Supplementation with Lactiplantibacillus brevis GKEX Combined with Resistance Exercise Training Improves Muscle Mass, Strength Performance, and Body Fat Condition in Healthy Humans
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Participants
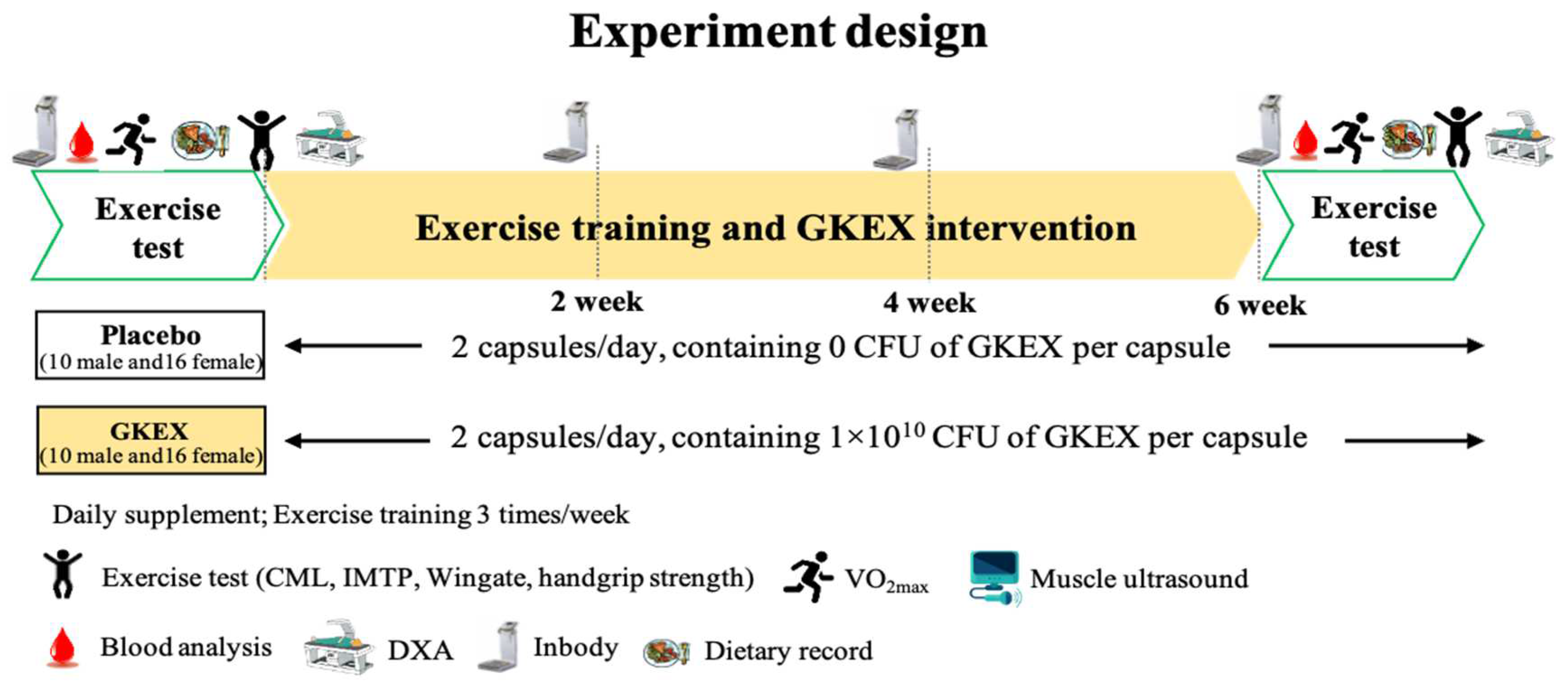
2.3. Experimental Design
2.4. Resistance Exercise Training
2.5. Maximal Oxygen Uptake (VO2max)
2.6. Handgrip Strength Test
2.7. Countermovement Jump Assessment (CMJ)
2.8. Isometric Mid-Thigh Pull (IMTP)
2.9. Wingate Anaerobic Test (WAnT)
2.10. Body Composition
2.11. Ultrasonic Measurement of Muscle and Fascia Thickness
2.12. Clinical Biochemistry and Hematology Analysis
2.13. Statistics
3. Results
3.1. Effects of 6-Week Supplementation with GKEX on Body Composition Change
3.2. Effects of 6-Week Supplementation with GKEX on Muscle Thickness and Fascia
3.3. Effects of 6-Week Supplementation with GKEX on Maximum Handgrip Strength
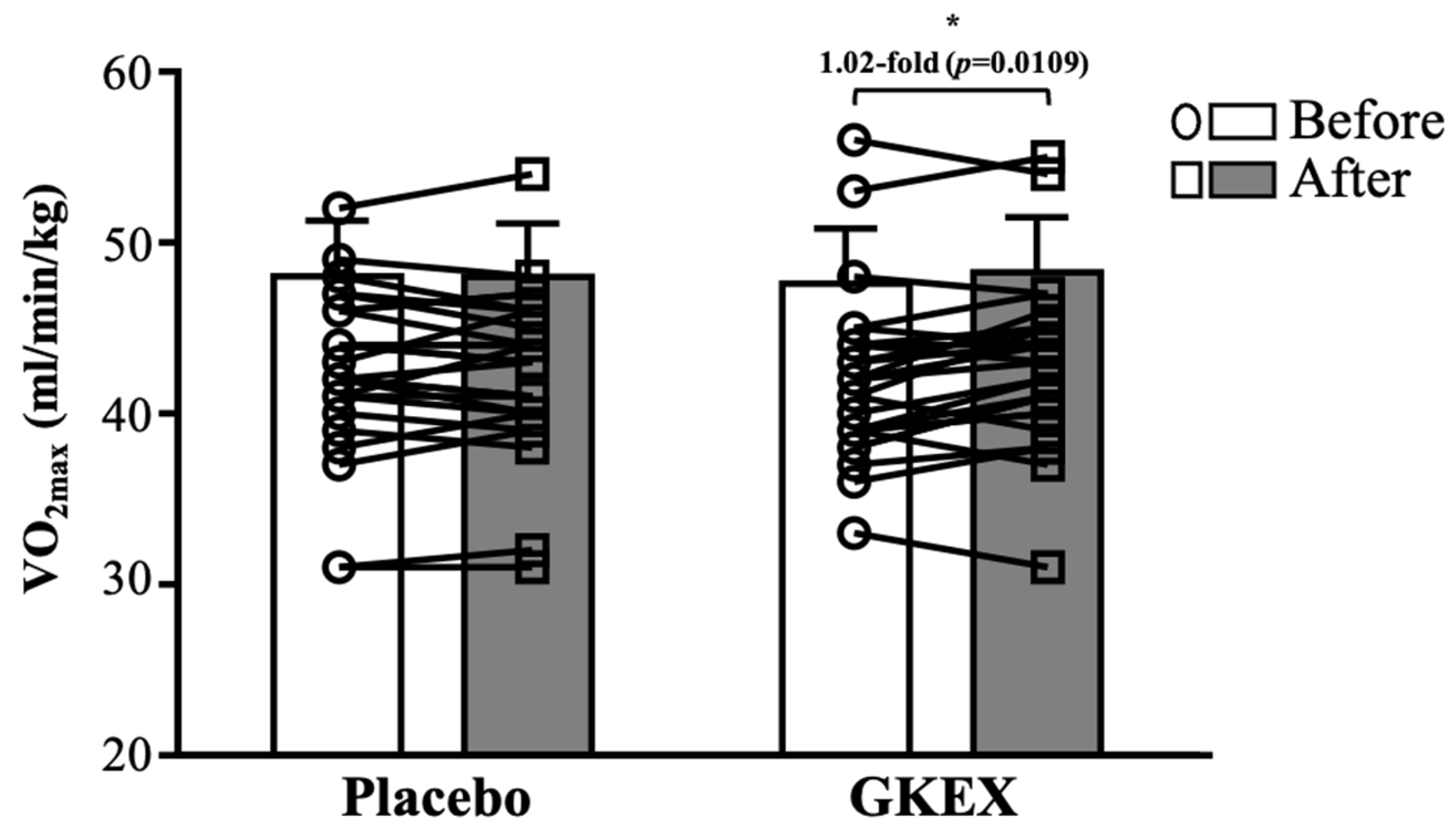
3.4. Effects of 6-Week Supplementation with GKEX on Maximal Oxygen Uptake (VO2max)
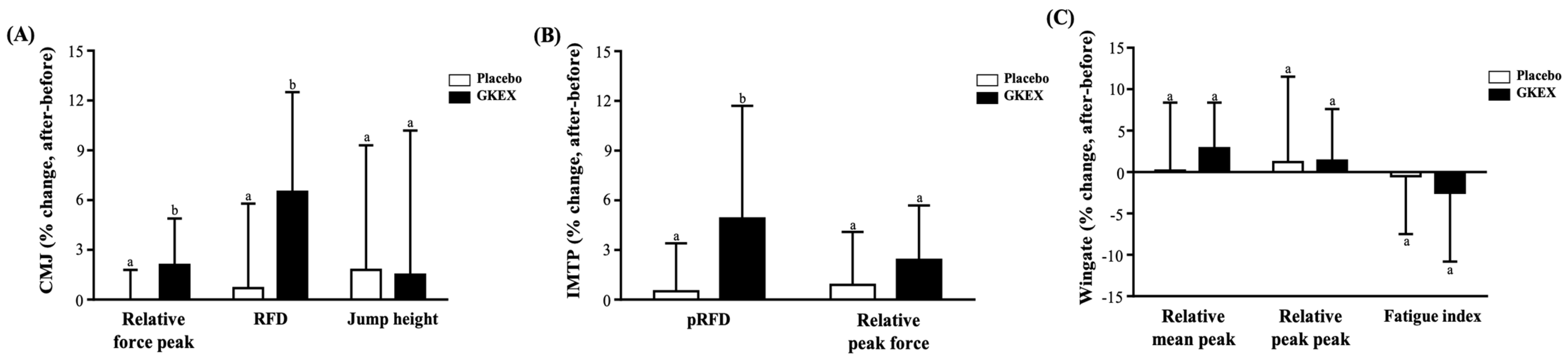
3.5. Effects of 6-Week Supplementation with GKEX on Maximum Vertical Jump Height of Exercise Performance
3.6. Effects of 6-Week Supplementation with GKEX on Isotonic Muscle Strength of Exercise Performance
3.7. Effects of 6-Week Supplementation with GKEX on Anaerobic Exercise Performance
3.8. Effects of 6-Week GKEX Intervention on Biochemical and Hematological Characteristics of Subjects
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anderson, E.; Durstine, J.L. Physical activity, exercise, and chronic diseases: A brief review. Sports Med. Health Sci. 2019, 1, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.B.; Park, J.H.; Park, H.S.; Kim, H.J.; Park, J.J. Effects of whey protein supplement on 4-week resistance exercise-induced improvements in muscle mass and isokinetic muscular function under dietary control. Nutrients 2023, 15, 1003. [Google Scholar] [CrossRef] [PubMed]
- Prokopidis, K.; Giannos, P.; Kirwan, R.; Ispoglou, T.; Galli, F.; Witard, O.C.; Triantafyllidis, K.K.; Kechagias, K.S.; Morwani-Mangnani, J.; Ticinesi, A.; et al. Impact of probiotics on muscle mass, muscle strength and lean mass: A systematic review and meta-analysis of randomized controlled trials. J. Cachexia Sarcopenia Muscle 2023, 14, 30–44. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Lee, M.C.; Lee, C.C.; Ng, K.S.; Hsu, Y.J.; Tsai, T.Y.; Young, S.L.; Lin, J.S.; Huang, C.C. Effect of Lactobacillus plantarum TWK10 on exercise physiological adaptation, performance, and body composition in healthy humans. Nutrients 2019, 11, 2836. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The international scientific association for probiotics and prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: Dietary supplements and the high-performance athlete. Br. J. Sports Med. 2018, 52, 439–455. [Google Scholar] [CrossRef] [PubMed]
