The Role of Methionine Restriction in Gastric Cancer: A Summary of Mechanisms and a Discussion on Tumor Heterogeneity
Abstract
1. Introduction
2. The Relationship between Met and the Methionine Cycle
3. Mechanisms of Met in the Pathophysiology of Gastric Cancer
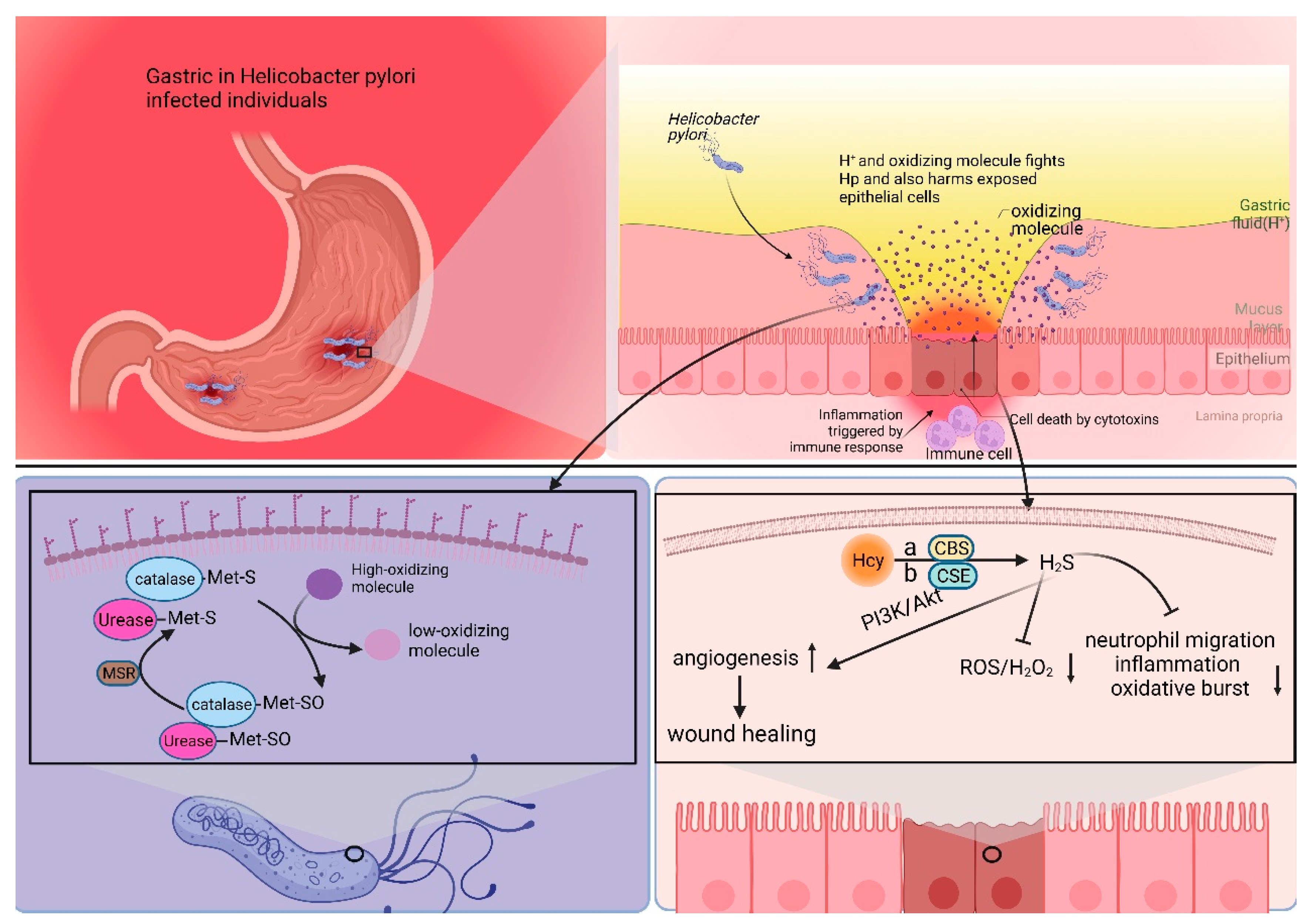
3.1. Mechanisms of Met’s Role in Helicobacter pylori Survival and Infection
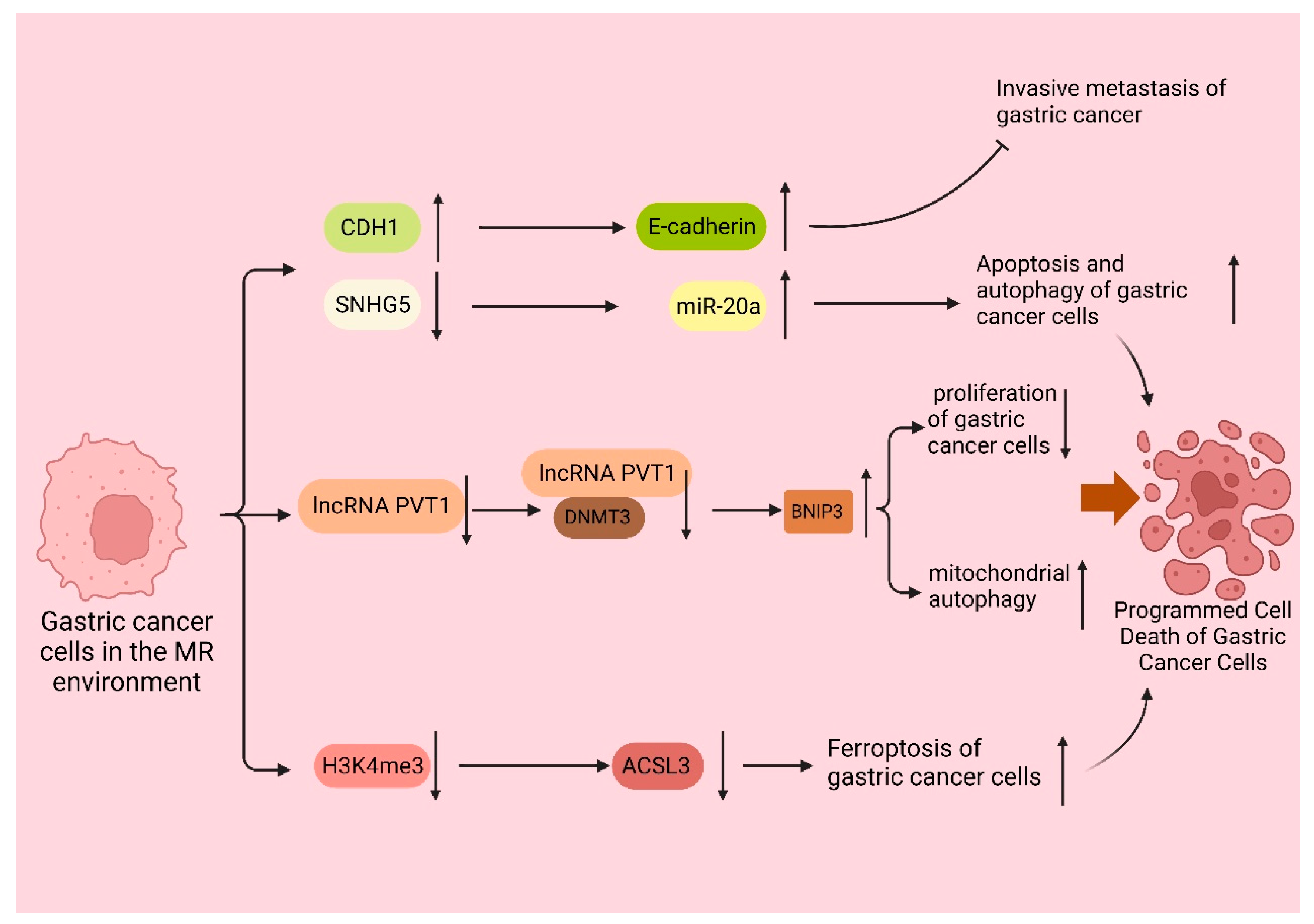
3.2. The Mechanism of Met in Programmed Cell Death of Gastric Cancer Cells
3.3. The Mechanism of Met’s Role in the Immune Response to Gastric Cancer
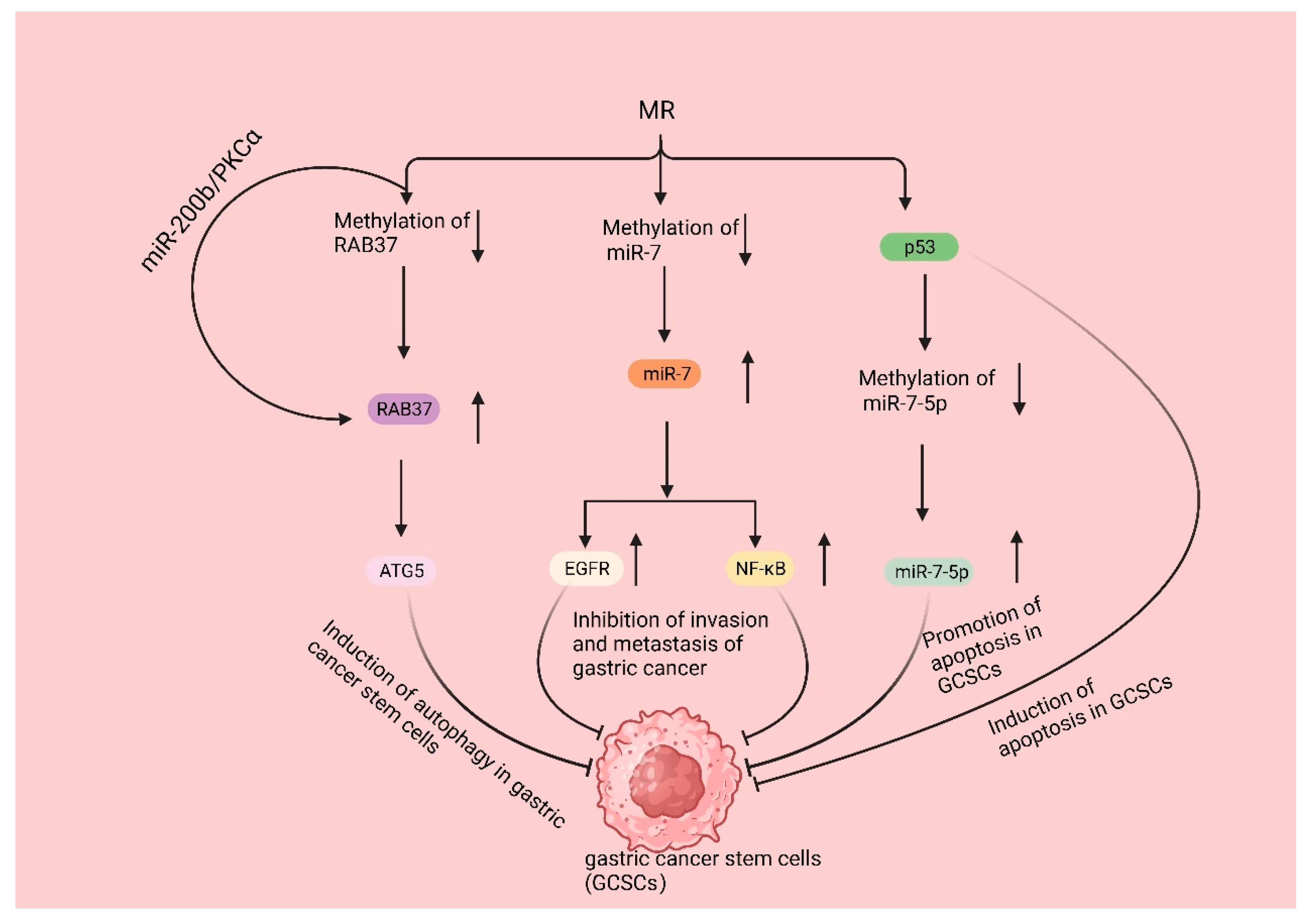
3.4. Mechanisms Underlying the Impact of Met on Gastric Cancer Stem Cells
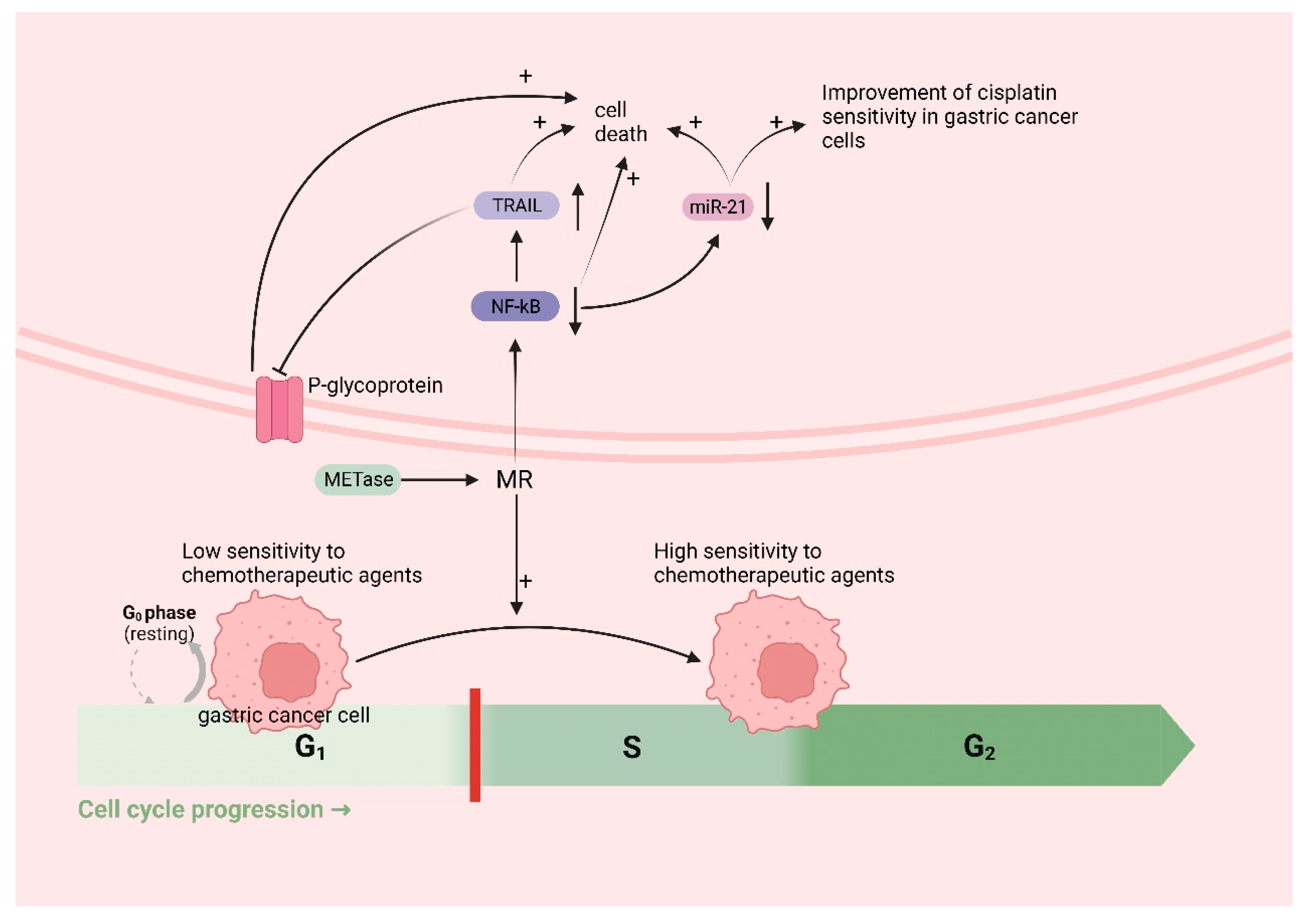
3.5. The Role and Mechanism of Met in Gastric Cancer Chemotherapy
4. Conclusions and Prospects
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| MR | methionine restriction |
| EOGC | early-onset gastric cancer |
| EBV | Epstein–Barr virus, |
| MSI | microsatellite instability |
| CIN | chromosomal instability |
| Met | methionine |
| SAM | S-adenosyl methionine |
| MAT | methionine adenosyltransferase |
| SAH | S-adenosyl homocysteine |
| Hcy | homocysteine |
| MTases | methyltransferases |
| DNMTs | DNA methyltransferases |
| HMTs | histone methyltransferases |
| GSH | glutathione |
| ROS | reactive oxygen species |
| GSSG | oxidized glutathione |
| Hp | Helicobacter pylori |
| IARC | International Agency for Research on Cancer |
| NO | nitric oxide |
| Msr | methionine sulfoxide reductase |
| H2S | hydrogen sulfide |
| CBS | cystathionine β-synthase |
| CSE | cystathionine γ-lyase |
| 3MST | 3-mercaptopyruvate sulfotransferase |
| METase | methioninase |
| p-Akt | phosphorylated Akt |
| t-Akt | total Akt |
| rMetase | recombinant methioninase |
| lncRNA | long non-coding RNA |
| MAT2A | Met adenylyltransferase 2A |
| H3K4me3 | trimethylation of histone H3 at lysine 4 |
| IL-17 | interleukin-17 |
| IFN-γ | interferon-gamma |
