2. Materials and Methods
The electrical properties of interfacial water next to hydrophilic surfaces were measured with and without coronal discharge. Nafion 117 perfluorinated membranes (0.007 in. thick, Sigma-Aldrich, Saint Louis, MO, USA) were used as the hydrophilic surface. Before use, the Nafion membrane was immersed in deionized water for 10 min at room temperature.
Deionized water was obtained from a NANOpure Diamond ultrapure water system. The purity of water from this system was certified by a resistivity value up to 18.2 MΩ-cm, which exceeded American Society for Testing and Materials (ASTM), College of American Pathologist/ National Committee for Clinical Laboratory Standards (CAP/NCCLS) type I water requirements. In addition, the deionized water was passed through a 0.2-μm hollow fiber filter to ensure bacteria- and particle-free water before filling the glass chamber (dimensions: 7 cm × 4.5 cm × 2.5 cm) to a consistent height of 1 cm for all experiments.
As the probing electrode, we used standard glass microelectrodes pulled by a micropipette puller (P-87, Sutter Instrument, Novato, CA, USA) and filled with 3M KCl. Diameters of the microelectrode tips were less than 1 μm. To check for electrode quality, the probing and reference electrodes were immersed in 3M KCl prior to each experiment. In 3M KCl, the potential difference between the two electrodes was negligible. However, in deionized water, a DC offset almost always appeared due to deionized water’s poor conductivity. The DC offset could be anywhere between −20 mV to +20 mV. To measure the electrical potential distribution in the water in the vicinity of Nafion, we used a motor to advance the probe electrode. The motor was controlled by software written in Labview, and had a spatial resolution of 1.25 μm.
A high voltage generator (ES30P-5W, Gamma High Voltage Research) was used to create the coronal discharge, which produced positive air ions.
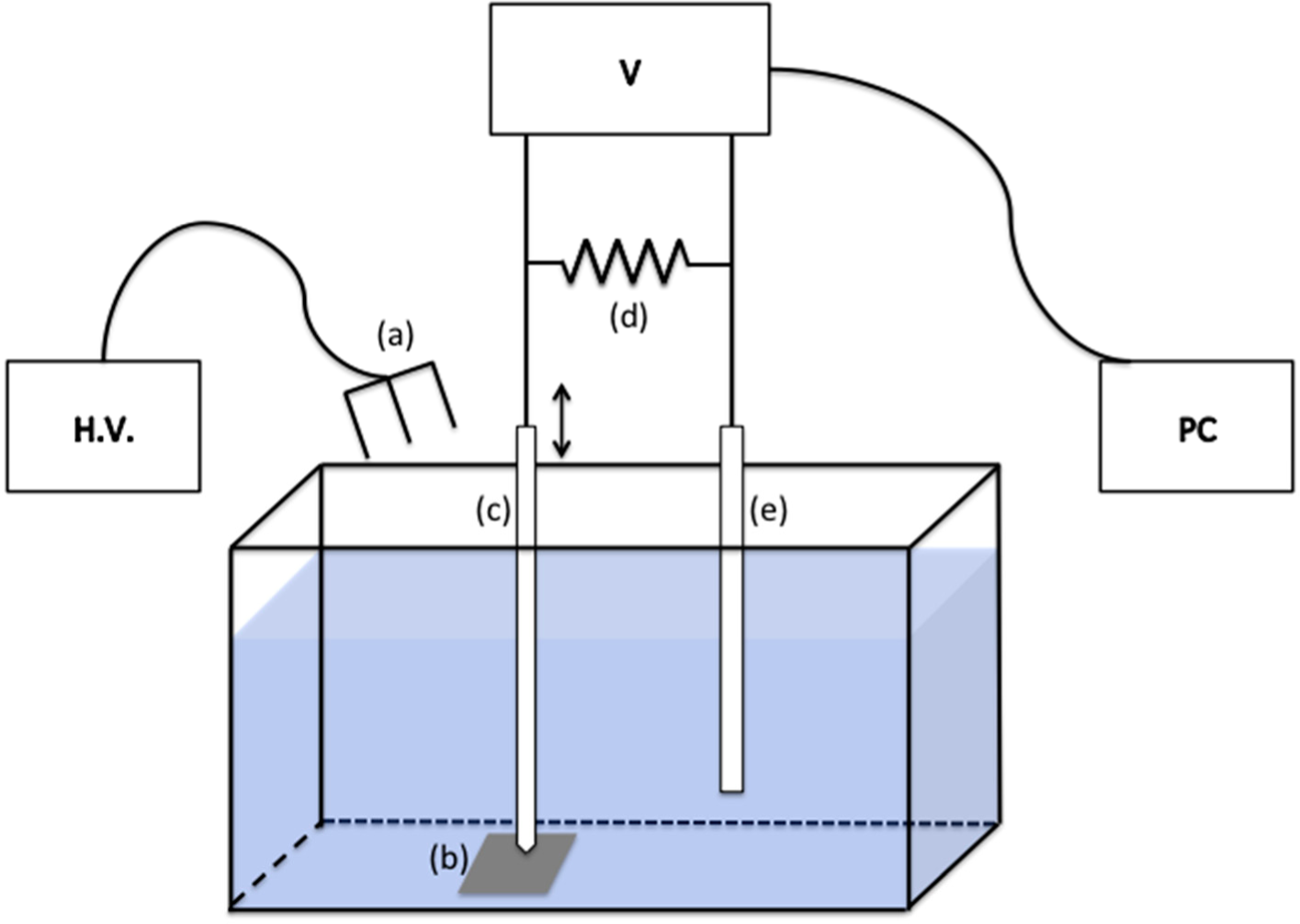
The experimental setup is shown in