- Dukes, A.; Davis, C.; El Refaey, M.; Upadhyay, S.; Mork, S.; Arounleut, P.; Johnson, M.H.; Hill, W.D.; Isales, C.M.; Hamrick, M.W. The aromatic amino acid tryptophan stimulates skeletal muscle IGF1/p70s6k/mTor signaling in vivo and the expression of myogenic genes in vitro. Nutrition 2015, 31, 1018–1024. [Google Scholar] [CrossRef] [PubMed]
- Jäger, R.; Zaragoza, J.; Purpura, M.; Iametti, S.; Marengo, M.; Tinsley, G.M.; Anzalone, A.J.; Oliver, J.M.; Fiore, W.; Biffi, A.; et al. Probiotic Administration increases amino acid absorption from plant protein: A placebo-controlled, randomized, double-blind, multicenter, crossover study. Probiotics Antimicrob. Proteins 2020, 12, 1330–1339. [Google Scholar] [CrossRef] [PubMed]
- Prokopidis, K.; Chambers, E.; Ni Lochlainn, M.; Witard, O.C. Mechanisms linking the gut-muscle axis with muscle protein metabolism and anabolic resistance: Implications for older adults at risk of sarcopenia. Front. Physiol. 2021, 12, 770455. [Google Scholar] [CrossRef]
- Storelli, G.; Defaye, A.; Erkosar, B.; Hols, P.; Royet, J.; Leulier, F. Lactobacillus plantarum promotes drosophila systemic growth by modulating hormonal signals through TOR-dependent nutrient sensing. Cell Metabolism 2011, 14, 403–414. [Google Scholar] [CrossRef]
- Chen, L.H.; Huang, S.Y.; Huang, K.C.; Hsu, C.C.; Yang, K.C.; Li, L.A.; Chan, C.H.; Huang, H.Y. Lactobacillus paracasei PS23 decelerated age-related muscle loss by ensuring mitochondrial function in SAMP8 mice. Aging 2019, 11, 756–770. [Google Scholar] [CrossRef] [PubMed]
- Shokri-Mashhadi, N.; Navab, F.; Ansari, S.; Rouhani, M.H.; Hajhashemy, Z.; Saraf-Bank, S. A meta-analysis of the effect of probiotic administration on age-related sarcopenia. Food Sci. Nutr. 2023, 11, 4975–4987. [Google Scholar] [CrossRef] [PubMed]
- Mante, E.S.; Sakyi-Dawson, E.; Amoa-Awua, W.K. Antimicrobial interactions of microbial species involved in the fermentation of cassava dough into agbelima with particular reference to the inhibitory effect of lactic acid bacteria on enteric pathogens. Int. J. Food Microbiol. 2003, 89, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Ji, H.F.; Zhang, D.Y.; Wang, S.X.; Wang, J.; Shan, D.C.; Wang, Y.M. Effects of Lactobacillus brevis preparation on growth performance, fecal microflora and serum profile in weaned pigs. Livest. Sci. 2015, 178, 251–254. [Google Scholar] [CrossRef]
- Zhang, Q.; Fan, X.Y.; Cao, Y.J.; Zheng, T.T.; Cheng, W.J.; Chen, L.J.; Lv, X.C.; Ni, L.; Rao, P.F.; Liang, P. The beneficial effects of Lactobacillus brevis FZU0713-fermented Laminaria japonica on lipid metabolism and intestinal microbiota in hyperlipidemic rats fed with a high-fat diet. Food Funct. 2021, 12, 7145–7160. [Google Scholar] [CrossRef] [PubMed]