| GCSCs | gastric cancer stem cells |
| ATG5 | autophagy-related gene 5 |
| EGFR | epidermal growth factor receptor |
| NF-Κb | nuclear factor-κB |
| TRAIL | Tumor-necrosis-factor-associated apoptosis-inducing ligand |
References
- Wang, F.H.; Zhang, X.T.; Li, Y.F.; Tang, L.; Qu, X.J.; Ying, J.E.; Zhang, J.; Sun, L.Y.; Lin, R.B.; Qiu, H.; et al. The Chinese Society of Clinical Oncology (CSCO): Clinical guidelines for the diagnosis and treatment of gastric cancer, 2021. Cancer Commun. 2021, 41, 747–795. [Google Scholar] [CrossRef]
- Cao, W.; Chen, H.D.; Yu, Y.W.; Li, N.; Chen, W.Q. Changing profiles of cancer burden worldwide and in China: A secondary analysis of the global cancer statistics 2020. Chin. Med. J. 2021, 134, 783–791. [Google Scholar] [CrossRef]
- Thrift, A.P.; Wenker, T.N.; El-Serag, H.B. Global burden of gastric cancer: Epidemiological trends, risk factors, screening and prevention. Nat. Rev. Clin. Oncol. 2023, 20, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Iarc, L. Schistosomes, liver flukes and Helicobacter pylori. IARC Monogr. Eval. Carcinog. Risks Hum. 1994, 61, 1–241. [Google Scholar]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. IARC monographs on the evaluation of carcinogenic risks to humans. Ingested nitrate and nitrite, and cyanobacterial peptide toxins. IARC Monogr. Eval. Carcinog. Risks Hum. 2010, 94, 1–412. [Google Scholar]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Tobacco smoke and involuntary smoking. IARC Monogr. Eval. Carcinog. Risks Hum. 2004, 83, 1–1438. [Google Scholar]
- Karimi, P.; Islami, F.; Anandasabapathy, S.; Freedman, N.D.; Kamangar, F. Gastric cancer: Descriptive epidemiology, risk factors, screening, and prevention. Cancer Epidemiol. Biomarkers Prev. 2014, 23, 700–713. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, L.; Wang, X.; Wang, J.; Yan, Z.; Cheng, J.; Gong, G.; Li, G. Body mass index and risk of gastric cancer: A meta-analysis of a population with more than ten million from 24 prospective studies. Cancer Epidemiol. Biomarkers Prev. 2013, 22, 1395–1408. [Google Scholar] [CrossRef]
