If It Works, Don’t Touch It? A Cell-Based Approach to Studying 2-[18F]FDG Metabolism
Abstract
:1. Introduction
2. Results
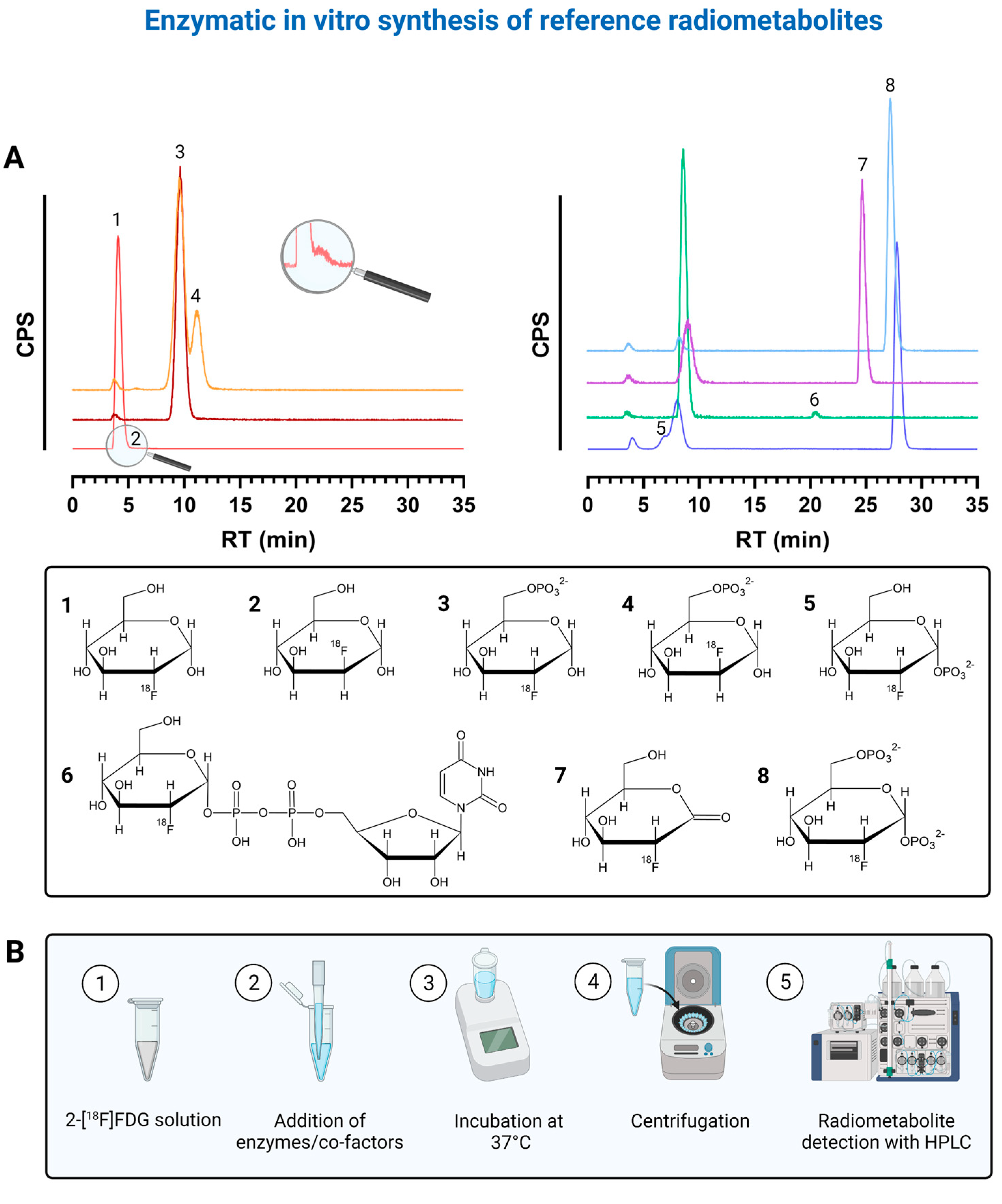
2.1. Enzymatic In Vitro Preparation and Verification of Radiometabolites
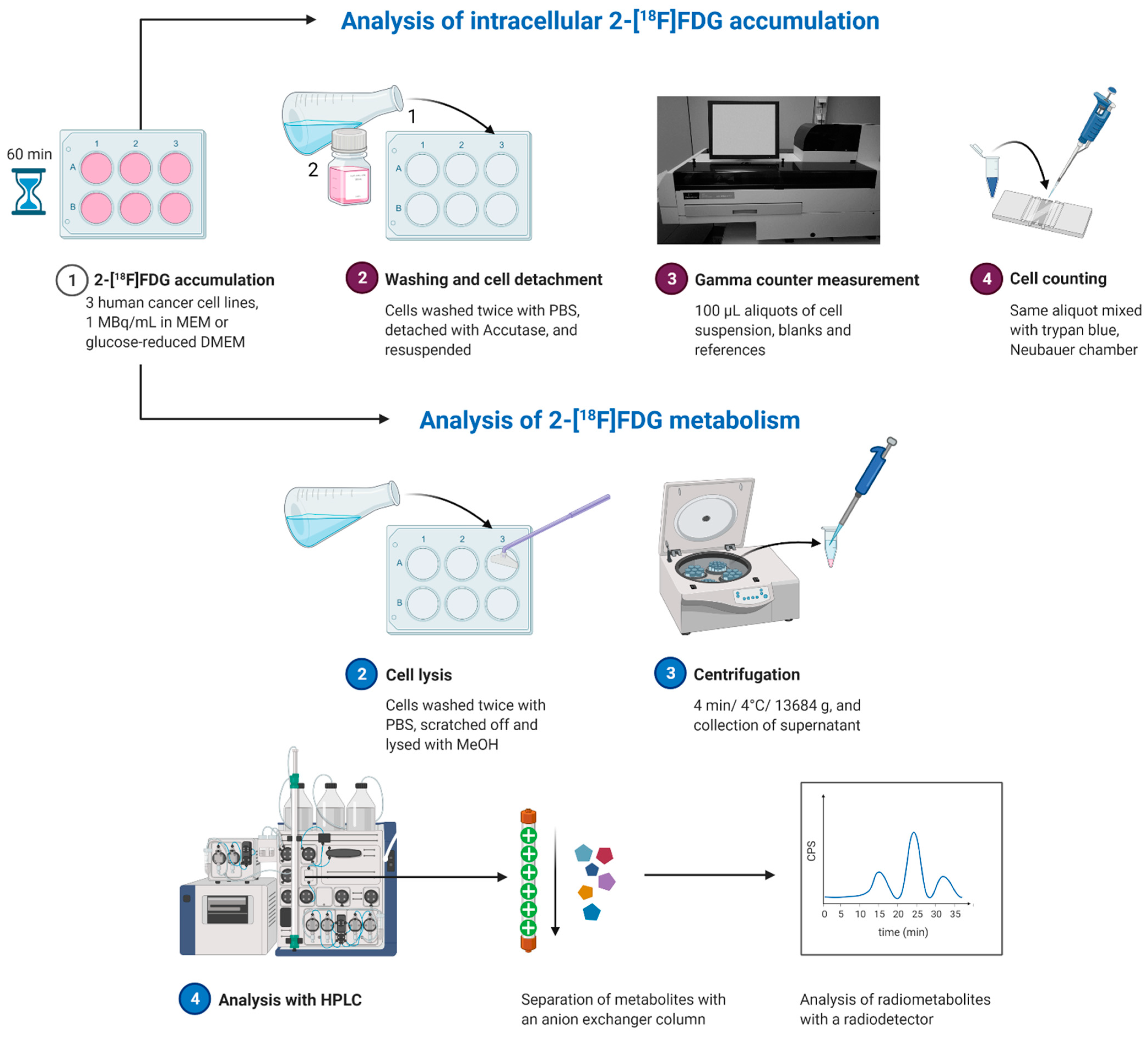
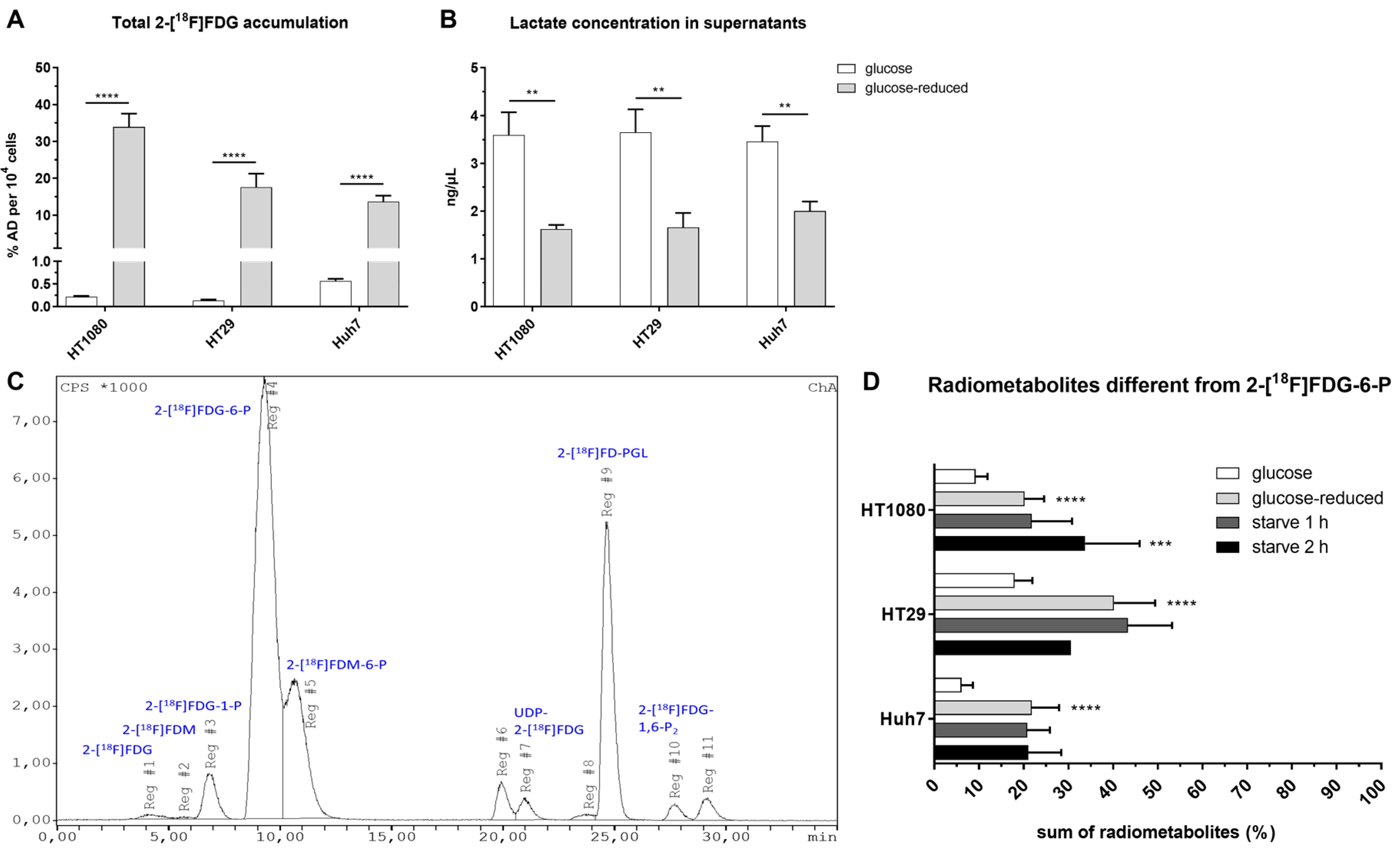
2.2. Intracellular Accumulation and Metabolism of 2-[18F]FDG in HT1080 (Fibrosarcoma), HT29 (Colorectal Adenocarcinoma) and Huh7 (Hepatocellular Carcinoma)
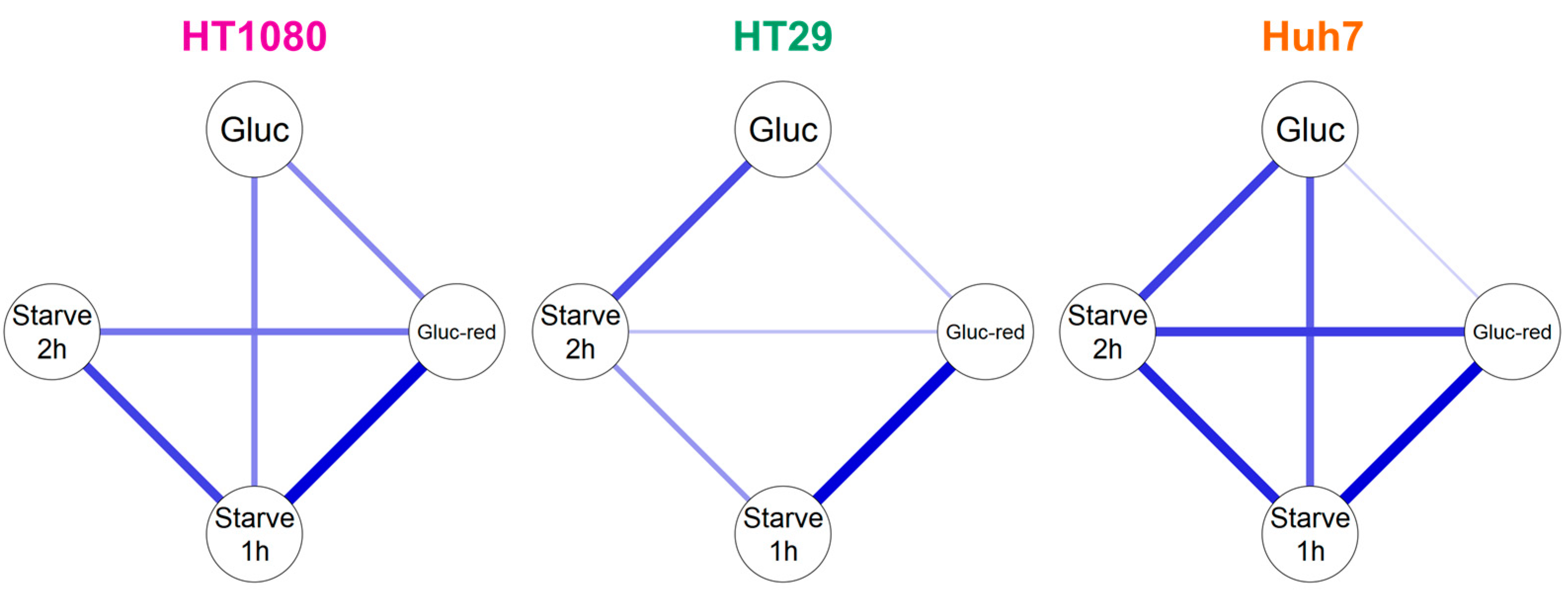
2.3. Efflux of Radioactivity in HT1080 (Fibrosarcoma) and HT29 (Colorectal Adenocarcinoma)
3. Discussion
4. Materials and Methods
4.1. General
4.2. Cell Culture
4.3. Enzymatic In Vitro Synthesis of 2-[18F]FDG Radiometabolites
4.4. 2-[18F]FDG-6-P
4.5. 2-[18F]FD-PGL
4.6. 2-[18F]FDG-1,6-P2
4.7. 2-[18F]FDG-1-P
4.8. UDP-2-[18F]FDG
4.9. 2-[18F]FDM-6-P
4.10. 2-[18F]FDG Accumulation Experiments
4.11. Radiometabolite Detection with HPLC
4.12. Efflux Experiments
4.13. Statistics
4.13.1. General
4.13.2. Gaussian Model
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wadsak, W.; Mitterhauser, M. Basics and principles of radiopharmaceuticals for PET/CT. Eur. J. Radiol. 2010, 73, 461–469. [Google Scholar] [CrossRef]
- Pichler, V.; Berroterán-Infante, N.; Philippe, C.; Vraka, C.; Klebermass, E.-M.; Balber, T.; Pfaff, S.; Nics, L.; Mitterhauser, M.; Wadsak, W. An Overview of PET Radiochemistry, Part 1: The Covalent Labels18F,11C, and13N. J. Nucl. Med. 2018, 59, 1350–1354. [Google Scholar] [CrossRef] [Green Version]
- Potter, M.; Newport, E.; Morten, K.J. The Warburg effect: 80 years on. Biochem. Soc. Trans. 2016, 44, 1499–1505. [Google Scholar] [CrossRef] [Green Version]
