Evolution of Brains and Computers: The Roads Not Taken
Abstract
1. Introduction
2. Contingent versus Convergent Evolution
Some traits of human thinking will be common (as common as bilateral symmetry, segmentation, and tubular guts are in biology), but the possibility space of viable minds will likely contain traits far outside what we have evolved. It is not necessary that this type of thinking be faster than humans, greater, or deeper. In some cases it will be simpler. Our most important machines are not machines that do what humans do better, but machines that can do things we can’t do at all. Our most important thinking machines will not be machines that can think what we think faster, better, but those that think what we can’t think.
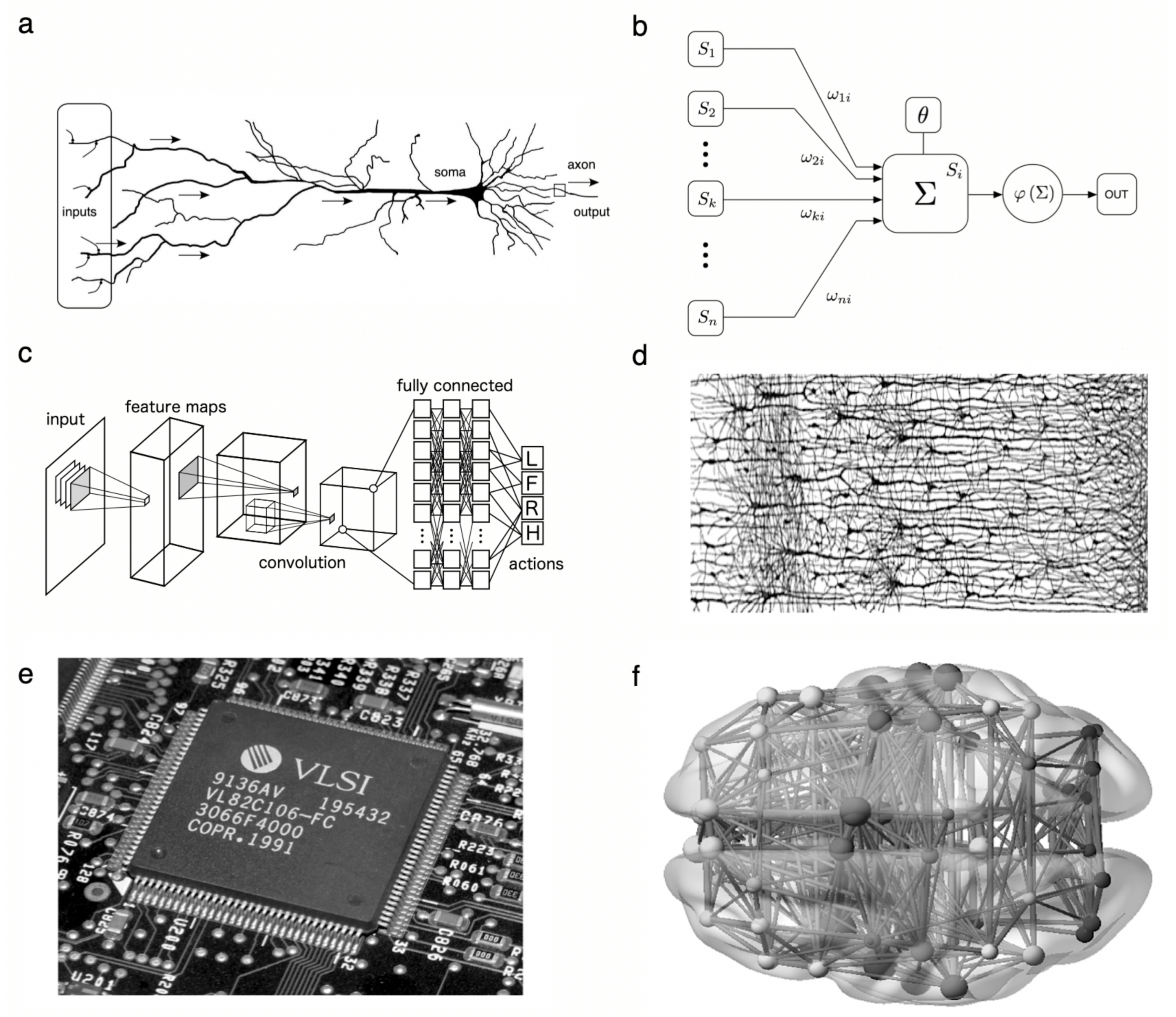
2.1. Threshold Units
2.2. Hierarchical Processing of Sensory Inputs
2.3. Wiring Cost Universals
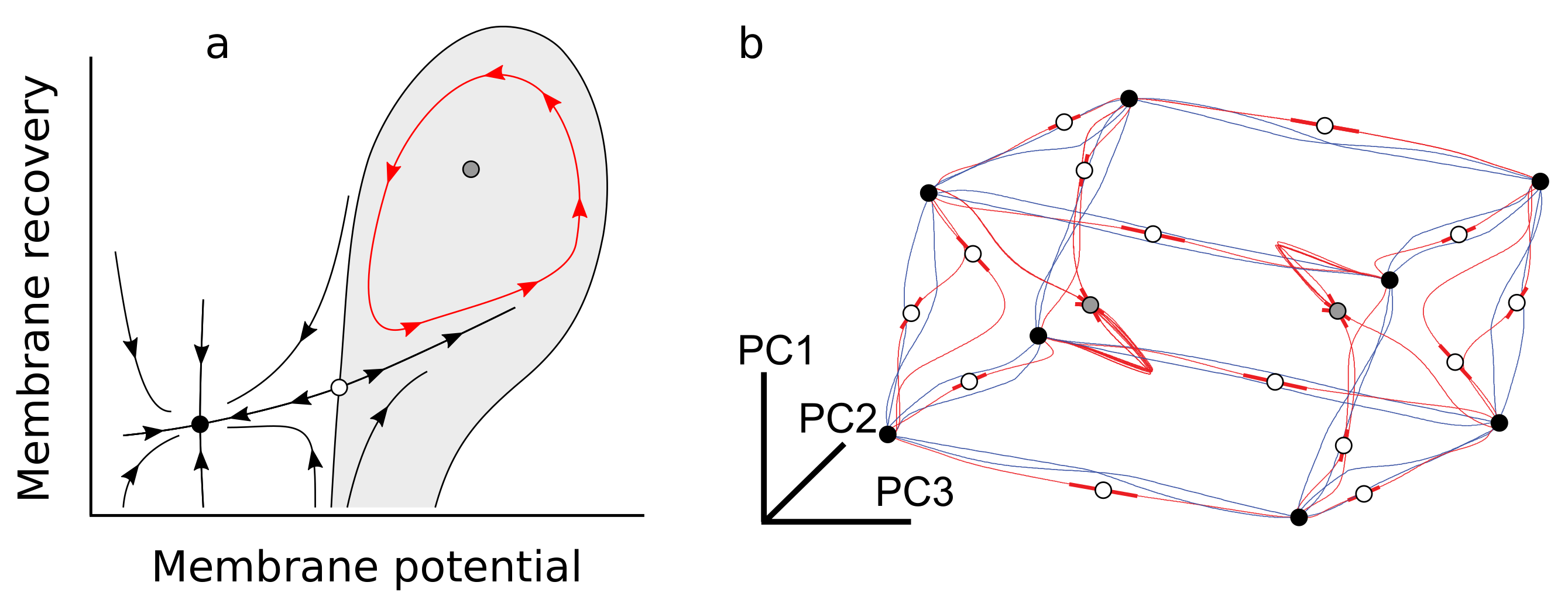
2.4. A Few Building Blocks of Dynamical Systems Enable Complex Cognition
2.5. Learning, Learning to Learn—Meta-Learning
3. The Gap
3.1. Language
3.2. Time Travel
3.3. Mind Reading
3.4. Right from Wrong
3.5. Extended Mind
3.6. Social Learning
4. A Space of Cognitive Complexity
- Computational complexity: This needs to be understood as some measure over the tasks performed by each kind of agent. That would include memory, learning, decision making, and other cognitive traits.
