Effects of Topical or Intravitreal Application of Anti-Vascular Endothelial Growth Factor on Density of Intestinal Blood Vessels of Mice
Abstract
1. Introduction
2. Materials and Methods
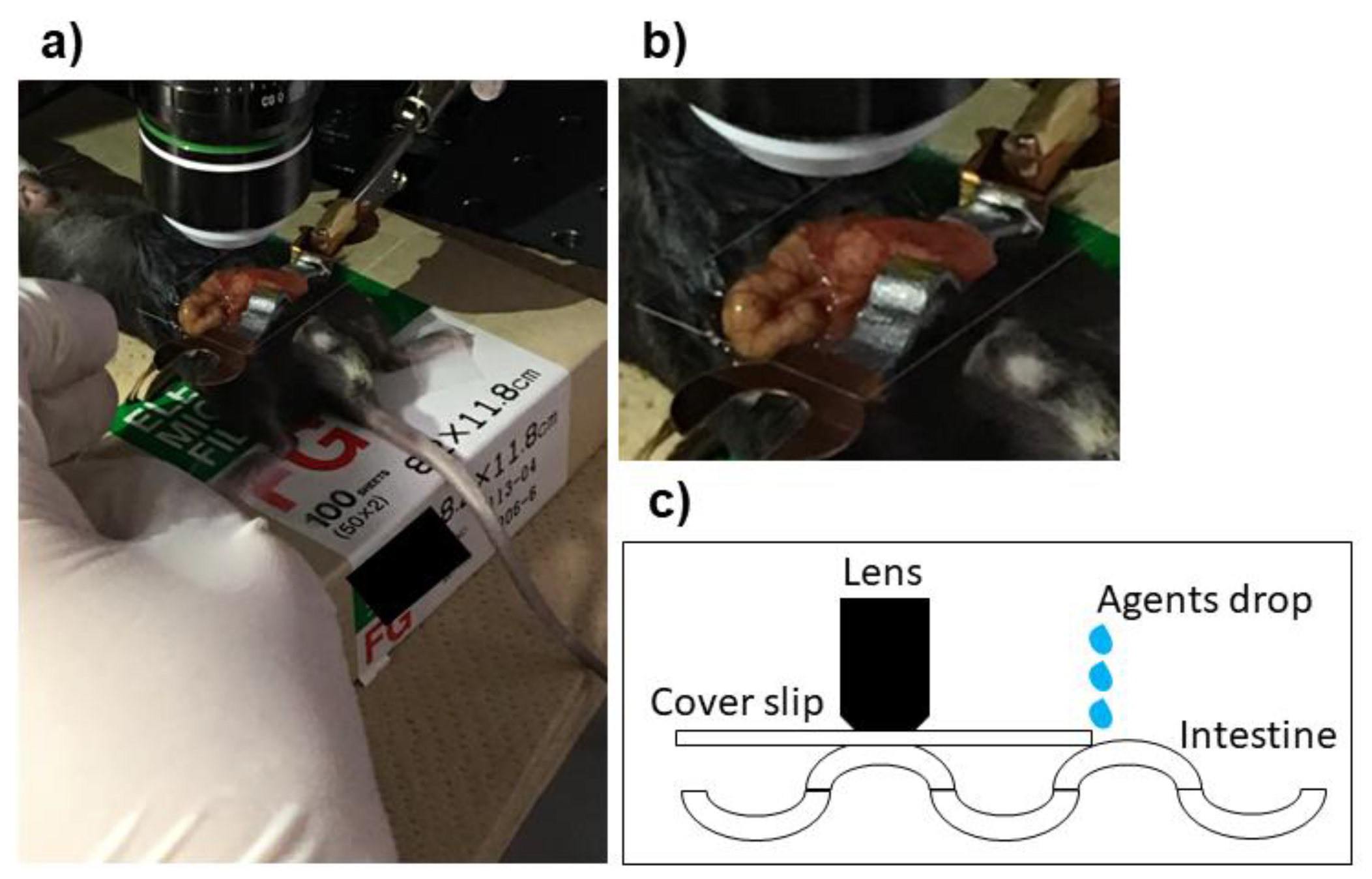
2.1. Preparation of Mice for Experiments
2.2. Surgical Procedure for Mice Experiment
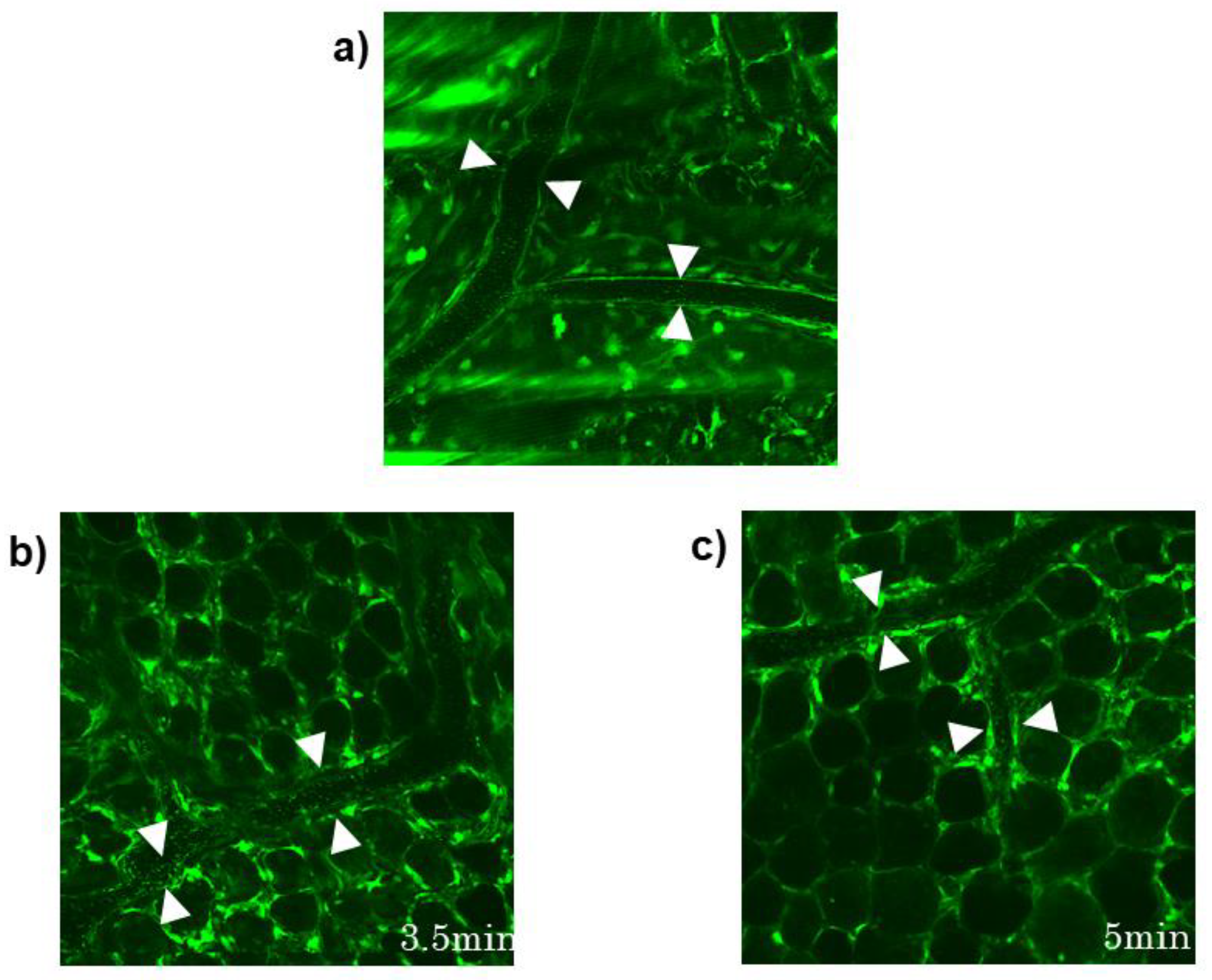
2.3. Preparation and Set Up of Microscope for Experiments
2.4. Administration of Anti-VEGF and Control Agents
2.5. Evaluations of Vascular Density
Statistical Analyses
3. Results
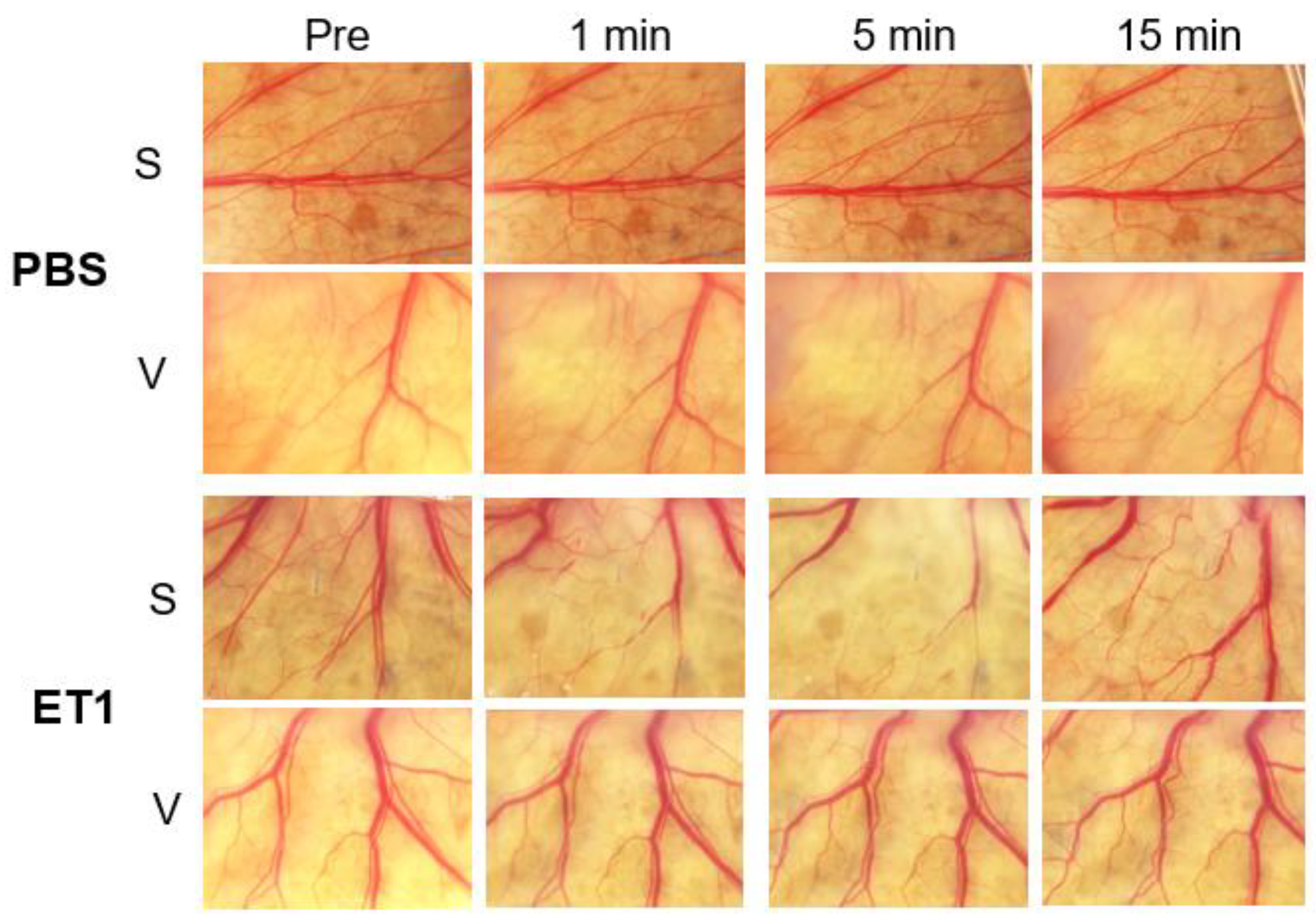
3.1. Vascular Density (VD) after Applications of Control Agents
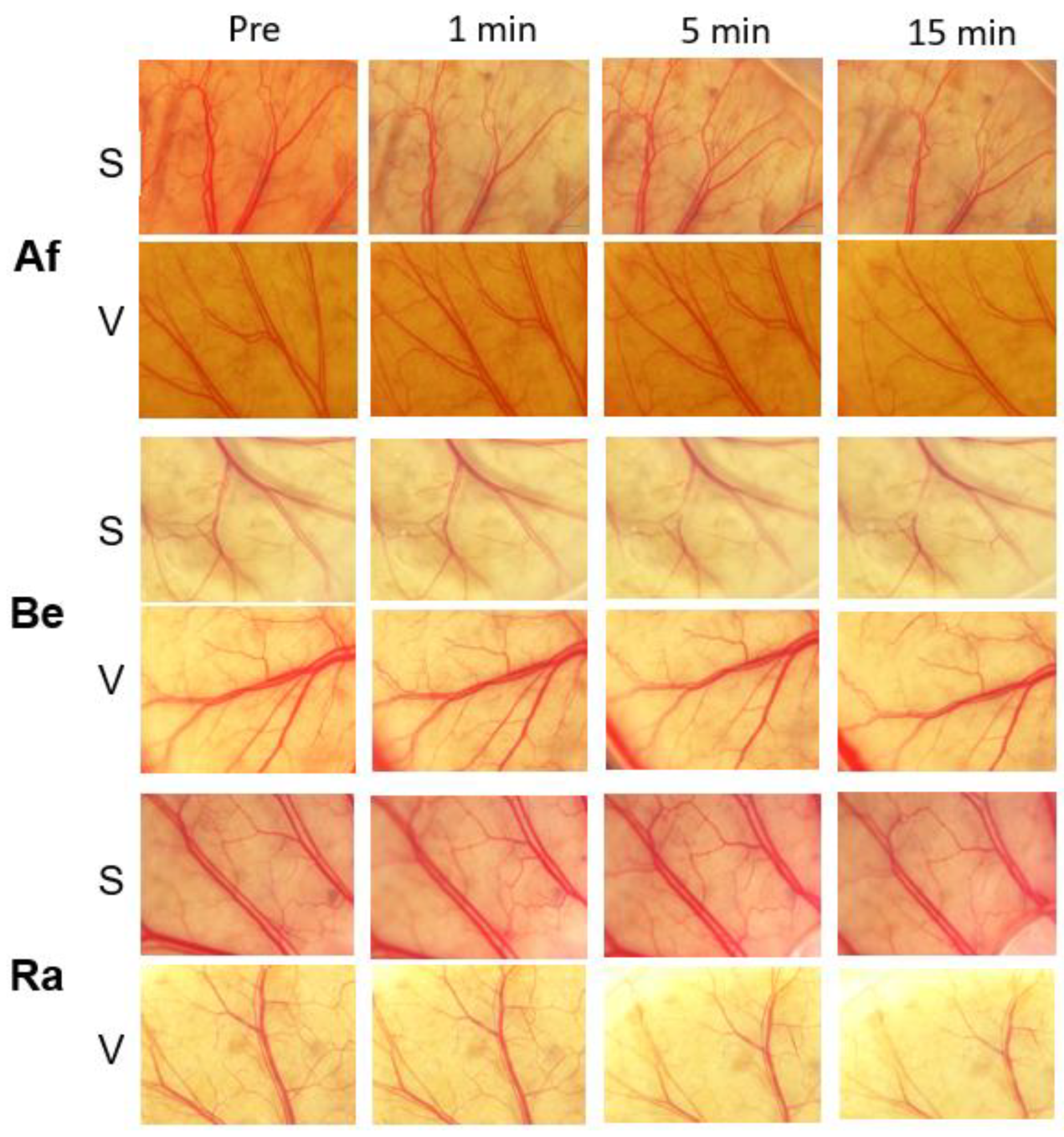
3.2. Evaluations of Vascular Density for Different Anti-VEGF Agents
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Nguyen, Q.D.; Brown, D.M.; Marcus, D.M.; Boyer, D.S.; Patel, S.; Feiner, L.; Gibson, A.; Sy, J.; Rundle, A.C.; Hopkins, J.J.; et al. Ranibizumab for diabetic macular edema: Results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology 2012, 119, 789–801. [Google Scholar] [CrossRef]
- Brown, D.M.; Schmidt-Erfurth, U.; Do, D.V.; Holz, F.G.; Boyer, D.S.; Midena, E.; Heier, J.S.; Terasaki, H.; Kaiser, P.K.; Marcus, D.M.; et al. Intravitreal aflibercept for diabetic macular edema: 100-week results from the VISTA and VIVID studies. Ophthalmology 2015, 122, 2044–2052. [Google Scholar] [CrossRef] [PubMed]