Figure 1 Electrical potentials were measured with an electrometer (Model 6512, Keithley Instruments, Cleveland, OH, USA) and recorded on the computer through a data acquisition device (USB-6009, National Instruments, Austin, TX, USA). A flexible reference electrode (WPI) was placed remotely from the interfacial region. A 1000 MΩ passive resistor was placed in parallel with the electrometer terminals to keep the load resistance constant and to facilitate readout of the current extracted from the interfacial water. The resistor also helped to reduce ambient electromagnetic noise.
We first positioned the probing microelectrode immediately above and just touching the Nafion surface to obtain the most stable and highest magnitude electrical potential readout. The reference electrode was placed remotely. Before turning on the high voltage source, we measured the potential profile as a function of distance from the Nafion surface. The probing microelectrode was moved away from the Nafion surface by increments of 1.25 μm until it reached 1 mm from the Nafion surface. The electrical potential was recorded at each pause. After the potential profile measurement was completed, the system automatically brought the probing microelectrode back to the Nafion surface. Thus, the data collected provides reference information to be compared with the experimental results obtained in the presence of positive air ions.
High-voltage discharge was used to produce air ions. We turned up the voltage from 0 kV to 10 kV at 1 kV intervals. The discharge electrodes were located 3 cm above the water surface. Potentials as a function of increasing voltage were measured by the probing electrode and recorded simultaneously. Then we maintained the high voltage at 10 kV for 10 min and measured the potential profile once again. We set the 10 kV limit because at about 10.5 to 11 kV, the discharge electrodes began generating electric arcs which interfered with all nearby electronic equipment and made measurements inconsistent thereafter.
The probing microelectrode was brought back to the Nafion surface automatically after the second potential profile measurement was completed. Then we reversed the procedure by turning down the high voltage gradually back to zero and recording the potential between the two electrodes at 1 kV intervals. At the end, the potential profile was recorded again to check the reversibility of the air-ion effect.
