Monocyclic Phenolic Acids; Hydroxy- and Polyhydroxybenzoic Acids: Occurrence and Recent Bioactivity Studies
Abstract
:1. Introduction
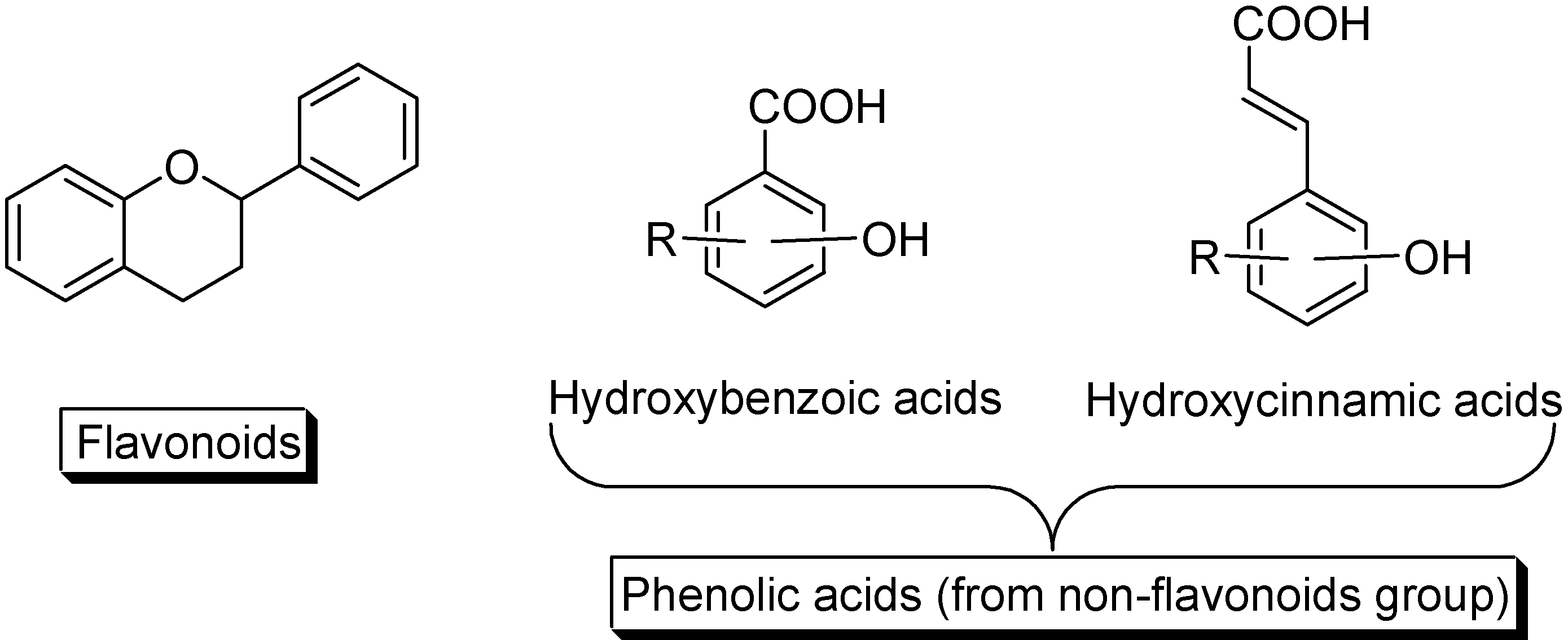
2. Results and Discussion





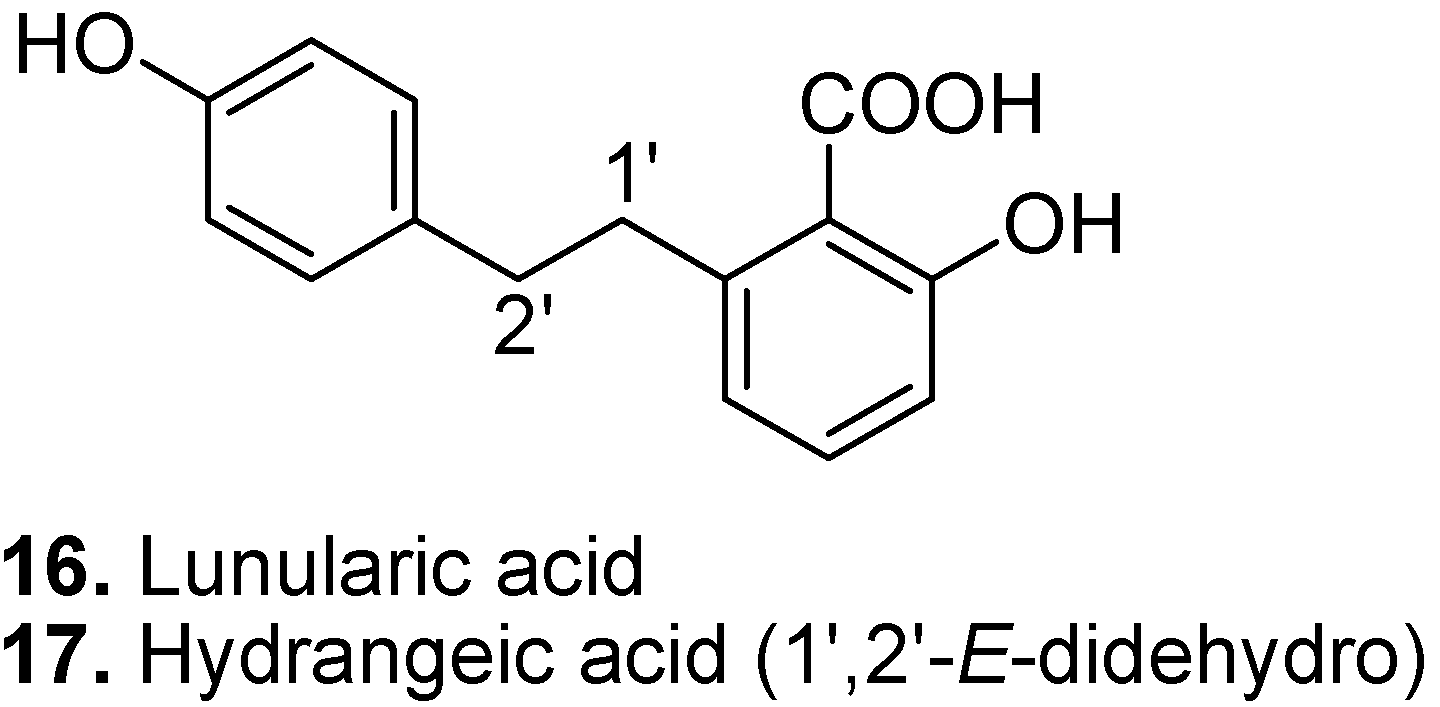
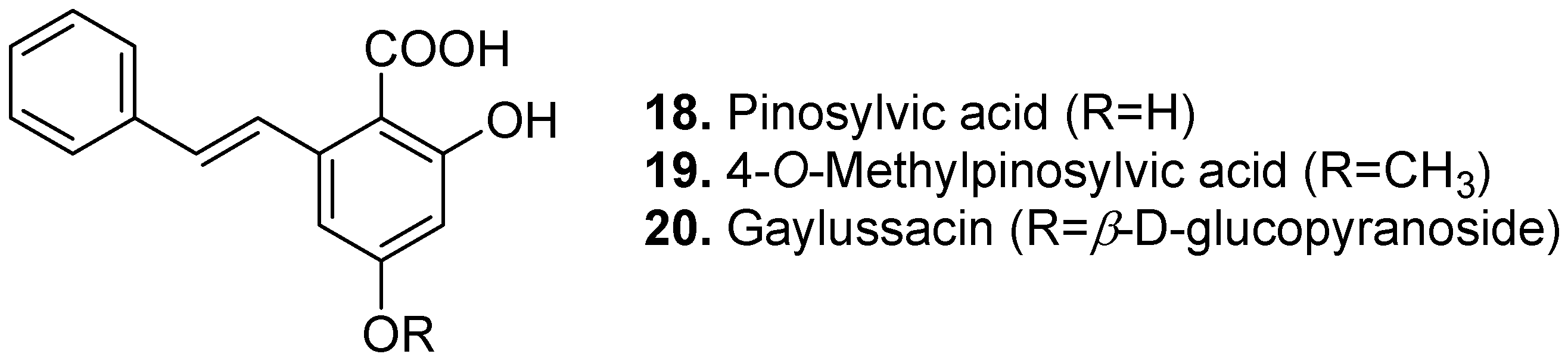
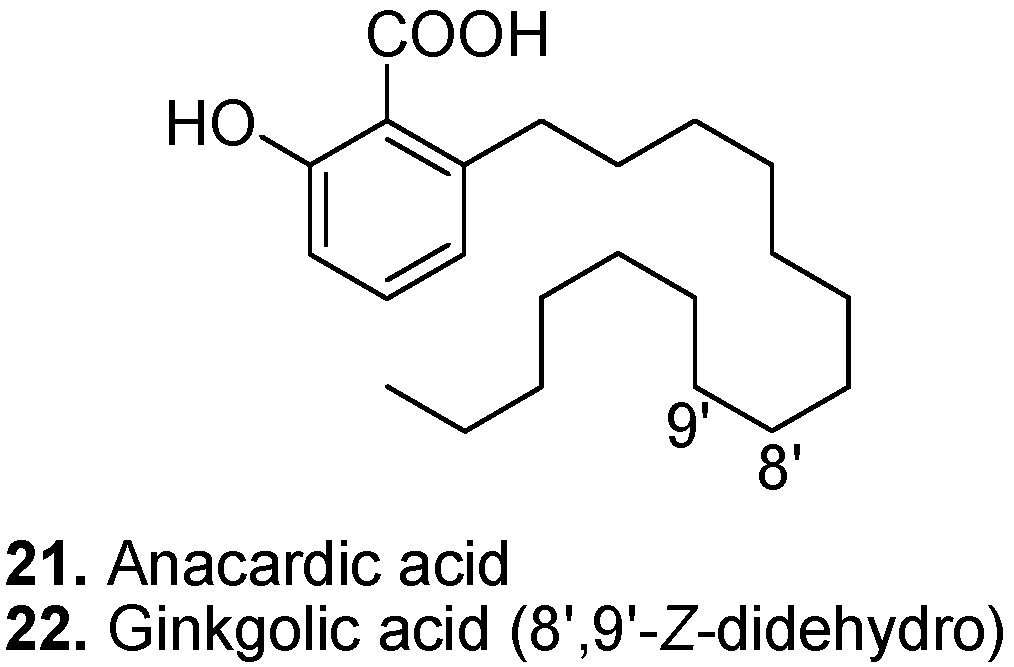
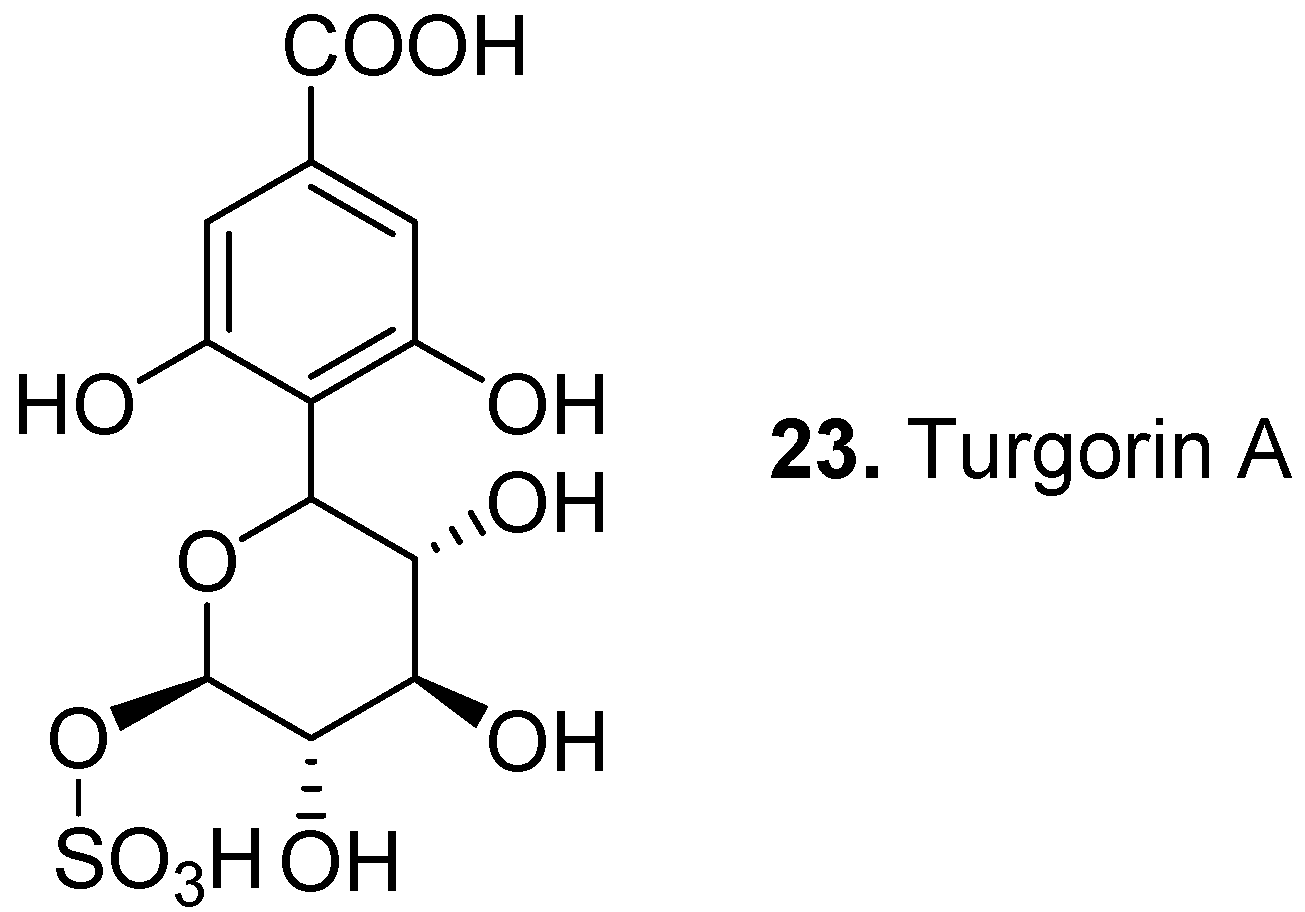
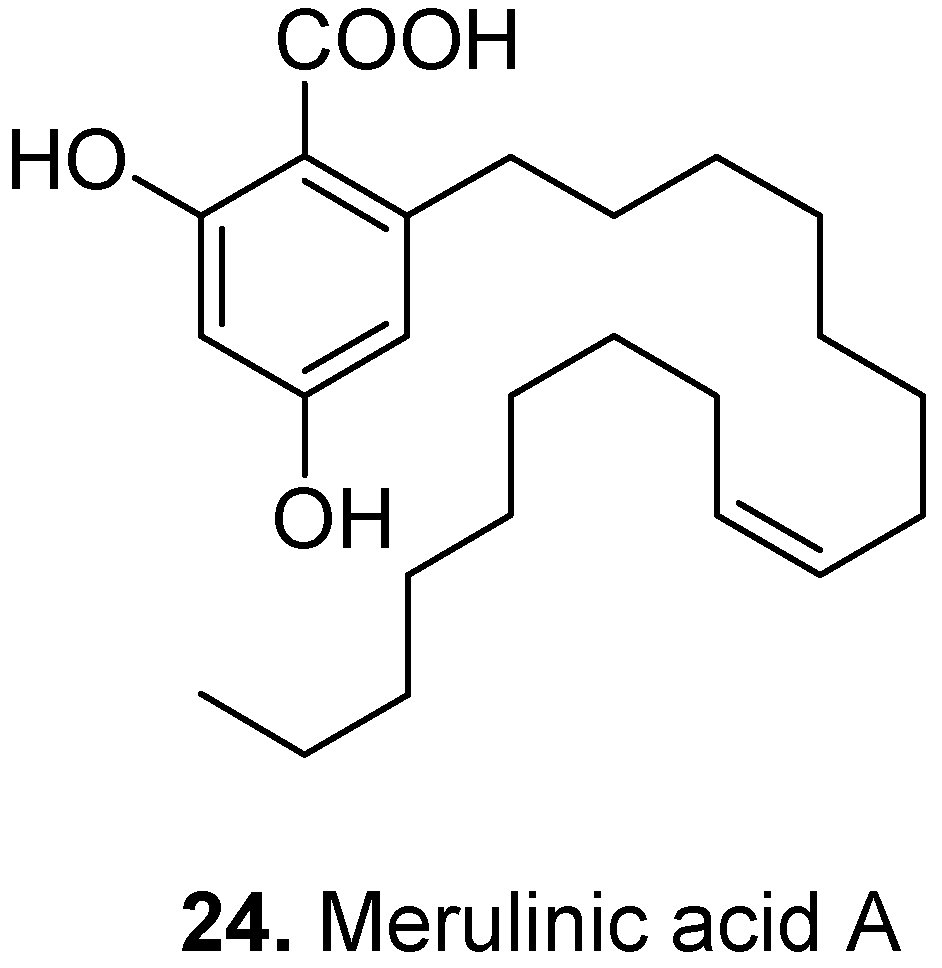
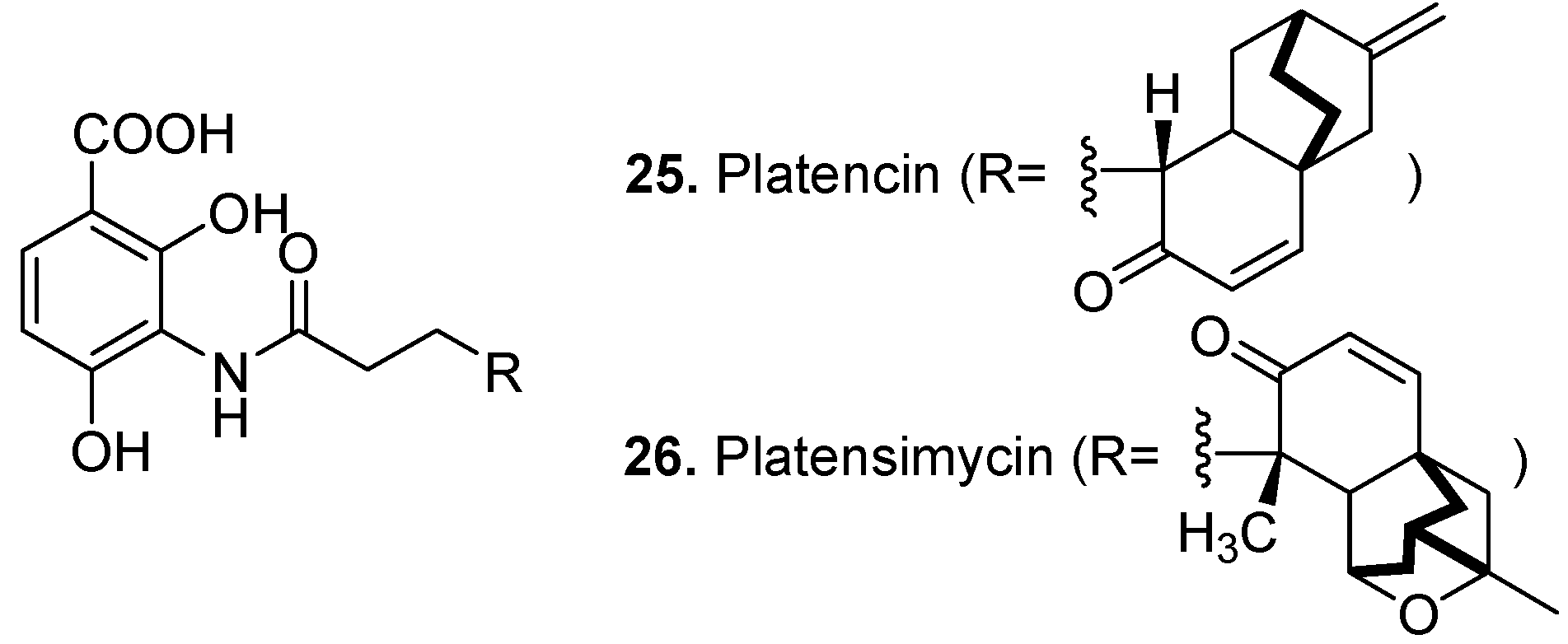
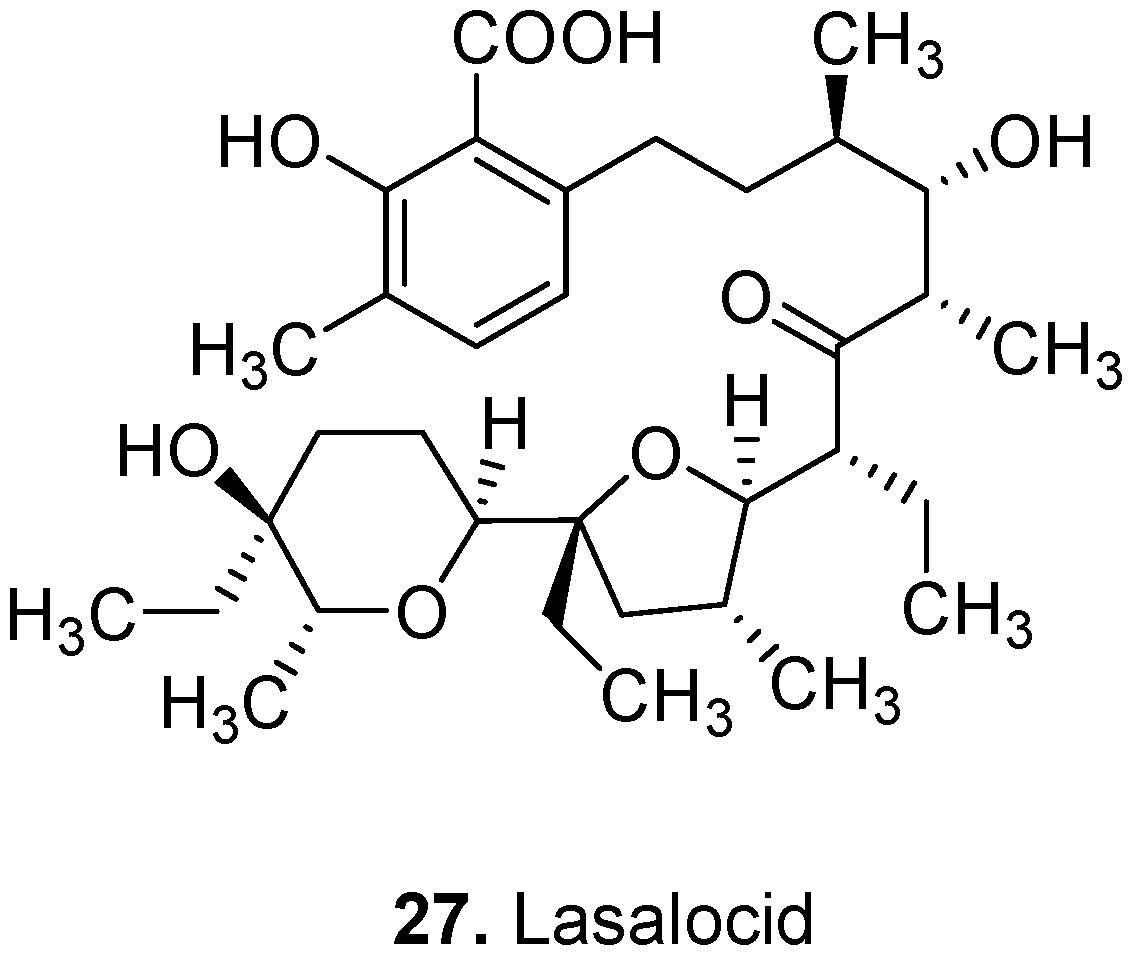
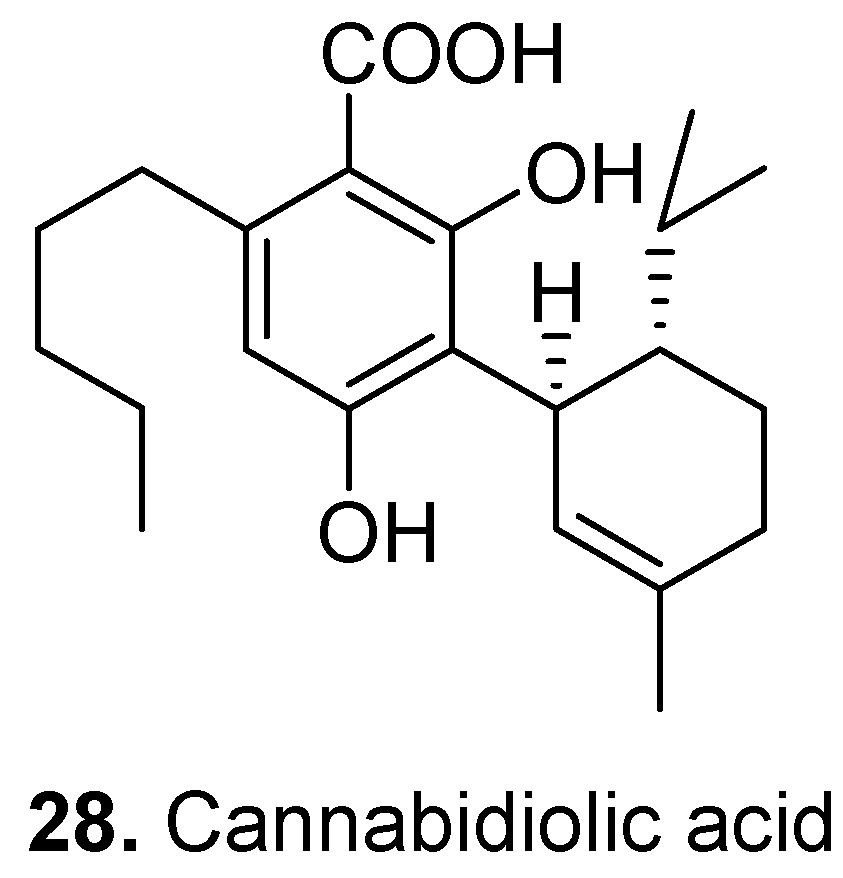
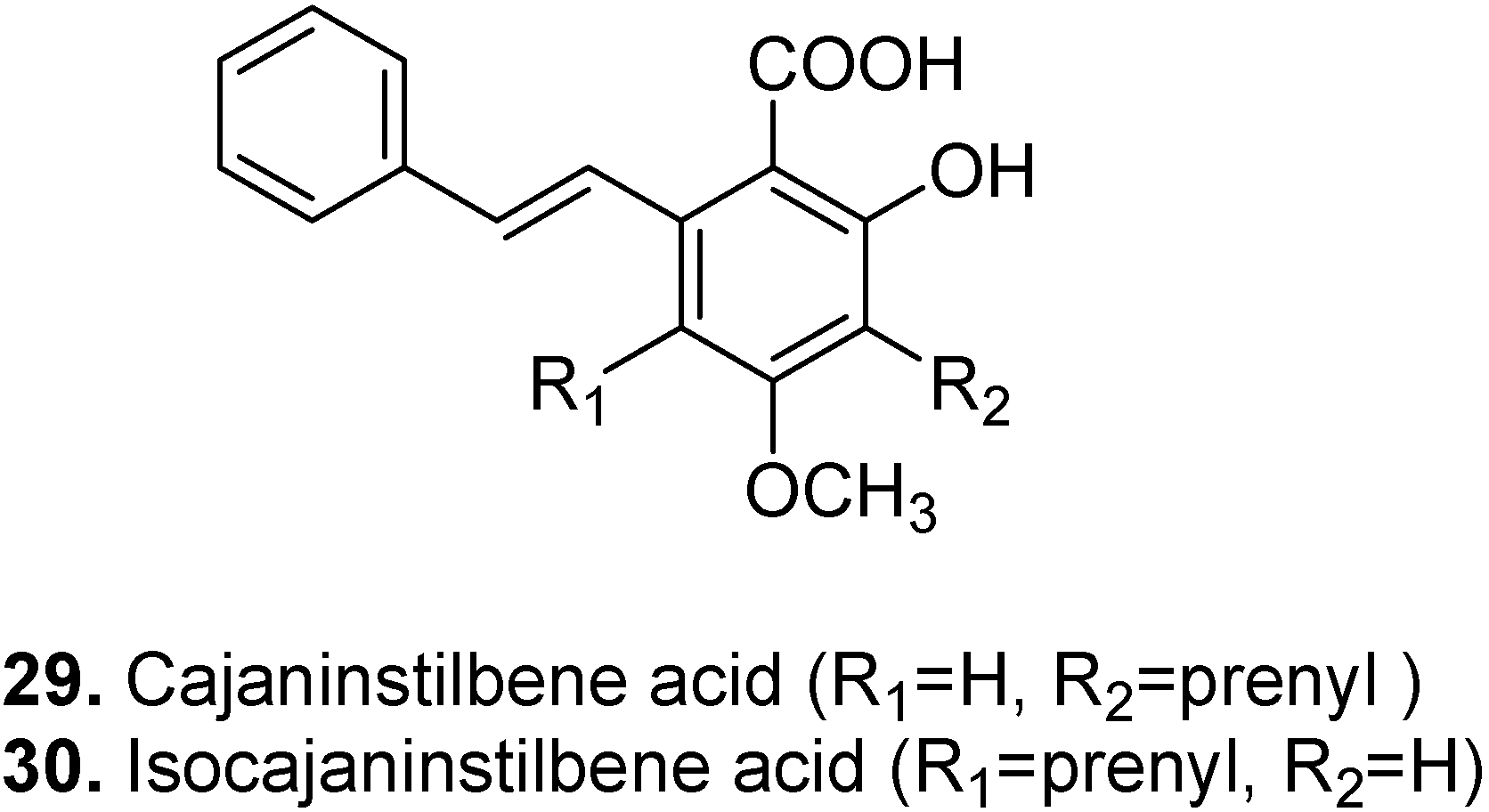
3. Conclusions
Acknowledgements
References
- Martin, K.R.; Appel, C.L. Polyphenols as dietary supplements: A double-edged sword. Nutr. Dietary Suppl. 2010, 2, 1–12. [Google Scholar]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef]
- Harris, C.S.; Mo, F.; Migahed, L.; Chepelev, L.; Haddad, P.S.; Wright, J.S.; Willmore, W.G.; Arnason, J.T.; Bennett, S.A.L. Plant phenolics regulate neoplastic cell growth and survival: a quantitative structure-activity and biochemical analysis. Can. J. Physiol. Pharmacol. 2007, 85, 1124–1138. [Google Scholar] [CrossRef]
- Huang, W.Y.; Cai, Y.Z.; Zhang, Y.B. Natural Phenolic Compounds From Medicinal Herbs and Dietary Plants: Potential Use for Cancer Prevention. Nutr. Cancer 2010, 62, 1–20. [Google Scholar]
- Liu, R.H. Potential synergy of phytochemicals in cancer prevention: Mechanism of action. J. Nutr. 2004, 134, 3479S–3485S. [Google Scholar]
- Stalikas, C.D. Extraction, separation, and detection methods for phenolic acids and flavonoids. J. Sep. Sci. 2007, 30, 3268–3295. [Google Scholar] [CrossRef]
- de la Rosa, L.A.; Alvarez-Parrilla, E.; Gonzalez-Aguilar, G.A. Fruit and Vegetable Phytochemicals- Chemistry, Nutritional Value, and Stability, 1st ed; Wiley-Blackwell: Ames, IA, USA, 2010. [Google Scholar]
- Lafay, S.; Gil-Izquierdo, A. Bioavailability of Phenolic acids. Phytochem. Rev. 2008, 7, 301–311. [Google Scholar] [CrossRef]
- Robbins, R.J. Phenolic acids in foods: An overview of analytical methodology. J. Agr. Food Chem. 2003, 51, 2866–2887. [Google Scholar] [CrossRef]
- Harborne, J.B.; Baxter, H.; Moss, G.P. Phytochemical Dictionary—A Handbook of Bioactive Compounds from Plants, 2nd ed; Taylor & Francis: London, UK, 1999; p. 976. [Google Scholar]
- Plant Phenolics.; Harborne, J.B. (Ed.) Academic Press: San Diego, CA, USA, 1989.
- CRC-DNP, The Dictionary of Natural Products; Chapman & Hall: London, UK, 2010.
- Bendini, A.; Cerretani, L.; Carrasco-Pancorbo, A.; Gomez-Caravaca, A.M.; Segura-Carretero, A.; Fernandez-Gutierrez, A.; Lercker, G. Phenolic molecules in virgin olive oils: a survey of their sensory properties, health effects, antioxidant activity and analytical method. An overview of the last decade. Molecules 2007, 12, 1679–1719. [Google Scholar] [CrossRef]
- Gruz, J.; Ayaz, F.A.; Torun, H.; Strand, M. Phenolic acid content and radical scavenging activity of extracts from medlar (Mespilus germanica L.) fruit at different stages of ripening. Food Chem. 2011, 124, 271–277. [Google Scholar] [CrossRef]
- Ford, C.M.; Hoj, P.B. Multiple glucosyltransferase activities in the grapevine Vitis vinifera L. Aust. J. Grape and Wine Res. 1998, 4, 48–58. [Google Scholar] [CrossRef]
- Sircar, D.; Mitra, A. Accumulation of p-hydroxybenzoic acid in hairy roots of Daucus carota 2: Confirming biosynthetic steps through feeding of inhibitors and precursors. J. Plant Physiol. 2009, 166, 1370–1380. [Google Scholar] [CrossRef]
- Chong, K.P.; Rossall, S.; Atong, M. In Vitro Antimicrobial Activity and Fungitoxicity of Syringic Acid, Caffeic Acid and 4-hydroxybenzoic Acid against Ganoderma Boninense. J. Agr. Sci. 2009, 1, 15–20. [Google Scholar]
- Ling, W.C.; Ahmad, F.; Mat Ali, R. Luteolin and 4-Hydroxybenzoic Acid from the Leaves Vitex negundo L. Malaysian J. Sci. 2005, 24, 133–135. [Google Scholar]
- Chakraborty, M.; Das, K.; Dey, G.; Mitra, A. Unusually high quantity of 4-hydroxybenzoic acid accumulation in cell wall of palm mesocarps. Biochem. Syst. Ecol. 2006, 34, 509–513. [Google Scholar] [CrossRef]
- Pugazhendhi, D.; Pope, G.S.; Darbre, P.D. Oestrogenic activity of p-hydroxybenzoic acid (common metabolite of paraben esters) and methylparaben in human breast cancer cell lines. J. Appl. Toxicol. 2005, 25, 301–309. [Google Scholar] [CrossRef]
