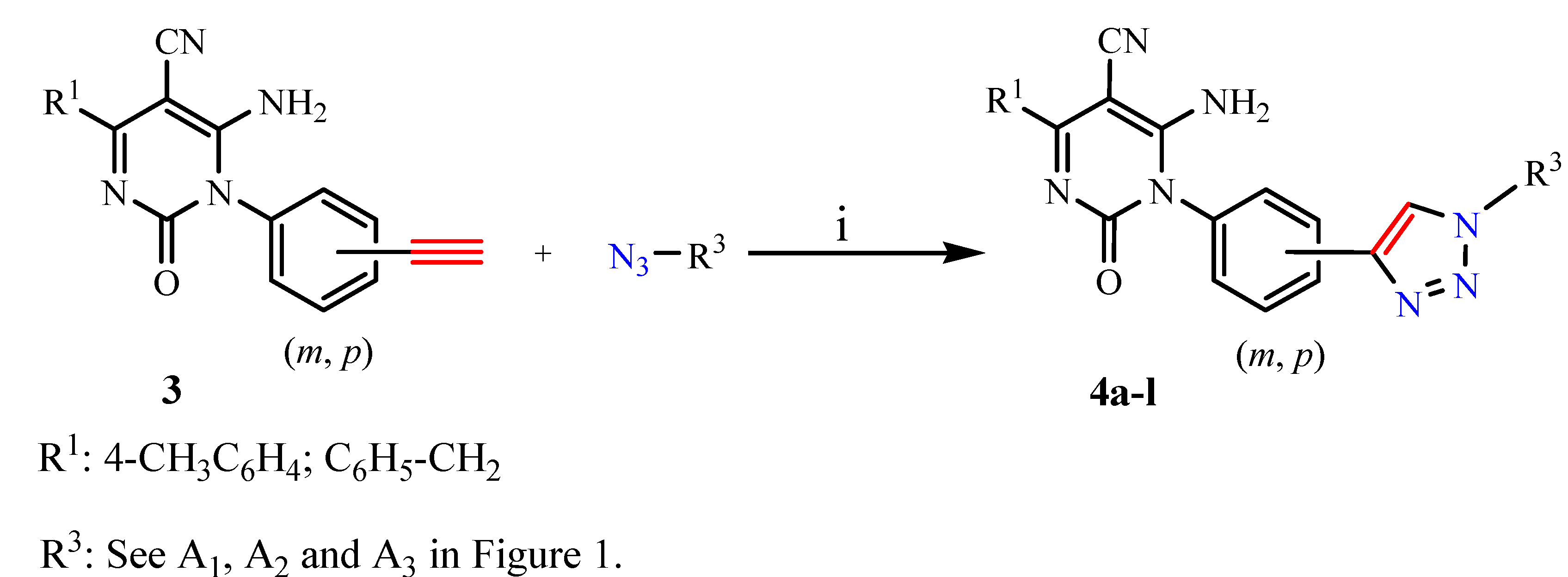Experimental
Commercially available reagent grade chemicals were used as received without additional purification. All reactions were followed by TLC (E. Merck Kieselgel 60 F-254), with detection by UV light at 254 nm. Column chromatography was performed on silica gel (60–200 mesh E. Merck). IR spectra were recorded on a Perkin-Elmer PARAGON 1000 FT-IR spectrometer. 1H- and 13C-NMR spectra were recorded on an AC Bruker spectrometer at 300 MHz (1H) and 75 MHz (13C) using (CD3)2SO as solvent with (CD3)2SO (δH 2.5) or (CD3)2SO (δC 39.5). Chemical shifts (δ) are reported in parts per million (ppm) relative to tetramethylsilane (0 ppm) as internal reference and the following multiplicity abbreviations were used: s, singlet; d, doublet; t, triplet; q, quadruplet; m, multiplet; J in hertz. The mass spectra were recorded on an ion trap mass spectrometer (Finnigan LCQ Deca XP Max) using electrospray as an ionization source. Melting points were determined on an Electrothermal 9300 capillary melting point apparatus and are uncorrected. UV-visible spectra were recorded on a Specord 205 Analytikjena spectrophotometer. The purity of all compounds was determined by LC-PDA-MS methods and was found to be in the range between 96–99%.
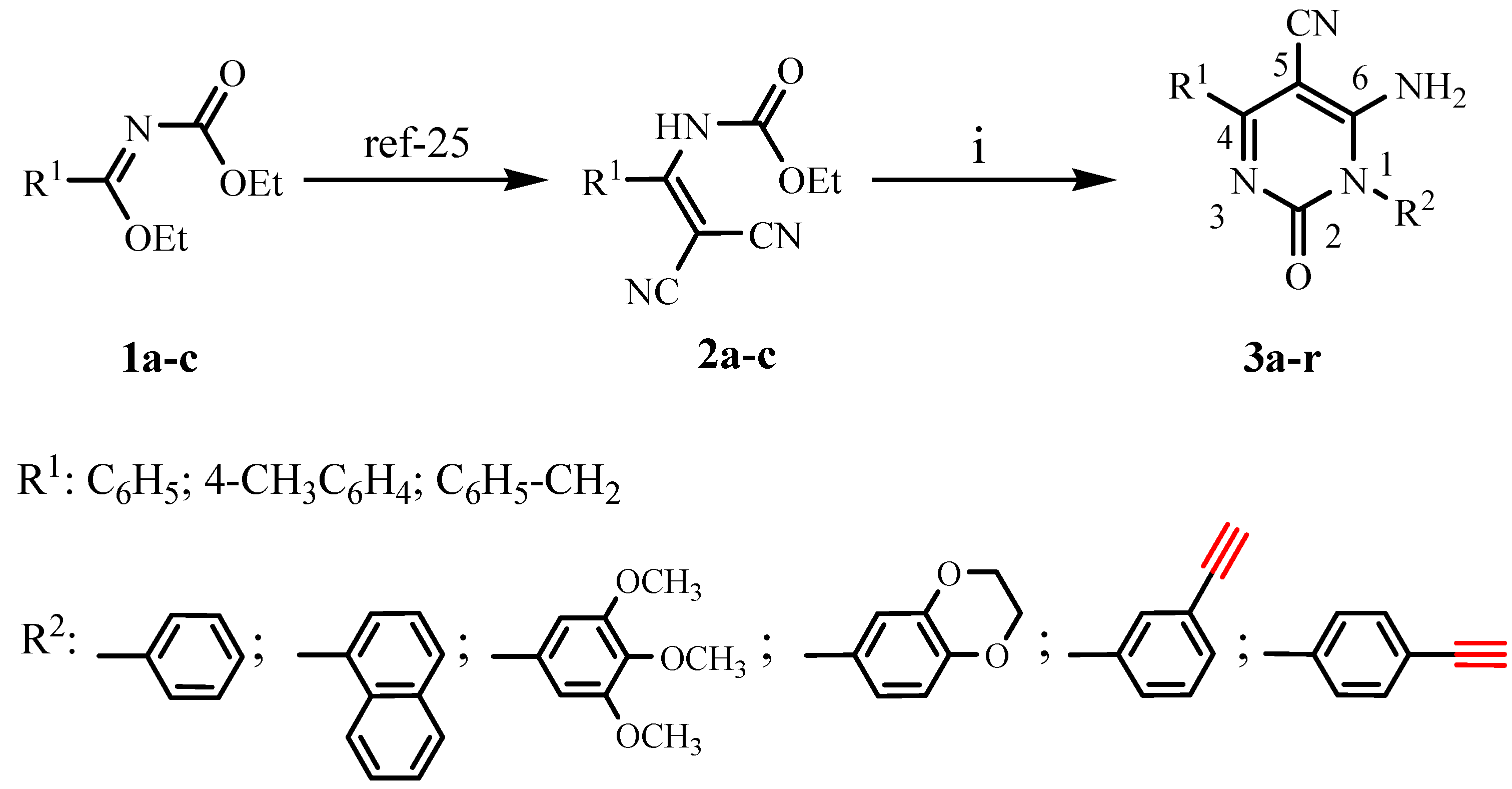
General experimental procedure for preparation of 6-amino-5-cyano-1,4-disubstituted-2(1H)-pyrimid-inones 3a-r
To a magnetically stirred solution of the ethyl 2,2-dicyanovinylcarbamate derivatives 2a-c (1 equiv) in chlorobenzene (25 mL), a primary aromatic amine (1.2 equiv) added and reaction mixture was stirred for 2~4 h at 110 °C. Reaction progress was monitored by TLC using the indicated eluents. The resulting mixture was allowed to cool at room temperature. The formed precipitate was isolated by filtration and washed with ethanol or with diethyl ether for 3e, 3f, 3k, 3l, 3q and 3r to give pure products.