Control experiments were conducted to clarify whether the effect on interfacial water was indeed from air ions or from electrical polarization due to the high voltage discharge electrodes or from the ozone generated during the coronal discharge. To test for electrical polarization, we used two identical reference electrodes in deionized water without the Nafion sheet. The two electrodes were placed side by side equally close to the high voltage discharge electrodes horizontally, at different heights in water. To test for the ozone, we used a copper metal mesh to cover the top of the chamber. Since the metal mesh sheet was grounded, no air ions could reach the water, while the electrically neutral ozone could still do so.
3. Results
Electrical potential measurements on the water-Nafion interface have been carried out many times with consistent results. When the probing electrode is very close to the Nafion surface, sometimes, even touching, and the reference electrode placed remotely, the magnitude of the potential difference between the two electrodes is the highest. It can range from −100 to −200 mV. Most often, its value is approximately −120 mV.
With the methods described in the Materials and Methods section, we measured the potential profile
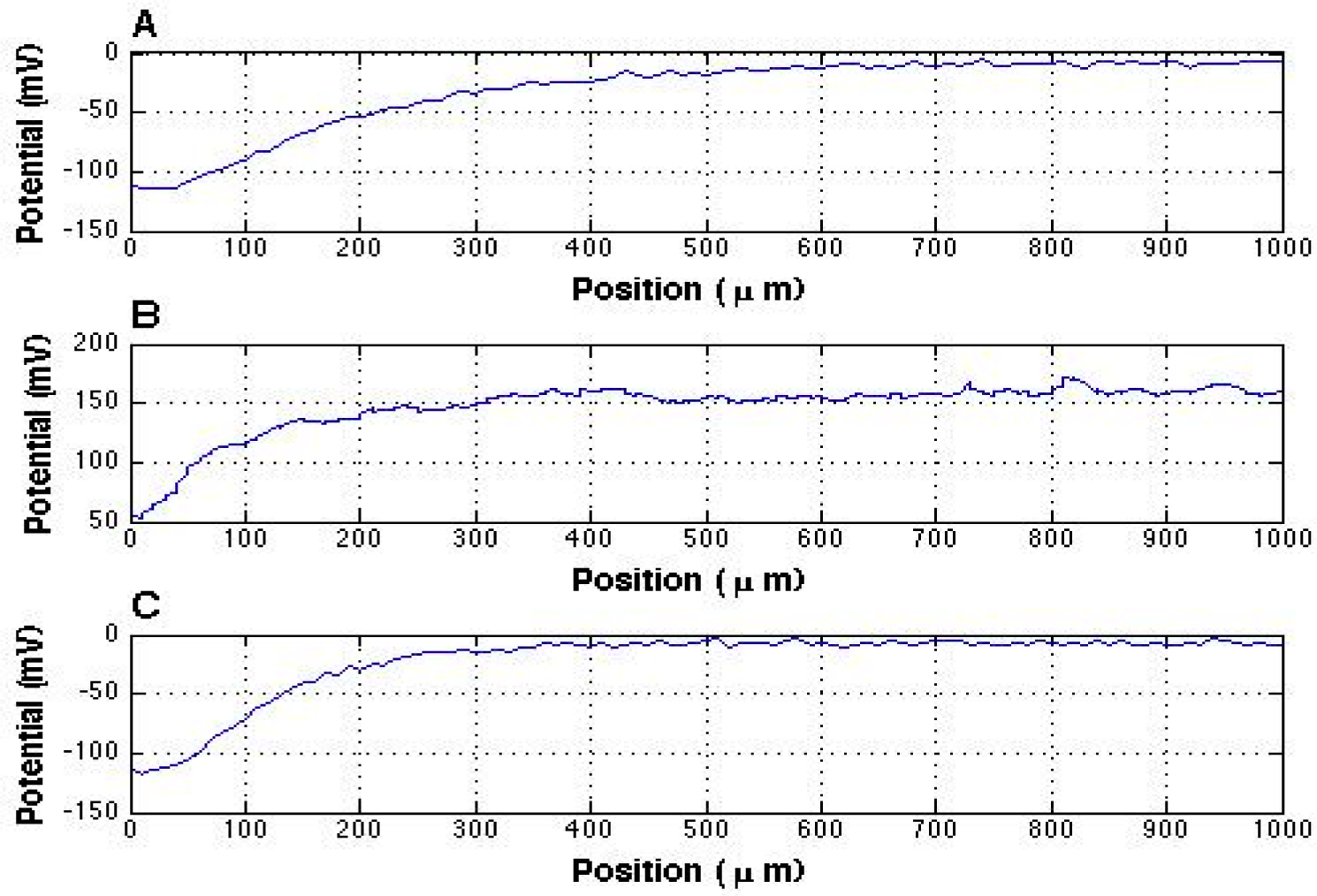
vs. distance from the Nafion surface as well as the highest potential before, during, and after applying the high voltage. Before turning on the high voltage supply, we obtained results similar to those found earlier. A representative potential profile is shown in
Figure 2A.
