Randomized Trial of General Strength and Conditioning versus Motor Control and Manual Therapy for Chronic Low Back Pain on Physical and Self-Report Outcomes
Abstract
1. Introduction
2. Materials and Methods
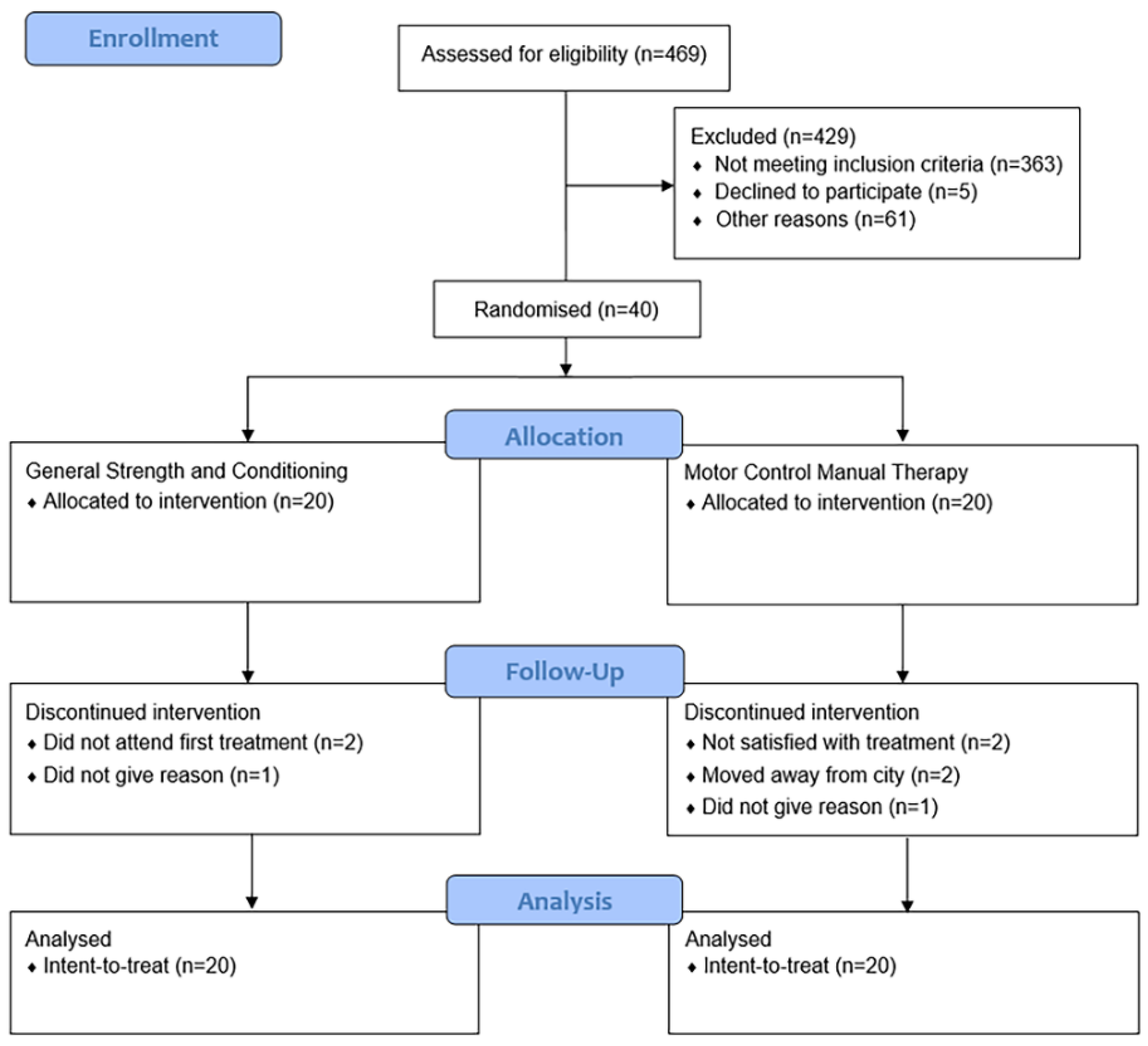
2.1. Participants
2.1.1. Inclusion and Exclusion
2.1.2. Randomization
2.2. Interventions
2.2.1. General Strength and Conditioning
2.2.2. Motor Control Exercise plus Manual Therapy
2.3. Outcomes
2.3.1. Physical Outcomes
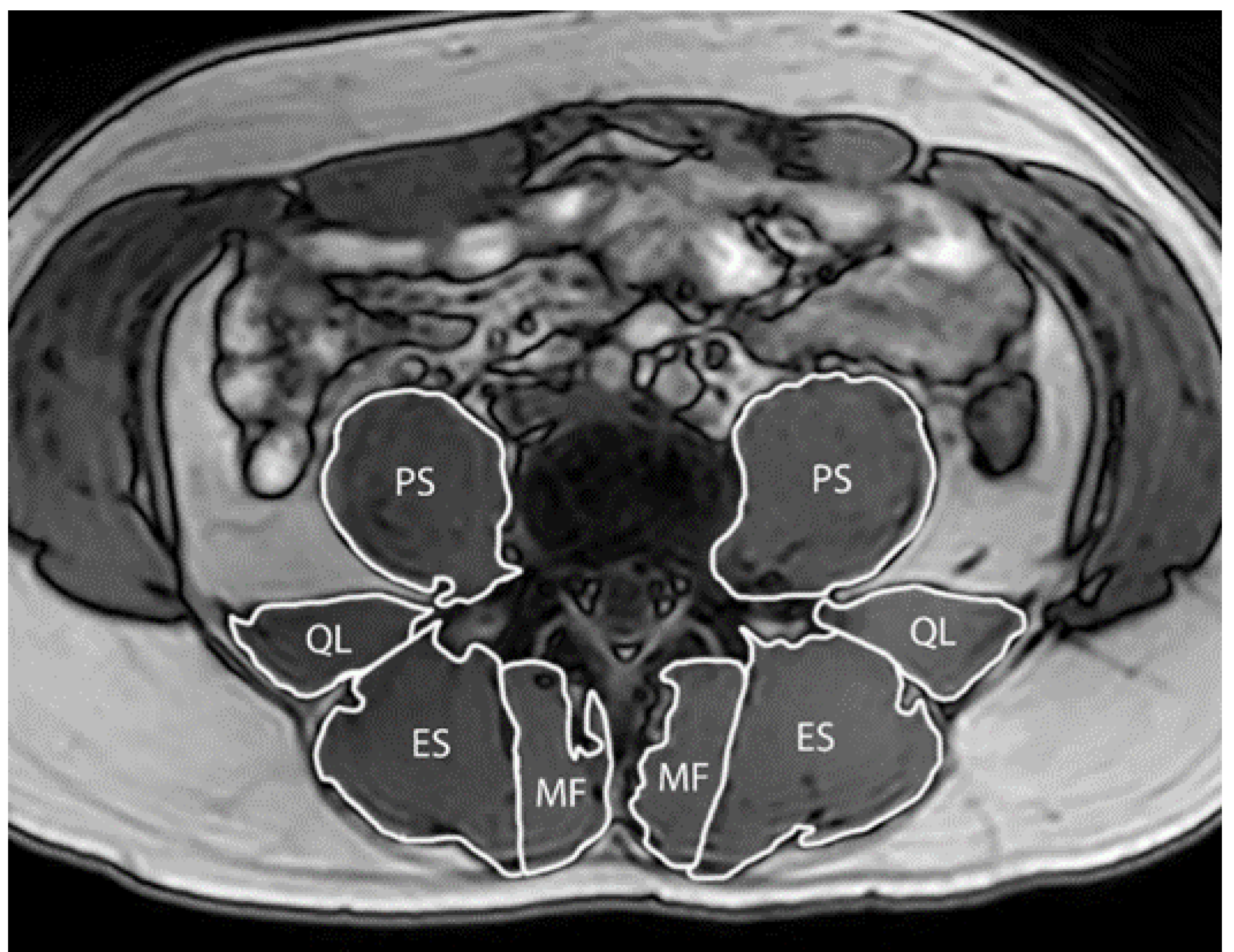
2.3.2. Paraspinal Muscle Size
2.3.3. Self-Report Outcomes
2.4. Statistical Analysis
3. Results
3.1. Descriptive Characteristics
3.2. Physical Outcomes
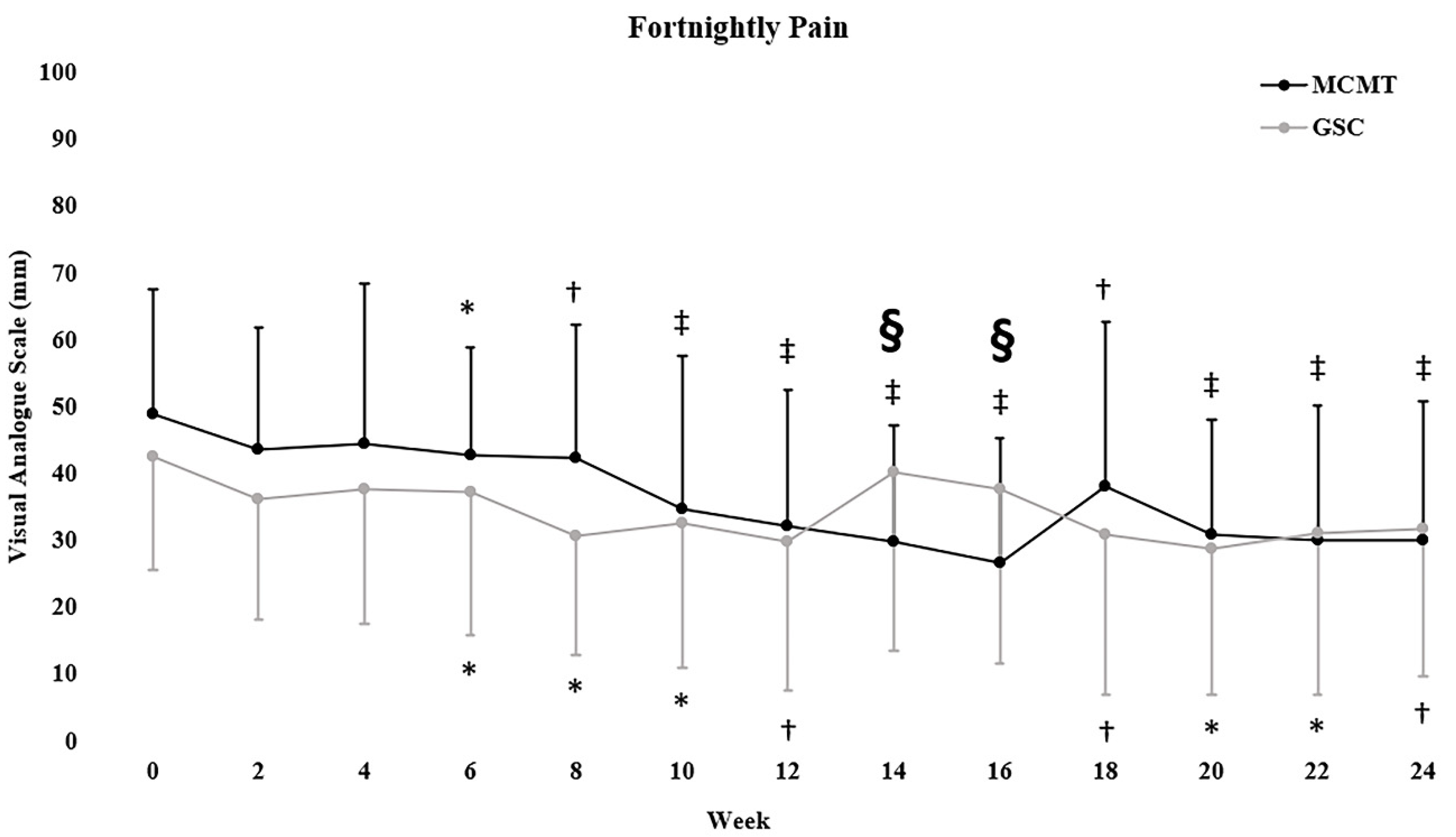
3.3. Self-Reported Outcomes
3.4. Paraspinal Muscle Size
4. Discussion
4.1. Physical Outcomes
4.2. Self-Reported Outcomes
4.3. Paraspinal Muscle Size
4.4. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethics Approval
Trial Registration
References
- Hoy, D.; Bain, C.; Williams, G.; March, L.; Brooks, P.; Blyth, F.; Woolf, A.; Vos, T.; Buchbinder, R. A systematic review of the global prevalence of low back pain. Arthritis Rheumatol. 2012, 64, 2028–2037. [Google Scholar] [CrossRef] [PubMed]
- Andersson, G.B. Epidemiological features of chronic low-back pain. Lancet 1999, 354, 581–585. [Google Scholar] [CrossRef]
- Vos, T.; Flaxman, A.D.; Naghavi, M.; Lozano, R.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; Aboyans, V.; et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: A systematic analysis for the global burden of disease study 2010. Lancet 2012, 380, 2163–2196. [Google Scholar] [CrossRef]
- Murray, C.J.; Vos, T.; Lozano, R.; Naghavi, M.; Flaxman, A.D.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; et al. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2013, 380, 2197–2223. [Google Scholar] [CrossRef]
- Walker, B.; Muller, R.; Grant, W. Low back pain in Australian adults: The economic burden. Asia Pac. J. Public Health 2003, 15, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Martin, B.I.; Deyo, R.A.; Mirza, S.K.; Turner, J.A.; Comstock, B.A.; Hollingworth, W.; Sullivan, S.D. Expenditures and health status among adults with back and neck problems. JAMA 2008, 299, 656–664. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.; Maher, C.G.; Hancock, M.J.; McAuley, J.H.; Herbert, R.D.; Costa, L.O. The prognosis of acute and persistent low-back pain: A meta-analysis. Can. Med. Assoc. J. 2012, 184, E613–E624. [Google Scholar] [CrossRef] [PubMed]
- Itz, C.J.; Geurts, J.; Van Kleef, M.; Nelemans, P. Clinical course of non-specific low back pain: A systematic review of prospective cohort studies set in primary care. Eur. J. Pain 2013, 17, 5–15. [Google Scholar] [CrossRef]