6-Amino-5-cyano-1,4-diphenyl-2(1H)-pyrimidinone (3a). White cristals, yield (75%), C17H12N4O, M = 288 gmol−1, mp 252–254 °C, Rf = 0.21 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 248 (32,400), 318 (11,232); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,212 (CN), 1,665 (C=O), 1,616 (C=N); 1H-NMR: (DMSO-d6): δ = 7.34–7.82 (m, 12H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 72.9 (C-5), 117.1 (CN), 128.8, 128.9, 130, 130.7, 131.5, 135.1, 137.5, 154.1 (C-2), 160.5 (C-4), 172 (C-6); MS-(+)ESI: m/z (%): 599 ([2M+Na]+, 35), 311 ([M+Na]+, 4), 289 ([M+H]+, 100).
6-Amino-5-cyano-1-(naphthalen-1-yl)-4-phenyl-2(1H)-pyrimidinone (3b). Greyish white solid, yield (68%), C21H14N4O, M = 338 gmol−1, mp 198–200 °C, Rf = 0.54 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 248 (37,518), 318 (14,196); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,211 (CN), 1,672 (C=O), 1,620 (C=N); 1H-NMR: (DMSO-d6): δ = 7.42–8.15 (m, 14H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 73.1 (C-5), 117.2 (CN), 122, 127.1, 127.2, 127.8, 128, 128.8, 129, 129.2, 129.7, 130.7, 131.5, 131.6, 135.1, 137.6, 154.2 (C-2), 160.9 (C-4), 172.6 (C-6); MS-(+)ESI: m/z (%): 699 ([2M+Na]+, 16), 339 ([M+H]+, 100).
6-Amino-5-cyano-1-(3,4,5-trimethoxyphenyl)-4-phenyl-2(1H)-pyrimidinone (3c). Yellowish white solid, yield (71%), C20H18N4O4, M = 378 gmol−1, mp 255–257 °C, Rf = 0.21 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε L.mol−1cm−1): 249 (51,030), 320 (14,742); IR (KBr) cm−1: 3,450-3,310 (NH2), 2,209 (CN), 1,687 (C=O), 1,620 (C=N); 1H-NMR: (DMSO-d6): δ = 3.74 (s, 6H, 2OCH3), 3.76 (s, 3H, OCH3), 7.39–7.60 (m, 9H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 56.5 (2C, 2OCH3), 60.4 (OCH3), 72.6 (C-5), 106.4, 117.1 (CN), 125.9, 128.6, 130.5, 136.9, 138, 138.4, 153.9, 154 (C-2), 160.7 (C-4), 172 (C-6); MS-(+)ESI: m/z (%): 779 ([2M+Na]+, 3), 379 ([M+H]+, 100).
6-Amino-5-cyano-1-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-4-phenyl-2(1H)-pyrimidinone (3d). Pale brown solid, yield (74%), C19H14N4O3, M = 346 gmol−1, mp 283–285 °C, Rf = 0.49 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 248 (37887), 318 (12456); IR (KBr) cm−1: 3,450-3,310 (NH2), 2,210 (CN), 1,696 (C=O), 1,662 (C=N); 1H-NMR: (DMSO-d6): δ = 4.30 (s, 4H, 2CH2), 6.80-7.59 (m, 10H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 64.5 (CH2), 64.6 (CH2), 72.6 (C-5), 117.2 (CN), 118.5, 121.7, 125.7, 127.6, 129.2, 131.9, 137.7, 138.1, 144.5, 144.6, 154.1 (C-2), 160.7 (C-4), 171.8 (C-6); MS-(+)ESI: m/z (%): 715 ([2M+Na]+, 8), 347 ([M+H]+, 100).
6-Amino-5-cyano-1-(3-ethynylphenyl)-4-phenyl-2(1H)-pyrimidinone (3e). Pale yellow solid, yield (62%), C19H12N4O, M = 312 gmol−1, mp 237–239 °C, Rf = 0.52 (ethyl acetate/dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 254 (36,972), 307 (14,508); IR (KBr) cm−1: 3,450–3,310 (NH2), 3,270 (≡C-H), 2,209 (CN), 1,665 (C=O), 1,636 (C=N); 1H-NMR: (DMSO-d6): δ = 4.32 (s, 1H, C≡CH), 7.27–7.86 (m, 11H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 73.7 (C-5), 82.4 (C≡CH), 83.2 (C≡CH), 117.2 (CN), 124.1, 128.6, 129.2, 129.9, 131.1, 132.4, 133.4, 134.5, 135.4, 141.5, 154 (C-2), 160.5 (C-4), 171.7 (C-6); MS-(+)ESI: m/z (%): 647 ([2M+Na]+, 21), 313 ([M+H]+, 100).