- Flavell, R.R.; Naeger, D.M.; Aparici, C.M.; Hawkins, R.A.; Pampaloni, M.H.; Behr, S.C. Malignancies with Low Fluorodeoxyglucose Uptake at PET/CT: Pitfalls and Prognostic Importance:Resident and Fellow Education Feature. Radiographics 2016, 36, 293–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.-F.; Du, Y.; Ma, Y.; Postel, G.C.; Civelek, A.C. 18F-Fluorodeoxyglucose Uptake and Tumor Hypoxia: Revisit 18F-Fluorodeoxyglucose in Oncology Application. Transl. Oncol. 2014, 7, 240–247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hensley, C.T.; Faubert, B.; Yuan, Q.; Lev-Cohain, N.; Jin, E.; Kim, J.; Jiang, L.; Ko, B.; Skelton, R.; Loudat, L.; et al. Metabolic Heterogeneity in Human Lung Tumors. Cell 2016, 164, 681–694. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kernstine, K.H.; Faubert, B.; Do, Q.; Rogers, T.J.; Hensley, C.T.; Cai, L.; Torrealba, J.; Oliver, D.; Wachsmann, J.W.; Lenkinski, R.E.; et al. Does Tumor FDG-PET Avidity Represent Enhanced Glycolytic Metabolism in Non-Small Cell Lung Cancer? Ann. Thorac. Surg. 2020, 109, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Wienhard, K. Measurement of glucose consumption using [18F] fluorodeoxyglucose. Methods 2002, 27, 218–225. [Google Scholar] [CrossRef]
- Izuishi, K.; Yamamoto, Y.; Mori, H.; Kameyama, R.; Fujihara, S.; Masaki, T.; Suzuki, Y. Molecular mechanisms of [18F]fluorodeoxyglucose accumulation in liver cancer. Oncol. Rep. 2013, 31, 701–706. [Google Scholar] [CrossRef]
- Wiebe, L.I. FDG metabolism: Quaecumque sunt vera. J. Nucl. Med. 2001, 42, 1679–1681. [Google Scholar]