- Degree of autonomy: This is a crucial attribute of adaptive complexity. We can define autonomy as “the property of a system that builds and actively maintains the rules that define itself, as well as the way it behaves in the world” [183].
- Interactions between agents: This third and no less relevant dimension might enable cognition capabilities that transcend the individual. Tight interactions between agents might be a pre-requisite for (or a consequence of) eusociality [184], as they might enable a switch of the selective focus of Darwinian selection.
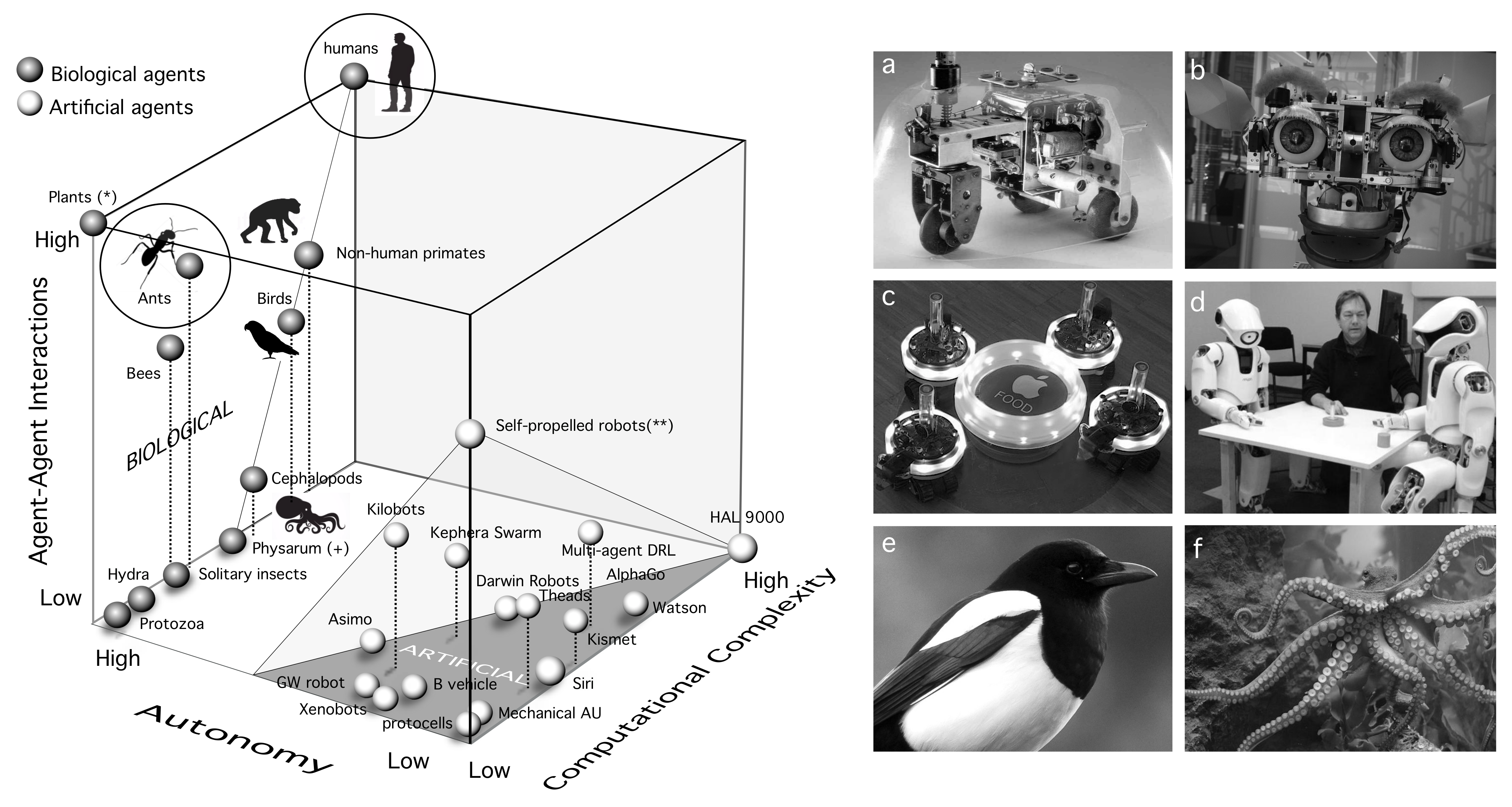
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Levin, M.; Dennett, D.C. Cognition All the Way Down. Aeon Essays. 2020. Available online: https://ase.tufts.edu/cogstud/dennett/papers/Levin_Dennett_Aeon.pdf (accessed on 2 May 2022).
- Friston, K. Does predictive coding have a future? Nat. Neurosci. 2018, 21, 1019–1021. [Google Scholar] [CrossRef] [PubMed]
- Jacob, F. Of Flies, Mice, and Men; Harvard University Press: Cambridge, MA, USA, 1998. [Google Scholar]
- Rao, R.P.; Ballard, D.H. Predictive coding in the visual cortex: A functional interpretation of some extra-classical receptive-field effects. Nat. Neurosci. 1999, 2, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Seoane, L.F.; Solé, R.V. Information theory, predictability and the emergence of complex life. R. Soc. Open Sci. 2018, 5, 172221. [Google Scholar] [CrossRef] [PubMed]
- Llinas, R.R. I of the Vortex: From Neurons to Self; MIT Press: Cambridge, MA, USA, 2001. [Google Scholar]
- The Cambrian Explosion; Roberts and Company: Genwodd, VL, USA, 2013.