- Kodjikian, L.; Bellocq, D.; Bandello, F.; Loewenstein, A.; Chakravarthy, U.; Koh, A.; Augustin, A.; de Smet, M.D.; Chhablani, J.; Tufail, A.; et al. First-line treatment algorithm and guidelines in center-involving diabetic macular edema. Eur. J. Ophthalmol. 2019, 29, 573–584. [Google Scholar] [CrossRef]
- Zarbin, M.A. Anti-VEGF Agents and the Risk of Arteriothrombotic Events. Asia-Pac. J. Ophthalmol. 2018, 7, 63–67. [Google Scholar]
- Azzolini, C.; Pagani, I.S.; Pirrone, C.; Borroni, D.; Donati, S.; Al Oum, M.; Pigni, D.; Chiaravalli, A.M.; Vinciguerra, R.; Simonelli, F.; et al. Expression of VEGF-A, Otx homeobox and p53 family genes in proliferative vitreoretinopathy. Mediat. Inflamm. 2013, 2013, 857380. [Google Scholar] [CrossRef] [PubMed]
- Kamba, T.; McDonald, D.M. Mechanisms of adverse effects of anti-VEGF therapy for cancer. Br. J. Cancer 2007, 96, 1788–1795. [Google Scholar] [CrossRef]
- Sugimoto, M.; Nunome, T.; Sakamoto, R.; Kobayashi, M.; Kondo, M. Effect of intravitreal ranibizumab on the ocular circulation of the untreated fellow eye. Graefes Arch. Clin. Exp. Ophthalmol. 2017, 255, 1543–1550. [Google Scholar] [CrossRef] [PubMed]
- Avery, R.L.; A Castellarin, A.; Steinle, N.C.; Dhoot, D.S.; Pieramici, D.J.; See, R.; Couvillion, S.; A Nasir, M.; Rabena, M.D.; Le, K.; et al. Systemic pharmacokinetics following intravitreal injections of ranibizumab, bevacizumab or aflibercept in patients with neovascular AMD. Br. J. Ophthalmol. 2014, 98, 1636–1641. [Google Scholar] [CrossRef]