- Kamaya, Y.; Tsuboi, S.; Takada, T.; Suzuki, K. Growth stimulation and inhibition effects of 4-hydroxybenzoic acid and some related compounds on the freshwater green alga Pseudokirchneriella subcapitata. Arch. Environ. Contam. Toxicol. 2006, 51, 537–541. [Google Scholar] [CrossRef]
- Muljono, R.A.B.; Darsono, F.L.; Scheffer, J.J.C.; Verpoorte, R. Assay of 2,3-dihydroxybenzoic acid and related compounds in plant materials by high-performance liquid chromatography. J. Chromatogr. A 2001, 927, 39–45. [Google Scholar] [CrossRef]
- Sugiyama, Y.; Hirota, A. New Potent DPPH Radical Scavengers from a Marine-Derived Actinomycete Strain USF-TC31. Biosci. Biotechnol. Biochem. 2009, 73, 2731–2734. [Google Scholar] [CrossRef]
- Onofrejova, L.; Vasickova, J.; Klejdus, B.; Stratil, P.; Misurcova, L.; Kracmar, S.; Kopecky, J.; Vacek, J. Bioactive phenols in algae: The application of pressurized-liquid and solid-phase extraction techniques. J. Pharm. Biomed. Anal. 2010, 51, 464–470. [Google Scholar] [CrossRef]
- Sroka, Z.; Cisowski, W. Hydrogen peroxide scavenging, antioxidant and anti-radical activity of some phenolic acids. Food Chem. Toxicol. 2003, 41, 753–758. [Google Scholar] [CrossRef]
- Parent, M.A.; Bellaire, B.H.; Murphy, E.A.; Roop, R.M.; Elzer, P.H.; Baldwin, C.L. Brucella abortus siderophore 2,3-dihydroxybenzoic acid (DHBA) facilitates intracellular survival of the bacteria. Microb. Pathog. 2002, 32, 239–248. [Google Scholar] [CrossRef]
- Ashidate, K.; Kawamura, M.; Mimura, D.; Tohda, H.; Miyazaki, S.; Teramoto, T.; Yamamoto, Y.; Hirata, Y. Gentisic acid, an aspirin metabolite, inhibits oxidation of low-density lipoprotein and the formation of cholesterol ester hydroperoxides in human plasma. Eur. J. Pharmacol. 2005, 513, 173–179. [Google Scholar] [CrossRef]
- Sharma, S.; Khan, N.; Sultana, S. Study on prevention of two-stage skin carcinogenesis by Hibiscus rosa sinensis extract and the role of its chemical constituent, gentisic acid, in the inhibition of tumour promotion response and oxidative stress in mice. Eur. J. Cancer Prev. 2004, 13, 53–63. [Google Scholar] [CrossRef]
- Fernandez, I.S.; Cuevas, P.; Angulo, J.; Lopez-Navajas, P.; Canales-Mayordomo, A.; Gonzalez-Corrochano, R.; Lozano, R.M.; Valverde, S.; Jimenez-Barbero, J.; Romero, A.; Gimenez-Gallego, G. Gentisic Acid, a Compound Associated with Plant Defense and a Metabolite of Aspirin, Heads a New Class of in vivo Fibroblast Growth Factor Inhibitors. J. Biol. Chem. 2010, 285, 11714–11729. [Google Scholar]
- Stochmal, A.; Kowalska, I.; Janda, B.; Perrone, A.; Piacente, S.; Oleszek, W. Gentisic acid conjugates of Medicago truncatula roots. Phytochemistry 2009, 70, 1272–1276. [Google Scholar] [CrossRef]
- Sultana, N.; Akhter, M.; Khatoon, Z. Nematicidal natural products from the aerial parts of Rubus niveus. Nat. Prod. Res. 2010, 24, 407–415. [Google Scholar] [CrossRef]
- Lin, A.N.; Nakatsui, T. Salicylic acid revisited. Int. J. Dermatol. 1998, 37, 335–342. [Google Scholar] [CrossRef]
- Shah, J. The salicylic acid loop in plant defense. Curr. Opin. Plant Biol. 2003, 6, 365–371. [Google Scholar] [CrossRef]
- Loake, G.; Grant, M. Salicylic acid in plant defence-the players and protagonists. Curr. Opin. Plant Biol. 2007, 10, 466–472. [Google Scholar] [CrossRef]
- Wattallachaisaereekul, S.; Lantz, A.E.; Nielsen, M.L.; Andresson, O.S.; Nielsen, J. Optimization of heterologous production of the polyketide 6-MSA in Saccharomyces cerevisiae. Biotechnol. Bioeng. 2007, 97, 893–900. [Google Scholar] [CrossRef]
- Stuart, A.E.; Brooks, C.J.W.; Prescott, R.J.; Blackwell, A. Repellent and antifeedant activity of salicylic acid and related compounds against the biting midge, Culicoides impunctatus (Diptera : Ceratopogonidae). J. Med. Entomol. 2000, 37, 222–227. [Google Scholar] [CrossRef]
- Haschek, W.M.; Wallig, M.A.; Rousseaux, C. Fundamentals of Toxicologic Pathology, 2nd ed; Academic Press: San Diego, CA, USA, 2010. [Google Scholar]
- Hur, J.M.; Park, J.G.; Yang, K.H.; Park, J.C.; Park, J.R.; Chun, S.S.; Choi, J.S.; Choi, J.W. Effect of methanol extract of Zanthoxylum piperitum leaves and of its compound, protocatechuic acid, on hepatic drug metabolizing enzymes and lipid peroxidation in rats. Biosci. Biotechnol. Biochem. 2003, 67, 945–950. [Google Scholar] [CrossRef]
- Pan, Y.L.; Zhang, L.; Chen, G.N. Separation and determination of protocatechuic aldehyde and protocatechuic acid in Salivia miltorrhrza by capillary electrophoresis with amperometric detection. Analyst 2001, 126, 1519–1523. [Google Scholar] [CrossRef]