- González, C.A.; Agudo, A. Carcinogenesis, prevention and early detection of gastric cancer: Where we are and where we should go. Int. J. Cancer 2012, 130, 745–753. [Google Scholar] [CrossRef]
- Boeing, H. Epidemiological research in stomach cancer: Progress over the last ten years. J. Cancer Res. Clin. Oncol. 1991, 117, 133–143. [Google Scholar] [CrossRef]
- Machlowska, J.; Baj, J.; Sitarz, M.; Maciejewski, R.; Sitarz, R. Gastric Cancer: Epidemiology, Risk Factors, Classification, Genomic Characteristics and Treatment Strategies. Int. J. Mol. Sci. 2020, 21, 4012. [Google Scholar] [CrossRef]
- Lin, Y.; Lin, S.; Chen, J.; Chen, L.; Tao, T.; Wu, S. Time Trends and Age-Period-Cohort Effects on the Incidence of Gastric Cancer in Changle from 2003 to 2012. Cancer Manag. Res. 2019, 11, 8885–8892. [Google Scholar] [CrossRef]
- Wiseman, M. The second World Cancer Research Fund/American Institute for Cancer Research expert report. Food, nutrition, physical activity, and the prevention of cancer: A global perspective. Proc. Nutr. Soc. 2008, 67, 253–256. [Google Scholar] [CrossRef]
- Davidson, M.; Cafferkey, C.; Goode, E.F.; Kouvelakis, K.; Hughes, D.; Reguera, P.; Kalaitzaki, E.; Peckitt, C.; Rao, S.; Watkins, D.; et al. Survival in Advanced Esophagogastric Adenocarcinoma Improves with Use of Multiple Lines of Therapy: Results from an Analysis of More than 500 Patients. Clin. Colorectal Cancer 2018, 17, 223–230. [Google Scholar] [CrossRef]
- Li, Y.; Lu, H.L.; Cao, T.H.; Duan, Z.P.; Huang, W.X. Research on risk factors of gastric carcinoma in the young. Guide China Med. 2016, 14, 14–15. [Google Scholar]
- Jin, P.; Tian, Y.T. Lifestyle Change in Terms of High Risk Factors for Gastric Cancer. Med. Recapitul. 2020, 26, 3329–3332+3337. Available online: https://link.cnki.net/urlid/11.3553.R.20200831.1012.002 (accessed on 23 December 2023).
