First Synthesis and Isolation of the E- and Z-Isomers of Some New Schiff Bases. Reactions of 6-Azido-5-Formyl-2-Pyridone with Aromatic Amines
Abstract
:Introduction
Results and Discussion
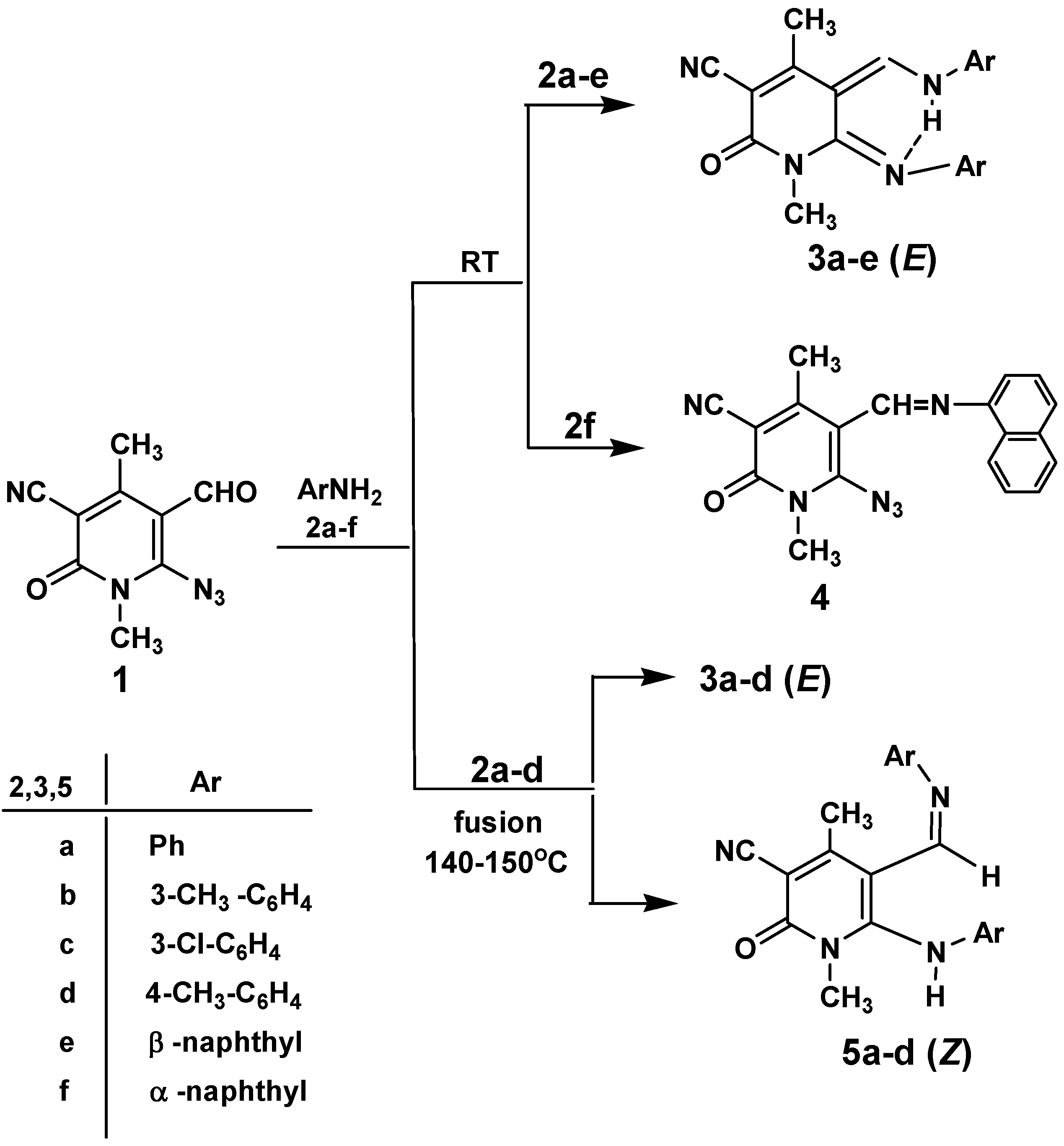
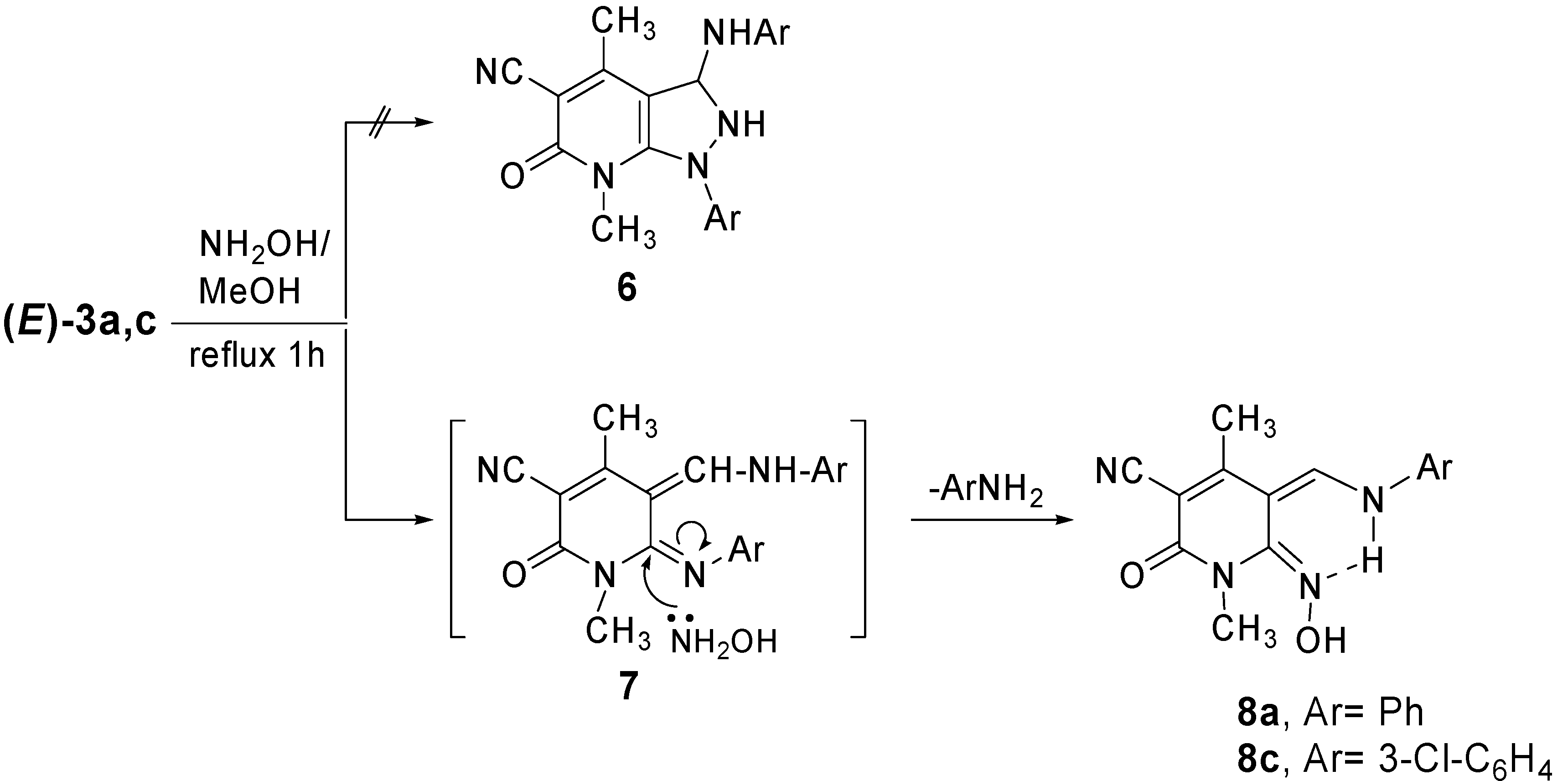
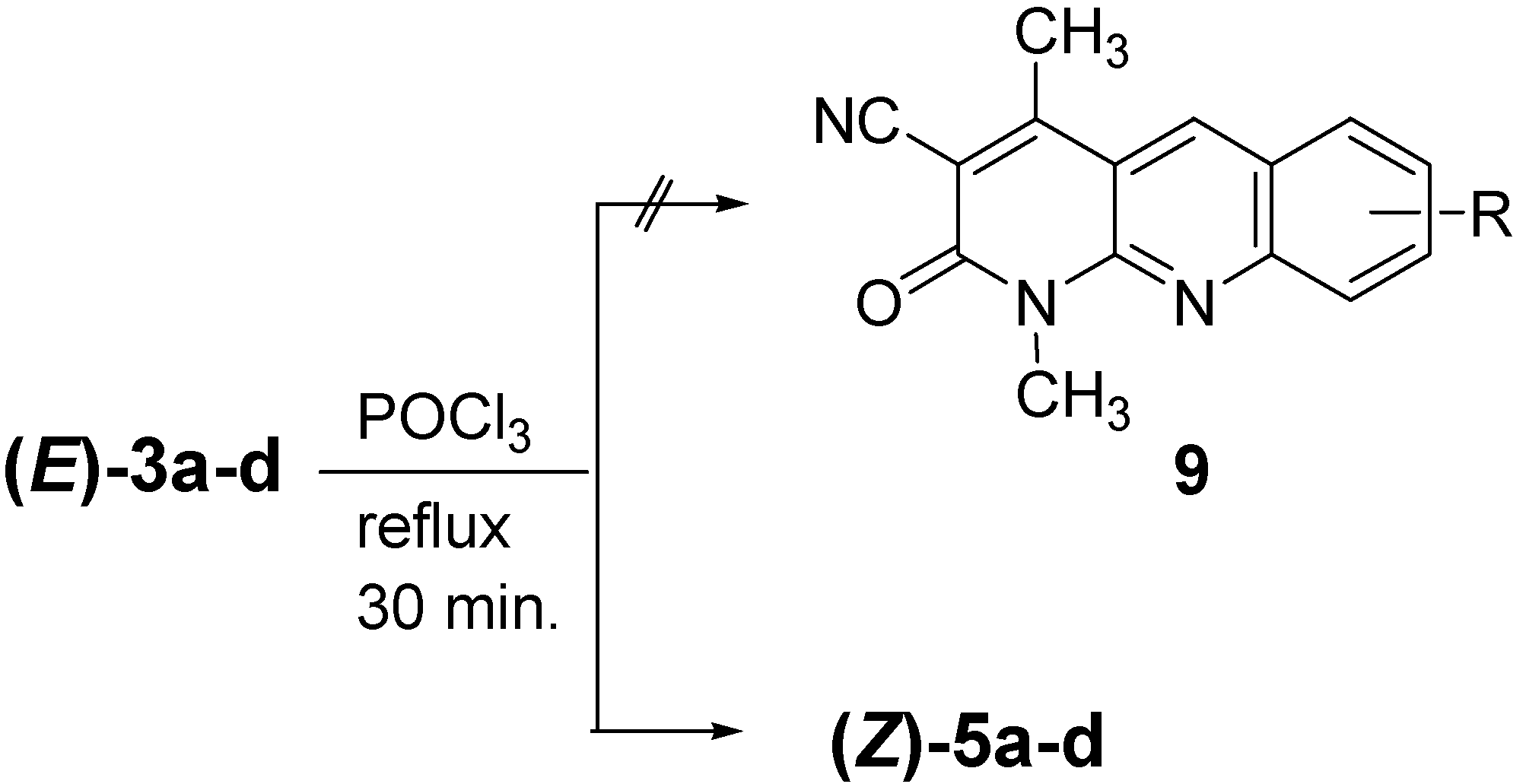
Conclusions
Experimental
General
References
- Karia, F. D.; Parsania, P. H. Synthesis, biological and thermal properties of Schiff bases of bisphenol-C. Asian J. Chem. 1999, 11, 991–995. [Google Scholar]
- Samadhiya, S.; Halve, A. Synthetic utility of Schiff Bases as potential herbicidal agents. Orient. J. Chem. 2001, 17, 119–122. [Google Scholar]
- Singh, W. M.; Dash, B. C. Synthesis of some new Schiff bases containing thiazole and oxazole nuclei and their fungicidal activity. Pesticides 1988, 22, 33–37. [Google Scholar]
- Pandeya, S. N.; Sriram, D.; Nath, G.; De Clercq, E. Synthesis and antimicrobial activity of Schiff and Mannich bases of isatin and its derivatives with pyrimidine. Farmaco 1999, 54, 624–628. [Google Scholar] [CrossRef]
- Baseer, M. A.; Jadhav, V. D.; Phule, R. M.; Archana, Y. V.; Vibhute, Y. B. Synthesis and antimicrobial activity of some new Schiff bases. Orient. J. Chem. 2000, 16, 553–556. [Google Scholar]
- El-Masry, A. H.; Fahmy, H. H.; Abdelwahed, S. H. A. Synthesis and antimicrobial activity of some new benzimidazole derivatives. Molecules 2000, 5, 1429–1438. [Google Scholar] [CrossRef]
