Recent Status and Prospects on Thermochemical Heat Storage Processes and Applications
Abstract
1. Introduction
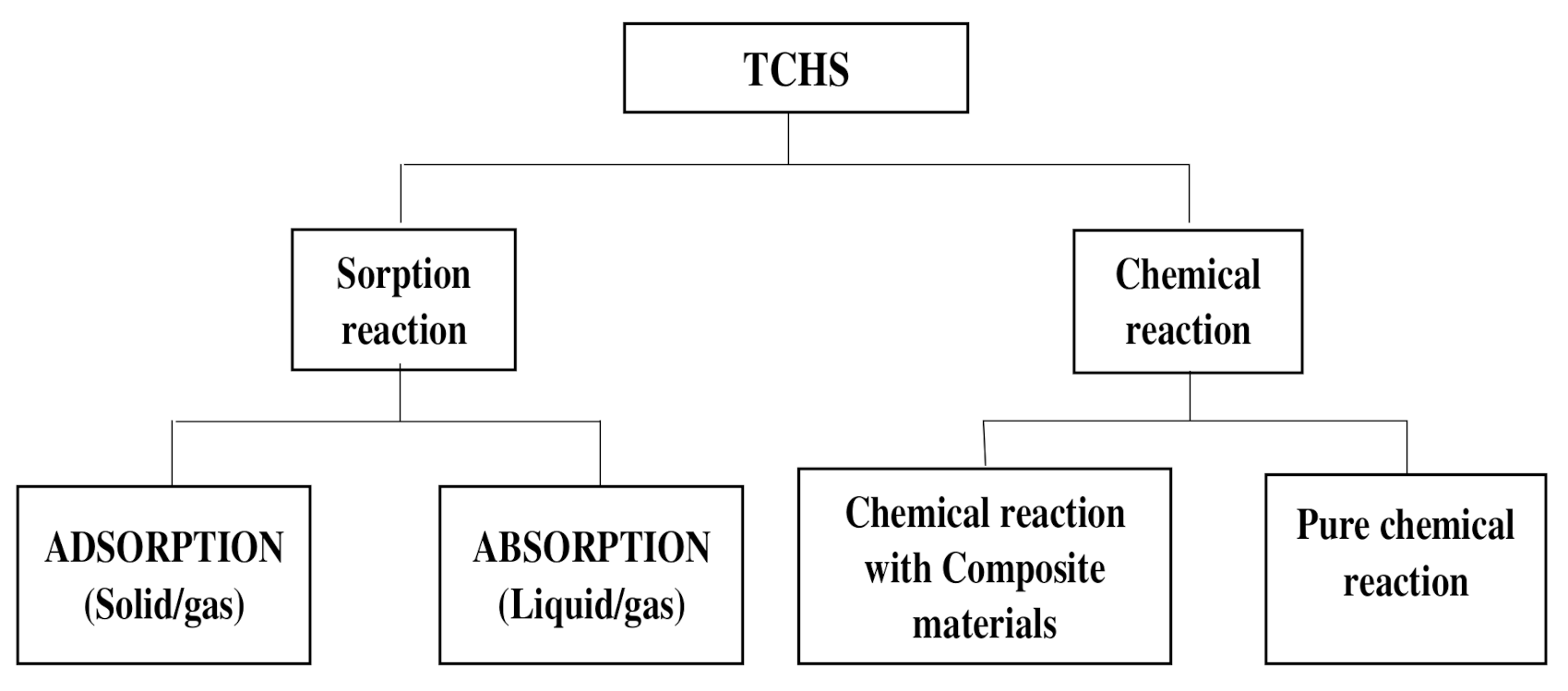
2. General Concepts of TCHS
3. The Different Processes for TCHS
- Storage capacity: this defines how much energy the system can store and varies significantly according to the process, the device, and the scale of the system involved.
- Power: it defines the speed at which the energy stored in the system can be released (and loaded).
- Efficiency: it is the balance between the energy received by the user and the energy required to charge the storage unit. It takes into account the energy losses during the storing process.
- Storage period: this is the total time that energy is stored, ranging from few hours to several months. This enables characteristics such as short- or long-term storage.
- Charge/discharge time: it defines the duration needed to fully charge and discharge the system.
- Cost of the technology: it evaluates the cost price of the system by referring either to the capacity (cost/kWh) or the power (cost/kW) of the system and fluctuates according to the investment and operating costs of the equipment involved and their life span.
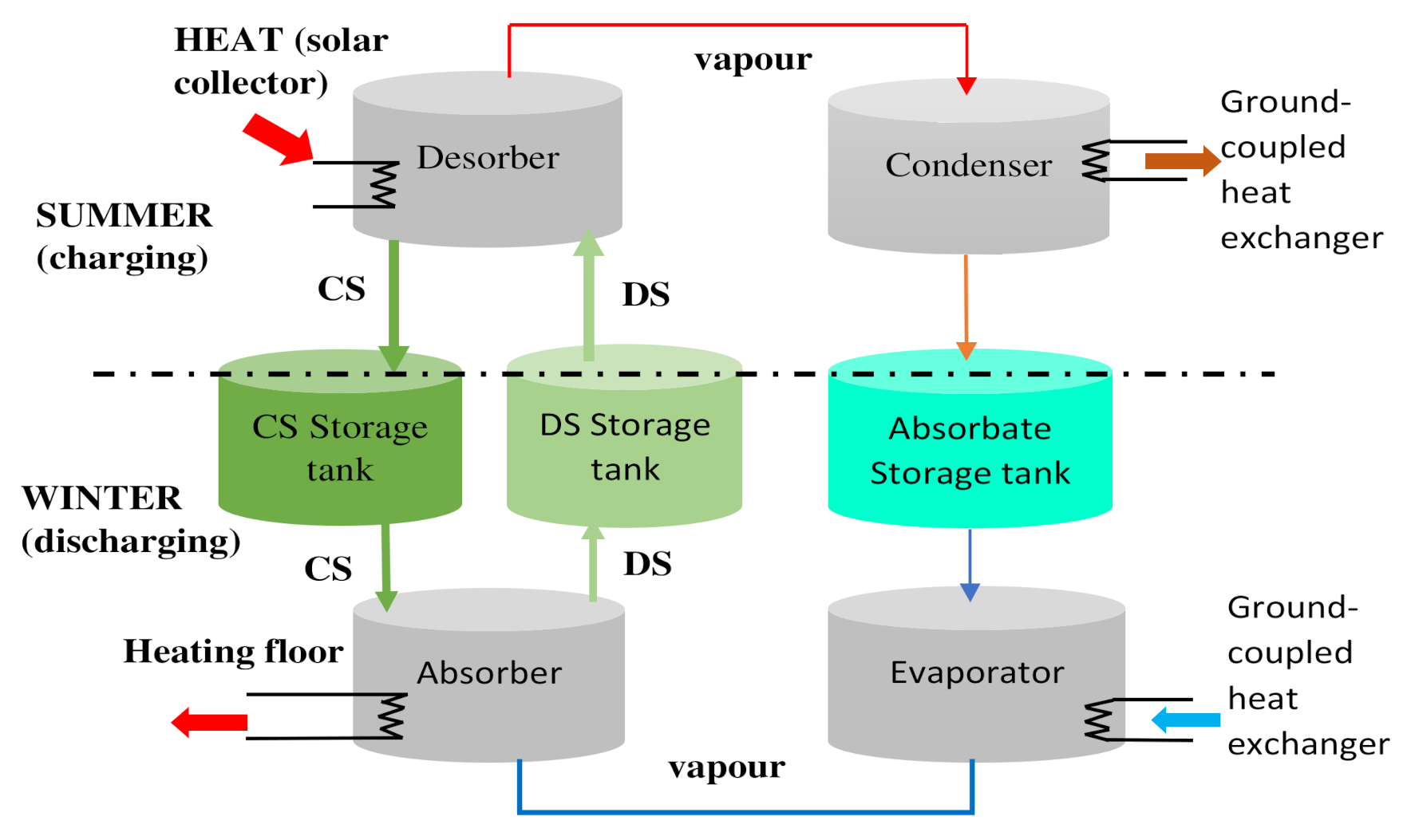
3.1. Absorption Storage Process
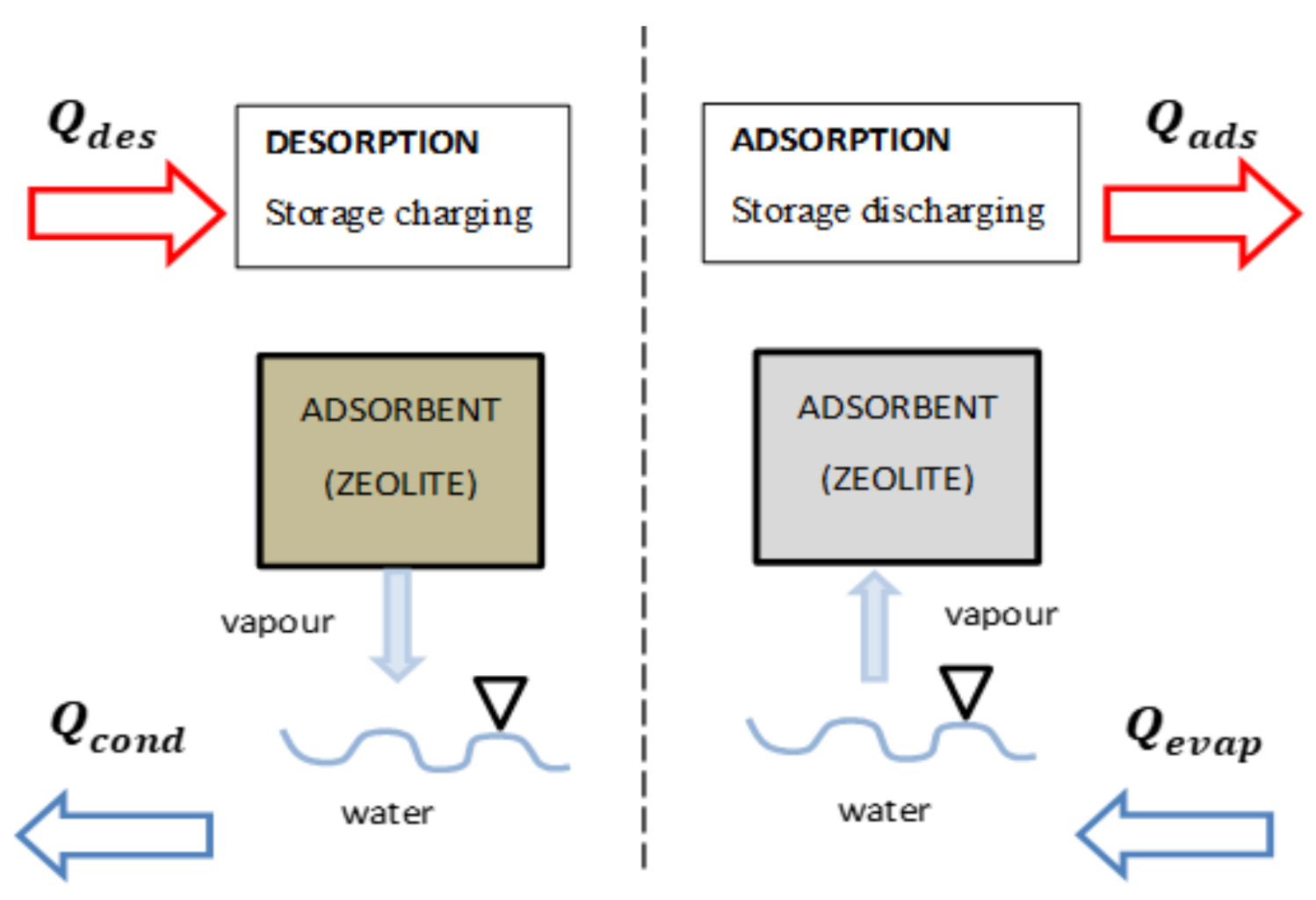
3.2. Adsorption Storage Process
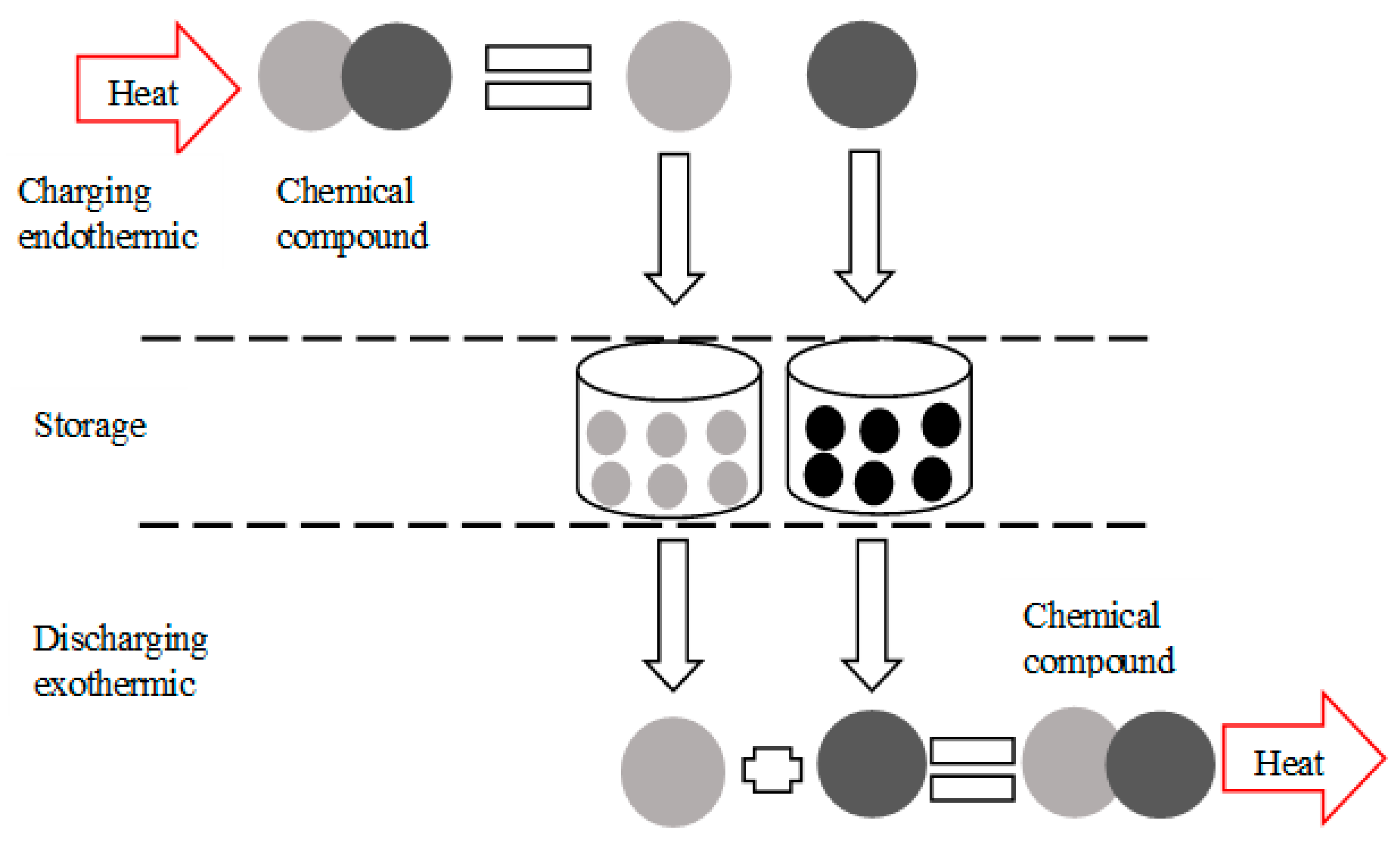
3.3. Chemical Reactions Storage Process
- The energy density is, respectively, higher than for adsorption and absorption storage systems.