- Wu, S.L.; Zhang, Y.; Fu, Y.; Li, J.; Wang, J.S. Gastric cancer incidence, mortality and burden in adolescents and young adults: A time-trend analysis and comparison among China, South Korea, Japan and the USA. BMJ Open 2022, 12, e061038. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Kawazoe, A.; Lordick, F.; Janjigian, Y.Y.; Shitara, K. Biomarker-targeted therapies for advanced-stage gastric and gastro-oesophageal junction cancers: An emerging paradigm. Nat. Rev. Clin. Oncol. 2021, 18, 473–487. [Google Scholar] [CrossRef]
- Alsina, M.; Arrazubi, V.; Diez, M.; Tabernero, J. Current developments in gastric cancer: From molecular profiling to treatment strategy. Nat. Rev. Gastroenterol. Hepatol. 2023, 20, 155–170. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Lauren, P. The Two Histological Main Types of Gastric Carcinoma: Diffuse and So-Called Intestinal-Type Carcinoma. An Attempt at a Histo-Clinical Classification. Acta Pathol. Microbiol. Scand. 1965, 64, 31–49. [Google Scholar] [CrossRef]
- The Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 2014, 513, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Seevaratnam, R.; Cardoso, R.; McGregor, C.; Lourenco, L.; Mahar, A.; Sutradhar, R.; Law, C.; Paszat, L.; Coburn, N. How useful is preoperative imaging for tumor, node, metastasis (TNM) staging of gastric cancer? A meta-analysis. Gastric Cancer 2012, 15 (Suppl. S1), S3–S18. [Google Scholar] [CrossRef] [PubMed]
- Nagtegaal, I.D.; Odze, R.D.; Klimstra, D.; Paradis, V.; Rugge, M.; Schirmacher, P.; Washington, K.M.; Carneiro, F.; Cree, I.A. The 2019 WHO classification of tumours of the digestive system. Histopathology 2020, 76, 182–188. [Google Scholar] [CrossRef]
- Benson, A.B.; Venook, A.P.; Al-Hawary, M.M.; Arain, M.A.; Chen, Y.J.; Ciombor, K.K.; Cohen, S.A.; Cooper, H.S.; Deming, D.A.; Garrido-Laguna, I.; et al. Small Bowel Adenocarcinoma, Version 1.2020, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc. Netw. 2019, 17, 1109–1133. [Google Scholar] [CrossRef] [PubMed]
- Takei, S.; Kawazoe, A.; Shitara, K. The New Era of Immunotherapy in Gastric Cancer. Cancers 2022, 14, 1054. [Google Scholar] [CrossRef]
- Hoffman, R.M. Clinical Studies of Methionine-Restricted Diets for Cancer Patients. Methods Mol. Biol. 2019, 1866, 95–105. [Google Scholar] [CrossRef]
- Minalyan, A.; Benhammou, J.N.; Artashesyan, A.; Lewis, M.S.; Pisegna, J.R. Autoimmune atrophic gastritis: Current perspectives. Clin. Exp. Gastroenterol. 2017, 10, 19–27. [Google Scholar] [CrossRef]
- Catenacci, D.V.T.; Tebbutt, N.C.; Davidenko, I.; Murad, A.M.; Al-Batran, S.E.; Ilson, D.H.; Tjulandin, S.; Gotovkin, E.; Karaszewska, B.; Bondarenko, I.; et al. Rilotumumab plus epirubicin, cisplatin, and capecitabine as first-line therapy in advanced MET-positive gastric or gastro-oesophageal junction cancer (RILOMET-1): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2017, 18, 1467–1482. [Google Scholar] [CrossRef] [PubMed]
- Dutton, S.J.; Ferry, D.R.; Blazeby, J.M.; Abbas, H.; Dahle-Smith, A.; Mansoor, W.; Thompson, J.; Harrison, M.; Chatterjee, A.; Falk, S.; et al. Gefitinib for oesophageal cancer progressing after chemotherapy (COG): A phase 3, multicentre, double-blind, placebo-controlled randomised trial. Lancet Oncol. 2014, 15, 894–904. [Google Scholar] [CrossRef] [PubMed]
- Lordick, F.; Kang, Y.K.; Chung, H.C.; Salman, P.; Oh, S.C.; Bodoky, G.; Kurteva, G.; Volovat, C.; Moiseyenko, V.M.; Gorbunova, V.; et al. Capecitabine and cisplatin with or without cetuximab for patients with previously untreated advanced gastric cancer (EXPAND): A randomised, open-label phase 3 trial. Lancet Oncol. 2013, 14, 490–499. [Google Scholar] [CrossRef]
- Waddell, T.; Chau, I.; Cunningham, D.; Gonzalez, D.; Okines, A.F.; Okines, C.; Wotherspoon, A.; Saffery, C.; Middleton, G.; Wadsley, J.; et al. Epirubicin, oxaliplatin, and capecitabine with or without panitumumab for patients with previously untreated advanced oesophagogastric cancer (REAL3): A randomised, open-label phase 3 trial. Lancet Oncol. 2013, 14, 481–489. [Google Scholar] [CrossRef]
- Thuss-Patience, P.C.; Shah, M.A.; Ohtsu, A.; Van Cutsem, E.; Ajani, J.A.; Castro, H.; Mansoor, W.; Chung, H.C.; Bodoky, G.; Shitara, K.; et al. Trastuzumab emtansine versus taxane use for previously treated HER2-positive locally advanced or metastatic gastric or gastro-oesophageal junction adenocarcinoma (GATSBY): An international randomised, open-label, adaptive, phase 2/3 study. Lancet Oncol. 2017, 18, 640–653. [Google Scholar] [CrossRef] [PubMed]
- Hecht, J.R.; Bang, Y.J.; Qin, S.K.; Chung, H.C.; Xu, J.M.; Park, J.O.; Jeziorski, K.; Shparyk, Y.; Hoff, P.M.; Sobrero, A.; et al. Lapatinib in Combination with Capecitabine Plus Oxaliplatin in Human Epidermal Growth Factor Receptor 2-Positive Advanced or Metastatic Gastric, Esophageal, or Gastroesophageal Adenocarcinoma: TRIO-013/LOGiC—A Randomized Phase III Trial. J. Clin. Oncol. 2016, 34, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Satoh, T.; Xu, R.H.; Chung, H.C.; Sun, G.P.; Doi, T.; Xu, J.M.; Tsuji, A.; Omuro, Y.; Li, J.; Wang, J.W.; et al. Lapatinib plus paclitaxel versus paclitaxel alone in the second-line treatment of HER2-amplified advanced gastric cancer in Asian populations: TyTAN—A randomized, phase III study. J. Clin. Oncol. 2014, 32, 2039–2049. [Google Scholar] [CrossRef] [PubMed]