- Qaseem, A.; Wilt, T.J.; McLean, R.M.; Forciea, M.A. Noninvasive treatments for acute, subacute, and chronic low back pain: A clinical practice guideline from the American College of Physicians. Ann. Intern. Med. 2017, 166, 514–530. [Google Scholar] [CrossRef]
- Koes, B.W.; van Tulder, M.; Lin, C.-W.C.; Macedo, L.G.; McAuley, J.; Maher, C. An updated overview of clinical guidelines for the management of non-specific low back pain in primary care. Eur. Spine J. 2010, 19, 2075–2094. [Google Scholar] [CrossRef]
- Dagenais, S.; Caro, J.; Haldeman, S. A systematic review of low back pain cost of illness studies in the United States and internationally. Spine J. 2008, 8, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Searle, A.; Spink, M.; Ho, A.; Chuter, V. Exercise interventions for the treatment of chronic low back pain: A systematic review and meta-analysis of randomised controlled trials. Clin. Rehabil. 2015, 29, 1155–1167. [Google Scholar] [CrossRef] [PubMed]
- Owen, P.J.; Miller, C.T.; Mundell, N.L.; Verswijveren, S.J.; Tagliaferri, S.D.; Brisby, H.; Bowe, S.J.; Belavy, D.L. Which specific modes of exercise training are most effective for treating low back pain? Network Meta-analysis. Br. J. Sports Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Rubinstein, S.M.; de Zoete, A.; van Middelkoop, M.; Assendelft, W.J.; de Boer, M.R.; van Tulder, M.W. Benefits and harms of spinal manipulative therapy for the treatment of chronic low back pain: Systematic review and meta-analysis of randomised controlled trials. BMJ 2019, 364, I689. [Google Scholar] [CrossRef] [PubMed]
- Chou, R.; Baisden, J.; Carragee, E.J.; Resnick, D.K.; Shaffer, W.O.; Loeser, J.D. Surgery for low back pain: A review of the evidence for an American Pain Society clinical practice guideline. Spine 2009, 34, 1094–1109. [Google Scholar] [CrossRef]
- O’Sulliva Caneiro, J.P.; O’Keeffe, M.; O’Sullivan, K. Unraveling the complexity of low back pain. J. Orthop. Sports Phys. Ther. 2016, 46, 932–937. [Google Scholar]
- Tagliaferri, S.D.; Miller, C.T.; Owen, P.J.; Mitchell, U.H.; Brisby, H.; Fitzgibbon, B.; Masse-Alarie, H.; Van Oosterwijck, J.; Belavy, D.L. Domains of chronic low back pain and assessing treatment effectiveness: A clinical perspective. Pain Pract. 2020, 20, 211–225. [Google Scholar] [CrossRef]
- Owen, P.J.; Miller, C.T.; Rantalainen, T.; Simson, K.J.; Connell, D.; Hahne, A.J.; Trudel, G.; Ford, J.J.; Belavy, D.L. Exercise for the intervertebral disc: A 6-month randomised controlled trial in chronic low back pain. Eur. Spine J. 2020. [Google Scholar] [CrossRef]
- Taylor, J.B.; Goode, A.P.; George, S.Z.; Cook, C.E. Incidence and risk factors for first-time incident low back pain: A systematic review and meta-analysis. Spine J. 2014, 14, 2299–2319. [Google Scholar] [CrossRef]
- Ranger, T.A.; Cicuttini, F.M.; Jensen, T.S.; Peiris, W.L.; Hussain, S.M.; Fairley, J.; Urquhart, D.M. Are the size and composition of the paraspinal muscles associated with low back pain? A systematic review. Spine J. 2017, 17, 1729–1748. [Google Scholar] [CrossRef]