6-Amino-5-cyano-1-(4-ethynylphenyl)-4-phenyl-2(1H)-pyrimidinone (3f). White solid, yield (60%), C19H12N4O, M = 312 gmol−1, mp 206–208 °C, Rf = 0.57 (ethyl acetate/dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 247 (38,844), 309 (13,572); IR (KBr) cm−1: 3,450-3,310 (NH2), 3,268 (≡C-H), 2,210 (CN), 1,676 (C=O), 1,635 (C=N); 1H-NMR: (DMSO-d6): δ = 4.31 (s, 1H, C≡CH), 7.34–7.91 (m, 11H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 73.7 (C-5), 82.3 (C≡CH), 83.2 (C≡CH), 117.2 (CN), 125, 128.5, 130.1, 131.2, 132.4, 133.4, 135.4, 141.3, 154 (C-2), 160 (C-4), 172.1 (C-6); MS-(+)ESI: m/z (%): 647 ([2M+Na]+, 10), 313 ([M+H]+, 100).
6-Amino-5-cyano-4-(4-methylphenyl)-1-phenyl-2(1H)-pyrimidinone (3g). White solid, yield (73%), C18H14N4O, M = 302 gmol−1, mp 257–259 °C, Rf = 0.47 (ethyl acetate/dichloromethane, 70:30, v/v);UV (MeOH) λmax nm (ε Lmol−1cm−1): 250 (26,727), 319 (11,325); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,211 (CN), 1,674 (C=O), 1,619 (C=N); 1H-NMR: (DMSO-d6): δ = 2.52 (s, 3H, CH3), 7,45–7,9 (m, 11H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 21.5 (CH3), 72.6 (C-5), 118.3 (CN), 128.8, 129, 130.1, 130.8, 131.5, 132.4, 135.1, 137.5, 154.3 (C-2), 160.8 (C-4), 172.5 (C-6); MS-(+)ESI: m/z (%): 627 ([2M+Na]+, 31), 325 ([M+Na]+, 3), 303 ([M+H]+, 100).
6-Amino-5-cyano-4-(4-methylphenyl)-1-(naphthalen-1-yl)-2(1H)-pyrimidinone (3h). Greyish green solid, yield (67%), C22H16N4O, M = 352 gmol−1, mp 261–263 °C, Rf = 0.57 (ethyl acetate/ dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 251 (26,928), 318 (12,672); IR (KBr) cm−1: 3,450-3,310 (NH2), 2,209 (CN), 1,686 (C=O), 1,617 (C=N); 1H-NMR: (DMSO-d6): δ = 2.43 (s, 3H, CH3), 7.38-8.15 (m, 13H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 21.5 (CH3), 72.4 (C-5), 118.3 (CN), 122.5, 127, 127.2, 127.9, 128.1, 128.9, 129.1, 129.3, 129.8, 130.7, 131.4, 132.1, 135.6, 137.7, 154.6 (C-2), 161 (C-4), 172.8 (C-6); MS-(+)ESI: m/z (%): 727 ([2M+Na]+, 15), 353 ([M+H]+, 100).
6-Amino-5-cyano-4-(4-methylphenyl)-1-(3,4,5-trimethoxyphenyl)-2(1H)-pyrimidinone (3i). Pale yellow solid, yield (75%), C21H20N4O4, M = 392 gmol−1, mp 292–294 °C, Rf = 0.53 (ethyl acetate/ dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 250 (52,920); IR (KBr) cm−1: 3,450-3,310 (NH2), 2,212 (CN), 1,671 (C=O), 1,625 (C=N); 1H-NMR: (DMSO-d6): δ = 2.41 (s, 3H, CH3), 3.74 (s, 6H, 2OCH3), 3.77 (s, 3H, OCH3), 7.4–7.59 (m, 8H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 21.4 (CH3), 56.6 (2C, 2OCH3), 60.4 (OCH3), 72.6 (C-5), 106.4, 117.1 (CN), 125.8, 128.6, 130.5, 137.5, 138.1, 138.5, 154, 154.5 (C-2), 160.6 (C-4), 171.9 (C-6); MS-(+)ESI: m/z (%): 393 ([M+H]+, 100).
6-Amino-5-cyano-1-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-4-(4-methylphenyl)-2(1H)-pyrimidinone (3j). Yellow solid, yield (72%), C20H16N4O3, M = 360 gmol−1, mp 284–286 °C, Rf = 0.54 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 251 (32,940), 280 (19,980), 318 (13,500); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,211 (CN), 1,658 (C=O), 1,634 (C=N); 1H-NMR: (DMSO-d6): δ = 2.40 (s, 3H, CH3), 4.30 (s, 4H, 2CH2), 6.79-7.60 (m, 9H, Ar-H + NH2); 13C- NMR (DMSO-d6): δ = 21.4 (CH3), 64.5 (CH2), 64.6 (CH2), 72.6 (C-5), 117.1 (CN), 117.9, 121.5, 125.8, 127.7, 128.5, 132, 137.4, 138, 144.8, 144.8, 154.1 (C-2), 160.7 (C-4), 171.8 (C-6); MS-(+)ESI: m/z (%): 743 ([2M+Na]+, 9), 361 ([M+H]+, 100).
6-Amino-5-cyano-1-(3-ethynylphenyl)-4-(4-methylphenyl)-2(1H)-pyrimidinone (3k). Pale yellow solid, yield (70%), C20H14N4O, M = 326 gmol−1, mp 247–249 °C, Rf = 0.61 (ethyl acetate/dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 252 (24,339), 307 (16,137); IR (KBr) cm−1: 3,450–3,310 (NH2), 3,271 (≡C-H), 2,211 (CN), 1,671 (C=O), 1,640 (C=N); 1H-NMR: (DMSO-d6): δ = 2.41 (s, 3H, CH3), 4.32 (s, 1H, C≡CH), 7.25–7.75 (m, 10H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 21.5 (CH3), 73.81 (C-5), 82.4 (C≡CH), 83.2 (C≡CH), 117.2 (CN), 124, 128.8, 129.3, 129.8, 131, 132.5, 133.3, 134.6, 135.6, 141.5, 153.9 (C-2), 160.5 (C-4), 171.8 (C-6); MS-(+)ESI: m/z (%): 675 ([2M+Na]+, 19), 327 ([M+H]+, 100).
6-Amino-5-cyano-1-(4-ethynylphenyl)-4-(4-methylphenyl)-2(1H)-pyrimidinone (3l). Brownish yellow solid, yield (61%), C20H14N4O, M = 326 gmol−1, mp 192–194 °C, Rf = 0.49 (ethyl acetate/ dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 250 (28,851), 308 (15,159); IR (KBr) cm−1: 3,450–3,310 (NH2), 3,270 (≡C-H), 2,209 (CN), 1,662 (C=O), 1,638 (C=N); 1H-NMR: (DMSO-d6): δ = 2.41 (s, 3H, CH3), 4.30 (s, 1H, C≡CH), 7.27–7.94 (m, 10H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 21.5 (CH3), 72.9 (C-5), 82.3 (C≡CH), 83.3 (C≡CH), 117.2 (CN), 124.1, 128.9, 129.3, 131.1, 132.5, 134.4, 135.8, 141.3, 153.9 (C-2), 160.1 (C-4), 171.6 (C-6); MS-(+)ESI: m/z (%): 675 ([2M+Na]+, 9), 327 ([M+H]+, 100).
