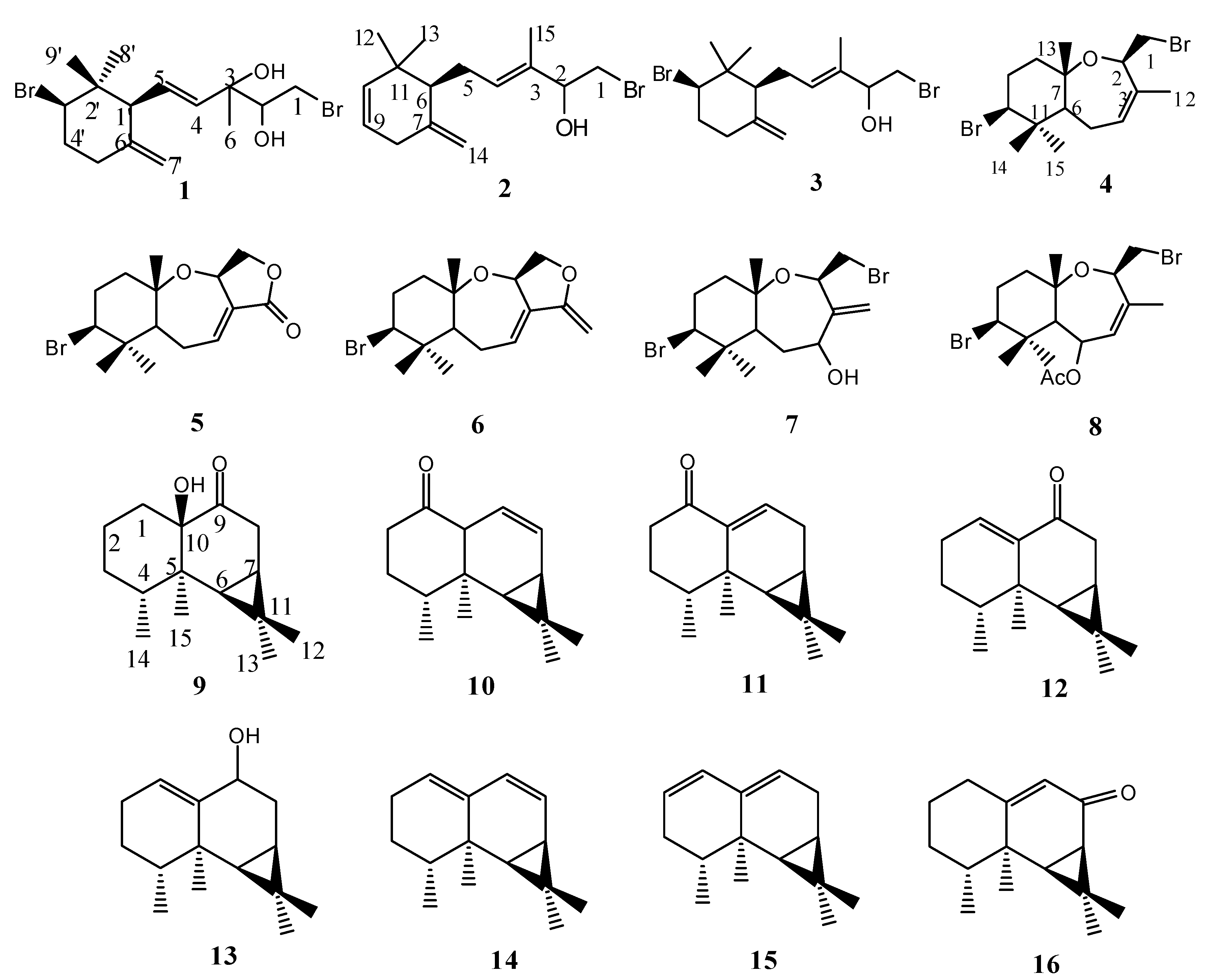
3.3. Extraction and Isolation
The air-dried alga L. similis (1.1 kg) was powdered and extracted with 95% EtOH at room temperature. The resulting extractive solution was filtered and evaporated under reduced pressure (<45 ℃) to yield a dark residue (34.2 g), and then partitioned between H2O and EtOAc. The EtOAc phase (28.5 g) was subjected to column chromatography (CC) over silica gel eluting with a gradient of increasing EtOAc in petroleum ether (PE, 0~100%) to give 7 fractions on the basis of TLC analysis. Fraction III (1.9 g) were further chromatographed over Sephadex LH-20 eluting with PE-CHCl3-MeOH (5:5:1) to yield compounds 1 (6.8 mg), 2 (5.7 mg), 4 (6.7 mg), 5 (7.3 mg) and 8 (7.3 mg) respectively. Fraction IV (0.6 g) was further chromatographed over Si gel eluting with a gradient of increasing EtOAc (35-100%) in PE to yield 3 (8.3 mg), 6 (7.9 mg), 10 (8.6 mg), 11 (10.0 mg), 14 (5.6 mg) and 16 (5.6 mg). Fraction Ⅴ (3.6 g) was decolored by column chromatography over Sephadex LH-20 eluting with PE-CHCl3-MeOH (5:5:1) and preparative TLC (PE/EtOAc 40:3) to afford compound 7 (7.3 mg), 9 (8.3 mg), 12 (11.1 mg), 13 (9.7 mg) and 15 (7.5 mg).