- Yoshida, I.; Shiba, T.; Taniguchi, H.; Takahashi, M.; Murano, T.; Hiruta, N.; Hori, Y.; Bujo, H.; Maeno, T. Evaluation of plasma vascular endothelial growth factor levels after intravitreal injection of ranibizumab and aflibercept for exudative age-related macular degeneration. Graefes Arch. Clin. Exp. Ophthalmol. 2014, 252, 1483–1489. [Google Scholar] [CrossRef]
- Wang, X.; Sawada, T.; Sawada, O.; Saishin, Y.; Liu, P.; Ohji, M. Serum and Plasma Vascular Endothelial Growth Factor Concentrations Before and After Intravitreal Injection of Aflibercept or Ranibizumab for Age-Related Macular Degeneration. Am. J. Ophthalmol. 2014, 158, 738–744. [Google Scholar] [CrossRef]
- Avery, R.L.; Castellarin, A.A.; Steinle, N.C.; Dhoot, D.S.; Pieramici, D.J.; See, R.; Couvillion, S.; Nasir, M.A.; Rabena, M.D.; Maia, M.; et al. Systemic pharmacokinetics and pharmacodynamics of intravitreal aflibercept, bevacizumab abd ranibizumab. Retina 2017, 37, 1847–1858. [Google Scholar] [CrossRef] [PubMed]
- Avery, R.L.; Gordon, G.M. Systemic safety of prolonged monthly anti-vascular endothelial growth factor therapy for diabetic macular edema: A systematic review and meta-analysis. JAMA Ophthalmol. 2016, 134, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Thulliez, M.; Angoulvant, D.; Pisella, P.-J.; Bejan-Angoulvant, T. Overview of systematic reviews and meta-analyses on systemic adverse events associated with intravitreal anti-vascular endothelial growth factor medication use. JAMA Ophthalmol. 2018, 136, 557–566. [Google Scholar] [CrossRef]