- An, L.J.; Guan, S.; Shi, G.F.; Bao, Y.M.; Duan, Y.L.; Jiang, B. Protocatechuic acid from Alpinia oxyphylla against MPP+-induced neurotoxicity in PC12 cells. Food Chem. Toxicol. 2006, 44, 436–443. [Google Scholar] [CrossRef]
- Gutzeit, D.; Wray, V.; Winterhalter, P.; Jerz, G. Preparative isolation and purification of flavonoids and protocatechuic acid from sea buckthorn juice concentrate (Hippophae rhamnoides L. ssp rhamnoides) by high-speed counter-current chromatography. Chromatographia 2007, 65, 1–7. [Google Scholar]
- Yip, E.C.H.; Chan, A.S.L.; Pang, H.; Tam, Y.K.; Wong, Y.H. Protocatechuic acid induces cell death in HepG2 hepatocellular carcinoma cells through a c-Jun N-terminal kinase-dependent mechanism. Cell Biol. Toxicol. 2006, 22, 293–302. [Google Scholar] [CrossRef]
- Fukuji, T.S.; Tonin, F.G.; Tavares, M.F.M. Optimization of a method for determination of phenolic acids in exotic fruits by capillary electrophoresis. J. Pharm. Biomed. Anal. 2010, 51, 430–438. [Google Scholar] [CrossRef]
- Vari, R.; D'Archivio, M.; Filesi, C.; Carotenuto, S.; Scazzocchio, B.; Santangelo, C.; Giovannini, C.; Masella, R. Protocatechuic acid induces antioxidant/detoxifying enzyme expression through JNK-mediated Nrf2 activation in murine macrophages. J. Nutr. Biochem. 2010, in press. [Google Scholar]
- Guan, S.; Jiang, B.; Bao, Y.M.; An, L.J. Protocatechuic acid suppresses MPP+-induced mitochondrial dysfunction and apoptotic cell death in PC12 cells. Food Chem. Toxicol. 2006, 44, 1659–1666. [Google Scholar] [CrossRef]
- Liu, Y.M.; Jiang, B.; Bao, Y.M.; An, L.J. Protocatechuic acid inhibits apoptosis by mitochondrial dysfunction in rotenone-induced PC12 cells. Toxicol. In Vitro 2008, 22, 430–437. [Google Scholar] [CrossRef]
- Yin, M.C.; Lin, C.C.; Wu, H.C.; Tsao, S.M.; Hsu, C.K. Apoptotic Effects of Protocatechuic Acid in Human Breast, Lung, Liver, Cervix, and Prostate Cancer Cells: Potential Mechanisms of Action. J. Agr. Food Chem. 2009, 57, 6468–6473. [Google Scholar] [CrossRef]
- Galvano, F.; Vitaglione, P.; Volti, G.L.; Di Giacomo, C.; Gazzo, D.; Vanella, L.; La Fauci, L.; Fogliano, V. Protocatechuic acid: The missing human cyanidins' metabolite. Mol. Nutr. Food Res. 2008, 52, 386–387. [Google Scholar] [CrossRef]
- Vitaglione, P.; Donnarumma, G.; Napolitano, A.; Galvano, F.; Gallo, A.; Scalfi, L.; Fogliano, V. Protocatechuic acid is the major human metabolite of cyanidin-glucosides. J. Nutr. 2007, 137, 2043–2048. [Google Scholar]
- Itoh, A.; Isoda, K.; Kondoh, M.; Kawase, M.; Kobayashi, M.; Tamesada, M.; Yagi, K. Hepatoprotective Effect of Syringic Acid and Vanillic Acid on Concanavalin A-Induced Liver Injury. Biol. Pharm. Bull. 2009, 32, 1215–1219. [Google Scholar] [CrossRef]
- Itoh, A.; Isoda, K.; Kondoh, M.; Kawase, M.; Watari, A.; Kobayashi, M.; Tamesada, M.; Yagi, K. Hepatoprotective Effect of Syringic Acid and Vanillic Acid on CCl4-Induced Liver Injury. Biol. Pharm. Bull. 2010, 33, 983–987. [Google Scholar] [CrossRef]
- Dhananjaya, B.L.; Nataraju, A.; Gowda, C.D.R.; Sharath, B.K.; D'Souza, C.J.M. Vanillic acid as a novel specific inhibitor of snake venom 5'-nucleotidase: A pharmacological tool in evaluating the role of the enzyme in snake envenomation. Biochem.-Moscow 2009, 74, 1315–1319. [Google Scholar] [CrossRef]
- Li, X.; Lin, L.; Wu, P.; Liu, M.; Wei, X. Chemical Constituents from Barks of Endospermum chinense Benth. J. Trop. Subtrop. Bot. 2007, 1, Q946. [Google Scholar]
- Zhao, X.; Ye, X.; Zhu, D. Isolation and identification of chemical constituents from peels of Citrus changshan-huyou Y.B. Chang. J. Peking Univ. 2009, 5, 575–577. [Google Scholar]
- Lai, Y.; Zhou, H. Isolation and Identification of the Chemical Constituents of Folium Fici Microcarpae. Food Drug 2008, 3, 11–13. [Google Scholar]
- Saroglou, V.; Karioti, A.; Rancic, A.; Dimas, K.; Koukoulitsa, C.; Zervou, M.; Skaltsa, H. Sesquiterpene Lactones from Anthemis melanolepis and Their Antibacterial and Cytotoxic Activities. Prediction of Their Pharmacokinetic Profile. J. Nat. Prod. 2010, 73, 242–246. [Google Scholar] [CrossRef]
- Zhong, H.; Ruan, J.L.; Yao, Q.Q. Two new 4-arylcoumarins from the seeds of Calophyllum polyanthum. J. Asian Nat. Prod. Res. 2010, 12, 562–568. [Google Scholar] [CrossRef]
