Pentacyclic Triterpenoids from the Medicinal Herb, Centella asiatica (L.) Urban
Abstract
:Introduction
Terpenoids as Natural Products and Secondary Metabolites
Chemical Diversity of Terpenoids
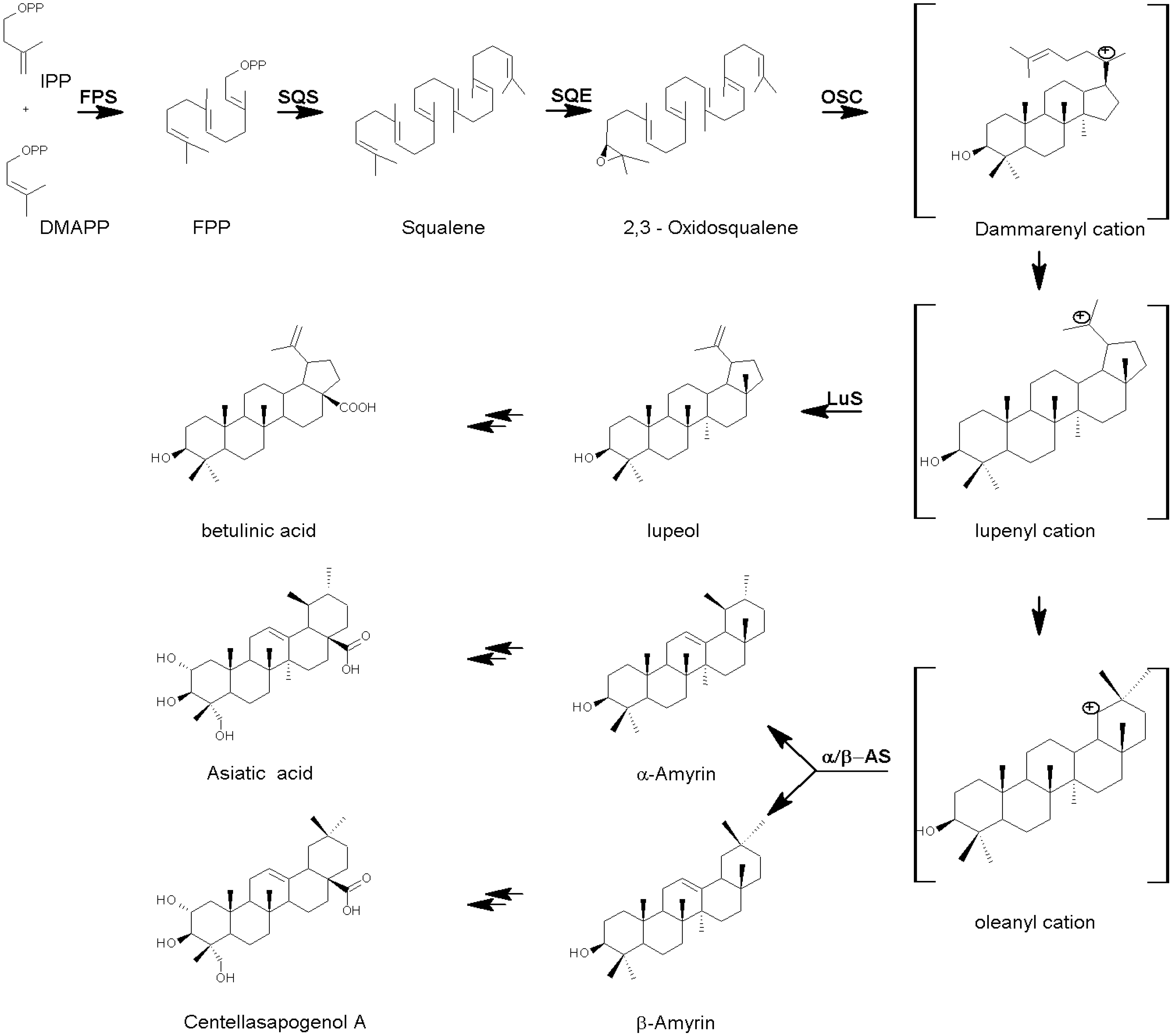
The Biosynthesis of Centella Triterpenes and Triterpenoids
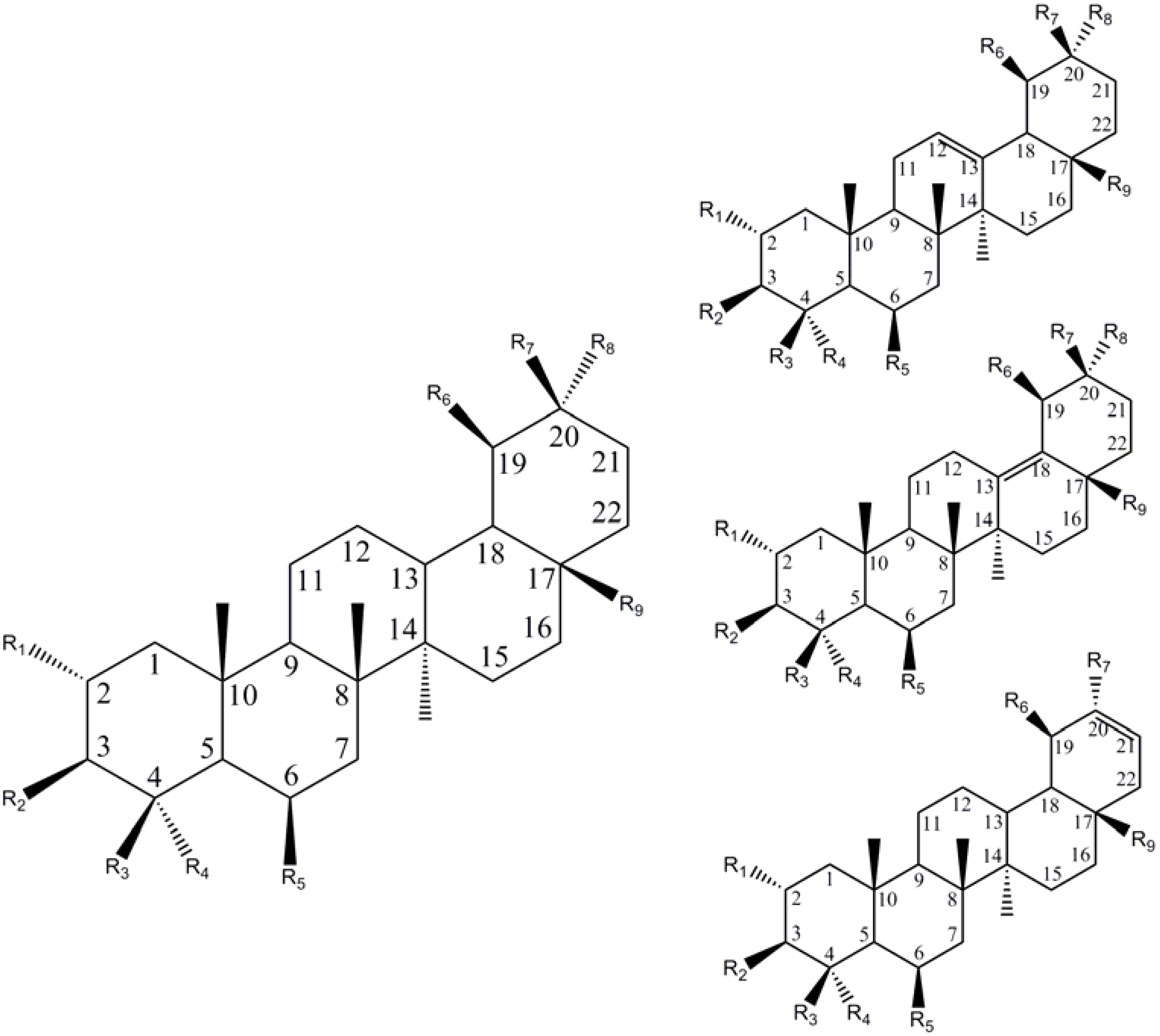
| R1 | R2 | R3 | R4 | R5 | R6 | R7 | R8 | R9 | C=C | Name | Ref |
| Ursane (C19,C20 – dimethyl) | |||||||||||
| OH | OH | CH3 | CH2OH | H | CH3 | CH3 | OH | COOH | - | 2α,3ß,20,23-tetrahydroxy-urs-28-oic acid | [46] |
| OH | OH | CH3 | CH2OH | H | CH3 | CH3 | H | COOH | 20-21 | 2α,3ß,23-trihydroxy-urs-20-en-28-oic acid | [46] |
| OH | OH | CH3 | CH2OH | H | CH3 | CH3 | H | COO-glc(1-6)glc(1-4)rha | 20-21 | Scheffuroside B | [46] |
| 2α,3ß,23-trihydroxy-urs-20-en-28-oic acid O-α-L-rhamnopyranosyl- (1-4)-O- β-D-glucopyranosyl-(1-6)-O- β-D-glucopyranosyl ester | [47] | ||||||||||
| OH | OH | CH3 | CH2OH | H | CH3 | CH3 | H | COOH | 12-13 | Asiatic acid | [36,48] |
| 2α,3ß,23-trihydroxy-urs-12-en-28-oic acid | |||||||||||
| OH | OH | CH3 | CH2OH | H | CH3 | CH3 | H | COOCH3 | 12-13 | Methyl asiatate | [48] |
| OH | OH | CH3 | CH2OH | H | CH3 | CH3 | H | COO-glc(1-6)glc(1-4)rha | 12-13 | Asiaticoside | [34,35,49] |
| 2α,3ß,23-trihydroxy-urs-12-en-28-oic acid O-α-L-rhamnopyranosyl- (1-4)-O- ß-D-glucopyranosyl-(1-6)-O- ß-D-glucopyranosyl ester | |||||||||||
| OH | -OH | CH3 | CH2OAc | H | CH3 | CH3 | H | COO-glc(1-6)glc(1-4)rha | 12-13 | Asiaticoside C | [47] |
| OH | -OH | CH3 | CH3 | H | CH3 | CH3 | H | COO-glc(1-6)glc(1-4)rha | 12-13 | Asiaticoside D | [47] |
| OH | OH | CH3 | CH2OH | H | CH3 | CH3 | H | COO-glc(1-6)glc | 12-13 | Asiaticoside E | [47] |
| H | OH | CH3 | CH2OH | H | CH3 | CH3 | H | COO-glc(1-6)glc(1-4)rha | 12-13 | Asiaticoside F | [47] |
| OH | -OH | CH3 | CH2OH | OH | CH3 | CH3 | H | COOH | 12-13 | Brahmic acid, Madecassic acid | [34,35,40,48,50] |
| (6ß-hydroxy-asiatic acid) | |||||||||||
| OH | OCH3 | CH3 | CH2OH | OH | CH3 | CH3 | H | COOCH3 | 12-13 | Methyl brahmate | [48] |
| OH | OH | CH3 | CH2OH | OH | CH3 | CH3 | H | CH2OH | 12-13 | Brahmol | [48] |
| OH | OH | CH3 | CH2OH | OH | CH3 | CH3 | H | COO-glc(1-6)glc | 12-13 | Centellasaponin B | [34] |