Next, the high voltage power supply was turned on and the potential between the two electrodes was recorded while the voltage was increased in 1 kV increments. This probe electrode remained at a fixed position in the water near the Nafion surface.
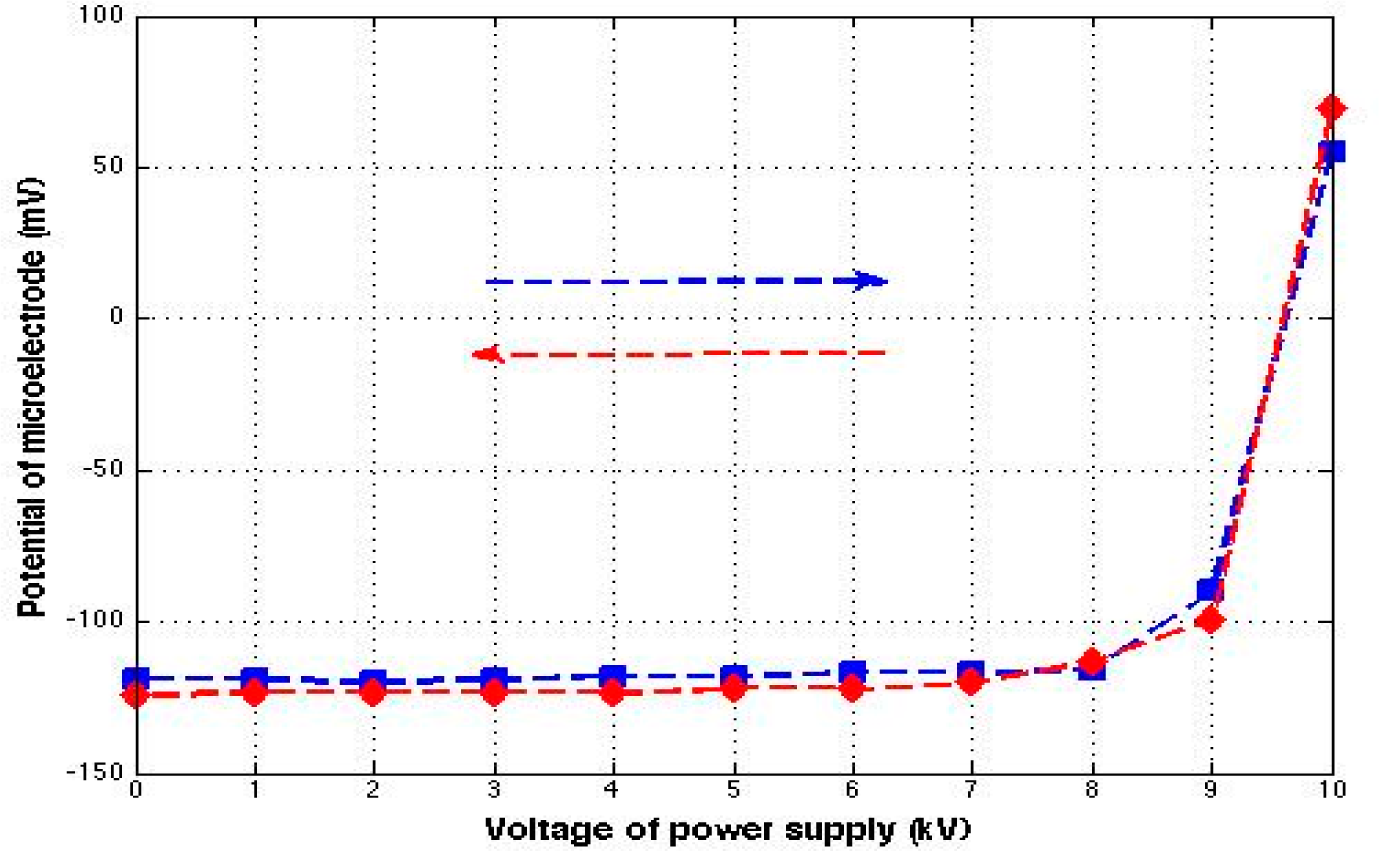
Figure 3 (blue line) shows that the electrical potential remained the same (less than 3% change) at all voltages below 8 kV. However, starting from around 9 kV, the electrical properties of interfacial water began changing noticeably. At the end, even the polarity changed from negative to positive.
Figure 2B depicts the potential profile that was obtained ten minutes after the voltage of the power supply reached 10 kV. As can be observed, the potential profile already shifted in the positive zone while the microelectrode was probing in the interfacial water region. This implies that the positive air ions can transfer through to the interfacial water, thus changing its’ electrical properties.
The altered electrical properties of the interfacial water were fully reversible.
Figure 3 (red line) shows the result obtained during successive reductions of the high voltage. The results fit in nicely with those obtained during the voltage upturn (blue line), except that the order was reversed. The polarity returned to negative between 10 and 9 kV, with no obvious change seen from 8 to 0 kV. Upon reaching zero, the final potential profile measurement showed a return to the values obtained before turning on the high voltage supply (
Figure 2C).
Shutting down the high voltage supply directly from 10 kV produced similar results: the potential between electrodes promptly jumped back approximately to the initial value within just a few seconds. This was an interesting find because positive ions dissolved in water are not expected to neutralize so fast. However, our previous work has shown that interfacial water forms rapidly, implying that the return to negative potential may be due to the rapid re-building of negatively charged interfacial water.
Next, we investigated if the altered electrical properties genuinely arose from the air ions, or from some other features such as polarization of water or ozone produced from coronal discharge.