- Duque, I.; Parra, J.-H.; Duvallet, A. Maximal aerobic power in patients with chronic low back pain: A comparison with healthy subjects. Eur. Spine J. 2011, 20, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Chou, R.; Shekelle, P. Will this patient develop persistent disabling low back pain? JAMA 2010, 303, 1295–1302. [Google Scholar] [CrossRef] [PubMed]
- Lamé, I.E.; Peters, M.L.; Vlaeyen, J.W.; Kleef, M.V.; Patijn, J. Quality of life in chronic pain is more associated with beliefs about pain, than with pain intensity. Eur. J. Pain 2005, 9, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Kell, R.T.; Asmundson, G.J. A comparison of two forms of periodized exercise rehabilitation programs in the management of chronic nonspecific low-back pain. J. Strength Cond. Res. 2009, 23, 513–523. [Google Scholar] [CrossRef]
- Kell, R.T.; Risi, A.D.; Barden, J.M. The response of persons with chronic nonspecific low back pain to three different volumes of periodized musculoskeletal rehabilitation. J. Strength Cond. Res. 2011, 25, 1052–1064. [Google Scholar] [CrossRef]
- Ford, .J.J.; Hahne, A.J.; Surkitt, L.D.; Chan, A.Y.; Richards, M.C.; Slater, S.L.; Hinman, R.S.; Pizzari, T.; Davidson, M.; Taylor, N.F. Individualised physiotherapy as an adjunct to guideline-based advice for low back disorders in primary care: A randomised controlled trial. Br. J. Sports Med. 2016, 50, 237–245. [Google Scholar] [CrossRef]
- Simson, K.J.; Miller, C.T.; Ford, J.; Hahne, A.; Main, L.; Rantalainen, T.; Teo, W.P.; Teychenne, M.; Connell, D.; Trudel, G.; et al. Optimising conservative management of chronic low back pain: Study protocol for a randomised controlled trial. Trials 2017, 18, 184. [Google Scholar] [CrossRef]
- Macedo, L.G.; Latimer, J.; Maher, C.G.; Hodges, P.W.; McAuley, J.H.; Nicholas, M.K.; Tonkin, L.; Stanton, C.J.; Stanton, T.R.; Stafford, R. Effect of motor control exercises versus graded activity in patients with chronic nonspecific low back pain: A randomized controlled trial. Phys. Ther. 2012, 92, 363–377. [Google Scholar] [CrossRef]
- Clarke, C.L.; Ryan, C.G.; Martin, D.J. Pain neurophysiology education for the management of individuals with chronic low back pain: A systematic review and meta-analysis. Man. Ther. 2011, 16, 544–549. [Google Scholar] [CrossRef]
- Kodesh, E.; Weissman-Fogel, I. Exercise-induced hypoalgesia–interval versus continuous mode. Appl. Physiol. Nutr. Metab. 2014, 39, 829–834. [Google Scholar] [CrossRef]
- Caneiro, J.; O’Sullivan, P.; Lipp, O.V.; Mitchinson, L.; Oeveraas, N.; Bhalvani, P.; Abrugiato, R.; Thorkildsen, S.; Smith, A. Evaluation of implicit associations between back posture and safety of bending and lifting in people without pain. Scand. J. Pain 2018, 18, 719–728. [Google Scholar] [CrossRef] [PubMed]
