3.2. Chemistry
3.2.1. Synthesis of compounds 5a-h
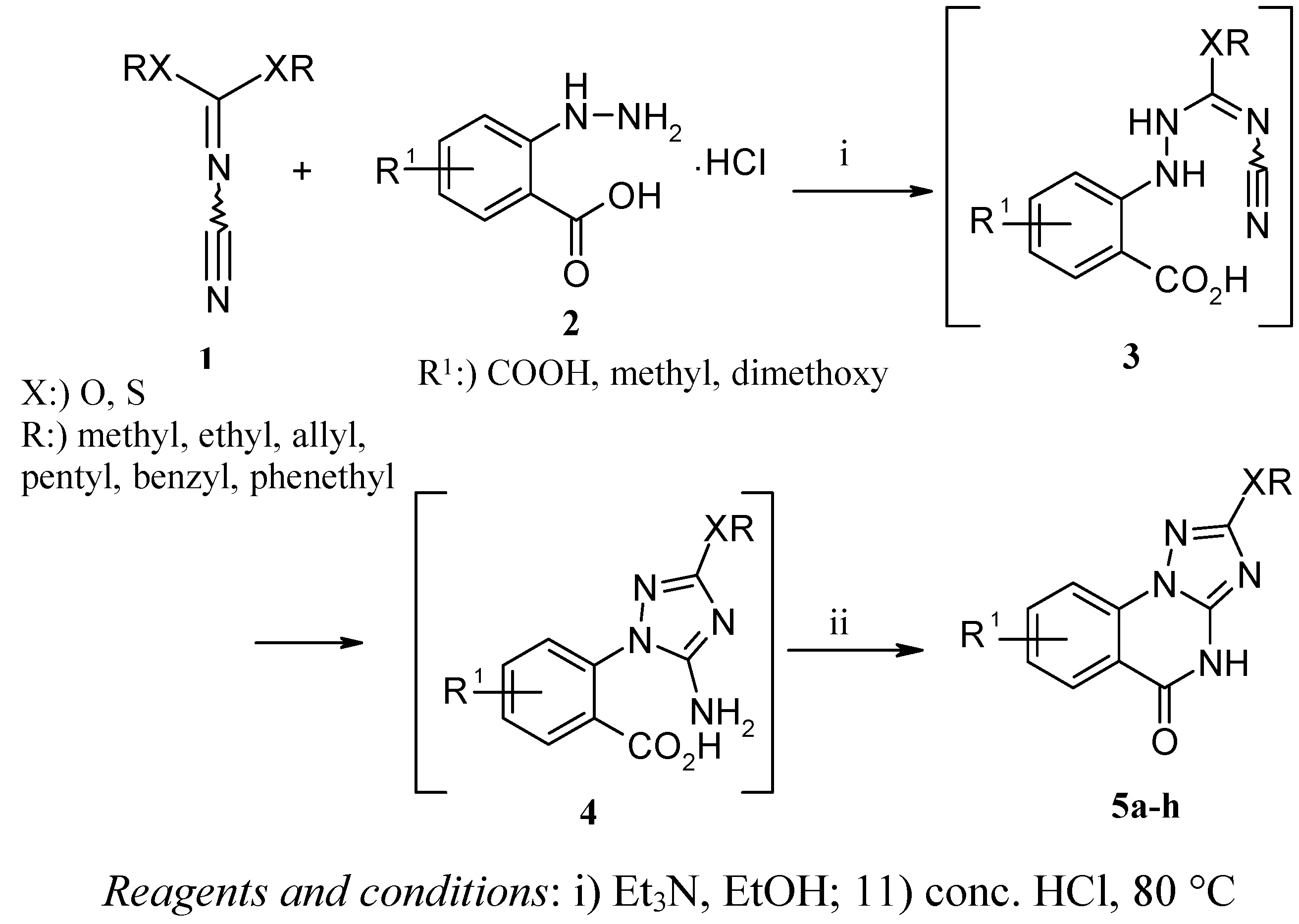
10 mmol of substituted hydrazinobenzoic acid 2 was added portionwise to a stirred solution of 1 (10 mmol) in EtOH (20 mL) at 0°C. Afterwards triethylamine (30 mmol) was added dropwise over a period of 30 min. After the addition was complete, the reaction mixture was left to stir overnight at room temperature. Acidification of the mixture was performed by conc. HCl under ice cooling followed by refluxing for 1-3 h. After cooling, the mixture was poured into ice/water, the resulting solid was filtered, washed with water and dried. Recrystallization from THF gave analytically pure colored cpmpounds 5a-h.
8-Carboxylic acid-2-methoxy-4H-[1,2,4]triazolo[[1,5-a]]quinazolin-5-one (5a). IR (cm−1): ν 1,685, 1,712 (C=O). 1H-NMR (DMSO-d6): δ 3.19 (s, 1H, OH), 3.99 (s, 3H, OCH3), 7.48-8.05 (m, 3H, Ar-H), 13.15 (s, 1H, NH). 13C-NMR: 57.16, 114.31, 116.53, 125.58, 128.08, 135.12, 136.18, 147.87, 159.70, 161.83, 168.02. MS, m/z (%): 260 (M+, 100).
8-Carboxylic acid-2-ethoxy-4H-[1,2,4]triazolo[[1,5-a]]quinazolin-5-one (5b). IR (cm−1): ν 1,689, 1,703 (C=O). 1H-NMR (DMSO-d6): δ 1.38 (t, J = 7.02 Hz, 3H, OCH2CH3), 3.34 (s, 1H, OH), 4.35 (q, J = 14.10 Hz, 2H, OCH2CH3), 7.67-8.12 (m, 3H, Ar-H), 13.01 (s, 1H, NH). 13C-NMR: 14.86, 65.64, 114.29, 116.79, 125.43, 128.25, 135.62, 136.12, 147.24, 156.14, 159.86, 167.52. MS, m/z (%): 274 (M+, 95).
8-Methyl-2-pentyloxy-4H-[1,2,4]triazolo[1,5-a]quinazolin-5-one (5c). IR (cm−1): ν 1,690 (C=O). 1H-NMR (DMSO-d6): δ 0.98 (t, J = 7.32 Hz, 3H, OCH2CH2CH2CH2CH3), 1.37-1.44 (m, 4H, OCH2CH2CH2CH2CH3), 1.63-1.79 (m, 2H, OCH2CH2CH2CH2CH3), 2.78 (s, 3H, CH3), 4.42 (t, J = 7.41 Hz, 2H, OCH2CH2CH2CH2CH3), 7.45-8.51 (m, 3H, Ar-H), 12.83 (s, 1H, NH). 13C-NMR: 13.39, 14.47, 22.24, 27.73, 28.07, 69.74, 114.65, 116.80, 125.45, 128.68, 135.72, 136.11, 147.74, 159.91, 167.70. MS, m/z (%): 286 (M+, 85).