| OH | OH | CH3 | CH2OH | OH | CH3 | CH3 | H | COO-glc(1-6) glc(1-4)rha | 12-13 | Brahminoside | [34,35] |
| Madecassoside | [51] | ||||||||||
| OH | OH | CH3 | CH3 | OH | CH3 | CH3 | H | COO-glc(1-6) glc(1-4)rha | 12-13 | Centellasaponin C | [34] |
| OH | O-L-Ara | CH3 | CH2OH | OH | CH3 | CH3 | H | COOH | 12-13 | Arabinoside | [50] |
| 3-O-[α-L-ara]-2α,3ß,6ß,23-tetrahydroxy-urs-12-en-28-oic acid | |||||||||||
| H | OH | H | CH2OH | OH | CH3 | CH3 | H | COOH | 12-13 | Isothankunic acid | [42] |
| 3α,5α,6ß,24-tetrahydroxy-urs-12-en-28-oic acid | |||||||||||
| H | OH | H | CH2OH | OH | CH3 | CH3 | H | COO-glc(1-6) glc(1-4)rha | 12-13 | Isothankuniside | [38] |
| OH | OH | CH3 | CH3 | OH | CH3 | CH3 | H | COOH | 12-13 | Madasiatic acid | [34] |
| Oleanane (C20, C20-dimethyl) | |||||||||||
| OH | OH | CH3 | CH2OH | H | H | CH3 | CH3 | COOH | 12-13 | 2α,3ß,23-trihydroxy-olean-12-en-28-oic acid | [35] |
| OH | OH | CH3 | CH2OH | OH | H | CH3 | CH3 | COOH | 12-13 | Terminolic acid | [3,40] |
| OH | -OH | CH3 | CH2OH | OH | H | CH3 | CH3 | COO-glc(1-6) glc(1-4)rha | 12-13 | Asiaticoside B | [3,50,34] |
| OH | -OH | CH3 | CH2OH | H | H | CH3 | CH3 | -COOH | 13-18 | Centellasapogenol A | [35] |
| 2α,3ß,23-trihydroxy-olean-13-en-28-oic acid) | |||||||||||
| OH | OH | CH3 | CH2OH | H | H | CH3 | CH3 | COO-glc(1-6) glc(1-4)rha | 13-18 | Centellasaponin A | [34,35] |
| Scheffoleoside A | |||||||||||
| H | OH | CH3 | CH2OH | OH | H | CH3 | CH3 | COOH | 12-13 | 3ß,6ß,23-trihydroxy-olean-12-en-28-oic acid | [34] |
| H | OH | CH3 | CH2OH | OH | H | CH3 | CH3 | COO-glc(1-6) glc(1-4)rha | 13-18 | Centellasaponin D | [34] |
Biological Activities of Centella Triterpenoid Saponins and Sapogenins
| Extract | Chemical composition | Applications |
|---|---|---|
| Asiatic acid | >95% Asiatic acid | Anti-ageing cosmetics, application after laser therapy, cosmeceutics |
| Titrated Extract of CentellaAsiatica (TECA) | 55-66% Genins | Anti-cellulite, slimming products, breast creams, stretch marks, scarred skin, anti-ageing cosmetics, moisturizing care |
| 34-44% Asiaticoside | ||
| TECA cosmetics | >40% Genins | Anti-cellulite, slimming products, breast creams, stretch marks, scarred skin, anti-ageing cosmetics, moisturizing care |
| > 36% Asiaticoside | ||