- Saragiotto, B.T.; Maher, C.G.; Yamato, T.P.; Costa, L.O.; Menezes Costa, L.C.; Ostelo, R.W.; Macedo, L.G. Motor control exercise for chronic non specific low back pain. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Ford, J.; Hahne, A.; Chan, A.Y.; Surkitt, L. A classification and treatment protocol for low back disorders: Part 3 - Functional restoration for intervertebral disc related disorders. Phys. Ther. Rev. 2012, 17, 55–75. [Google Scholar] [CrossRef]
- Maitland, G. The Maitland concept: Assessment, examination and treatment by passive movement. Physical therapy of the low back. Clin. Phys. Ther. 1987, 13, 135–156. [Google Scholar]
- Ito, T.; Shirado, O.; Suzuki, H.; Takahashi, M.; Kaneda, K.; Strax, T.E. Lumbar trunk muscle endurance testing: An inexpensive alternative to a machine for evaluation. Arch. Phys. Med. Rehabil. 1996, 77, 75–79. [Google Scholar] [CrossRef]
- Fritz, J.M.; Irrgang, J.J. A comparison of a modified Oswestry low back pain disability questionnaire and the Quebec back pain disability scale. Phys. Ther. 2001, 81, 776–788. [Google Scholar] [CrossRef]
- Miller, R.P.; Kori, S.H.; Todd, D.D. The Tampa scale: A measure of kinisophobia. Clin. J. Pain 1991, 7, 51. [Google Scholar] [CrossRef]
- Ware, J.E., Jr.; Sherbourne, C.D. The MOS 36-item short-form health survey (SF-36): Conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar] [CrossRef]
- Ware, J.E.; Keller, S.D.; Kosinski, M. SF-36 Physical and Mental Health Summary Scales: A User’s Manual; Health Assessment Lab: Boston, MA, USA, 1994. [Google Scholar]
- Ohnhaus, E.E.; Adler, R. Methodological problems in the measurement of pain: A comparison between the verbal rating scale and the visual analogue scale. Pain 1975, 1, 379–384. [Google Scholar] [CrossRef]
- Abraha, I.; Montedori, A. Modified intention to treat reporting in randomised controlled trials: Systematic review. BMJ 2010, 340, c2697. [Google Scholar] [CrossRef]
- Horton, N.J.; Kleinman, K.P. Much ado about nothing: A comparison of missing data methods and software to fit incomplete data regression models. Am. Stat. 2007, 61, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Javadian, Y.; Behtash, H.; Akbari, M.; Taghipour-Darzi, M.; Zekavat, H. The effects of stabilizing exercises on pain and disability of patients with lumbar segmental instability. J. Back Musculoskelet. Rehabil. 2012, 25, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Radaelli, R.; Fleck, S.J.; Leite, T.; Leite, R.D.; Pinto, R.S.; Fernandes, L.; Simão, R. Dose-response of 1, 3, and 5 sets of resistance exercise on strength, local muscular endurance, and hypertrophy. J. Strength Cond. Res. 2015, 29, 1349–1358. [Google Scholar] [CrossRef] [PubMed]
