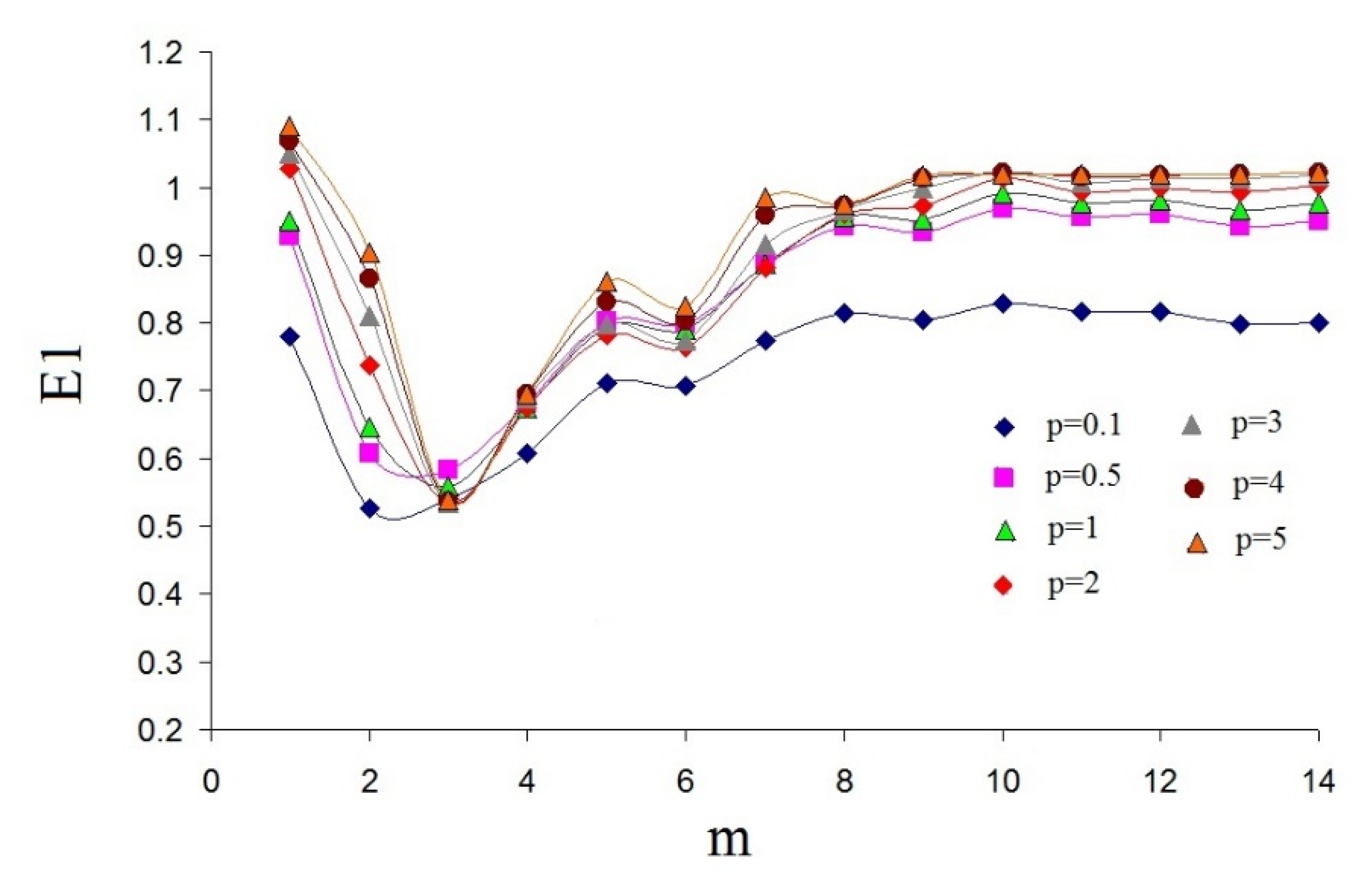
1. Introduction
In modern society, heart disease is one of the major causes of mortality [
1].Most clinical research in cardiology is based on the analysis of electrocardiograms (ECGs). One important characteristic of an ECG is the duration of one cardiac cycle, namely, the R-R interval. Indeed, R-R interval time series may contain information that indicates the presence of certain cardiovascular diseases. There has been considerable attention devoted to investigating the various aspects of the cardiac physiology using methods for nonlinear analysis, such as fractal analysis [
2,
3,
4], chaos theory [
5,
6,
7,
8,
9], and others [
10,
11]. One of the goals of such studies is to determine the most effective parameters for building expert systems for the diagnosis (and differentiation) of cardiac diseases. This is a particularly important issue, especially when creating modern portable devices for monitoring cardiac activity. Therefore, it is necessary to develop mathematical tools for non-linear analysis that can discriminate between healthy physiological and pathological R-R interval time series.
Many biomedical signals, such as cardiac signals, have complex structures representing a combination of simpler structures. These interactions reflect the influence of numerous vital processes. For example, recent studies have shown that many biomedical signals have a multifractal structure [
12,
13,
14,
15]. Such signals represent a complex fractal structure, which cannot be sufficiently characterized by a single summary value (e.g., the Hurst exponent). Multifractal signals are a combination of various simpler monofractal structures, each characterized by a single Hurst exponent. Therefore, to characterize a multifractal signal, the spectrum of generalized Hurst exponents is used, where each generalized Hurst exponent characterizes a certain monofractal structure.