- Southworth, R.; Parry, C.R.; Parkes, H.G.; Medina, R.A.; Garlick, P.B. Tissue-specific differences in 2-fluoro-2-deoxyglucose metabolism beyond FDG-6-P: a19F NMR spectroscopy study in the rat. NMR Biomed. 2003, 16, 494–502. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, Y.; Yamane, H.; Shinohara, S.; Kuribayashi, S.; Momozono, Y.; Yamato, Y.; Kojima, M.; Masuda, K. 2-Deoxy-2-Fluoro-d-Glucose as a Functional Probe for NMR: The Unique Metabolism Beyond Its 6-Phosphate. J. Neurochem. 2002, 66, 2113–2120. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, Y.; Umayahara, K.; Shimmura, T.; Yamashita, T. 19F NMR of 2-Deoxy-2-fluoro-D-glucose for Tumor Diagnosis in Mice. An NDP-Bound Hexose Analog as a New NMR Target for Imaging. NMR Biomed. 1997, 10, 35–41. [Google Scholar] [CrossRef]
- Suolinna, E.-M.; Haaparanta, M.; Paul, R.; Härkönen, P.; Solin, O.; Sipilä, H. Metabolism of 2-[18F]fluoro-2-deoxyglucose in tumor-bearing rats: Chromatographic and enzymatic studies. Int. J. Radiat. Appl. Instrument. Part B Nucl. Med. Biol. 1986, 13, 577–581. [Google Scholar] [CrossRef]
- Kaarstad, K.; Bender, D.; Bentzen, L.; Munk, O.L.; Keiding, S. Metabolic fate of 18F-FDG in mice bearing either SCCVII squamous cell carcinoma or C3H mammary carcinoma. J. Nucl. Med. 2002, 43, 940–947. [Google Scholar] [PubMed]
- Marini, C.; Ravera, S.; Buschiazzo, A.; Bianchi, G.; Orengo, A.M.; Bruno, S.; Bottoni, G.; Emionite, L.; Pastorino, F.; Monteverde, E.; et al. Discovery of a novel glucose metabolism in cancer: The role of endoplasmic reticulum beyond glycolysis and pentose phosphate shunt. Sci. Rep. 2016, 6, 25092. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sambuceti, G.; Cossu, V.; Bauckneht, M.; Morbelli, S.; Orengo, A.M.; Carta, S.; Ravera, S.; Bruno, S.; Marini, C. 18F-fluoro-2-deoxy-d-glucose (FDG) uptake. What are we looking at? Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 1278–1286. [Google Scholar] [CrossRef]