- Komakine, N.; Okasaka, M.; Takaishi, Y.; Kawazoe, K.; Murakami, K.; Yamada, Y. New dammarane-type saponin from roots of Panax notoginseng. J. Nat. Med. 2006, 60, 135–137. [Google Scholar] [CrossRef]
- Wu, J.H.; Tung, Y.T.; Wang, S.Y.; Shyur, L.F.; Kuo, Y.H.; Chang, S.T. Phenolic antioxidants from the heartwood of Acacia confusa. J. Agr. Food Chem. 2005, 53, 5917–5921. [Google Scholar] [CrossRef]
- Tung, Y.T.; Wu, J.H.; Kuo, Y.H.; Chang, S.T. Antioxidant activities of natural phenolic compounds from Acacia confusa bark. Bioresour. Technol. 2007, 98, 1120–1123. [Google Scholar] [CrossRef]
- Kuete, V.; Metuno, R.; Ngameni, B.; Tsafack, A.M.; Ngandeu, F.; Fotso, G.W.; Bezabih, M.; Etoa, F.X.; Ngadjui, B.T.; Abegaz, B.M.; Beng, V.P. Antimicrobial activity of the methanolic extracts and compounds from Treculia obovoidea (Moraceae). J. Ethnopharmacol. 2007, 112, 531–536. [Google Scholar] [CrossRef]
- Li, C.Y.; Lee, E.J.; Wu, T.S. Antityrosinase principles and constituents of the petals of Crocus sativus. J. Nat. Prod. 2004, 67, 437–440. [Google Scholar] [CrossRef]
- Wang, H.F.; Provan, G.J.; Helliwell, K. Determination of hamamelitannin, catechins and gallic acid in witch hazel bark, twig and leaf by HPLC. J. Pharm. Biomed. Anal. 2003, 33, 539–544. [Google Scholar] [CrossRef]
- Chen, H.M.; Wu, Y.C.; Chia, Y.C.; Chang, F.R.; Hsu, H.K.; Hsieh, Y.C.; Chen, C.C.; Yuan, S.S. Gallic acid, a major component of Toona sinensis leaf extracts, contains a ROS-mediated anti-cancer activity in human prostate cancer cells. Cancer Lett. 2009, 286, 161–171. [Google Scholar] [CrossRef]
- Kim, Y.J. Antimelanogenic and antioxidant properties of gallic acid. Biol. Pharm. Bull. 2007, 30, 1052–1055. [Google Scholar] [CrossRef]
- Dalla Pellegrina, C.; Padovani, G.; Mainente, F.; Zoccatelli, G.; Bissoli, G.; Mosconi, S.; Veneri, G.; Peruffo, A.; Andrighetto, G.; Rizzi, C.; Chignola, R. Anti-tumour potential of a gallic acid-containing phenolic fraction from Oenothera biennis. Cancer Lett. 2005, 226, 17–25. [Google Scholar] [CrossRef]
- Agarwal, C.; Tyagi, A.; Agarwal, R. Gallic acid causes inactivating phosphorylation of cdc25A/cdc25C-cdc2 via ATM-Chk2 activation, leading to cell cycle arrest, and induces apoptosis in human prostate carcinoma DU145 cells. Mol. Cancer Ther. 2006, 5, 3294–3302. [Google Scholar] [CrossRef]
- Kaur, M.; Velmurugan, B.; Rajamanickam, S.; Agarwal, R.; Agarwal, C. Gallic Acid, an Active Constituent of Grape Seed Extract, Exhibits Anti-proliferative, Pro-apoptotic and Anti-tumorigenic Effects Against Prostate Carcinoma Xenograft Growth in Nude Mice. Pharm. Res. 2009, 26, 2133–2140. [Google Scholar] [CrossRef]
- Liu, Z.J.; Schwimer, J.; Liu, D.; Lewis, J.; Greenway, F.L.; York, D.A.; Woltering, E.A. Gallic acid is partially responsible for the antiangiogenic activities of Rubus leaf extract. Phytother. Res. 2006, 20, 806–813. [Google Scholar] [CrossRef]
- Gupta, N.; Gupta, S.; Mahmood, A. Gallic acid inhibits brush border disaccharidases in mammalian intestine. Nutr. Res. 2007, 27, 230–235. [Google Scholar] [CrossRef]
- Kratz, J.M.; Andrighetti-Frohner, C.R.; Leal, P.C.; Nunes, R.J.; Yunes, R.A.; Trybala, E.; Bergstrom, T.; Barardi, C.R.M.; Simoes, C.M.O. Evaluation of anti-HSV-2 activity of gallic acid and pentyl gallate. Biol. Pharm. Bull. 2008, 31, 903–907. [Google Scholar] [CrossRef]
- Lu, Y.; Jiang, F.; Jiang, H.; Wu, K.; Zheng, X.; Cai, Y.; Katakowski, M.; Chopp, M.; To, S.S.T. Gallic acid suppresses cell viability, proliferation, invasion and angiogenesis in human glioma cells. Eur. J. Pharmacol. 2010, 641, 102–107. [Google Scholar] [CrossRef]
- You, B.R.; Moon, H.J.; Han, Y.H.; Park, W.H. Gallic acid inhibits the growth of HeLa cervical cancer cells via apoptosis and/or necrosis. Food Chem. Toxicol. 2010, 48, 1334–1340. [Google Scholar] [CrossRef]
- Zgorka, G.; Glowniak, K. Variation of free phenolic acids in medicinal plants belonging to the Lamiaceae family. J. Pharm. Biomed. Anal. 2001, 26, 79–87. [Google Scholar] [CrossRef]
- Matysik, G.; Wojciak-Kosior, M.; Paduch, R. The influence of Calendulae officinalis flos extracts on cell cultures, and the chromatographic analysis of extracts. J. Pharm. Biomed. Anal. 2005, 38, 285–292. [Google Scholar] [CrossRef]
- Mo, S.Y.; Wang, S.J.; Zhou, G.X.; Yang, Y.C.; Li, Y.; Chen, X.G.; Shi, J.G. Phelligridins C-F: Cytotoxic pyrano[4,3-c][2]benzopyran-1,6-dione and furo[3,2-c]pyran-4-one derivatives from the fungus Phellinus igniarius. J. Nat. Prod. 2004, 67, 823–828. [Google Scholar] [CrossRef]