To obtain this spectrum from the signal, Kennel et al. [
16] suggested applying the
q-th order fluctuation function, where
q 0. This procedure is called a multifractal detrended fluctuation analysis (MFDFA). Therefore, the MFDFA procedure for a stochastic time series {
X(
t)} consists of calculating the
q-th order fluctuation function [
12,
16]:
where the square bias
is defined over each sliding window of interval
, and time series {
} is determined in the following way:
where
defines the cumulative moving average for
, …,
;
—a line of best fit over each sliding window of length
;
is the number of points; and
, where
= 2, 3, 4, …,
[
12].The value
depends on the maximum size of the time series. In order to reduce the saturation effects owing to the finite size, it should be
[
12]. The ln–ln plot of
, as a function of
, yields a straight line with slope
h(
q), defined as the generalized Hurst exponent
h(
q).
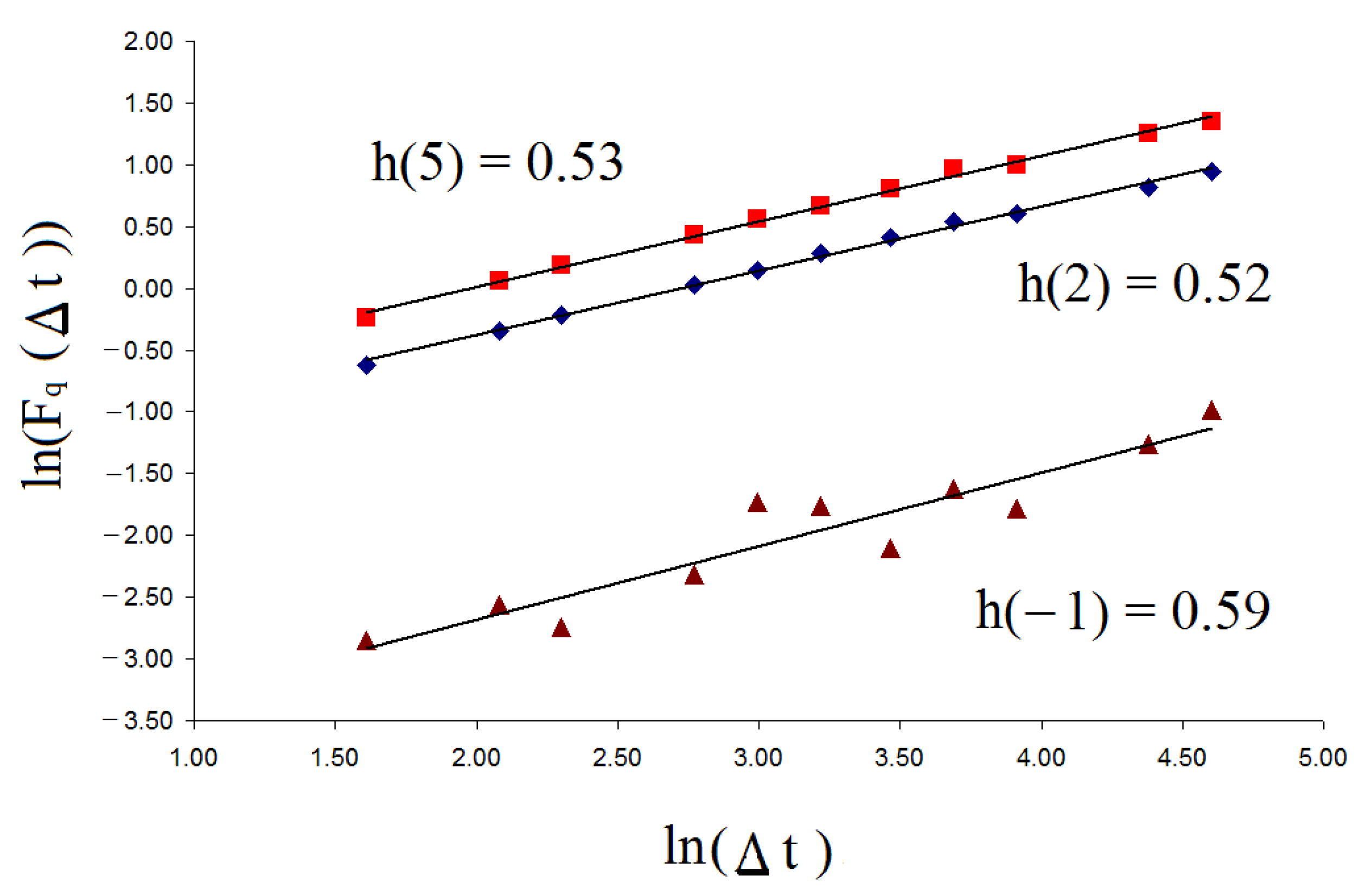
Figure 1 shows the ln–ln plot of
for random time series data (white noise) for
q = −1, 2, and 5.
The generalized Hurst exponent
h(
q) indicates the multifractal property of a signal. For a monofractal signal,
h(
q) is a constant, equaling the Hurst exponent
h(
q) =
H. Conversely, for a multifractal signal,
h(
q) decreases as
q increases. The singularity spectrum is assessed using the succeeding relation:
where
defines the strength of a singularity spectrum and
is the fractal dimension of a points set with a particular
value. For a monofractal signal, the series
is converted into a single point. The measure of degree of multifractality is estimated by the width of its spectrum, which evaluates the range of
where
:
where
. Larger values for the width indicates a higher level of multifractality of the spectrum [
12,
16,
17].
This approach is a powerful tool for analyzing biomedical signals. In [
13], MFDFA was applied to a human gait time series to compare and contrast the pathology and non-pathology group. The results of the research suggested that the degree of multifractality is higher for non-pathology groups. Using the multifractal spectrum of electroencephalogram (EEG) signals, the authors of [
14] developed a technique for the automated detection of epilepsy, and in [
15], the analysis reveals interesting results on neural activation of the alpha (8–12 Hertz) and theta (4–8 Hertz) brain rhythms while listening to simple acoustical stimuli. This research demonstrates the ability to qualify emotions using MFDFA.