- The temperature can rise to 1000 °C for a certain application.
- The heat can be restored at a constant temperature.
- The storage time, as well as the transport distance of the reagents, is theoretically unlimited since the products are stored under ambient temperature. This process is the most suitable heat storage process for seasonal storage, i.e., storing energy in the summer and releasing it in the winter for a long duration.
4. TCHS Materials
- The high affinity of the sorbent for the sorbate: impacts the rate of the reaction.
- Better volatility of the sorbate than the sorbent in absorption.
- High thermal conductivity and high heat transfer with the heat transfer fluid in the case adsorption.
- Desorption temperature as low as possible and suitable permeability.
- Environmental safety, non-toxicity, low global warming potential and ozone depletion potential, and low cost.
- Non-corrosiveness of materials and a low recovery temperature to ensure high solar fractions.
- Good thermal and molecular stability under assigned operating conditions (temperature, pressure).
- Moderate operating pressure range, no excessive pressure conditions, and especially no high vacuum.
4.1. Materials for Absorption and Adsorption Storage Processes
4.2. Materials for Chemical Reaction Energy Storage Process
5. TCES Reactors
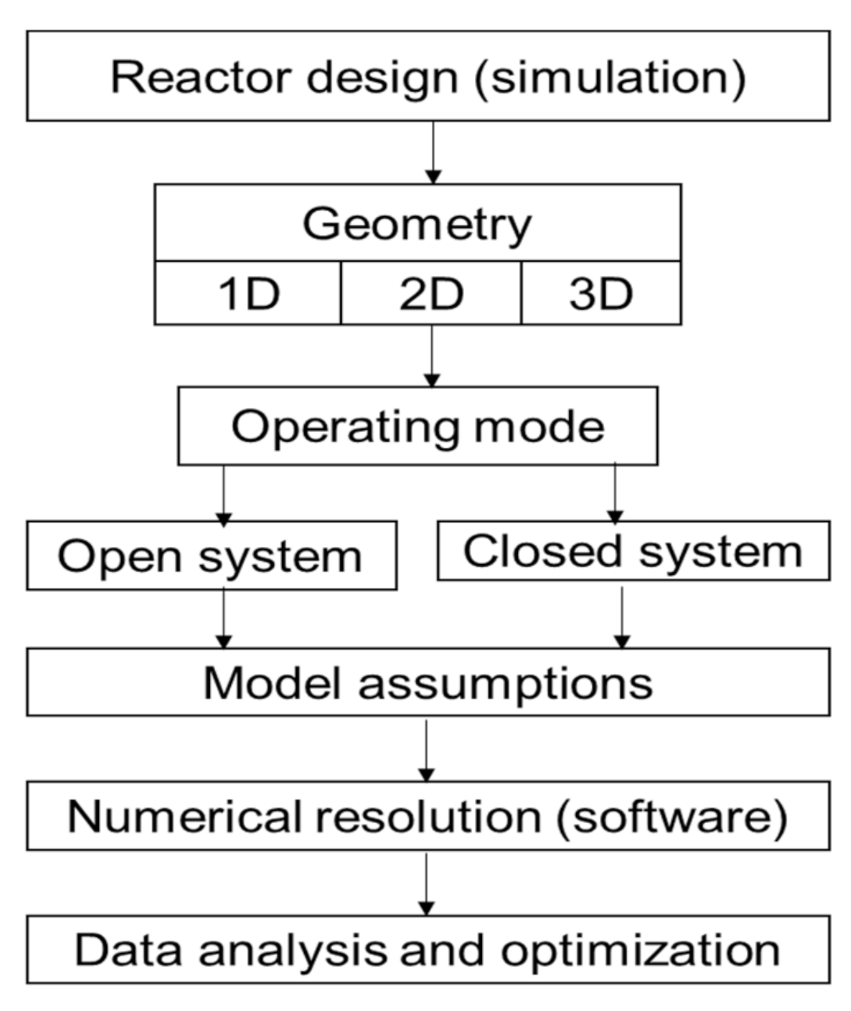
5.1. TCES Reactor Sizing Analysis Criteria and Assumptions
5.2. TCES Reactor Classification
- The very low heat and mass transfer efficiency of the system.
- The non-uniform distribution of the energy to be stored within the material reservoir.
- The uncontrolled corrosion of internal components.
- The high cost of the conception, which reduces the number of available prototypes.
- The need for additional gas for fluidization in the case of fluidized reactors.
- The difficulty of maintaining reactor components.
- The very high-pressure drops of the transfer fluid at the outlet.
- The limited number of available simulations works that do not allow a thorough understanding of the physical phenomena involved.
6. TCHS Systems Prototypes and Projects
7. Conclusions and Prospects
- TCHS is a wide area of investigation more than as discussed in one paper.
- Absorption and adsorption processes are commonly used for space heating purposes and applications that require a low or middle grade of temperature, whereas the chemical reaction process is used for high energy density and high temperature.
- Magnesium chloride has received considerable interest in recent work with the increasing use of open-circuit reactors for building heating applications and inorganic hydroxide material for high-temperature applications.
- The shape of the reactors, as well as the correct choice of the reactive bed, appeared to be very important. It should therefore be emphasized that before any experimental work and any prototype design, a numerical simulation through the above-mentioned software must be performed, and the simulation works should be clear enough and realistic to allow the implementation in a prototype device for experimentations
- The sizing of heat exchangers is an integral part of reactor sizing, and their efficiency has an impact on the reactor performance.
- Before any design of the system, it is necessary to take into account the real application required (building heating, industrial hot temperature process, etc.).
- Closed TCHS reactors require a heat exchanger to provide or remove the heat of the reaction. It involves a lot of technical components but allows better control of the reactor and offers better reaction kinetics.
- The open TCHS reactor, because it operates at atmospheric pressure, overcomes these constraints, offering a more simplified and less economical design, but it cannot provide better control of the reactor. New technology must therefore be found to combine these two operating systems. We suggest the type of open system capable of operating with a heat exchanger connected to an external open system.
- For both TCHS reactors, many barriers remain to be overcome. Particularly on the geometries of the devices, which do not ensure an optimal operation. Heat exchanger selection criteria, the control of the transfer phenomena within the reactors, and the problems of recycling materials after the reactions are some. While the cycles and lifetimes of the materials altered after the reactions, the problem of corrosion within the reactors as well as the high thermal losses deteriorates the performance of the equipment. The problems of adapting specific devices for each type of application involved, and the difficulties of maintenance of the systems due to their complexity complete the barrier list.
- Numerical simulations are of crucial importance in the dimensioning of thermochemical heat storage systems. In this study, a dashboard has been prosed for this purpose.
- For the visualization of physical phenomena occurring within the reactor, more recently the COMSOL Multiphysics software is increasingly being used since it offers a 3D model for the resolution of equations with a very satisfactory mesh system and calculation accuracy. The Trnsys Simulation software is also used in the case of a macro-scale simulation involving the production or storage of energy until its end-use. It should therefore be emphasized that before any experimental work and any prototype design, a numerical simulation through microscale and macroscale system software must be performed.
- Future research directions must take into account:
- The problems that hinder TCHS technology separately: microscopic aspect and macroscopic aspect.
- The enhancement of the thermal conductivity of the storage materials and the stability of the reaction cycles.
- The development of several models of the reactive bed, allowing better storage and a total reaction rate of the materials.
- The investigation of coupling between the physics of heat transfer in porous media, chemical reactions, and the transport of diluted species in solution.
- The analysis of corrosion problems inside reactors.
- The numerical simulation of a varied range of a combination of reactor and storage materials, to have a consistent database for technological choices.
- Intensifying of experimentation efforts to validate and promote numerical models and simulations works, and application of TCHS technology through technical and economic feasibility analysis.