| Heteroside | >55% Madecassoside | Slow release effect, anti-ageing cosmetics, for moisturizing night-creams |
| >14% Asiaticoside | ||
| Asiaticoside | >95% Asiaticoside | Anti-inflammatory, against irritated and reddened skin, anti-allergic |
| Genins | >25% Asiatic acid | Natural antibiotic, antibacterial properties, for anti-acne products, intimate hygiene |
| >60% Madecassic acid |
Variation in Triterpene Production in C. asiatica Chemotypes
| Medical claim | Description of treatment | Ref. | ||||
|---|---|---|---|---|---|---|
| Skin ailments |
|
| ||||
| Circulation | Acts as a complementary treatment of ulcers of venous origin. |
| ||||
| Arthritis and rheumatism | Extracts are taken orally to relieve symptoms of venous and lymphatic vessel insufficiency. | |||||
| Memory enhancement, vitality and longitivity. | In India, for the past 30 000 years of Ayurvedic medicine, it has been used from wound healing, a mild diuretic, increasing concentration and alertness, and well as for the treatment of anxiety and stress. | |||||
| Cancer |
|
| ||||
| A general health tonic, an aphrodisiac and immune booster | Centella assists in destroying toxic accumulation in the brain as well as in the nerves, while it helps to clear the body from heavy metals as well as drugs – including recreational drugs. | [61] | ||||
| Respiratory ailments |
| |||||
| Detoxifying the body | Stimulates lipolysis and blood microcirculation and are thus used in the management of local adiposity or cellulite. | |||||
| Slimming | ||||||
| Diuretic | ||||||
| Treatment of liver and kidneys |
|
| ||||
| Sedative, Anti-stress, anti-anxiety and the treatment of depression |
| |||||
| Antifungal properties | ||||||
| Insect anti-feedantMosquito repellent |
| |||||
| Antibacterial activity |
| |||||
| Location / Source | Glycosides | Associated triterpene acids | ||
|---|---|---|---|---|
| Saponin | Sapogenin | Sugar | ||
|
|
| Glucose and rhamnose | |
|
|
| Glucose and rhammose |
|
|
|
| Glucose and rhamnose |
|
| ||||
|
| Glucose and rhamnose |
| |
| Glucose, rhamnose and arabinose | ||||
|
| Glucose and rhamnose |
| |
|
| Glucose and rhamnose |
| |
Manipulation of Centelloside Production in Cell and Tissue Culture
Conclusions
Acknowledgements
- Sample availability: Not available.