- Frampton, J.; Murphy, K.G.; Frost, G.; Chambers, E.S. Short-chain fatty acids as potential regulators of skeletal muscle metabolism and function. Nat. Metab. 2020, 2, 840–848. [Google Scholar] [CrossRef] [PubMed]
- den Besten, G.; Gerding, A.; van Dijk, T.H.; Ciapaite, J.; Bleeker, A.; van Eunen, K.; Havinga, R.; Groen, A.K.; Reijngoud, D.J.; Bakker, B.M. Protection against the metabolic syndrome by guar gum-derived short-chain fatty acids depends on peroxisome proliferator-activated receptor γ and glucagon-like peptide-1. PLoS ONE 2015, 10, e0136364. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.E.; Bhattacharya, A.; Sataranatarajan, K.; Qaisar, R.; Sloane, L.; Rahman, M.M.; Kinter, M.; Van Remmen, H. The histone deacetylase inhibitor butyrate improves metabolism and reduces muscle atrophy during aging. Aging Cell 2015, 14, 957–970. [Google Scholar] [CrossRef]
- Li, H.; Qiu, T.; Huang, G.; Cao, Y. Production of gamma-aminobutyric acid by Lactobacillus brevis NCL912 using fed-batch fermentation. Microb. Cell Fact. 2010, 9, 85. [Google Scholar] [CrossRef]
- Godfrey, R.J.; Madgwick, Z.; Whyte, G.P. The exercise-induced growth hormone response in athletes. Sports Med. 2003, 33, 599–613. [Google Scholar] [CrossRef]
- Sakashita, M.; Nakamura, U.; Horie, N.; Yokoyama, Y.; Kim, M.; Fujita, S. Oral supplementation using gamma-aminobutyric acid and whey protein improves whole body fat-free mass in men after resistance training. J. Clin. Med. Res. 2019, 11, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Arvanitoyannis, I.S. HACCP and ISO 22000: Application to Foods of Animal Origin; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Brzycki, M. Strength testing—Predicting a one-rep max from reps-to-fatigue. J. Phys. Educ. Recreat. Dance 1993, 64, 88–90. [Google Scholar] [CrossRef]
- Bruce, R.A.; Kusumi, F.; Hosmer, D. Maximal oxygen intake and nomographic assessment of functional aerobic impairment in cardiovascular disease. Am. Heart J. 1973, 85, 546–562. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Ho, C.S.; Hsu, Y.J.; Wu, M.F.; Huang, C.C. Effect of 8-week frequency-specific electrical muscle stimulation combined with resistance exercise training on muscle mass, strength, and body composition in men and women: A feasibility and safety study. PeerJ 2023, 11, e16303. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Ho, C.S.; Hsu, Y.J.; Huang, C.C. Live and Heat-Killed Probiotic Lactobacillus paracasei PS23 accelerated the improvement and recovery of strength and damage biomarkers after exercise-induced muscle damage. Nutrients 2022, 14, 4563. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Hsu, Y.J.; Ho, C.S.; Chang, C.H.; Liu, C.W.; Huang, C.C.; Chiang, W.D. Evaluation of the efficacy of supplementation with planox® lemon verbena extract in improving oxidative stress and muscle damage: A randomized double-blind controlled trial. Int. J. Med. Sci. 2021, 18, 2641–2652. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Lee, M.C.; Ho, C.S.; Hsu, Y.J.; Ho, C.C.; Kan, N.W. Protective and recovery effects of resveratrol supplementation on exercise performance and muscle damage following acute plyometric exercise. Nutrients 2021, 13, 3217. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Ikezoe, T.; Takeno, Y.; Ichihashi, N. Acute and prolonged effect of static stretching on the passive stiffness of the human gastrocnemius muscle tendon unit in vivo. J. Orthop. Res. 2011, 29, 1759–1763. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Qu, F.; Zhu, L.H. Isolation of genomic DNAs from plants, fungi and bacteria using benzyl chloride. Nucleic Acids Res. 1993, 21, 5279–5280. [Google Scholar] [CrossRef]
- Chew, W.; Lim, Y.P.; Lim, W.S.; Chambers, E.S.; Frost, G.; Wong, S.H.; Ali, Y. Gut-muscle crosstalk. A perspective on influence of microbes on muscle function. Front. Med. 2023, 9, 1065365. [Google Scholar] [CrossRef]
- Przewłócka, K.; Folwarski, M.; Kaźmierczak-Siedlecka, K.; Skonieczna-Żydecka, K.; Kaczor, J.J. Gut-muscle axisexists and may affect skeletal muscle adaptation to training. Nutrients 2020, 12, 1451. [Google Scholar] [CrossRef] [PubMed]
- Bycura, D.; Santos, A.C.; Shiffer, A.; Kyman, S.; Winfree, K.; Sutliffe, J.; Pearson, T.; Sonderegger, D.; Cope, E.; Caporaso, J.G. Impact of different exercise modalities on the human gut microbiome. Sports 2021, 9, 14. [Google Scholar] [CrossRef]
- Clarke, S.F.; Murphy, E.F.; O’Sullivan, O.; Lucey, A.J.; Humphreys, M.; Hogan, A.; Hayes, P.; O’Reilly, M.; Jeffery, I.B.; Wood-Martin, R.; et al. Exercise and associated dietary extremes impact on gut microbial diversity. Gut 2014, 63, 1913–1920. [Google Scholar] [CrossRef]
- Guan, L.; Cao, Z.; Pan, Z.; Zhao, C.; Xue, M.; Yang, F.; Chen, J. Butyrate promotes C2C12 myoblast proliferation by activating ERK/MAPK pathway. Mol. Omics 2023, 19, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Hsu, Y.J.; Ho, H.H.; Kuo, Y.W.; Lin, W.Y.; Tsai, S.Y.; Chen, W.L.; Lin, C.L.; Huang, C.C. Effectiveness of human-origin Lactobacillus plantarum PL-02 in improving muscle mass, exercise performance and anti-fatigue. Sci Rep. 2021, 11, 19469. [Google Scholar] [CrossRef]
- Chen, L.; Nelson, D.R.; Zhao, Y.; Cui, Z.; Johnston, J.A. Relationship between muscle mass and muscle strength, and the impact of comorbidities: A population-based, cross-sectional study of older adults in the United States. BMC Geriatr. 2013, 13, 74. [Google Scholar] [CrossRef] [PubMed]
- Maughan, R.J.; Watson, J.S.; Weir, J. Strength and cross-sectional area of human skeletal muscle. J. Physiol. 1983, 338, 37–49. [Google Scholar] [CrossRef]