According to standard electrostatic induction theory, any positive charge placed above a body of water (a dielectric material) should induce negative charge on the top part of the water, while the bottom part becomes more positive. However, the air ion effect did the opposite: it made the water more positive on top than the bottom (
Figure 2B). Apparently, positive charge from the high voltage source must have entered the water.
To test this idea, we removed the Nafion and placed two identical reference electrodes in the deionized water. One electrode was kept at mid-height of the chamber, while the other was moved vertically, to the top, middle and bottom. When the high voltage source was turned on, the top of the water was clearly more positive than the bottom.
In order to rule out the influence of ozone produced from coronal discharge as the possible cause of the electrical changes, a grounded copper metal mesh was positioned on top of the chamber to block any charged air ions from entering into the water, while allowing the uncharged ozone to pass through. We then elicited coronal discharge by turning up the voltage to 10 kV, the highest value possible before creating an electric arc. The potential difference between the two electrodes changed by less than 3 %. This observation confirmed that without entry of the charged ions, the electrical properties of interfacial water would not change, implying that the observed potential changes were indeed caused by the positive ions and not by artifacts such as ozone or other byproducts of coronal discharge.
A question is whether naturally occurring changes of atmospheric charge have similar effects on interfacial water. During our earlier experiments on the electrical properties of interfacial water, we noticed small variations in the electrical potential’s periodical amplitude. After investigating, we found that those variations were related to the airflow from the laboratory air conditioner: we used an acoustic microphone placed at the air conditioner outlet, and confirmed that the higher the airflow, the louder the noise.
