The Development of Design and Manufacture Techniques for Bioresorbable Coronary Artery Stents
Abstract
:1. Introduction
2. Stent Material and Design
2.1. Stent Design
2.2. Stent Material
3. Mechanical and Degradation Performance
3.1. Mechanical Performance
3.2. Degradation Properties
3.3. Clinical Trial
4. Biocompatibility
4.1. Surface Modification
4.2. Drug Coating
5. Stent Manufacture Techniques
6. Conclusions and Future Work
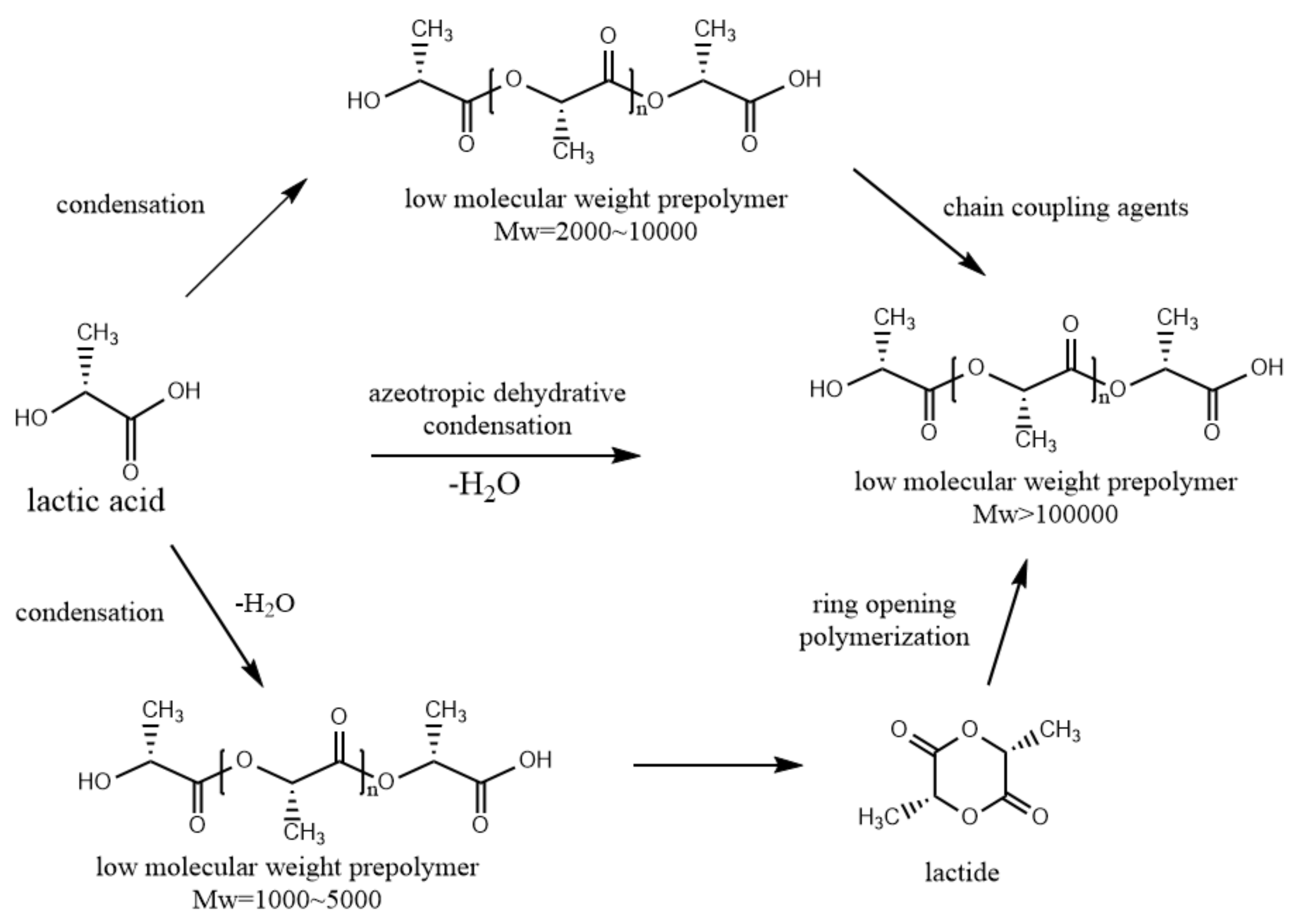
- The development of biodegradable materials plays an important role in the development of biodegradable scaffolds. Compared with metallic scaffolds, biodegradable scaffolds still have many deficiencies in radial strength and other mechanical properties that need to be continuously improved upon and developed in the future. Good mechanical properties can prolong the service life of a stent and provide strong support at the lesion and injury site, which is conducive to the recovery of patients. The material processing method can significantly influence the mechanical properties of the scaffold. Exploring new stent processing methods has become a hot research direction;
- The degradability of bioresorbable scaffolds is also a key property characteristic. The assessment of degradation performance is generally divided into in vivo and in vitro degradation experiments. In vitro degradation experiments are usually conducted in pH- and temperature-specific solutions such as tetrahydrofuran solution with a pH of 7 at 37 °C. In vivo degradation experiments can also be divided into animal experiments and human experiments. Animal studies have been carried out in rabbits, mice, pigs and sheep to assess whether stents cause severe inflammation and cellular problems. Similarly, stents can be implanted in humans to evaluate their six-months, one-year or long-term performance after implantation.
- Excellent biocompatibility plays a vital role in the development of medical implants. Stents come into direct contact with the cardiovascular and blood vessels after implantation, which is a major cause of clinical complications. Surface modification and drug coating of scaffolds should be proposed to improve stent biocompatibility. The surface texture of the scaffold can be modified using physical and chemical methods to intuitively reduce the contact between the scaffold and the blood vessels. Stent drug loading can reduce complications and control the degradation rate of stents through drug release, which is also the direction of future development.
- Additive manufacturing has been a hot topic in the medical field in recent years. Many studies have shown that 3D printing may be an alternative scaffold fabrication method through developing intelligent polymer materials. Shape memory materials are also a new development direction. The development of biodegradable stents with shape memory performance can simplify the complex process of stent implantation and provide more convenient services for doctors and patients.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Iqbal, J.; Onuma, Y.; Ormiston, J.; Abizaid, A.; Waksman, R.; Serruys, P. Bioresorbable scaffolds: Rationale, current status, challenges, and future. Eur. Heart. J. 2014, 35, 765–776. [Google Scholar] [CrossRef] [Green Version]
- Dyet, J.F.; Watts, W.G.; Ettles, D.F.; Nicholson, A.A. Mechanical properties of metallic stents: How do these properties influence the choice of stent for specific lesions. Cardiovasc. Inter. Rad. 2000, 23, 47–54. [Google Scholar] [CrossRef]
- Soares, J.S.; Moore, J.E., Jr. Biomechanical Challenges to Polymeric Biodegradable Stents. Ann. Biomed. Eng. 2016, 44, 560–579. [Google Scholar] [CrossRef]
- Onuki, Y.; Bhardwaj, U.; Papadimitrakopoulos, F.; Burgess, D.J. A review of the biocompatibility of implantable devices: Current challenges to overcome foreign body response. J. Dia. Sci. Tech. 2008, 2, 1003–1015. [Google Scholar] [CrossRef]
- Park, S.A.; Lee, S.J.; Lim, K.S.; Bae, I.H.; Lee, J.H.; Kim, W.D.; Jeong, M.H.; Park, J.-K. In vivo evaluation and characterization of a bio-absorbable drug-coated stent fabricated using a 3D-printing system. Mater. Lett. 2015, 141, 355–358. [Google Scholar] [CrossRef]
- Senatov, F.S.; Niaza, K.V.; Zadorozhnyy, M.Y.; Maksimkin, A.V.; Kaloshkin, S.D.; Estrin, Y.Z. Mechanical properties and shape memory effect of 3D-printed PLA-based porous scaffolds. J. Mech. Behav. Biomed. 2016, 57, 139–148. [Google Scholar] [CrossRef]
- Zarek, M.; Mansour, N.; Shapira, S.; Cohn, D. 4D Printing of Shape Memory-Based Personalized Endoluminal Medical Devices. Macromol. Rapid. Comm. 2017, 38, 1600628–1600634. [Google Scholar] [CrossRef]
- McMahon, S.; Bertollo, N.; Cearbhaill, E.D.O.; Salber, J.; Pierucci, L.; Duffy, P.; Dürig, T.; Bi, V.; Wang, W. Bio-resorbable polymer stents: A review of material progress and prospects. Prog. Polym. Sci. 2018, 83, 79–96. [Google Scholar] [CrossRef]
- Capodanno, D. Bioresorbable Scaffolds in Coronary Intervention: Unmet Needs and Evolution. Korean Circ. J. 2018, 48, 24–35. [Google Scholar] [CrossRef] [Green Version]
- Mattesini, A.; Bartolini, S.; Sorini Dini, C.; Valente, S.; Parodi, G.; Stolcova, M.; Meucci, F.; Di Mario, C. The DESolve novolimus bioresorbable Scaffold: From bench to bedside. J. Thorac. Dis. 2017, 9, S950–S958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramcharitar, S.; Serruys, P.W. Fully Biodegradable Coronary Stents Progress to Date. Am. J. Cardiovasc. Drugs 2008, 8, 305–314. [Google Scholar] [CrossRef]
- Abizaid, A.; Carrie, D.; Frey, N.; Lutz, M.; Weber-Albers, J.; Dudek, D.; Chevalier, B.; Weng, S.C.; Costa, R.A.; Anderson, J.; et al. 6-Month Clinical and Angiographic Outcomes of a Novel Radiopaque Sirolimus-Eluting Bioresorbable Vascular Scaffold: The FANTOM II Study. JACC Cardiovasc. Interv. 2017, 10, 1832–1838. [Google Scholar] [CrossRef] [PubMed]