- Rokka, J.; Grönroos, T.J.; Viljanen, T.; Solin, O.; Haaparanta-Solin, M. HPLC and TLC methods for analysis of [ 18 F]FDG and its metabolites from biological samples. J. Chromatogr. B 2017, 1048, 140–149. [Google Scholar] [CrossRef]
- Meyer, G.-J.; Matzke, K.H.; Hamacher, K.; Füchtner, F.; Steinbach, J.; Notohamiprodjo, G.; Zijlstra, S. The stability of 2-[18F]fluoro-deoxy-d-glucose towards epimerisation under alkaline conditions. Appl. Radiat. Isot. 1999, 51, 37–41. [Google Scholar] [CrossRef]
- Horecker, B.L.; Smyrniotis, P.Z. Reversibility of glucose-6-phosphate oxidation. Biochim. Biophys. Acta Bioenerg. 1953, 12, 98–102. [Google Scholar] [CrossRef]
- McSheehy, P.M.; Leach, M.O.; Judson, I.R.; Griffiths, J.R. Metabolites of 2′-fluoro-2′-deoxy-d-glucose detected by 19F magnetic resonance spectroscopy in vivo predict response of murine RIF-1 tumors to 5-fluorouracil. Cancer Res. 2000, 60, 2122–2127. [Google Scholar]
- Shinohara, S.; Kanazawa, Y.; Kojima, M. Evaluation of energy metabolism in brain using epimerization of 2-deoxy-2-fluoro-d-glucose by 19F NMR: The effect of anesthesia. Magn. Reson. Med. 1991, 21, 191–196. [Google Scholar] [CrossRef]
- Keppler, D.; Keppler, A. Methods of Enzymatic Analysis, 3rd ed.; Verlag Chemie: Weinheim, Germany, 1984; p. 203. [Google Scholar]
- Chou, J.Y.; Jun, H.S.; Mansfield, B.C. The SLC37 family of phosphate-linked sugar phosphate antiporters. Mol. Asp. Med. 2013, 34, 601–611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cossu, V.; Marini, C.; Piccioli, P.; Rocchi, A.; Bruno, S.; Orengo, A.M.; Emionite, L.; Bauckneht, M.; Grillo, F.; Capitanio, S.; et al. Obligatory role of endoplasmic reticulum in brain FDG uptake. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1184–1196. [Google Scholar] [CrossRef]