6-Amino-4-benzyl-5-cyano-1-phenyl-2(1H)-pyrimidinone (3m). White solid, yield (76%), C18H14N4O, M = 302 gmol−1, mp 276–278 °C, Rf = 0.31 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 248 (22,197), 308 (14,043); IR (KBr) cm−1: 3,450-3,310 (NH2), 2,211 (CN), 1,680 (C=O), 1,614 (C=N); 1H-NMR: (DMSO-d6): δ = 3.9 (s, 2H, CH2), 7.3–7.53 (m, 12H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 43.4 (CH2), 73.7 (C-5), 116.7 (CN), 127.3, 128.9, 129, 129.4, 129.7, 130.7, 135, 137.1, 154.1 (C-2), 159.7 (C-4), 175.4 (C-6); MS-(+)ESI: m/z (%): 627 ([2M+Na]+, 20), 325 ([M+Na]+, 3), 303 ([M+H]+, 100).
6-Amino-4-benzyl-5-cyano-1-(naphthalen-1-yl)-2(1H)-pyrimidinone (3n). Pale violet solid, yield (59%), C22H16N4O, M = 352 gmol−1, mp 252–254 °C, Rf = 0.47 (ethyl acetate/dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 248 (24,288), 296 (17,952); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,212 (CN), 1,678 (C=O), 1,618 (C=N); 1H-NMR: (DMSO-d6): δ = 3.97 (s, 2H, CH2), 7.27–8.27 (m, 14H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 43.6 (CH2), 73.7 (C-5), 116.6 (CN), 117.9, 121.7, 126.5, 126.9, 127.4, 127.8, 127.9, 129.1, 129.6, 129.7, 131.3, 134.3, 135, 137.1, 154.1 (C-2), 160 (C-4), 175.9 (C-6); MS-(+)ESI: m/z (%): 727 ([2M+Na]+, 10), 353 ([M+H]+, 100).
6-Amino-4-benzyl-5-cyano-1-(3,4,5-trimethoxyphenyl)-2(1H)-pyrimidinone (3o). Yellow solid, yield (65%), C21H20N4O4, M = 392 gmol−1, mp 224–226 °C, Rf = 0.26 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε L.mol−1cm−1): 247 (39,396), 307 (19,404); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,208 (CN), 1,687 (C=O), 1,627 (C=N); 1H-NMR: (DMSO-d6): δ = 3.91 (s, 2H, CH2), 3.75 (s, 6H, 2OCH3), 3.77 (s, 3H, OCH3), 7.47–8.62 (m, 9H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 43.4 (CH2), 56.6 (2C, 2OCH3), 60.4 (OCH3), 72.7 (C-5), 107.1, 116.9 (CN), 126, 128.6, 129.9, 137.1, 138.3, 138.5, 153.6, 154.4 (C-2), 160.7 (C-4), 175 (C-6); MS-(+)ESI: m/z (%): 393 ([M+H]+, 100).
6-Amino-4-benzyl-5-cyano-1-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-2(1H)-pyrimidinone (3p). Dark brown solid, yield (55%), C20H16N4O3, M = 360 gmol−1, mp 287–289 °C, Rf = 0.41 (ethyl acetate/ dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 251 (31,320), 318 (15,120); IR (KBr) cm−1: 3,450-3,310 (NH2), 2,210 (CN), 1,661 (C=O), 1,609 (C=N); 1H-NMR: (DMSO-d6): δ = 3.9 (s, 2H, CH2), 4.32 (s, 4H, 2CH2), 6.79–8.05 (m, 10H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 43.4 (CH2), 64.5 (CH2), 64.8 (CH2), 72.6 (C-5), 116.9 (CN), 117.4, 121.7, 126, 127.5, 128.1, 131.5, 136.9, 138.1, 143.9, 144.2, 153.9 (C-2), 160.5 (C-4), 175.4 (C-6); MS-(+)ESI: m/z (%): 743 ([2M+Na]+, 11), 361 ([M+H]+, 100).
6-Amino-4-benzyl-5-cyano-1-(3-ethynylphenyl)-2(1H)-pyrimidinone (3q). White solid, yield (62%), C20H14N4O, M = 326 gmol−1, mp 224–226 °C, Rf = 0.47 (ethyl acetate/dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 248 (40,587), 308 (15,160); IR (KBr) cm−1: 3,450–3,310 (NH2), 3,271 (≡C-H), 2,209 (CN), 1,662 (C=O), 1,625 (C=N); 1H-NMR: (DMSO-d6): δ = 3.9 (s, 2H, CH2), 4.3 (s, 1H, C≡CH), 7.32–7.83 (m, 11H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 43.4 (CH2), 73.8 (C-5), 82.3 (C≡CH), 83.2 (C≡CH), 116.5 (CN), 123.9, 128.5, 129.2, 130.2, 131.4, 132.7, 132.9, 134.3, 136.4, 142.1, 154.1 (C-2), 160.8 (C-4), 174.9 (C-6); MS-(+)ESI: m/z (%): 675 ([2M+Na]+, 17), 327 ([M+H]+, 100).
6-Amino-4-benzyl-5-cyano-1-(4-ethynylphenyl)-2(1H)-pyrimidinone (3r). White solid, yield (58%), C20H14N4O, M = 326 gmol−1, mp 252–254 °C, Rf = 0.56 (ethyl acetate/dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 250 (44,010), 318 (12,714); IR (KBr) cm−1: 3,450–3,310 (NH2), 3,269 (≡C-H), 2,211 (CN), 1,668 (C=O), 1,639 (C=N); 1H-NMR: (DMSO-d6): δ = 3.89 (s, 2H, CH2), 4.31 (s, 1H, C≡CH), 7.27–7.63 (m, 11H, Ar-H + NH2); 13C-NMR (DMSO-d6): δ = 43.4 (CH2), 73.8 (C-5), 82.3 (C≡CH), 83.4 (C≡CH), 116.5 (CN), 123.4, 127.2, 128.9, 129.5, 129.6, 133.9, 135.5, 137, 153.9 (C-2), 159.6 (C-4), 175.5 (C-6); MS-(+)ESI: m/z (%): 675 ([2M+Na]+, 8), 327 ([M+H]+, 100).