2-Allyloxy-8-methyl-4H-[1,2,4]triazolo[1,5-a]quinazolin-5-one (5d). IR (cm−1): ν 1,697 (C=O). 1H-NMR (DMSO-d6): δ 3.39 (s, 3H, CH3), 4.86 (d, J = 5.68 Hz, 2H, CH2=CHCH2), 5.42-5.61 (m, 2H, CH2=CHCH2), 6.05-6.15 (m, 1H, CH2=CHCH2), 7.68-8.25 (m, 3H, Ar-H), 13.41 (s, 1H, NH). 13C-NMR: 23.89, 69.60, 113.82, 116.44, 118.25, 125.16, 128.13, 134.11, 135.30, 135.62, 147.30, 159.45, 166.92. MS, m/z (%): 256 (M+, 100).
2-Benzyloxy-7,8-dimethoxy-4H-[1,2,4]triazolo[1,5-a]quinazolin-5-one (5e IR (cm−1): ν 1,710 (C=O). 1H-NMR (DMSO-d6): δ 3.48 (s, 3H, OCH3), 3.98 (s, 3H, OCH3), 5.39 (s, 2H, OCH2Ph), 7.37-8.16 (m, 7H, Ar-H), 13.44 (s, 1H, NH). 13C-NMR: 54.23, 58.09, 71.18, 114.34, 116.81, 125.53, 127.74, 128.03, 128.85, 135.75, 136.11, 136.77, 147.11, 160.40, 167.58. MS, m/z (%): 352 (M+, 92).
7,8-Dimethoxy-2-phenethyloxy-4H-[1,2,4]triazolo[1,5-a]quinazolin-5-one (5f). IR (cm−1): ν 1,689 (C=O). 1H-NMR (DMSO-d6): δ 3.09 (t, J = 7.44 Hz, 2H, OCH2CH2Ph), 3.80 (s, 3H, OCH3), 4.01 (s, 3H, OCH3), 4.50 (t, J = 7.41 Hz, 2H, OCH2CH2Ph), 7.20-8.19 (m, 7H, Ar-H), 13.75 (s, 1H, NH). 13C-NMR: 34.91, 51.73, 56.71, 70.23, 116.81, 114.32, 126.80, 125.51, 128.29, 128.74, 129.37, 136.14, 138.33, 147.72, 159.91, 167.57. MS, m/z (%): 366 (M+, 53).
8-Methyl-2-methylsulfanyl-4H-[1,2,4]triazolo[1,5-a]quinazolin-5-one (5g). IR (cm−1): ν 1,687 (C=O). 1H-NMR (DMSO-d6): δ 2.94 (s, 3H, CH3), 3.27 (s, 3H, SCH3) 7.64-8.25 (m, 3H, Ar-H), 13.68 (s, 1H, NH). 13C-NMR: 13.92, 24.60, 114.65, 116.23, 125.50, 128.58, 135.72, 136.12, 149.11, 159.90, 162.30. MS, m/z (%): 246 (M+, 87).
7,8-Dimethoxy-2-methylsulfanyl-4H-[1,2,4]triazolo[1,5-a]quinazolin-5-one (5h). IR (cm−1): ν 1,698 (C=O). 1H-NMR (DMSO-d6): δ 2.87 (s, 3H, SCH3), 3.07 (s, 3H, OCH3), 3.84 (s, 3H, OCH3), 7.59-8.36 (m, 2H, Ar-H), 13.90 (s, 1H, NH). 13C-NMR: 13.78, 56.45, 58.01, 114.05, 115.91, 126.34, 129.08, 135.09, 136.52, 149.11, 159.72, 165.30. MS, m/z (%): 292 (M+, 100).
3.2.2. Synthesis of compounds 6a-d
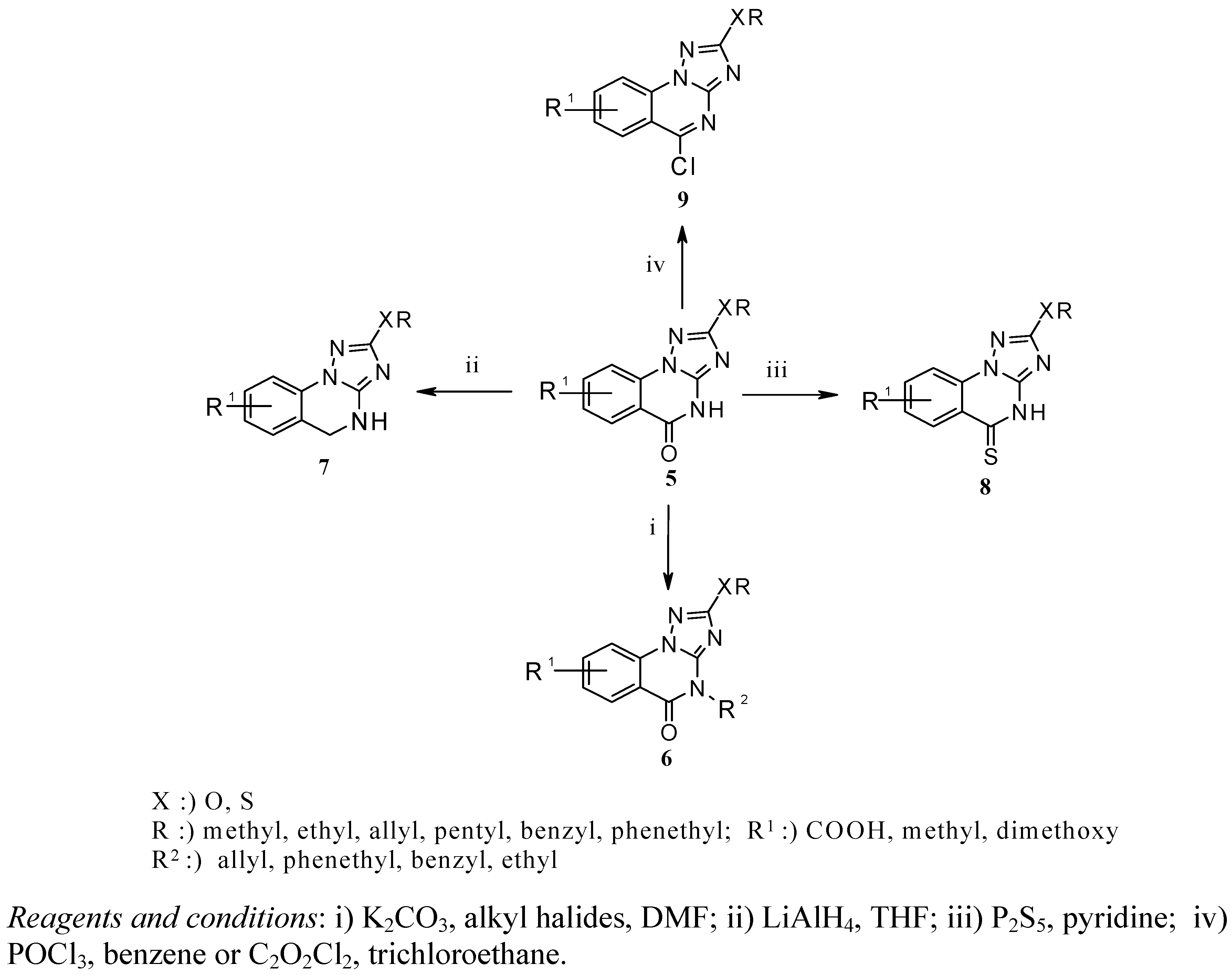
To a solution of 5 (1 mmol) in DMF (5 mL) was added potassium carbonate (1.2 mmol) portion wise over a period of 10 min at room temperature. After stirring for 20 min, the appropriate alkyl halide (1.5 mmol) was added dropwise and the reaction mixture was stirred for 18 h at room temperature. The mixture was poured into ice/water, the precipitate was filtered off, washed with water and dried. Analytically pure products 6a-d were obtained after recrystallization from THF.