- Wu, Q.; Fu, D.X.; Hou, A.J.; Lei, G.Q.; Llu, Z.J.; Chen, J.K.; Zhou, T.S. Antioxidative phenols and phenolic glycosides from Curculigo orchioides. Chem. Pharm. Bull. 2005, 53, 1065–1067. [Google Scholar] [CrossRef]
- Al-Farsi, M.; Alasalvar, C.; Morris, A.; Baron, M.; Shahidi, F. Comparison of antioxidant activity, anthocyanins, carotenoids, and phenolics of three native fresh and sun-dried date (Phoenix dactylifera L.) varieties grown in Oman. J. Agr. Food Chem. 2005, 53, 7592–7599. [Google Scholar] [CrossRef]
- Chen, J.J.; Huang, S.Y.; Duh, C.Y.; Chen, I.S.; Wang, T.C.; Fang, H.Y. A new cytotoxic amide from the stem wood of Hibiscus tiliaceus. Planta Med. 2006, 72, 935–938. [Google Scholar] [CrossRef]
- Germano, M.P.; D'Angelo, V.; Biasini, T.; Sanogo, R.; De Pasquale, R.; Catania, S. Evaluation of the antioxidant properties and bioavailability of free and bound phenolic acids from Trichilia emetica Vahl. J. Ethnopharmacol. 2006, 105, 368–373. [Google Scholar] [CrossRef]
- Nakajima, Y.; Sato, Y.; Konishi, T. Antioxidant small phenolic ingredients in Inonotus obliquus (persoon) Pilat (Chaga). Chem. Pharm. Bull. 2007, 55, 1222–1226. [Google Scholar] [CrossRef]
- Atta Ur, R.; Zareen, S.; Choudhary, M.I.; Akhtar, M.N.; Khan, S.N. alpha-glucosidase inhibitory activity of triterpenoids from Cichorium intybus. J. Nat. Prod. 2008, 71, 910–913. [Google Scholar] [CrossRef]
- Chethan, S.; Dharmesh, S.M.; Malleshi, N.G. Inhibition of aldose reductase from cataracted eye lenses by finger millet (Eleusine coracana) polyphenols. Bioorg. Med. Chem. 2008, 16, 10085–10090. [Google Scholar] [CrossRef]
- Kong, W.K.; Zhao, Y.L.; Shan, L.M.; Xiao, X.H.; Guo, W.Y. Thermochemical studies on the quantity - Antibacterial effect relationship of four organic acids from Radix Isatidis on Escherichia coli growth. Biol. Pharm. Bull. 2008, 31, 1301–1305. [Google Scholar] [CrossRef]
- Rastogi, S.; Pandey, M.M.; Rawat, A.K.S. High-Performance Thin-Layer Chromatography Densitometric Method for the Simultaneous Determination of Three Phenolic Acids in Syzygium aromaticum (L.) Merr. & Perry. J. AOAC Int. 2008, 91, 1169–1173. [Google Scholar]
- Khallouki, F.; Hull, W.E.; Owen, R.W. Characterization of a rare triterpenoid and minor phenolic compounds in the root bark of Anisophyllea dichostyla R. Br. Food Chem. Toxicol. 2009, 47, 2007–2012. [Google Scholar] [CrossRef]
- Ksouri, R.; Falleh, H.; Megdiche, W.; Trabelsi, N.; Mhamdi, B.; Chaieb, K.; Bakrouf, A.; Magne, C.; Abdelly, C. Antioxidant and antimicrobial activities of the edible medicinal halophyte Tamarix gallica L. and related polyphenolic constituents. Food Chem. Toxicol. 2009, 47, 2083–2091. [Google Scholar] [CrossRef]
- da Silveira, C.V.; Trevisan, M.T.S.; Rios, J.B.; Erben, G.; Haubner, R.; Pfundstein, B.; Owen, R.W. Secondary plant substances in various extracts of the leaves, fruits, stem and bark of Caraipa densifolia Mart. Food Chem. Toxicol. 2010, 48, 1597–1606. [Google Scholar] [CrossRef]
- Gulcin, I.; Bursal, E.; Sehitoglu, M.H.; Bilsel, M.; Goren, A.C. Polyphenol contents and antioxidant activity of lyophilized aqueous extract of propolis from Erzurum, Turkey. Food Chem. Toxicol. 2010, 48, 2227–2238. [Google Scholar] [CrossRef]
- Sharma, N.; Sharma, U.K.; Gupta, A.P.; Sinha, A.K. Simultaneous determination of epicatechin, syringic acid, quercetin-3-O-galactoside and quercitrin in the leaves of Rhododendron species by using a validated HPTLC method. J. Food Compos. Anal. 2010, 23, 214–219. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, L.W. Phenolic compounds in cereal grains and their health benefits. Cereal. Foods World 2007, 52, 105–111. [Google Scholar]
- Jin, Y.S.; Heo, S.I.; Lee, M.J.; Rhee, H.I.; Wang, M.H. Free radical scavenging and hepatoprotective actions of Quercus aliena acorn extract against CCl4-induced liver. Free Radic. Res. 2005, 39, 1351–1358. [Google Scholar] [CrossRef]
- Masibo, M.; He, Q. Major mango polyphenols and their potential significance to human health. Compr. Rev. Food. Sci. Food Saf. 2008, 7, 309–319. [Google Scholar] [CrossRef]
- Abbasi, A.M.; Khan, M.A.; Ahmad, M.; Zafar, M.; Khan, H.; Muhammad, N.; Sultana, S. Medicinal plants used for the treatment of jaundice and hepatitis based on socio-economic documentation. Afr. J. Biotechnol. 2009, 8, 1643–1650. [Google Scholar]