References
- Liu, M.; van Wyk, B.E.; Tilney, P.-M. A Taxonomic Evaluation of Fruit Structure in the Family Apiaceae.
- Brinkhaus, B.; Linder, M.; Schuppan, D.; Hahn, E.G. Chemical, pharmacological and clinical profile of the East African medicinal plant Centella asiatica. Phytomedicine 2000, 7, 427–448. [Google Scholar] [CrossRef]
- Schaneberg, B.T.; Mikell, J.R.; Bedir, E.; Khan, I.A. An improved HPLC method for quantitive determination of six triterpenes in Centella asiatica extracts and commercial products. Pharmazie 2003, 58, 381–384. [Google Scholar]
- Verma, R.K.; Bhartariya, K.G.; Gupta, M.M.; Kumar, S. Reverse-phase high performance liquid chromatography if asiaticoseide in Centella asiatica. Phytochem. Anal. 1999, 10, 191–193. [Google Scholar]
- Zheng, C.-J.; Qin, L.-P. Chemical components of Centella asiatica and their bioactivities. Chin. J. Integr. Med. 2007, 5, 348–351. [Google Scholar]
- Adamson, R.S. On some species of Centella. II. S. Afr. J. Bot. 1950, 15, 93–95. [Google Scholar]
- Bruneton, J. Pharmacognosy, Phytochemistry, Medicinal Plants, 2nd ed; Lavoisier Publishing: Paris, France, 1995. [Google Scholar]
- World Health Organisation, Herbae Centellae. In WHO Monographs on Selected Medicinal Plants; World Health Organisation: Geneva, Switzerland, 1999; Volume 1, pp. 77–85.
- Hanson, J.R. The biosynthesis of secondary metabolites. In Natural Products, the Secondary Metabolites; The Royal Society of Chemistry: Cambridge, UK, 2003; pp. 112–121. [Google Scholar]
- Gershenzon, J.; Kreis, W. Biosynthesis of monoterpenes, sesquiterpenes, diterpenes, sterols, cardiac glycosides and steroid saponins. In Biochemistry of Plant Secondary Metabolites. Annual Plant Reviews; Wink, M., Ed.; Sheffield Academic Press: Sheffield, UK, 1999; Volume 2, pp. 222–299. [Google Scholar]
- Devon, T.K.; Scott, A.I. Handbook of Natural Occurring Compounds; Academic Press: New York, NY, USA, 1972; Volume 2. [Google Scholar]
- Darnley-Gibbs, R. Chemotaxonomy of Flowering Plants. II; McGill-Queen's University Press: Montreal and London, UK, 1974. [Google Scholar]
- Bell, E.A.; Charlwood, B.V. Secondary plant products. In Encyclopedia of Plant Physiology; Springer Verlag: New York, NY, USA, 1980; p. 674. [Google Scholar]
- Ruzicka, L. The isoprene rule and the biogenesis of terpenic compounds. Experimentia 1953, 9, 357–367. [Google Scholar]
- Gershenzon, J.; Dudareva, N. The function of terpene natural products in the natural world. Nat. Chem. Biol. 2007, 3, 408–414. [Google Scholar] [CrossRef]
- Sparg, S.G.; Light, M.E.; van Staden, J. Biological activities and distribution of plant saponins. J. Ethnopharmacol. 2004, 94, 219–243. [Google Scholar] [CrossRef]
- Papadopoulou, K.; Melton, R.E.; Leggerr, M.; Daniels, M.J.; Osborurn, A.E. Compromised disease resistance in saponin-deficient plants. Proc. Natl. Acad. Sci. USA 1999, 96, 12923–12928. [Google Scholar]
- Liu, J.; Henkel, T. Traditional Chinese medicine (TCM): Are polyphenols and saponins the key ingredients triggering biological activities? Cur. Med. Chem. 2002, 9, 1483–1485. [Google Scholar]
- Phillips, D.R.; Rasberry, J.M.; Bartel, B.; Matsuda, S.P.T. Biosynthestic diversity in plant triterpene cyclization. Curr. Opin. Plant Biol. 2006, 9, 305–314. [Google Scholar] [CrossRef]
- Jenner, H.; Townsend, B.; Osbourn, A. Unravelling triterpene glycoside synthesis in plants: Phytochemistry and functional genomics join forces. Planta 2005, 220, 503–506. [Google Scholar] [CrossRef]
- Haralampidis, K.; Trojanowska, M.; Osbourn, A.E. Biosynthesis of triterpenoid saponins in plants. In Advances in Biochemical Engineering/Biotechnology; Scheper, T, Ed.; Springer Verlag: Berlin, Heidelberg, Germany, 2002; Volume 75, pp. 32–49. [Google Scholar]