- Jackson, J.K.; Shepherd, T.R.; Kell, R.T. The influence of periodized resistance training on recreationally active males with chronic nonspecific low back pain. J. Strength Cond. Res. 2011, 25, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Aasa, B.; Berglund, L.; Michaelson, P.; Aasa, U. Individualized low-load motor control exercises and education versus a high-load lifting exercise and education to improve activity, pain intensity, and physical performance in patients with low back pain: A randomized controlled trial. J. Orthop. Sports Phys. Ther. 2015, 45, 77–85. [Google Scholar] [CrossRef]
- Grgic, J.; Schoenfeld, B.J.; Davies, T.B.; Lazinica, B.; Krieger, J.W.; Pedisic, Z. Effect of resistance training frequency on gains in muscular strength: A systematic review and meta-analysis. Sports Med. 2018, 48, 1207–1220. [Google Scholar] [CrossRef]
- Schoenfeld, B.J.; Grgic, J.; Ogborn, D.; Krieger, J.W. Strength and hypertrophy adaptations between low-vs. high-load resistance training: A systematic review and meta-analysis. J. Strength Cond. Res. 2017, 31, 3508–3523. [Google Scholar] [CrossRef]
- Chan, C.W.; Mok, N.W.; Yeung, E.W. Aerobic exercise training in addition to conventional physiotherapy for chronic low back pain: A randomized controlled trial. Arch. Phys. Med. Rehabil. 2011, 92, 1681–1685. [Google Scholar] [CrossRef]
- Shvartz, E.; Reibold, R. Aerobic fitness norms for males and females aged 6 to 75 years: A review. Aviat. Space Environ. Med. 1990, 61, 3–11. [Google Scholar]
- Jordan, J.L.; Holden, M.A.; Mason, E.E.; Foster, N.E. Interventions to improve adherence to exercise for chronic musculoskeletal pain in adults. Cochrane Database Syst. Rev. 2010. [Google Scholar] [CrossRef]
- Pettitt, R.W.; Udermann, B.E.; Reineke, D.M.; Wright, G.A.; Battista, R.A.; Mayer, J.M.; Murray, S.R. Time-course of delayed onset muscle soreness evoked by three intensities of lumbar eccentric exercise. Athl. Train. Sports Health Care 2010, 2, 171–176. [Google Scholar] [CrossRef]
- Veqar, Z.; Kalra, R. Causes and management of delayed onset muscle soreness: A review. Elixir Hum. Physio 2013, 55, 13205–13211. [Google Scholar]
- Mannion, A.F.; Balagué, F.; Pellisé, F.; Cedraschi, C. Pain measurement in patients with low back pain. Nat. Rev. Rheumatol. 2007, 3, 610. [Google Scholar] [CrossRef] [PubMed]
- Vianin, M. Psychometric properties and clinical usefulness of the Oswestry Disability Index. J. Chiropr. Med. 2008, 7, 161–163. [Google Scholar] [CrossRef]
- Gomes-Neto, M.; Lopes, J.M.; Conceição, C.S.; Araujo, A.; Brasileiro, A.; Sousa, C.; Carvalho, V.O.; Arcanjo, F.L. Stabilization exercise compared to general exercises or manual therapy for the management of low back pain: A systematic review and meta-analysis. Phys. Ther. Sport 2017, 23, 136–142. [Google Scholar] [CrossRef]
- Crombez, G.; Vlaeyen, J.W.; Heuts, P.H.; Lysens, R. Pain-related fear is more disabling than pain itself: Evidence on the role of pain-related fear in chronic back pain disability. Pain 1999, 80, 329–339. [Google Scholar] [CrossRef]