The work [
12] shows the high efficiency of applying the multifractal approach for analyzing R-R interval time series. Therefore, using the
q-th order fluctuation function, we built a two-factor logistic regression model for diagnosis of the pathology of cardiac signals with an area under the receiver operating characteristic curve (ROC curve) of 0.96 (95% CI 0.92–0.99).These results serve as inspiration for creating a four-factor logistic regression model that is able to differentiate between congestive heart failure (chf) signals from other pathologies, namely, the atrial fibrillation (af) and sudden death (sd) groups. Here, the area under the ROC curve is 0.91 (95% CI 0.84–0.97).
However, unlike fractal analysis, which permits the structure of signals to be evaluated, chaos analysis explores the base dynamics of the system, which forms the observed signal. Therefore, fractal analysis does not allow a more detailed study of the dynamic properties of the system [
18]. Therefore, it is necessary to develop mathematical tools for chaotic analysis which has the power to differentiate between healthy and pathological signals (and moreover, finer differentiation across different subclasses of the pathology).
Chaotic analysis usually begins with reconstructing the phase space of a dynamical system. For reconstructing the phase space of a dynamical system, two parameters, specifically, the embedding dimension
m and time delay
J, are used [
19,
20].The time delay
J is usually estimated either by examining the autocorrelation function (acf) [
20] or mutual information (MI) [
21]. The estimate of the time delay
J is the smallest possible value that produces reconstructed attractors whose coordinates are as independent as possible (as measured by correlation or MI). Using the acf, the time delay
J is generally chosen in accordance with the lag, where the absolute value of the acf first attains a zero (or close to it) and then flattens out. This method is quite simple and does not require massive calculations. Moreover, for Gaussian signals, a zero autocorrelation for some lag
J or beyond implies the independence of observations at time points with absolute lag of at least
J. However, for non-Gaussian signals, a zero correlation does not necessarily imply independence. For this reason, the delay
J derived from the autocorrelation may produce misleading results.
Fraser and Swinney [
21] have demonstrated that the MI function is a more accurate measure of independence compared to the acf. In the method proposed by Fraser and Swinney, the optimal time delay
J is selected according to the first minimum MI function. Liebert and Schuster [
22] demonstrated that the minima of the MI function match the minima of the correlation integral function, which requires less computation than the MI function [
22]. Therefore, the application of the correlation integral function is more convenient for practical use.
The following approaches can be used to estimate the embedding dimension: The false nearest neighbor (FNN) method [
23], Cao’s method [
24], or the correlation dimension method [
25,
26]. The idea of the FNN method was developed using the factthat when the embedding dimension is small, points that are distant in the original phase space are brought together in the reconstruction space [
24]. If
m is defined as an embedding dimension by the embedding theorems [
27,
28], then the
true neighbors are any two points which are close in the
m-dimensional reconstructed space and that remain close in the (
m + 1)-dimensional reconstructed space. On the contrary, there are
false neighbors. However, the FNN method is not accurate enough to determine the parameters
and
.Therefore, this approach may lead to incorrect estimation of the embedding dimension. Cao’s method [
24] overcomes the shortcomings of the nearest neighbor method, which makes it more attractive for practical applications such as cardiac signals.
One of the conditions for the chaotic state of a dynamic system is sensitivity to the values of the initial conditions [
19,
20]. The largest Lyapunov exponent (
LLE) is often considered as one quantitative measure of this sensitivity. The largest Lyapunov exponent characterizes the degree of exponential divergence of close trajectories [
20]. The presence of a positive Lyapunov exponent in the system indicates that any two close trajectories quickly diverge over time, that is, there is sensitivity to the values of the initial conditions. Therefore, determination of the Lyapunov exponent recognizes the existence of chaotic behavior [
19,
20].
It is understood that many biomedical signals have a complex chaotic structure, which is the result of the interaction of various chaotic systems involved in the regulation of vital processes in the body. This complex structure is a combination of simpler chaotic structures. To calculate the characteristic values of these chaotic structures, the non-Euclidean norms can be used. The family of Minkowski norms [
29,
30] are parameterized by their exponents
p = 1,2,…:
Generalizations of Minkowski norms are presented in [
29] for the event where
p is a positive real number. For
p 1, those extensions are actually norms, but for 0 <
p < 1, the triangle inequality does not remain and they cannot be regarded as norms. Therefore,
p-norms with
p < 1 were named fractional norms. Below, we denote the Minkowski norm and a fractional norm as
p-norm
.
In this paper, to characterize the chaotic structure, we propose only considering the largest Lyapunov exponent. Therefore, we will not consider the entire spectrum of values of the Lyapunov exponent for a given chaotic structure, calculated for the corresponding p-norm , which will greatly simplify the calculations. Each p value of p-norm will correspond to its largest Lyapunov exponent LLE(p). Therefore, a complex chaotic structure will be characterized by the spectrum of LLE(p) for p(0;). If the entire spectrum of LLE(p) values is characterized by a single value, then this structure will have mono-chaotic behavior. If some variability of the LLE(p) values is observed, then the considered structure is said to possess multi-chaotic behavior.
We believe that the R-R interval time series is a combination of various chaotic structures that results in the interaction with the regulation of cardiac activity. To evaluate the chaotic structure, we propose estimating the largest Lyapunov exponent using the
p-norm
, based on the well-known method proposed by Rosenstein et al. in [
20].This approach can allow us to identify the multi-chaotic behavior of R-R intervals. Consequently, we anticipate improvement in the differentiation of the chaotic properties of R-R intervals for healthy and non-healthy subjects compared to the standard approach, proposed by Rosenstein et al. in [
20]. To demonstrate the effectiveness of the new approach, we will construct logistic models for differentiating R-R intervals for healthy and non-healthy subjects. In summary, the goal of this study is to design a new approach for identifying the multi-chaotic behavior of time series and demonstrate its effectiveness for evaluating the multi-chaotic properties of R-R intervals.