- Nishio, S.; Kosuga, K.; Igaki, K.; Okada, M.; Kyo, E.; Tsuji, T.; Takeuchi, E.; Inuzuka, Y.; Takeda, S.; Hata, T.; et al. Long-Term (>10 Years) Clinical Outcomes of First-in-Human Biodegradable Poly-l-Lactic Acid Coronary Stents Igaki-Tamai Stents. Circulation 2012, 125, 2343–2352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamai, H.; Igaki, K.; Kyo, E.; Kosuga, K.; Kawashima, A.; Matsui, S.; Komori, H.; Tsuji, T.; Motohara, S.; Uehata, H. Initial and 6-month results of biodegradable poly-l-lactic acid coronary stents in humans. Circulation 2000, 102, 399–404. [Google Scholar] [CrossRef] [Green Version]
- Cerrato, E.; Barbero, U.; Gil Romero, J.A.; Quadri, G.; Mejia-Renteria, H.; Tomassini, F.; Ferrari, F.; Varbella, F.; Gonzalo, N.; Escaned, J. Magmaris™ resorbable magnesium scaffold:state-of-art review. Future Cardiol. 2019, 15, 267–279. [Google Scholar] [CrossRef] [Green Version]
- Eling, B.; Gogolewski, S.; Pennings, A.J. Biodegadable materials of poly(l-lactic acid): 1. Melt-spun and solution spun fibers. Polymer 1982, 23, 1587–1593. [Google Scholar] [CrossRef]
- Lunt, J.; Shafer, A.L. Polylactic acid polymers from corn: Applications in the textiles industry. J. Ind. Text. 2000, 29, 191–205. [Google Scholar] [CrossRef]
- Lunt, J. Large scale production, properties and commercial applications of polylactic acid polymers. Polym. Deg. Stab. 1998, 59, 145–152. [Google Scholar] [CrossRef]
- Garlotta, D. A Literature Review of Poly (Lactic Acid). J. Polym. Environ. 2001, 9, 63–84. [Google Scholar] [CrossRef]
- Sabino, M.A.; Albuerne, J.; Muller, A.J.; Brisson, J. Influence of in vitro hydrolytic degradation on the morphology and crystallization behavior of poly(pdioxanone). Biomacromolecules 2004, 2, 358–370. [Google Scholar] [CrossRef]
- Bartkowiak-Jowsa, M.; Będziński, R.; Kozłowska, A.; Filipiak, J.; Pezowicz, C. Mechanical, rheological, fatigue, and degradation behavior of PLLA, PGLA and PDGLA as materials for vascular implants. Meccanica 2012, 48, 721–731. [Google Scholar] [CrossRef] [Green Version]
- Hashima, K.S.N.; Inoue, T. Structure-properties of super-tough PLA alloy with excellent heat resistance. Polymer 2010, 17, 3934–3939. [Google Scholar] [CrossRef]
- El Fray, M.; Altstädt, V. Fatigue behaviour of multiblock thermoplastic elastomers. 2. Dynamic creep of poly(aliphatic/aromatic-ester) copolymers. Polymer 2013, 16, 4643–4650. [Google Scholar] [CrossRef]
- Hu, T.; Yang, J.; Cui, K.; Rao, Q.; Yin, T.; Tan, L.; Zhang, Y.; Li, Z.; Wang, G. Controlled Slow-Release Drug-Eluting Stents for the Prevention of Coronary Restenosis: Recent Progress and Future Prospects. ACS Appl. Mater. Inter. 2015, 7, 11695–11712. [Google Scholar] [CrossRef] [PubMed]
- Kwon, D.; Kim, J.; Kim, D.; Kang, H.; Lee, B.; Lee, K.; Kim, M. Biodegradable stent. J. Biomed. Sci. Eng. 2012, 5, 208–216. [Google Scholar] [CrossRef] [Green Version]
- Nair, L.S.; Laurencin, C.T. Biodegradable polymers as biomaterials. Prog. Polym. Sci. 2007, 32, 762–798. [Google Scholar] [CrossRef]
- Vroman, I.; Tighzert, L. Biodegradable Polymers. Materials 2009, 2, 307–344. [Google Scholar] [CrossRef]
- Maurus, P.B.; Kaeding, C.C. Bioabsorbable implant material review. Oper. Techn. Sport. Med. 2004, 12, 158–160. [Google Scholar] [CrossRef]
- Wypych, G. Handbook of Polymers, 2nd ed.; ChemTec Publishing: Toronto, Canada, 2016; pp. 419–421. [Google Scholar]
- Terasaka, S.; Iwasaki, Y.; Shinya, N.; Uchida, T. Fibrin glue and polyglycolic acid nonwoven fabric as a biocompatible dural substitute. Oper. Neurosurg. 2006, 58, 131–134. [Google Scholar] [CrossRef]
- Mochizuki, M.; Hirami, M. Structural effects on the biodegradation of aliphatic polyesters. Polym. Advan. Technol. 1997, 8, 203–209. [Google Scholar] [CrossRef]
- Gunatillake, P.; Mayadunne, R.; Adhikari, R. Recent developments in biodegradable synthetic polymers. Biotech. Ann. Rev. 2006, 12, 301–347. [Google Scholar]
- Wang, M.; Chen, W.; Zhang, H.; Li, X.; Zhang, Y.; Yao, K.; Yao, F. Synthesis and characterization of PLLA-PLCA-PEG multiblock copolymers and their applications in modifying PLLA porous scaffolds. Eur. Polym. J. 2007, 43, 4683–4694. [Google Scholar] [CrossRef]
- Xia, N.; Yu, W.; Wang, Y.; Han, Y.; Zheng, P.; Wang, W.; Sakaguchi, G.; Matsuda, K.; Saijo, K.; Takenaka, M.; et al. Inserting polyoxomolybdate cluster into poly(epsilon-caprolactone) to create a class of new heteropolymer: Synthesis and supramolecular structures. Polymer 2011, 52, 1772–1780. [Google Scholar] [CrossRef]
- Hu, T.; Yang, C.; Lin, S.; Yu, Q.; Wang, G. Biodegradable stents for coronary artery disease treatment: Recent advances and future perspectives. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 91, 163–178. [Google Scholar] [CrossRef]
- Vanwachem, P.B.; Beugeling, T.; Feijen, J.; Bantjes, A.; Detmers, J.P.; Vanaken, W.G. Interaction of cultured human-endothelial cells with polymeric surfaces of different wettabilities. Biomaterials 1985, 6, 403–408. [Google Scholar] [CrossRef] [Green Version]
- Eberhart, R.C.; Su, S.H.; Nguyen, K.T.; Zilberman, M.; Tang, L.P.; Nelson, K.D.; Frenkel, P. Bioresorbable polymeric stents: Current status and future promise. J. Biomat. Sci. Polym. Ed. 2003, 14, 299–312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farah, S.; Anderson, D.G.; Langer, R. Physical and mechanical properties of PLA, and their functions in widespread applications—A comprehensive review. Adv. Drug. Deliv. Rev. 2016, 107, 367–392. [Google Scholar] [CrossRef] [Green Version]
- Kitabata, H.; Waksman, R.; Warnack, B. Bioresorbable metal scaffold for cardiovascular application: Current knowledge and future perspectives. Cardio. Revascular. Med. 2014, 15, 109–116. [Google Scholar] [CrossRef]
- Pache, J.; Kastrati, A.; Mehilli, J.; Schuhlen, H.; Dotzer, F.; Hausleiter, J.; Fleckenstein, M.; Neumann, F.J.; Sattelberger, U.; Schmitt, C.; et al. Intracoronary stenting and angiographic results: Strut thickness effect on restenosis outcome (ISAR-STEREO-2) trial. J. Am. Coll. Cardiol. 2003, 41, 1283–1288. [Google Scholar] [CrossRef] [Green Version]
- Kastrati, A.; Mehilli, J.; Dirschinger, J.; Dotzer, F.; Schuhlen, H.; Neumann, F.J.; Fleckenstein, M.; Pfafferott, C.; Seyfarth, M.; Schomig, A. Intracoronary stenting and angiographic results—Strut thickness effect on restenosis outcome (ISAR-STEREO) trial. Circulation 2001, 103, 2816–2821. [Google Scholar] [CrossRef] [Green Version]