- DeFelipe, J. The evolution of the brain, the human nature of cortical circuits, and intellectual creativity. Front. Neuroanat. 2011, 5, 29. [Google Scholar] [CrossRef] [PubMed]
- Yuste, R. From the neuron doctrine to neural networks. Nat. Rev. Neurosci. 2015, 16, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Valverde, S. Major transitions in information technology. Phil. Trans. R. Soc. B 2016, 371, 20150450. [Google Scholar] [CrossRef] [PubMed]
- Von Neumann, J. The Computer and the Brain; Yale University Press: London, UK, 1958. [Google Scholar]
- Ackley, D.H.; Hinton, G.E.; Sejnowski, T.J. A learning algorithm for Boltzmann machines. Cognitive Sci. 1985, 9, 147–169. [Google Scholar] [CrossRef]
- Hopfield, J.J. Neural networks and physical systems with emergent collective computational abilities. Proc. Natl. Acad. Sci. USA 1982, 79, 2554–2558. [Google Scholar] [CrossRef]
- Kelleher, J.D. Deep Learning; MIT Press: Cambridge, MA, USA, 2019. [Google Scholar]
- Mitchell, M. Artificial Intelligence: A Guide for Thinking Humans; Penguin: London, UK, 2019. [Google Scholar]
- Qu, X.; Sun, Z.; Ong, Y.S.; Gupta, A.; Wei, P. Minimalistic attacks: How little it takes to fool deep reinforcement learning policies. IEEE Trans. Cogn. Develop. Syst. 2020, 13, 806–817. [Google Scholar] [CrossRef]
- Kelly, K. What Do You Think about Machines that Think? 2015. Available online: https://www.edge.org/response-detail/26097 (accessed on 2 May 2022).
- Solé, R.V. Synthetic transitions: Towards a new synthesis. Phil. Trans. R. Soc. B 2016, 371, 20150438. [Google Scholar] [CrossRef]
- Gould, S.J. Wonderful Life: The Burgess Shale and the Nature of History; WW Norton & Company: New York, NY, USA, 1990. [Google Scholar]
- Lane, N. Life Ascending: The Ten Great Inventions of Evolution; Norton and Co.: New York, NY, USA, 2009. [Google Scholar]
- McGhee, G.R. Convergent Evolution: Limited Forms Most Beautiful; MIT Press: Cambridge, MA, USA, 2011. [Google Scholar]
- Emery, N.J.; Clayton, N.S. The mentality of crows: Convergent evolution of intelligence in corvids and apes. Science 2004, 306, 1903–1907. [Google Scholar] [CrossRef]
- Prior, H.; Schwarz, A.; Güntürkün, O. Mirror-induced behavior in the magpie (Pica pica): Evidence of self-recognition. PLoS Biol. 2008, 6, e202. [Google Scholar] [CrossRef]
- Evolutionary convergence and biologically embodied cognition. Interface Focus 2017, 7, 20160123. [CrossRef]
- Powell, R.; Mikhalevich, I.; Logan, C.; Clayton, N.S. Convergent minds: The evolution of cognitive complexity in nature. Interface Focus 2017, 7, 20170029. [Google Scholar] [CrossRef]
- Van Duijn, M. Phylogenetic origins of biological cognition: Convergent patterns in the early evolution of learning. Interface Focus 2017, 7, 20160158. [Google Scholar] [CrossRef]
- Morris, S.C. Life’s Solution: Inevitable Humans in a Lonely Universe; Cambridge University Press: Cambridge, UK, 2003. [Google Scholar]
- Godfrey-Smith, P. Other Minds: The Octopus and the Evolution of Intelligent Life; William Collins: London, UK, 2016. [Google Scholar]
- Shannon, C.E. A symbolic analysis of relay and switching circuits. Electr. Eng. 1938, 57, 713–723. [Google Scholar] [CrossRef]
- McCulloch, W.S.; Pitts, W. A logical calculus of the ideas immanent in nervous activity. Bull. Math. Biophys. 1943, 5, 115–133. [Google Scholar] [CrossRef]
- Rashevsky, N. The neural mechanism of logical thinking. Bull. Math. Biophys. 1946, 8, 29–40. [Google Scholar] [CrossRef]
- Rashevsky, N. Mathematical Biophysics: Physico-Mathematical Foundations of Biology, 3rd ed.; Dover P., Inc.: New York, NY, USA, 1960. [Google Scholar]
- Pitts, W. Some observations on the simple neuron circuit. Bull. Math. Biophys. 1942, 4, 121–129. [Google Scholar] [CrossRef]
- Gidon, A.; Zolnik, T.A.; Fidzinski, P.; Bolduan, F.; Papoutsi, A.; Poirazi, P.; Holtkamp, M.; Vida, I.; Larkum, M.E. Dendritic action potentials and computation in human layer 2/3 cortical neurons. Science 2020, 367, 83–87. [Google Scholar] [CrossRef]
- Hertz, J.; Krogh, A.; Palmer, R.G. Introduction to the Theory of Neural Computation. Volume I; Addison-Wesley: Redwood City, CA, USA, 1991. [Google Scholar]
- Peretto, P. An Introduction to the Modeling of Neural Networks; Cambridge University Press: Cambridge, UK, 1992. [Google Scholar]
- Rojas, R. Neural Networks: A Systematic Introduction; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Bornholdt, S. Boolean network models of cellular regulation: Prospects and limitations. J. R. Soc. Interface 2008, 5, S85–S94. [Google Scholar] [CrossRef]
- Glass, L.; Kauffman, S.A. The logical analysis of continuous, non-linear biochemical control networks. J. Theor. Biol. 1973, 39, 103–129. [Google Scholar] [CrossRef]
- Kurten, K.E. Correspondence between neural threshold networks and Kauffman Boolean cellular automata. J. Phys. A 1988, 21, L615. [Google Scholar] [CrossRef]
- Luque, B.; Solé, R.V. Phase transitions in random networks: Simple analytic determination of critical points. Phys. Rev. E 1997, 55, 257. [Google Scholar] [CrossRef]
- Hubel, D.H.; Wiesel, T.N. Receptive fields of single neurones in the cat’s striate cortex. J. Physiol. 1959, 148, 574. [Google Scholar] [CrossRef]
- Hubel, D.H.; Wiesel, T.N. Receptive fields, binocular interaction and functional architecture in the cat’s visual cortex. J. Physiol. 1962, 160, 106. [Google Scholar] [CrossRef]
- Churchl, P.S.; Sejnowski, T.J. The Computational Brain; MIT Press: Cambridge, MA, USA, 1994. [Google Scholar]
- Livingstone, M.; Hubel, D. Segregation of form, color, movement, and depth: Anatomy, physiology, and perception. Science 1988, 240, 740–749. [Google Scholar] [CrossRef]
- Rolls, E.T.; Deco, G. Computational Neuroscience of Vision; Oxford University Press: Oxford, UK, 2007. [Google Scholar]
- Levick, W.R. Receptive fields and trigger features of ganglion cells in the visual streak of the rabbit’s retina. J. Physiol. 1967, 188, 285. [Google Scholar] [CrossRef]
- Russell, T.L.; Werblin, F.S. Retinal synaptic pathways underlying the response of the rabbit local edge detector. J. Neurophysiol. 2010, 103, 2757–2769. [Google Scholar] [CrossRef]
- Marr, D.; Hildreth, E. Theory of edge detection. Ser. B Biol. Sci. 1980, 207, 187–217. [Google Scholar]
- Marr, D. Vision: A Computational Investigation into the Human Representation and Processing of Visual Information; MIT Press: Cambridge, MA, USA, 1982. [Google Scholar]
- Fukushima, K. Neocognitron: A hierarchical neural network capable of visual pattern recognition. Neural Netw. 1988, 1, 119–130. [Google Scholar] [CrossRef]