8-Methyl-2-methylsulfanyl-4-phenethyl[1,2,4]triazolo[1,5-a]quinazolin-5-one (6a). IR (cm−1): ν 1,671 (C=O). 1H-NMR (DMSO-d6): δ 2.98 (s, 3H, SCH3), 3.37 (t, J = 7.54 Hz, 2H, NCH2CH2Ph), 4.02 (s, 3H, CH3), 4.31 (t, J = 7.51 Hz, 2H, NCH2CH2Ph), 7.22-8.20 (m, 8H, Ar-H). 13C-NMR: 13.98, 24.74, 34.48, 64.26, 114.68, 116.13, 125.82, 126.88, 128.87, 135.42, 135.84, 138.39, 147.59, 158.76, 167.91. MS, m/z (%): 350 (M+, 90).
2-Benzyloxy-7,8-dimethoxy-4-allyl[1,2,4]triazolo[1,5-a]quinazolin-5-one (6b). IR (cm−1): ν 1,678 (C=O). 1H-NMR (DMSO-d6): δ 3.11 (s, 3H, OCH3), 3.90 (s, 3H, OCH3), 4.45 (d, J = 5.62 Hz, 2H, CH2=CHCH2), 5.17-5.29 (m, 2H, CH2=CHCH2), 5.31 (s, 2H, CH2), 6.25-6.33 (m, 1H, CH2=CHCH2), 7.50-8.30 (m, 7H, Ar-H). 13C-NMR: 24.11, 49.89, 57.34, 69.60, 113.82, 116.44, 118.25, 124.03, 124.98, 125.16, 128.13, 131.54, 134.91, 135.30, 135.62, 147.30, 159.05, 165.82. MS, m/z (%): 392 (M+, 79).
8-Carboxylic acid-4-ethyl-2-methoxy[1,2,4]triazolo[1,5-a]quinazolin-5-one (6c). IR (cm−1): ν 1,675, 1,682 (C=O). 1H-NMR (DMSO-d6): δ 1.37 (t, J = 7.02 Hz, 3H, NCH2CH3), 3.52 (s, 1H, OH), 4.09 (q, J =14.22 Hz, 2H, NCH2CH3), 4.32 (s, 3H, OCH3), 7.58-8.09 (m, 3H, Ar-H). 13C-NMR: 14.23, 52.34, 57.18, 114.13, 116.27, 125.71, 128.79, 135.20, 135.42, 148.49, 158.64, 162.34, 167.99. MS, m/z (%): 288 (M+, 67).
2-Allyloxy-4-benzyl-8-methyl[1,2,4]triazolo[1,5-a]quinazolin-5-one (6d). IR (cm−1): ν 1,670 (C=O). 1H-NMR (DMSO-d6): δ 3.57 (s, 3H, CH3), 4.83 (d, J = 4.60 Hz, 2H, CH2=CHCH2), 5.20 (s, 2H, CH2), 5.30-5.42 (m, 2H, CH2=CHCH2), 6.04-6.14 (m, 1H, CH2=CHCH2), 7.46-8.15 (m, 8H, Ar-H). 13C-NMR: 24.29, 44.53, 63.14, 114.47, 116.10, 117.18, 125.83, 128.82, 130.23, 131.66, 134.45, 134.90, 135.32, 135.86, 148.79, 157.81, 168.43. MS, m/z (%): 346 (M+, 80).
3.2.3. Synthesis of compounds 7a-d
A solution of 5 (1 mmol) in dry THF (5 mL) was added dropwise to a stirred suspension of LiAlH4 (3 mmol) in dry THF (10 mL). After stirring at room temperature for 3 h, water (0.4 mL) was added carefully and the mixture was stirred for an additional 30 min. The reaction mixture was filtered and the solvent removed under reduced pressure, the residue was dissolved in THF and passed through a short column chromatography, the solvent was removed under reduced pressure, and the obtained solid was recrystallized from EtOAc/n-hexane.
4,5-Dihydro-8-methyl-2-methylsulanyl[1,2,4]triazolo[1,5-a]quinazoline (7a). IR (cm−1): ν 3,167, (NH). 1H-NMR (DMSO-d6): δ 2.83 (s, 3H, CH3), 3.50 (s, 3H, SCH3), 4.20 (s, 2H, CH2-quinazoline), 7.28-7.82 (m, 3H, Ar-H), 7.95 (s, 1H, NH). 13C-NMR: 13.23, 25.23, 43.22, 112.72, 119.64, 124.50, 126.23, 130.75, 134.16, 155.18, 165.29. MS, m/z (%): 232 (M+, 100).
2-Allyloxy-4,5-dihydro-8-methyl[1,2,4]triazolo[1,5-a]quinazoline (7b). IR (cm-1): ν 3,153, (NH). 1H-NMR (DMSO-d6): δ 2.76 (s, 3H, CH3), 4.76 (d, J = 6.74 Hz, 2H, CH2=CHCH2), 4.92 (s, 2H, CH2-quinazoline), 5.22-5.33 (m, 2H, CH2=CHCH2), 6.09-6.16 (m, 1H, CH2=CHCH2), 7.48-8.10 (m, 3H, Ar-H), 8.25,(s, 1H, NH). 13C-NMR: 23.45, 69.63, 113.87, 116.45, 118.20, 119.24, 125.33, 128.12, 134.57, 135.25, 135.51, 159.47, 167.70. MS, m/z (%): 242 (M+, 89).