- Koskinas, K.C.; Chatzizisis, Y.S.; Antoniadis, A.P.; Giannoglou, G.D. Role of endothelial shear stress in stent restenosis and thrombosis: Pathophysiologic mechanisms and implications for clinical translation. J. Am. Coll. Cardiol. 2012, 59, 1337–1349. [Google Scholar] [CrossRef] [Green Version]
- Carlier, S.G.; van Damme, L.C.A.; Blommerde, C.P.; Wentzel, J.J.; van Langehove, G.; Verheye, S.; Kockx, M.M.; Knaapen, M.W.M.; Cheng, C.; Gijsen, F.; et al. Augmentation ofwall shear stress inhibits neointimal hyperplasia after stent implantation:inhibition through reduction of inflammation. Circulation 2003, 107, 2741–2746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, H.Y.; Hermiller, J.; Sinha, A.K.; Sturek, M.; Zhu, L.; Kassab, G.S. Effects of stent sizing on endothelial and vessel wall stress: Potential mechanisms for in-stent restenosis. J. Appl. Physiol. 2009, 106, 1686–1691. [Google Scholar] [CrossRef] [Green Version]
- Jiménez, J.M.; Davies, P.F. Hemodynamically driven stent strut design. Ann. Biomed. Eng. 2009, 37, 1483–1494. [Google Scholar] [CrossRef]
- LaDisa, J.F., Jr.; Olson, L.E.; Molthen, R.C.; Hettrick, D.A.; Pratt, P.F.; Hardel, M.D.; Kersten, J.R.; Warltier, D.C.; Pagel, P.S. Alterations in wall shear stress predict sites of neointimal hyperplasia after stent implantation in rabbit iliac arteries. Am. J. Physiol. Heart Circ. Physiol. 2005, 288, H2465–H2475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mejia, J.; Ruzzeh, B.; Mongrain, R.; Leask, R.; Bertrand, O.F. Evaluation of the effect of stent strut profile on shear stress distribution using statistical moments. Biomed. Eng. Online 2009, 8, 8–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bourantas, C.V.; Papafaklis, M.I.; Kotsia, A.; Farooq, V.; Muramatsu, T.; Gomez-Lara, J.; Zhang, Y.J.; Iqbal, J.; Kalatzis, F.G.; Naka, K.K.; et al. Effect of the endothelial shear stress patterns on neointimal proliferation following drug-eluting bioresorbable vascular scaffold implantation: An optical coherence tomography study. JACC Cardiovasc. Interv. 2014, 7, 315–324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tenekecioglu, E.; Sotomi, Y.; Torii, R.; Bourantas, C.; Miyazaki, Y.; Collet, C.; Crake, T.; Su, S.; Onuma, Y.; Serruys, P.W. Strut protrusion and shape impact on endothelial shear stress: Insights from pre-clinical study comparing Mirage and Absorb bioresorbable scaffolds. Int. J. Cardiovasc. Imaging 2017, 33, 1313–1322. [Google Scholar] [CrossRef] [Green Version]
- Venkatraman, S.; Poh, T.L.; Vinalia, T.; Mak, K.H.; Boey, F. Collapse pressures of biodegradable stents. Biomaterials 2003, 24, 2105–2111. [Google Scholar] [CrossRef]
- Venkatraman, S.S.; Tan, L.P.; Joso, J.F.; Boey, Y.C.; Wang, X. Biodegradable stents with elastic memory. Biomaterials 2006, 27, 1573–1578. [Google Scholar] [CrossRef]
- Grabow, N.; Schlun, M.; Sternberg, K.; Hakansson, N.; Kramer, S.; Schmitz, K.P. Mechanical properties of laser cut poly(L-lactide) micro-specimens: Implications for stent design, manufacture, and sterilization. J. Biomech. Eng. 2005, 127, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Weir, N.A.; Buchanan, F.J.; Orr, J.F.; Farrar, D.F.; Boyd, A. Processing, annealing and sterilisation of poly-l-lactide. Biomaterials 2004, 25, 3939–3949. [Google Scholar] [CrossRef]
- Athanasiou, K.A.; Niederauer, G.G.; Agrawal, C.M. Sterilization, toxicity, biocompatibility and clinical applications of polylactic acid/polyglycolic acid copolymers. Biomoterials 1996, 17, 93–102. [Google Scholar] [CrossRef]
- Kimberly, A.; Hooper, J.D.C.; Joachim, K. Comparison of the Effect of Ethylene Oxide and g-Irradiation on Selected Tyrosine-Derived Polycarbonates and Poly(L-lactic acid). J. Appl. Polym. Sci. 1996, 63, 1499–1510. [Google Scholar]
- Carrasco, F.; Pagès, P.; Gámez-Pérez, J.; Santana, O.O.; Maspoch, M.L. Processing of poly(lactic acid): Characterization of chemical structure, thermal stability and mechanical properties. Polym. Degrad. Stabil. 2010, 95, 116–125. [Google Scholar] [CrossRef]
- Hua, R.; Tian, Y.; Cheng, J.; Wu, G.; Jiang, W.; Ni, Z.; Zhao, G. The effect of intrinsic characteristics on mechanical properties of poly(l-lactic acid) bioresorbable vascular stents. Med. Eng. Phys. 2020, 81, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Bergstrom, J.S.; Hayman, D. An Overview of Mechanical Properties and Material Modeling of Polylactide (PLA) for Medical Applications. Ann. Biomed. Eng. 2016, 44, 330–340. [Google Scholar] [CrossRef]
- Gupta, B.; Revagade, N.; Hilborn, J. Poly(lactic acid) fiber: An overview. Prog. Polym. Sci. 2007, 32, 455–482. [Google Scholar] [CrossRef]
- Lu, L.; Peter, S.J.; Lyman, M.D.; Lai, H.-L.; Leite, S.M.; Tamada, J.A.; Vacanti, J.P.; Langer, R.; Mikos, A.G. In vitro degradation of porous poly(l-lactic acid) foams. Biomaterials 2000, 21, 1595–1605. [Google Scholar] [CrossRef]
- Goonoo, N.; Bhaw-Luximon, A.; Jhurry, D. Biodegradable polymer blends: Miscibility, physicochemical properties and biological response of scaffolds. Polym. Int. 2015, 64, 1289–1302. [Google Scholar] [CrossRef]
- Naseem, R.; Zhao, L.; Silberschmidt, V.; Liu, Y.; Scaife, O.; Willcock, H.; Eswaran, S.; Hossainy, S. Mechanical and chemical characterisation of bioresorbable polymeric stent over two-year in vitro degradation. J. Biomater. Appl. 2019, 34, 61–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaona, L.A.; Gómez Ribelles, J.L.; Perilla, J.E.; Lebourg, M. Hydrolytic degradation of PLLA/PCL microporous membranes prepared by freeze extraction. Polym. Degrad. Stabil. 2012, 97, 1621–1632. [Google Scholar] [CrossRef]
- Matta, A.K.; Rao, R.U.; Suman, K.N.S.; Rambabu, V. Preparation and Characterization of Biodegradable PLA/PCL Polymeric Blends. Proc. Mater. Sci. 2014, 6, 1266–1270. [Google Scholar] [CrossRef] [Green Version]
- Bobel, A.C.; Lohfeld, S.; Shirazi, R.N.; McHugh, P.E. Experimental mechanical testing of Poly (l-Lactide) (PLLA) to facilitate pre-degradation characteristics for application in cardiovascular stenting. Polym. Test. 2016, 54, 150–158. [Google Scholar] [CrossRef] [Green Version]