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| preexponential factor [1/s] | |
| concentration of the material [mol/m3] | |
| specific molar heat capacity [J/(mol·K] | |
| vapor diffusion coefficient | |
| activation energy | |
| acceleration of gravity | |
| permeability | |
| M | Molecular mass |
| mass of the material | |
| number of the moles in the bed | |
| p | pressure [Pa] |
| thermal power | |
| reference pressure [1 bar] | |
| amount of heat consumption | |
| Ideal gas constant | |
| s | entropy |
| Sink or source | |
| gas | |
| solid | |
| s | sorbent |
| t | total |
| t | time [s] |
| velocity vector | |
| atomic volume | |
| advancement of the reaction [-] | |
| free Gibbs energy | |
| free standard Gibbs energy | |
| reaction standard entropy | |
| reaction standard enthalpy | |
| TCHS | Thermochemical Heat Storage |
| density | |
| air velocity | |
| stoichiometric coefficient | |
| Subscript | |
| 0 | anhydrous state |
| 1 | hydrate state |
| eff | effective |
| eq | equilibrium |
| i | initial |
| Greek symbols | |
| extent conversion [%] | |
| heating rate [K/min] | |
| Porosity [%] | |
| λ | thermal conductivity [W/(m·K)] |
| equilibrium constant [-] | |
References
- Cabeza, L.F.; Solé, A.; Barreneche, C. Review on sorption materials and technologies for heat pumps and thermal energy storage. Renew. Energy 2017, 110, 3–39. [Google Scholar] [CrossRef]
- Palomba, V.; Frazzica, A. Recent advancements in sorption technology for solar thermal energy storage applications. Sol. Energy 2019, 192, 69–105. [Google Scholar] [CrossRef]
- Prasad, J.S.; Muthukumar, P.; Desai, F.; Basu, D.N.; Rahman, M.M. A critical review of high-temperature reversible thermochemical energy storage systems. Appl. Energy 2019, 254, 113733. [Google Scholar] [CrossRef]
- Desai, F.; Prasad, J.S.; Muthukumar, P.; Rahman, M.M. Thermochemical energy storage system for cooling and process heating applications: A review. Energy Convers. Manag. 2021, 229, 113617. [Google Scholar] [CrossRef]
- N’Tsoukpoe, K.E.; Liu, H.; Le Pierrès, N.; Luo, L. A review on long-term sorption solar energy storage. Renew. Sustain. Energy Rev. 2009, 13, 2385–2396. [Google Scholar] [CrossRef]
- Tatsidjodoung, P.; Le Pierrès, N.; Luo, L. A review of potential materials for thermal energy storage in building applications. Renew. Sustain. Energy Rev. 2013, 18, 327–349. [Google Scholar] [CrossRef]
- Cot-Gores, J.; Castell, A.; Cabeza, L.F. Thermochemical energy storage and conversion: A-state-of-the-art review of the experimental research under practical conditions. Renew. Sustain. Energy Rev. 2012, 16, 5207–5224. [Google Scholar] [CrossRef]
- Yu, N.; Wang, R.; Wang, L. Sorption thermal storage for solar energy. Prog. Energy Combust. Sci. 2013, 39, 489–514. [Google Scholar] [CrossRef]
- Solé, A.; Martorell, I.; Cabeza, L.F. State of the art on gas–Solid thermochemical energy storage systems and reactors for building applications. Renew. Sustain. Energy Rev. 2015, 47, 386–398. [Google Scholar] [CrossRef]
- Fopah-Lele, A.; Tamba, J.G. A review on the use of SrBr2·6H2O as a potential material for low temperature energy storage systems and building applications. Sol. Energy Mater. Sol. Cells 2017, 164, 175–187. [Google Scholar] [CrossRef]
- Krese, G.; Koželj, R.; Butala, V.; Stritih, U. Thermochemical seasonal solar energy storage for heating and cooling of buildings. Energy Build. 2018, 164, 239–253. [Google Scholar] [CrossRef]
- Lizana, J.; Chacartegui, R.; Barrios-Padura, A.; Ortiz, C. Advanced low-carbon energy measures based on thermal energy storage in buildings: A review. Renew. Sustain. Energy Rev. 2018, 82, 3705–3749. [Google Scholar] [CrossRef]
- Kuznik, F.; Johannes, K.; Obrecht, C.; David, D. A review on recent developments in physisorption thermal energy storage for building applications. Renew. Sustain. Energy Rev. 2018, 94, 576–586. [Google Scholar] [CrossRef]
- Chen, M.; Sun, X.; Christensen, R.N.; Skavdahl, I.; Utgikar, V.; Sabharwall, P. Dynamic behavior of a high-temperature printed circuit heat exchanger: Numerical modeling and experimental investigation. Appl. Therm. Eng. 2018, 135, 246–256. [Google Scholar] [CrossRef]
- Dicaire, D.; Tezel, F.H. Use of adsorbents for thermal energy storage of solar or excess heat: Improvement of energy density. Int. J. Energy Res. 2013, 37, 1059–1068. [Google Scholar] [CrossRef]
- Haldorai, S.; Gurusamy, S.; Pradhapraj, M. A review on thermal energy storage systems in solar air heaters. Int. J. Energy Res. 2019, 43, 6061–6077. [Google Scholar] [CrossRef]
- Hongois, S. Stockage de Chaleur Inter-Saisonnier par Voie Thermochimique pour le Chauffage Solaire de la Maison Individuelle. Ph.D. Thesis, Université de Perpignan, Perpignan, France, 2012. [Google Scholar]
- Liu, F.; Dong, F.; Li, Y.; Jia, L. Study on the heating performance and optimal intermediate temperature of a series gas engine compression-absorption heat pump system. Appl. Therm. Eng. 2018, 135, 34–40. [Google Scholar] [CrossRef]
- Ma, T.; Ren, T.; Chen, H.; Zhu, Y.; Li, S.; Ji, G. Thermal performance of a solar high temperature thermochemical reactor powered by a solar simulator. Appl. Therm. Eng. 2019, 146, 881–888. [Google Scholar] [CrossRef]
- Cao, D.L.; Hong, G.; Le, A.T. Applying chemical heat storage to saving exhaust gas energy in diesel engines: Principle, design and experiment. J. Energy Storage 2020, 28, 101311. [Google Scholar] [CrossRef]
- Kumar, A.; Shukla, S. A Review on thermal energy storage unit for solar thermal power plant application. Energy Procedia 2015, 74, 462–469. [Google Scholar] [CrossRef]
- Vasiliev, L.; Kanonchik, L.; Tsitovich, A. Adsorption system with heat pipe thermal control for mobile storage of gaseous fuel. Int. J. Therm. Sci. 2017, 120, 252–262. [Google Scholar] [CrossRef]
- Jiang, H.; Qu, J.; Lu, R.Y.; Wang, J.-A.J. Grid-to-rod flow-induced impact study for PWR fuel in reactor. Prog. Nucl. Energy 2016, 91, 355–361. [Google Scholar] [CrossRef]
- Graves, C.; Ebbesen, S.D.; Mogensen, M.B.; Lackner, K.S. Sustainable hydrocarbon fuels by recycling CO2 and H2O with renewable or nuclear energy. Renew. Sustain. Energy Rev. 2011, 15, 1–23. [Google Scholar] [CrossRef]
- Kastanya, D. DERMAGA—An alternative tool to generate random patterned-channel-age in CANDU fuel management analyses part II: Robustness against varying basic pattern size. Prog. Nucl. Energy 2016, 91, 362–372. [Google Scholar] [CrossRef]
- Haeussler, A.; Abanades, S.; Julbe, A.; Jouannaux, J.; Cartoixa, B. Solar thermochemical fuel production from H2O and CO2 splitting via two-step redox cycling of reticulated porous ceria structures integrated in a monolithic cavity-type reactor. Energy 2020, 201, 117649. [Google Scholar] [CrossRef]
- Mamani, V.; Gutiérrez, A.; Fernández, A.; Ushak, S. Industrial carnallite-waste for thermochemical energy storage application. Appl. Energy 2020, 265, 114738. [Google Scholar] [CrossRef]
- Xu, J.; Wang, R.; Li, Y. A review of available technologies for seasonal thermal energy storage. Sol. Energy 2014, 103, 610–638. [Google Scholar] [CrossRef]
- Golparvar, B.; Niazmand, H. Adsorption cooling systems for heavy trucks A/C applications driven by exhaust and coolant waste heats. Appl. Therm. Eng. 2018, 135, 158–169. [Google Scholar] [CrossRef]
- Hou, H.; Shao, G.; Yang, W.; Wong, W.-Y. One-dimensional mesoporous inorganic nanostructures and their applications in energy, sensor, catalysis and adsorption. Prog. Mater. Sci. 2020, 113, 100671. [Google Scholar] [CrossRef]
- Petrushenko, I.; Petrushenko, K. Physical adsorption of hydrogen molecules on single-walled carbon nanotubes and carbon-boron-nitrogen heteronanotubes: A comparative DFT study. Vacuum 2019, 167, 280–286. [Google Scholar] [CrossRef]
- Imponenti, L.; Albrecht, K.J.; Wands, J.W.; Sanders, M.; Jackson, G.S. Thermochemical energy storage in strontium-doped calcium manganites for concentrating solar power applications. Sol. Energy 2017, 151, 1–13. [Google Scholar] [CrossRef]
- Pan, Z.; Zhao, C. Gas–solid thermochemical heat storage reactors for high-temperature applications. Energy 2017, 130, 155–173. [Google Scholar] [CrossRef]
- Li, Y.; Li, Z.; Lei, L.; Lan, T.; Li, Y.; Li, P.; Lin, X.; Liu, R.; Huang, Z.; Fen, X.; et al. Chemical vapor deposition-grown carbon nanotubes/graphene hybrids for electrochemical energy storage and conversion. FlatChem 2019, 15, 100091. [Google Scholar] [CrossRef]
- Janghel, D.; Karagadde, S.; Saha, S.K. Thermal performance analysis of phase change material based thermal storage system usineneralized numerical model with volumetric expansion and shrinkage. Appl. Therm. Eng. 2020, 180, 115826. [Google Scholar] [CrossRef]
- Cabeza, L.F.; Solé, A.; Fontanet, X.; Barreneche, C.; Jové, A.; Gallas, M.; Prieto, C.; Fernández, A.I. Thermochemical energy storage by consecutive reactions for higher efficient concentrated solar power plants (CSP): Proof of concept. Appl. Energy 2017, 185, 836–845. [Google Scholar] [CrossRef]
- Aydin, D.; Casey, S.P.; Riffat, S. The latest advancements on thermochemical heat storage systems. Renew. Sustain. Energy Rev. 2015, 41, 356–367. [Google Scholar] [CrossRef]
- Farcot, L.; Le Pierrès, N.; Fourmigué, J.-F. Experimental investigation of a moving-bed heat storage thermochemical reactor with SrBr2/H2O couple. J. Energy Storage 2019, 26, 101009. [Google Scholar] [CrossRef]