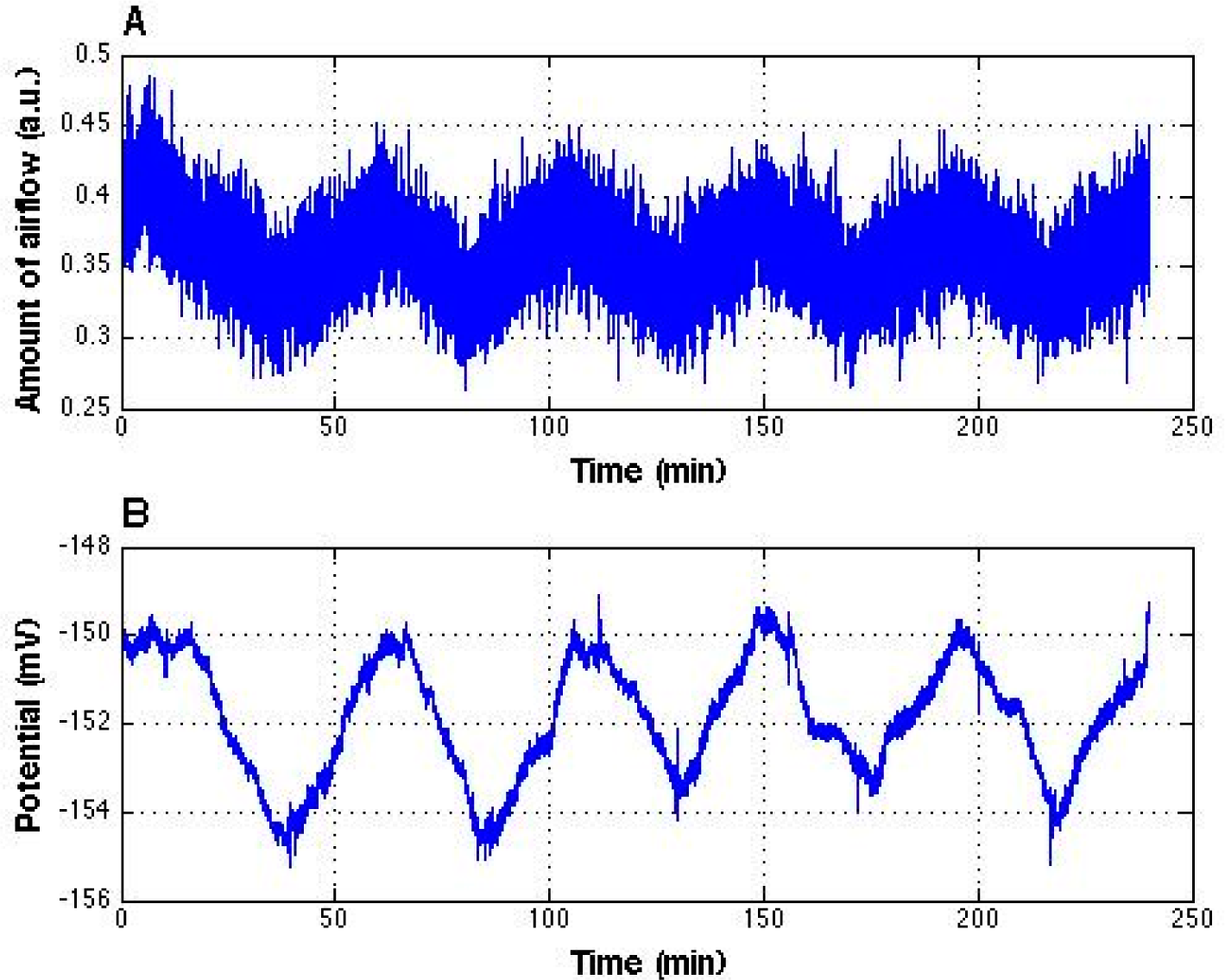
We therefore recorded the airflow over an extended period (4 h) while simultaneously measuring the electrical potential of interfacial water. We confirmed an excellent alignment of the periodic change in potential (about 4 mV in amplitude) with the airflow from the air conditioner (
Figure 4). The period was approximately 50 min. Throughout the experiment, the measured change of water temperature was less than one degree Celsius.