- Krizhevsky, A.; Sutskever, I.; Hinton, G.E. Imagenet classification with deep convolutional neural networks. Adv. Neural Inf. Process. Syst. 2012, 25, 1–9. [Google Scholar] [CrossRef]
- Yamins, D.L.; DiCarlo, J.J. Using goal-driven deep learning models to understand sensory cortex. Nat. Neurosci. 2016, 19, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.E.; Bower, J.M. Brain maps and parallel computers. Trends Neurosci. 1990, 13, 403–408. [Google Scholar] [CrossRef]
- Khaligh-Razavi, S.M.; Kriegeskorte, N. Deep supervised, but not unsupervised, models may explain IT cortical representation. PLoS Comput. Biol. 2014, 10, e1003915. [Google Scholar] [CrossRef]
- Schrimpf, M.; Kubilius, J.; Hong, H.; Majaj, N.J.; Rajalingham, R.; Issa, E.B.; Kar, K.; Bashivan, P.; Prescott-Roy, J.; Geiger, F.; et al. Brain-score: Which artificial neural network for object recognition is most brain-like? BioRxiv 2020, 407007. [Google Scholar] [CrossRef]
- Yamins, D.L.; Hong, H.; Cadieu, C.F.; Solomon, E.A.; Seibert, D.; DiCarlo, J.J. Performance-optimized hierarchical models predict neural responses in higher visual cortex. Proc. Nat. Acad. Sci. USA 2014, 111, 8619–8624. [Google Scholar] [CrossRef]
- Zhuang, C.; Yan, S.; Nayebi, A.; Schrimpf, M.; Frank, M.C.; DiCarlo, J.J.; Yamins, D.L. Unsupervised neural network models of the ventral visual stream. Proc. Nat. Acad. Sci. USA 2021, 118, e2014196118. [Google Scholar] [CrossRef]
- Kell, A.J.; Yamins, D.L.; Shook, E.N.; Norman-Haignere, S.V.; McDermott, J.H. A task-optimized neural network replicates human auditory behavior, predicts brain responses, and reveals a cortical processing hierarchy. Neuron 2018, 98, 630–644. [Google Scholar] [CrossRef]
- Caucheteux, C.; King, J.R. Brains and algorithms partially converge in natural language processing. Commun. Biol. 2022, 5, 1–10. [Google Scholar] [CrossRef]
- Thiry, L.; Arbel, M.; Belilovsky, E.; Oyallon, E. The unreasonable effectiveness of patches in deep convolutional kernels methods. arXiv 2021, arXiv:2101.07528. [Google Scholar]
- Trockman, A.; Kolter, J.Z. Patches Are All You Need? arXiv 2022, arXiv:2201.09792. [Google Scholar]
- Stephens, G.J.; Mora, T.; Tkačik, G.; Bialek, W. Statistical thermodynamics of natural images. Phys. Rev. Let. 2013, 110, 018701. [Google Scholar] [CrossRef]
- Seoane, L.F.; Solé, R. Criticality in pareto optimal grammars? Entropy 2020, 22, 165. [Google Scholar] [CrossRef]
- Christie, P.; Stroob, T.D. The interpretation and application of Rent’s rule. IEEE T. VLSI Syst. 2000, 8, 639–648. [Google Scholar] [CrossRef]
- Bassett, D.S.; Greenfield, D.L.; Meyer-Lindenberg, A.; Weinberger, D.R.; Moore, S.W.; Bullmore, E.T. Efficient physical embedding of topologically complex information processing networks in brains and computer circuits. PLoS Comput. Biol. 2010, 6, e1000748. [Google Scholar] [CrossRef]
- Moses, M.E.; Forrest, S.; Davis, A.L.; Lodder, M.A.; Brown, J.H. Scaling theory for information networks. J. Royal Soc. Interface 2008, 5, 1469–1480. [Google Scholar] [CrossRef]
- Moses, M.; Bezerra, G.; Edwards, B.; Brown, J.; Forrest, S. Energy and time determine scaling in biological and computer designs. Phil. Trans. R.Soc. B 2016, 371, 20150446. [Google Scholar] [CrossRef]
- Howard, A.S.; Ler, M.; Chu, G.; Chen, L.C.; Chen, B.; Tan, M.; Wang, W.; Zhu, Y.; Pang, R.; Vasudevan, V.; et al. Searching for mobilenetv3. In Proceedings of the 2019 IEEE/CVF International Conference on Computer Vision (ICCV), Seoul, Korea, 27 October–2 November 2019; pp. 1314–1324. [Google Scholar]
- Mirhoseini, A.; Goldie, A.; Yazgan, M.; Jiang, J.W.; Songhori, E.; Wang, S.; Lee, Y.J.; Johnson, E.; Pathak, O.; Nazi, A.; et al. A graph placement methodology for fast chip design. Nature 2021, 594, 207–212. [Google Scholar] [CrossRef]
- Sussillo, D.; Barak, O. Opening the black box: Low-dimensional dynamics in high-dimensional recurrent neural networks. Neural Comput. 2013, 25, 626–649. [Google Scholar] [CrossRef]
- Izhikevich, E.M. Dynamical Systems in Neuroscience; MIT Press: Cambridge, MA, USA, 2007. [Google Scholar]
- Hodgkin, A.L. The local electric changes associated with repetitive action in a non-medullated axon. J. Physiol. 1948, 107, 165. [Google Scholar] [CrossRef] [PubMed]
- Hodgkin, A.L.; Huxley, A.F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J. Physiol. 1952, 117, 500. [Google Scholar] [CrossRef] [PubMed]
- FitzHugh, R. Impulses and physiological states in theoretical models of nerve membrane. Biophys. J. 1961, 1, 445–466. [Google Scholar] [CrossRef]
- Izhikevich, E.M. Neural excitability, spiking and bursting. Int. J. Bifurcat. Chaos 2000, 10, 1171–1266. [Google Scholar] [CrossRef]
- Nagumo, J.; Arimoto, S.; Yoshizawa, S. An active pulse transmission line simulating nerve axon. Proc. IRE 1962, 50, 2061–2070. [Google Scholar] [CrossRef]
- Rinzel, J.; Ermentrout, G.B. Analysis of Neural Excitability and Oscillations. In Methods in Neuronal Modeling; Koch, C., Segev, I., Eds.; MIT Press: Cambridge, MA, USA, 1998; pp. 251–292. [Google Scholar]
- Gallego, J.A.; Perich, M.G.; Miller, L.E.; Solla, S.A. Neural manifolds for the control of movement. Neuron 2017, 94, 978–984. [Google Scholar] [CrossRef]
- Gardner, R.J.; Hermansen, E.; Pachitariu, M.; Burak, Y.; Baas, N.A.; Dunn, B.A.; Moser, M.B.; Moser, E.I. Toroidal topology of population activity in grid cells. Nature 2022, 602, 123–128. [Google Scholar] [CrossRef]
- Gao, P.; Ganguli, S. On simplicity and complexity in the brave new world of large-scale neuroscience. Curr. Opin. Neurobiol. 2015, 32, 148–155. [Google Scholar] [CrossRef]
- Kaufman, M.T.; Churchl, M.M.; Ryu, S.I.; Shenoy, K.V. Cortical activity in the null space: Permitting preparation without movement. Nature Neurosci. 2014, 17, 440–448. [Google Scholar] [CrossRef]
- Mante, V.; Sussillo, D.; Shenoy, K.V.; Newsome, W.T. Context-dependent computation by recurrent dynamics in prefrontal cortex. Nature 2013, 503, 78–84. [Google Scholar] [CrossRef]
- Kandel, E.R. In Search of Memory: The Emergence of a New Science of Mind; WW Norton & Company: New York, NY, USA, 2007. [Google Scholar]
- Hebb, D.O. The Organization of Behavior; Wiley & Sons: New York, NY, USA, 1949. [Google Scholar]
- Rumelhart, D.E.; Hinton, G.E.; Williams, R.J. Learning Internal Representations by Error Propagation; Technical Report (DTIC Document); 1985. Available online: https://web.stanford.edu/class/psych209a/ReadingsByDate/02_06/PDPVolIChapter8.pdf (accessed on 2 May 2022).