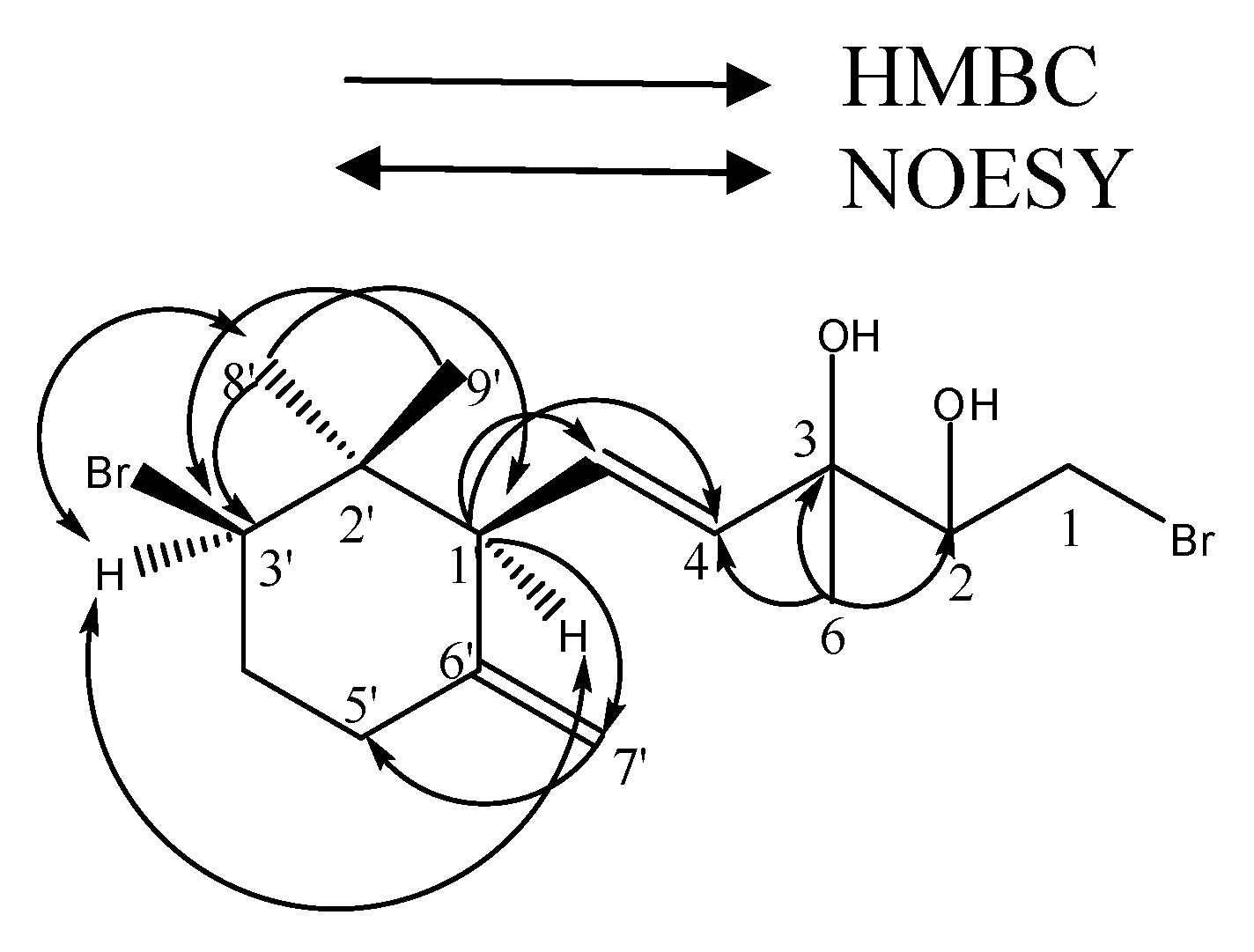
(4E)-1-bromo-5-[(1'S*, 3'R*)-3'-bromo-2', 2'-dimethyl-6'-methylenecyclohexyl]-3-methylpent-4-ene-2, 3-diol (
1): colorless oil; IR (film) v
max 3410, 3012, 2921, 1440, 1065, 990 cm
-1; [α]20D +18.4 (CHCl
3,
c 0.30);
1H NMR (CDCl
3) (see
Table 1);
13C NMR data (CDCl
3) (see
Table 1). ESI-MS:
m/z 399/401/403 (1:2:1) [M-H
2O+Na]
+. HR-ESI-MS:
m/z 401.1441([M-H
2O+Na]
+) (calcd for 401.1426, C
15H
22Br
2ONa).
Isopalisol (2): C15H23BrO, colorless oil; EI-MS m/z(%): 282 (3), 280 (3, M-H2O), 265 (8), 201 (15), 121 (100), 105 (48), 93 (55); 1H-NMR(CDCl3, 500MHz) δ: 3.45 (2H, m, H2-1), 4.22 (1H, t, J=5.0Hz, H-2), 5.48 (1H, m, H-4), 2.25 (1H, m, H-5α), 2.90 (1H, m, H-5β), 2.00 (1H, m, H-6), 2.64 (2H, m, H2-8), 5.52 (1H, m, H-9), 5.37 (1H, m, H-10), 0.95 (3H, s, H3-12), 1.02 (3H, s, H3-13), 4.60 (1H, s, H-14α), 4.86 (1H, s, H-14β),1.65 (3H, s, H3-15); 13C-NMR(CDCl3, 125MHz) δ: 38.5 (t, C-1), 76.5 (d, C-2), 133.4 (s, C-3), 128.4 (d, C-4), 25.2 (t, C-5), 52.4 (d, C-6), 145.5 (s, C-7), 32.4 (t, C-8), 123.1 (d, C-9), 136.9 (q, C-10), 37.1 (s, C-11), 25.1 (q, C-12), 30.3 (q, C-13), 109.7 (t, C-14), 12.2 (q, C-15).
Luzonensol (3): C15H24Br2O, colorless oil; EI-MS m/z: 364 (3), 362 (6), 360 (3, M-H2O), 281 (100), 201 (47),147 (30), 121 (80); 1H-NMR(CDCl3, 500MHz) δ: 3.49 (1H, dd, J=9.0, 10.0 Hz, H-1α), 3.40 (1H, dd, J=4.5, 10.0 Hz, H-1β), 4.74 (1H, dd, J=4.5, 9.0 Hz, H-2), 5.29 (1H, m, H-4), 2.45 (1H, m, H-5α), 2.25 (1H, m, H-5β), 1.83 (1H, m, H-6), 2.04 (1H, m, H-8α), 2.35 (1H, m, H-8β), 2.08 (1H, m, H-9α), 2.30 (1H, m, H-9β), 4.15 (1H, dd, J=4.5, 11.5Hz, H-10), 0.87 (3H, s, H3-12), 1.20 (3H, s, H3-13), 4.56 (1H, s, H-14α), 4.91 (1H, s, H-14β), 1.69 (3H, s, H3-15); 13C-NMR(CDCl3,125MHz) δ: 36.9 (t, C-1), 70.1 (d, C-2), 133.1 (s, C-3), 129.9 (d, C-4), 24.7 (t, C-5), 53.2 (d, C-6), 145.3 (s, C-7), 37.2 (t, C-8), 35.6 (t, C-9), 66.7 (d, C-10), 41.6 (s, C-11), 16.6 (q, C-12), 28.4 (q, C-13), 110.0 (t, C-14), 17.4 (q, C-15).