- Ma, Z.; Bao, H.; Roskilly, A.P. Seasonal solar thermal energy storage using thermochemical sorption in domestic dwellings in the UK. Energy 2019, 166, 213–222. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, R. Sorption thermal energy storage: Concept, process, applications and perspectives. Energy Storage Mater. 2020, 27, 352–369. [Google Scholar] [CrossRef]
- Le Pierrès, N.; N’Tsoukpoe, K.E. Procédé de stockage de chaleur solaire intersaisonnier par absorption LiBr-H2O. In Proceedings of the 20th Congrès Français de Mécanique, Besançon, France, 28 August–2 September 2011; p. 6. [Google Scholar]
- Ponshanmugakumar, A.; Rajavel, R. Experimental analysis of vapour absorption generator integrated with thermal energy storage system. Mater. Today Proc. 2019, 16, 1158–1167. [Google Scholar] [CrossRef]
- Gao, J.; Xu, Z.; Wang, R. Experimental study on a double-stage absorption solar thermal storage system with enhanced energy storage density. Appl. Energy 2020, 262, 114476. [Google Scholar] [CrossRef]
- Ibrahim, N.I.; Khan, M.M.A.; Mahbubul, I.; Saidur, R.; Al-Sulaiman, F.A. Experimental testing of the performance of a solar absorption cooling system assisted with ice-storage for an office space. Energy Convers. Manag. 2017, 148, 1399–1408. [Google Scholar] [CrossRef]
- Borri, E.; Tafone, A.; Comodi, G.; Romagnoli, A. Improving liquefaction process of microgrid scale Liquid Air Energy Storage (LAES) through Waste Heat Recovery (WHR) and absorption chiller. Energy Procedia 2017, 143, 699–704. [Google Scholar] [CrossRef]
- Berdiyeva, P.; Karabanova, A.; Makowska, M.; Johnsen, R.E.; Blanchard, D.; Hauback, B.C.; Deledda, S. In-situ neutron imaging study of NH3 absorption and desorption in SrCl2 within a heat storage prototype reactor. J. Energy Storage 2020, 29, 101388. [Google Scholar] [CrossRef]
- Razmi, A.; Soltani, M.; Torabi, M. Investigation of an efficient and environmentally-friendly CCHP system based on CAES, ORC and compression-absorption refrigeration cycle: Energy and exergy analysis. Energy Convers. Manag. 2019, 195, 1199–1211. [Google Scholar] [CrossRef]
- Kee, S.Y.; Wong, J.L.O.; Munusamy, Y.; Ong, K.S.; Choong, Y.C. Light absorptive polymeric form-stable composite phase change material for thermal storage. Appl. Therm. Eng. 2020, 172, 114673. [Google Scholar] [CrossRef]
- Tafone, A.; Borri, E.; Comodi, G.; Broek, M.V.D.; Romagnoli, A. Liquid Air Energy Storage performance enhancement by means of organic rankine cycle and absorption chiller. Appl. Energy 2018, 228, 1810–1821. [Google Scholar] [CrossRef]
- Le Pierrès, N.; Huaylla, F.; Stutz, B.; Perraud, J. Long-term solar heat storage process by absorption with the KCOOH/H2O couple: Experimental investigation. Energy 2017, 141, 1313–1323. [Google Scholar] [CrossRef]
- Wu, W. Low-temperature compression-assisted absorption thermal energy storage using ionic liquids. Energy Built Environ. 2020, 1, 139–148. [Google Scholar] [CrossRef]
- Kaushik, S.; Lam, K.; Chandra, S.; Tomar, C. Mass and energy storage analysis of an absorption heat pump with simulated time dependent generator heat input. Energy Convers. Manag. 1982, 22, 183–196. [Google Scholar] [CrossRef]
- Mohamed, H.; Ben Brahim, A. Modeling of the absorption phase of a cycle with solar absorption using the couple NH3–H2O for in-sight energy storage. Int. J. Hydrog. Energy 2017, 42, 8624–8630. [Google Scholar] [CrossRef]
- Guloglu, G.E.; Hamidi, Y.K.; Altan, M.C. Moisture absorption of composites with interfacial storage. Compos. Part A Appl. Sci. Manuf. 2020, 134, 105908. [Google Scholar] [CrossRef]
- Qian, M.; Xu, C.; Gao, Y. Open-air combustion synthesis of three-dimensional graphene for oil absorption and energy storage. Mater. Sci. Eng. B 2018, 238-239, 149–154. [Google Scholar] [CrossRef]
- Wang, L.; Xiao, F.; Cui, B.; Hu, M.; Lu, T. Performance analysis of absorption thermal energy storage for distributed energy systems. Energy Procedia 2019, 158, 3152–3157. [Google Scholar] [CrossRef]
- Ibrahim, N.I.; Al-Sulaiman, F.A.; Ani, F.N. Performance characteristics of a solar driven lithium bromide-water absorption chiller integrated with absorption energy storage. Energy Convers. Manag. 2017, 150, 188–200. [Google Scholar] [CrossRef]
- De, R.K.; Ganguly, A. Performance comparison of solar-driven single and double-effect LiBr-water vapor absorption system based cold storage. Therm. Sci. Eng. Prog. 2020, 17, 100488. [Google Scholar] [CrossRef]
- Hirmiz, R.; Lightstone, M.; Cotton, J. Performance enhancement of solar absorption cooling systems using thermal energy storage with phase change materials. Appl. Energy 2018, 223, 11–29. [Google Scholar] [CrossRef]
- Ibrahim, N.I.; Al-Sulaiman, F.A.; Ani, F.N. Solar absorption systems with integrated absorption energy storage–A review. Renew. Sustain. Energy Rev. 2018, 82, 1602–1610. [Google Scholar] [CrossRef]
- Leonzio, G. Solar systems integrated with absorption heat pumps and thermal energy storages: State of art. Renew. Sustain. Energy Rev. 2017, 70, 492–505. [Google Scholar] [CrossRef]
- Bi, Y.; Zang, G.; Qin, L.; Li, H.; Wang, H. Study on the characteristics of charging/discharging processes in three-phase energy storage coupling in solar air conditioning system. Energy Build. 2019, 204, 109456. [Google Scholar] [CrossRef]
- Mehari, A.; Xu, Z.; Wang, R. Thermal energy storage using absorption cycle and system: A comprehensive review. Energy Convers. Manag. 2020, 206, 112482. [Google Scholar] [CrossRef]
- Rodriguez-Hidalgo, M.; Rodriguez-Aumente, P.; Lecuona-Neumann, A.; Legrand, M. Thermo-chemical storage for renewable energies based on absorption: Getting a uniform injection into the grid. Appl. Therm. Eng. 2019, 146, 338–345. [Google Scholar] [CrossRef]
- Peng, X.; She, X.; Li, Y.; Ding, Y. Thermodynamic analysis of Liquid Air Energy Storage integrated witerial system of organic rankine and absorption refrigeration cycles driven by compression heat. Energy Procedia 2017, 142, 3440–3446. [Google Scholar] [CrossRef]
- Razmi, A.; Soltani, M.; Aghanajafi, C.; Torabi, M. Thermodynamic and economic investigation of a novel integration of the absorption-recompression refrigeration system with compressed air energy storage (CAES). Energy Convers. Manag. 2019, 187, 262–273. [Google Scholar] [CrossRef]
- Fernandes, M.; Gaspar, A.; Costa, V.; Costa, J.J.; Brites, G. Optimization of a thermal energy storage system provided with an adsorption module–A GenOpt application in a TRNSYS/MATLAB model. Energy Convers. Manag. 2018, 162, 90–97. [Google Scholar] [CrossRef]
- Wang, H.; Hao, Y. Thermodynamic study of solar thermochemical methane steam reforming with alternating H2 and CO2 permeation membranes reactors. Energy Procedia 2017, 105, 1980–1985. [Google Scholar] [CrossRef]
- Loni, R.; Pavlovic, S.; Bellos, E.; Tzivanidis, C.; Asli-Ardeh, E.A. Thermal and exergy performance of a nanofluid-based solar dish collector with spiral cavity receiver. Appl. Therm. Eng. 2018, 135, 206–217. [Google Scholar] [CrossRef]
- Zhang, M.; Wei, Z.; Wang, T.; Muhammad, S.; Zhu, J.; Liu, J.; Hu, J. Nickel-iron layered double hydroxides and reduced graphene oxide composite with robust lithium ion adsorption ability for high-capacity energy storage systems. Electrochim. Acta 2019, 296, 190–197. [Google Scholar] [CrossRef]
- Smejkal, T.; Mikyška, J.; Fučík, R. Numerical modelling of adsorption and desorption of water vapor in zeolite 13X using a two-temperature model and mixed-hybrid finite element method numerical solver. Int. J. Heat Mass Transf. 2020, 148, 119050. [Google Scholar] [CrossRef]
- Alby, D.; Salles, F.; Fullenwarth, J.; Zajac, J. On the use of metal cation-exchanged zeolites in sorption thermochemical storage: Some practical aspects in reference to the mechanism of water vapor adsorption. Sol. Energy Mater. Sol. Cells 2018, 179, 223–230. [Google Scholar] [CrossRef]
- Ng, S.W.L.; Yilmaz, G.; Ong, W.L.; Ho, G.W. One-step activation towards spontaneous etching of hollow and hierarchical porous carbon nanospheres for enhanced pollutant adsorption and energy storage. Appl. Catal. B Environ. 2018, 220, 533–541. [Google Scholar] [CrossRef]
- Narayanan, S.; Yang, S.; Kim, H.; Wang, E.N. Optimization of adsorption processes for climate control and thermal energy storage. Int. J. Heat Mass Transf. 2014, 77, 288–300. [Google Scholar] [CrossRef]
- Ntsoane, M.L.; Jalali, A.; Römer, J.; Duewel, K.; Göller, C.; Kühn, R.; Mähne, K.; Geyer, M.; Sivakumar, D.; Mahajan, P.V. Performance evaluation of silica gel-water adsorption based cooling system for mango fruit storage in Sub-Saharan Africa. Postharvest Biol. Technol. 2019, 149, 195–199. [Google Scholar] [CrossRef]
- Zhang, C.; Kong, R.; Wang, X.; Xu, Y.; Wang, F.; Ren, W.; Wang, Y.; Su, F.; Jiang, J.-X. Porous carbons derived from hypercrosslinked porous polymers for gas adsorption and energy storage. Carbon 2017, 114, 608–618. [Google Scholar] [CrossRef]
- Schreiber, H.; Lanzerath, F.; Bardow, A. Predicting performance of adsorption thermal energy storage: From experiments to validated dynamic models. Appl. Therm. Eng. 2018, 141, 548–557. [Google Scholar] [CrossRef]
- Jia, H.; Zhang, H.; Wan, S.; Sun, J.; Xie, X.; Sun, L. Preparation of nitrogen-doped porous carbon via adsorption-doping for highly efficient energy storage. J. Power Sources 2019, 433, 226712. [Google Scholar] [CrossRef]
- Rani, S.; Padmanabhan, E.; Prusty, B.K. Review of gas adsorption in shales for enhanced methane recovery and CO2 storage. J. Pet. Sci. Eng. 2019, 175, 634–643. [Google Scholar] [CrossRef]