- Boland, E.L.; Shine, R.; Kelly, N.; Sweeney, C.A.; McHugh, P.E. A Review of Material Degradation Modelling for the Analysis and Design of Bioabsorbable Stents. Ann. Biomed. Eng. 2016, 44, 341–356. [Google Scholar] [CrossRef] [PubMed]
- Karanasiou, G.S.; Papafaklis, M.I.; Conway, C.; Michalis, L.K.; Tzafriri, R.; Edelman, E.R.; Fotiadis, D.I. Stents: Biomechanics, Biomaterials, and Insights from Computational Modeling. Ann. Biomed. Eng. 2017, 45, 853–872. [Google Scholar] [CrossRef] [PubMed]
- Rajagopal, K.R.; Wineman, A.S. A constitutive equation for nonlinear solids which undergo deformation induced microstructural changes. Int. J. Plast. 1992, 8, 385–395. [Google Scholar] [CrossRef] [Green Version]
- Rajagopal, K.R.; Srinivasa, A.R.; Wineman, A.S. On the shear and bending of a degrading polymer beam. Int. J. Plast. 2007, 23, 1618–1636. [Google Scholar] [CrossRef]
- Soares, J.S.; Moore, J.E., Jr.; Rajagopal, K.R. Constitutive framework for biodegradable polymers with applications to biodegradable stents. Asaio J. 2008, 54, 295–301. [Google Scholar] [CrossRef]
- Soares, J.S.; Rajagopal, K.; Moore, J.E., Jr. Theoretical modeling of cyclically loaded, biodegradable cylinders. In European Conference on Computational Mechanics, 3rd ed.; Motasoares, C.A., Martins, J.A.C., Rodrigues, H.C., Ambrosio, J.C., Pina, C.A.B., Motasoares, C.M., Pereira, E.B.R., Folgado, J., Eds.; Springer: Lisbon, Portugal, 2006; p. 207. [Google Scholar]
- Muliana, A.; Rajagopal, K.R. Modeling the response of nonlinear viscoelastic biodegradable polymeric stents. Int. J. Solids Struct. 2012, 49, 989–1000. [Google Scholar] [CrossRef] [Green Version]
- Luo, Q.; Liu, X.; Li, Z.; Huang, C.; Zhang, W.; Meng, J.; Chang, Z.; Hua, Z. Degradation Model of Bioabsorbable Cardiovascular Stents. PLoS ONE 2014, 9, e110278. [Google Scholar]
- Shazly, T.; Kolachalama, V.B.; Ferdous, J.; Oberhauser, J.P.; Hossainy, S.; Edelman, E.R. Assessment of Material By-Product Fate from Bioresorbable Vascular Scaffolds. Ann. Biomed. Eng. 2012, 40, 955–965. [Google Scholar] [CrossRef]
- Khan, K.A.; El-Sayed, T. A phenomenological constitutive model for the nonlinear viscoelastic responses of biodegradable polymers. Acta Mech. 2013, 224, 287–305. [Google Scholar] [CrossRef]
- Lin, S.; Dong, P.; Zhou, C.; Dallan, L.A.P.; Zimin, V.N.; Pereira, G.T.R.; Lee, J.; Gharaibeh, Y.; Wilson, D.L.; Bezerra, H.G.; et al. Degradation modeling of poly-l-lactide acid (PLLA) bioresorbable vascular scaffold within a coronary artery. Nanotechnol. Rev. 2020, 9, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Ormiston, J.A.; Serruys, P.W.; Regar, E.; Dudek, D.; Thuesen, L.; Webster, M.W.I.; Onuma, Y.; Garcia-Garcia, H.M.; McGreevy, R.; Veldhof, S. A bioabsorbable everolimus-eluting coronary stent system for patients with single de-novo coronary artery lesions (ABSORB): A prospective open-label trial. Lancet 2008, 371, 899–907. [Google Scholar] [CrossRef]
- Serruys, P.W.; Chevalier, B.; Dudek, D.; Cequier, A.; Carrié, D.; Iniguez, A.; Dominici, M.; van der Schaaf, R.J.; Haude, M.; Wasungu, L.; et al. A bioresorbable everolimus-eluting scaffold versus a metallic everolimus-eluting stent for ischaemic heart disease caused by de-novo native coronary artery lesions (ABSORB II): An interim 1-year analysis of clinical and procedural secondary outcomes from a randomised controlled trial. Lancet 2015, 385, 43–54. [Google Scholar] [PubMed]
- Stone, G.W.; Ellis, S.G.; Gori, T.; Metzger, D.C.; Stein, B.; Erickson, M.; Torzewski, J.; Williams, J.; Lawson, W.; Broderick, T.M.; et al. Blinded outcomes and angina assessment of coronary bioresorbable scaffolds: 30-day and 1-year results from the ABSORB IV randomised trial. Lancet 2018, 392, 1530–1540. [Google Scholar] [CrossRef]
- Olonisakin, K.; Fan, M.Z.; Xin-Xiang, Z.; Ran, L.; Lin, W.S.; Zhang, W.F.; Yang, W.B. Key Improvements in Interfacial Adhesion and Dispersion of Fibers/Fillers in Polymer Matrix Composites; Focus on PLA Matrix Composites. Compos. Interfaces 2021, 1–50. [Google Scholar] [CrossRef]
- Xu, K.; Fu, G.; Xu, B.; Zhou, Y.; Su, X.; Liu, H.; Zhang, Z.; Yu, B.; Wang, X.; Han, Y.; et al. Safety and efficacy of the novel sirolimus-eluting bioresorbable scaffold for the treatment of de novo coronary artery disease: One-year results from a prospective patient-level pooled analysis of NeoVas trials. Catheter. Cardiovasc. Interv. 2019, 93, 832–838. [Google Scholar] [CrossRef]
- Feng, G.; Qin, C.; Sha, F.; Lyu, Y.; Xia, J.; Jiang, X.; Villarreal-Gómez, L.J. Evaluation of Inflammatory and Calcification after Implantation of Bioabsorbable Poly-L-Lactic Acid/Amorphous Calcium Phosphate Scaffolds in Porcine Coronary Arteries. J. Nanomater. 2021, 2021, 6652648. [Google Scholar] [CrossRef]
- Wu, Y.; Yin, J.; Chen, J.; Yao, Z.; Qian, J.; Shen, L.; Ge, L.; Ge, J. Final report of the 5-year clinical outcomes of the XINSORB bioresorbable sirolimus-eluting scaffold in the treatment of single de novo coronary lesions in a first-in-human study. Ann. Transl. Med. 2020, 8, 1162–1173. [Google Scholar] [CrossRef] [PubMed]
- Li, C.J.; Xu, B.; Song, L.; Yu, M.Y.; Yan, H.B.; Qiu, H.; Mu, C.W.; Cui, J.G.; Guan, C.D.; Sun, Z.W.; et al. The safety and efficacy of Firesorb bioresorbable scaffold in first-in-man study for coronary artery disease: The four-year outcomes. Zhonghua Xin Xue Guan Bing Za Zhi 2021, 49, 128–135. [Google Scholar] [PubMed]
- Rickert, D.; Lendlein, A.; Peters, I.; Moses, M.A.; Franke, R.P. Biocompatibility testing of novel multifunctional polymeric biomaterials for tissue engineering applications in head and neck surgery: An overview. Eur. Arch. Otorhinolaryngol. 2006, 263, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.M.; Langone, J.J. Issues and perspectives on the biocompatibility and immunotoxicity 1 evaluation of implanted controlled release systems. J. Control. Release 1999, 57, 107–113. [Google Scholar] [CrossRef]
- Mendes, S.C.; Reis, R.L.; Bovell, Y.P.; Cunha, A.M.; van Blitterswijk, C.A.; de Bruijn, J.D. Biocompatibility testing of novel starch-based materials with potential application in orthopaedic surgery: A preliminary study. Biomaterials 2001, 22, 2057–2064. [Google Scholar] [CrossRef]