General experimental procedure for preparation of 1,4-disubstituted-1,2,3-triazoles 4a-l
The mixture of alkyne 3 (1 mmol) and azides (1 mmol) was suspended in a mixture of THF/t-BuOH/H2O (3:1:1, v/v/v, 6/2/2 mL). Sodium ascorbate (89 mg, 0.45 equiv) was added followed by addition of CuSO45H2O (16 mg, 0.1 equiv). The heterogeneous mixture was stirred vigorously for 2 days, at which time TLC showed complete conversion. The reaction mixture was concentrated under vacuum and the residue was treated with H2O (50 mL) and extracted with dichloromethane (3 × 15 mL). The combined organic extracts were dried over anhydrous Na2SO4, filtered and evaporated under reduced pressure to give a crude mass. Column chromatography purification using ethyl acetate/dichloromethane as eluent gave the clicked product 4.
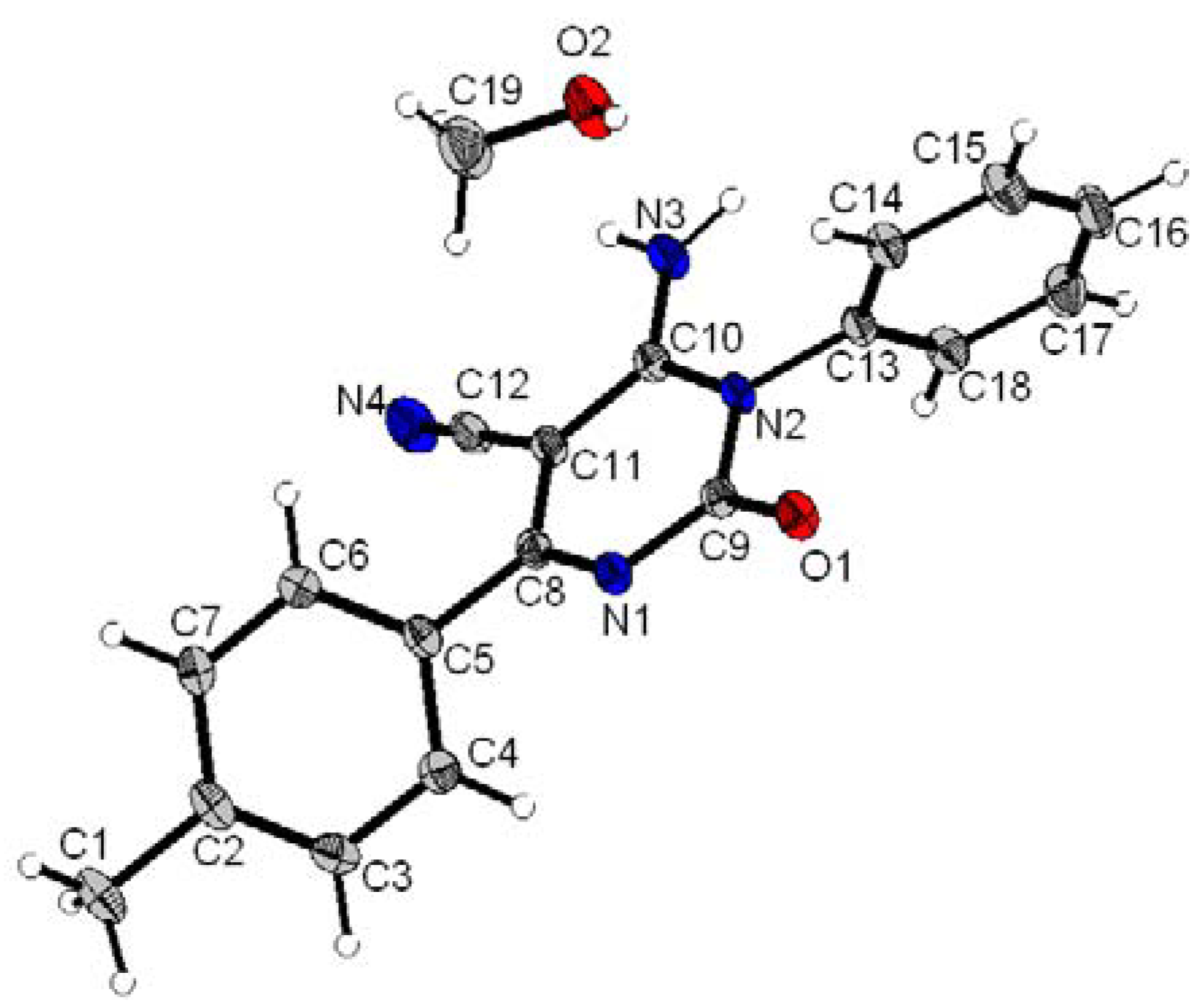
6-Amino-5-cyano-4-(4-methylphenyl)-1-(3-(1-(phenylthiomethyl)-1H-1,2,3-triazol-4-yl)phenyl)-2(1H)-pyrimidinone (4a). White solid, yield (82%), C27H21N7OS, M = 491 gmol−1, mp 220–222 °C, Rf = 0.34 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 253 (57,447), 319 (13,993); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,225 (CN), 1,677 (C=O), 1,643 (C=N); 1H-NMR: (DMSO-d6): δ = 2.41 (s, 3H, CH3), 6.02 (s, 2H, CH2), 7.30–7.98 (m, 15H, Ar-H + NH2), 8.65 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 21 (CH3), 52.1 (CH2), 72.1 (C-5), 116.7 (CN), 121.3 (CHar-triazole), 125.3, 125.9, 127.8, 127.9, 128.3, 128.8, 129.3, 130.6, 130.7, 132.2, 132.4, 134.1, 135.3, 141, 146 (Cq-triazole), 153.5 (C-2), 160 (C-4), 171.3 (C-6); MS-(+)ESI: m/z (%): 983 ([2M + H]+, 19), 514 ([M + Na]+, 7), 492 ([M+H]+, 100), MS-(-)ESI: m/z (%): 464 (26), 354 (8).
6-Amino-5-cyano-4-(4-methylphenyl)-1-(4-(1-(phenylthiomethyl)-1H-1,2,3-triazol-4-yl)phenyl)-2(1H)-pyrimidinone (4b). White solid, yield (72%), C27H21N7OS, M = 491 gmol−1, mp 259–261 °C, Rf = 0.27 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 251 (46,399), 318 (15,466); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,226 (CN), 1,681 (C=O), 1,648 (C=N); 1H-NMR: (DMSO-d6): δ = 2.4 (s, 3H, CH3), 6.08 (s, 2H, CH2), 7.29–8.11 (m, 15H, Ar-H + NH2), 8.72 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 21 (CH3), 52.3 (CH2), 72.7 (C-5), 116.5 (CN), 121.7 (CHar-triazole), 125.6, 127.4, 127.7, 128.6, 129.7, 130.5, 130.9, 132.1, 132.5, 133.9, 135.4, 140.9, 146.1 (Cq-triazole), 154 (C-2), 160.1 (C-4), 171.4 (C-6); MS-(+)ESI: m/z (%): 983 ([2M + H]+, 20), 514 ([M + Na]+, 8), 492 ([M+H]+, 100), MS-(-)ESI: m/z (%): 464 (22), 354 (9).