2-Benzyloxy-4,5-dihydro-7,8-dimethoxy[1,2,4]triazolo[1,5-a]quinazoline (7c). IR (cm−1): ν 3,189, (NH). 1H-NMR (DMSO-d6): δ 2.93 (s, 3H, OCH3), 3.30 (s, 3H, OCH3), 4.50 (s, 2H, CH2-quinazoline), 5.26 (s, 2H, OCH2Ph), 7.01-7.56 (m, 7H, Ar-H), 7.91 (s, 1H, NH). 13C-NMR: 52.07, 55.39, 69.94, 112.27, 119.23, 124.10, 126.30, 127.12, 127.75, 128.23, 128.85, 133.27, 136.44, 154.55, 166.87. MS, m/z (%): 338 (M+, 93).
4,5-Dihydro-7,8-dimethoxy-2-phenethyloxy[1,2,4]triazolo[1,5-a]quinazoline (7d). IR (cm−1): ν 3,180, (NH). 1H-NMR (DMSO-d6): δ 2.89 (s, 3H, OCH3), 3.2 2 (s, 3H, OCH3), 3.44 (t, J = 7.45 Hz, 2H, OCH2CH2Ph), 4.39 (t, J = 7.41 Hz, 2H, OCH2CH2Ph), 4.48 (s, 2H, CH2-quinazoline), 7.10-7.52 (m, 7H, Ar-H), 7.77 (s, 1H, NH). 13C-NMR: 34.90, 45.07, 49.38, 68.95, 113.33, 119.27, 124.23, 126.80, 128.51, 129.37, 131.09, 135.74, 136.11, 138.33, 154.90, 166.85. MS, m/z (%): 352 (M+, 90).
3.2.4. Synthesis of compounds 8a-d
Compound 5 (1 mmol) was refluxed with phosphorus pentasulfide (1 mmol) in absolute pyridine (5 mL) for 2 h. Afterwards the reaction mixture was cooled and poured into ice/water, the yellow precipitate was separated by filtration and washed thoroughly with water. Recrystallization from aqueous DMF furnished analytically pure 8a-d.
7,8-Dimethoxy-2-methylsulfanyl-4H-[1,2,4]triazolo[1,5-a]quinazolin-5-thione (8a). IR (cm−1): ν 1,268 (C=S). 1H-NMR (DMSO-d6): δ 3.32 (s, 3H, SCH3), 3.70 (s, 3H, OCH3), 4.02 (s, 3H, OCH3), 7.52-7.96 (m, 2H, Ar-H), 14.72 (s, 1H, NH). 13C-NMR: 13.72, 54.43, 56.84, 114.21, 122.43, 125.83, 132.41, 135.88, 149.59, 162.78, 185.71. MS, m/z (%): 308 (M+, 100).
2-Allyloxy-8-methyl-4H-[1,2,4]triazolo[1,5-a]quinazolin-5-thione (8b). IR (cm−1): ν 1,258 (C=S). 1H-NMR (DMSO-d6): δ 2.86 (s, 3H, CH3), 4.85 (d, J = 6.36 Hz, 2H, CH2=CHCH2), 5.31-5.46 (m, 2H, CH2=CHCH2), 6.08-6.15 (m, 1H, CH2=CHCH2), 7.48-8.12 (m, 3H, Ar-H), 14.48 (s, 1H, NH). 13C-NMR: 25.09, 69.92, 114.27, 118.39, 122.53, 125.92, 128.21, 131.83, 132.42 , 135.92, 145.75, 167.31, 184.91. MS, m/z (%): 272 (M+, 94).
2-Benzyloxy-7,8-dimethoxy-4H-[1,2,4]triazolo[1,5-a]quinazolin-5-thione (8c). IR (cm−1): ν 1,255 (C=S). 1H-NMR (DMSO-d6): δ 3.20 (s, 3H, OCH3), 3.78 (s, 3H, OCH3), 5.42 (s, 2H, OCH2Ph), 7.37-8.62 (m, 7H, Ar-H), 14.74 (s, 1H, NH). 13C-NMR: 45.21, 48.34, 70.60, 114.24, 122.40, 125.37, 128.06, 128.30, 128.90, 131.72, 132.33, 135.38, 145.90, 167.34, 185.11. MS, m/z (%): 368 (M+, 65).
7,8-Dimethoxy-2-phenethyloxy-4H-[1,2,4]triazolo[1,5-a]quinazolin-5-thione (8d). IR (cm−1): ν 1,249 (C=S). 1H-NMR (DMSO-d6): δ 2.95 (s, 3H, OCH3), 3.11 (t, J = 6.35 Hz, 2H, OCH2CH2Ph), 3.58 (s, 3H, OCH3), 4.55 (t, J = 6.63 Hz, 2H, OCH2CH2Ph), 7.24-8.61 (m, 7H, Ar-H), 14.70 (s, 1H, NH). 13C-NMR: 34.45, 47.21, 53.34, 69.72, 114.23, 122.42, 125.00, 125.82, 126.30, 128.85, 131.76, 135.83, 137.80, 145.63, 165.20, 186.62. MS, m/z (%): 382 (M+, 78).
3.2.5. Synthesis of compounds 9a-f
Method-A: Compound 5 (2 mmol) was refluxed with oxalyl chloride (6 mmol) in 1,1,2-trichloroethane (12 mL) for 19 h at 105°C. The solution was cooled and MeOH (0.2 mL) was added drop- wise, the obtained solid was filtered, washed with hexane, dried and recrystallized from THF-hexane.
Method-B: Compound 5 (1 mmol) was refluxed with Phosphorus oxychloride (1 mL) in benzene (7 mL) for 2 h. The solvent was evaporated and the residue was treated with saturated aqueous solution of potassium carbonate. The solid was filtered, washed thoroughly with water, dried and recrystallized from THF-hexane.
8-Carboxylic acid-5-chloro-2-ethoxy[1,2,4]triazolo[1,5-a]quinazoline (9a). IR (cm−1): ν 1,683 (C=O). 1H-NMR (DMSO-d6): δ 1.30 (t, J = 7.07 Hz, 3H, OCH2CH3), 3.03 (s, 1H, OH), 4.34 (q, J = 14.10 Hz, 2H, OCH2CH3), 7.49-8.15 (m, 3H, Ar-H). 13C-NMR: 14.56, 64.57, 114.38, 116.05, 125.39, 128.22, 135.58, 136.43, 142.70, 154.32, 159.38. MS, m/z (%): 292 (M+, 88).
5-Chloro-8-methyl-2-pentyloxy[1,2,4]triazolo[1,5-a]quinazoline (9b). 1H-NMR (DMSO-d6): δ 0.81 (t, J = 7.45 Hz, 3H, OCH2CH2CH2CH2CH3), 1.46-1.63 (m, 4H, OCH2CH2CH2CH2CH3), 1.83-1.89 (m, 2H, OCH2CH2CH2CH2CH3), 3.11 (s, 3H, CH3), 4.43 (t, J = 7.60 Hz, 2H, OCH2CH2CH2CH2CH3), 7.45-8.16 (m, 3H, Ar-H). 13C-NMR: 13.75, 21.70, 27.35, 28.16, 45.82, 69.52, 114.70, 116.81, 126.54, 127.95, 136.57, 146.63, 155.33, 160.07. MS, m/z (%): 304 (M+, 91).