- Ikai, M.; Fukunaga, T. A study on training effect on strength per unit cross-sectional area of muscle by means of ultrasonic measurement. Int. Z. Angew. Physiol. 1970, 28, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Wilke, J.; Macchi, V.; De Caro, R.; Stecco, C. Fascia thickness, aging and flexibility: Is there an association? J. Anat. 2019, 234, 43–49. [Google Scholar] [CrossRef]
- Jia, S.; Wu, Q.; Wang, S.; Kan, J.; Zhang, Z.; Zhang, X.; Zhang, X.; Li, J.; Xu, W.; Du, J.; et al. Pea Peptide Supplementation in Conjunction with Resistance Exercise Promotes Gains in Muscle Mass and Strength. Front. Nutr. 2022, 9, 878229. [Google Scholar] [CrossRef]
- Suchomel, T.J.; Nimphius, S.; Bellon, C.R.; Stone, M.H. The importance of muscular strength: Training considerations. Sports Med. 2018, 48, 765–785. [Google Scholar] [CrossRef] [PubMed]
- Marián, V.; Katarína, L.; Dávid, O.; Matúš, K.; Simon, W. Improved maximum strength, vertical jump and sprint performance after 8 weeks of jump squat training with individualized loads. J. Sports Sci. Med. 2016, 15, 492–500. [Google Scholar] [PubMed]
- Karim, A.; Muhammad, T.; Shahid Iqbal, M.; Qaisar, R. A multistrain probiotic improves handgrip strength and functional capacity in patients with COPD: A randomized controlled trial. Arch. Gerontol. Geriatr. 2022, 102, 104721. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.R.; Hoffman, M.W.; Zelicha, H.; Gepner, Y.; Willoughby, D.S.; Feinstein, U.; Ostfeld, I. the effect of 2 weeks of inactivated probiotic Bacillus coagulans on endocrine, inflammatory, and performance responses during self-defense training in soldiers. J. Strength Cond. Res. 2019, 33, 2330–2337. [Google Scholar] [CrossRef] [PubMed]
- Vaisberg, M.; Paixão, V.; Almeida, E.B.; Santos, J.M.B.; Foster, R.; Rossi, M.; Pithon-Curi, T.C.; Gorjão, R.; Momesso, C.M.; Andrade, M.S.; et al. Daily intake of fermented milk containing Lactobacillus casei shirota (lcs) modulates systemic and upper airways immune/inflammatory responses in marathon runners. Nutrients 2019, 11, 1678. [Google Scholar] [CrossRef] [PubMed]
- Salarkia, N.; Ghadamli, L.; Zaeri, F.; Sabaghian Rad, L. Effects of probiotic yogurt on performance, respiratory and digestive systems of young adult female endurance swimmers: A randomized controlled trial. Med. J. Islam. Repub. Iran 2013, 27, 141–146. [Google Scholar] [PubMed]
- Chen, Y.M.; Wei, L.; Chiu, Y.S.; Hsu, Y.J.; Tsai, T.Y.; Wang, M.F.; Huang, C.C. Lactobacillus plantarum TWK10 supplementation improves exercise performance and increases muscle mass in mice. Nutrients 2016, 8, 205. [Google Scholar] [CrossRef]
- Murray, B.; Rosenbloom, C. Fundamentals of glycogen metabolism for coaches and athletes. Nutr. Rev. 2018, 76, 243–259. [Google Scholar] [CrossRef]
- Zhang, W.Q.; Zhao, T.T.; Gui, D.K.; Gao, C.L.; Gu, J.L.; Gan, W.J.; Huang, W.; Xu, Y.; Zhou, H.; Chen, W.N.; et al. Sodium butyrate improves liver glycogen metabolism in type 2 diabetes mellitus. J. Agric. Food Chem. 2019, 67, 7694–7705. [Google Scholar] [CrossRef]