- Sugimoto, M.; Handa, C.; Hirano, K.; Sunaya, T.; Kondo, M. Intravitreal aflibercept for diabetic macular edema in real-world clinical practice in Japan: 24-month outcomes. Graefes Arch. Clin. Exp. Ophthalmol. 2022, 260, 3489–3498. [Google Scholar] [CrossRef] [PubMed]
- Koike, Y.; Li, B.; Chen, Y.; Miyake, H.; Lee, C.; Chi, L.; Wu, R.; Inoue, M.; Uchida, K.; Kusunoki, M.; et al. Live imaging of fetal intra-abdominal organs using Two-photon laser-scanning microscopy. Methods Mol. Biol. 2018, 1752, 63–69. [Google Scholar]
- Fukuda, Y.; Hirata, Y.; Yoshimi, H.; Kojima, T.; Kobayashi, Y.; Yanagisawa, M.; Masaki, T. Endothelin is a potent secretagogue for atrial natriuretic peptide in cultured rat atrial myocytes. Biochem. Biophys. Res. Commun. 1988, 155, 167–172. [Google Scholar] [CrossRef]
- Zudaire, E.; Gambardella, L.; Kurcz, C.; Vermeren, S. A computational tool for quantitative analysis of vascular networks. PLoS ONE 2011, 6, e27385. [Google Scholar]
- Thulliez, M.; Angoulvant, D.; Le Lez, M.L.; Jonville-Bera, A.P.; Pisella, P.J.; Gueyffier, F.; Bejan-Angoulvant, T. Cardiovascular events and bleeding risk associated with intravitreal antivascular endothelial growth factor monoclonal antibodies: Systematic review and meta-analysis. JAMA Ophthalmol. 2014, 132, 1317–1326. [Google Scholar] [CrossRef]
- Kitchens, J.W.; Do, D.V.; Boyer, D.S.; Thompson, D.; Gibson, A.; Saroj, N.; Vitti, R.; Berliner, A.J.; Kaiser, P.K. Comprehensive review of ocular and systemic safety events with intravitreal aflibercept injection in randomized controlled trials. Ophthalmology 2016, 123, 1511–1520. [Google Scholar] [CrossRef]