2-Benzyloxy-5-chloro-7,8-dimethoxy[1,2,4]triazolo[1,5-a]quinazoline (9c). 1H-NMR (DMSO-d6): δ 3.23 (s, 3H, OCH3), 3.64 (s, 3H, OCH3), 5.79 (s, 2H, OCH2Ph), 7.37-8.45 (m, 7H, Ar-H). 13C-NMR: 50.01, 53.74, 71.34, 115.20, 117.42, 125.50, 126.71, 127.14, 128.25, 128.70, 132.41, 135.90, 136.11, 136.77, 155.93, 162.65. MS, m/z (%): 370 (M+, 69).
2-Allyloxy-5-chloro-8-methyl[1,2,4]triazolo[1,5-a]quinazoline (9d). 1H-NMR (DMSO-d6): δ 3.01 (s, 3H, CH3), 4.21 (d, J = 5.50 Hz, 2H, CH2=CHCH2), 5.33-5.52 (m, 2H, CH2=CHCH2), 6.10-6.16 (m, 1H, CH2=CHCH2), 7.73-8.34 (m, 3H, Ar-H). 13C-NMR: 25.89, 69.60, 113.82, 116.44, 118.25, 125.16, 128.13, 134.11, 135.30, 135.62, 145.87, 158.33, 163.12. MS, m/z (%): 274 (M+, 100).
5-Chloro-7,8-dimethoxy-2-phenethyloxy[1,2,4]triazolo[1,5-a]quinazoline (9e): 1H-NMR (DMSO-d6): δ 3.09 (s, 3H, OCH3), 3.21 (t, J = 7.50 Hz, 2H, OCH2CH2Ph), 3.52 (s, 3H, OCH3), 4.65 (t, J = 7.51 Hz, 2H, OCH2CH2Ph), 7.22-8.37 (m, 7H, Ar-H). 13C-NMR: 35.09, 49.60, 54.11, 69.61, 114.59, 124.40, 124.83, 126.72, 128.74, 129.30, 134.29, 134.94, 138.49, 153.37, 156.84, 161.31. MS, m/z (%): 384 (M+, 100).
5-Chloro-8-methyl-2-methylsulfanyl[1,2,4]triazolo[1,5-a]quinazoline (9f). 1H-NMR (DMSO-d6): δ 3.12 (s, 3H, CH3), 3.72 (s, 3H, SCH3) 7.34-8.15 (m, 3H, Ar-H). 13C-NMR: 13.62, 24.00, 115.15, 117.83, 125.36, 128.18, 136.02, 136.92, 149.80, 152.24, 158.93. MS, m/z (%): 264 (M+, 93).
3.2.6. Synthesis of compounds 10a-d
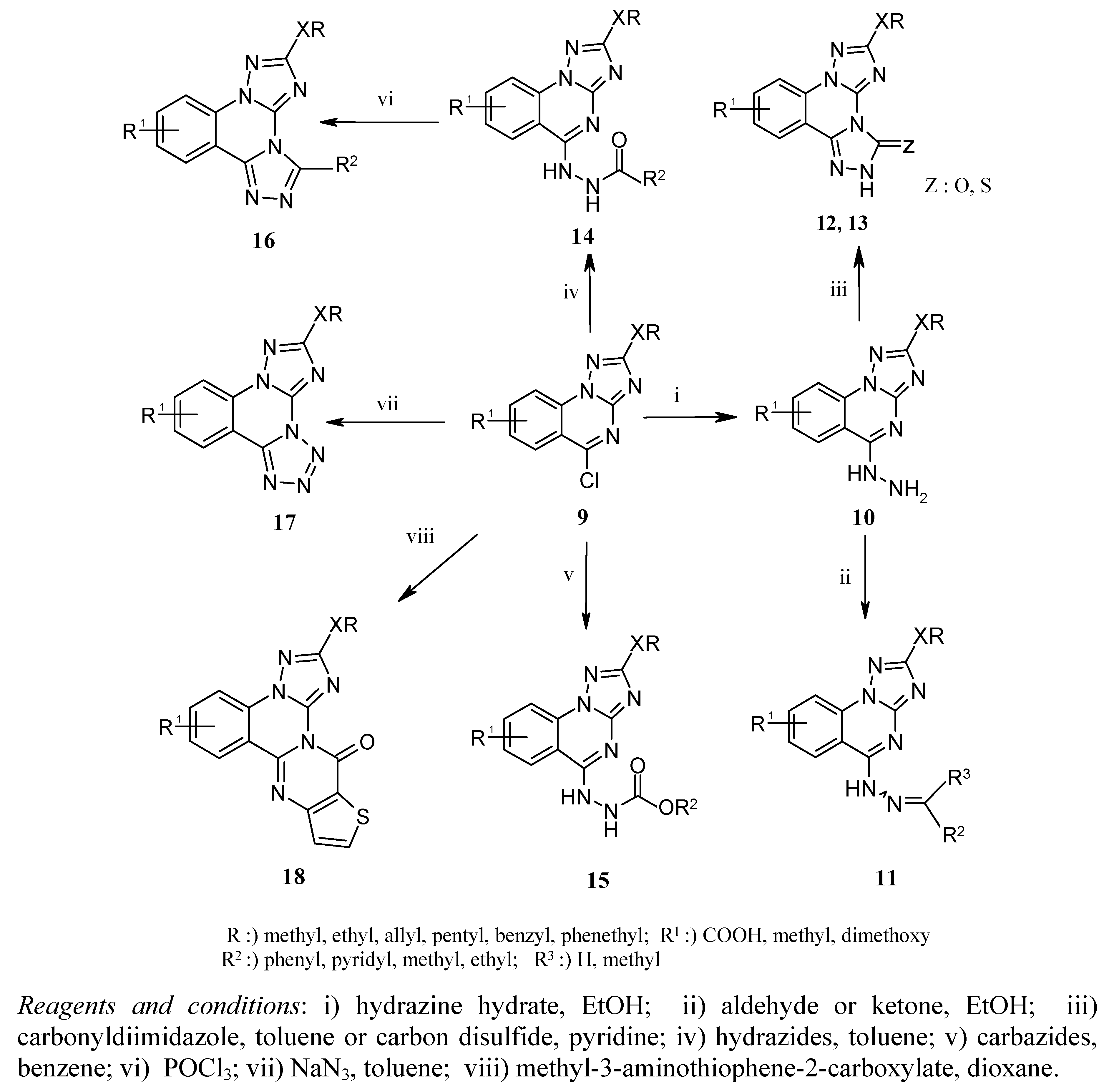
Compound 9 (1 mmol) was heated under reflux with hydrazine hydrate (5 mmol) in EtOH (8 mL) for 3 h. After cooling, the precipitate was filtered off and washted with water. Recrystallization from EtOH afforded 10a-d as colored pure solids.