- Monticone, M.; Ferrante, S.; Rocca, B.; Baiardi, P.; Dal Farra, F.; Foti, C. Effect of a long-lasting multidisciplinary program on disability and fear-avoidance behaviors in patients with chronic low back pain: Results of a randomized controlled trial. Clin. J. Pain 2013, 29, 929–938. [Google Scholar] [CrossRef]
- Henschke, N.; Ostelo, R.W.; van Tulder, M.W.; Vlaeyen, J.W.; Morley, S.; Assendelft, W.J.; Main, C.J. Behavioural treatment for chronic low back pain. Cochrane Database Syst. Rev. 2010. [Google Scholar] [CrossRef]
- Danneels, L.A.; Vanderstraeten, G.G.; Cambier, D.C.; Witvrouw, E.E.; Bourgois, J.; Dankaerts, W.; De Cuyper, H.J. Effects of three different training modalities on the cross sectional area of the lumbar multifidus muscle in patients with chronic low back pain. Br. J. Sports Med. 2001, 35, 186–191. [Google Scholar] [CrossRef]
- Chung, S.; Lee, J.; Yoon, J. Effects of stabilization exercise using a ball on mutifidus cross-sectional area in patients with chronic low back pain. J. Sports Sci. Med. 2013, 12, 533. [Google Scholar]
- Gupta, S.K. Intention-to-treat concept: A review. Perspect. Clin. Res. 2011, 2, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Berglund, L.; Aasa, B.; Michaelson, P.; Aasa, U. Effects of Low-Load Motor Control Exercises and a High-Load Lifting Exercise on Lumbar Multifidus Thickness. Spine 2017, 42, E876–E882. [Google Scholar] [CrossRef]
- Higgins, J.P.; Green, S. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions 5.1.0; The Cochrane Collaboration: London, UK, 2011; pp. 33–49. [Google Scholar]
- Hoffmann, T.C.; Maher, C.G.; Briffa, T.; Sherrington, C.; Bennell, K.; Alison, J.; Singh, M.F.; Glasziou, P.P. Prescribing exercise interventions for patients with chronic conditions. Can. Med. Assoc. J. 2016, 188, 510–518. [Google Scholar] [CrossRef] [PubMed]
- Kamper, S.J. Generalizability: Linking evidence to practice. J. Orthop. Sports Phys. Ther. 2020, 50, 45–46. [Google Scholar] [CrossRef] [PubMed]
- Saragiotto, B.T.; Maher, C.G.; Hancock, M.J.; Koes, B.W. Subgrouping patients with nonspecific low back pain: Hope or hype? J. Orthop. Sports Phys. Ther. 2017, 47, 44–48. [Google Scholar] [CrossRef] [PubMed]
| GSC (n = 20) | MCMT (n = 20) | |
|---|---|---|
| Age, years | 34.8 (4.9) | 34.6 (7.2) |
| Male, n (%) Female, n (%) | 11 (55) 9 (45) | 10 (50) 10 (50) |
| Height, cm | 169.6 (7.7) | 172.5 (9.1) |
| Weight, kg | 77.8 (13.5) | 76.9 (16.8) |
| Body Mass Index, (kg/m2) | 27.1 (4.9) | 25.4 (4.2) |
| Medication, n (%) b | 0 (0) | 1 (5) |
| Low Back Pain (0–100) c | 41.4 (17.8) | 48.9 (19.1) |
| Baseline Values and Within-Group Changes | ||||||
|---|---|---|---|---|---|---|
| GSC | MCMT | GSC vs. MCMT | ||||
| Mean (SD) Mean (95% CI) | p-Value | Mean (SD) Mean (95% CI) | p-Value | Net Difference (95% CI) | Group × Time | |