- Markowetz, F.; Spang, R. Inferring cellular networks—A review. BMC Bioinform. 2007, 8, S5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kulterer, O.C.; Niederstaetter, L.; Herz, C.T.; Haug, A.R.; Bileck, A.; Pils, D.; Kautzky-Willer, A.; Gerner, C.; Kiefer, F.W. The Presence of Active Brown Adipose Tissue Determines Cold-Induced Energy Expenditure and Oxylipin Profiles in Humans. J. Clin. Endocrinol. Metab. 2020, 105, 2203–2216. [Google Scholar] [CrossRef] [PubMed]
- Zois, C.E.; Harris, A.L. Glycogen metabolism has a key role in the cancer microenvironment and provides new targets for cancer therapy. J. Mol. Med. 2016, 94, 137–154. [Google Scholar] [CrossRef] [Green Version]
- Spitz, D.R.; Sim, J.E.; Ridnour, L.A.; Galoforo, S.S.; Lee, Y.J. Glucose deprivation-induced oxidative stress in human tumor cells. A fundamental defect in metabolism? Ann. N. Y. Acad. Sci. 2006, 899, 349–362. [Google Scholar] [CrossRef] [PubMed]
- Cherkas, A.; Holota, S.; Mdzinarashvili, T.; Gabbianelli, R.; Zarkovic, N. Glucose as a Major Antioxidant: When, what for and Why It Fails? Antioxidants 2020, 9, 140. [Google Scholar] [CrossRef] [Green Version]
- Sprinz, C.; Altmayer, S.; Zanon, M.; Watte, G.; Irion, K.; Marchiori, E.; Hochhegger, B. Effects of blood glucose level on 18F-FDG uptake for PET/CT in normal organs: A systematic review. PLoS ONE 2018, 13, e0193140. [Google Scholar] [CrossRef]