3. Results
We will now investigate the feasibility of applying multi-chaotic analysis to reveal any possible difference among R-R interval time series of healthy subjects (hs), subjects diagnosed with congestive heart failure (chf), and subjects diagnosed with atrial fibrillation (af). To estimate the delay time
J along with the widely used methods of the acf and the MI function, we used the correlation integral function
, derived from [
20]. The embedded dimension
m was evaluated by applying the method in [
24].
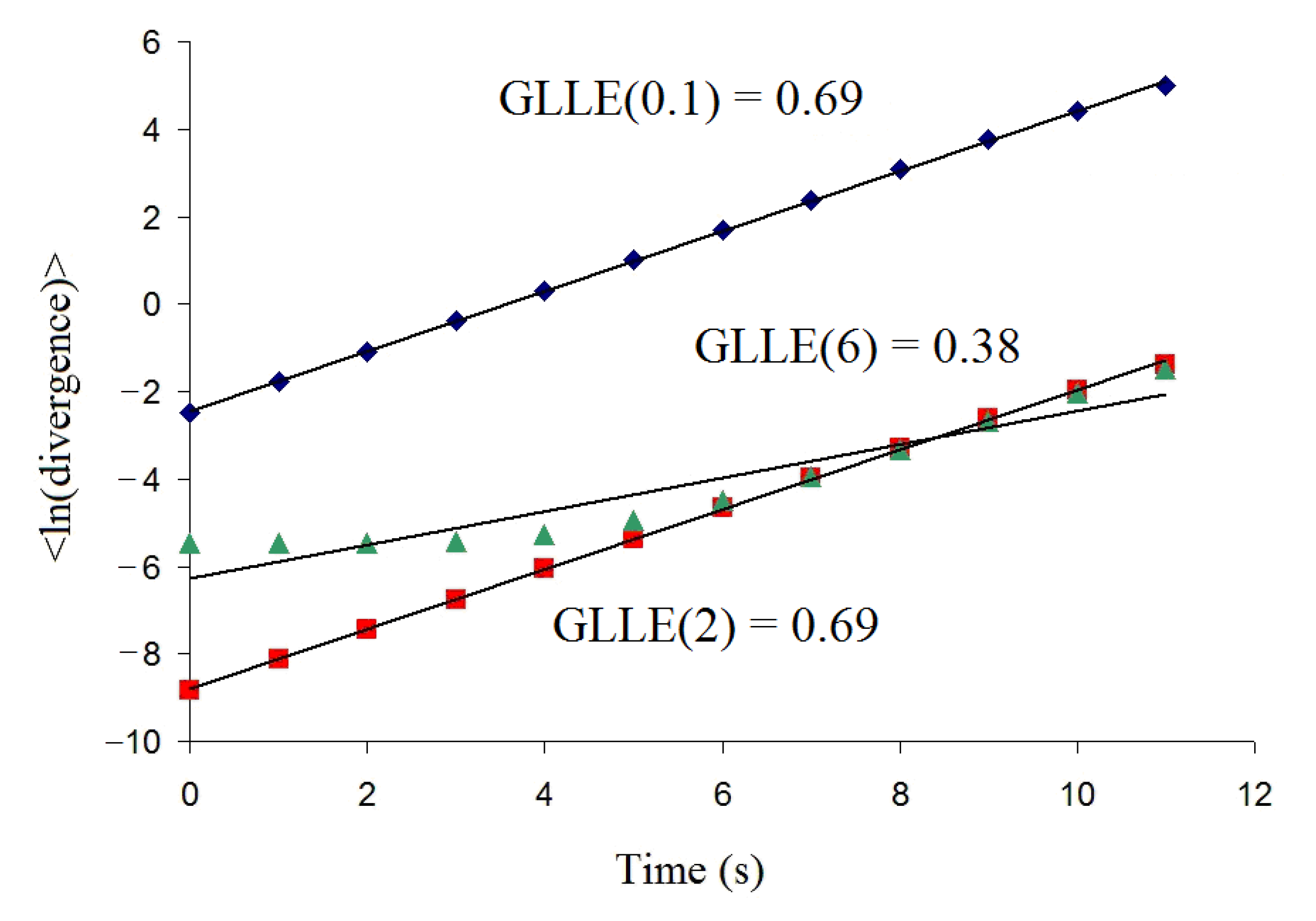
Appendix A contains graphs demonstrating the results of applying this method for one of the R-R intervals recorded. Preliminary estimation of the interval of
p-norm values showed us that
p[0.1; 5] is the most appropriate interval for assessing the
GLLE (p) of R-R interval time series.