| Trunk Extension Endurance (s) * | ||||||
| Baseline | 101.9 (58.2) | <0.001 | 88.8 (35.9) | 0.305 | 51.5 (4.5, 98.4) | 0.007 |
| ∆ 3 months | 74.8 (38.8, 110.8) | 23.3 (−6.0, 52.6) | ||||
| ∆ 6 months | 133.7 (97.7, 169.7) | <0.001 | 51.8 (22.5, 81.1) | <0.001 | 81.8 (34.8, 128.8) | 0.004 |
| Trunk Flexion Endurance (s) * | ||||||
| Baseline | 81.4 (71.5) | 0.001 | 60.4 (40.5) | 0.864 | 19.3 (−11.7, 50.5) | 0.034 |
| ∆ 3 months | 30.4 (5.6, 55.1) | 11.0 (−6.7, 28.7) | ||||
| ∆ 6 months | 68.4 (43.6, 93.2) | <0.001 | 16.8 (−0.9, 34.5) | 0.053 | 51.5 (20.5, 82.6) | 0.004 |
| 1-RM Leg Press (kg) | ||||||
| Baseline | 141.1 (44.2) | <0.001 | 130.4 (43.6) | 0.040 | 16.6 (−4.5, 37.7) | 0.123 |
| ∆ 3 months | 29.6 (13.0, 46.2) | 13.1 (0.6, 25.5) | ||||
| ∆ 6 months | 47.7 (30.7, 64.7) | <0.001 | 23.1 (10.6, 35.5) | <0.001 | 24.7 (3.4, 46.0) | 0.023 |
| Leg Press Endurance (repetitions) | ||||||
| Baseline | 18.6 (5.4) | 0.001 | 21.9 (5.8) | 0.280 | 8.7 (1.4, 15.9) | 0.019 |
| ∆ 3 months | 10.7 (4.6, 16.9) | 1.9 (−1.5, 5.4) | ||||
| ∆ 6 months | 15.4 (9.1, 21.7) | <0.001 | 6.2 (2.7, 9.7) | 0.001 | 9.1 (1.7, 16.4) | 0.015 |
| Peak Oxygen Consumption (mL/kg/min) | ||||||
| Baseline | 38.0 (8.4) | 0.027 | 38.8 (6.7) | 0.333 | 3.2 (0.4, 6.0) | 0.025 |
| ∆ 3 months | 2.2 (0.2, 4.1) | −1.0 (−3.0, 1.0) | ||||
| ∆ 6 months | 2.4 (0.4, 4.3) | 0.019 | 1.1 (−1.0, 3.2) | 0.295 | 1.3 (−1.6, 4.2) | 0.380 |
| Baseline Values and Within-Group Changes | ||||||
|---|---|---|---|---|---|---|
| GSC | MCMT | GSC vs MCMT | ||||
| Mean (SD) Mean (95% CI) | p-Value | Mean (SD) Mean (95% CI) | p-Value | Net Difference (95% CI) | Group × Time | |
| Oswestry Disability Index (0–100) | ||||||
| Baseline | 24.5 (12.1) | <0.001 <0.001 | 23.4 (8.5) | 0.031 <0.001 | −6.7 (−12.2, −1.3) −5.7 (−11.2, −0.2) | 0.015 0.041 |
| ∆ 3 months | −11.3 (−15.0, −7.6) | −4.5 (−8.5, −0.4) | ||||
| ∆ 6 months | −13.5 (−17.2, −9.8) | −7.7 (−11.8, −3.5) | ||||
| Tampa Scale of Kinesiophobia (17–68) | ||||||
| Baseline | 38.7 (5.6) | <0.001 <0.001 | 38.6 (6.0) | 0.658 0.321 | −6.7 (−10.0, −3.4) −6.6 (−9.9, −3.2) | <0.001 <0.001 |
| ∆ 3 months | −6.0 (−8.7, −3.4) | 0.4 (−1.5, 2.4) | ||||
| ∆ 6 months | −7.4 (−10.0, −4.7) | −1.0 (−3.0, 1.0) | ||||
| SF-36 Physical Health (0–100) | ||||||
| Baseline | 42.6 (8.4) | <0.001 <0.001 | 41.2 (9.6) | 0.001 <0.001 | −0.7 (−6.7, 5.3) 0.6 (−5.5, 6.7) | 0.813 0.843 |
| ∆ 3 months | 7.5 (3.7, 11.3) | 8.2 (3.5, 12.9) | ||||
| ∆ 6 months | 10.4 (6.6, 14.2) | 9.7 (4.9, 14.5) | ||||
| SF-36 Mental Health (0–100) | ||||||
| Baseline | 31.5 (12.4) | 0.098 0.001 | 33.4 (13.9) | 0.959 0.106 | 7.1 (−3.8, 18.0) 7.5 (−3.5, 18.6) | 0.202 0.179 |
| ∆ 3 months | 6.8 (−1.2, 14.7) | −0.2 (−7.6, 7.2) | ||||
| ∆ 6 months | 13.7 (5.7, 21.6) | 6.2 (−1.3, 13.7) | ||||
| Baseline Values and Within-Group Changes | ||||||
|---|---|---|---|---|---|---|