6-Amino-4-benzyl-5-cyano-1-(3-(1-(phenylthiomethyl)-1H-1,2,3-triazol-4-yl)phenyl)-2(1H)-pyrimidin-one (4c). White solid, yield (80%), C27H21N7OS, M = 491 gmol−1, mp 235–237 °C, Rf = 0.36 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 250 (62,602), 318 (19,939); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,208 (CN), 1,668 (C=O), 1,616 (C=N); 1H-NMR: (DMSO-d6): δ = 3.90 (s, 2H, CH2), 6.13 (s, 2H, CH2), 7.27–7.65 (m, 16H, Ar-H + NH2), 8.85 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 43.4 (CH2), 52.2 (CH2), 73.4 (C-5), 117 (CN), 122.3 (CHar-triazole), 124.9, 125.6, 126.9, 127.7, 128.3, 129.2, 129.6, 131.2, 131.8, 133.1, 133.4, 134.7, 136.5, 140.9, 147.3 (Cq-triazole), 154 (C-2), 159.6 (C-4), 175.7 (C-6); MS-(+)ESI: m/z (%): 983 ([2M + H]+, 19), 514 ([M + Na]+, 7), 492 ([M+H]+, 100), MS-(-)ESI: m/z (%): 464 (2), 354 (8).
6-Amino-4-benzyl-5-cyano-1-(4-(1-(phenylthiomethyl)-1H-1,2,3-triazol-4-yl)phenyl)-2(1H)-pyrimidin-one (4d). White solid, yield (75%), C27H21N7OS, M = 491 gmol−1, mp 262–264 °C, Rf = 0.30 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 248 (61,129), 319 (18,412); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,226 (CN), 1,685 (C=O), 1,647 (C=N); 1H-NMR: (DMSO-d6): δ = 3.9 (s, 2H, CH2), 6.23 (s, 2H, CH2), 7.2–7.91 (m, 16H, Ar-H + NH2), 8.64 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 43.5 (CH2), 51.9 (CH2), 73.7 (C-5), 116.5 (CN), 120.5 (CHar-triazole), 126, 126.9, 127.2, 129.1, 129.9, 129.9, 129.9, 131.9, 132.2, 134.2, 136.4, 141.2, 146.7 (Cq-triazole), 153.5 (C-2), 159.3 (C-4), 175.6 (C-6); MS-(+)ESI: m/z (%): 983 ([2M + H]+, 22), 514 ([M + Na]+, 7), 492 ([M+H]+, 100), MS-(-)ESI: m/z (%): 464 (25), 354 (9).
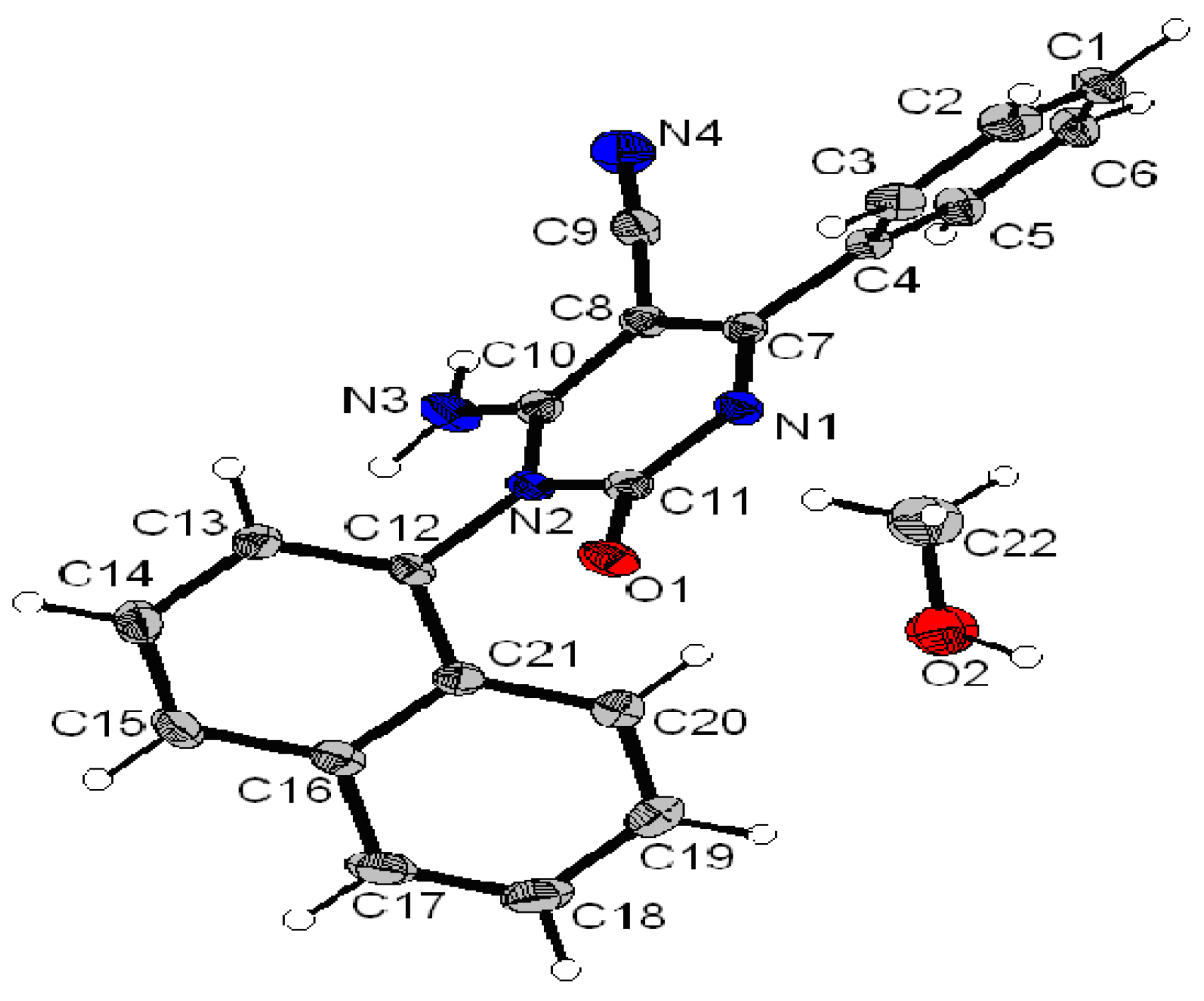
6-Amino-5-cyano-1-(3-(1-(4-(isopropylamino)-6-(methylthio)-1,3,5-triazin-2-yl)-1H-1,2,3-triazol-4-yl)-phenyl)-4-(4-methylphenyl)-2(1H)-pyrimidinone (4e). Pale yellow solid, yield (73%), C27H25N11OS, M = 551 gmol−1, mp 230–232 °C, Rf = 0.39 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 248 (71,905), 318 (19,009); IR (KBr) cm−1: 3,450–3,310 (NH2 + NH), 2,208 (CN), 1,671 (C=O), 1,627 (C=N); 1H-NMR: (DMSO-d6): δ = 1.21 (d, 3H, J = 9 Hz, CH3), 1.23 (d, 3H, J = 9 Hz, CH3), 2.41 (s, 3H, CH3), 2.59 (s, 3H, -SCH3), 4.3 (m, 1H, CH), 7.25–7.77 (m, 11H, Ar-H + NH2 + NH), 8.1 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 21.2 (SCH3), 21.5 (CH3), 22.3 (CH3), 22.6 (CH3), 42.8 (CH), 73.8 (C-5), 117.2 (CN), 120.7 (CHar-triazole), 126.3, 128.8, 129.3, 129.4, 131.3, 132.1, 132.1, 134.6, 135.9, 141.5, 146.4 (Cq-triazole), 154 (C-2), 160.5 (C-4), 164, 171.8 (C-6), 182.1, 183.1; MS-(+)ESI: m/z (%): 574 ([M + Na]+, 7), 552 ([M+H]+, 100), MS-(-)ESI: m/z (%): 524 (64), 482 (7).