- Lillicrap, T.P.; Santoro, A.; Marris, L.; Akerman, C.J.; Hinton, G. Backpropagation and the brain. Nat. Rev. Neurosci. 2020, 21, 335–346. [Google Scholar] [CrossRef]
- Mnih, V.; Kavukcuoglu, K.; Silver, D.; Rusu, A.A.; Veness, J.; Bellemare, M.G.; Graves, A.; Riedmiller, M.; Fidjel, A.K.; Ostrovski, G.; et al. Human-level control through deep reinforcement learning. Nature 2015, 518, 529–533. [Google Scholar] [CrossRef]
- Moravčík, M.; Schmid, M.; Burch, N.; Lisý, V.; Morrill, D.; Bard, N.; Davis, T.; Waugh, K.; Johanson, M.; Bowling, M. Deepstack: Expert-level artificial intelligence in heads-up no-limit poker. Science 2017, 356, 508–513. [Google Scholar] [CrossRef]
- Silver, D.; Huang, A.; Maddison, C.J.; Guez, A.; Sifre, L.; Van Den Driessche, G.; Schrittwieser, J.; Antonoglou, I.; Panneershelvam, V.; Lanctot, M.; et al. Mastering the game of Go with deep neural networks and tree search. Nature 2016, 529, 484–489. [Google Scholar] [CrossRef]
- Silver, D.; Schrittwieser, J.; Simonyan, K.; Antonoglou, I.; Huang, A.; Guez, A.; Hubert, T.; Baker, L.; Lai, M.; Bolton, A.; et al. Mastering the game of go without human knowledge. Nature 2017, 550, 354–359. [Google Scholar] [CrossRef]
- Bellec, G.; Scherr, F.; Subramoney, A.; Hajek, E.; Salaj, D.; Legenstein, R.; Maass, W. A solution to the learning dilemma for recurrent networks of spiking neurons. Nat. Commun. 2020, 11, 3625. [Google Scholar] [CrossRef]
- Lillicrap, T.P.; Cownden, D.; Tweed, D.B.; Akerman, C.J. Random synaptic feedback weights support error backpropagation for deep learning. Nat. Commun. 2016, 7, 13276. [Google Scholar] [CrossRef]
- Sutton, R.S.; Barto, A.G. Reinforcement Learning: An Introduction; MIT Press: Cambridge, MA, USA, 2018. [Google Scholar]
- Foster, D.J. Replay comes of age. Annu. Rev. Neurosci. 2017, 40, 581–602. [Google Scholar] [CrossRef]
- Foster, D.J.; Wilson, M.A. Reverse replay of behavioural sequences in hippocampal place cells during the awake state. Nature 2006, 440, 680–683. [Google Scholar] [CrossRef]
- Penagos, H.; Varela, C.; Wilson, M.A. Oscillations, neural computations and learning during wake and sleep. Curr. Opin. Neurobiol. 2017, 44, 193–201. [Google Scholar] [CrossRef]
- Subramoney, A.; Scherr, F.; Maass, W. Reservoirs learn to learn. In Reservoir Computing; Springer: Singapore, 2021; pp. 59–76. [Google Scholar]
- Wang, J.X.; Kurth-Nelson, Z.; Kumaran, D.; Tirumala, D.; Soyer, H.; Leibo, J.Z.; Hassabis, D.; Botvinick, M. Prefrontal cortex as a meta-reinforcement learning system. Nat. Neurosci. 2018, 21, 860–868. [Google Scholar] [CrossRef]
- Suddendorf, T. The Gap: The Science of What Separates Us from Other Animals; Basic Books: New York, NY, USA, 2013. [Google Scholar]
- Niyogi, P. The Computational Nature of Language Learning and Evolution; MIT Press: Cambridge, MA, USA, 2006. [Google Scholar]
- Solé, R.V.; Corominas-Murtra, B.; Valverde, S.; Steels, L. Language networks: Their structure, function, and evolution. Complexity 2010, 15, 20–26. [Google Scholar] [CrossRef]
- Dehaene, S.; Meyniel, F.; Wacongne, C.; Wang, L.; Pallier, C. The neural representation of sequences: From transition probabilities to algebraic patterns and linguistic trees. Neuron 2015, 88, 2–19. [Google Scholar] [CrossRef]
- Bickerton, D. Language and Species; University of Chicago Press: Chicago, IL, USA, 1990. [Google Scholar]
- Berwick, R.C.; Chomsky, N. Why Only Us: Language and Evolution; MIT Press: Cambridge, MA, USA, 2016. [Google Scholar]
- Hauser, M.D.; Chomsky, N.; Fitch, W.T. The faculty of language: What is it, who has it, and how did it evolve? Science 2002, 298, 1569–1579. [Google Scholar] [CrossRef]
- Bickerton, D. More Than Nature Needs; Harvard University Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Arbib, M.A. From monkey-like action recognition to human language: An evolutionary framework for neurolinguistics. Behav. Brain Sci. 2005, 28, 105–124. [Google Scholar] [CrossRef]
- Corballis, M.C. Language as gesture. Hum. Movement Sci. 2009, 28, 556–565. [Google Scholar] [CrossRef]