Palisadin B (4): C15H24OBr2, colorless oil; ESI-MS m/z: 378/380/382; 1H-NMR (CDCl3, 500MHz) δ: 3.41 (1H, dd, J=7, 11Hz, H-1α), 3.73 (1H, dd, J=3, 11Hz, H-1β), 4.54 (1H, m, H-2), 5.63 (1H, d, J=8 Hz, H-4), 2.05 (3H, m, H2-5 and H-6), 1.77 (2H, m, H2-8), 2.25 (2H, m, H2-9), 3.95 (1H, dd, J=5, 12 Hz, H-10), 1.27 (3H, s, H3-12), 1.36 (3H, s, H3-13), 0.95 (3H, s, H3-14), 1.15 (3H, s, H3-15); 13C-NMR(CDCl3,125MHz) δ: 36.2 (t, C-1), 70.7 (d, C-2), 136.1 (s, C-3), 129.4 (d, C-4), 25.9 (t, C-5), 52.8 (d, C-6), 77.5 (s, C-7), 36.7 (t, C-8), 32.9 (t, C-9), 66.3 (d, C-10), 40.8 (s, C-11), 21.0 (q, C-12), 22.0 (q, C-13), 17.9 (q, C-14), 30.7 (q, C-15).
Aplysistatin (5): C15H21BrO3, colorless oil; EI-MS m/z: 330/328[M]+ (1), 300/298 (4), 286/284(8), 249(5), 218/216(8), 204/202(33), 189/187(19), 139(24), 123(100), 107(39); 1H-NMR (CDCl3, 500MHz) δ: 0.96(3H, s, H3-13), 1.18(3H, s, H3-15), 1.30(3H, s, H3-14), 3.86(1H, dd, J = 7.0 , 9.0 Hz, H-1α), 3.91(1H, m, H-10), 4.48(1H, t, J = 9.0, 9.0 Hz, H-1β), 5.13(1H, m, H-2), 6.95(1H, m, H-4). 13C-NMR (CDCl3,125MHz) δ: 69.9(C-1), 66.8(C-2), 132.0(C-3), 143.0(C-4),27.2(C-5), 51.3(C-6), 79.0(C-7), 32.4(C-8), 37.7(C-9), 65.1(C-10), 41.0(C-11), 169.3(C-12), 18.0(C-13), 30.7(C-14), 21.7(C-15).
Palisadin A (6): C15H23BrO2, colorless oil; EI-MS m/z: 314/316; 1H-NMR (CDCl3, 500MHz) δ: 1.00(3H, s, H3-14), 1.25(3H, s, H3-15), 1.37(3H, s, H3-13), 1.55(1H, m, H-8α), 1.85(1H, m, H-8β), 2.25(2H, m, H2-9), 2.40(2H, m, H2-5), 2.40(1H, m, H-6), 3.93(1H, dd, J = 8.0, 8.0 Hz, H-1α), 3.95(1H, dd, J = 5.0, 12.0 Hz, H-10), 4.10(1H, dd, J = 8.0, 8.0 Hz, H-1β), 4.45(2H, m, H2-12), 4.83(1H, m, H-2), 5.63(1H, br s, H-4). 13C-NMR (CDCl3, 125MHz) δ: 75.4(C-1), 71.0(C-2), 141.9(C-3), 121.1(C-4), 26.3(C-5), 51.8(C-6), 78.0(C-7), 37.6(C-8), 32.7(C-9), 66.2(C-10), 41.0(C-11), 72.0(C-12), 21.9(C-13), 18.0(C-14), 30.8(C-15).