6-Amino-5-cyano-1-(4-(1-(4-(isopropylamino)-6-(methylthio)-1,3,5-triazin-2-yl)-1H-1,2,3-triazol-4-yl)-phenyl)-4-(4-methylphenyl)-2(1H)-pyrimidinone (4f). white solid, yield (94%), C27H25N11OS, M = 551 gmol−1, mp 253–255 °C, Rf = 0.31 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 251 (64,467), 319 (15,703); IR (KBr) cm−1: 3,450–3,310 (NH2 + NH), 2,225 (CN), 1,663 (C=O), 1,638 (C=N); 1H-NMR: (DMSO-d6): δ = 1.21 (d, 3H, J = 9 Hz, CH3), 1.23 (d, 3H, J = 9 Hz, CH3), 2.4 (s, 3H, CH3), 2.61 (s, 3H, -SCH3), 4.31 (m, 1H, CH), 7.21–7.75 (m, 11H, Ar-H + NH2 + NH), 8.21 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 21.1 (SCH3), 21.5 (CH3), 22.3 (CH3), 22.5 (CH3), 42.9 (CH), 73.8 (C-5), 116.9 (CN), 120.6 (CHar-triazole), 124.2, 128.8, 129.6, 131.4, 133.3, 134.6, 136, 141.3, 146.7 (Cq-triazole), 153.9 (C-2), 160.3 (C-4), 163.9, 171.7 (C-6), 182.1, 183.2; MS-(+)ESI: m/z (%): 574 ([M + Na]+, 7), 552 ([M+H]+, 100), MS-(-)ESI: m/z (%): 524 (54), 482 (9).
6-Amino-4-benzyl-5-cyano-1-(3-(1-(4-(isopropylamino)-6-(methylthio)-1,3,5-triazin-2-yl)-1H-1,2,3-triazol-4-yl)phenyl)-2(1H)-pyrimidinone (4g). Yellowish solid, yield (88%), C27H25N11OS, M = 551 gmol−1, mp 241–243 °C, Rf = 0.37 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 249 (50,416), 307 (27,274); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,206 (CN), 1,670 (C=O), 1,629 (C=N); 1H-NMR: (DMSO-d6): δ = 1.21 (d, 3H, J = 9 Hz, CH3), 1.23 (d, 3H, J = 9 Hz, CH3), 2.59 (s, 3H, -SCH3), 3.9 (s, 2H, CH2), 4.3 (m, 1H, CH), 7.27–7.43 (m, 12H, Ar-H + NH2 + NH), 8.29 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 22.2 (SCH3), 22.3 (CH3), 22.5 (CH3), 42.8 (CH), 43.4 (CH2), 73.8 (C-5), 116.6 (CN), 120.8 (CHar-triazole), 127.4, 128, 128.3, 129, 129.8, 130, 131.3, 135.2, 137.3, 146.5 (Cq-triazole), 153.9 (C-2), 160.2 (C-4), 163.8, 164.2, 175.4 (C-6), 182.1, 183; MS-(+)ESI: m/z (%): 574 ([M + Na]+, 9), 552 ([M+H]+, 100), MS-(-)ESI: m/z (%): 524 (61), 482 (7).
6-Amino-4-benzyl-5-cyano-1-(4-(1-(4-(isopropylamino)-6-(methylthio)-1,3,5-triazin-2-yl)-1H-1,2,3-triazol-4-yl)phenyl)-2(1H)-pyrimidinone (4h). White solid, yield (71%), C27H25N11OS, M = 551 gmol−1, mp 274–276 °C, Rf = 0.32 (ethyl acetate/dichloromethane, 70:30, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 250 (52,069), 308 (25,621); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,226 (CN), 1,685 (C=O), 1,642 (C=N); 1H-NMR: (DMSO-d6): δ = 1.21 (d, 3H, J = 9 Hz, CH3), 1.23 (d, 3H, J = 9 Hz, CH3), 2.60 (s, 3H, -SCH3), 3.9 (s, 2H, CH2), 4.3 (m, 1H, CH), 7.36–7.47 (m, 12H, Ar-H + NH2 + NH), 8.3 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 21.5 (SCH3), 22.3 (CH3), 22.5 (CH3), 42.8 (CH), 43.4 (CH2), 73.8 (C-5), 116.5 (CN), 120.7 (CHar-triazole), 127.2, 127.8, 128.9, 129.6, 131.1, 135, 137.1, 146.5 (Cq-triazole), 154.1 (C-2), 159.7 (C-4), 163.6, 164, 175.4 (C-6), 182.1, 183.1; MS-(+)ESI: m/z (%): 574 ([M + Na]+, 7), 552 ([M+H]+, 100), MS-(-)ESI: m/z (%): 524 (56), 482 (7).