- Travaini, L.; Trifiro, G.; Paganelli, G. [18F] FDG uptake: Pay attention to candies. Ecancermedicalscience 2007, 1, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rabkin, Z.; Israel, O.; Keidar, Z. Do Hyperglycemia and Diabetes Affect the Incidence of False-Negative 18F-FDG PET/CT Studies in Patients Evaluated for Infection or Inflammation and Cancer? A Comparative Analysis. J. Nucl. Med. 2010, 51, 1015–1020. [Google Scholar] [CrossRef] [Green Version]
- Jahromi, A.H.; Fallahzadeh, M.K.; Takalkar, A.; Sheng, J.; Zibari, G.; Amiri, H.S. Impact of Plasma Glucose Level at the Time of Fluorodeoxyglucose Administration on the Accuracy of FDG-PET/CT in the Diagnosis of Pancreatic Lesions. Int. J. Endocrinol. Metab. 2014, 12, e16429. [Google Scholar] [CrossRef] [Green Version]
- Boellaard, R.; Delgado-Bolton, R.; Oyen, W.J.G.; Giammarile, F.; Tatsch, K.; Eschner, W.; Verzijlbergen, F.J.; Barrington, S.F.; Pike, L.C.; Weber, W.A.; et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: Version 2.0. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 328–354. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, M.R.; Heiden, M.G.V. Determinants of nutrient limitation in cancer. Crit. Rev. Biochem. Mol. Biol. 2019, 54, 193–207. [Google Scholar] [CrossRef] [PubMed]
- Lindl, T.; Gstraunthaler, G. Zell- und Gewebekultur, 6th ed.; Spektrum Akademischer Verlag: Heidelberg, Germany, 2008; p. 95. [Google Scholar]
- González, O.; Alonso, R.M. Chapter 6—Validation of bioanalytical chromatographic methods for the quantification of drugs in biological fluids. In Handbook of Analytical Separations; Hempel, G., Ed.; Elsevier Science B.V.: Amsterdam, The Netherlands, 2020; Volume 7, pp. 115–134. [Google Scholar]
- Perisic, O. Heterodimer Binding Scaffolds Recognition via the Analysis of Kinetically Hot Residues. Pharmaceuticals 2018, 11, 29. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| HT1080 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Radiometabolites | RT (min) | Glucose | Glucose-Reduced | Starve (1 h) | Starve (2 h) | ||||