4-hydroxyl-Palisudin C (7): C15H24Br2O2, colorless oil; EIMS: m/z 394 396 398 [M]+ (1:2:1), 376 378 380 [M-H2O]+ (1:2:1); 1H-NMR (CDCl3, 500MHz) δ: 3.31 (1H, t, J = 10.5 Hz, H-1α), 3.44 (1H, dd, J = 8.8, 10.5 Hz, H-1β), 4.43 (1H, d, J = 7.7 Hz, H-2), 4.86 (1H, t, J = 8.1 Hz, H-4), 1.43–1.46 (1H, m, H-5α), 2.20–2.24 (1H, m, H-5β), 1.61–1.62 (1H, m, H-6), 1.59–1.60 (1H, m, H-8α), 1.70–1.73 (1H, m, H-8β), 2.05–2.07 (1H, m, H-9α), 2.22–2.23 (1H, m, H-9β), 3.90 (1H, dd, J = 4.3,12.8 Hz, H-10), 4.91 (1H, s, H-12α), 5.23 (1H, s, H-12β), 1.26 (3H, s, H3-13), 0.97 (3H, s, H3-14), 1.10 (3H, s, H3-15); 13C-NMR (CDCl3, 125MHz) δ: 37.3 (t, C-1), 70.9 (d, C-2), 153.2 (s, C-3), 68.8 (d, C-4), 34.9 (t, C-5), 48.8 (d, C-6), 77.5 (s, C-7), 36.9 (t, C-8), 32.3 (t, C-9), 66.1 (d, C-10), 40.3 (s, C-11), 107.1 (t, C-12), 22.3 (q, C-13), 17.8 (q, C-14), 30.8 (q, C-15).
5-Acetoxypalisadin B (8): C17H26Br2O3, colorless oil; EI-MS m/z(%): 436/438/440; 1H-NMR (CDCl3, 500MHz) δ: 1.01(3H, s, H3-14), 1.20(3H, s, H3-15), 1.63(3H, s, H3-13), 1.80(3H, s, H3-12), 2.08(3H, s, H3-OAc), 1.58(1H, m, H-8α), 1.84 (1H, m, H-8β), 1.73(1H, m, H-6), 2.15~2.31(2H, m, H2-9), 3.41(1H, dd, J = 8.0, 11.0 Hz, H-1α), 3.70(1H, dd, J = 3.0, 11.0 Hz, H-1β), 3.87(1H, dd, J = 4.0, 12.0 Hz, H-10), 4.44(1H, d, J = 10.0 Hz, H-2), 5.69(1H, d, J = 8.0 Hz H-4), 5.78(1H, d, J = 8.0 Hz, H-5); 13C-NMR(CDCl3,125MHz) δ: [25.2, 170.3(C-OAc)], 34.8(C-1), 70.1(C-2), 142.5(C-3), 127.0(C-4), 69.6(C-5), 53.7(C-6), 77.8(C-7), 39.3(C-8), 32.7(C-9), 66.1(C-10), 41.3(C-11), 21.2(C-12), 21.5(C-13), 18.7(C-14), 30.8(C-15).
10-Hydroxyaristolan-9-one (9): C15H24O2, colorless oil; FAB-MS(pos.): 236 [M]+; 1H-NMR (CDCl3, 500MHz) δ: 0.80(1H, d, J = 9.1 Hz, H-6), 0.94(3H, d, J = 6.7 Hz, H3-14), 1.05(3H, s, H3-12), 1.06(3H, s, H3-13), 1.20(3H, s, H3-15), 1.22~1.33(3H, m, H2-3 and H-7), 1.38(1H, m, H-1α), 1.47 (1H, m, H-4), 1.56 (1H, m, H-2α), 1.65 (1H, m, H-2β), 1.92(1H, m, H-1β), 2.26 (1H, d, J=17.0 Hz, H-8α), 3.09 (1H, dd, J = 9.0, 17.0 Hz, H-8β); 13C-NMR (CDCl3,125MHz) δ: 29.5 (t, C-1), 22.8 (t, C-2), 29.1 (t, C-3), 34.8 (d, C-4), 46.8 (s, C-5), 29.0 (d, C-6),22.9 (d, C-7), 34.5 (t, C-8),215.9 (s, C-9), 77.7(s, C-10), 21.0 (t, C-11), 16.8 (q, C-12), 30.6 (q, C-13),17.0 (q, C-14), 18.4 (q, C-15).