- Lawler, T.P.; Cialdella-Kam, L. Non-carbohydrate Dietary Factors and Their Influence on Post-Exercise Glycogen Storage: A Review. Curr. Nutr. Rep. 2020, 9, 394–404. [Google Scholar] [CrossRef]
- Ansdell, P.; Thomas, K.; Hicks, K.M.; Hunter, S.K.; Howatson, G.; Goodall, S. Physiological sex differences affect the integrative response to exercise: Acute and chronic implications. Exp. Physiol. 2020, 105, 2007–2021. [Google Scholar] [CrossRef] [PubMed]
- de Paiva, A.K.F.; de Oliveira, E.P.; Mancini, L.; Paoli, A.; Mota, J.F. Effects of probiotic supplementation on performance of resistance and aerobic exercises: A systematic review. Nutr. Rev. 2023, 81, 153–167. [Google Scholar] [CrossRef] [PubMed]
- Mohr, A.E.; Pugh, J.; O’Sullivan, O.; Black, K.; Townsend, J.R.; Pyne, D.B.; Wardenaar, F.C.; West, N.P.; Whisner, C.M.; McFarland, L.V. Best practices for probiotic research in athletic and physically active populations: Guidance for future randomized controlled trials. Front. Nutr. 2022, 9, 809983. [Google Scholar] [CrossRef] [PubMed]

| Dietary Intake | Before | After | ||
|---|---|---|---|---|
| Placebo | GKEX | Placebo | GKEX | |
| Carbohydrates (g/day) | 164 ± 38 | 164 ± 42 | 163 ± 44 | 165 ± 43 |
| Proteins (g/day) | 124 ± 547 | 126 ± 39 | 125 ± 55 | 125 ± 42 |
| Fats (g/day) | 70 ± 26 | 71 ± 19 | 70 ± 25 | 72 ± 19 |
| Total calories (kcal/day) | 1785 ± 521 | 1803 ± 376 | 1784 ± 522 | 1805 ± 378 |
| Body Composition | Week | Placebo | GKEX | Delta | |
|---|---|---|---|---|---|
| Placebo | GKEX | ||||
| BW (kg) | Before | 61.9 ± 9.2 a | 65.3 ± 12.6 a | 0.0 ± 0.0 a | 0.0 ± 0.0 a |
| Week 2 | 62.2 ± 9.4 a,* | 65.0 ± 12.4 a | 0.4 ± 0.9 b | −0.3 ± 0.8 a | |
| Week 4 | 62.4 ± 9.5 a,* | 64.9 ± 12.4 a | 0.5 ± 0.8 b | −0.3 ± 1.1 a | |
| After | 62.2 ± 9.5 a | 64.7 ± 12.3 a | 0.3 ± 0.9 b | −0.5 ± 1.4 a | |
| BMI (kg/m2) | Before | 22.4 ± 2.7 a | 22.8 ± 2.6 a | 0.0 ± 0.0 a | 0.0 ± 0.0 a |
| Week 2 | 22.5 ± 2.7 a | 22.7 ± 2.5 a | 0.1 ± 0.3 b | −0.1 ± 0.3 a | |
| Week 4 | 22.5 ± 2.7 a,* | 22.6 ± 2.5 a | 0.2 ± 0.3 b | −0.1 ± 0.4 a | |
| After | 22.5 ± 2.7 a | 22.6 ± 2.5 a | 0.1 ± 0.3 b | −0.2 ± 0.4 a | |
| Muscle Weight (kg) | Before | 27.0 ± 4.9 a | 28.6 ± 6.9 a | 0.0 ± 0.0 a | 0.0 ± 0.0 a |
| Week 2 | 26.9 ± 4.9 a | 28.8 ± 6.8 a,* | −0.1 ± 0.4 a | 0.3 ± 0.3 b | |
| Week 4 | 27.0 ± 5.0 a | 29.1 ± 6.8 a,* | 0.0 ± 0.3 a | 0.5 ± 0.4 b | |
| After | 27.0 ± 5.0 a | 29.3 ± 6.9 a,* | 0.0 ± 0.5 a | 0.8 ± 0.5 b | |
| Fat (%) | Before | 21.2 ± 6.9 a | 22.4 ± 5.4 a | 0.0 ± 0.0 a | 0.0 ± 0.0 a |
| Week 2 | 21.4 ± 6.7 a,* | 22.1 ± 5.5 a,* | 0.2 ± 0.7 b | −0.4 ± 0.7 a | |
| Week 4 | 21.4 ± 6.9 a,* | 21.4 ± 5.4 a,* | 0.2 ± 0.8 b | −1.1 ± 1.2 a | |
| After | 21.1 ± 6.9 a | 21.3 ± 5.3 a,* | −0.1 ± 1.0 b | −1.2 ± 1.3 a | |
| Body Composition | Before | After | Delta | |||