8-Methyl-2-methylsulfanyl[1,2,4]triazolo[[1,5-a]]quinazolin-5-yl-hydrazine (10a). IR (cm−1): ν 3,189, 3,231 (NH-NH2). 1H-NMR (DMSO-d6): δ 2.80 (s, 3H, CH3), 3.78 (s, 3H, SCH3), 4.84 (s, 2H, NH2), 7.97-8.30 (m, 3H, Ar-H), 9.37 (s, 1H, NH). 13C-NMR: 13.87, 26.48, 114.48, 124.06, 124.77, 125.40, 127.35, 134.24, 134.88, 153.31, 169.73. MS, m/z (%): 260 (M+, 100).
8-Carboxylic acid-2-ethoxy[1,2,4]triazolo[[1,5-a]]quinazolin-5-yl-hydrazine (10b). IR (cm−1): ν 3,182, 3,201 (NH-NH2), 1,686 (C=O). 1H-NMR (DMSO-d6): δ 1.32 (t, J = 7.07 Hz, 3H, OCH2CH3), 3.34 (s, 1H, OH), 4.37 (q, J = 14.15 Hz, 2H, OCH2CH3), 4.65 (s, 2H, NH2), 7.89-8.05 (m, 3H, Ar-H), 9.42 (s, 1H, NH). 13C-NMR: 14.73, 65.61, 114.12, 116.45, 125.62, 128.43, 135.13, 136.29, 145.38, 157.82, 167.92. MS, m/z (%): 288 (M+, 78).
2-Allyloxy-8-methyl[1,2,4]triazolo[[1,5-a]]quinazolin-5-yl-hydrazine (10c). IR (cm−1): ν 3,210, 3,267 (NH-NH2). 1H-NMR (DMSO-d6): δ 3.21 (s, 3H, CH3), 4.81 (s, 2H, NH2), 4.85 (d, J = 5.30 Hz, 2H, CH2=CHCH2), 5.29-5.43 (m, 2H, CH2=CHCH2), 6.06-6.12 (m, 1H, CH2=CHCH2) 7.87-8.31 (m, 3H, Ar-H), 9.90 (s, 1H, NH). 13C-NMR: 25.34, 69.53, 70.55, 113.12, 114.59, 118.32, 124.41, 133.52, 134.26, 134.95, 150.72, 161.12, 168.50. MS, m/z (%): 270 (M+, 94).
2-Benzyloxy-7,8-dimethoxy[1,2,4]triazolo[[1,5-a]]quinazolin-5-yl-hydrazine (10d). IR (cm−1): ν 3,205, 3,286 (NH-NH2). 1H-NMR (DMSO-d6): δ 3.08 (s, 3H, OCH3), 3.43 (s, 3H, OCH3), 4.82 (s, 2H, OCH2Ph), 5.40 (s, 2H, NH2), 7.93-8.32 (m, 7H, Ar-H), 9.84 (s, 1H, NH). 13C-NMR: 43.21, 47.87, 69.63, 113.85, 116.43, 118.20, 125.12, 128.15, 132.62, 135.15, 135.66, 147.34, 159.45, 166.93. MS, m/z (%): 366 (M+, 72).
3.2.7. Synthesis of compounds 11a-d
A mixture of 10 (1 mmol) and aldehyde or ketone (1 mmol) was refluxed in EtOH (10 mL) for 3 h. The solvent was removed under reduced pressure, and the resulting solids were recrystallized from EtOH.
N-Isopropylidene-N'-(8-methyl-2-methylsulfanyl[1,2,4]triazolo[[1,5-a]]quinazolin-5-yl)hydrazine (11a). 1H-NMR (DMSO-d6): δ 2.21 (s, 3H, CH3-isopropyl), 2.63 (s, 3H, CH3-isopropyl), 2.85 (s, 3H, CH3), 3.45 (s, 3H, SCH3), 7.37-7.94 (m, 3H, Ar-H), 10.45 (s, 1H, NH). 13C-NMR: 13.80, 18.67, 25.27, 45.34, 115.08, 124.90, 125.75, 126.06, 134.24, 134.96, 162.37, 164.54. MS, m/z (%): 300 (M+, 79).
N-Benzylidene-N'-(8-methy-2-methylsulfanyl[1,2,4]triazolo[[1,5-a]]-quinazolin-5-yl)hydrazine (11b). 1H-NMR (DMSO-d6): δ 2.92 (s, 3H, CH3), 3.34 (s, 3H, SCH3), 4.33 (s, 1H, CH-benzylidene), 7.45-8.05 (m, 8H, Ar-H), 11.83 (s, 1H, NH). 13C-NMR: 13.78, 25.17, 69.79, 110.23, 114.12, 115.37, 124.65, 127.54, 129.20, 131.76, 133.54, 141.32, 154.20, 168.97. MS, m/z (%): 348 (M+, 90).
N-(2-Benzyloxy-7,8-dimethoxy[1,2,4]triazolo[1,5-a]quinazolin-5-yl)-N'-isopropylidene-hydrazine (11c). 1H-NMR (DMSO-d6): δ 2.12 (s, 3H, CH3-isopropyl), 2.30 (s, 3H, CH3-isopropyl ), 2.87 (s, 3H, OCH3), 3.33 (s, 3H, OCH3), 5.54 (s, 2H, OCH2Ph), 6.34 (s, 1H, NH), 7.37-8.63 (m, 7H, Ar-H). 13C-NMR: 12.71, 13.43, 52.65, 58.43, 70.88, 103.16, 109.37, 114.05, 121.53, 124.69, 128.18, 128.76, 136.08, 136.74, 143.30, 150.85, 152.63, 169.21. MS, m/z (%): 406 (M+, 80).
N-(2-Phenethyloxy)-7,8-dimethoxy[1,2,4]triazolo[1,5-a]quinazolin-5-yl)-N'-(1-phenyl-ethylidene)-hydrazine (11d). 1H-NMR (DMSO-d6): δ 2.56 (s, 3H, OCH3), 3.17 (t, J = 7.74 Hz, 2H, OCH2CH2Ph), 3.34 (s, 3Η, CH3-ethylidene), 3.83 (s, 3H, OCH3), 4.77 (t, J = 7.71 Hz, 2H, OCH2CH2Ph), 7.25-8.55 (m, 12H, Ar-H), 9.91 (s, 1H, NH). 13C-NMR: 14.65, 35.10, 51.11, 56.43, 69.79, 110.73, 114.62, 116.73, 124.65, 125.33, 126.23, 128.11, 129.20, 131.11, 131.58, 132.27, 135.78, 139.52, 141.32, 145.34, 152.20, 161.57. MS, m/z (%): 482 (M+, 64).