- Njoku, O.V.; Obi, C. Phytochemical constituents of some selected medicinal plants. African J. Pure Appl. Chem. 2009, 3, 228–233. [Google Scholar]
- Ma, H.B.; Diao, Y.P.; Zhao, D.Y.; Li, K.; Kang, T.G. A new alternative to treat swine influenza A virus infection: extracts from Terminalia chebula Retz. Afr. J. Microbiol. Res. 2010, 4, 497–499. [Google Scholar]
- Bhouri, W.; Derbel, S.; Skandrani, I.; Boubaker, J.; Bouhlel, I.; Sghaier, M.B.; Kilani, S.; Mariotte, A.M.; Dijoux-Franca, M.G.; Ghedira, K.; Chekir-Ghedira, L. Study of genotoxic, antigenotoxic and antioxidant activities of the digallic acid isolated from Pistacia lentiscus fruits. Toxicol. Vitro 2010, 24, 509–515. [Google Scholar] [CrossRef]
- Eckermann, C.; Schroder, G.; Eckermann, S.; Strack, D.; Schmidt, E.; Schneider, B.; Schroder, J. Stilbenecarboxylate biosynthesis: a new function in the family of chalcone synthase-related proteins. Phytochemistry 2003, 62, 271–286. [Google Scholar]
- Asakawa, Y. Chemosystematics of the Hepaticae. Phytochemistry 2004, 65, 623–669. [Google Scholar] [CrossRef]
- Zhou, K.; Wu, B.; Zhuang, Y.; Ding, L.; Liu, Z.; Qiu, F. Chemical constituents of fresh celery. Zhongguo Zhong Yao Za Zhi 2009, 34, 1512–1515. [Google Scholar]
- Yoshikawa, H.; Ichiki, Y.; Sakakibara, K.; Tamura, H.; Suiko, M. The biological and structural similarity between lunularic acid and abscisic acid. Biosci. Biotechnol. Biochem. 2002, 66, 840–846. [Google Scholar] [CrossRef]
- Asakawa, Y. Biologically active compounds from bryophytes. Pure Appl. Chem. 2007, 79, 557–580. [Google Scholar] [CrossRef]
- Zhang, H.L.; Matsuda, H.; Yamashita, C.; Nakamura, S.; Yoshikawa, M. Hydrangeic acid from the processed leaves of Hydrangea macrophylla var. thunbergii as a new type of anti-diabetic compound. Eur. J. Pharmacol. 2009, 606, 255–261. [Google Scholar] [CrossRef]
- Shang, X.F.; He, X.R.; He, X.Y.; Li, M.X.; Zhang, R.X.; Fan, P.C.; Zhang, Q.L.; Jia, Z.P. The genus Scutellaria an ethnopharmacological and phytochemical review. J. Ethnopharmacol. 2010, 128, 279–313. [Google Scholar] [CrossRef]
- Yannai, S. Dictionary of Food Compounds- Additives, Flavors, and Ingredients, 1st ed; Chapman & Hall/CRC: Boca Raton, FL, USA, 2004; p. 1784. [Google Scholar]
- Malikov, V.M.; Yuldashev, M.P. Phenolic compounds of plants of the Scutellaria genus. Distribution, structure, and properties. Chem. Nat. Compd. 2002, 38, 473–519. [Google Scholar]
- Philip, J.Y.N.; Francisco, J.D.; Dey, E.S.; Buchweishaija, J.; Mkayula, L.L.; Ye, L. Isolation of Anacardic Acid from Natural Cashew Nut Shell Liquid (CNSL) Using Supercritical Carbon Dioxide. J. Agr. Food Chem. 2008, 56, 9350–9354. [Google Scholar]
- Kubo, I.; Nihei, K.I.; Tsujimoto, K. Antibacterial action of anacardic acids against methicillin resistant Staphylococcus aureus (MRSA). J. Agr. Food Chem. 2003, 51, 7624–7628. [Google Scholar] [CrossRef]
- Ha, T.J.; Kubo, I. Lipoxygenase inhibitory activity of anacardic acids. J. Agr. Food Chem. 2005, 53, 4350–4354. [Google Scholar] [CrossRef]
- Kubo, I.; Masuoka, N.; Ha, T.J.; Tsujimoto, K. Antioxidant activity of anacardic acids. Food Chem. 2006, 99, 555–562. [Google Scholar] [CrossRef]
- Schultz, D.J.; Olsen, C.; Cobbs, G.A.; Stolowich, N.J.; Parrott, M.M. Bioactivity of anacardic acid against Colorado potato beetle (Leptinotarsa decemlineata) larvae. J. Agr. Food Chem. 2006, 54, 7522–7529. [Google Scholar] [CrossRef]
- Castillo-Juarez, I.; Rivero-Cruz, F.; Celis, H.; Romero, I. Anti-Helicobacter pylori activity of anacardic acids from Amphipterygium adstringens. J. Ethnopharmacol. 2007, 114, 72–77. [Google Scholar] [CrossRef]
- van Beek, T.A.; Montoro, P. Chemical analysis and quality control of Ginkgo biloba leaves, extracts, and phytopharmaceuticals. J. Chromatogr. A 2009, 1216, 2002–2032. [Google Scholar] [CrossRef]
- van Beek, T.A. Chemical analysis of Ginkgo biloba leaves and extracts. J. Chromatogr. A 2002, 967, 21–55. [Google Scholar] [CrossRef]
- Trevisan, M.T.S.; Pfundstein, B.; Haubner, R.; Wurtele, G.; Spiegelhalder, B.; Bartsch, H.; Owen, R.W. Characterization of alkyl phenols in cashew (Anacardium occidentale) products and assay of their antioxidant capacity. Food Chem. Toxicol. 2006, 44, 188–197. [Google Scholar] [CrossRef]
- Fukuda, I.; Ito, A.; Hirai, G.; Nishimura, S.; Kawasaki, H.; Saitoh, H.; Kimura, K.; Sodeoka, M.; Yoshida, M. Ginkgolic Acid Inhibits Protein SUMOylation by Blocking Formation of the E1-SUMO Intermediate. Chem. Biol. 2009, 16, 133–140. [Google Scholar]