These observations may be explained by the fact that air conditioners are sources of charged air. When air is forced to flow through pipes and filters, it becomes positively charged due to friction. In fact, air is the most positive substance of the triboelectric series [
14]. The air’s positive charge can help explain how the airflow from the air conditioner can impact the electrical features of interfacial water.
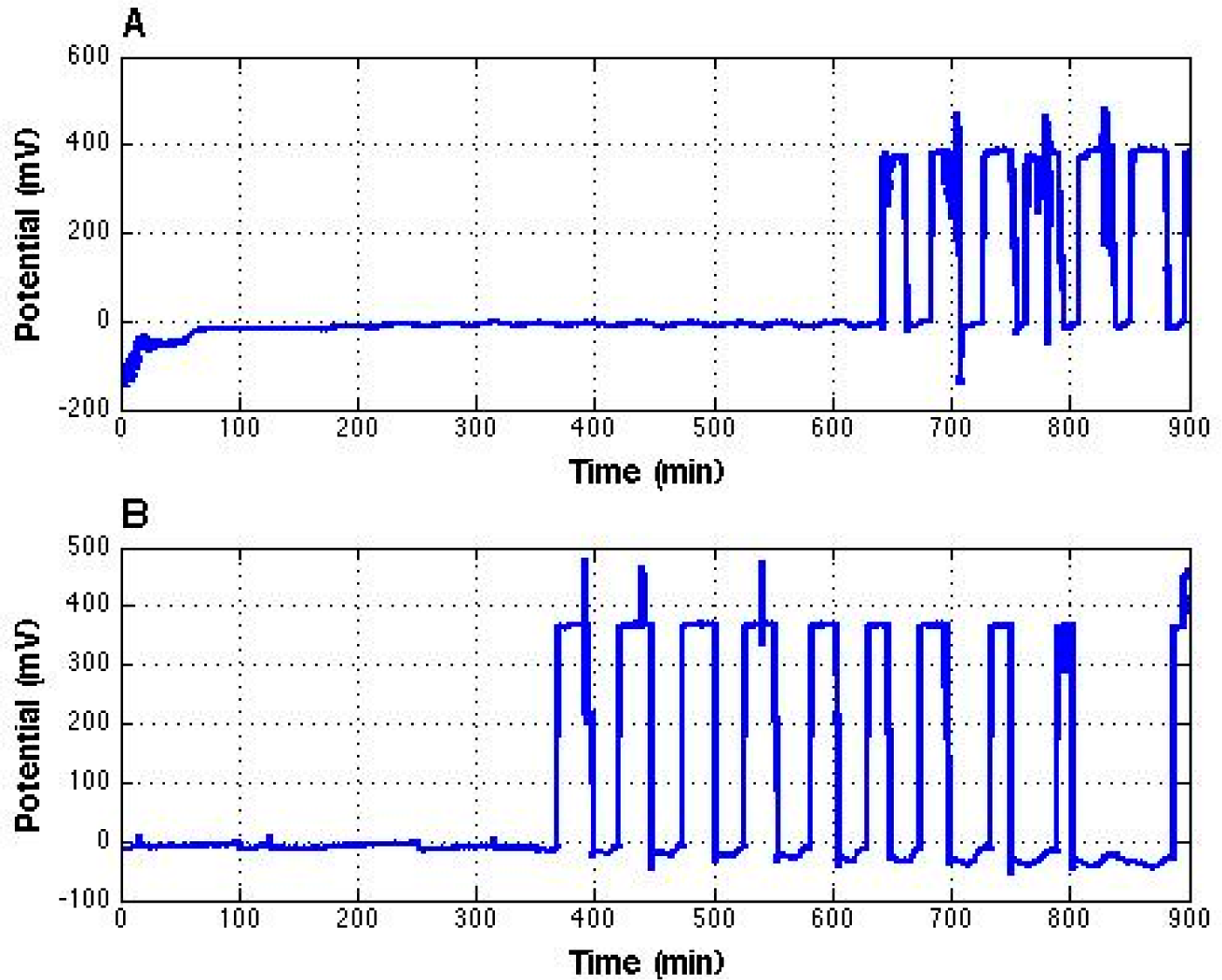
While the effects of the laboratory air conditioner were generally modest, one occasion produced a rather astonishing observation.