- Rizzolatti, G.; Arbib, M.A. Language within our grasp. Trends Neurosci. 1998, 21, 188–194. [Google Scholar] [CrossRef]
- Koelsch, S. Neural substrates of processing syntax and semantics in music. In Music that Works; Springer Science + Business: New York, NY, USA, 2009; pp. 143–153. [Google Scholar]
- Armstrong, D.F.; Stokoe, W.C.; Wilcox, S.E. Gesture and the Nature of Language; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- De Heer, W.A.; Huth, A.G.; Griffiths, T.L.; Gallant, J.L.; Theunissen, F.E. The hierarchical cortical organization of human speech processing. J. Neurosci. 2017, 37, 6539–6557. [Google Scholar] [CrossRef]
- Huth, A.G.; Nishimoto, S.; Vu, A.T.; Gallant, J.L. A continuous semantic space describes the representation of thousands of object and action categories across the human brain. Neuron 2012, 76, 1210–1224. [Google Scholar] [CrossRef]
- Blank, I.; Balewski, Z.; Mahowald, K.; Fedorenko, E. Syntactic processing is distributed across the language system. Neuroimage 2016, 127, 307–323. [Google Scholar] [CrossRef]
- Catani, M.; Jones, D.K.; Ffytche, D.H. Perisylvian language networks of the human brain. Ann. Neurol. 2005, 57, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Fedorenko, E.; Kanwisher, N. Neuroimaging of language: Why hasn’t a clearer picture emerged? Lang. Linguist. 2009, 3, 839–865. [Google Scholar] [CrossRef]
- Fedorenko, E.; Nieto-Castanon, A.; Kanwisher, N. Lexical and syntactic representations in the brain: An fMRI investigation with multi-voxel pattern analyses. Neuropsychologia 2012, 50, 499–513. [Google Scholar] [CrossRef] [PubMed]
- Fedorenko, E.; Thompson-Schill, S.L. Reworking the language network. Trends Cogn. Sci. 2014, 18, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Geschwind, N. Language and the brain. Sci. Am. 1972, 226, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Ayyash, D.; Malik-Moraleda, S.; Gallée, J.; Affourtit, J.; Hoffman, M.; Mineroff, Z.; Jouravlev, O.; Fedorenko, E. The universal language network: A cross-linguistic investigation spanning 45 languages and 11 language families. bioRxiv 2022. [Google Scholar] [CrossRef]
- Chomsky, N. Knowledge of Language: Its Nature, Origin, and Use; Greenwood Publishing Group: Westport, CT, USA, 1986. [Google Scholar]
- Gibson, E.; Futrell, R.; Piantadosi, S.P.; Dautriche, I.; Mahowald, K.; Bergen, L.; Levy, R. How efficiency shapes human language. Trends Cogn. Sci. 2019, 23, 389–407. [Google Scholar] [CrossRef]
- Evans, N.; Levinson, S.C. The myth of language universals: Language diversity and its importance for cognitive science. Behav. Brain Sci. 2009, 32, 429–448. [Google Scholar] [CrossRef]
- Solé, R.; Seoane, L.F. Ambiguity in language networks. Linguist. Rev. 2015, 32, 5–35. [Google Scholar] [CrossRef]
- Ferrer i Cancho, R.; Solé, R.V. Least effort and the origins of scaling in human language. Proc. Natl. Acad. Sci. USA 2003, 100, 788–791. [Google Scholar] [CrossRef]
- Seoane, L.F.; Solé, R. The morphospace of language networks. Sci. Rep. 2018, 8, 1–14. [Google Scholar] [CrossRef]
- Fellbaum, C. (Ed.) WordNet: An Electronic Lexical Database; MIT Press: Cambridge, MA, USA, 1998. [Google Scholar]
- Miller, G.A. WordNet: A lexical database for English. Commun. ACM 1995, 38, 39–41. [Google Scholar] [CrossRef]
- Goñi, J.; Arrondo, G.; Sepulcre, J.; Martincorena, I.; Vélez de Mendizábal, N.; Corominas-Murtra, B.; Bejarano, B.; Ardanza-Trevijano, S.; Peraita, H.; Wall, D.P.; et al. The semantic organization of the animal category: Evidence from semantic verbal fluency and network theory. Cogn. Process. 2011, 12, 183–196. [Google Scholar] [CrossRef]
- Steyvers, M.; Tenenbaum, J.B. The large-scale structure of semantic networks: Statistical analyses and a model of semantic growth. Cognitive Sci. 2005, 29, 41–78. [Google Scholar] [CrossRef]
- Motter, A.E.; De Moura, A.P.; Lai, Y.C.; Dasgupta, P. Topology of the conceptual network of language. Phys. Rev. E 2002, 65, 065102. [Google Scholar] [CrossRef]
- Steels, L. The Talking Heads Experiment. 1999. Available online: https://langsci-press.org/catalog/book/49 (accessed on 2 May 2022).