- Shitara, K.; Doi, T.; Hosaka, H.; Thuss-Patience, P.; Santoro, A.; Longo, F.; Ozyilkan, O.; Cicin, I.; Park, D.; Zaanan, A.; et al. Efficacy and safety of trifluridine/tipiracil in older and younger patients with metastatic gastric or gastroesophageal junction cancer: Subgroup analysis of a randomized phase 3 study (TAGS). Gastric Cancer 2022, 25, 586–597. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Shang, D.; Cheng, H.; Zhou, R. The small GTPase RAB37 functions as an organizer for autophagosome biogenesis. Autophagy 2018, 14, 727–729. [Google Scholar] [CrossRef]
- Li, Y.F.; Zhang, H.T.; Xin, L. Hyaluronic acid-modified polyamidoamine dendrimer G5-entrapped gold nanoparticles delivering METase gene inhibits gastric tumor growth via targeting CD44+ gastric cancer cells. J. Cancer Res. Clin. Oncol. 2018, 144, 1463–1473. [Google Scholar] [CrossRef] [PubMed]
- Bauerle, M.R.; Schwalm, E.L.; Booker, S.J. Mechanistic diversity of radical S-adenosylmethionine (SAM)-dependent methylation. J. Biol. Chem. 2015, 290, 3995–4002. [Google Scholar] [CrossRef] [PubMed]
- Xin, L.; Li, S.H.; Liu, C.; Zeng, F.; Cao, J.Q.; Zhou, L.Q.; Zhou, Q.; Yuan, Y.W. Methionine represses the autophagy of gastric cancer stem cells via promoting the methylation and phosphorylation of RAB37. Cell Cycle 2020, 19, 2644–2652. [Google Scholar] [CrossRef]
- Alom, S. DNA methylation of angiogenic gene in the gastric cancer and fate of S-adenosyl-l-methionine, the donor of the methyl group. Gene 2023, 848, 146903. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, J.S.; Guo, M.Z.; Feng, B.S.; Zhang, J.P. Inhibitory effect of S-adenosylmethionine on the growth of human gastric cancer cells in vivo and in vitro. Chin. J. Cancer 2010, 29, 752–760. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Niu, F.; Wu, J.; Chen, F.; Yang, H.; Li, J.; Jin, T.; Wu, Y. Association study between genetic polymorphisms in folate metabolism and gastric cancer susceptibility in Chinese Han population: A case-control study. Mol. Genet. Genom. Med. 2019, 7, e633. [Google Scholar] [CrossRef]
- Ma, M.; Kong, P.; Huang, Y.; Wang, J.; Liu, X.; Hu, Y.; Chen, X.; Du, C.; Yang, H. Activation of MAT2A-ACSL3 pathway protects cells from ferroptosis in gastric cancer. Free Radic. Biol. Med. 2022, 181, 288–299. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Li, S.; Liu, L.; Zhou, Q.; Yuan, Y.; Xin, L. Recombinant methioninase regulates PI3K/Akt/Glut-1 pathway and inhibits aerobic glycolysis to promote apoptosis of gastric cancer cells. Nan Fang Yi Ke Da Xue Xue Bao 2020, 40, 27–33. [Google Scholar] [CrossRef]
- Xin, L.; Zhou, L.Q.; Liu, L.; Yuan, Y.W.; Zhang, H.T.; Zeng, F. METase promotes cell autophagy via promoting SNHG5 and suppressing miR-20a in gastric cancer. Int. J. Biol. Macromol. 2019, 122, 1046–1052. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Liu, Z.; Wei, G.; Yang, P.; Hu, X.E.; Qu, F.; Lu, J.; He, X.; Bao, G. Genetic polymorphisms in folate-metabolizing genes associated with gastric cancer prognosis in northwest China subjects. J. Cancer 2020, 11, 6413–6420. [Google Scholar] [CrossRef]
- Feng, M.; Namanja-Magliano, H.; Rajagopalan, S.; Mishra, T.; Ducati, R.G.; Hirsch, B.M.; Kelly, L.; Szymczak, W.; Fajardo, J.E.; Sidoli, S.; et al. MAT Gain of Activity Mutation in Helicobacter pylori Is Associated with Resistance to MTAN Transition State Analogues. ACS Infect. Dis. 2023, 9, 966–978. [Google Scholar] [CrossRef]
- Schmalstig, A.A.; Benoit, S.L.; Misra, S.K.; Sharp, J.S.; Maier, R.J. Noncatalytic Antioxidant Role for Helicobacter pylori Urease. J. Bacteriol. 2018, 200, 10–128. [Google Scholar] [CrossRef]
- Nardone, G.; Compare, D. Epigenetic alterations due to diet and Helicobacter pylori infection in gastric carcinogenesis. Expert. Rev. Gastroenterol. Hepatol. 2008, 2, 243–248. [Google Scholar] [CrossRef]
- Zhao, T.; Xu, Z.; Gu, D.; Wu, P.; Huo, X.; Wei, X.; Tang, Y.; Gong, W.; He, M.L.; Chen, J. The effects of genomic polymorphisms in one-carbon metabolism pathways on survival of gastric cancer patients received fluorouracil-based adjuvant therapy. Sci. Rep. 2016, 6, 28019. [Google Scholar] [CrossRef]
- Xin, L.; Yang, W.F.; Zhang, H.T.; Li, Y.F.; Liu, C. The mechanism study of lentiviral vector carrying methioninase enhances the sensitivity of drug-resistant gastric cancer cells to Cisplatin. Br. J. Cancer 2018, 118, 1189–1199. [Google Scholar] [CrossRef]
- Miyake, M.; Miyake, K.; Han, Q.; Igarashi, K.; Kawaguchi, K.; Barangi, M.; Kiyuna, T.; Sugisawa, N.; Higuchi, T.; Oshiro, H.; et al. Synergy of oral recombinant methioninase (rMETase) and 5-fluorouracil on poorly differentiated gastric cancer. Biochem. Biophys. Res. Commun. 2023, 643, 48–54. [Google Scholar] [CrossRef]
- Hoffman, D.R.; Cornatzer, W.E.; Duerre, J.A. Relationship between tissue levels of S-adenosylmethionine, S-adenylhomocysteine, and transmethylation reactions. Can. J. Biochem. 1979, 57, 56–65. [Google Scholar] [CrossRef]
- Sanderson, S.M.; Gao, X.; Dai, Z.; Locasale, J.W. Methionine metabolism in health and cancer: A nexus of diet and precision medicine. Nat. Rev. Cancer 2019, 19, 625–637. [Google Scholar] [CrossRef]
- Miller, A.L. The methionine-homocysteine cycle and its effects on cognitive diseases. Altern. Med. Rev. 2003, 8, 7–19. [Google Scholar]
- Chen, D.H.; Wu, K.T.; Hung, C.J.; Hsieh, M.; Li, C. Effects of adenosine dialdehyde treatment on in vitro and in vivo stable protein methylation in HeLa cells. J. Biochem. 2004, 136, 371–376. [Google Scholar] [CrossRef]
- Koundouros, N.; Poulogiannis, G. Phosphoinositide 3-Kinase/Akt Signaling and Redox Metabolism in Cancer. Front. Oncol. 2018, 8, 160. [Google Scholar] [CrossRef] [PubMed]