- More, P. G.; Bhalvankar, R. B.; Pattar, S. C. Synthesis and biological activities of Schiff bases of aminothiazoles. J. Indian Chem. Soc. 2001, 78, 474–475. [Google Scholar]
- Pathak, P.; Jolly, V. S.; Sharma, K. P. Synthesis and biological activities of some new substituted arylazo Schiff bases. Orient. J. Chem. 2000, 16, 161–162. [Google Scholar]
- Desai, S. B.; Desai, P. B.; Desai, K. R. Synthesis of some Schiff bases, thiazollidones, and azetidinones derived from 2,6-diaminobenzo[1,2-d:4,5-d,]bisthiazole and their anticancer activities. Heterocycl. Commun. 2001, 7, 83–90. [Google Scholar]
- Aydogan, F.; Öcal, N.; Turgut, Z.; Yolacan, C. Transformations of aldimines derived from pyrrole-2-carboxaldehyde. Synthesis of thiazolidino-fused compounds. Bull. Kor. Chem. Soc. 2001, 22, 476–480. [Google Scholar]
- Cozzi, P. G. Metal-Salen Schiff base complexes in catalysis: practical aspects. Chem. Soc. Rev. 2004, 33, 410–421. [Google Scholar] [CrossRef]
- Mekheimer, R. A. A new approach to the synthesis of polyfunctionally substituted 1,8-naphthyridin-2-one derivatives from 6-azidopyridones. A novel thermal decomposition to 6-aminopyridones. Synthesis 2001, 103–107. [Google Scholar] [CrossRef]
- Mekheimer, R. Synthesis of aminopyridines from azidopyridines and tetrazolo[1,5-a]- pyridines. Pharmazie 1994, 49, 322–324. [Google Scholar]