Aristol-8-en-1-one (10): C15H22O, colorless oil; FAB-MS (pos.): 218 [M]+; 1H-NMR (CDCl3, 500MHz) δ: 0.56 (3H, s, H3-15), 0.78 (1H, d, J = 8.5 Hz, H-6), 1.01 (3H, s, H3-12), 1.05 (3H, d, J = 6.6 Hz, H3-14), 1.16 (3H, s, H3-13), 1.23 (1H, dd, J = 5.6, 8.5 Hz, H-7), 1.69 (1H, m, H-3α), 1.94 (1H, m, H-3β), 1.96 (1H, m, H-4), 2.36 (2H, m, H2-2), 2.58 (1H, br s, H-10), 5.86 (1H, dd, J = 1.9, 10.1 Hz, H-9), 6.02 (1H, m, H-8, H-8). 13C-NMR (CDCl3, 125MHz) δ: 209.9 (s, C-1),40.5 (t, C-2), 31.4 (t, C-3), 40.1 (d, C-4),39.1 (s, C-5), 35.1 (d, C-6), 24.7 (d, C-7), 127.3 (d, C-8), 118.8 (d, C-9), 54.7 (d, C-10), 26.1 (s, C-11), 16.0 (q, C-12),30.5 (q, C-13), 15.5 (q, C-14), 15.9 (q, C-15).
Aristolan-9-en-1-one (11): C15H22O, colorless oil; 1H-NMR (CDCl3, 500MHz) δ: 0.71 (1H, d, J = 9.4 Hz, H-6), 0.87 (1H, m, H-7), 0.99 (3H, H, s, H3-15), 1.03 (3H, s, H3-12), 1.06 (3H, d, J = 6.8 Hz, H3-14), 1.07 (3H, s, H3-13), 1.73 (2H, m, H2-3), 1.98 (1H, m, H-4), 2.28 (1H, m, H-2α), 2.34 (1H, m, H-8α), 2.50 (1H, m, H-2β), 2.55 (1H, m, H- 8β), 6.41 (1H, m, H-9α). 13C-NMR (CDCl3, 125MHz) δ: 202.0 (s, C-1), 40.0 (t, C-2), 27.8 (t, C-3), 35.9 (d, C-4), 37.8 (s, C-5), 32.0 (d, C-6), 19.3 (d, C-7), 22.5 (t, C-8), 132.5 (d, C-9), 143.5 (s, C-10), 18.8 (s, C-11), 15.3 (q, C-12), 29.8 (q, C-13),15.4 (q, C-14), 22.6 (q, C-15).
Aristolan-1(10)-en-9-one (12): C15H22O, colorless oil; 1H-NMR (CDCl3, 500MHz) δ: 0.90 (1H, d, J = 9.4 Hz, H-6), 0.96 (1H, m, H-7), 0.99 (3H, s, H3-12), 1.02(3H, s, H3-15), 1.04 (3H, d, J = 5.8 Hz, H3-14), 1.05 (3H, s, H3-13), 1.50 (2H, m, H2-3), 1.76 (1H, m, H-4), 2.24 (2H, m, H2-2), 2.66 (1H, dd, J = 1.1, 20.0Hz, H-8α), 2.78 (1H, dd, J = 6.1, 20.0 Hz, H-8β), 6.71 (1H, m, H-1). 13C-NMR (CDCl3, 125MHz) δ: 136.4 (d, C-1), 25.9 (t, C-2), 26.1 (t, C-3), 36.7 (d, C-4), 37.6 (s, C-5),29.8 (d, C-6), 19.0 (d, C-7), 34.6 (t, C-8), 200.2 (s, C-9), 142.5 (s, C-10),18.7 (s, C-11), 14.8 (q, C-12), 30.1 (q, C-13),15.9 (q, C-14), 24.0 (q, C-15).
Aristolan-1(10)-en-9-ol (13): C15H24O, colorless needles ; 1H-NMR (CDCl3, 500MHz) δ: 0.53 (1H, d, J = 9.1 Hz, H-6), 0.75 (1H, m, H-7), 0.97 (3H, s, H3-12), 1.07(3H, s, H3-15), 0.95 (3H, d, J = 6.9 Hz, H3-14), 0.98 (3H, s, H3-13), 1.38 (2H, m, H2-3), 1.75 (1H, m, H-4), 2.01 (2H, m, H2-2), 1.22 (1H, m, H-8α), 2.32 (1H, m, H-8β), 5.53 (1H, m, H-1). 13C-NMR (CDCl3, 125MHz) δ: 116.4 (d, C-1), 25.4 (t, C-2), 26.7 (t, C-3), 36.9 (d, C-4), 38.8 (s, C-5), 32.9 (d, C-6),18.4 (d, C-7), 30.7 (t, C-8), 67.4 (d, C-9), 145.7 (s, C-10), 18.6 (s, C-11), 16.1 (q, C-12), 29.8 (q, C-13),16.6 (q, C-14),23.9 (q, C-15).