- Kaushal, I.; Saharan, P.; Kumar, V.; Sharma, A.K.; Umar, A. Superb sono-adsorption and energy storage potential of multifunctional Ag-Biochar composite. J. Alloy. Compd. 2019, 785, 240–249. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, H. Surface adsorption and encapsulated storage of H2 molecules in a cagelike (MgO)12 cluster. Int. J. Hydrog. Energy 2018, 43, 16609–16616. [Google Scholar] [CrossRef]
- Olsson, E.; Hussain, T.; Karton, A.; Cai, Q. The adsorption and migration behavior of divalent metals (Mg, Ca, and Zn) on pristine and defective graphene. Carbon 2020, 163, 276–287. [Google Scholar] [CrossRef]
- Helaly, H.O.; Awad, M.M.; El-Sharkawy, I.I.; Hamed, A.M. Theoretical and experimental investigation of the performance of adsorption heat storage system. Appl. Therm. Eng. 2019, 147, 10–28. [Google Scholar] [CrossRef]
- Brancato, V.; Gordeeva, L.G.; Grekova, A.D.; Sapienza, A.; Vasta, S.; Frazzica, A.; Aristov, Y.I. Water adsorption equilibrium and dynamics of LICL/MWCNT/PVA composite for adsorptive heat storage. Sol. Energy Mater. Sol. Cells 2019, 193, 133–140. [Google Scholar] [CrossRef]
- Lehmann, C.; Beckert, S.; Nonnen, T.; Gläser, R.; Kolditz, O.; Nagel, T. Water loading lift and heat storage density prediction of adsorption heat storage systems using Dubinin-Polanyi theory—Comparison with experimental results. Appl. Energy 2017, 207, 274–282. [Google Scholar] [CrossRef]
- Bales, C. Laboratory Tests of Chemical Reactions and Prototype Sorption Storage Units: Report of B4 of Subtask B. A Report of IEA Solar Heating and Cooling Programme. January 2008. Available online: http://www.iea-shc.org/data/sites/1/publications/task32-b4.pdf (accessed on 18 July 2021).
- Kerskes, H.; Mette, B.; Bertsch, F.; Asenbeck, S.; Drück, H. Chemical energy storage using reversible solid/gas-reactions (CWS)—Results of the research project. Energy Procedia 2012, 30, 294–304. [Google Scholar] [CrossRef]
- N’Tsoukpoe, K.E.; Schmidt, T.; Rammelberg, H.U.; Watts, B.A.; Ruck, W.K. A systematic multi-step screening of numerous salt hydrates for low temperature thermochemical energy storage. Appl. Energy 2014, 124, 1–16. [Google Scholar] [CrossRef]
- Shao, H.; Nagel, T.; Roßkopf, C.; Linder, M.; Wörner, A.; Kolditz, O. Non-equilibrium thermo-chemical heat storage in porous media: Part 2—A 1D computational model for a calcium hydroxide reaction system. Energy 2013, 60, 271–282. [Google Scholar] [CrossRef]
- De Valeria, M.K.; Michaelides, E.E.; Michaelides, D.N. Energy and thermal storage in clusters of grid-independent buildings. Energy 2020, 190, 116440. [Google Scholar] [CrossRef]
- Lim, K.-W.; Peddigari, M.; Annapureddy, V.; Hwang, G.-T.; Choi, J.-J.; Kim, G.-Y.; Yi, S.N.; Ryu, J. Energy storage characteristics of {001} oriented Pb(Zr0.52Ti0.48)O3 thin film grown by chemical solution deposition. Thin Solid Films 2018, 660, 434–438. [Google Scholar] [CrossRef]
- Uebbing, J.; Rihko-Struckmann, L.K.; Sundmacher, K. Exergetic assessment of CO2 methanation processes for the chemical storage of renewable energies. Appl. Energy 2019, 233-234, 271–282. [Google Scholar] [CrossRef]
- Silakhori, M.; Jafarian, M.; Arjomandi, M.; Nathan, G.J. Experimental assessment of copper oxide for liquid chemical looping for thermal energy storage. J. Energy Storage 2019, 21, 216–221. [Google Scholar] [CrossRef]
- Liu, J.; Baeyens, J.; Deng, Y.; Wang, X.; Zhang, H. High temperature Mn2O3/Mn3O4 and Co3O4/CoO systems for thermo-chemical energy storage. J. Environ. Manag. 2020, 267, 110582. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Fan, S.; Lang, X.; Wang, Y.; Li, G.; Wang, S. Hydrogen and chemical energy storage in gas hydrate at mild conditions. Int. J. Hydrog. Energy 2020, 45, 14915–14921. [Google Scholar] [CrossRef]
- Cui, C.; Pu, Y. Improvement of energy storage density with trace amounts of ZrO2 additives fabricated by wet-chemical method. J. Alloy. Compd. 2018, 747, 495–504. [Google Scholar] [CrossRef]
- Atinafu, D.G.; Wang, C.; Dong, W.; Chen, X.; Du, M.; Gao, H.; Wang, G. In-situ derived graphene from solid sodium acetate for enhanced photothermal conversion, thermal conductivity, and energy storage capacity of phase change materials. Sol. Energy Mater. Sol. Cells 2020, 205, 110269. [Google Scholar] [CrossRef]
- Tesio, U.; Guelpa, E.; Verda, V. Integration of thermochemical energy storage in concentrated solar power. Part 1: Energy and economic analysis/optimization. Energy Convers. Manag. X 2020, 6, 100039. [Google Scholar] [CrossRef]
- Mehrpooya, M.; Pakzad, P. Introducing a hybrid mechanical—Chemical energy storage system: Process development and energy/exergy analysis. Energy Convers. Manag. 2020, 211, 112784. [Google Scholar] [CrossRef]
- Gao, J.; Tian, Y.; Wang, L.; Zhang, X.; An, G. Investigation on bi-salt chemisorption system for long term energy storage. Chem. Eng. Sci. 2020, 221, 115699. [Google Scholar] [CrossRef]
- Wang, Y.; Ji, L.; Li, B.; Wang, L.; Bai, Y.; Chen, H.; Ding, Y. Investigation on the thermal energy storage characteristics in a spouted bed based on different nozzle numbers. Energy Rep. 2020, 6, 127–136. [Google Scholar] [CrossRef]
- Brancato, V.; Calabrese, L.; Palomba, V.; Frazzica, A.; Fullana-Puig, M.; Solé, A.; Cabeza, L.F. MgSO4·7H2O filled macro cellular foams: An innovative composite sorbent for thermo-chemical energy storage applications for solar buildings. Sol. Energy 2018, 173, 1278–1286. [Google Scholar] [CrossRef]
- Ding, H.; Song, Z.; Zhang, H.; Li, X. Niobium-based oxide anodes toward fast and safe energy storage: A review. Mater. Today Nano 2020, 11, 100082. [Google Scholar] [CrossRef]
- Palys, M.J.; Daoutidis, P. Using hydrogen and ammonia for renewable energy storage: A geographically comprehensive techno-economic study. Comput. Chem. Eng. 2020, 136, 106785. [Google Scholar] [CrossRef]
- Miller, H.A.; Lavacchi, A.; Vizza, F. Storage of renewable energy in fuels and chemicals through electrochemical reforming of bioalcohols. Curr. Opin. Electrochem. 2020, 21, 140–145. [Google Scholar] [CrossRef]
- Silakhori, M.; Jafarian, M.; Arjomandi, M.; Nathan, G.J. The energetic performance of a liquid chemical looping cycle with solar thermal energy storage. Energy 2019, 170, 93–101. [Google Scholar] [CrossRef]
- Li, B.; Li, Z.; Pang, Q.; Zhuang, Q.; Zhu, J.; Tsiakaras, P.; Shen, P.K. Synthesis and characterization of activated 3D graphene via catalytic growth and chemical activation for electrochemical energy storage in supercapacitors. Electrochim. Acta 2019, 324, 134878. [Google Scholar] [CrossRef]
- Li, S.; Liu, J.; Tan, T.; Nie, J.; Zhang, H. Optimization of LiNO3–Mg(OH)2 composites as thermo-chemical energy storage materials. J. Environ. Manag. 2020, 262, 110258. [Google Scholar] [CrossRef] [PubMed]
- Funayama, S.; Takasu, H.; Zamengo, M.; Kariya, J.; Kim, S.T.; Kato, Y. Composite material for high-temperature thermochemical energy storage using calcium hydroxide and ceramic foam. Energy Storage 2019, 1, e53. [Google Scholar] [CrossRef]
- D’Ans, P.; Hohenauer, W.; Courbon, E. Monitoring of thermal properties of a composite material used in thermochemical heat storage. In Proceedings of the Eurotherm Seminar 99—Advances in Thermal Energy Storage, Lleida, Spain, 28–30 May 2014; pp. 1–9. [Google Scholar]
- Wang, J.-X.; Li, Y.-Z.; Mao, Y.-F.; Li, E.-H.; Ning, X.; Ji, X.-Y. Comparative study of the heating surface impact on porous-material-involved spray system for electronic cooling—An experimental approach. Appl. Therm. Eng. 2018, 135, 537–548. [Google Scholar] [CrossRef]
- Wu, S.; Li, T.; Yan, T.; Wang, R. Advanced thermochemical resorption heat transformer for high-efficiency energy storage and heat transformation. Energy 2019, 175, 1222–1233. [Google Scholar] [CrossRef]
- Malley-Ernewein, A.; Lorente, S. Constructal design of thermochemical energy storage. Int. J. Heat Mass Transf. 2019, 130, 1299–1306. [Google Scholar] [CrossRef]
- Frazzica, A.; Brancato, V. Verification of hydrothermal stability of adsorbent materials for thermal energy storage. Int. J. Energy Res. 2019, 43, 6161–6170. [Google Scholar] [CrossRef]
- Dizaji, H.B.; Hosseini, H. A review of material screening in pure and mixed-metal oxide thermochemical energy storage (TCES) systems for concentrated solar power (CSP) applications. Renew. Sustain. Energy Rev. 2018, 98, 9–26. [Google Scholar] [CrossRef]
- Van Helden, W. European Projects on Seasonal Solar Thermal Storage Applications; AEE—Institute for Sustainable Technologies: Gleisdorf, Austria, 2014. [Google Scholar]
- Van Helden, W. Compact thermal storage R&D in IEA T4224 and EU COMTES project. In Proceedings of the IEA ECES–Energy Conservation through Energy Storage, Annex Proposal Workshop, Paris, France, 18–19 September 2012. [Google Scholar]
- Hauer, A. Adsorption systems for TES—Design and demonstration projects. In Proceedings of the Properties and Applications of Nanocrystalline Alloys from Amorphous Precursors, Budmerice, Slovakia, 9–15 June 2003; Springer: Berlin, Germany, 2007; pp. 409–427. [Google Scholar]
- Lu, Y.; Wang, R.; Zhang, M.; Jiangzhou, S. Adsorption cold storage system with zeolite—Water working pair used for locomotive air conditioning. Energy Convers. Manag. 2003, 44, 1733–1743. [Google Scholar] [CrossRef]
- Schreiber, H.; Graf, S.; Lanzerath, F.; Bardow, A. Adsorption thermal energy storage for cogeneration in industrial batch processes: Experiment, dynamic modeling and system analysis. Appl. Therm. Eng. 2015, 89, 485–493. [Google Scholar] [CrossRef]