- Fournier, E.P.C.; Montero-Menei, C.N.; Benoit, J.P. Biocompatibility of implantable synthetic polymeric drug carriers: Focus on brain biocompatibility. Biomaterials 2003, 19, 3311–3331. [Google Scholar] [CrossRef]
- Laurencin, C.T.; Elgendy, H. The Biocompatibility and Toxicity of Degradable Polymeric Materials: Implication for Drug Delivery; John Wiley Sons: New York, NY, USA, 1994; pp. 27–46. [Google Scholar]
- Sieminski, A.L.; Gooch, K.J. Biomaterial-microvasculature interactions. Biomaterials 2000, 21, 2232–2241. [Google Scholar] [CrossRef]
- Ríhová, B. Immunocompatibility and biocompatibility of cell delivery systems. Adv. Drug Deliv. Rev. 2000, 42, 65–80. [Google Scholar] [CrossRef]
- Mikos, A.G.; McIntire, L.V.; Anderson, J.M.; Babensee, J.E. Host response to tissue engineered devices. Adv. Drug Deliv. Rev. 1998, 33, 111–139. [Google Scholar]
- Gaspardone, A.V.F. Coronary stenting and inflammation. Am. J. Cardiol. 2005, 96, 65L–75L. [Google Scholar] [CrossRef]
- Kornowski, R.H.M.; Tio, F.O.; Bramwell, O.; Wu, H.; Leon, M. In-stent restenosis: Contributions of inflammatory responses and arterial injury to neointimal hyperplasia. J. Am. Coll. Cardiol. 1998, 31, 224–230. [Google Scholar] [CrossRef] [Green Version]
- Van der Giessen, W.J.; Lincoff, A.M.; Schwartz, R.S.; van Beusekom, H.M.; Serruys, P.W.; Holmes DRJr Ellis, S.G.; Topol, E.J. Marked inflammatory sequelae to implantation of biodegradable and nonbiodegradable polymers in porcine coronary arteries. Circulation 1996, 94, 1690–1697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, S.S.; Landau, C.; Chao, R.Y.N.; Timmons, R.; Meidell, R.; Tang, L.P.; Eberhart, R. Expandable, bioresorbable endovascular stent with anti-platelet and anti-inflammation treatments. Circulation 2001, 104, 2401. [Google Scholar]
- Hietala, E.S.; Stahls, A.; Valimaa, T.; Maasilta, P.; Tormala, P.; Nieminen, M.S.; Harjula, A.L.J. Biodegradation of the copolymeric polylactide stent—Long-term follow-up in a rabbit aorta model. J. Vasc. Res. 2001, 38, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, T.; Shandas, R. A Survey of Surface Modification Techniques for Next-Generation Shape Memory Polymer Stent Devices. Polymers 2014, 6, 2309–2331. [Google Scholar] [CrossRef]
- Martinez, A.W.; Chaikof, E.L. Microfabrication and nanotechnology in stent design. Wires. Nanomed. Nanobiotechnol. 2011, 3, 256–268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boura, C.; Menu, P.; Payan, E.; Picart, C.; Voegel, J.C.; Muller, S.; Stoltz, J.F. Endothelial cells grown on thin polyelectrolyte mutlilayered films: An evaluation of a new versatile surface modification. Biomaterials 2003, 24, 3521–3530. [Google Scholar] [CrossRef]
- Angelova, N.; Hunkeler, D. Rationalizing the design of polymeric biomaterials. Trends Biotechnol. 1999, 17, 409–421. [Google Scholar] [CrossRef]
- Ma, Z.; Mao, Z.; Gao, C. Surface modification and property analysis of biomedical polymers used for tissue engineering. Colloids Surf. B. 2007, 60, 137–157. [Google Scholar] [CrossRef]
- Prasad, C.K.; Muraleedharan, C.V.; Krishnan, L.K. Bio-mimetic composite matrix that promotes endothelial cell growth for modification of biomaterial surface. J. Biomed. Mater. Res. A 2007, 80A, 644–654. [Google Scholar] [CrossRef]
- Pavithra, D.; Doble, M. Biofilm formation, bacterial adhesion and host response on polymeric implants—Issues and prevention. Biomed. Mater. 2008, 3, 034003. [Google Scholar] [CrossRef]
- Helmus, M.N.; Gibbons, D.F.; Cebon, D. Biocompatibility: Meeting a Key Functional Requirement of Next- Generation Medical Devices. Toxicol. Pathol. 2008, 36, 70–80. [Google Scholar] [CrossRef]
- Arima, Y.; Iwata, H. Effect of wettability and surface functional groups on protein adsorption and cell adhesion using well-defined mixed self-assembled monolayers. Biomaterials 2007, 28, 3074–3082. [Google Scholar] [CrossRef]
- Vanwachem, P.B.; Hogt, A.H.; Beugeling, T.; Feijen, J.; Bantjes, A.; Detmers, J.P.; Vanaken, W.G. Adhesion of cultured human-endothelial cells onto methacrylate polymers with varying surface wettability and charge. Biomaterials 1987, 8, 323–328. [Google Scholar] [CrossRef] [Green Version]
- Chu, P.K.; Chen, J.Y.; Wang, L.P.; Huang, N. Plasma-surface modification of biomaterials. Mater. Sci. Eng. R Rep. 2002, 36, 143–206. [Google Scholar] [CrossRef] [Green Version]
- Ratner, B.D. Surface modification of polymers—Chemical, biological and surface analytical challenges. Biosens. Bioelectro. 1995, 10, 797–804. [Google Scholar] [CrossRef]
- Lyu, S.P.; Cernohous, J.J.; Bates, F.S.; Macosko, C.W. Interfacial reaction induced roughening in polymer blends. Macromolecules 1999, 32, 106–110. [Google Scholar] [CrossRef]
- Curtis, A.; Wilkinson, C. Topographical control of cells. Biomaterials 1997, 18, 1573–1583. [Google Scholar] [CrossRef]
- Meredith, J.C.; Sormana, J.L.; Keselowsky, B.G.; Garcia, A.J.; Tona, A.; Karim, A.; Amis, E.J. Combinatorial characterization of cell interactions with polymer surfaces. J. Biomed. Mater. Res. A 2003, 66A, 483–490. [Google Scholar] [CrossRef]
- Fare, S.; Valtulina, V.; Petrini, P.; Alessandrini, E.; Pietrocola, G.; Tanzi, M.C.; Speziale, P.; Visai, L. In vitro interaction of human fibroblasts and platelets with a shape-memory polyurethane. J. Biomed. Mater. Res. A 2005, 73A, 1–11. [Google Scholar] [CrossRef]
- Craighead, H.G.; James, C.D.; Turner, A.M.P. Chemical and topographical patterning for directed cell attachment. Curr. Opin. Solid State Mater. Sci. 2001, 5, 177–184. [Google Scholar] [CrossRef]
- Noeske, M.; Degenhardt, J.; Strudthoff, S.; Lommatzsch, U. Plasma jet treatment of five polymers at atmospheric pressure: Surface modifications and the relevance for adhesion. Int. J. Adhes. Adhes. 2004, 24, 171–177. [Google Scholar] [CrossRef]
- Falconnet, D.; Csucs, G.; Grandin, H.M.; Textor, M. Surface engineering approaches to micropattern surfaces for cell-based assays. Biomaterials 2006, 27, 3044–3063. [Google Scholar] [CrossRef] [PubMed]