- Sugimoto, M.; Wakamatsu, Y.; Miyata, R.; Nunome, T.; Tenma, Y.; Matsubara, H.; Kondo, M.; Wada, H.; Nakatani, K. Expression of vascular infarction-related molecules after anti-vascular endothelium growth factor treatment for diabetic macular edema. Sci. Rep. 2019, 9, 12373. [Google Scholar] [CrossRef]
- Shweiki, D.; Itin, A.; Neufeld, G.; Gitay-Goren, H.; Keshet, E. Patterns of expression of vascular endothelial growth factor (VEGF) and VEGF receptors in mice suggest a role in hormonally regulated angiogenesis. J. Clin. Investig. 1993, 91, 2235–2243. [Google Scholar] [CrossRef]
- Ropert, S.; Vignaux, O.; Mir, O.; Goldwasser, F. VEGF pathway inhibition by anticancer agent sunitinib and susceptibility to atherosclerosis plaque disruption. Investig. New Drugs 2011, 29, 1497–1499. [Google Scholar] [CrossRef] [PubMed]
- Boswell, C.A.; Ferl, G.Z.; Mundo, E.E.; Bumbaca, D.; Schweiger, M.G.; Theil, F.-P.; Fielder, P.J.; Khawli, L.A. Effects of anti-VEGF on predicted antibody biodistribution: Roles of vascular volume, interstitial volume, and blood flow. PLoS ONE 2011, 6, e17874. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Wu, X.; Cheng, Z.; Lee, C.V.; LeCouter, J.; Campa, C.; Fuh, G.; Lowman, H.; Ferrara, N. Interaction between bevacizumab and murine VEGF-A: A reassessment. Investig. Ophthalmol. Vis. Sci. 2008, 49, 522–527. [Google Scholar] [CrossRef] [PubMed]
- Miki, K.; Miki, A.; Matsuoka, M.; Muramatsu, D.; Hackett, S.F.; Campochiaro, P.A. Effects of intraocular ranibizumab and bevacizumab in transgenic mice expressing human vascular endothelial growth factor. Ophthalmology 2009, 116, 1748–1754. [Google Scholar] [CrossRef]