- Knez, Ž.; Novak, Z. Adsorption of water vapor on silica, alumina, and their mixed oxide aerogels. J. Chem. Eng. Data 2001, 46, 858–860. [Google Scholar] [CrossRef]
- Kohler, T.; Biedermann, T.; Müller, K. Experimental study of MgCl2 ⋅ 6H2O as thermochemical energy storage material. Energy Technol. 2018, 6, 1935–1940. [Google Scholar] [CrossRef]
- Seiler, M.; Kühn, A.; Ziegler, F.; Wang, X. Sustainable cooling strategies using new chemical system solutions. Ind. Eng. Chem. Res. 2013, 52, 16519–16546. [Google Scholar] [CrossRef]
- Guerrero, M.B.; Sarrion, B.; Perejon, A.; Sanchez-Jimenez, P.E.; Perez-Maqueda, L.A.; Valverde, J.M. Large-scale high-temperature solar energy storage using natural minerals. Sol. Energy Mater. Sol. Cells 2017, 168, 14–21. [Google Scholar] [CrossRef]
- Ristić, A.; Maučec, D.; Henninger, S.K.; Kaučič, V. New two-component water sorbent CaCl2-FeKIL2 for solar thermal energy storage. Microporous Mesoporous Mater. 2012, 164, 266–272. [Google Scholar] [CrossRef]
- Xu, C.; Ju, X.; Yu, Z.; Ma, X. A review of salt hydrate-based sorption technologies for long-term thermal energy storage. Chin. Sci. Bull. 2015, 60, 3569–3579. [Google Scholar] [CrossRef][Green Version]
- Zettl, B.; Englmair, G.; Steinmaurer, G. Development of a revolving drum reactor for open-sorption heat storage processes. Appl. Therm. Eng. 2014, 70, 42–49. [Google Scholar] [CrossRef]
- Liu, D.; Xin-Feng, L.; Bo, L.; Si-Quan, Z.; Yan, X. Progress in thermochemical energy storage for concentrated solar power: A review. Int. J. Energy Res. 2018, 42, 4546–4561. [Google Scholar] [CrossRef]
- Kodama, T.; Bellan, S.; Gokon, N.; Cho, H.S. Particle reactors for solar thermochemical processes. Sol. Energy 2017, 156, 113–132. [Google Scholar] [CrossRef]
- Abanades, S.; André, L. Design and demonstration of a high temperature solar-heated rotary tube reactor for continuous particles calcination. Appl. Energy 2018, 212, 1310–1320. [Google Scholar] [CrossRef]
- Li, T.; Wang, R.; Kiplagat, J.; Wang, L.; Oliveira, R. A conceptual design and performance analysis of a triple-effect solid-gas thermochemical sorption refrigeration system with internal heat recovery. Chem. Eng. Sci. 2009, 64, 3376–3384. [Google Scholar] [CrossRef]
- Schmoldt, A.; Benthe, H.F.; Haberland, G. Digitoxin metabolism by rat liver microsomes. Biochem. Pharmacol. 1975, 24, 1639–1641. [Google Scholar] [CrossRef]
- Salviati, S.; Carosio, F.; Saracco, G.; Fina, A. Hydrated salt/graphite/polyelectrolyte organic-inorganic hybrids for efficient thermochemical storage. Nanomaterials 2019, 9, 420. [Google Scholar] [CrossRef]
- Leng, G.; Navarro, H.; Yu, Q.; Wellio, G.; Qiao, G.; Li, C.; Huang, Y.; Zhao, Y.; Zhang, G.; Meng, Y.; et al. Design of composite materials/devices for thermal storage—A critical review. Veruscript Funct. Nanomater. 2018, 2, 1–28. [Google Scholar] [CrossRef]
- Alonso, E.; Romero, M. Review of experimental investigation on directly irradiated particles solar reactors. Renew. Sustain. Energy Rev. 2015, 41, 53–67. [Google Scholar] [CrossRef]
- Lu, Y.R.; Nikrityuk, P. A fixed-bed reactor for energy storage in chemicals (E2C): Proof of concept. Appl. Energy 2018, 228, 593–607. [Google Scholar] [CrossRef]
- Ranjha, Q.; Oztekin, A. Numerical analyses of three-dimensional fixed reaction bed for thermochemical energy storage. Renew. Energy 2017, 111, 825–835. [Google Scholar] [CrossRef]
- Fopah-Lele, A.; Kuznik, F.; Osterland, T.; Ruck, W.K. Thermal synthesis of a thermochemical heat storage with heat exchanger optimization. Appl. Therm. Eng. 2016, 101, 669–677. [Google Scholar] [CrossRef]
- Tabatabaei, M.; Aghbashlo, M.; Dehhaghi, M.; Panahi, H.K.S.; Mollahosseini, A.; Hosseini, M.; Soufiyan, M.M. Reactor technologies for biodiesel production and processing: A review. Prog. Energy Combust. Sci. 2019, 74, 239–303. [Google Scholar] [CrossRef]
- Melchior, T.; Perkins, C.; Lichty, P.; Weimer, A.W.; Steinfeld, A. Solar-driven biochar gasification in a particle-flow reactor. Chem. Eng. Process. Process. Intensif. 2009, 48, 1279–1287. [Google Scholar] [CrossRef]
- Melchior, T.; Perkins, C.; Weimer, A.W.; Steinfeld, A. A cavity-receiver containing a tubular absorber for high-temperature thermochemical processing using concentrated solar energy. Int. J. Therm. Sci. 2008, 47, 1496–1503. [Google Scholar] [CrossRef]
- Wieckert, C.; Palumbo, R.; Frommherz, U. A two-cavity reactor for solar chemical processes: Heat transfer model and application to carbothermic reduction of ZnO. Energy 2004, 29, 771–787. [Google Scholar] [CrossRef]
- Nick, A.; Peter, K. Solar Thermochemical energy storage. In Chemical Engineering Process; American Institute of Chemical Engineers: New York, NY, USA, 2017; pp. 19–43. ISSN 0360-7275. [Google Scholar]
- Bush, H.E.; Datta, R.; Loutzenhiser, P.G. Aluminum-doped strontium ferrites for a two-step solar thermochemical air separation cycle: Thermodynamic characterization and cycle analysis. Sol. Energy 2019, 188, 775–786. [Google Scholar] [CrossRef]
- Kodama, T.; Gokon, N.; Yamamoto, R. Thermochemical two-step water splitting by ZrO2-supported NixFe3−xO4 for solar hydrogen production. Sol. Energy 2008, 82, 73–79. [Google Scholar] [CrossRef]
- Koepf, E.; Advani, S.; Prasad, A.K.; Steinfeld, A. Experimental investigation of the carbothermal reduction of ZnO usineam-down, gravity-fed solar reactor. Ind. Eng. Chem. Res. 2015, 54, 8319–8332. [Google Scholar] [CrossRef]
- Zgraggen, A.; Haueter, P.; Trommer, D.; Romero, M.; DeJesus, J.; Steinfeld, A. Hydrogen production by steam-gasification of petroleum coke using concentrated solar power—II Reactor design, testing, and modeling. Int. J. Hydrog. Energy 2006, 31, 797–811. [Google Scholar] [CrossRef]
- Steinfeld, A. Solar thermal production of zinc and syngas via combined ZnO-reduction and CH4-reforming processes. Int. J. Hydrog. Energy 1995, 20, 793–804. [Google Scholar] [CrossRef]
- Agrafiotis, C.; Roeb, M.; Sattler, C. A review on solar thermal syngas production via redox pair-based water/carbon dioxide splitting thermochemical cycles. Renew. Sustain. Energy Rev. 2015, 42, 254–285. [Google Scholar] [CrossRef]
- Schmidt, M.; Gollsch, M.; Giger, F.; Grün, M.; Linder, M. Development of a moving bed pilot plant for thermochemical energy storage with CaO/Ca(OH). AIP Conf. Proc. 2016, 1734, 50041. [Google Scholar] [CrossRef]
- Li, K.; Wu, H.; Wei, J.; Qiu, G.; Wei, C.; Cheng, D.; Zhong, L. Simultaneous decarburization, nitrification and denitrification (SDCND) in coking wastewater treatment using an integrated fluidized-bed reactor. J. Environ. Manag. 2019, 252, 109661. [Google Scholar] [CrossRef] [PubMed]
- Vidal, A.; Gonzalez, A.; Denk, T. A 100 kW cavity-receiver reactor with an integrated two-step thermochemical cycle: Thermal performance under solar transients. Renew. Energy 2020, 153, 270–279. [Google Scholar] [CrossRef]
- Pilehvar, A.; Esteki, M.; Ansarifar, G.; Hedayat, A. Stability analysis and parametric study of natural circulation integrated self-pressurized water reactor. Ann. Nucl. Energy 2020, 139, 107279. [Google Scholar] [CrossRef]
- Shkatulov, A.; Houben, J.; Fischer, H.; Huinink, H. Stabilization of K2CO3 in vermiculite for thermochemical energy storage. Renew. Energy 2020, 150, 990–1000. [Google Scholar] [CrossRef]
- Gonzalez-Tineo, P.A.; Durán-Hinojosa, U.; Delgadillo-Mirquez, L.R.; Meza-Escalante, E.R.; Gortáres-Moroyoqui, P.; Ulloa-Mercado, R.G.; Serrano-Palacios, D. Performance improvement of an integrated anaerobic-aerobic hybrid reactor for the treatment of swine wastewater. J. Water Process. Eng. 2020, 34, 101164. [Google Scholar] [CrossRef]
- Gabriel, K.; Finney, L.; Dolloso, P. Preliminary results of the integrated hydrolysis reactor in the Cu-Cl hydrogen production cycle. Int. J. Hydrog. Energy 2019, 44, 9743–9752. [Google Scholar] [CrossRef]
- Xing, W.; Zhang, W.; Li, D.; Li, J.; Jia, F.; Cui, Y.; Ren, F. An integrated O/A two-stage packed-bed reactor (INT-PBR) for total nitrogen removal from low organic carbon wastewater. Chem. Eng. J. 2017, 328, 894–903. [Google Scholar] [CrossRef]
- Hu, C.; Wang, M.-S.; Chen, C.-H.; Chen, Y.-R.; Huang, P.-H.; Tung, K.-L. Phosphorus-doped g-C3N4 integrated photocatalytic membrane reactor for wastewater treatment. J. Membr. Sci. 2019, 580, 1–11. [Google Scholar] [CrossRef]
- Hao, R.; Wang, X.; Mao, X.; Tian, B.; Zhao, Y.; Yuan, B.; Tao, Z.; Shen, Y. An integrated dual-reactor system for simultaneous removal of SO2 and NO: Factors assessment, reaction mechanism and application prospect. Fuel 2018, 220, 240–247. [Google Scholar] [CrossRef]
- Fernández, J.; Marín, P.; Díez, F.V.; Ordóñez, S. Combustion of coal mine ventilation air methane in a regenerative combustor with integrated adsorption: Reactor design and optimization. Appl. Therm. Eng. 2016, 102, 167–175. [Google Scholar] [CrossRef]
- Wang, X.; Wang, X.; Shao, Y.; Jin, B. Coal-fueled separated gasification chemical looping combustion under auto-thermal condition in a two-stage reactor system. Chem. Eng. J. 2020, 390, 124641. [Google Scholar] [CrossRef]
- Kiss, A.A. Versatile biodiesel production by catalytic separative reactors. In Computer Aided Chemical Engineering; Elsevier: Amsterdam, The Netherlands, 2009; Volume 27, pp. 1689–1694. [Google Scholar]
- Baloch, M.; Akunna, J.; Kierans, M.; Collier, P. Structural analysis of anaerobic granules in a phase separated reactor by electron microscopy. Bioresour. Technol. 2008, 99, 922–929. [Google Scholar] [CrossRef]