3.2.8. Synthesis of compounds 12a,b
A mixture of 10 (0.5 mmol) and 1,1'-carbonyldiimidazole (0.6 mmol) was refluxed in absolute toluene (7 mL) for 3 h. The solvent was removed under reduced pressure and the residue was treated with CHCl3. The resulting solid was separated by filtration and recrystallized from EtOH.
2-Allyloxy-8-methyl-bis[1,2,4]triazolo[1,5-a:4',3'-c]quinazolin-3-one (12a). IR (cm−1): ν 1,702 (C=O). 1H-NMR (DMSO-d6): δ 3.01 (s, 3H, CH3), 4.60 (d, J = 5.54 Hz, 2H, CH2=CHCH2), 5.20-5.39 (m, 2H, CH2=CHCH2), 6.10-6.18 (m, 1H, CH2=CHCH2), 7.31-7.92 (m, 3H, Ar-H), 12.24 (s, 1H, NH). 13C-NMR: 24.74, 65.09, 114.92, 125.12, 127.64, 129.03, 131.23, 135.66, 136.76, 148.73, 157.43, 168.37. MS, m/z (%): 296 (M+, 75).
2-Benzyloxy-7,8-dimethoxy-bis[1,2,4]triazolo[1,5-a:4',3'-c]quinazolin-3-one (12b). IR (cm−1): ν 1,711 (C=O). 1H-NMR (DMSO-d6): δ 3.75 (s, 3H, OCH3), 4.03 (s, 3H, OCH3), 5.41 (s, 2H, OCH2Ph), 7.38-8.22 (m, 7H, Ar-H), 12.87 (s, 1H, NH). 13C-NMR: 54.76, 56.65, 70.53, 110.38, 114.59, 120.67, 124.87, 128.48, 133.33, 134.30, 134.89, 136.89, 147.67, 153.38, 156.80, 168.62. MS, m/z (%): 392 (M+, 82).
3.2.9. Synthesis of compounds 13a,b
A mixture of 10 (0.5 mmol) and CS2 (2.5 mmol) in pyridine (5 mL) was refluxed for 2 h. After cooling, the mixture was poured into ice/water, the yellow precipitate was filtered off, washed with water and recrystallized from MeOH.
2-Benzyloxy-7,8-dimethoxy-bis[1,2,4]triazolo[1,5-a:4',3'-c]quinazolin-3-thione (13a). 1H-NMR (DMSO-d6): δ 3.88 (s, 3H, OCH3), 4.43 (s, 3H, OCH3), 5.11 (s, 2H, OCH2Ph), 7.43-8.18 (m, 7H, Ar-H), 14.60 (s, 1H, NH). 13C-NMR: 49.06, 55.78, 71.52, 112.05, 115.19, 124.16, 125.63, 126.87, 128.34, 128.83, 133.33, 134.42, 136.15, 142.06, 157.12, 163.17, 185.73. MS, m/z (%): 408 (M+, 90).
8-Methyl-2-methylsulfanyl-bis[1,2,4]triazolo[1,5-a:4',3'-c]quinazolin-3-thione (13b). 1H-NMR (DMSO-d6): δ 2.82 (s, 3H, CH3), 3.90 (s, 3H, SCH3) 7.74-8.15 (m, 3H, Ar-H), 14.68 (s, 1H, NH). 13C-NMR: 13.72, 24.60, 114.75, 115.23, 126.57, 129.53, 1350, 136.32, 148.91, 159.94, 162.30, 185.05. MS, m/z (%): 302 (M+, 83).
3.2.10. Synthesis of compounds 14a,b
A mixture of 9 (1 mmol) and the corresponding carbohydrazide (2.2 mmol) was refluxed in toluene (10 mL) for 2.5 h. After cooling, the solid was collected by filtration. Analytically pure products 14a,b were obtained by recrystallization from MeOH.
8-Methyl-N-(2-methylsulfanyl[1,2,4]triazolo[[1,5-a]]quinazolin-5-yl)-benzohydrazide (14a). IR (cm−1): ν 1,660 (C=O), 3,184 (NH). 1H-NMR (DMSO-d6): δ 2.98 (s, 3H, CH3), 4.01(s, 3H, SCH3), 7.53-8.51 (m, 8H, Ar-H), 10.39 (s, 1H, NH), 10.97 (s, 1H, NH). 13C-NMR: 13.78, 25.67, 109.83, 114.88, 124.93, 125.29, 127.41, 127.99, 128.87, 129.07, 132.21, 132.81, 133.04, 135.09, 157.07, 168.00. MS, m/z (%): 364 (M+, 92).
8-Methyl-N-(2-methylsulfanyl[1,2,4]triazolo[[1,5-a]]quinazolin-5-yl)-isonicotinichydrazide (14b). IR (cm−1): ν 1,673 (C=O), 3,207 (NH). 1H-NMR (DMSO-d6): δ 3.08 (s, 3H, CH3), 3.94 (s, 3H, SCH3), 7.65-8.50 (m, 7H, Ar-H), 10.66 (s, 1H, NH), 11.14 (s, 1H, NH). 13C-NMR: 13.56, 24.69, 109.70, 114.94, 121.73, 124.88, 125.36, 128.45, 133.20, 135.21, 139.74, 150.94, 155.50, 164.82. MS, m/z (%): 365 (M+, 76).
3.2.11. Synthesis of compounds 15a,b
A mixture of 9 (1 mmol) and benzyl carbazate or ethyl carbazate (2.2 mmol) was refluxed in benzene (10 mL) for 2.5 h. The solvent was removed under reduced pressure, the resulting solid was filtered off and recrystallized from MeOH.
Ethyl-N-(2-allyloxy-8-methyl[1,2,4]triazolo[[1,5-a]]quinazolin-5-yl)-hydrazine-carboxylate (15a). IR (cm−1): ν 1708 (C=O), 3198 (NH). 1H-NMR (DMSO-d6): δ 1.13 (t, J = 7.61 Hz, 3H, OCH2CH3), 2.87 (s, 3H, CH3), 4.08 (q, J = 10.12 Hz, 2H, OCH2CH3), 4.60 (d, J = 5.54 Hz, 2H, CH2=CHCH2), 5.07-5.19 (m, 2H, CH2=CHCH2), 6.06-6.13 (m, 1H, CH2=CHCH2) 7.54-7.93 (m, 3H, Ar-H), 9.50 (s, 1H, NH), 10.34 (s, 1H, NH). 13C-NMR: 14.92, 23.89, 57.33, 63.75, 109.26, 114.60, 124.79, 126.21, 127.07, 134.70, 137.15, 156.69, 169.23. MS, m/z (%): 342 (M+, 80).