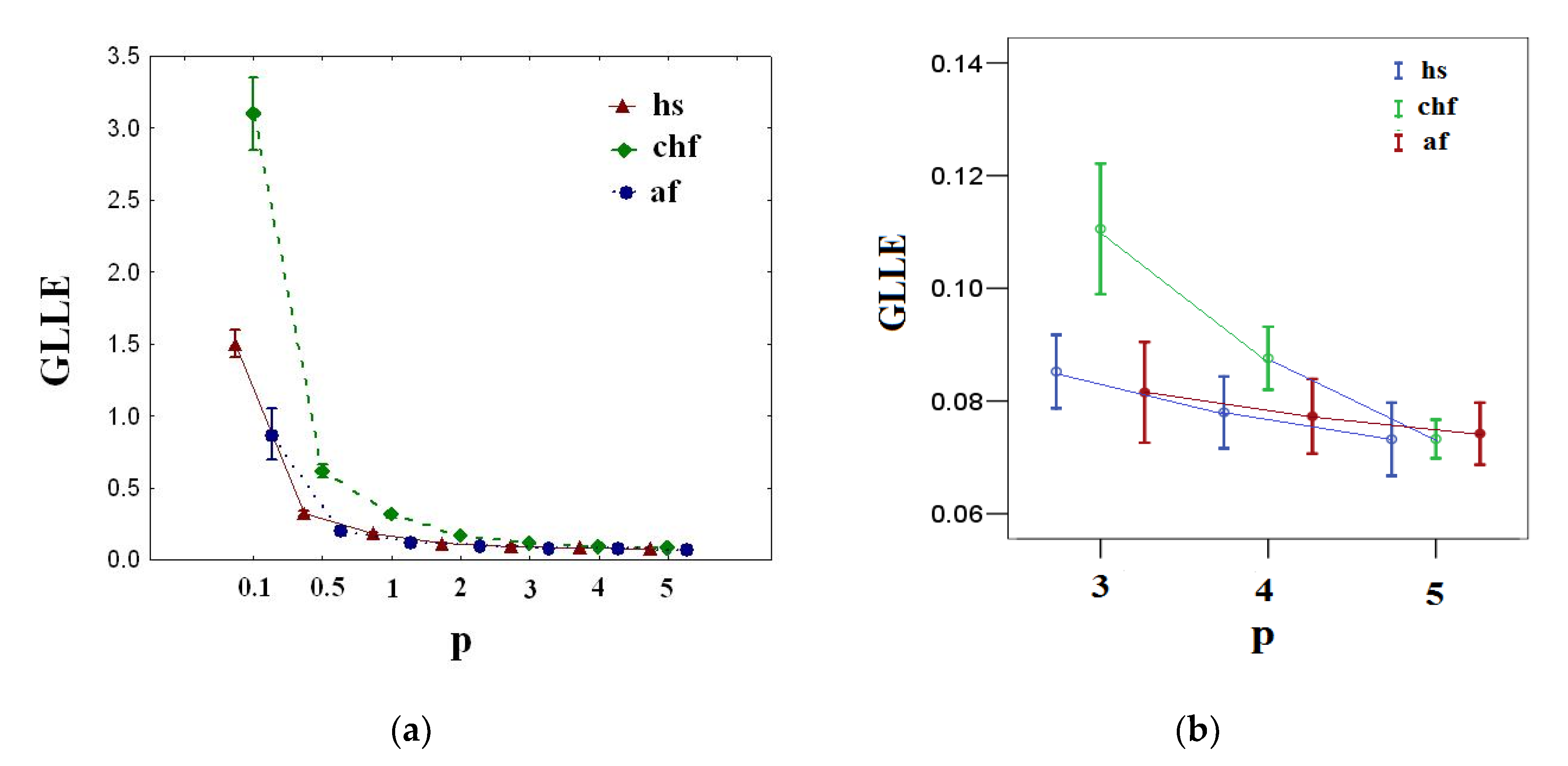
Figure 3 shows the results of calculating
GLLE(p) for healthy (hs), congestive heart failure (chf), and atrial fibrillation (af) groups.
From
Figure 3a, it is clear that there is variation of
GLLE(p) with
p for healthy (hs), congestive heart failure (chf), and atrial fibrillation (af) groups, where the values of
GLLE(p) decrease with increasing
p. This indicates the multi-chaotic behavior of the R-R interval time series considered.
Figure 3b demonstrates that there is no statistically significant difference (
p-value > 0.05) among groups for
GLLE (5). Therefore, for multi-chaotic analysis of the considered signals, it is sufficient to use
p-norm
5.
To characterize multi-chaotic behavior, let us consider estimation of the spectrum width
W = GLLE−
GLLE. Since standard approaches analyze chaotic behavior using the Euclidean norm, given the importance of this issue for the chaotic analysis of R-R interval time series, we separately consider the values of the largest Lyapunov exponent
GLLE(2). Table 4 shows the results achieved for the largest Lyapunov exponent
GLLE(2) and estimation of the spectrum width
W. Since the empirical distribution of the estimated values differs from a normal distribution (
p-value < 0.05, Kolmogorov–Smirnov test), we proceeded to use the median (
Me) in order to evaluate the central values of
GLLE(2) and
W, and applied the first quartile
and third quartile
to evaluate the variation or spread of the distribution of the considered values.
From
Table 4, note that the chf group
GLLE(2) = 0.15(0.12–0.20) is characterized by higher values (
p-value < 0.05, ANOVA F-test) than hs
GLLE(2) = 0.11(0.10–0.13). The af group
GLLE(2) = 0.08(0.07–0.10) has lower values (
p-value < 0.05, ANOVA F-test) compared to the hs group
GLLE(2). These results clearly demonstrate that for the Euclidean norm, the chf heart rate control system is more sensitive to the initial conditions, whereas the af heart rate control system is less sensitive to the initial conditions compared to the hs heart rate control system. Nevertheless,
Figure 3b demonstrates that when increasing the
p-norm, these differences degrade, which indicates the important role of the SF component in the differentiation of the R-R interval time series.
Table 4 shows that pathology can be characterized by different spectral widths. Using the ANOVA method, we conclude that the spectrum width of the congestive heart failure group
= 2.61(1.70–4.23) is significantly greater (
p-value < 0.05, ANOVA F-test) than each ofthe healthy group
= 1.28(0.78–1.95) and atrial fibrillation group
= 0.42(0.26–0.79). The spectrum width
for the atrial fibrillation group has significantly lower values (
p-value < 0.05, ANOVA) than the healthy group
.Therefore, the R-R interval time series of the chf group is characterized by the largest difference between the SF and LF components, and the af group is characterized by the smallest difference. This result leads us to make an assumption about the different level of the random component in the groups of the studied signals. Therefore, we can infer from these findings that the random component dominates in the af R-R interval time series compared to the hs R-R interval time series. On the contrary, the contribution of the random component in the congestive heart failure R-R interval time series is smaller compared to in the hs R-R interval time series intervals.