| GSC | MCMT | GSC vs. MCMT | ||||
| Mean (SD) Mean (95% CI) | p-Value | Mean (SD) Mean (95% CI) | p-Value | Net Difference (95% CI) | Group × Time | |
| Multifidus Volume (cm3) | ||||||
| Baseline | 18.2 (4.0) | 0.102 | 18.0 (5.5) | 0.477 | 0.6 (−0.1, 1.4) | 0.096 |
| ∆ 3 months | 0.4 (−0.1, 1.0) | −0.2 (−0.7, 0.3) | ||||
| ∆ 6 months | 0.8 (0.3, 1.3) | 0.003 | 0.2 (−0.3, 0.7) | 0.463 | 0.6 (−0.1, 1.4) | 0.116 |
| Erector Spinae Volume (cm3) | ||||||
| Baseline | 45.9 (11.2) | 0.506 | 48.7 (15.3) | 0.884 | 0.5 (−1.2, 2.2) | 0.570 |
| ∆ 3 months | 0.4 (−0.8, 1.6) | −0.1 (−1.3, 1.1) | ||||
| ∆ 6 months | 0.1 (−1.1, 1.3) | 0.810 | 0.1 (−1.1, 1.4) | 0.822 | 0.0 (−1.7, 1.7) | 0.998 |
| Psoas Major Volume (cm3) | ||||||
| Baseline | 32.8 (9.7) | 0.697 | 32.5 (11.5) | 0.157 | 0.3 (−1.1, 1.7) | 0.677 |
| ∆ 3 months | −0.2 (−1.4, 0.9) | −0.5 (−1.3, 0.2) | ||||
| ∆ 6 months | 0.2 (−0.9, 1.4) | 0.686 | 0.0 (−0.7, 0.8) | 0.967 | 0.2 (−1.2, 1.7) | 0.761 |
| Quadratus Lumborum Volume (cm3) | ||||||
| Baseline | 11.2 (3.4) | 0.428 | 11.6 (4.4) | 0.697 | 0.0 (−0.8, 0.9) | 0.933 |
| ∆ 3 months | 0.2 (−0.3, 0.6) | 0.1 (−0.6, 0.9) | ||||
| ∆ 6 months | 0.1 (−0.4, 0.5) | 0.813 | 0.3 (−0.5, 1.0) | 0.472 | −0.2 (−1.0, 0.6) | 0.614 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tagliaferri, S.D.; Miller, C.T.; Ford, J.J.; Hahne, A.J.; Main, L.C.; Rantalainen, T.; Connell, D.A.; Simson, K.J.; Owen, P.J.; Belavy, D.L. Randomized Trial of General Strength and Conditioning versus Motor Control and Manual Therapy for Chronic Low Back Pain on Physical and Self-Report Outcomes. J. Clin. Med. 2020, 9, 1726. https://doi.org/10.3390/jcm9061726
Tagliaferri SD, Miller CT, Ford JJ, Hahne AJ, Main LC, Rantalainen T, Connell DA, Simson KJ, Owen PJ, Belavy DL. Randomized Trial of General Strength and Conditioning versus Motor Control and Manual Therapy for Chronic Low Back Pain on Physical and Self-Report Outcomes. Journal of Clinical Medicine. 2020; 9(6):1726. https://doi.org/10.3390/jcm9061726
Chicago/Turabian StyleTagliaferri, Scott D., Clint T. Miller, Jon J. Ford, Andrew J. Hahne, Luana C. Main, Timo Rantalainen, David A. Connell, Katherine J. Simson, Patrick J. Owen, and Daniel L. Belavy. 2020. "Randomized Trial of General Strength and Conditioning versus Motor Control and Manual Therapy for Chronic Low Back Pain on Physical and Self-Report Outcomes" Journal of Clinical Medicine 9, no. 6: 1726. https://doi.org/10.3390/jcm9061726
APA StyleTagliaferri, S. D., Miller, C. T., Ford, J. J., Hahne, A. J., Main, L. C., Rantalainen, T., Connell, D. A., Simson, K. J., Owen, P. J., & Belavy, D. L. (2020). Randomized Trial of General Strength and Conditioning versus Motor Control and Manual Therapy for Chronic Low Back Pain on Physical and Self-Report Outcomes. Journal of Clinical Medicine, 9(6), 1726. https://doi.org/10.3390/jcm9061726