- Collins, H.A. Secondary product formation in plant tissue cultures. Plant Growth Regul. 2001, 34, 119–134. [Google Scholar] [CrossRef]
- Benveniste, P. Biosynthesis and accumulation of sterols. Annu. Rev. Plant Biol. 2004, 55, 429–457. [Google Scholar]
- Kalinowska, M.; Zimowski, J.; Paczkowski, C.; Zdzislaw, A. The formation of sugar chains in triterpenoid saponins in glycoalkaloids. Phytochemistry Rev. 2005, 4, 237–257. [Google Scholar]
- Mangas, S.; Bonfill, M.; Osuna, L.; Moyano, E.; Tortoriello, J.; Cusido, R.M.; Pinol, M.T.; Palaźom, J. The effect of methyl jasmonate on triterpene and steriol metabolisms of Centella asiatica, Ruscus aculeatus and Galphimia glauca cultured plants. Phytochemistry 2006, 67, 2041–2049. [Google Scholar] [CrossRef]
- Zhang, H.; Shibuya, M.; Yokota, S.; Ebizuka, Y. Oxidosqualene cyclases from cell suspension cultures of Betula playphylla var. Japonica: Molecular evolution of oxidosqualene cyclases in higher plants. Biol. Pharm. Bull. 2003, 26, 642–650. [Google Scholar] [CrossRef]
- Morrissey, J.P.; Osbourn, A.E. Fungal resistance to plant antibiotics as a mechanism of pathogenesis. Microbiol. Mol. Biol. Rev. 1999, 63, 708–724. [Google Scholar]
- Achnine, L.; Huhman, D.V.; Farag, M.A.; Sumner, L.W.; Blount, J.W.; ixon, R.A. Genomics-based selection and functional characterization of triterpene glycosyltransferases from the model legume Medicago truncatula. Plant J. 2005, 41, 875–887. [Google Scholar] [CrossRef]
- Oyedeji, O.A.; Afolayan, A.J. Chemical composition and antibacterial activity of the essential oil of Centella asiatica growing in South Africa. Pharmaceut. Biol. 2005, 43, 249–252. [Google Scholar] [CrossRef]
- Kim, O.T.; Kim, M.Y.; Hong, M.H.; Ahn, J.C.; Oh, M.H.; Hwang, B. Production of triterpene glycosides from whole plant cultures of Centella asiatica (L.) Urban. Kor. J. Plant Biotechnol. 2002a, 29, 275–279. [Google Scholar]
- Aziz, Z.A.; Davey, M.R.; Power, J.B.; Anthony, P.; Smith, R.M.; Lowe, K.C. Production of asiaticoside and madecassocide in Centella asiatica in vitro and in vivo. Biol Plant. 2007, 51, 34–42. [Google Scholar]
- James, J.T.; Meyer, R.; Dubery, I.A. Characterisation of two phenotypes of Centella asiatica in Southern Africa through the composition of four triterpenoids in callus, cell suspensions and leaves. Plant Cell Tiss.Organ Cult. 2008, 94, 91–99. [Google Scholar] [CrossRef]
- Kim, O.T.; Kim, M.Y.; Huh, S.M.; Bai, D.G.; Ahn, J.C.; Hwang, B. Cloning of a cDNA probably encoding oxidosqualene cyclase associated with asiaticoside biosynthesis from Centella asiatica (L.) Urban. Plant Cell Rep. 2005, 24, 304–311. [Google Scholar] [CrossRef]
- Matsuda, H.; Morikawa, T.; Ueda, H.; Yokhikawa, M. Medicinal foodstuffs XXVII. Saponin constituents of Gotu Kola (2): Structures of new ursane- and oleanane-type triterpene oligoglycerides, Centella saponins B, C, and D, from Centella asiatica cultivated in Sri Lanka. Chem. Pharm. Bull. 2001a, 49, 1368–1371. [Google Scholar] [CrossRef]
- Matsuda, H.; Morikawa, T.; Ueda, H.; Yoshikawa, M. Medicinal foodstuffs. XXVI. Inhibitors of aldose reductase and new triterpene and its oligoglycoside, Centellasapogenol A and Centellasaponin A, from Centella asiatica (Gotu kola). Hetrocycles 2001b, 55, 1499–1504. [Google Scholar] [CrossRef]
- Labadie, R.P.; De Silva, K.T.D. Centella asiatica (L.) Urban in perspective: An evaluative account. In Studies in Indian Medicinal History; Wujastyk, D., Zysk, K.G., Eds.; Motilal Banarsidass: New Delhi, India, 2001; Volume V, pp. 191–205. [Google Scholar]
- Rastogi, R.P.; Dhar, M.L. Chemical examination of Centella asiatica Linn. II. Brahmoside and brahminoside. Indian J. Chem. 1963, 1, 267–269. [Google Scholar]