- Steels, L. The Talking Heads Experiment: Words and Meanings; VUB Artificial Intelligence Laboratory: Brussels, Belgium, 1999. [Google Scholar]
- Steels, L. Evolving grounded communication for robots. Trends Cogn. Sci. 2003, 7, 308–312. [Google Scholar] [CrossRef]
- Steels, L. Agent-based models for the emergence and evolution of grammar. Philos. Trans. R. Soc. B 2016, 371, 20150447. [Google Scholar] [CrossRef]
- Steels, L. (Ed.) Design Patterns in Fluid Construction Grammar; John Benjamins Publishin: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Buonomano, D. Your Brain Is a Time Machine: The Neuroscience and Physics of Time; WW Norton & Company: New York, NY, USA, 2017. [Google Scholar]
- Roberts, W.A. Are animals stuck in time? Psychol. Bull. 2002, 128, 473. [Google Scholar] [CrossRef]
- Suddendorf, T.; Corballis, M.C. The evolution of foresight: What is mental time travel, and is it unique to humans? Behav. Brain Sci. 2013, 30, 299–313. [Google Scholar] [CrossRef]
- Schacter, D.L.; Addis, D.R.; Buckner, R.L. Remembering the past to imagine the future: The prospective brain. Nat. Rev. Neurosci. 2007, 8, 657–661. [Google Scholar] [CrossRef]
- Maniadakis, M.; Trahanias, P. Temporal cognition: A key ingredient of intelligent systems. Front. Neurorobotics 2011, 5, 2. [Google Scholar] [CrossRef]
- Elman, J.L. Finding structure in time. Cogn. Sci. 1990, 14, 179–211. [Google Scholar] [CrossRef]
- Ha, D.; Schmidhuber, J. World models. arXiv 2018, arXiv:1803.10122. [Google Scholar]
- Kaiser, L.; Babaeizadeh, M.; Milos, P.; Osinski, B.; Campbell, R.H.; Czechowski, K.; Erhan, D.; Finn, C.; Kozakowski, P.; Levine, S.; et al. Model-based reinforcement learning for atari. arXiv 2019, arXiv:1903.00374. [Google Scholar]
- Kanwisher, N.; McDermott, J.; Chun, M.M. The fusiform face area: A module in human extrastriate cortex specialized for face perception. J. Neurosci. 1997, 17, 4302–4311. [Google Scholar] [CrossRef]
- Ramachandran, V.S. The Tell-Tale Brain: Unlocking the Mystery of Human Nature; Random House: New York, NY, USA, 2012. [Google Scholar]
- Rizzolatti, G.; Fadiga, L.; Gallese, V.; Fogassi, L. Premotor cortex and the recognition of motor actions. Cognit. Brain Res. 1996, 3, 131–141. [Google Scholar] [CrossRef]
- Tomasello, M.; Vaish, A. Origins of human cooperation and morality. Ann. Rev. Psychol. 2013, 64, 231–255. [Google Scholar] [CrossRef]
- Dennett, D.C. Darwin’s Dangerous Idea; Simon & Schuster: New York, NY, USA, 1995. [Google Scholar]
- Harris, S. The Moral Landscape: How Science Can Determine Human Values; Simon and Schuster: New York, NY, USA, 2011. [Google Scholar]
- Gordon, J.S. Building moral robots: Ethical pitfalls and challenges. Sci. Eng. Ethics 2020, 26, 141–157. [Google Scholar] [CrossRef]
- Wallach, W.; Allen, C. Moral Machines: Teaching Robots Right from Wrong; Oxford University Press: Oxford, UK, 2008. [Google Scholar]
- Clark, A.; Chalmers, D. The extended mind. Analysis 1998, 58, 7–19. [Google Scholar] [CrossRef]
- Japayasu, H.F.; Lal, K.N. Extended spider cognition. Anim. Cogn. 2017, 20, 375–395. [Google Scholar] [CrossRef]
- Bonabeau, E.; Theraulaz, G.; Deneubourg, J.L.; Aron, S.; Camazine, S. Self-organization in social insects. Trends Ecol. Evol. 1997, 12, 188–193. [Google Scholar] [CrossRef]
- Gupta, A.; Savarese, S.; Ganguli, S.; Fei-Fei, L. Embodied intelligence via learning and evolution. Nat. Commun. 2021, 12, 5721. [Google Scholar] [CrossRef] [PubMed]
- Ha, D. Reinforcement learning for improving agent design. Artif. Life 2019, 25, 352–365. [Google Scholar] [CrossRef] [PubMed]
- Nakano, R.; Hilton, J.; Balaji, S.; Wu, J.; Ouyang, L.; Kim, C.; Hesse, C.; Jain, S.; Kosaraju, V.; Saunders, W.; et al. WebGPT: Browser-assisted question-answering with human feedback. arXiv 2021, arXiv:2112.09332. [Google Scholar]
- Schaal, S. Is imitation learning the route to humanoid robots? Trends Cogn. Sci. 1999, 3, 233–242. [Google Scholar] [CrossRef]
- Fong, T.; Nourbakhsh, I.; Dautenhahn, K. A survey of socially interactive robots. Robot. Auton. Syst. 2003, 42, 143–166. [Google Scholar] [CrossRef]
- Breazeal, C.; Brian, S. Robots that imitate humans. Trends Cog. Sci. 2002, 6, 481–487. [Google Scholar] [CrossRef]
- Breazeal, C. Toward sociable robots. Robot. Auton. Syst. 2003, 42, 167–175. [Google Scholar] [CrossRef]
- Arbib, M.; Ganesh, V.; Gasser, B. Dyadic brain modelling, mirror systems and the ontogenetic ritualization of ape gesture. Phil. Trans. R. Soc. B 2014, 369, 20130414. [Google Scholar] [CrossRef]
- Ramachandran, V.S. Mirror neurons and imitation learning as the driving force behind “the great leap forward” in human evolution. Edge 2000, 29. Available online: https://www.edge.org/conversation/mirror-neurons-and-imitation-learning-as-the-driving-force-behind-the-great-leap-forward-in-human-evolution (accessed on 2 May 2022).
- Ecoffet, A.; Lehman, J. Reinforcement learning under moral uncertainty. In Proceedings of the International Conference on Machine Learning, Virtual, 18–24 July 2021; pp. 2926–2936. [Google Scholar]
- Jaques, N.; Lazaridou, A.; Hughes, E.; Gulcehre, C.; Ortega, P.; Strouse, D.J.; Leibo, J.Z.; De Freitas, N. Social influence as intrinsic motivation for multi-agent deep reinforcement learning. In Proceedings of the International Conference on Machine Learning, Long Beach, CA, USA, 9–15 June 2019; pp. 3040–3049. [Google Scholar]
- Rabinowitz, N.; Perbet, F.; Song, F.; Zhang, C.; Eslami, S.A.; Botvinick, M. Machine theory of mind. In Proceedings of the International Conference on Machine Learning, Stockholm, Sweden, 10–15 July 2018; pp. 4218–4227. [Google Scholar]
- Raup, D.M. Geometric analysis of shell coiling: General problems. J. Paleontol. 1966, 40, 1178–1190. [Google Scholar]
- McGhee, G.R. The Geometry of Evolution: Adaptive Landscapes and Theoretical Morphospaces; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Niklas, K.J. Computer models of early land plant evolution. Annu. Rev. Earth Planet. Sci. 2004, 32, 47–66. [Google Scholar] [CrossRef]
- Tyszka, J. Morphospace of foraminiferal shells: Results from the moving reference model. Lethaia 2006, 39, 1–12. [Google Scholar] [CrossRef]
- Avena-Koenigsberger, A.; Goñi, J.; Solé, R.; Sporns, O. Network morphospace. J. R. Soc. Interface 2015, 12, 20140881. [Google Scholar] [CrossRef]