It is widely accepted that a good indicator of the level of the random component of a chaotic signal is the correlation dimension
. Argyris et al. [
33] demonstrated that the correlation dimension increases when increasing the level of the random component of the chaotic signal. We now use this property of the correlation dimension to test our assumption about the level of the random component in healthy, congestive heart failure, and atrial fibrillation groups.
Table 5 shows the results achieved for the correlation dimension
.
Table 5 demonstrates that the correlation dimension of the af group
= 0.93(0.69–1.00) is significantly greater (
p-value < 0.05, ANOVA F-test) than each of the hs group
= 0.57(0.50−0.66) and chf group
= 0.55(0.17−0.85). This confirms our assumption that the variance contribution of the random component is larger in the af group R-R interval time series compared to the hs group R-R interval time series. However, no statistically significant difference (
p-value < 0.05, ANOVA F-test) was found between the correlation dimension of the hs and chf groups.
By setting up a logistic regression model [
34], we explored the capacity of multi-chaos analysis to discriminate between healthy group R-R interval time series and the pathological group R-R interval time series. Predictors of the logistic regression model were the attributes (GLLE_p) GLLE_0.1, GLLE_0.5, GLLE_1, GLLE_2, GLLE_3, GLLE_4, and GLLE_5. Considering that the input parameters of the model have a small range of changes, we multiplied them by a hundred. Therefore, the odds ratio can be estimated more efficiently. The data were separated into the training set (85 cases) and the testing set (38 cases). The output variable is denoted as Y = 0 if the subject belongs to the healthy group and, accordingly, Y = 1 if the subject belongs to the pathology group. To choose the minimum set of factor attributes, a stepwise-variable selection (SVS) method was applied [
12,
34]. Therefore, five attributes, introduced in
Table 6, were selected. The estimated parameters of the logistic regression model (model-1) for the log odds that a signal belongs to the pathology group is given by
where
P is the probability of belonging to the pathology group.
Table 6 indicates that an increase of one unit in the GLLE_0.1 (from baseline) indicates that the odds of being in the pathology group is 2.78 times the odds at the baseline, keeping the other factors constant. Similarly, increasing GLLE_4 by 1.0 unit leads to an increased odds of belonging to the pathology group. On the contrary, keeping all attributes constant, increasing GLLE_0.5, GLLE_1, or GLLE_5 by 1.0 unit leads to an increased odds of being in the healthy group.
Since we had unbalanced samples, we used the Precision-Recall Curve (PRC) to evaluate the performance of the classifier [
35].
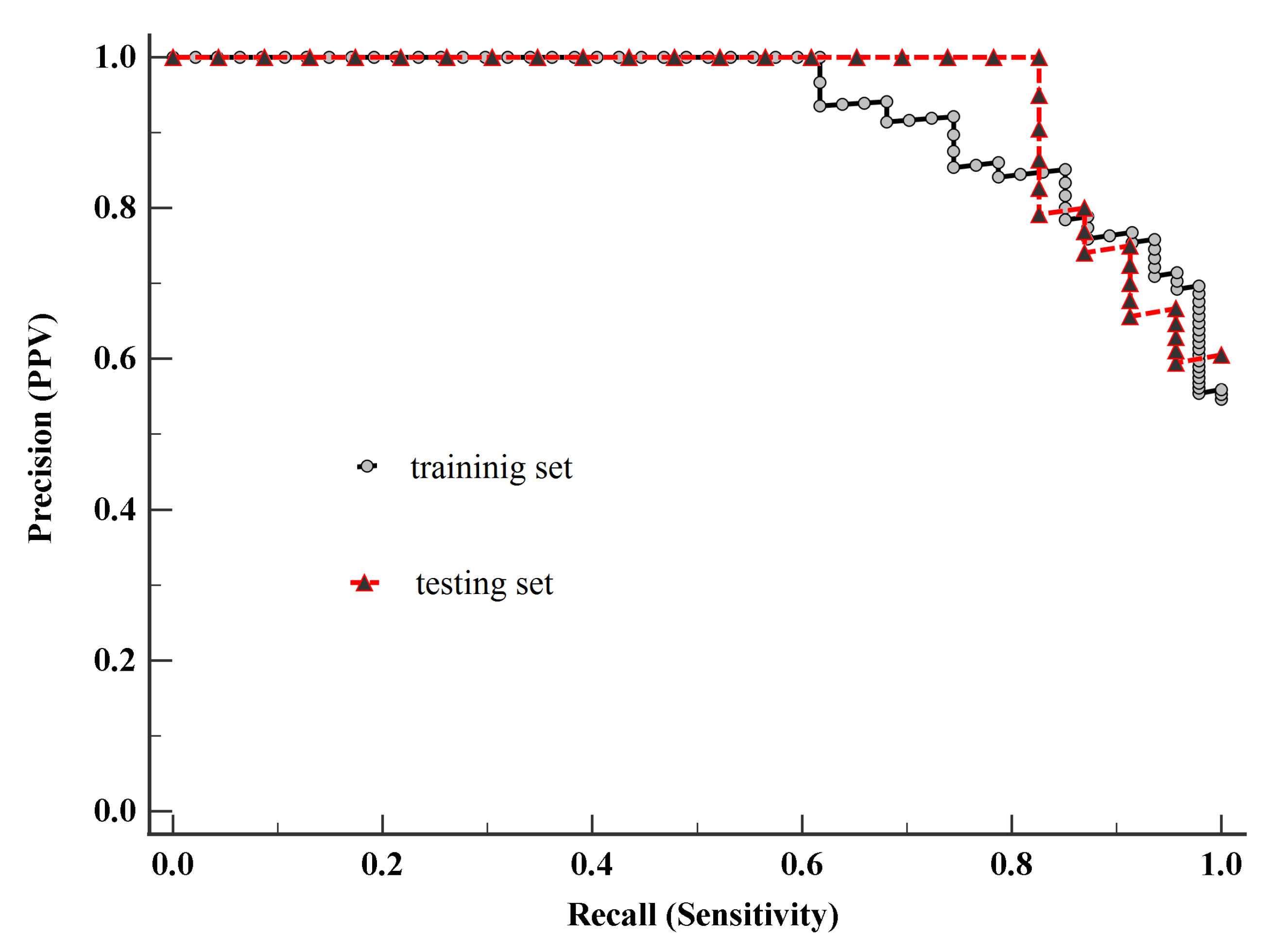
Figure 4 demonstrates the PRC curve of model-1 built on the training set. The area under the PRC curve (AUC) equals 0.93 (95% CI 0.88–0.97), suggesting a good performance. Analyzing the PRC curve allowed us to determine the associated criterion, defining the threshold of model-1, which is greater than 0.46.