- Elgemeie, H. G.; El-Fahham, H. A.; Mekheimer, R. Novel synthesis of condensed pyridin-2(1H)-thione derivatives from arylidenecyanothioacetamides and cycloalkanones. Sulfur Lett. 1988, 8, 187–198. [Google Scholar]
- Mekheimer, R.; Mohamed, H. N.; Sadek, K. U. Synthesis of functionalized 4H-pyrano[3,2-c]pyridines from 4-hydroxy-6-methyl-2-pyridone and their reactions. Unexpected new routes to 3,3\-benzylidene bis[4-hydroxy-6-methyl-2(1H)-3-pyridinone]s. Bull. Chem. Soc. Jpn. 1997, 70, 1625–1630. [Google Scholar] [CrossRef]
- Mekheimer, R.; Shaker, R.; Sadek, K. U.; Otto, H. H. A novel synthesis of benzo[g]- imidazo[1,2-a]pyridines: The reactivity of arylidine-1H-benzimidazole-2-acetonitrile with electron poor olefines and dimethylacetylene dicarboxylate under microwave irradiation. Heterocycl. Commun. 1997, 3, 217–221. [Google Scholar]
- Van Tinh, D.; Fischer, M.; Stadlbauer, W. Ring closure reactions of cyclic 2-arylamino-methylene-1,3-diones. J. Heterocyclic Chem. 1996, 33, 905–910. [Google Scholar] [CrossRef]
- Prasad, D.; Prasad, N.; Prasad, R. M.; Ferrier, R. J.; Milgate, S. M. 13C-NMR Examination of ethyl cyano(arylhydrazono)acetates. J. Chem. Soc. Perkin Trans. 1 1984, 1397–1399. [Google Scholar]