- Dutta, T.; Basu, U.P. Triterpenoids: Part I. Thankuniside and Thankunic acid – a new triterpene glycoside and acid from Centella asiatica Linn. (Urb.). J. Sci. Ind. Res. 1962, B12, 239. [Google Scholar]
- Bhattacharyya, S.C. Constituents of Centella asiatica Part II. Structure of the terpene acids. J. Indian Chem. Soc. 1956, 33, 630–634. [Google Scholar]
- Jian, P.; Guiqing, K.; Chuanxun, Y.; Beibei, Z.; Risheng, J.; Yuan, Y. Separation and determination of madecassic acid extracts of Centella asiatica using High Performance Liquid Chromatography with β-cyclodextrin as mobile phase additive. Chin. J. Chromat. 2007, 25, 316–318. [Google Scholar] [CrossRef]
- Rao, P.S.; Seshadri, T.R. Variation in the chemical composition of Indian samples of Centella asiatica. Curr. Sci. 1970, 38, 77–79. [Google Scholar]
- Dutta, T.; Basu, U.P. Isothankunic acid–A new triterpene acid from Centella asiatica (URB). Bull. Nat. Inst. Sci. India 1968, 37, 178–184. [Google Scholar]
- Hegnauer, R. Chemotaxinomie der Pflanzen. VI; Birkhaűser Verlag: Basel, Switzerland, 1966; p. 551. [Google Scholar]
- Castellani, C.; Marai, A.; Vacchi, P. The Centella asiatica. Bull. Chim. Farm. 1981, 120, 570–605. [Google Scholar]
- Yu, Q.-L.; Duan, H.-Q.; Takaishi, Y.; Gao, W.-Y. A novel triterpene from Centella asiatica. Molecules 2006, 11, 661–665. [Google Scholar]
- Yu, Q.L.; Duan, H.Q.; Gao, W.Y.; Takaishi, Y. A new triterpene and a saponin from Centella asiatica. Chin. Chem. Lett. 2007, 18, 62–64. [Google Scholar]
- Jiang, Z.Y.; Zhang, X.M.; Zhou, J. New triterpenoid glycosides from Centella asiatica. Helv.Chim Acta. 2005, 88, 297–204. [Google Scholar] [CrossRef]
- Singh, B.; Rastogi, R.P. A reinvestigation of the triterpenes of Centella asiatica. Phytochemistry 1969, 8, 917–921. [Google Scholar] [CrossRef]
- Polonsky, J.; Sach, E.; Lederer, E. Sur la Constitution Chimique de la partie glucidique de l’asiatocid. Bull. Soc. Chim. Fr. 1959, 880–887. [Google Scholar]
- Shukla, Y.N.; Srivastava, R.; Tripathi, A.K.; Prajapati, V. Characterization of an ursane triterpenoid from Centella asiatica with growth inhibitory activity against Spirarctia bliqua. Pharm. Biol. 2000, 38, 262–267. [Google Scholar]
- Pinhas, H.; Bondiou, J.C. Sur la constitution chimique de la partie glucidique du madecassoside. Bull. Soc. Chim. Fr. 1967, 6, 1888–1890. [Google Scholar]
- Yoshinori, A.; Reiko, M.; Tsunematsu, T. Mono- and sesquiterpenoids from Hydrocotyle and Centella species. Phytochemistry 1982, 21, 2590–2592. [Google Scholar] [CrossRef]
- Rajkumar, S.; Jebanesan, A. Repellent activity of selected plant essential oils against the malarial fever mosquito Anopheles stephensi. Trop. Biomed. 2007, 24, 71–75. [Google Scholar]
- Kartnig, T. Clinical applications of Centella asiatica (L.) Urban. In Herbs, Spices, and Medicinal Plants: Recent Advances in Botany, Horticulture, and Pharmacology; Cracker, L.E., Simon, J.E., Eds.; Oryx Press: Phoenix, AZ, USA, 1988; Volume 3, pp. 145–173. [Google Scholar]
- Pointel, J.P.; Boccalon, M.D.; Cloarec, M.; Ledevehat, M.D.; Joubert, M. Titrated extract of Centella asiaitica (TECA) in the treatment of venous insufficiency of the lower limbs. Angiology 1987, 38, 46–50. [Google Scholar] [CrossRef]
- Montecchio, G.P.; Samaden, A.; Carbone, S.; Vigotti, M.; Siragusa, S.; Piovella, F. Centella asiatica triterpenic fraction (CATTF) reduces the number of circulating endothelial cells in subjects with post phlebitis syndrome. Haematology 1991, 76, 256–259. [Google Scholar]
- Advanced Cosmeceutical Technology. ACT’s Power Products list. 2006. Available online: http://www.in-cosmetics.com/ExhibitorLibrary/383/ACT_brochure.pdf accessed on 6 November 2008.