| mean (%) | SD | mean (%) | SD | mean (%) | SD | mean (%) | SD | ||
| 2-[18F]FDG | ~4 | 11 | 3 | 0.3 | 0.1 | 0.40 | 0.09 | 0.62 | 0.28 |
| 2-[18F]FDG-1-P | ~7 | 0 | 0 | 1.6 | 0.7 | 3.18 | 2.61 | 5.7 | 3.5 |
| 2-[18F]FDG-6-P | ~9.5 | 79 | 2 | 79 | 4 | 78 | 9 | 66 | 12 |
| 2-[18F]FDM-6-P | ~12 | 0 | 0 | 9 | 5 | 6 | 5 | 12 | 1 |
| radiometabolite 5 | ~19.5 | 0 | 0 | 0.53 | 0.44 | 0.56 | 0.25 | 2 | 1 |
| UDP-2-[18F]FDG | ~21 | 0 | 0 | 0.51 | 0.23 | 0.69 | 0.40 | 1.7 | 0.8 |
| 2-[18F]FD-PGL | ~25 | 0.0 | 0.0 | 4 | 2 | 7 | 4 | 12 | 7 |
| 2-[18F]FDG-1,6-P2 | ~28 | 8 | 2 | 3 | 2 | 3 | 2 | 0.47 | 0.11 |
| radiometabolite 12 | ~29.5 | 1.1 | 0.6 | 0.95 | 0.60 | 0.96 | 0.82 | 0.22 | 0.02 |
| HT29 | |||||||||
| Radiometabolites | RT (min) | Glucose | Glucose-Reduced | Starve (1 h) | Starve (2 h) | ||||
| mean (%) | SD | mean (%) | SD | mean (%) | SD | mean (%) | SD | ||
| 2-[18F]FDG | ~4 | 3 | 1 | 0.4 | 0.2 | 0.57 | 0.14 | 0.4 | 0.1 |
| 2-[18F]FDG-1-P | ~7 | 1.2 | 0.5 | 3 | 1 | 3 | 2 | 1.2 | 0.2 |
| 2-[18F]FDG-6-P | ~9.5 | 79 | 4 | 59 | 9 | 56 | 10 | 69.0 | 0.7 |
| 2-[18F]FDM-6-P | ~12 | 7 | 2 | 14 | 3 | 17 | 3 | 19 | 1 |
| radiometabolite 5 | ~19.5 | 0.1 | 0.1 | 1.5 | 0.9 | 2.9 | 1.0 | 2.2 | 0.4 |
| UDP-2-[18F]FDG | ~21 | 0.1 | 0.2 | 1.0 | 0.6 | 2.1 | 0.5 | 2.0 | 0.3 |
| 2-[18F]FD-PGL | ~25 | 3 | 1 | 15 | 7 | 15 | 8 | 5 | 2 |
| 2-[18F]FDG-1,6-P2 | ~28 | 4 | 3 | 3 | 3 | 1 | 1 | 0.3 | 0.1 |
| radiometabolite 12 | ~29.5 | 3 | 1 | 3 | 1 | 2 | 1 | 0.7 | 0.2 |
| Huh7 | |||||||||
| Radiometabolites | RT (min) | Glucose | Glucose-reduced | Starve (1 h) | Starve (2 h) | ||||