Aristolan-1(10)-8-diene (14): C15H22, colorless needles; 1H-NMR (CDCl3, 500MHz) δ: 0.94 (1H, d, J = 8.4 Hz, H-6), 1.17 (1H, dd, J = 5.3, 8.3 Hz, H-7), 0.92 (3H, s, H3-12), 0.96(3H, s, H3-15), 0.99 (3H, d, J = 6.9 Hz, H3-14), 1.11 (3H, s, H3-13), 1.53 (1H, m, H-3α), 1.67 (2H, m, H-3β and H-4), 2.16 (2H, m, H2-2), 5.73 (1H, dd, J = 5.3, 9.7 Hz,, H-8), 5.28 (1H, m, H-1), 5.85 (1H, d, J = 9.7Hz, , H-9). 13C-NMR (CDCl3, 125MHz) δ: 122.0 (d, C-1), 25.2 (t, C-2), 27.7 (t, C-3), 35.8(d, C-4), 34.6 (s, C-5), 34.5 (d, C-6), 24.4 (d, C-7), 124.3 (d, C-8), 126.7 (d, C-9), 141.7 (s, C-10), 26.3(s, C-11), 14.7(q, C-12), 29.3 (q, C-13), 15.9 (q, C-14), 23.0 (q, C-15).
Aristolan-1,9-diene (15): C15H22, colorless needles; 1H-NMR (CDCl3, 500MHz) δ: 0.67 (1H, d, J = 9.3 Hz, H-6), 0.81 (1H, dd, J = 7.1, 9.3 Hz, H-7), 1.03 (3H, m, H3-12), 0.96(3H, s, H3-15), 0.99 (3H, d, J = 6.9 Hz, H3-14), 1.04 (3H, m, H3-13), 1.90 (1H, dd, J = 11.2, 18.3 Hz, H-3α), 2.01(1H, m, H-3β), 5.55 (1H, dd, J = 5.6,9.8Hz, H-2), 2.23(1H, dd, J = 5.3, 20.1 Hz,, H-8α), 2.55(1H, dd, J = 7.1, 20.1 Hz, H-8β), 5.88 (1H, d, J = 9.8 Hz, H-1), 5.26 (1H, m, H-9). 13C-NMR (CDCl3, 125MHz) δ: 129.1(d, C-1), 125.2 (d, C-2), 32.3 (t, C-3), 34.1(d, C-4), 35.1 (s, C-5), 31.4 (d, C-6),19.7 (d, C-7), 22.4 (t, C-8), 122.7 (d, C-9),140.7 (s, C-10), 18.3(s, C-11),15.0(q, C-12), 29.9 (q, C-13),15.1 (q, C-14), 21.1 (q, C-15).
Aristofone (16): C15H22O, colorless needles; 1H-NMR (CDCl3, 500MHz) δ: 1.29~1.36 (3H, m, H2-1 and H-3β), 1.48 (1H, m, H-2β), 1.74~.178 (2H, m, H-2α and H-3α), 1.65 (1H, d, J = 8.0Hz, H-4), 2.18 (1H, d, J = 8.0 Hz, H-6), 2.34~2.40 (1H, m, H-7), 5.64 (1H, s, H-9), 1.40 (3H, s, H3-12), 1.92 (3H, s, H3-13), 0.99 (3H, dd, J = 2.4, 6.8 Hz, H3-14), 1.12 (3H, s, H3-15). 13C-NMR (CDCl3,125MHz) δ: 33.0 (t, C-l), 30.4 (t, C-2), 26.0 (t, C-3), 38.5 (d, C-4), 39.4 (s, C-5), 39.0(d, C-6), 35.4 (d, C-7), 195.9(s, C-8), 124.1 (d, C-9), 167.3 (s, C-l0), 24.1 (s, C-11), 16.2 (q, C-12), 29.5 (q, C-13), 22.4 (q, C-14), 16.3 (q, C-15); EIMS: 218 [M]+.