- Ueda, M.; Yamamura, S. Chemistry and biology of plant leaf movements. Angew. Chem.-Int. Edit. 2000, 39, 1400–1414. [Google Scholar]
- Ueda, M.; Nakamura, Y. Chemical basis of plant leaf movement. Plant Cell Physiol. 2007, 48, 900–907. [Google Scholar] [CrossRef]
- Ueda, M.; Sugimoto, T.; Sawai, Y.; Ohnuki, T.; Yamamura, S. Chemical studies on plant leaf movement controlled by a biological clock. Pure Appl. Chem. 2003, 75, 353–358. [Google Scholar] [CrossRef]
- Ueda, M.; Shigemori, H.; Sata, N.; Yamamura, S. The diversity of chemical substances controlling the nyctinastic leaf-movement in plants. Phytochemistry 2000, 53, 39–44. [Google Scholar]
- Sontag, B.; Dasenbrock, J.; Arnold, N.; Steglich, W. Metabolites from the wood-rotting basidiomycete Hapalopilus mutans (Aphyllophorales). Eur. J. Org. Chem. 1999, 5, 1051–1055. [Google Scholar]
- Zjawiony, J.K. Biologically active compounds from aphyllophorales (polypore) fungi. J. Nat. Prod. 2004, 67, 300–310. [Google Scholar] [CrossRef]
- Stasiuk, M.; Jaromin, A.; Kozubek, A. The effect of merulinic acid on biomembranes. Biochim. Biophys. Acta-Biomembr 2004, 1667, 215–221. [Google Scholar] [CrossRef]
- Wang, J.; Kodali, S.; Lee, S.H.; Galgoci, A.; Painter, R.; Dorso, K.; Racine, F.; Motyl, M.; Hernandez, L.; Tinney, E.; Colletti, S.L.; Herath, K.; Cummings, R.; Salazar, O.; Gonzalez, I.; Basilio, A.; Vicente, F.; Genilloud, O.; Pelaez, F.; Jayasuriya, H.; Young, K.; Cully, D.F.; Singh, S.B. Discovery of platencin, a dual FabF and FabH inhibitor with in vivo antibiotic properties. Proc. Natl. Acad. Sci. USA 2007, 104, 7612–7616. [Google Scholar]
- Zhang, C.W.; Ondeyka, J.; Dietrich, L.; Gailliot, F.P.; Hesse, M.; Lester, M.; Dorso, K.; Motyl, M.; Ha, S.N.; Wang, J.; Singh, S.B. Isolation, structure and biological activities of platencin A(2)-A(4) from Streptomyces platensis. Bioorg. Med. Chem. 2010, 18, 2602–2610. [Google Scholar] [CrossRef]
- Singh, S.B.; Ondeyka, J.G.; Herath, K.B.; Zhang, C.W.; Jayasuriya, H.; Zink, D.L.; Parthasarathy, G.; Becker, J.W.; Wang, J.; Soisson, S.M. Isolation, enzyme-bound structure and antibacterial activity of platencin A(1) from Streptomyces platensis. Bioorg. Med. Chem. Lett. 2009, 19, 4756–4759. [Google Scholar]
- Jayasuriya, H.; Herath, K.B.; Zhang, C.; Zink, D.L.; Basilio, A.; Genilloud, O.; Diez, M.T.; Vicente, F.; Gonzalez, I.; Salazar, O.; Pelaez, F.; Cummings, R.; Ha, S.; Wang, J.; Singh, S.B. Isolation and structure of platencin: A FabH and FabF dual inhibitor with potent broad-spectrum antibiotic activity. Angew. Chem.-Int. Edit. 2007, 46, 4684–4688. [Google Scholar]
- Palanichamy, K.; Kaliappan, K.P. Discovery and Syntheses of "Superbug Challengers"-Platensimycin and Platencin. Chem.-Asian J. 2010, 5, 668–703. [Google Scholar] [CrossRef]
- Wang, J.; Soisson, S.M.; Young, K.; Shoop, W.; Kodali, S.; Galgoci, A.; Painter, R.; Parthasarathy, G.; Tang, Y.S.; Cummings, R.; Ha, S.; Dorso, K.; Motyl, M.; Jayasuriya, H.; Ondeyka, J.; Herath, K.; Zhang, C.W.; Hernandez, L.; Allocco, J.; Basilio, A.; Tormo, J.R.; Genilloud, O.; Vicente, F.; Pelaez, F.; Colwell, L.; Lee, S. H.; Michael, B.; Felcetto, T.; Gill, C.; Silver, L.L.; Hermes, J.D.; Bartizal, K.; Barrett, J.; Schmatz, D.; Becker, J.W.; Cully, D.; Singh, S.B. Platensimycin is a selective FabF inhibitor with potent antibiotic properties. Nature 2006, 441, 358–361. [Google Scholar]
- Singh, S.B.; Jayasuriya, H.; Ondeyka, J.G.; Herath, K.B.; Zhang, C.W.; Zink, D.L.; Tsou, N.N.; Ball, R.G.; Basilio, A.; Genilloud, O.; Diez, M.T.; Vicente, F.; Pelaez, F.; Young, K.; Wang, J. Isolation, structure, and absolute stereochemistry of platensimycin, a broad spectrum antibiotic discovered using an antisense differential sensitivity strategy. J. Am. Chem. Soc. 2006, 128, 11916–11920. [Google Scholar]
- Habich, D.; von Nussbaum, F. Platensimycin, a new antibiotic and "superbug challenger" from nature. ChemMedChem 2006, 1, 951–954. [Google Scholar] [CrossRef]
- Manallack, D.T.; Crosby, I.T.; Khakham, Y.; Capuano, B. Platensimycin: A promising antimicrobial targeting fatty acid synthesis. Curr. Med. Chem. 2008, 15, 705–710. [Google Scholar] [CrossRef]