Figure 5 shows an overnight potential recording with the same set-up as
Figure 1 but without the high voltage power supply. The amplitude change was over 400 mV, and the period was about 50 min, the same as the air conditioner (compare
Figure 4). Because the period is similar, we believe the “skyscraper” oscillations arose from the positively charged air ions coming from the air conditioner.
While there were small periodic changes (similar to those of
Figure 4) during the first ten hours of recording, suddenly at about 10.5 h, the potential dramatically changed to a high positive value close to 400 mV (
Figure 5A). This kind of threshold effect fits the experimental results described earlier in this paper. We later recorded the potential of interfacial water for another 15 h without changing the experimental setup and observed similar results (
Figure 5B). The one missing skyscraper oscillation near the end of the recording implied that the air conditioner might not have supplied sufficient positive air ions for threshold crossing, the same as during early periods in the two recordings.
These observations confirmed our findings that airflow from ordinary air conditioners can appreciably change the electrical properties of interfacial water.
4. Discussions
Generally speaking, negative charge is believed to promote good health, whereas positive charge compromises health [
7–
9,
15–
17]. Nature provides a reasonably balanced supply of air ions; however more negative ions are found at places such as waterfalls and forests [
18], which often elicit sensations of well-being. The question arises whether these effects are largely psychogenic or whether some physical mechanism exists to explain the effects.
One common denominator of function may be cell water. By volume, our bodies are roughly 2/3 of water, and much of that water is interfacial;
i.e., it resides next to some hydrophilic interface [
12]. More recent experimental observations of interfacial water have shown that interfacial water is negatively charged [
10,
11]. This negative charge could be reduced by atmospheric positivity and thereby cause adverse health effects. In other words, if this water, along with the proteins it envelops, is vital for function, then compromising the amount of interfacial water could impair function.
The experimental results were consistent with this hypothesis. In the experiments involving high voltage, a threshold voltage was required to produce enough air ions to have appreciable impact on the interfacial water. When the discharge electrodes were placed 3 cm from the water surface, the threshold voltage required to produce enough air ions was approximately 8 kV. From this result we infer that positive ions in the air may change concentration appreciably without having adverse health effects. However, beyond a threshold concentration, the ill effects of positive charge may become appreciable— even wiping out the negativity of interfacial water and replacing it with the many positive ions in the water.
The rapid rebuilding of interfacial water once the positive ions were removed is consistent with earlier results. Interfacial water builds rapidly. Once the inhibiting positive ions are removed, the structure can easily and quickly rebuild. In terms of health impact, this suggests that restoration of function can occur rapidly once the positive air ions are removed. This phenomenon can potentially explain the “refreshing” sensation that people often feel when stepping out of a crowded room with positive ions from exhalation, and into fresh air.
We obtained similar results with positive ions produced from an air conditioner. When the conditions are right, enough air ions could be produced to change the electrical properties of the interfacial water. The example of dramatically changed electrical potential indicated that the structure of interfacial water could be completed destroyed by positive air ions, perhaps explaining the serious consequences people can sometimes suffer in sufficiently foul air. More generally, this result may explain the natural craving to escape from air-conditioned environments and into natural air.