|---|---|---|---|---|---|---|
| Placebo | GKEX | Placebo | GKEX | Placebo | GKEX | |
| LBM (kg) | 46.1 ± 7.6 a | 47.7 ± 10.2 a | 46.4 ± 7.8 a | 48.3 ± 10.1 a,* | 0.3 ± 0.9 a | 0.6 ± 1.2 a |
| FBM (kg) | 21.1 ± 6.9 a | 22.4 ± 5.6 a | 21.1 ± 6.9 a | 21.3 ± 5.3 a,* | −0.2 ± 5.5 b | −1.1 ± 1.3 a |
| Mineral Weight (kg) | 2.5 ± 0.4 a | 2.7 ± 0.5 a | 2.5 ± 0.4 a | 2.7 ± 0.5 a | 0.0 ± 0.0 a | 0.0 ± 0.0 a |
| Muscle Ultrasound | Before | After | Delta | |||
|---|---|---|---|---|---|---|
| Placebo | GKEX | Placebo | GKEX | Placebo | GKEX | |
| Muscle thickness (cm) | ||||||
| Right biceps | 2.19 ± 0.40 a | 2.12 ± 0.41a | 2.21 ± 0.43 a,* | 2.29 ± 0.42 a,* | 0.02 ± 0.05 a | 0.16 ± 0.07 b |
| Right quadriceps | 2.35 ± 0.29 a | 2.44 ± 0.30 a | 2.35 ± 0.28 a | 2.57 ± 0.32 a,* | 0.00 ± 0.07 a | 0.12 ± 0.07 b |
| Left biceps | 2.13 ± 0.38 a | 2.13 ± 0.42 a | 1.95 ± 0.37 a | 2.29 ± 0.51a,* | 0.00 ± 0.08 a | 0.16 ± 0.14 b |
| Left quadriceps | 2.31 ± 0.26 a | 2.44 ± 0.36 a | 2.31 ± 0.26 a | 2.52 ± 0.37 b,* | 0.00 ± 0.06 a | 0.08 ± 0.12 b |
| Fascia (cm) | ||||||
| Right biceps | 0.13 ± 0.07 a | 0.11 ± 0.09 a | 0.12 ± 0.07 a | 0.13 ± 0.09 a,* | 0.00 ± 0.02 a | 0.02 ± 0.02 b |
| Right quadriceps | 0.15 ± 0.06 a | 0.16 ± 0.06 a | 0.16 ± 0.06 a | 0.18 ± 0.07 a,* | 0.01 ± 0.02 a | 0.02 ± 0.04 a |
| Left biceps | 0.13 ± 0.06 a | 0.15 ± 0.08 a | 0.14 ± 0.06 a,* | 0.17 ± 0.08 b,* | 0.01 ± 0.02 a | 0.01 ± 0.02 a |
| Left quadriceps | 0.16 ± 0.06 a | 0.19 ± 0.07 b | 0.16 ± 0.06 a | 0.20 ± 0.06 b | 0.01 ± 0.03 a | 0.01 ± 0.03 a |
| Exercise Performance | Before | After | ||
|---|---|---|---|---|
| Placebo | GKEX | Placebo | GKEX | |
| CMJ | ||||
| Relative peak force (N/kg) | 14.2 ± 2.1 a | 14.3 ± 1.9 a | 14.2 ± 2.2 a | 14.6 ± 1.9 a,* |
| RFD (N/kg/s) | 7.5 ± 1.7 a | 7.4 ± 1.6 a | 7.5 ± 1.7 a | 7.9 ± 1.6 a,* |
| Jump height (cm) | 28.4 ± 6.0 a | 29.7 ± 5.4 a | 29.1 ± 6.4 a | 30.3 ± 5.9 a |
| IMTP | ||||
| Relative peak force (N/kg) | 13.5 ± 2.7 a | 13.2 ± 3.0 a | 13.6 ± 2.7 a | 13.9 ± 3.2 a,* |
| RFD (N/s) | 8039 ± 940 a | 8057 ± 855 a | 8120 ± 976 a | 8261 ± 892 a,* |
| Wingate | ||||
| Relative mean power (W/kg) | 5.8 ± 1.0 a | 5.9 ± 1.0 a | 5.8 ± 1.0 a | 6.1 ± 1.0 a,* |
| Relative peak power (W/kg) | 8.5 ± 1.9 a | 8.9 ± 1.7 a | 8.6 ± 1.8 a | 9.0 ± 1.6 a |
| Fatigue index (%) | 48.1 ± 7.5 a | 46.1 ± 11.1 a | 47.9 ± 6.9 a | 45.2 ± 11.0 a |
| Characteristics | Before | After | ||
|---|---|---|---|---|
| Placebo | GKEX | Placebo | GKEX | |
| Blood Biochemical Parameters | ||||
| AST (U/L) | 18 ± 4 | 18 ± 4 | 18 ± 3 | 18 ± 4 |
| ALT (U/L) | 17 ± 3 | 16 ± 4 | 16 ± 2 | 15 ± 4 |
| TC (mg/dL) | 161 ± 13 | 162 ± 12 | 160 ± 13 | 161 ± 13 |
| TG (mg/dL) | 89 ± 17 | 88 ± 22 | 88 ± 18 | 87 ± 17 |
| HDL (mg/dL) | 57.4 ± 6.9 | 56.6 ± 8.1 | 57.7 ± 7.2 | 56.9 ± 7.5 |
| LDL (mg/dL) | 85.2 ± 14.4 | 87.8 ± 8.7 | 85.0 ± 14.3 | 86.0 ± 10.6 |