- Chai, E.Z.; Siveen, K.S.; Shanmugam, M.K.; Arfuso, F.; Sethi, G. Analysis of the intricate relationship between chronic inflammation and cancer. Biochem. J. 2015, 468, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Judde, J.G.; Frost, P. Patterns of methionine auxotrophy in normal and neoplastic cells: The methionine independence of lymphocyte mitogenesis and low frequency of the methionine-dependent phenotype in human tumors. Cancer Res. 1988, 48, 6775–6779. [Google Scholar] [PubMed]
- Martínez, Y.; Li, X.; Liu, G.; Bin, P.; Yan, W.; Más, D.; Valdivié, M.; Hu, C.A.; Ren, W.; Yin, Y. The role of methionine on metabolism, oxidative stress, and diseases. Amino Acids 2017, 49, 2091–2098. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, S.; Hoffman, R.M.; Bertino, J.R. Exploiting methionine restriction for cancer treatment. Biochem. Pharmacol. 2018, 154, 170–173. [Google Scholar] [CrossRef]
- Gao, X.; Sanderson, S.M.; Dai, Z.; Reid, M.A.; Cooper, D.E.; Lu, M.; Richie, J.P., Jr.; Ciccarella, A.; Calcagnotto, A.; Mikhael, P.G.; et al. Dietary methionine influences therapy in mouse cancer models and alters human metabolism. Nature 2019, 572, 397–401. [Google Scholar] [CrossRef]
- Pokrovsky, V.S.; Qoura, L.A.; Demidova, E.A.; Han, Q.; Hoffman, R.M. Targeting Methionine Addiction of Cancer Cells with Methioninase. Biochem. (Mosc.) 2023, 88, 944–952. [Google Scholar] [CrossRef] [PubMed]
- Nardone, G.; Compare, D.; De Colibus, P.; de Nucci, G.; Rocco, A. Helicobacter pylori and epigenetic mechanisms underlying gastric carcinogenesis. Dig. Dis. 2007, 25, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.O.; Peng, J.Z.; Lam, S.K.; Lai, K.C.; Yuen, M.F.; Cheung, H.K.; Kwong, Y.L.; Rashid, A.; Chan, C.K.; Wong, B.C. Eradication of Helicobacter pylori infection reverses E-cadherin promoter hypermethylation. Gut 2006, 55, 463–468. [Google Scholar] [CrossRef]
- Perri, F.; Cotugno, R.; Piepoli, A.; Merla, A.; Quitadamo, M.; Gentile, A.; Pilotto, A.; Annese, V.; Andriulli, A. Aberrant DNA methylation in non-neoplastic gastric mucosa of H. Pylori infected patients and effect of eradication. Am. J. Gastroenterol. 2007, 102, 1361–1371. [Google Scholar] [CrossRef]
- Scott, D.R.; Marcus, E.A.; Weeks, D.L.; Sachs, G. Mechanisms of acid resistance due to the urease system of Helicobacter pylori. Gastroenterology 2002, 123, 187–195. [Google Scholar] [CrossRef]
- Scott, D.R.; Marcus, E.A.; Wen, Y.; Singh, S.; Feng, J.; Sachs, G. Cytoplasmic histidine kinase (HP0244)-regulated assembly of urease with UreI, a channel for urea and its metabolites, CO2, NH3, and NH4(+), is necessary for acid survival of Helicobacter pylori. J. Bacteriol. 2010, 192, 94–103. [Google Scholar] [CrossRef]
- Olivera-Severo, D.; Uberti, A.F.; Marques, M.S.; Pinto, M.T.; Gomez-Lazaro, M.; Figueiredo, C.; Leite, M.; Carlini, C.R. A New Role for Helicobacter pylori Urease: Contributions to Angiogenesis. Front. Microbiol. 2017, 8, 1883. [Google Scholar] [CrossRef]
- Benoit, S.L.; Maier, R.J. Helicobacter Catalase Devoid of Catalytic Activity Protects the Bacterium against Oxidative Stress. J. Biol. Chem. 2016, 291, 23366–23373. [Google Scholar] [CrossRef]
- Kimura, H. Hydrogen sulfide and polysulfides as signaling molecules. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2015, 91, 131–159. [Google Scholar] [CrossRef] [PubMed]
- Greabu, M.; Totan, A.; Miricescu, D.; Radulescu, R.; Virlan, J.; Calenic, B. Hydrogen Sulfide, Oxidative Stress and Periodontal Diseases: A Concise Review. Antioxidants 2016, 5, 3. [Google Scholar] [CrossRef] [PubMed]
- Kimura, H.; Shibuya, N.; Kimura, Y. Hydrogen sulfide is a signaling molecule and a cytoprotectant. Antioxid. Redox Signal 2012, 17, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, Y.; Hirashita, Y.; Tamura, C.; Kudo, Y.; Sakai, K.; Togo, K.; Fukuda, K.; Matsunari, O.; Okamoto, K.; Ogawa, R.; et al. Helicobacter pylori infection modulates endogenous hydrogen sulfide production in gastric cancer AGS cells. Helicobacter 2020, 25, e12732. [Google Scholar] [CrossRef]
- Robert, K.; Nehmé, J.; Bourdon, E.; Pivert, G.; Friguet, B.; Delcayre, C.; Delabar, J.M.; Janel, N. Cystathionine beta synthase deficiency promotes oxidative stress, fibrosis, and steatosis in mice liver. Gastroenterology 2005, 128, 1405–1415. [Google Scholar] [CrossRef] [PubMed]
- Bourque, C.; Zhang, Y.; Fu, M.; Racine, M.; Greasley, A.; Pei, Y.; Wu, L.; Wang, R.; Yang, G. H2S protects lipopolysaccharide-induced inflammation by blocking NFκB transactivation in endothelial cells. Toxicol. Appl. Pharmacol. 2018, 338, 20–29. [Google Scholar] [CrossRef]
- Paul, B.D.; Snyder, S.H. H2S signalling through protein sulfhydration and beyond. Nat. Rev. Mol. Cell Biol. 2012, 13, 499–507. [Google Scholar] [CrossRef]
- Guo, F.F.; Yu, T.C.; Hong, J.; Fang, J.Y. Emerging Roles of Hydrogen Sulfide in Inflammatory and Neoplastic Colonic Diseases. Front. Physiol. 2016, 7, 156. [Google Scholar] [CrossRef]
- Graziosi, L.; Mencarelli, A.; Renga, B.; D’Amore, C.; Bruno, A.; Santorelli, C.; Cavazzoni, E.; Cantarella, F.; Rosati, E.; Donini, A.; et al. Epigenetic modulation by methionine deficiency attenuates the potential for gastric cancer cell dissemination. J. Gastrointest. Surg. 2013, 17, 39–49. [Google Scholar] [CrossRef]
- Di Bartolomeo, M.; Pietrantonio, F.; Pellegrinelli, A.; Martinetti, A.; Mariani, L.; Daidone, M.G.; Bajetta, E.; Pelosi, G.; de Braud, F.; Floriani, I.; et al. Osteopontin, E-cadherin, and β-catenin expression as prognostic biomarkers in patients with radically resected gastric cancer. Gastric Cancer 2016, 19, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Zhan, T.; Rindtorff, N.; Boutros, M. Wnt signaling in cancer. Oncogene 2017, 36, 1461–1473. [Google Scholar] [CrossRef]