Taking into account the values of the associated criterion and Equation (14), classification results of model-1 for the training and testing sets were determined. The classification results of model-1 are given in
Table 7.
To assess the performance of the classifier, the Matthews correlation coefficient (MCC) [
35] was calculated, based on the training set, to be MCC= 0.65 and based on the testing set, to be MCC = 0.57. These results indicate the good quality of the model in terms of both the training set and testing set. Furthermore, evaluating the quality of the model, its PRC curve was constructed based on the testing set. This PRC curve is shown in
Figure 4, where AUC equals 0.95 (95% CI 0.88–0.98), demonstrating a good performance. Since that there is no statistically significant difference (
p-value > 0.05) between the AUC of the PRC curve built on the testing set and the AUC of PRC curve built on the training set, it can be assumed that this model has the potential to be used for diagnostic purposes. Nevertheless, for use in diagnostics, this model requires additional research based on a larger sample.
Given the difference in multi-chaotic behavior in the RR intervals time series of the chf and af, the next step is to identify the values that differentiate between the congestive heart failure and the atrial fibrillation groups. Unfortunately, due to the small sample size, we did not split the sample into a training and testing set. Therefore, the results of this model are preliminary and require further additional research with a larger sample size.
By constructing a logistic regression model as a classifier based on the attributes (GLLE_p) GLLE_0.1, GLLE_0.5, GLLE_1, GLLE_2, GLLE_3, GLLE_4, and GLLE_5, we defined statistically significant values and assessed their impact. To avoid uncertainty in the form of infinity, we multiplied the attributes by a hundred. The output variable is denoted by Y = 0 for the congestive heart failure group and, accordingly, Y = 1 for the atrial fibrillation group. To choose the minimum set of factor attributes, the SVS method was applied [
12,
34]. Consequently, three factor values were identified, which are introduced in
Table 8.The estimated parameters of the logistic regression model (model-2) for the log odds that a signal belongs to the atrial fibrillation group are given by
Table 8 indicates that an increase of one unit in the GLLE_4 (from the baseline) indicates that the odds of being in the atrial fibrillation group is 7.62 × 10
times the odds at the baseline, keeping the other factors constant. On the contrary, when keeping all attributes constant, increasing GLLE_1 or GLLE_5 by 1.0 unit leads to the odds of being in the congestive heart failure group being increased.
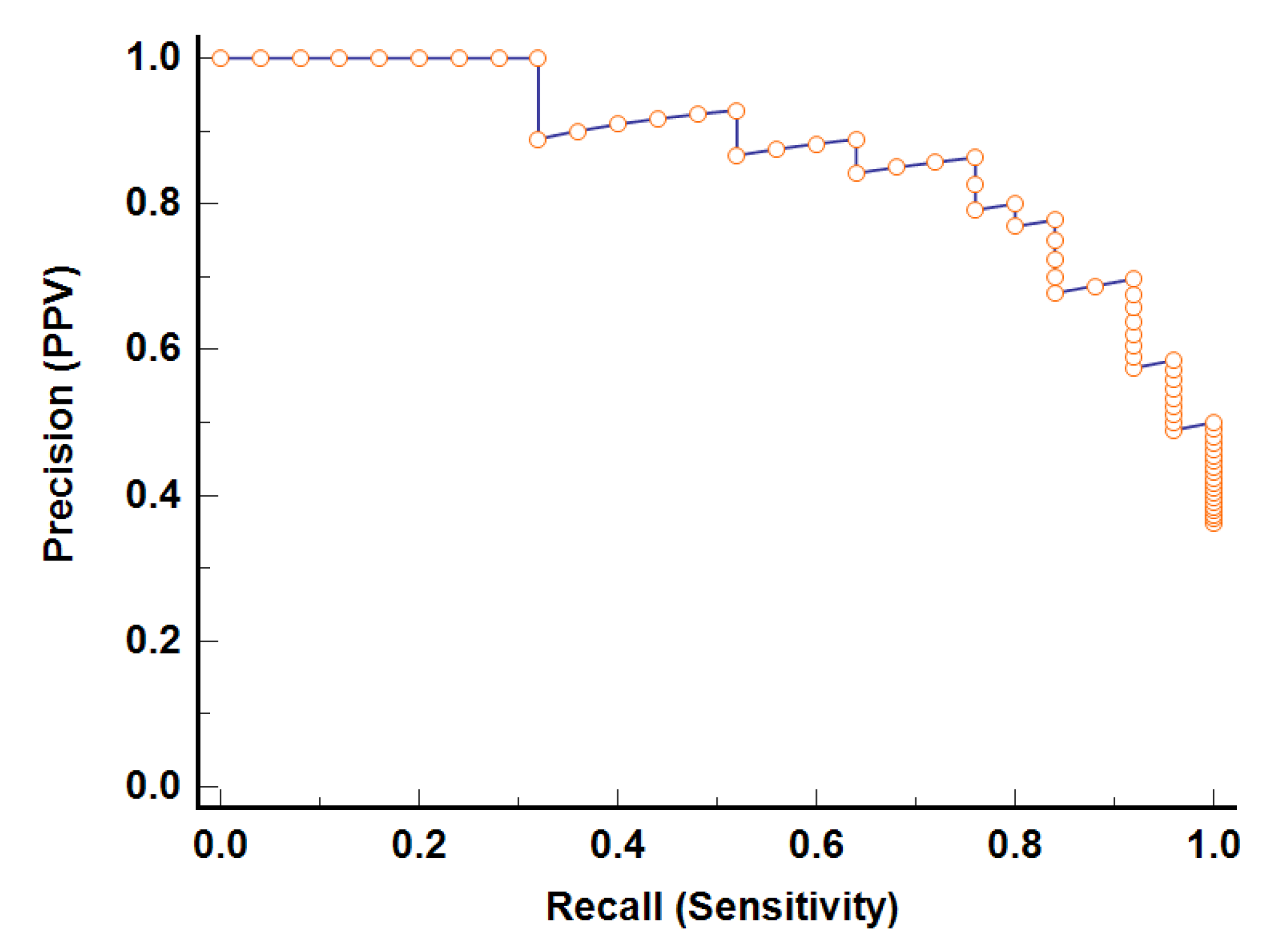
Figure 5 shows the PRC curve of model-2. The area under the PRC curve equals 0.87 (95% CI 0.74–0.95), demonstrating good performance.
The associated criterion, defining the threshold of model-2, is greater than 0.48. In view of the values of the associated criterion and Equation (15), the classification results of model-2 were defined. The classification results of model-2 are given in
Table 9.
The Matthews correlation coefficient equals 0.68. These results indicate the good performance of model-2. Therefore, the preliminary model demonstrated the good potential of the proposed approach to discriminate between the congestive heart failure and atrial fibrillation patients. However, this study should be continued with a larger sample.
4. Discussion