| mean (%) | SD | mean (%) | SD | mean (%) | SD | mean (%) | SD | ||
| 2-[18F]FDG | ~4 | 8 | 2 | 2 | 1 | 3 | 2 | 2 | 1 |
| 2-[18F]FDG-1-P | ~7 | 0.7 | 0.4 | 4 | 2 | 3 | 2 | 4 | 2 |
| 2-[18F]FDG-6-P | ~9.5 | 86 | 3 | 76 | 6 | 76 | 5 | 77 | 6 |
| 2-[18F]FDM-6-P | ~12 | 4 | 2 | 13 | 4 | 10 | 6 | 8 | 5 |
| radiometabolite 5 | ~19.5 | 0.2 | 0.3 | 0.5 | 0.3 | 1.5 | 0.5 | 2 | 1 |
| UDP-2-[18F]FDG | ~21 | 0.1 | 0.2 | 1.1 | 0.5 | 1.8 | 0.7 | 2.7 | 1.0 |
| 2-[18F]FD-PGL | ~25 | 0.4 | 0.8 | 3 | 1 | 3.4 | 0.8 | 3 | 2 |
| 2-[18F]FDG-1,6-P2 | ~28 | 0.7 | 0.3 | 0.2 | 0.2 | 0.1 | 0.1 | 0.4 | 0.1 |
| radiometabolite 12 | ~29.5 | 0.2 | 0.2 | 0.1 | 0.2 | 0.06 | 0.07 | 0.2 | 0.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klebermass, E.-M.; Mahmudi, M.; Geist, B.K.; Pichler, V.; Vraka, C.; Balber, T.; Miller, A.; Haschemi, A.; Viernstein, H.; Rohr-Udilova, N.; et al. If It Works, Don’t Touch It? A Cell-Based Approach to Studying 2-[18F]FDG Metabolism. Pharmaceuticals 2021, 14, 910. https://doi.org/10.3390/ph14090910
Klebermass E-M, Mahmudi M, Geist BK, Pichler V, Vraka C, Balber T, Miller A, Haschemi A, Viernstein H, Rohr-Udilova N, et al. If It Works, Don’t Touch It? A Cell-Based Approach to Studying 2-[18F]FDG Metabolism. Pharmaceuticals. 2021; 14(9):910. https://doi.org/10.3390/ph14090910
Chicago/Turabian StyleKlebermass, Eva-Maria, Mahshid Mahmudi, Barbara Katharina Geist, Verena Pichler, Chrysoula Vraka, Theresa Balber, Anne Miller, Arvand Haschemi, Helmut Viernstein, Nataliya Rohr-Udilova, and et al. 2021. "If It Works, Don’t Touch It? A Cell-Based Approach to Studying 2-[18F]FDG Metabolism" Pharmaceuticals 14, no. 9: 910. https://doi.org/10.3390/ph14090910