- Shi, L.; Zhu, W.; Huang, Y.; Zhuo, L.; Wang, S.; Chen, S.; Zhang, B.; Ke, B. Cancer-associated fibroblast-derived exosomal microRNA-20a suppresses the PTEN/PI3K-AKT pathway to promote the progression and chemoresistance of non-small cell lung cancer. Clin. Transl. Med. 2022, 12, e989. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Han, T.; Wei, G.; Wang, Y. Inhibition of microRNA-17/20a suppresses cell proliferation in gastric cancer by modulating UBE2C expression. Oncol. Rep. 2015, 33, 2529–2536. [Google Scholar] [CrossRef][Green Version]
- Yang, R.; Fu, Y.; Zeng, Y.; Xiang, M.; Yin, Y.; Li, L.; Xu, H.; Zhong, J.; Zeng, X. Serum miR-20a is a promising biomarker for gastric cancer. Biomed. Rep. 2017, 6, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Chen, Q.; Li, Y.; Lei, G.; Scott, A.; Huo, L.; Li, C.Y.; Estrella, J.S.; Correa, A.; Pizzi, M.P.; et al. Targeting cancer stem cells with a pan-BCL-2 inhibitor in preclinical and clinical settings in patients with gastroesophageal carcinoma. Gut 2021, 70, 2238–2248. [Google Scholar] [CrossRef]
- Erkan, M.; Kleeff, J.; Esposito, I.; Giese, T.; Ketterer, K.; Büchler, M.W.; Giese, N.A.; Friess, H. Loss of BNIP3 expression is a late event in pancreatic cancer contributing to chemoresistance and worsened prognosis. Oncogene 2005, 24, 4421–4432. [Google Scholar] [CrossRef] [PubMed]
- Sugita, H.; Iida, S.; Inokuchi, M.; Kato, K.; Ishiguro, M.; Ishikawa, T.; Takagi, Y.; Enjoji, M.; Yamada, H.; Uetake, H.; et al. Methylation of BNIP3 and DAPK indicates lower response to chemotherapy and poor prognosis in gastric cancer. Oncol. Rep. 2011, 25, 513–518. [Google Scholar] [CrossRef]
- Xin, L.; Lu, H.; Liu, C.; Zeng, F.; Yuan, Y.W.; Wu, Y.; Wang, J.L.; Wu, D.Z.; Zhou, L.Q. Methionine deficiency promoted mitophagy via lncRNA PVT1-mediated promoter demethylation of BNIP3 in gastric cancer. Int. J. Biochem. Cell Biol. 2021, 141, 106100. [Google Scholar] [CrossRef]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An iron-dependent form of nonapoptotic cell death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef]
- Jiang, X.; Stockwell, B.R.; Conrad, M. Ferroptosis: Mechanisms, biology and role in disease. Nat. Rev. Mol. Cell Biol. 2021, 22, 266–282. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lin, J.; Li, Z.; Wang, M. In what area of biology has a “new” type of cell death been discovered? Biochim. Biophys. Acta Rev. Cancer 2023, 1878, 188955. [Google Scholar] [CrossRef]
- Magtanong, L.; Ko, P.J.; To, M.; Cao, J.Y.; Forcina, G.C.; Tarangelo, A.; Ward, C.C.; Cho, K.; Patti, G.J.; Nomura, D.K.; et al. Exogenous Monounsaturated Fatty Acids Promote a Ferroptosis-Resistant Cell State. Cell Chem. Biol. 2019, 26, 420–432.e9. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Martínez, R.; Cruz-Gil, S.; Gómez de Cedrón, M.; Álvarez-Fernández, M.; Vargas, T.; Molina, S.; García, B.; Herranz, J.; Moreno-Rubio, J.; Reglero, G.; et al. A link between lipid metabolism and epithelial-mesenchymal transition provides a target for colon cancer therapy. Oncotarget 2015, 6, 38719–38736. [Google Scholar] [CrossRef]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef]
- Dvorak, H.F. Tumors: Wounds that do not heal. Similarities between tumor stroma generation and wound healing. N. Engl. J. Med. 1986, 315, 1650–1659. [Google Scholar] [CrossRef]
- Grivennikov, S.I.; Greten, F.R.; Karin, M. Immunity, inflammation, and cancer. Cell 2010, 140, 883–899. [Google Scholar] [CrossRef] [PubMed]
- Wellenstein, M.D.; Coffelt, S.B.; Duits, D.E.M.; van Miltenburg, M.H.; Slagter, M.; de Rink, I.; Henneman, L.; Kas, S.M.; Prekovic, S.; Hau, C.S.; et al. Loss of p53 triggers WNT-dependent systemic inflammation to drive breast cancer metastasis. Nature 2019, 572, 538–542. [Google Scholar] [CrossRef]
- Bezzi, M.; Seitzer, N.; Ishikawa, T.; Reschke, M.; Chen, M.; Wang, G.; Mitchell, C.; Ng, C.; Katon, J.; Lunardi, A.; et al. Diverse genetic-driven immune landscapes dictate tumor progression through distinct mechanisms. Nat. Med. 2018, 24, 165–175. [Google Scholar] [CrossRef]
- Wellenstein, M.D.; de Visser, K.E. Cancer-Cell-Intrinsic Mechanisms Shaping the Tumor Immune Landscape. Immunity 2018, 48, 399–416. [Google Scholar] [CrossRef]
- Apetoh, L.; Ghiringhelli, F.; Tesniere, A.; Obeid, M.; Ortiz, C.; Criollo, A.; Mignot, G.; Maiuri, M.C.; Ullrich, E.; Saulnier, P.; et al. Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat. Med. 2007, 13, 1050–1059. [Google Scholar] [CrossRef] [PubMed]
- He, S.J.; Cheng, J.; Feng, X.; Yu, Y.; Tian, L.; Huang, Q. The dual role and therapeutic potential of high-mobility group box 1 in cancer. Oncotarget 2017, 8, 64534–64550. [Google Scholar] [CrossRef] [PubMed]
- Halliday, G.M.; Patel, A.; Hunt, M.J.; Tefany, F.J.; Barnetson, R.S. Spontaneous regression of human melanoma/nonmelanoma skin cancer: Association with infiltrating CD4+ T cells. World J. Surg. 1995, 19, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Waldman, A.D.; Fritz, J.M.; Lenardo, M.J. A guide to cancer immunotherapy: From T cell basic science to clinical practice. Nat. Rev. Immunol. 2020, 20, 651–668. [Google Scholar] [CrossRef] [PubMed]