- Criado, Y.; Alonso, M.; Abanades, J.C.; Anxionnaz-Minvielle, Z. Conceptual process design of a CaO/Ca(OH)2 thermochemical energy storage system using fluidized bed reactors. Appl. Therm. Eng. 2014, 73, 1087–1094. [Google Scholar] [CrossRef]
- Kasesaz, Y.; Bavarnegin, E.; Golshanian, M.; Khajeali, A.; Jarahi, H.; Mirvakili, S.; Khalafi, H. BNCT project at Tehran Research Reactor: Current and prospective plans. Prog. Nucl. Energy 2016, 91, 107–115. [Google Scholar] [CrossRef]
- Wyttenbach, J.; Bougard, J.; Descy, G.; Skrylnyk, O.; Courbon, E.; Frère, M.; Bruyat, F. Performances and modelling of a circular moving bed thermochemical reactor for seasonal storage. Appl. Energy 2018, 230, 803–815. [Google Scholar] [CrossRef]
- Li, W.; Li, X.; Peng, Y.; Wang, Y.; Tu, J. Experimental and numerical investigations on heat transfer in stratified subsurface materials. Appl. Therm. Eng. 2018, 135, 228–237. [Google Scholar] [CrossRef]
- Straits Research. Thermal Energy Storage Market: Information by Storage Material, Technology, Application, and End-Use—Forecast Till 2026; Straits Research: New York, NY, USA, 2019; Available online: https://straitsresearch.com/report/thermal-energy-storage-market/ (accessed on 18 July 2021).
- Zhang, X.; Li, M.; Shi, W.; Wang, B.; Li, X. Experimental investigation on charging and discharging performance of absorption thermal energy storage system. Energy Convers. Manag. 2014, 85, 425–434. [Google Scholar] [CrossRef]
- Fumey, B.; Weber, R.; Gantenbein, P.; Daguenet-Frick, X.; Williamson, T.; Dorer, V. Closed sorption heat storage based on aqueous sodium hydroxide. Energy Procedia 2014, 48, 337–346. [Google Scholar] [CrossRef]
- Chen, C.; Li, Y.; Zhou, M.; Zhou, S.; Jin, W. Design of a methanation reactor for producing high-temperature supercritical carbon dioxide in solar thermochemical energy storage. Sol. Energy 2018, 176, 220–229. [Google Scholar] [CrossRef]
- Edwards, S.E.; Materić, V. Calcium looping in solar power generation plants. Sol. Energy 2012, 86, 2494–2503. [Google Scholar] [CrossRef]
- Wong, B. Sulfur Based Thermochemical Heat Storage for Baseload Concentrating Power; General Atomics: San Diego, CA, USA, 2011. [Google Scholar]
- Lin, J.; Zhao, Q.; Huang, H.; Mao, H.; Liu, Y.; Xiao, Y. Applications of low-temperature thermochemical energy storage systems for salt hydrates based on material classification: A review. Sol. Energy 2021, 214, 149–178. [Google Scholar] [CrossRef]
- N’Tsoukpoe, K.E.; Osterland, T.; Opel, O.; Ruck, W.K. Cascade thermochemical storage with internal condensation heat recovery for better energy and exergy efficiencies. Appl. Energy 2016, 181, 562–574. [Google Scholar] [CrossRef]
- N’Tsoukpoe, K.E.; Kuznik, F. A reality check on long-term thermochemical heat storage for household applications. Renew. Sustain. Energy Rev. 2021, 139, 110683. [Google Scholar] [CrossRef]
| Author(s) | Highlights | Refs |
|---|---|---|
| N’Tsoukpoe et al., (2009); Tatsidjodoung et al., (2013) |
| [5,6] |
| Cot-Gores et al., (2012) |
| [7] |
| Yu. et al., (2013) |
| [8] |
| Sole et al., (2015); Fopah-Lele and Tamba (2017) |
| [9,10] |
| Krese et al., (2018) |
| [11] |
| Lizana et al., (2018) |
| [12] |
| Kuznik et al., (2018) |
| [13] |
| Sunku Prasad et al., (2019) |
| [3] |
| Desai et al., (2020) |
| [4] |
| Adsorption | |||||||
|---|---|---|---|---|---|---|---|
| Material | Operating Temperatures | Energy Density of the Bed | Prototype Energy Density | Storage Capacity kWh | Discharge Power kW | Discharge Time h | References |
| Mesoporous silicates | Charge: 88 °C Discharge: 42 °C | 119 | 52.3 | 27.4 (808 kg of anhydrous silica gel). | 2.87 | 9.5 | [118] |
| Charge: 88 °C Discharge: 42 °C | 50 | 33.3 | 13 | 0.5–1 | 13–26 | [6,121] | |
| Charge: 180 °C Discharge: 30 °C | 180 | 57.8 | 1 | 0.8–1.8 | 0.56–1.25 | [6,122] | |
| Zeolite 4A–H2O | Charge: 180 °C Discharge: 35 °C | 160 | 120 | 12 kWh (70 kg of anhydrous 4A zeolite) | 1–1.5 | 8–12 | [6] |
| Zeolite 13X–H2O | Charge: 130 °C Discharge: 65 °C | 124 | NA | 1300 | 135 | 9.6 | [121,123] |
| Zeolite H2O | Charge: 135 °C Discharge:140 °C | NA | NA | 2400 (14 t of dehydrated zeolite) | NA | 10 | [124] |
| Zeolite H2O | Charge: 180 °C Discharge: 60–50 °C | 140–220 | NA | NA | 134 | NA | [29,118,125] |
| Activated carbon Methanol | Charge: 95 °C Discharge: 35 °C | 60 (Simulated, bed energy density) | NA | NA | NA | NA | [126,127] |
| Absorption | |||||||
| NaOH/H2O | Charge:100–150 °C Discharge: 40–65 °C | 250 | 5 | 8.9 | 1 | 8.9 | [41] |
| LiCl/H2O | Charge: 46–87 °C Discharge: 30 °C | 253 | 85 | 35 | 8 | 4.4 | [84] |
| CaCl2/H2O | Charge: 70–80 °C Discharge: 21 °C | NA | 116 (data from simulations) | 15 | 0.03 −0.560 | 27–500 | [121] |
| LiBr/H2O | Charge: 75–90 °C Discharge: 30–38 °C | 251 | NA | 8 | 1 | 8 | [89,128] |
| CaCl2/H2O | Charge: 95 °C Discharge: 35 °C | NA | 200 (Simulation, prototype energy density) | NA | NA | NA | [8] |
| Reaction Equations | Operating Conditions °C | Heat Storage Density | Characterization Level |
|---|---|---|---|
| MgSO4·7H2O → MgSO4 + 7H2O | Charge: 122–150 Discharge: 122 | 1512 of MgSO4 (theoretical) | Material scale ECN project: Characterization, experimental tests, a sample of 10 mg |
| MgCl2·6H2O → MgCl2·2H2O + 4H2O | Charge: 115–130 Discharge: 35 | 2170.8 of MgCl2·2H2O | Material scale IEC Project: Material characterization. Sample of 250 mg. Stabilization with zeolite 4A is to be further considered. |
| MgCl2·6H2O → MgCl2·H2O + 5H2O | Charge: 150 Discharge: 30–50 | - | Material scale ECN project: Material characterization, a sample of 300 g of material. |
| CuSO4·5H2O → CuSO4·H2O + 5H2O | Discharge: 40–60 (heat supply at T ≥ 40 to ignite discharge) | 2066.4 of CuSO4·H2O (theoretical) | ITW project: Material characterization, a sample of 100 mg |
| CaCl2·2.3H2O → CaCl2 + 2.3H2O | Charge: 150 Discharge (temperature lift): T = 6.2 (reactor and evaporator both at 25) T = 10 (reactor at 50 and evaporator at 10) | - | ECN project: Material characterization, a sample of 40 g |
| Bentonite + CaCl2 | Discharge: 35 | 667 of composite material | ITW Project: Material characterization |
| Kal(SO4)2·12H2O → Kal(SO4)2·3H2O + 3H2O | Charge: 65 Discharge: 25 | 864 of Kal(SO4)2·3H2O | Reactor scale: PROMES CEA-INES Project, 25 kg of Kal(SO4)2·12H2O |
| Al2(SO4)3·18H2O → Al2(SO4)3·5H2O + 13H2O | Charge: 150 Discharge (temperature lift): T = 9.8 (reactor and evaporator both at 25) T = 10 (reactor at 50 and evaporator at 10) | - | Reactor scale. ECN Project: Sample of 40 g |
| Na2S·5H2O → Na2S·1.5H2O + 4.5H2O | Charge: 83 Discharge: 35 | 2808 of Na2S·1.5H2O | Reactor scale, ECN project: SWEAT prototype, 3 kg of material. |
| SrBr2·6H2O → SrBr2·H2O + 5H2O | Charge: 70–80 Discharge: 35 | 216 of SrBr2·H2O | Reactor scale, PROMES CEA- INES Project: SOLUX |
| CaCl2·2H2O → CaCl2·H2O + H2O | Charge: 95 Discharge: 35 | 720 of CaCl2·H2O | Reactor scale, BEMS: a theoretical study |
| Reaction Equations | Reaction Temperature, °C | Energy Storage Density | Advantages | Drawbacks |
|---|---|---|---|---|
| Ammonia decomposition material | 400–700 | 67 kJ·mol−1 |
|
|
| Inorganic hydroxide material | 350–900 | 300 kWh·m−3 |
|
|
| Inorganic hydroxide material | 100–167 | 380 kWh·m−3 | ||
| Methane reforming material | 700–860 | 247 kWh·m−3 |
|
|
| Methane reforming material | 600–950 | 250 kWh·m−3 | ||
| Carbonate decomposition material | 700–1000 | 692 kWh·m−3 |
|
|
| Metal hydride material | 250–500 | 75 kJ·mol−1 |
|
|
| Redox material | 127–1027 | 77 kJ·mol−1 |
|
|
| Redox material | 700–850 | 205 kJ·mol−1 |
| Dashboard 1: Open System | Dashboard 2: Closed System |
|---|---|
| Kinetic equation Mass equation Energy equation | Kinetic equation Mass equation Energy equation |
| Classification | Reactors | Subclassification | Reactor Description | Reactor Efficiency | Whole System Efficiency | Prototypes References |
|---|---|---|---|---|---|---|
| System involved | Indirect reactor | - | The material reservoir is directly heated to either heat the material or the heat transfer fluid by conduction. | 21—41% | 10–20% | [140,141,142,143] |
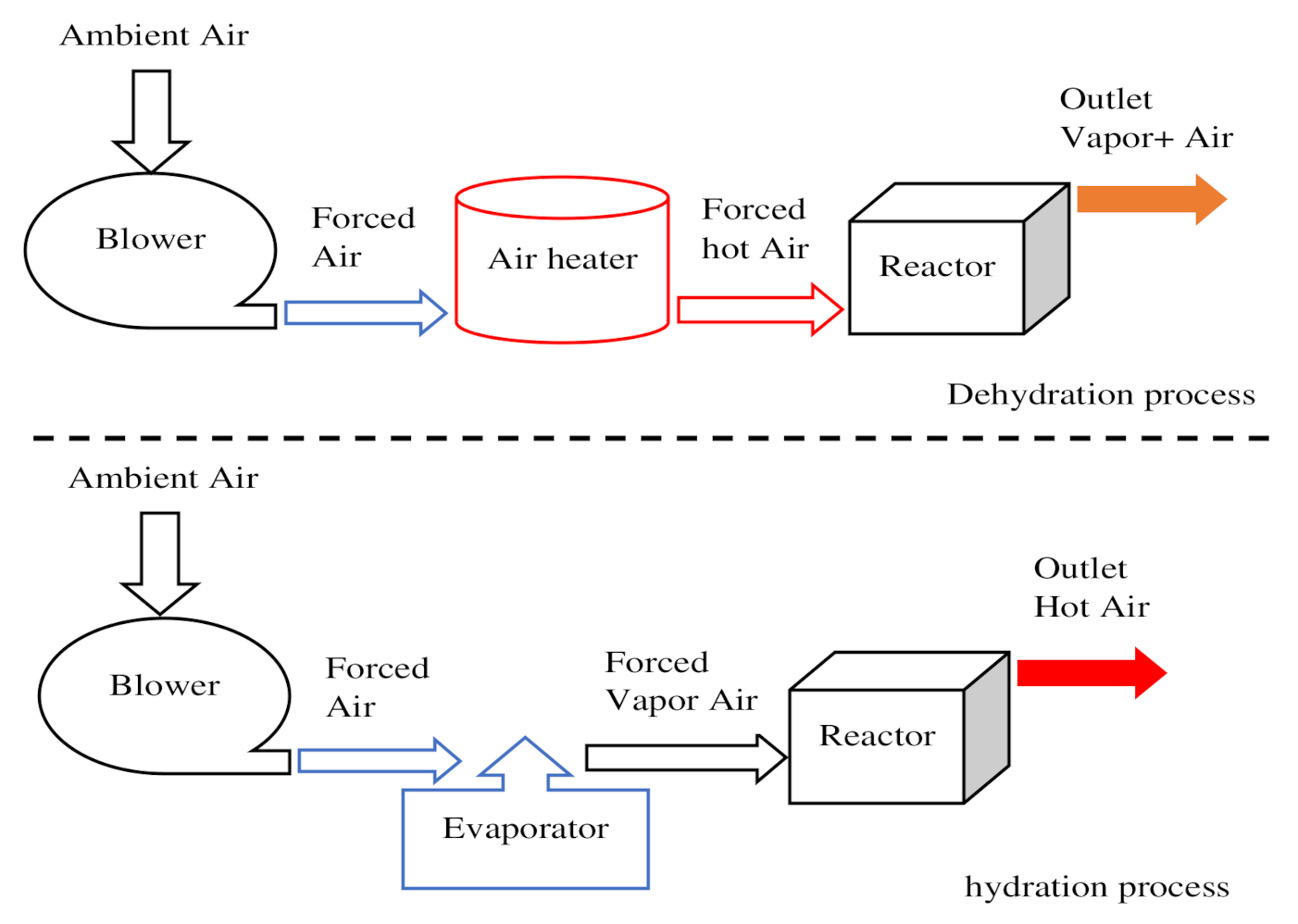
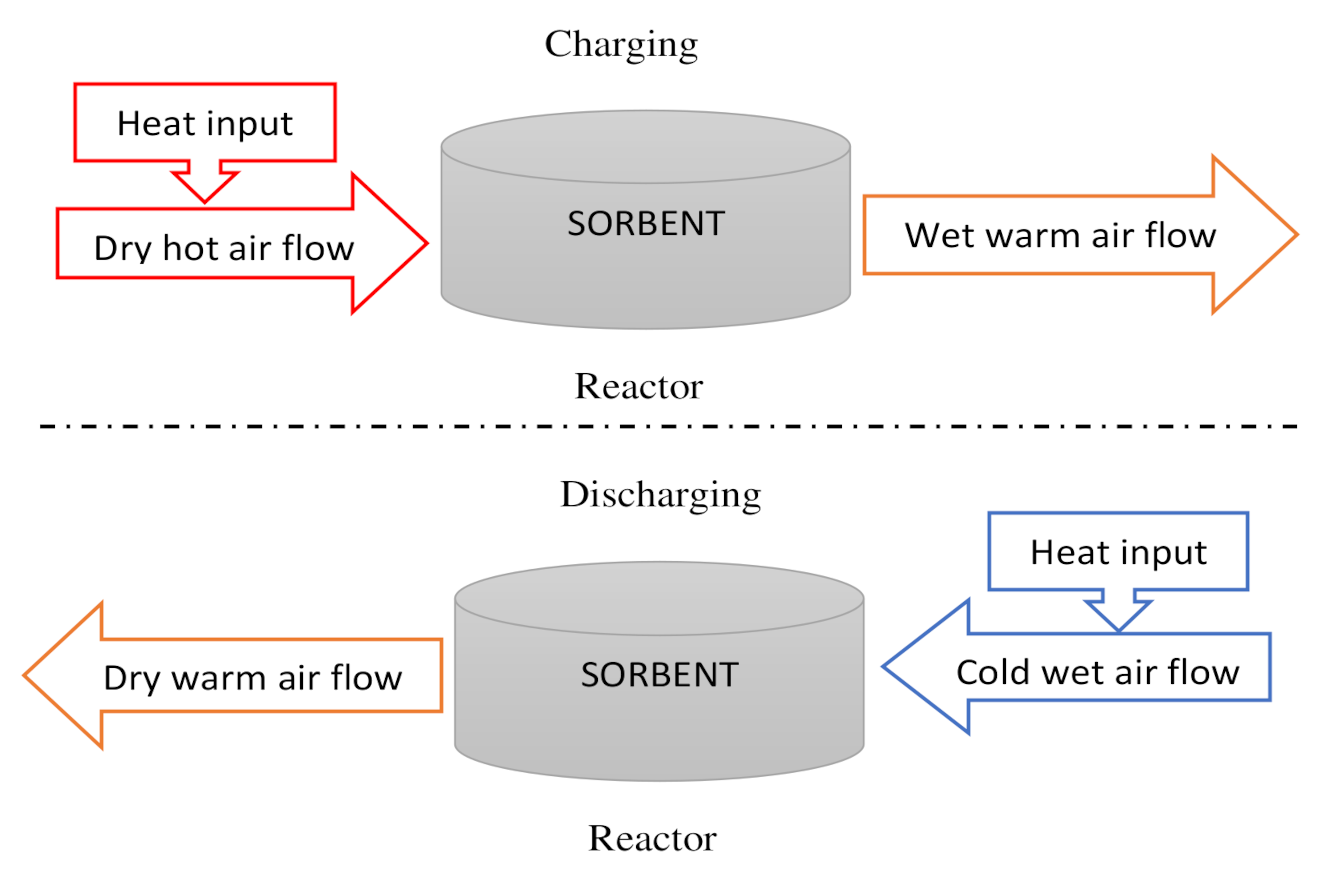
| Direct reactor | Open reactor | Reactants are heated by heat source input through an opened receiver aperture. Moist air atmospheric acts as a mass and heat carrier fluid. Use of gas diffusers to supply or collect the moist air (Figure 6 and Figure 7). Use of external heat exchanger to carry out the heat. | 25–50% | 15–35% | [144,145] | |
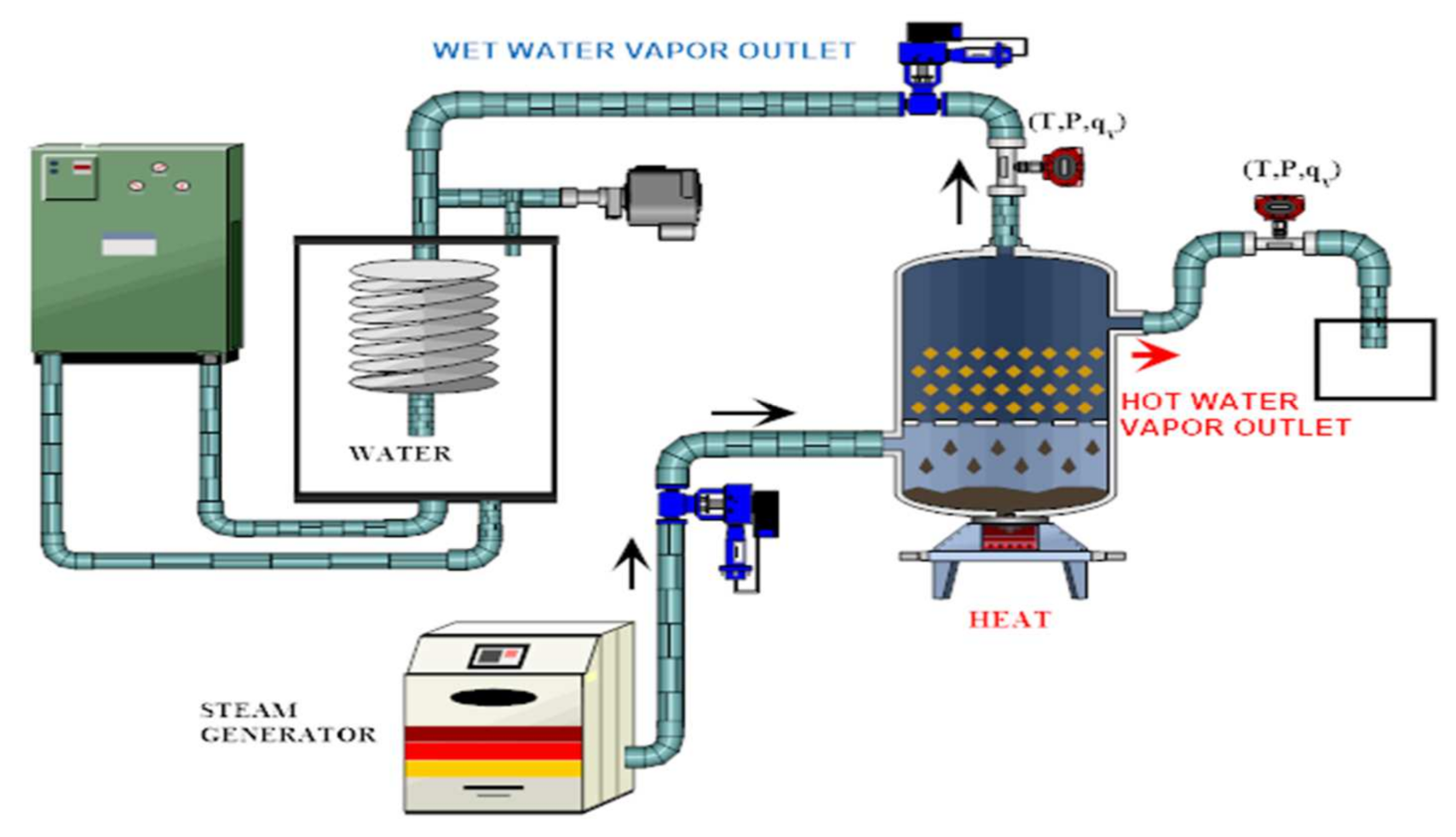
| Closed reactor | Reactants are heated by heat source input through a closed receiver aperture and the system is isolated from the atmospheric environment. Use of a gas diffuser and an internal heat exchanger to collect or supply the heat of the reaction. An evaporator is required to generate steam for the hydration phase and a heat source (Figure 8) | 40–64% | 15–40% | [146,147,148,149] | ||
| Reactors type | Stacked bed reactor | Fixed bed reactor Mobile bed reactor Rotary bed reactor | The material is packed inside the reactor heated by air/HTF flowing through the material bed/heat exchanger. The heat exchanger is used to carry out the heat of the reaction, and the material is replaced after full conversion is achieved. The HTF can flow cocurrent upflow/downflow or countercurrent. | 12–69% | 12–42% | [26,127,130,145,150] |
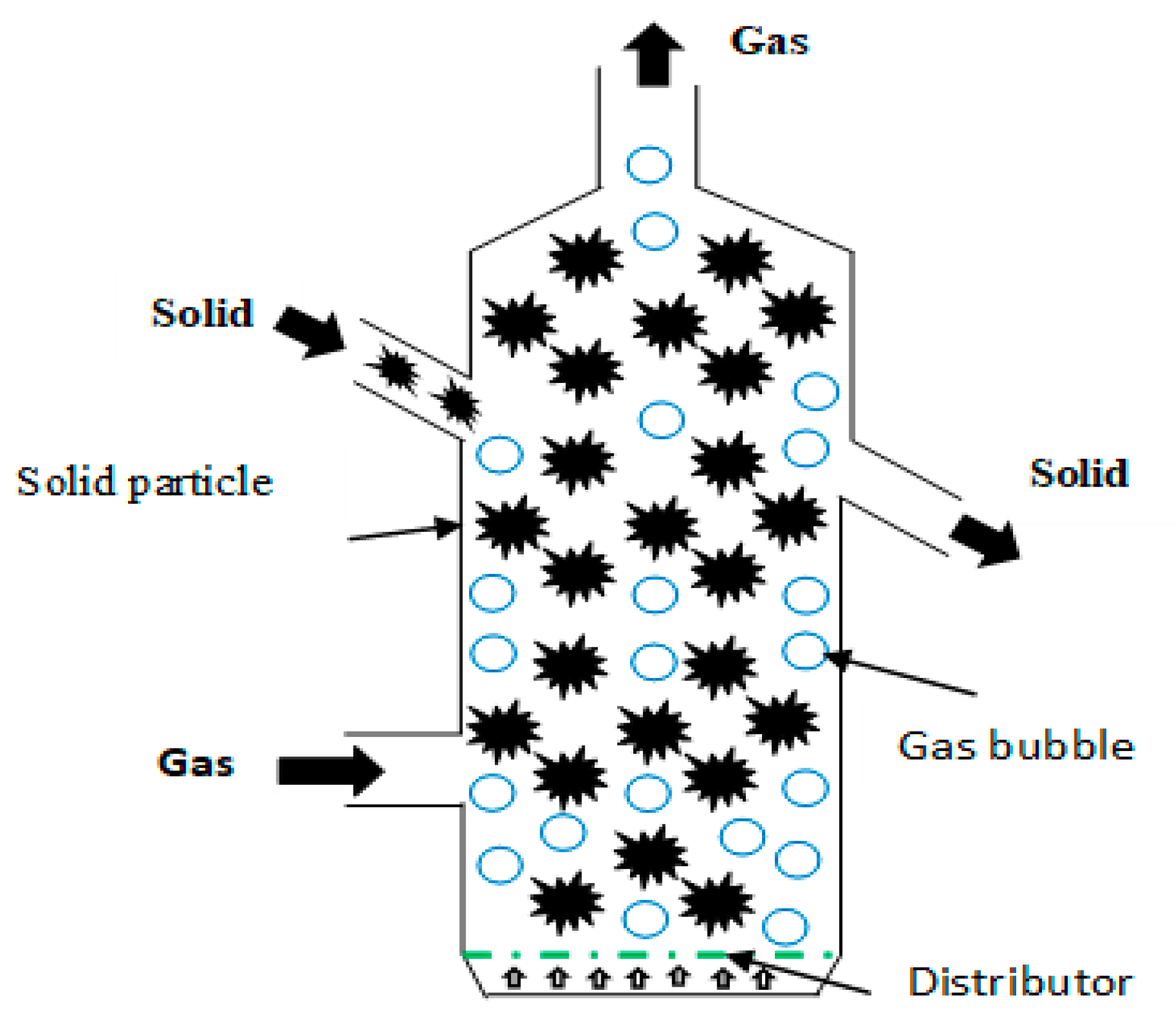