- Del Campo, A.; Arzt, E. Fabrication approaches for generating complex micro- and nanopatterns on polymeric surfaces. Chem. Rev. 2008, 108, 911–945. [Google Scholar] [CrossRef] [PubMed]
- Nie, Z.; Kumacheva, E. Patterning surfaces with functional polymers. Nat. Mater. 2008, 7, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.Y.; Yang, F.; Wang, S.; Ramakrishna, S. In vitro study of human vascular endothelial cell function on materials with various surface roughness. J. Biomed. Mater. Res. A 2004, 71A, 154–161. [Google Scholar] [CrossRef]
- Kane, R.S.; Takayama, S.; Ostuni, E.; Ingber, D.E.; Whitesides, G.M. Patterning proteins and cells using soft lithography. Biomaterials 1999, 20, 2363–2376. [Google Scholar] [CrossRef]
- Lahann, J.; Balcells, M.; Rodon, T.; Lee, J.; Choi, I.S.; Jensen, K.F.; Langer, R. Reactive polymer coatings: A platform for patterning proteins and mammalian cells onto a broad range of materials. Langmuir 2002, 18, 3632–3638. [Google Scholar] [CrossRef]
- Yamato, M.; Konno, C.; Utsumi, M.; Kikuchi, A.; Okano, T. Thermally responsive polymer-grafted surfaces facilitate patterned cell seeding and co-culture. Biomaterials 2002, 23, 561–567. [Google Scholar] [CrossRef]
- Zhang, M.Q.; Desai, T.; Ferrari, M. Proteins and cells on PEG immobilized silicon surfaces. Biomaterials 1998, 19, 953–960. [Google Scholar] [CrossRef]
- Mrksich, M.; Whitesides, G.M. Using self-assembled monolayers to understand the interactions of man-made surfaces with proteins and cells. Ann. Rev. Biophy. Biomol. Struct. 1996, 25, 55–78. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Azzaroni, O.; Zhou, F.; Huck, W.T.S. Topography printing to locally control wettability. J. Am. Chem. Soc. 2006, 128, 7730–7731. [Google Scholar] [CrossRef]
- Shi, J.X.; Li, H.; He, P.S. Micro- and nano-patterning of polymers. Chin. Sci. Bull. 2004, 49, 1431–1436. [Google Scholar] [CrossRef]
- Yang, F.; Wornyo, E.; Gall, K.; King, W.P. Nanoscale indent formation in shape memory polymers using a heated probe tip. Nanotechnology 2007, 18, 285302–285311. [Google Scholar] [CrossRef]
- Wornyo, E.; Gall, K.; Yang, F.; King, W. Nanoindentation of shape memory polymer networks. Polymer 2007, 48, 3213–3225. [Google Scholar] [CrossRef]
- Hinz, M.; Kleiner, A.; Hild, S.; Marti, O.; Durig, U.; Gotsmann, B.; Drechsler, U.; Albrecht, T.R.; Vettiger, P. Temperature dependent nano indentation of thin polymer films with the scanning force microscope. Eur. Polym. J. 2004, 40, 957–964. [Google Scholar] [CrossRef]
- Lee, H.; Dellatore, S.M.; Miller, W.M.; Messersmith, P.B. Mussel-inspired surface chemistry for multifunctional coatings. Science 2007, 318, 426–430. [Google Scholar] [CrossRef] [Green Version]
- Favia, P.; d’Agostino, R. Plasma treatments and plasma deposition of polymers for biomedical applications. Surf. Coat. Technol. 1998, 98, 1102–1106. [Google Scholar] [CrossRef]
- Chandy, T.; Das, G.S.; Wilson, R.F.; Rao, G.H.R. Use of plasma glow for surface-engineering biomolecules to enhance bloodcompatibility of Dacron and PTFE vascular prosthesis. Biomaterials 2000, 21, 699–712. [Google Scholar] [CrossRef]
- Pareta, R.A.; Reising, A.B.; Miller, T.; Storey, D.; Webster, T.J. Increased Endothelial Cell Adhesion on Plasma Modified Nanostructured Polymeric and Metallic Surfaces for Vascular Stent Applications. Biotechnol. Bioeng. 2009, 103, 459–471. [Google Scholar] [CrossRef]
- Chu, C.F.L.; Lu, A.; Liszkowski, M.; Sipehia, R. Enhanced growth of animal and human endothelial cells on biodegradable polymers. Bba. Gen. Subj. 1999, 1472, 479–485. [Google Scholar] [CrossRef]
- Bilek, M.M.M.; Bax, D.V.; Kondyurin, A.; Yin, Y.; Nosworthy, N.J.; Fisher, K.; Waterhouse, A.; Weiss, A.S.; dos Remedios, C.G.; McKenzie, D.R. Free radical functionalization of surfaces to prevent adverse responses to biomedical devices. Proc. Natl. Acad. Sci. USA 2011, 108, 14405–14410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, Y.; Zhao, H.; Behl, M.; Lendlein, A.; Guo, J.; Yang, D. Grafting of poly (ethylene glycol) monoacrylates on polycarbonateurethane by UV initiated polymerization for improving hemocompatibility. J. Mater. Sci. Mater. Med. 2013, 24, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Chaudhury, M.K. Self-Assembled Monolayers on Polymer Surfaces. Biosens. Bioelectron. 1995, 10, 785–788. [Google Scholar] [CrossRef]
- Liang, C.L.; Li, L.; Mao, C.; Zhang, J.; Shen, J. Synthesis and characterization of shape-memory polyurethane films with blood compatibility. In Proceedings of the Second International Conference on Smart Materials and Nanotechnology in Engineering, Weihai, China, 20 October 2009; Volume 7493, p. 749338. [Google Scholar]
- Luescher, T.F.; Steffel, J.; Eberli, F.R.; Joner, M.; Nakazawa, G.; Tanner, F.C.; Virmani, R. Drug-eluting stent and coronary thrombosis—Biological mechanisms and clinical implications. Circulation 2007, 115, 1051–1058. [Google Scholar] [CrossRef] [Green Version]
- Camici, G.G.; Steffel, J.; Akhmedov, A.; Schafer, N.; Baldinger, J.; Schulz, U.; Shojaati, K.; Matter, C.M.; Yang, Z.; Luescher, T.F.; et al. Dimethyl sulfoxide inhibits tissue factor expression, thrombus formation, and vascular smooth muscle cell activation—A potential treatment strategy for drug-eluting stents. Circulation 2006, 114, 1512–1521. [Google Scholar] [CrossRef] [Green Version]
- Sun, T.L.; Tan, H.; Han, D.; Fu, Q.; Jiang, L. No platelet can adhere—Largely improved blood compatibility on nanostructured superhydrophobic surfaces. Small 2005, 1, 959–963. [Google Scholar] [CrossRef]
- Tirrell, M.; Kokkoli, E.; Biesalski, M. The role of surface science in bioengineered materials. Surf. Sci. 2002, 500, 61–83. [Google Scholar] [CrossRef]
- Pakalns, T.; Haverstick, K.L.; Fields, G.B.; McCarthy, J.B.; Mooradian, D.L.; Tirrell, M. Cellular recognition of synthetic peptide amphiphiles in self-assembled monolayer films. Biomaterials 1999, 20, 2265–2279. [Google Scholar] [CrossRef]