Benzyl-N-(8-methyl-2-methylsulfanyl[1,2,4]triazolo[[1,5-a]]quinazolin-5-yl)-hydrazine-carboxylate (15b). IR (cm−1): ν 1718 (C=O), 3261 (NH). 1H-NMR (DMSO-d6): δ 3.74 (s, 3H, CH3), 4.11 (s, 3H, SCH3), 5.35 (s, 2H, OCH2Ph), 7.25-8.43 (m, 8H, Ar-H), 9.88 (s, 1H, NH), 10.44 (s, 1H, NH). 13C-NMR: 13.54, 25.03, 66.48, 109.62, 114.85, 124.75, 125.25, 126.25, 128.54, 128.81, 135.11, 135.33, 136.98, 152.74, 157.10, 168.42. MS, m/z (%): 394 (M+, 100).
3.2.12. Synthesis of compounds 16a,b
A mixture of 14 (0.5 mmol) and POCl3 (5 mL) was refluxed at 100°C for 2 h. After cooling, the excess of POCl3 was removed under reduced pressure and the residue was treated with saturated aqueous solution of K2CO3 under ice cooling. The resulting solids were collected by filtration and recrystallized from MeOH to afford 16a,b as colored pure products.
8-Methyl-2-methylsulfanyl-3-phenyl-bis[1,2,4]triazolo[1,5-a:4',3'-c]quinazoline (16a). 1H-NMR (DMSO-d6): δ 3.33 (s, 3H, CH3), 3.91 (s, 3H, SCH3), 7.74-8.48 (m, 8H, Ar-H). 13C-NMR: 13.65, 24.06, 111.94, 115.17, 124.56, 126.12, 127.09, 128.35, 129.80, 130.59, 131.09, 141.82, 143.67, 149.47, 167.34. MS, m/z (%): 346 (M+, 82).
8-Methyl-2-methylsulfanyl-3-pyridyl-bis[1,2,4]triazolo[1,5-a:4',3'-c]quinazoline (16b). 1H-NMR (DMSO-d6): δ 3.66 (s, 3H, CH3), 4.19 (s, 3H, SCH3), 7.53-8.41 (m, 7H, Ar-H). 13C-NMR: 13.42, 25.03, 111.24, 115.23, 124.39, 125.75, 128.21, 129.33, 131.16, 133.35, 142.52, 145.67, 150.03, 161.24. MS, m/z (%): 347 (M+, 95).
3.2.13. Synthesis of compounds 17a-c
A mixture of 9 (1 mmol) and NaN3 (1.2 mmol) in absolute DMF (5 mL) was heated at 90 °C in a nitrogen atmosphere for 24 h. After cooling, the reaction mixture was poured into water and saturated with brine solution. The resulting solid was filtered off, dried and recrystallized from MeOH.
8-Methyl-2-methylsulfanyl-tetrazolo[4,3-c][1,2,4]triazolo[[1,5-a]]quinazoline (17a). 1H-NMR (DMSO-d6): δ 3.32 (s, 3H, CH3), 3.99 (s, 3H, SCH3), 7.48-7.95 (m, 3H, Ar-H). 13C-NMR: 13.89, 24.67, 114.67, 116.23, 125.81, 128.27, 134.74, 136.49, 145.01, 157.32, 167.54. MS, m/z (%): 271 (M+, 77).
2-Benzyloxy-7,8-dimethoxy-tetrazolo[4,3-c][1,2,4]triazolo[[1,5-a]]quinazoline (17b). 1H-NMR (DMSO-d6): δ 3.48 (s, 3H, OCH3), 4.50 (s, 3H, OCH3), 5.75 (s, 2H, OCH2Ph), 7.44-8.28 (m, 7H, Ar-H). 13C-NMR: 45.98, 51.72, 71.57, 109.58, 115.08, 125.5, 127.72, 128.23, 130.36, 134.21, 134.53, 135.47, 148.17, 160.43, 167.16. MS, m/z (%): 377 (M+, 90).
7,8-Dimethoxy-2-phenethyloxy-tetrazolo[4,3-c][1,2,4]triazolo[[1,5-a]]quinazoline (17c). 1H-NMR (DMSO-d6): δ 3.42 (t, J = 7.50 Hz, 2H, OCH2CH2Ph), 3.62 (s, 3H, OCH3), 4.53 (s, 3H, OCH3), 4.79 (t, J = 7.51 Hz, 2H, OCH2CH2Ph), 7.23-8.67 (m, 7H, Ar-H). 13C-NMR: 41.43, 44.76, 64.83, 71.21, 115.57, 124.39, 126.10, 126.87, 127.56, 128.80, 129.35, 130.23, 134.99, 138.27, 142.32, 156.34, 167.54. MS, m/z (%): 391 (M+, 100).
3.2.14. Synthesis of compounds 18a,b
A mixture of 9 (1 mmol) and 3-aminothiophene-2-methylcarboxylic acid ester (2.2 mmol) in absolute dioxane (10 mL) was refluxed in the presence of NaH (0.4 mmol) for 21 h. The solvent was removed under reduced pressure and the residue was treated with water and MeOH. The resulting solid was filtered off and dried.
8-Methyl-2-pentyloxy-(3H-thieno[3,2-d]pyrimidin-4-one[4,3-c][1,2,4]triazolo[1,5-a]quinazoline (18a). IR (cm−1): ν 1,677 (C=O). 1H-NMR (DMSO-d6): δ 0.73 (t, J = 7.54 Hz, 3H, OCH2CH2CH2CH2CH3), 1.14-1.20 (m, 4H, OCH2CH2CH2CH2CH3), 1.48-1.67 (m, 2H, OCH2CH2CH2CH2CH3), 2.87 (s, 3H, CH3), 4.08 (t, J = 7.60 Hz, 2H, OCH2CH2CH2CH2CH3), 6.78-8.35 (m, 5H, Ar-H). 13C-NMR: 14.63, 22.18, 24.65, 27.69, 28.63, 69.35, 114.82, 123.71, 124.25, 124.80, 134.33, 134.89, 147.72, 153.60, 167.84. MS, m/z (%): 393 (M+, 61).
7,8-Dimethoxy-2-phenethyloxy-(3H-thieno[3,2-d]pyrimidin-4-one[4,3-c][1,2,4]triazolo[1,5-a]-quinazoline (18b). IR (cm−1): ν 1,670 (C=O). 1H-NMR (DMSO-d6): δ 3.67 (s, 3H, OCH3), 3.90 (t, J = 7.61 Hz, 2H, OCH2CH2Ph), 4.31 (s, 3H, OCH3), 4.60 (t, J = 7.63 Hz, 2H, OCH2CH2Ph), 6.38-8.15 (m, 9H, Ar-H). 13C-NMR: 36.40, 40.61, 54.70, 63.57, 109.08, 116.08, 124.25, 127.02, 128.13, 128.75, 131.36, 131.93, 133.23, 133.91, 134.53, 135.47, 148.17, 160.43, 167.16. MS, m/z (%): 473 (M+, 70).