- Irani, Y.; Scotney, P.; Nash, A.; Williams, K.A. Species cross-reactivity of antibodies used to treat ophthalmic conditions. Investig. Ophthalmol. Vis. Sci. 2016, 57, 586–591. [Google Scholar] [CrossRef]
- Feng, F.; Cheng, Y.; Liu, Q.-H. Bevacizumab treatment reduces retinal neovascularization in a mouse model of retinopathy of prematurity. Int. J. Ophthalmol. 2014, 7, 608–613. [Google Scholar]
- Akkoyun, I.; Karabay, G.; Haberal, N.; Dagdeviren, A.; Yilmaz, G.; Oto, S.; Erkanli, L.; Akova, Y.A. Structural consequences after intravitreal bevacizumab injection without increasing apoptotic cell death in a retinopathy of prematurity mouse model. Acta Ophthalmol. 2012, 90, 564–570. [Google Scholar] [CrossRef]
- Koike, Y.; Li, B.; Chen, Y.; Ganji, N.; Alganabi, M.; Miyake, H.; Lee, C.; Hock, A.; Wu, R.; Uchida, K.; et al. Live intravital intestine with blood flow visualization in neonatal mice using Two-photon laser scanning microscopy. Bio-Protocol 2021, 11, e3937. [Google Scholar] [CrossRef]

| Pre (%) | 1 min (%) | 5 min (%) | 15 min (%) | ||
|---|---|---|---|---|---|
| PBS | S | 46.3 ± 1.1 | 44.5 ± 0.8 | 44.8 ± 0.4 | 43.2 ± 1.4 |
| V | 44.8 ± 0.4 | 45.2 ± 0.6 | 45.4 ± 0.3 | 45.7 ± 0.1 | |
| ET1 | S | 46.7 ± 5.7 | 28.1 ± 8.6 ** | 32.1 ± 15.2 * | 34.0 ± 3.8 |
| V | 45.4 ± 5.7 | 43.5 ± 4.2 | 38.7 ± 8.2 | 40.7 ± 6.7 | |
| Pre (%) | 1 min (%) | 5 min (%) | 15 min (%) | ||
|---|---|---|---|---|---|