| Fluidized bed reactor | Vibrated bed reactor Blown bed reactor | The material is fed in the reactor and the fluid is fed through granular solid material. The high velocity of the fluid generates a suspension of material particles that act as if they were a fluid, which increases the quality of the heat transfer (Figure 9). | 15–75% | 20–60% | ||
| System components | Separate reactor | Dissociation between the thermal power and the storage capacity. There is no need for a steam or heat exchanger integrated into the reactor. Only the required amount of reactant is heated. | 28–75% | 20–65% | [26,151,152,153,154,155,156,157,158,159,160,161,162,163] | |
| Integrated reactor | Two phases reactor | Contains the material and the sorption pair reactant (air or steam). Need of heat exchanger to carry out the heat of the reaction. | 14–65% | 15–55% | [127,164,165,166] | |
| Three phases reactor | Contains the material and the sorption pair reactant (air or steam). The addition of a second working fluid is disconnected from the other and acts as a heat exchanger. Addition of material transport system. | 14–85% | 15–65% |
| Operating Circuit | Operating Pressure | Heat and Mass Transfers and Heat Storage | Design and Dimensioning |
|---|---|---|---|
| Closed system | The necessity of an evaporator to produce steam during hydration reaction |
| Strong technological constraints and manufacturing for the reactor and the evapo/condenser design. |
| Open system | The steam is provided by moist air coming from the environment |
|
|
| Investigation (Authors) & Years | Nature and Purpose | Storage Process | Progress & Contribution | Storage Materials | Reactor Type | Storage Density/ Storage Temperature | Descriptions | Refs. |
|---|---|---|---|---|---|---|---|---|
| Zhang et al., (2014) Shanghai Jiao Tong University (2020) | Space heating and domestic hot water Lab-scale | Absorption | Single-stage absorption; Multiple functions using: production of child water at 7 °C, heating water at 43 °C for space heating and domestic hot water at 65 °C | Integrated | 42, 88, and 110 respectively for child, heating, and hot water | Single or multiple storage tanks (Two or more) can be integrated with the absorption chiller/heat pump. Both the refrigerant and solution might be stored simultaneously. The refrigerant storage is in association with a condenser. The weak solution storage is in association with an absorber, and the strong solution is in association with the generator. | [43,169] | |
| French National Research Agency (2017) | Space heating Reactor scale, prototype, commercial | Absorption | Development of low-temperature heat storage system | Closed | = 60 °C | Four main components: a desorber, an absorber, a condenser, and an evaporator, Two solution storage tanks (for the diluted and concentrated solution) and an absorbate storage tank. The solution can crystalize in the solution storage tank in this process. A sandwich grooved vertical plate configuration was then chosen for the heat exchanger | [50] | |
| EMPA, COMTES (2014) | Space heating Reactor scale, prototype, commercial | Absorption | Development of commercial prototype energy storage system Achievement of an increase in volumetric energy density compared to hot water storage | Closed | 250 | Two chambers are connected Chamber 1 contains the sorbent and functions as desorber during charging and as absorber during discharging. Chamber 2 works as condenser and evaporator, respectively, containing the sorbate. | [170] | |
| Fopah-Lele (2015) | Numerical investigation Space heating | Adsorption | Modeling of the charging and discharging phase of a storage system with low thermal energy. Study of the influence of the performance of parameters such as temperature, pressure, and heat transfer coefficient on the charging process. 3D view of the heat transfer and the behaviors of the reactor during the process. | Closed | 531.77 | Numerical analysis of two types of heat exchanger: plate-fin and helical coil heat exchangers embedded in a . A reactor based on a honeycomb heat exchanger concept was design. The model is solved with the COMSOL Multiphysics software. The analytic results were implemented on a lab scale and prototype for validation of the model. | [138] | |
| Q. Ranjha (2017) | Numerical investigation Industrial process | Adsorption | Heat and mass transfer 3D simulation using a novel structure of reactor with powders. Optimization technic for selecting the appropriate structure of the reactor. | Closed | = 550 °C | Indirectly heated fixed reaction beds of circular and rectangular cross-sections; heat transfer fluid could flow in co-current, counter-current, or cross-flow to the reaction gas; COMSOL Multiphysics software is used for the simulation of the reactor. | [137] | |
| COMTES (2012) | Lab-scale prototype Space heating | Adsorption Absorption | Development of liquid and solid sorption systems for seasonal heat storage purposes | Zeolite 13XBF | Closed fixed bed | = 75 °C | Closed modular solid sorption system with an additional backup heater; prototype with a reactor of approximately 300 L and 164 kg. | [117] |
| Shanghai Jiao Tong University (2017) | Pilot-scale Space heating | Adsorption | Locomotive air conditioning system enhancement | Zeolite 13X | Closed | = 125 °C | Closed sorption system; the heat source is provided by high-temperature gas exhausted from an internal combustion engine; use of one adsorber and a cold storage tank; the cooling effect is transferred to the cabin by chilled water. | [119] |
| Lauren Farcot et al., (2019) | Building heating | Adsorption | Study of the impact of the air humidity at the reactor inlet on the reactor performances. The feasibility of continuous thermochemical heat storage in a moving bed reactor with hydrated salt. | / | Open moving bed | = 41 °C | Moving bed reactor with a wall in stainless steel to avoid corrosion. The air diffuser and collector are separated from the reactor by stainless steel. The cross-section of the reactor can be adjusted (reduced) by adding stainless steel walls along with the mesh. A stainless-steel reservoir receives the salt that falls from the rotary valve at the bottom of the reactor. Air can be flown through the reactor at a temperature between 0 and 100 °C. | [38] |
| Abanades (2018) | Industrial process | Chemical | Design and demonstration of a high temperature solar-heated rotary tube reactor for continuous particle calcination. | Indirect rotary tube | 500–1600 °C | The reactor is composed of a cavity-type solar receiver for radiation absorption; external heating by concentrated solar energy is provided; indirect heating of the reactants; the heat is transferred to a rotary tube, and the reactive particles are continuously injected into the rotary tube. | [130] | |
| Zhejiang University of Technology (2018) | Power generation | Chemical | Design of a methanation reactor for producing high-temperature supercritical carbon dioxide in solar thermochemical energy storage | Tubular packed bed | 700 °C | The system is composed of an endothermic and exothermic reactor at the inlet and outlet, respectively, each connected to a heat exchanger. Two materials vessels are used. The first to provide the methane to the endothermic reactor for decomposition under solar heating. The second is to store the product of the decomposition, which is pumping in the second reactor for production. | [171] | |
| Y. A. Criado (2014) | Power generation | Chemical | Analyze a thermochemical energy storage process using a hydroxide calcium chemical loop. | Fluidized bed reactor | 260 | Use of a single fluidized bed reactor alternating from hydration to dehydration conditions and two solid storage silos feeding solids continuously to the fluidized bed. Operation at atmospheric pressure is assumed for simplicity. | [164] | |
| Edwards et al., (2012) | solar power plants | Chemical | Developed a calcium looping CSP plant. Determined the operating conditions required to achieve satisfactory operation of the plant. | Fluidized bed reactor | 875 °C | Use of two reactor units operating independently. A solar calciner using concentrated solar energy to start the reaction. A carbonator recombines the product of the reaction thus releasing heat to the turbine. | [172] | |
| General Atomics (2O11) | Power generation | Chemical | Modified a storage cycle to yield elemental sulfur as a by-product, which is then stored and later used as a combustible to generate power. | Sulfur-Based | 1200 °C | Use of two distinct turbines. The first one is actuated by the flue gas leaving the combustion chamber. Use of heat exchanger to drive the heat to the second turbine powering a Rankine cycle. | [173] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gbenou, T.R.S.; Fopah-Lele, A.; Wang, K. Recent Status and Prospects on Thermochemical Heat Storage Processes and Applications. Entropy 2021, 23, 953. https://doi.org/10.3390/e23080953
Gbenou TRS, Fopah-Lele A, Wang K. Recent Status and Prospects on Thermochemical Heat Storage Processes and Applications. Entropy. 2021; 23(8):953. https://doi.org/10.3390/e23080953
Chicago/Turabian StyleGbenou, Tadagbe Roger Sylvanus, Armand Fopah-Lele, and Kejian Wang. 2021. "Recent Status and Prospects on Thermochemical Heat Storage Processes and Applications" Entropy 23, no. 8: 953. https://doi.org/10.3390/e23080953
APA StyleGbenou, T. R. S., Fopah-Lele, A., & Wang, K. (2021). Recent Status and Prospects on Thermochemical Heat Storage Processes and Applications. Entropy, 23(8), 953. https://doi.org/10.3390/e23080953