- Corominas-Murtra, B.; Goñi, J.; Solé, R.V.; Rodríguez-Caso, C. On the origins of hierarchy in complex networks. Proc. Nat. Acad. Sci. USA 2013, 110, 13316–13321. [Google Scholar] [CrossRef]
- Goñi, J.; Avena-Koenigsberger, A.; Velez de Mendizabal, N.; Van den Heuvel, M.P.; Betzel, R.F.; Sporns, O. Exploring the morphospace of communication efficiency in complex networks. PLoS ONE 2013, 8, e58070. [Google Scholar] [CrossRef]
- Arsiwalla, X.D.; Solé, R.; Moulin-Frier, C.; Herreros, I.; Sanchez-Fibla, M.; Verschure, P. The morphospace of consciousness. arXiv 2017, arXiv:1705.11190. [Google Scholar]
- Duong-Tran, D.; Abbas, K.; Amico, E.; Corominas-Murtra, B.; Dzemidzic, M.; Kareken, D.; Ventresca, M.; Goñi, J. A morphospace of functional configuration to assess configural breadth based on brain functional networks. Netw. Neurosci. 2021, 5, 666–688. [Google Scholar] [CrossRef]
- Ollé-Vila, A.; Seoane, L.F.; Solé, R. Ageing, computation and the evolution of neural regeneration processes. J. R. Soc. Interface 2020, 17, 20200181. [Google Scholar] [CrossRef]
- Seoane, L.F. Evolutionary aspects of reservoir computing. Philos. Trans. R. Soc. B 2019, 374, 20180377. [Google Scholar] [CrossRef]
- Seoane, L.F. Fate of Duplicated Neural Structures. Entropy 2020, 22, 928. [Google Scholar] [CrossRef] [PubMed]
- Seoane, L.F. Evolutionary paths to lateralization of complex brain functions. arXiv 2021, arXiv:2112.00221. [Google Scholar]
- Dennett, D.C. From Bacteria to Bach and Back: The Evolution of Minds; WW Norton & Company: New York, NY, USA, 2017. [Google Scholar]
- Ruiz-Mirazo, K.; Moreno, A. Autonomy in evolution: From minimal to complex life. Synthese 2012, 185, 21–52. [Google Scholar] [CrossRef]
- Wilson, E.O. The Social Conquest of Earth; W.W. Norton & Co.: New York, NY, USA, 2012. [Google Scholar]
- Dawkins, R. The Selfish Gene; Oxford University Press: Oxford, UK, 1976. [Google Scholar]
- Haig, D. From Darwin to Derrida: Selfish Genes, Social Selves, and the Meanings of Life; MIT Press: Cambridge, MA, USA, 2020. [Google Scholar]
- Seth, A.K.; McKinstry, J.L.; Edelman, G.M.; Krichmar, J.L. Visual binding through reentrant connectivity and dynamic synchronization in a brain-based device. Cereb. Cortex 2004, 14, 1185–1199. [Google Scholar] [CrossRef]
- Rubenstein, M.; Ahler, C.; Nagpal, R. A low cost scalable robot system for collective behaviors. In Proceedings of the 2012 IEEE International Conference on Robotics and Automation, St. Paul, MN, USA, 14–18 May 2012; pp. 3293–3298. [Google Scholar]
- Slavkov, I.; Carrillo-Zapata, D.; Carranza, N.; Diego, X.; Jansson, F.; Kaorp, J.; Hauert, S.; Sharpe, J. Morphogenesis in robot swarms. Sci. Robot. 2018, 3, eaau9178. [Google Scholar] [CrossRef]
- Deblais, A.; Barois, T.; Guerin, T.; Delville, P.H.; Vaudaine, R.; Lintuvuori, J.S.; Boudet, J.F.; Baret, J.C.; Kellay, H. Boundaries control collective dynamics of inertial self-propelled robots. Phys. Rev. Let. 2018, 120, 188002. [Google Scholar] [CrossRef]
- Pinero, J.; Solé, R. Statistical physics of liquid brains. Philos. Trans. R. Soc. B 2019, 374, 20180376. [Google Scholar] [CrossRef]
- Solé, R.; Moses, M.; Forrest, S. Liquid brains, solid brains. Philos. Trans. R. Soc. B 2019, 374, 20190040. [Google Scholar] [CrossRef]
- Baluška, F.; Levin, M. On having no head: Cognition throughout biological systems. Front. Psychol. 2016, 7, 902. [Google Scholar] [CrossRef]
- Werfel, J.; Petersen, K.; Nagpal, R. Designing collective behavior in a termite-inspired robot construction team. Science 2014, 343, 754–758. [Google Scholar] [CrossRef]
- Perolat, J.; Leibo, J.Z.; Zambaldi, V.; Beattie, C.; Tuyls, K.; Graepel, T. A multi-agent reinforcement learning model of common-pool resource appropriation. Adv. Neural Inf. Process. Syst. 2017, 30, 3646–3655. [Google Scholar]
- Zhou, J.; Lai, J.; Menda, G.; Stafstrom, J.A.; Miles, C.I.; Hoy, R.R.; Miles, R.N. Outsourced hearing in an orb-weaving spider that uses its web as an auditory sensor. Proc. Natl. Acad. Sci. USA 2022, 119, e2122789119. [Google Scholar] [CrossRef]
- Dawkins, R. Climbing Mount Improbable; WW Norton and Company: New York, NY, USA, 1997. [Google Scholar]
- Lake, B.M.; Ullman, T.D.; Tenenbaum, J.B.; Gershman, S.J. Building machines that learn and think like people. Behav. Brain Sci. 2017, 40, e253. [Google Scholar] [CrossRef]
- Floreano, D.; Mitri, S.; Magnenat, S.; Keller, L. Evolutionary conditions for the emergence of communication in robots. Curr. Biol. 2007, 17, 514–519. [Google Scholar] [CrossRef]
- Mitri, S.; Floreano, D.; Keller, L. The evolution of information suppression in communicating robots with conflicting interests. Proc. Natl. Acad. Sci. USA 2009, 106, 15786–15790. [Google Scholar] [CrossRef]
- Alberch, P. The logic of monsters: Evidence for internal constraint in development and evolution. Geobios 1989, 22, 21–57. [Google Scholar] [CrossRef]


| Human Brains | NH Vertebrate Brains | Deep AN Networks | EVOL-Neurorobotics | |
|---|---|---|---|---|
| Wiring | Hierarchical-nested | Hierarchical-nested | Feed-forward | FF, programmed |
| Basic units | Neurons | Neurons | Threshold units | Threshold units |
| Internal dynamics | Critical | Critical | Point attractors | Sensorimotor control |
| Time travel | Yes | Limited | None | None |
| Generalisation | Yes | Limited | No | No |
| Language | Syntactic | Simple | None | Proto-grammar |
| Meta-learning | Yes | Limited | Learning To learn | None |
| Mind readers | Yes | Limited | No | Emotion detector |
| Right ≠ wrong | Yes | Yes | Built Ethics | Built Ethics |
| Extended mind | Vast | Limited | No | Embodiment |
| Social Learning | Dominant | Limited | No | Imitation learning |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solé, R.; Seoane, L.F. Evolution of Brains and Computers: The Roads Not Taken. Entropy 2022, 24, 665. https://doi.org/10.3390/e24050665
Solé R, Seoane LF. Evolution of Brains and Computers: The Roads Not Taken. Entropy. 2022; 24(5):665. https://doi.org/10.3390/e24050665
Chicago/Turabian StyleSolé, Ricard, and Luís F. Seoane. 2022. "Evolution of Brains and Computers: The Roads Not Taken" Entropy 24, no. 5: 665. https://doi.org/10.3390/e24050665
APA StyleSolé, R., & Seoane, L. F. (2022). Evolution of Brains and Computers: The Roads Not Taken. Entropy, 24(5), 665. https://doi.org/10.3390/e24050665