We developed a new method for analyzing and evaluating the behavior of nonlinear time series. This method is based on the assumption that some biological time series have a complex chaotic structure, which is formed of a combination of simpler chaotic structures. These types of time series cannot be adequately characterized by a single LLE value, but have to be characterized by a spectrum of LLE values. To determine this spectrum of LLE values, p-norms were used. The application of p-norms allows the contribution of the SF component or LF component to the estimate of the distance between the pair of nearest neighbors in the m-dimensional space to be strengthened. Obviously, a larger difference between SF and LF components indicates a more complex chaotic structure of the studied signal. The difference between SF and LF components defines the spectrum width W.
As a result of using this approach for analyzing the Logistic map, Henon map, and Lorenz attractor, we note that these attractors produce the estimation of the spectrum width of W > 0 and, accordingly, are characterized by multi-chaotic behavior. However, the Rossler attractor, having W ≈ 0, is characterized by mono-chaotic behavior. Therefore, the time series can be characterized by mono-chaotic or multi-chaotic behavior.
Since the biomedical signal is a product of an interaction of a large number of different biological systems, this leads to the presence of a random component in the biomedical signal. Therefore, we conducted studies on the influence of the random component on the multi-chaotic behavior of the investigated chaotic systems. As a result, we found that as the variance contribution of the random component increases, the spectrum width W decreases.
This approach has been applied to discover possible differences among R-R interval time series of healthy subjects (hs), subjects diagnosed with congestive heart failure (chf), and subjects diagnosed with atrial fibrillation (af).As a result of the study, it was found that for the Euclidean norm, the heart rate control system in chf subjects is more sensitive to the initial conditions than in hs subjects. Moreover, for the Euclidean norm, the heart rate control system in af subjects is less sensitive to the initial conditions than in hs subjects. We believe that these distinctions are related to the difference in the chaos control strategy [
7].
Studies have demonstrated that estimation of the spectrum width can be used as one of the parameters to evaluate the level of the random component of the chaotic biomedical signal. As a result of evaluating the spectrum width and the correlation dimension , it was found that the contribution of the random component in the atrial fibrillation group R-R interval time series is larger compared to in the hs R-R interval time series and chf R-R interval time series.
By applying multi-chaos theory, we built a five-factor logistic model that is able to distinguish the hs signal from the pathological groups, i.e., the chf and af groups. This model has been demonstrated to have a good diagnostic performance. However, in order to use this model for diagnostic purposes, it should be tested at the appropriate medical center. This approach will allow the sample size to be significantly increased and improve the quality of the model. Since multi-chaotic analysis revealed the difference in multi-chaotic behavior within the pathological group, this allowed us to build a preliminary three-factor logistic model that is able to distinguish the chf from the af signal. This model has demonstrated a good performance, which makes this approach promising for building an expert system to discriminate between congestive heart failure and atrial fibrillation signals. However, to study the diagnostic characteristics of this approach, additional research with a larger sample is required.
In our study, multi-chaotic analysis exhibited a good performance, allowing the underlying dynamics of the system that generates the heart beat to be examined. This approach has a good potential for being used in the construction of expert systems for the diagnosis of cardiac pathologies.