| BUN (mg/dL) | 15.2 ± 2.0 | 15.5 ± 2.4 | 15.0 ± 2.1 | 15.0 ± 2.9 |
| CREA (mg/dL) | 0.98 ± 0.12 | 0.97 ± 0.12 | 0.98 ± 0.12 | 0.97 ± 0.10 |
| UA (mg/dL) | 5.2 ± 0.7 | 5.0 ± 0.6 | 5.3 ± 0.7 | 5.0 ± 0.6 |
| TP (g/dL) | 7.2 ± 0.3 | 7.3 ± 0.3 | 7.2 ± 0.4 | 7.3 ± 0.3 |
| Glucose (mg/dL) | 85 ± 7 | 86 ± 5 | 84 ± 7 | 85 ± 5 |
| Insulin (μU/mL) | 10.93 ± 2.24 | 11.00 ± 3.1 | 10.81 ± 2.46 | 10.93 ± 3.10 |
| Lactate (mmol/L) | 1.57 ± 0.27 | 1.64 ± 0.19 | 1.56 ± 0.29 | 1.63 ± 0.20 |
| NH3 (μmol/L) | 56.8 ± 9.9 | 53.3 ± 9.3 | 56.7 ± 10.3 | 53.1 ± 9.6 |
| Complete Blood Count | ||||
| WBC (103 cumm) | 6.0 ± 0.9 | 6.5 ± 1.1 | 6.2 ± 0.9 | 6.5 ± 1.5 |
| Neutrophils (%) | 53.4 ± 7.2 | 55 ± 6.4 | 55.4 ± 6.9 | 53 ± 6.6 |
| Lymphocytes (%) | 36.2 ± 6.6 | 33.8 ± 6.4 | 34.0 ± 5.9 | 36.4 ± 6.0 |
| Monocytes (%) | 7.5 ± 1.5 | 7.8 ± 1.7 | 7.9 ± 1.7 | 7.3 ± 1.8 |
| Eosinophils (%) | 2.3 ± 1.3 | 2.7 ± 1.5 | 2.0 ± 1.2 | 2.5 ± 1.2 |
| Basophils (%) | 0.6 ± 0.2 | 0.6 ± 0.3 | 0.6 ± 0.3 | 0.7 ± 0.3 |
| RBC (MIL/cumm) | 4.8 ± 0.4 | 4.7 ± 0.4 | 4.8 ± 0.4 | 15.0 ± 2.9 |
| Hemoglobin (gm/dL) | 14.1 ± 1.2 | 14.0 ± 1.2 | 14.1 ± 1.3 | 13.9 ± 1.1 |
| Hematocrit (%) | 42.1 ± 2.9 | 41.9 ± 3.7 | 41.9 ± 3.8 | 42.2 ± 3.8 |
| M.C.V (fl) | 88.0 ± 2.7 | 89.1 ± 2.5 | 88.1 ± 2.5 | 89.1 ± 2.6 |
| M.C.H (pg) | 29.7 ± 1.1 | 29.9 ± 0.9 | 29.5 ± 1.1 | 29.8 ± 1.0 |
| M.C.H.C (%) | 33.7 ± 0.6 | 33.4 ± 0.7 | 33.6 ± 0.7 | 33.4 ± 0.6 |
| Platelet count (103/Cumm) | 273 ± 51 | 278 ± 51 | 262 ± 62 | 273 ± 55 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, M.-C.; Hsu, Y.-J.; Ho, C.-S.; Tsai, Y.-S.; Chen, C.-C.; Huang, C.-C. Supplementation with Lactiplantibacillus brevis GKEX Combined with Resistance Exercise Training Improves Muscle Mass, Strength Performance, and Body Fat Condition in Healthy Humans. Foods 2024, 13, 1030. https://doi.org/10.3390/foods13071030
Lee M-C, Hsu Y-J, Ho C-S, Tsai Y-S, Chen C-C, Huang C-C. Supplementation with Lactiplantibacillus brevis GKEX Combined with Resistance Exercise Training Improves Muscle Mass, Strength Performance, and Body Fat Condition in Healthy Humans. Foods. 2024; 13(7):1030. https://doi.org/10.3390/foods13071030
Chicago/Turabian StyleLee, Mon-Chien, Yi-Ju Hsu, Chin-Shan Ho, You-Shan Tsai, Chin-Chu Chen, and Chi-Chang Huang. 2024. "Supplementation with Lactiplantibacillus brevis GKEX Combined with Resistance Exercise Training Improves Muscle Mass, Strength Performance, and Body Fat Condition in Healthy Humans" Foods 13, no. 7: 1030. https://doi.org/10.3390/foods13071030
APA StyleLee, M.-C., Hsu, Y.-J., Ho, C.-S., Tsai, Y.-S., Chen, C.-C., & Huang, C.-C. (2024). Supplementation with Lactiplantibacillus brevis GKEX Combined with Resistance Exercise Training Improves Muscle Mass, Strength Performance, and Body Fat Condition in Healthy Humans. Foods, 13(7), 1030. https://doi.org/10.3390/foods13071030