(Z)-Ethyl-2-(4-(4-(3-(6-amino-5-cyano-4-(4-methylphenyl)-2-oxopyrimidin-1(2H)-yl)phenyl)-1H-1,2,3-triazol-1-yl)benzylidene)-7-methyl-3-oxo-5-phenyl-3,5-dihydro-2H-thiazolo[3,2-a]pyrimidine-6-carboxylate (4i). Golden yellow solid, yield (84%), C43H33N9O4S, M = 771 gmol−1, mp 285–287 °C, Rf = 0.28 (ethyl acetate/dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 248 (95,989), 308 (35,851); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,211 (CN), 1,715 (C=O, ester), 1,677 (C=O); 1H-NMR: (DMSO-d6): δ = 1.13 (t, 3H, J = 6 Hz, CH3), 2.40 (s, 3H, CH3), 2.41 (s, 3H, CH3), 4.04 (q, 2H, J = 6 Hz, CH2), 6.06 (s, 1H, C-CH-N), 7.31–8.14 (m, 20H, Ar-H + NH2), 9.47 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 14.3 (CH3-CH2), 21.5 (CH3), 22.9 (CH3), 55.5 (C-CH-N), 60.7 (CH3-CH2), 72.6 (C-5), 109.4, 117.1 (CN), 120.4, 120.8 (CHar-triazole), 121.3, 125.9, 126.7, 127.9, 128.8, 129.1, 129.2, 129.3, 131.5, 131.9, 132.1, 132.5, 133.4, 134.6, 135.9, 137.7, 140.7, 141.5, 147.3 (Cq-triazole), 151.5, 154 (C-2), 155.7, 160.5 (C-4), 164.6 (C=O), 165.3, 171.9 (C-6); MS-(+)ESI: m/z (%): 794 ([M + Na]+, 3), 772 ([M+H]+, 100), MS-(-)ESI: m/z (%): 744 (10).
(Z)-Ethyl-2-(4-(4-(4-(6-amino-5-cyano-4-(4-methylphenyl)-2-oxopyrimidin-1(2H)-yl)phenyl)-1H-1,2,3-triazol-1-yl)benzylidene)-7-methyl-3-oxo-5-phenyl-3,5-dihydro-2H-thiazolo[3,2-a]pyrimidine-6-carboxylate (4j). Golden yellow solid, yield (72%), C43H33N9O4S, M = 771 gmol−1, mp 292–294 °C, Rf = 0.25 (ethyl acetate/dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 249 (84,424), 307 (38,164); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,225 (CN), 1,727 (C=O, ester), 1,685 (C=O); 1H-NMR: (DMSO-d6): δ = 1.15 (t, 3H, J = 6 Hz, CH3), 2.41 (s, 3H, CH3), 2.43 (s, 3H, CH3), 4.11 (q, 2H, J = 6 Hz, CH2), 6.02 (s, 1H, C-CH-N), 7.29–8.20 (m, 20H, Ar-H + NH2), 9.32 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 14.3 (CH3-CH2), 21.5 (CH3), 22.9 (CH3), 55.5 (C-CH-N), 60.6 (CH3-CH2), 72.6 (C-5), 109.4, 117.4 (CN), 120.7, 120.8 (CHar-triazole), 122, 126.2, 126.7, 128.1, 129, 129.3, 131.6, 132, 132.2, 132.6, 133.6, 134.5, 136.1, 137.7, 140.6, 141.4, 147.5 (Cq-triazole), 151.7, 152.2 (C-2), 156.2, 161.2 (C-4), 165.1 (C=O), 165.6, 172.5 (C-6); MS-(+)ESI: m/z (%): 794 ([M + Na]+, 3), 772 ([M+H]+, 100), MS-(-)ESI: m/z (%): 744 (7).
(Z)-Ethyl-2-(4-(4-(3-(6-amino-4-benzyl-5-cyano-2-oxopyrimidin-1(2H)-yl)phenyl)-1H-1,2,3-triazol-1-yl)benzylidene)-7-methyl-3-oxo-5-phenyl-3,5-dihydro-2H-thiazolo[3,2-a]pyrimidine-6-carboxylate (4k). Golden yellow solid, yield (81%), C43H33N9O4S, M = 771 gmol−1, mp 277–279 °C, Rf = 0.32 (ethyl acetate/dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 250 (72,859), 318 (27,756); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,212 (CN), 1,721 (C=O, ester), 1,671 (C=O); 1H-NMR: (DMSO-d6): δ = 1.12 (t, 3H, J = 6 Hz, CH3), 2.40 (s, 3H, CH3), 3.9 (s, 2H, CH2), 4.09 (q, 2H, J = 6 Hz, CH2), 5.98 (s, 1H, C-CH-N), 7.33–8.02 (m, 21H, Ar-H + NH2), 9.33 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 14.3 (CH3-CH2), 22.9 (CH3), 43.5 (CH2), 55.4 (C-CH-N), 60.7 (CH3-CH2), 73.4 (C-5), 110.2, 116.5 (CN), 119.9, 121.2 (CHar-triazole), 122, 126, 126.6, 128.3, 128.9, 129, 129.8, 130.1, 131.5, 132, 132.4, 132.9, 134.2, 134.7, 136.1, 137.5, 139.9, 142, 146.7 (Cq-triazole), 150.9, 153.6 (C-2), 156.2, 159.6 (C-4), 164.5 (C=O), 166.2, 175.8 (C-6); MS-(+)ESI: m/z (%): 794 ([M + Na]+, 3), 772 ([M+H]+, 100), MS-(-)ESI: m/z (%): 744 (9).
(Z)-ethyl-2-(4-(4-(4-(6-amino-4-benzyl-5-cyano-2-oxopyrimidin-1(2H)-yl)phenyl)-1H-1,2,3-triazol-1-yl)benzylidene)-7-methyl-3-oxo-5-phenyl-3,5-dihydro-2H-thiazolo[3,2-a]pyrimidine-6-carboxylate (4l). Golden yellow solid, yield (68%), C43H33N9O4S, M = 771 gmol−1, mp 281–283 °C, Rf = 0.61 (ethyl acetate/dichloromethane, 50:50, v/v); UV (MeOH) λmax nm (ε Lmol−1cm−1): 253 (90,207), 319 (15,381); IR (KBr) cm−1: 3,450–3,310 (NH2), 2,226 (CN), 1,728 (C=O, ester), 1,676 (C=O); 1H-NMR: (DMSO-d6): δ = 1.15 (t, 3H, J = 6 Hz, CH3), 2.41 (s, 3H, CH3), 3.89 (s, 2H, CH2), 4.12 (q, 2H, J = 6 Hz, CH2), 6.00 (s, 1H, C-CH-N), 7.28–8.46 (m, 21H, Ar-H + NH2), 9.40 (s, 1H, CHar-triazole); 13C-NMR (DMSO-d6): δ = 14.3 (CH3-CH2), 22.9 (CH3), 43.5 (CH2), 56.2 (C-CH-N), 60.7 (CH3-CH2), 73.8 (C-5), 112.5, 117.2 (CN), 120.3, 120.9 (CHar-triazole), 121.9, 126.3, 128.6, 129.4, 129.5, 129.9, 131.6, 131.8, 133.5, 133, 135.2, 135.4, 135.9, 137.6, 140.1, 142.2, 147.7 (Cq-triazole), 149.5, 153.9 (C-2), 157, 159.9 (C-4), 164.6 (C=O), 167.2, 174.9 (C-6); MS-(+)ESI: m/z (%): 794 ([M + Na]+, 3), 772 ([M+H]+, 100), MS-(-)ESI: m/z (%): 744 (6).