- Abou-Chaar, C.I. New drugs from higher plants recently introduced into therapeutics. Lebanese Pharm. 1963, 8, 15–37. [Google Scholar]
- Boely, C. Indications of titrated extract of Centella asiaticia in phlebology. Gaz. Med. Fr. 1975, 82, 741–744. [Google Scholar]
- Wolfram, Von St. Erfahrungen mit Maddecasol bei der behandling ulzererõserser Hautverãnderungen. Wien. Med. Wschr. 1965, 115, 439–442. [Google Scholar]
- Bonfill, M.; Mangas, S.; Cusido, R.M.; Osuna, L.; Pinol, M.T.; Palazon, J. Identification of triterpenoid compounds of Centella asiatica by thin–layer chromatography and mass spectrometry. Biomed. Chromatogr. 2005, 20, 151–153. [Google Scholar]
- Ayyanar, M.; Ignacimuthu, S. Medicinal uses and pharmalogical actions of five commonly used Indian medicinal plants: a mini-review. Iran. J. Pharmacol. Therap. 2008, 7, 104–114. [Google Scholar]
- Mathur, S.; Verma, R.K.; Gupta, M.M.; Ram, M.; Sharma, S.; Kumar, S. Screening of genetic resources of the medicinal-vegetable plant Centella asiatica for herb and asiaticoside yield under shaded and full sunlight. J. Hortic. Sci. Biotechnol. 2000, 755, 551–554. [Google Scholar]
- Sampson, J.H.; Raman, A.; Karlsen, G.; Navsaria, H.; Leigh, I.M. In vitro keratinocyte antiproliferant effect of Centella asiatica extract and triterpenoid saponins. Phytomedicine 2001, 8, 230–235. [Google Scholar] [CrossRef]
- Babu, T.D.; Kuttan, G.; Padikkala, J. Cytotoxic and anti-tumor properties of certain taxa of Umbelliferae with special reference to Centella asiatica (L.) Urban. J. Enthopharmacol. 1995, 48, 53–57. [Google Scholar] [CrossRef]
- Cristoni, A, Di Pierro. Management of local adiposity with botanicals. Chimica Oggi/Chemistry Today 1998, 16, 11–14. [Google Scholar]
- Sharma, D.N.K.; Khosa, R.L.; Chansauria, J.P.N.; Sahai, M. Antistress activity of Itinospora cordifolia and Centella asiatica. Phytother. Res. 1996, 10, 181–183. [Google Scholar] [CrossRef]
- Lucia, R.D.; Sertie, J.A.A.; Camargo, E.A.; Panizza, S. Phamacological and toxicological studies on Centella asiatica. Fitotherapia 1997, LXVIII, 413–416. [Google Scholar]
- Ponglux, D.; Wong, S.; Phadungcharoen, T.; Ruangrungsri, N.; Likhitwitayawuid, K. In Medicinal Plants; International Congress on National Products: Bangkok, Thailand, 1987; pp. 83–85. [Google Scholar]
- Jatisatienr, C.; Tragoolpua, K. Effect of the extract from eight species of medicinal plants on growth of selected plant pathogenic molds and dermatophyte. In International Symposium on Breeding Research on Medicinal and Aromatic Plants, Quedlingberg, Germany, June 30–July 4, 1996.
- Srivastava, R.; Shukla, Y.N.; Tripathi, A.K. Antifeedant compounds from Centella asiatica. Fitotherapia 68, 93–94.
- Newell, C.A.; Linda, A. Herbal Medicines: A Guide for Health Care Professionals, 6th ed; The Pharmacautical Press: London, UK, 1996; pp. 224–239. [Google Scholar]
- Srivastava, R.; Shukla, Y.N.; Darokar, M.P. Antibacterial activity of Centella asiatica. Fitotherapia 1997, 68, 466–467. [Google Scholar]
- Ullah, M.O.; Sultana, S.; Haque, A.; Tasmin, S. Antimicrobial, cytotoxic and antioxidant activity of Centella asiatica. Eur. J. Sci. Res. 2009, 30, 260–264. [Google Scholar]
- Pentacyclic triterpenes. US Patent 6303589, 2002.