| Bevacizumab | S | 46.1 ± 0.4 | 46.7 ± 0.4 | 46.7 ± 0.4 | 46.3 ± 0.6 |
| V | 45.0 ± 0.5 | 45.4 ± 0.6 | 44.7 ± 0.6 | 45.6 ± 0.4 | |
| Ranibizumab | S | 44.7 ± 1.3 | 45.0 ± 2.1 | 44.7 ± 2.1 | 44.6 ± 2.6 |
| V | 45.8 ± 0.5 | 46.2 ± 0.4 | 45.2 ± 1.3 | 45.6 ± 0.3 | |
| Aflibercept | S | 46.5 ± 1.2 | 46.2 ± 0.9 | 45.9 ± 1.1 | 46.1 ± 1.3 |
| V | 47.0 ± 0.2 | 46.9 ± 0.1 | 46.6 ± 0.7 | 46.9 ± 0.1 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ichio, A.; Sugimoto, M.; Koike, Y.; Toiyama, Y.; Kondo, M. Effects of Topical or Intravitreal Application of Anti-Vascular Endothelial Growth Factor on Density of Intestinal Blood Vessels of Mice. Medicina 2023, 59, 809. https://doi.org/10.3390/medicina59040809
Ichio A, Sugimoto M, Koike Y, Toiyama Y, Kondo M. Effects of Topical or Intravitreal Application of Anti-Vascular Endothelial Growth Factor on Density of Intestinal Blood Vessels of Mice. Medicina. 2023; 59(4):809. https://doi.org/10.3390/medicina59040809
Chicago/Turabian StyleIchio, Atsushi, Masahiko Sugimoto, Yuhki Koike, Yuji Toiyama, and Mineo Kondo. 2023. "Effects of Topical or Intravitreal Application of Anti-Vascular Endothelial Growth Factor on Density of Intestinal Blood Vessels of Mice" Medicina 59, no. 4: 809. https://doi.org/10.3390/medicina59040809
APA StyleIchio, A., Sugimoto, M., Koike, Y., Toiyama, Y., & Kondo, M. (2023). Effects of Topical or Intravitreal Application of Anti-Vascular Endothelial Growth Factor on Density of Intestinal Blood Vessels of Mice. Medicina, 59(4), 809. https://doi.org/10.3390/medicina59040809