- Sharif, F.; Hynes, S.O.; Cooney, R.; Howard, L.; McMahon, J.; Daly, K.; Crowley, J.; Barry, F.; O’Brien, T. Gene-eluting stents: Adenovirus-mediated delivery of eNOS to the blood vessel wall accelerates re-endothelialization and inhibits restenosis. Mol. Ther. 2008, 16, 1674–1680. [Google Scholar] [CrossRef]
- Goddard, J.M.; Hotchkiss, J.H. Polymer surface modification for the attachment of bioactive compounds. Prog. Polym. Sci. 2007, 32, 698–725. [Google Scholar] [CrossRef]
- Wang, Y.X.; Robertson, J.L.; Spillman, W.B.; Claus, R.O. Effects of the chemical structure and the surface properties of polymeric biomaterials on their biocompatibility. Pharm. Resdordr. 2004, 21, 1362–1373. [Google Scholar] [CrossRef]
- Mao, C.; Qiu, Y.Z.; Sang, H.B.; Mei, H.; Zhu, A.P.; Shen, J.; Lin, S.C. Various approaches to modify biomaterial surfaces for improving hemocompatibility. Adv. Colloid Interfac. 2004, 110, 5–17. [Google Scholar] [CrossRef]
- Costa, M.A.; Simon, D.I. Molecular basis of restenosis and drug-eluting stents. Circulation 2005, 111, 2257–2273. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Nguyen, K.T.; Brilakis, E.S.; Yang, J.; Fuh, E.; Banerjee, S. Enhanced Endothelialization of a New Stent Polymer Through Surface Enhancement and Incorporation of Growth Factor-Delivering Microparticles. J. Cardiovasc. Trans. 2012, 5, 519–527. [Google Scholar] [CrossRef]
- Garner, B.; Hodgson, A.J.; Wallace, G.G.; Underwood, P.A. Human endothelial cell attachment to and growth on polypyrrole-heparin is vitronectin dependent. J. Mater. Sci. Mater. Med. 1999, 10, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y. Surface micropatterning to regulate cell functions. Biomaterials 1999, 20, 2333–2342. [Google Scholar] [CrossRef]
- Wieneke, H.; Dirsch, O.; Sawitowski, T.; Gu, Y.L.; Brauer, H.; Dahmen, U.; Fischer, A.; Wnendt, S.; Erbel, R. Synergistic effects of a novel nanoporous stent coating and tacrolimus on intima proliferation in rabbits. Catheter. Cardiovasc. Interv. 2003, 60, 399–407. [Google Scholar] [CrossRef]
- Costa, J.R.; Abizaid, A.; Costa, R.; Feres, F.; Tanajura, L.F.; Abizaid, A.; Maldonado, G.; Staico, R.; Siqueira, D.; Sousa, A.G.M.R.; et al. 1-Year Results of the Hydroxyapatite Polymer-Free Sirolimus-Eluting Stent for the Treatment of Single De Novo Coronary Lesions The VESTASYNC I Trial. JACC Cardiovasc. Interv. 2009, 2, 422–427. [Google Scholar] [CrossRef] [Green Version]
- Bhargava, B.; Reddy, N.K.; Karthikeyan, G.; Raju, R.; Mishra, S.; Singh, S.; Waksman, R.; Virmani, R.; Somaraju, B. A novel paclitaxel-eluting porous carbon-carbon nanoparticle coated, nonpolymeric cobalt-chromium stent: Evaluation in a porcine model. Catheter. Cardiovasc. Interv. 2006, 67, 698–702. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, J.-H.; Jeon, O.; Kwon, I.C.; Park, K. Engineered polymers for advanced drug delivery. Eur. J. Pharm. Biopharm. 2009, 71, 420–430. [Google Scholar] [CrossRef] [Green Version]
- Liao, S.C.; Chan, C.K.; Ramakrishna, S. Stem cells and biomimetic materials strategies for tissue engineering. Mater. Sci. Eng. C 2008, 28, 1189–1202. [Google Scholar] [CrossRef]
- Rudolph, A.; Teske, M.; Illner, S.; Kiefel, V.; Sternberg, K.; Grabow, N.; Wree, A.; Hovakimyan, M. Surface Modification of Biodegradable Polymers towards Better Biocompatibility and Lower Thrombogenicity. PLoS ONE 2015, 10, e0142075. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.J.; Jo, H.H.; Lim, K.S.; Lim, D.; Lee, S.; Lee, J.H.; Kim, W.D.; Jeong, M.H.; Lim, J.Y.; Kwon, I.K.; et al. Heparin coating on 3D printed poly (l-lactic acid) biodegradable cardiovascular stent via mild surface modification approach for coronary artery implantation. Chem. Eng. J. 2019, 378, 122116. [Google Scholar] [CrossRef]
- Naseerali, C.P.; Hari, P.R.; Sreenivasan, K. The release kinetics of drug eluting stents containing sirolimus as coated drug: Role of release media. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2010, 878, 709–712. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Gu, Y.; Hua, R.; Ni, Z.; Zhao, G. In vitro release study of sirolimus from a PDLLA matrix on a bioresorbable drug-eluting stent. J. Drug Deliv. Sci. Technol. 2018, 48, 88–95. [Google Scholar] [CrossRef]
- Sojitra, P.; Kothwala, D.; Raval, A.; Kotadia, H.; Adeshara, S. Covalently Conjugation of Genistein with Biodegradable Poly L-Lactide. Trends Biomat. Artif. Org. 2010, 23, 144–148. [Google Scholar]
- Yamawaki, T.; Shimokawa, H.; Kozai, T.; Miyata, K.; Higo, T.; Tanaka, E.; Egashira, K.; Shiraishi, T.; Tamai, H.; Igaki, K.; et al. Intramural delivery of a specific tyrosine kinase inhibitor with biodegradable stent suppresses the restenotic changes of the coronary artery in pigs in vivo. J. Am. Coll. Cardiol. 1998, 32, 780–786. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; Mo, Z.; Guo, F.; Shi, D.; Han, Q.Q.; Liu, Q. Drug loaded nanoparticle coating on totally bioresorbable PLLA stents to prevent in-stent restenosis. J. Biomed. Mater. Res. B Appl. Biomater. 2018, 106, 88–95. [Google Scholar] [CrossRef]
- Alt, E.; Haehnel, I.; Beilharz, C.; Prietzel, K.; Preter, D.; Stemberger, A.; Fliedner, T.; Erhardt, W.; Schömig, A. Inhibition of Neointima Formation After Experimental Coronary Artery Stenting A New Biodegradable Stent Coating Releasing Hirudin and the Prostacyclin Analogue Iloprost. Circulation 2000, 101, 1453–1458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lincoff, A.F.J.; Ellis, S.; Tuch, R.; Topol, E. Sustained local delivery of dexamethasone by a novel intravascular eluting stent to prevent restenosis in the porcine coronary injury model. J. Am. Coll. Cardiol. 1997, 29, 808–816. [Google Scholar] [CrossRef] [Green Version]
- Karges, H.E.; Funk, K.A.; Rosenberger, H. Activity of coagulation and fibrinolysis parameters in animals. Drug Res. 1994, 44, 793–797. [Google Scholar]
- Hou, Z.; Yan, W.; Li, T.; Wu, W.; Cui, Y.; Zhang, X.; Chen, Y.P.; Yin, T.; Qiu, J.; Wang, G. Lactic acid-mediated endothelial to mesenchymal transition through TGF-beta1 contributes to in-stent stenosis in poly-L-lactic acid stent. Int. J. Biol. Macromol. 2020, 155, 1589–1598. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Weng, L.; Chen, C.; Zheng, J.; Wu, T.; Zeng, S.; Zhang, S.; Xiao, J. Inflammation and dysfunction in human aortic endothelial cells associated with poly-l-lactic acid degradation in vitro are alleviated by curcumin. J. Biomed. Mater. Res. A 2019, 107, 2756–2763. [Google Scholar] [CrossRef]