- Das, A.; Mallick, R. Correlation between genomic diversity and asiaticoside content in Centella asiatica (L.) Urban. Bot. Bull. Acad. Sin. 1991, 32, 1–8. [Google Scholar]
- Kim, O.T.; Kim, M.Y.; Hong, M.H.; Ahn, J.C.; Hwang, B. Stimulation of asiaticoside accumulation in the whole plant cultures of Centella asiatica (L.) Urban by elicitors. Plant Cell Rep. 2004, 23, 339–344. [Google Scholar] [CrossRef]
- Kim, O.T.; Kyong Hwan, B.; Shin, Y.-S.; Lee, M.-J.; Jung, S.-J.; Hyun, D.-Y.; Kim, Y.-C.; Seong, N.-S.; Cha, S.-W.; Hwang, B. Enhanced production of asiaticoside from hairy root cultures of Centella asiatica (L.) Urban elicited by methyl jasmonate. Plant Cell Rep. 2007, 26, 1941–1949. [Google Scholar]
- Inamdar, P.K.; Yeole, R.D.; Ghogare, A.B.; de Souza, N.J. Determination of biologically active constituents in Centella asiatica. J. Chromatogr. 1996, 742, 127–130. [Google Scholar]
- Monti, D.; Candido, A.; Manuel Cruz Silva, M.; Křen, V.; Riva, S.; Danieli, B. Biocatalyzed generation of molecular diversity: selective modification of the saponin asiaticoside. Adv. Synth. Catal. 2005, 347, 1168–1174. [Google Scholar] [CrossRef]
- Gupta, A.P.; Gupta, M.M.; Kumar, S. High performance thin layer chromatography of asiaticoside in Centella asiatica. J. Ind. Chem. Soc. 1999, 76, 321–322. [Google Scholar]
- Rouillard-Guellec, F.; Robin, J.R.; Ratsimamanga, A.R.; Ratsimamanga, S.; Rasaoanaivo, R. Comparative study of Centella asiatica of Madagascar origin and Indian origin. Acta Bot. Gall. 1997, 144, 489–493. [Google Scholar]
- Randriamampionona, D.; Diallo, B.; Rakotoniriana, F.; Rabemanantsoa, C.; Cheuk, K.; Corbisier, A.-M.; Mahillion, J.; Ratsimamanga, S.; Jaziri, M.E.J. Comparative analysis of active constituents in Centella asiatica samples from Madagascar: Application for ex situ conservation and clonal propagation. Fitoterapia 2007, 78, 482–489. [Google Scholar] [CrossRef]
- Pannizi, L.; Flamini, G.; Cironi, P.L.; Morelli, I. Composition and antimicrobial properties of essential oils of four Mediterranean Laminaceae. J. Ethnopharmacol. 1993, 39, 167–170. [Google Scholar]
- Giri, A.; Naraseu, M.L. Transgenic hairy roots: Recent trends and applications. Biotechnol. Adv. 2000, 18, 1–22. [Google Scholar]
- Gaines, J.L. Increasing alkaloid production from Catharanthus roseus suspensions through methyl jasmonate elicitation. Pharm. Eng. 2004, 24, 1–6. [Google Scholar]
- Kim, Y.; Wyslouzil, B.E.; Weathers, P.J. Secondary metabolism of hairy root cultures in bioreactors. In Vitro Cell. Dev. Biol. Plant. 38, 1–10.
- Samanani, N.; Facchini, P.J. Compartmentalization of plant secondary metabolism. In Recent Advances in Phytochemistry; Romeo, J.T., Ed.; Elsevier Ltd.: Amsterdam, the Netherland, 2006; Volume 40, p. 54. [Google Scholar]
- Bouhouche, N.; Solet, J.M.; Simon Ramiasa, A.; Bonal, J.; Cosson, L. Conversion of 3-dimethylthiocolchicine into thiocolchicoside by Centella asiatica suspension cultures. Phytochemistry 1998, 47, 743–747. [Google Scholar]
- Van der Heijden, R.; Verheij, E.R.; Schripsema, J.; Baerheim-Svebdsen, A.; Verpoorte, R.; Harkes, P.A.A. Induction of triterpene biosynthesis by elicitors in suspension cultures of Tabernamontana species. Plant Cell Rep. 1988, 7, 51–54. [Google Scholar] [CrossRef]
- Hayashi, H.; Huang, P.; Inoue, K. Up-regulation of soyasaponin biosynthesis by methyl jasmonate in cultured cells of Glycyrrhiza glabra. Plant Cell Physiol. 2003, 44, 404–411. [Google Scholar] [CrossRef]
- Roberts, S.C. Production and engineering of terpenoids in plant cell culture. Nat. Chem. Bio. 2007, 3, 387–395. [Google Scholar] [CrossRef]
- Degenhardt, J; Gershenzon, J.; Baldwin, I.T.; Kessler, A. Attracting friend to feast on foes: Engineering terpene emission to make crop plants more attractive to herbivore enemies. Curr. Opin. Biotechnol. 2003, 14, 169–176. [Google Scholar]
- Mayano, E.; Monfill, M.; Mangas, S.; Cusido, R.M.; Pinol, M.T.; Palazon, J. Centellosides production and expression level of genes encoding their synthesis in Centella asiatica in vitro cultures. J. Biotechnol. 2007, 131S, S43–S47. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
James, J.T.; Dubery, I.A. Pentacyclic Triterpenoids from the Medicinal Herb, Centella asiatica (L.) Urban. Molecules 2009, 14, 3922-3941. https://doi.org/10.3390/molecules14103922
James JT, Dubery IA. Pentacyclic Triterpenoids from the Medicinal Herb, Centella asiatica (L.) Urban. Molecules. 2009; 14(10):3922-3941. https://doi.org/10.3390/molecules14103922
Chicago/Turabian StyleJames, Jacinda T., and Ian A. Dubery. 2009. "Pentacyclic Triterpenoids from the Medicinal Herb, Centella asiatica (L.) Urban" Molecules 14, no. 10: 3922-3941. https://doi.org/10.3390/molecules14103922
APA StyleJames, J. T., & Dubery, I. A. (2009). Pentacyclic Triterpenoids from the Medicinal Herb, Centella asiatica (L.) Urban. Molecules, 14(10), 3922-3941. https://doi.org/10.3390/molecules14103922