- Edrington, T.S.; Callaway, T.R.; Varey, P.D.; Jung, Y.S.; Bischoff, K.M.; Elder, R.O.; Anderson, R.C.; Kutter, E.; Brabban, A.D.; Nisbet, D.J. Effects of the antibiotic ionophores monensin, lasalocid, laidlomycin propionate and bambermycin on Salmonella and E-coli O157 : H7 in vitro. J. Appl. Microbiol. 2003, 94, 207–213. [Google Scholar] [CrossRef]
- Armson, A.; Thompson, R.C.A.; Reynoldso, J.A. A review of chemotherapeutic approaches to the treatment of cryptosporidiosis. Expert Rev. Anti-infective Ther. 2003, 1, 297–305. [Google Scholar] [CrossRef]
- Izzo, A.A.; Borrelli, F.; Capasso, R.; Di Marzo, V.; Mechoulam, R. Non-psychotropic plant cannabinoids: new therapeutic opportunities from an ancient herb. Trends Pharmacol. Sci. 2009, 30, 515–527. [Google Scholar] [CrossRef]
- Sirikantaramas, S.; Taura, F.; Morimoto, S.; Shoyama, Y. Recent advances in Cannabis sativa research: Biosynthetic studies and its potential in biotechnology. Curr. Pharm. Biotechnol. 2007, 8, 237–243. [Google Scholar] [CrossRef]
- Takeda, S.; Misawa, K.; Yamamoto, I.; Watanabe, K. Cannabidiolic acid as a selective cyclooxygenase-2 inhibitory component in cannabis. Drug Metab. Dispos. 2008, 36, 1917–1921. [Google Scholar] [CrossRef]
- De Petrocellis, L.; Vellani, V.; Schiano-Moriello, A.; Marini, P.; Magherini, P.C.; Orlando, P.; Di Marzo, V. Plant-derived cannabinoids modulate the activity of transient receptor potential channels of ankyrin type-1 and melastatin type-8. J. Pharmacol. Exp. Ther. 2008, 325, 1007–1015. [Google Scholar] [CrossRef]
- Ligresti, A.; Moriello, A.S.; Starowicz, K.; Matias, I.; Pisanti, S.; De Petrocellis, L.; Laezza, C.; Portella, G.; Bifulco, M.; Di Marzo, V. Antitumor activity of plant cannabinoids with emphasis on the effect of cannabidiol on human breast carcinoma. J. Pharmacol. Exp. Ther. 2006, 318, 1375–1387. [Google Scholar] [CrossRef]
- Kong, Y.; Zu, Y.G.; Fu, Y.J.; Liu, W.; Chang, F.R.; Li, J.; Chen, Y.H.; Zhang, S.; Gu, C.B. Optimization of microwave-assisted extraction of cajaninstilbene acid and pinostrobin from pigeonpea leaves followed by RP-HPLC-DAD determination. J. Food Compos. Anal. 2010, 23, 382–388. [Google Scholar] [CrossRef]
- Inman, W.D.; Hoppe, D.C. Compositions containing hyportriglyceridemically active stilbenenoids. W. I. P. O. I. Bureau: Geneva, Switzerland, 2002. [Google Scholar]
- Inman, W.D.; Hoppe, D.C. Compositions containing hypoglucemically active stilbenoids. W. I. P. O. I. Bureau: Geneva, Switzerland, 2003. [Google Scholar]
- Kong, Y.; Fu, Y.J.; Zu, Y.G.; Liu, W.; Wang, W.; Hua, X.; Yang, M. Ethanol modified supercritical fluid extraction and antioxidant activity of cajaninstilbene acid and pinostrobin from pigeonpea [Cajanus cajan (L.) Millsp.] leaves. Food Chem. 2009, 117, 152–159. [Google Scholar] [CrossRef]
- Wu, N.; Fu, K.; Fu, Y.J.; Zu, Y.G.; Chang, F.R.; Chen, Y.H.; Liu, X.L.; Kong, Y.; Liu, W.; Gu, C.B. Antioxidant Activities of Extracts and Main Components of Pigeonpea [Cajanus cajan (L.) Millsp.] Leaves. Molecules 2009, 14, 1032–1043. [Google Scholar] [CrossRef]
- Sun, S.M.; Song, Y.M.; Liu, J. Studies on the pharmacology of Cajanin preparation. Chin. Trad. Herbal Drug. 1995, 26, 147–148. [Google Scholar]
- Zheng, Y.Y.; Yang, J.; Chen, D.H.; Sun, L. Effects of stilbene extracts from Cajanus cajan L. on ovariectomy-induced bone loss in rats. Acta Pharmacol. Sin. 2007, 42, 562–567. [Google Scholar]
- Cooksey, C.J.; Dahiya, J.S.; Garratt, P.J.; Strange, R.N. 2 Novel Stilbene-2-carboxylic acid Phytoalexins from Cajanus-Cajan. Phytochemistry 1982, 21, 2935–2938. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Khadem, S.; Marles, R.J. Monocyclic Phenolic Acids; Hydroxy- and Polyhydroxybenzoic Acids: Occurrence and Recent Bioactivity Studies. Molecules 2010, 15, 7985-8005. https://doi.org/10.3390/molecules15117985
Khadem S, Marles RJ. Monocyclic Phenolic Acids; Hydroxy- and Polyhydroxybenzoic Acids: Occurrence and Recent Bioactivity Studies. Molecules. 2010; 15(11):7985-8005. https://doi.org/10.3390/molecules15117985
Chicago/Turabian StyleKhadem, Shahriar, and Robin J. Marles. 2010. "Monocyclic Phenolic Acids; Hydroxy- and Polyhydroxybenzoic Acids: Occurrence and Recent Bioactivity Studies" Molecules 15, no. 11: 7985-8005. https://doi.org/10.3390/molecules15117985
APA StyleKhadem, S., & Marles, R. J. (2010). Monocyclic Phenolic Acids; Hydroxy- and Polyhydroxybenzoic Acids: Occurrence and Recent Bioactivity Studies. Molecules, 15(11), 7985-8005. https://doi.org/10.3390/molecules15117985