- Kathuria, Y.P. The Potential of Biocompatible Metallic Stents and Preventing Restenosis. Mater. Sci. Eng. A 2006, 417, 40–48. [Google Scholar] [CrossRef]
- Stepak, B.; Antonczak, A.J.; Bartkowiak-Jowsa, M.; Filipiak, J.; Pezowicz, C.; Abramski, K.M. Fabrication of a polymer-based biodegradable stent using a CO2 laser. Arc. Civ. Mech. Eng. 2014, 14, 317–326. [Google Scholar] [CrossRef]
- Guerra, A.J.; de Ciurana, J. Fibre laser cutting of polymer tubes for stents manufacturing. Proc. Manuf. 2017, 13, 190–196. [Google Scholar] [CrossRef]
- Tamrin, K.F.; Nukman, Y.; Choudhury, I.A.; Shirley, S. Multiple-objective optimization in precision laser cutting of different thermoplastics. Opt. Laser. Eng. 2015, 67, 57–65. [Google Scholar] [CrossRef]
- Zeina, I.; Hutmacher, D.W.; Tanc, K.C.; Teoha, S.H. Fused deposition modeling of novel scaffold architectures for tissue engineering applications. Biomaterials 2002, 23, 1169–1185. [Google Scholar] [CrossRef]
- Guerra, A.J.; Ciurana, J. 3D-printed bioabsordable polycaprolactone stent: The effect of process parameters on its physical features. Mater. Des. 2018, 137, 430–437. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, L.; Fang, Y.; Sun, W. Design, Characterization, and 3D Printing of Cardiovascular Stents with Zero Poisson’s Ratio in Longitudinal Deformation. Engineering 2020, in press. [Google Scholar] [CrossRef]
- Qiu, T.; Jiang, W.; Yan, P.; Jiao, L.; Wang, X. Development of 3D-Printed Sulfated Chitosan Modified Bioresorbable Stents for Coronary Artery Disease. Front. Bioeng. Biotechnol. 2020, 8, 462–474. [Google Scholar] [CrossRef] [PubMed]
- Guerra, A.J.; Cano, P.; Rabionet, M.; Puig, T.; Ciurana, J. 3D-Printed PCL/PLA Composite Stents: Towards a New Solution to Cardiovascular Problems. Materials 2018, 11, 1679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Z.; Zhao, J.; Wu, W.; Wang, P.; Wang, B.; Li, G.; Zhang, S. Radial Compressive Property and the Proof-of-Concept Study for Realizing Self-expansion of 3D Printing Polylactic Acid Vascular Stents with Negative Poisson’s Ratio Structure. Materials 2018, 11, 1357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Misra, S.K.; Ostadhossein, F.; Babu, R.; Kus, J.; Tankasala, D.; Sutrisno, A.; Walsh, K.A.; Bromfield, C.R.; Pan, D. 3D-Printed Multidrug-Eluting Stent from Graphene-Nanoplatelet-Doped Biodegradable Polymer Composite. Adv. Healthc. Mater. 2017, 6, 1700008–1700022. [Google Scholar] [CrossRef]

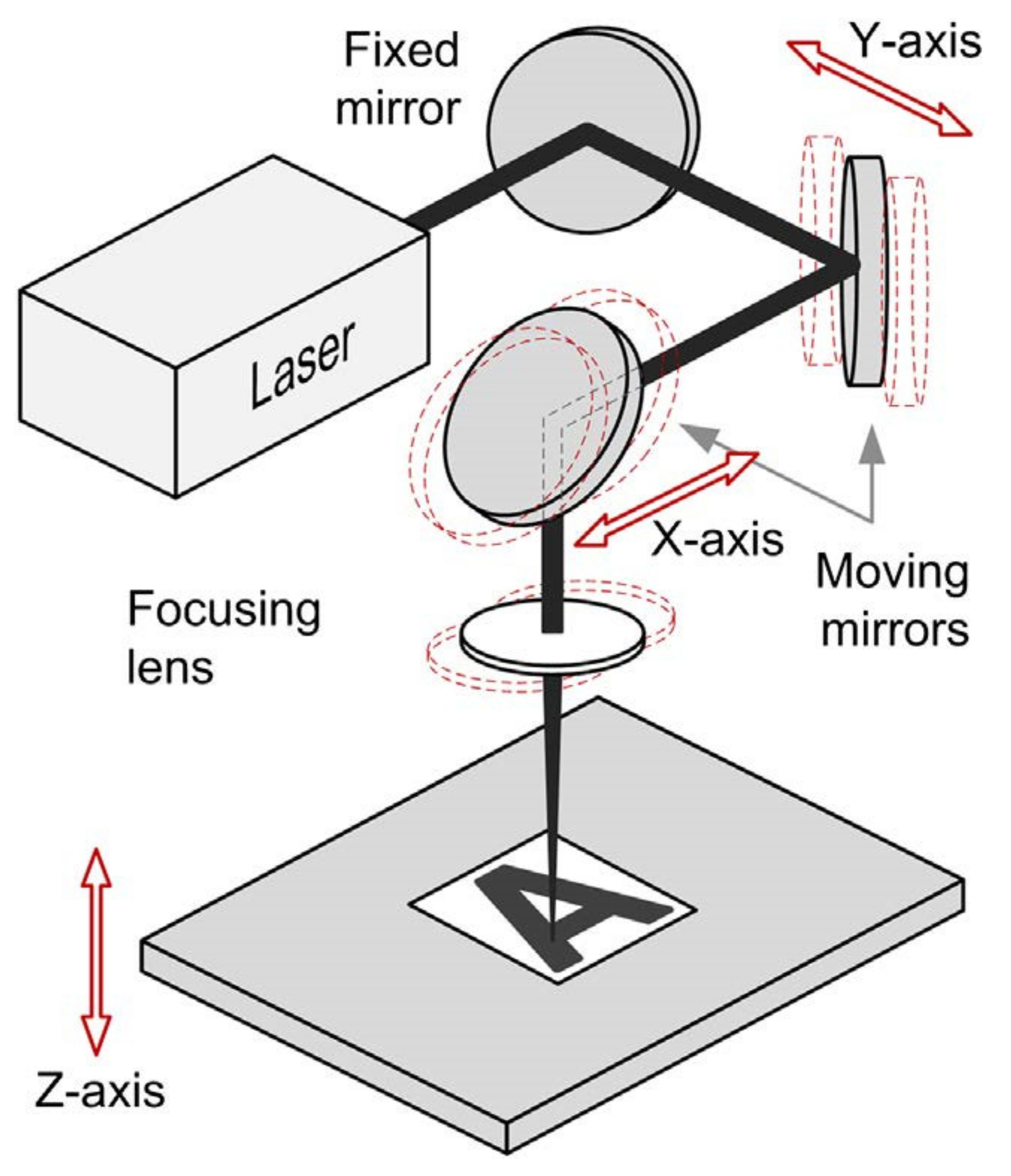

| Polymer | Tensile Strength (MPa) | Young’s Modulus (GP) | Yield Strength (MPa) | Melting Point (°C) | Elongation (%) |
|---|---|---|---|---|---|
| PLA | 21–60 | 4.0 | 70 | 150–162 | 4 |
| PLLA | 45–70 | 2.17 | 57 | 173–178 | 3.3 |
| PDLA | 46 | 2.16 | 46 | Amorphous | 2.6 |
| PDLLA | 40 | 1-3 | - | Amorphous | - |
| PGA | 77.3 | 3.33 | 77.3 | 220–225 | 3.9 |
| PCL | 20–35 | 0.4 | - | 58–63 | - |
| Methods | Principle | Function |
|---|---|---|
| Surface roughening [104,110,111,112,113,114,115] | Oxygen plasma deposition Argon plasma deposition Etching Sanding | Decrease cell migration No chemical alteration Increase surface area Restrict cell movement Enhance cell attachment |
| Surface patterning [102,114,116,117,118,119,120,121,122,123,124,125,126,127,128,129] | Lithography Microfluidic Self-assembled Monolayers Transfer printing Stencil-assisted printing Nanopatterning | Quell non-specific protein–surface interactions Enhance endothelial cell attachment Encourage vessel healing Promote anti-thrombotic properties |
| Chemical modification [99,104,121,130,131,132,133,134,135,136,137,138] | Chemical vapor deposition Plasma vapor deposition Grafting techniques Self-assembled monolayers | Enhance the functionality of the surface |
| Surface coatings and films [139,140,141,142,143] | Wet/solvent coating Langmuir-Blodgett films | Increase endothelial cell attachment Reduce blood coagulation and thrombosis |
| Attachment of pharmaceuticals or biopharmaceuticals to the surface [104,131,144,145,146,147,148,149,150,151] | Chemical vapor deposition Wet chemical surface modification Plasma treatment Nitric oxide or thrombomodulin Layer by layer Polypyrrole composites | Control cell behavior Direct cell signaling |
| Porous surfaces to facilitate drug delivery [152,153,154,155] | Drugs attached directly | Stimulate vessel healing Better incorporation with body |
| Company | Stent | Base Material | Strut Thickness (µm) | Stent Diameter (mm) | Degradation Time (Months) | Polymer-Based Coating |
|---|---|---|---|---|---|---|
| Meril Medical | MeRes | PLLA | >200 | 3.0 | 24 | - |
| MeRes | PLLA | 100 | 2.5, 3.0, 3.5 | - | PDLLA | |
| Amaranth | FORTITUDE | PLLA | 150–200 | 2.75 | 3–6 | - |
| Huaan Biotechnology Group | XINSORB | PLLA | 160 | 3.0 | 24–36 | PDLLA/PLLA |
| Manli Cardiology | Mirage | PLLA | 125–150 | 3.0-3.5 | 14 | PLA |
| Arterius | ArterioSorb 120 | PLLA | 120 | - | - | PDLA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Jiao, L.; Pang, S.; Yan, P.; Wang, X.; Qiu, T. The Development of Design and Manufacture Techniques for Bioresorbable Coronary Artery Stents. Micromachines 2021, 12, 990. https://doi.org/10.3390/mi12080990
Wang L, Jiao L, Pang S, Yan P, Wang X, Qiu T. The Development of Design and Manufacture Techniques for Bioresorbable Coronary Artery Stents. Micromachines. 2021; 12(8):990. https://doi.org/10.3390/mi12080990
Chicago/Turabian StyleWang, Liang, Li Jiao, Shuoshuo Pang, Pei Yan, Xibin Wang, and Tianyang Qiu. 2021. "The Development of Design and Manufacture Techniques for Bioresorbable Coronary Artery Stents" Micromachines 12, no. 8: 990. https://doi.org/10.3390/mi12080990
APA StyleWang, L., Jiao, L., Pang, S., Yan, P., Wang, X., & Qiu, T. (2021). The Development of Design and Manufacture Techniques for Bioresorbable Coronary Artery Stents. Micromachines, 12(8), 990. https://doi.org/10.3390/mi12080990