- Täuble, A. E.; Langhans, K.; Kappe, T.; Stadlbauer, W. Thermolytic ring closure of 4-azido-3-phenysulfanyl-and-4-azido-3-phenylsulfonyl-2-quinolones to 12H-quinolino[3,4-b][1,4]benzo- thiazin-6(5H)-ones (Organic azides in heterocyclic synthesis, part 31). J. Heterocyclic Chem. 2002, 39, 1259–1264. [Google Scholar] [CrossRef]
- Sample availability: Contact the authors.
© 2008 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Mekheimer, R.A.; Hameed, A.M.A.; Sadek, K.U. First Synthesis and Isolation of the E- and Z-Isomers of Some New Schiff Bases. Reactions of 6-Azido-5-Formyl-2-Pyridone with Aromatic Amines. Molecules 2008, 13, 195-203. https://doi.org/10.3390/molecules13010195
Mekheimer RA, Hameed AMA, Sadek KU. First Synthesis and Isolation of the E- and Z-Isomers of Some New Schiff Bases. Reactions of 6-Azido-5-Formyl-2-Pyridone with Aromatic Amines. Molecules. 2008; 13(1):195-203. https://doi.org/10.3390/molecules13010195
Chicago/Turabian StyleMekheimer, Ramadan A., Afaf M. Abdel Hameed, and Kamal U. Sadek. 2008. "First Synthesis and Isolation of the E- and Z-Isomers of Some New Schiff Bases. Reactions of 6-Azido-5-Formyl-2-Pyridone with Aromatic Amines" Molecules 13, no. 1: 195-203. https://doi.org/10.3390/molecules13010195
APA StyleMekheimer, R. A., Hameed, A. M. A., & Sadek, K. U. (2008). First Synthesis and Isolation of the E- and Z-Isomers of Some New Schiff Bases. Reactions of 6-Azido-5-Formyl-2-Pyridone with Aromatic Amines. Molecules, 13(1), 195-203. https://doi.org/10.3390/molecules13010195