- Bian, Y.; Li, W.; Kremer, D.M.; Sajjakulnukit, P.; Li, S.; Crespo, J.; Nwosu, Z.C.; Zhang, L.; Czerwonka, A.; Pawłowska, A.; et al. Cancer SLC43A2 alters T cell methionine metabolism and histone methylation. Nature 2020, 585, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Li, C.W.; Li, X.; Ding, Q.; Guo, L.; Liu, S.; Liu, C.; Lai, C.C.; Hsu, J.M.; Dong, Q.; et al. MET Inhibitors Promote Liver Tumor Evasion of the Immune Response by Stabilizing PDL1. Gastroenterology 2019, 156, 1849–1861.e13. [Google Scholar] [CrossRef] [PubMed]
- El-Kenawi, A.; Berglund, A.; Estrella, V.; Zhang, Y.; Liu, M.; Putney, R.M.; Yoder, S.J.; Johnson, J.; Brown, J.; Gatenby, R. Elevated Methionine Flux Drives Pyroptosis Evasion in Persister Cancer Cells. Cancer Res. 2023, 83, 720–734. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Tan, Y.T.; Chen, Y.X.; Zheng, X.J.; Wang, W.; Liao, K.; Mo, H.Y.; Lin, J.; Yang, W.; Piao, H.L.; et al. Methionine deficiency facilitates antitumour immunity by altering m(6)A methylation of immune checkpoint transcripts. Gut 2023, 72, 501–511. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, H.; Zhao, J.; Wan, P.; Hu, Y.; Lv, K.; Hu, Y.; Yang, X.; Ma, M. Activation of MAT2A-RIP1 signaling axis reprograms monocytes in gastric cancer. J. Immunother. Cancer 2021, 9, e001364. [Google Scholar] [CrossRef]
- Hinshaw, D.C.; Shevde, L.A. The Tumor Microenvironment Innately Modulates Cancer Progression. Cancer Res. 2019, 79, 4557–4566. [Google Scholar] [CrossRef]
- Chang, S.; Aune, T.M. Dynamic changes in histone-methylation ‘marks’ across the locus encoding interferon-gamma during the differentiation of T helper type 2 cells. Nat. Immunol. 2007, 8, 723–731. [Google Scholar] [CrossRef]
- Wei, G.; Wei, L.; Zhu, J.; Zang, C.; Hu-Li, J.; Yao, Z.; Cui, K.; Kanno, Y.; Roh, T.Y.; Watford, W.T.; et al. Global mapping of H3K4me3 and H3K27me3 reveals specificity and plasticity in lineage fate determination of differentiating CD4+ T cells. Immunity 2009, 30, 155–167. [Google Scholar] [CrossRef]
- Inoue, M.; Okamoto, K.; Terashima, A.; Nitta, T.; Muro, R.; Negishi-Koga, T.; Kitamura, T.; Nakashima, T.; Takayanagi, H. Arginine methylation controls the strength of γc-family cytokine signaling in T cell maintenance. Nat. Immunol. 2018, 19, 1265–1276. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Tong, Q.; Bishop, D.K.; Zhang, Y. Histone methyltransferase and histone methylation in inflammatory T-cell responses. Immunotherapy 2013, 5, 989–1004. [Google Scholar] [CrossRef]
- Correa, L.O.; Jordan, M.S.; Carty, S.A. DNA Methylation in T-Cell Development and Differentiation. Crit. Rev. Immunol. 2020, 40, 135–156. [Google Scholar] [CrossRef]
- Sinclair, L.V.; Howden, A.J.; Brenes, A.; Spinelli, L.; Hukelmann, J.L.; Macintyre, A.N.; Liu, X.; Thomson, S.; Taylor, P.M.; Rathmell, J.C.; et al. Antigen receptor control of methionine metabolism in T cells. eLife 2019, 8, e44210. [Google Scholar] [CrossRef]
- Moore, J.R.; Hubler, S.L.; Nelson, C.D.; Nashold, F.E.; Spanier, J.A.; Hayes, C.E. 1,25-Dihydroxyvitamin D(3) increases the methionine cycle, CD4(+) T cell DNA methylation and Helios(+)Foxp3(+) T regulatory cells to reverse autoimmune neurodegenerative disease. J. Neuroimmunol. 2018, 324, 100–114. [Google Scholar] [CrossRef]
- Feng, J.; Lü, S.; Ding, Y.; Zheng, M.; Wang, X. Homocysteine activates T cells by enhancing endoplasmic reticulum-mitochondria coupling and increasing mitochondrial respiration. Protein Cell 2016, 7, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Ni, S.J.; Zhao, L.Q.; Wang, X.F.; Wu, Z.H.; Hua, R.X.; Wan, C.H.; Zhang, J.Y.; Zhang, X.W.; Huang, M.Z.; Gan, L.; et al. CBX7 regulates stem cell-like properties of gastric cancer cells via p16 and AKT-NF-κB-miR-21 pathways. J. Hematol. Oncol. 2018, 11, 17. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.H.; Cheng, H.C.; Wang, Y.S.; Lin, P.; Jen, J.; Kuo, I.Y.; Chang, Y.H.; Liao, P.C.; Chen, R.H.; Yuan, W.C.; et al. Small GTPase Rab37 targets tissue inhibitor of metalloproteinase 1 for exocytosis and thus suppresses tumour metastasis. Nat. Commun. 2014, 5, 4804. [Google Scholar] [CrossRef]
- Ge, J.; Chen, Z.; Huang, J.; Chen, J.; Yuan, W.; Deng, Z.; Chen, Z. Upregulation of autophagy-related gene-5 (ATG-5) is associated with chemoresistance in human gastric cancer. PLoS ONE 2014, 9, e110293. [Google Scholar] [CrossRef]
- Sheng, Y.; Song, Y.; Li, Z.; Wang, Y.; Lin, H.; Cheng, H.; Zhou, R. RAB37 interacts directly with ATG5 and promotes autophagosome formation via regulating ATG5-12-16 complex assembly. Cell Death Differ. 2018, 25, 918–934. [Google Scholar] [CrossRef]
- Xie, J.; Chen, M.; Zhou, J.; Mo, M.S.; Zhu, L.H.; Liu, Y.P.; Gui, Q.J.; Zhang, L.; Li, G.Q. miR-7 inhibits the invasion and metastasis of gastric cancer cells by suppressing epidermal growth factor receptor expression. Oncol. Rep. 2014, 31, 1715–1722. [Google Scholar] [CrossRef]
- Zhao, X.D.; Lu, Y.Y.; Guo, H.; Xie, H.H.; He, L.J.; Shen, G.F.; Zhou, J.F.; Li, T.; Hu, S.J.; Zhou, L.; et al. MicroRNA-7/NF-κB signaling regulatory feedback circuit regulates gastric carcinogenesis. J. Cell Biol. 2015, 210, 613–627. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Liu, Z.; Liao, S.; Li, E.; Wu, X.; Zeng, W. Elevated microRNA-7 inhibits proliferation and tumor angiogenesis and promotes apoptosis of gastric cancer cells via repression of Raf-1. Cell Cycle 2020, 19, 2496–2508. [Google Scholar] [CrossRef] [PubMed]
- Shiraki, N.; Shiraki, Y.; Tsuyama, T.; Obata, F.; Miura, M.; Nagae, G.; Aburatani, H.; Kume, K.; Endo, F.; Kume, S. Methionine metabolism regulates maintenance and differentiation of human pluripotent stem cells. Cell Metab. 2014, 19, 780–794. [Google Scholar] [CrossRef] [PubMed]
- Xin, L.; Liu, L.; Liu, C.; Zhou, L.Q.; Zhou, Q.; Yuan, Y.W.; Li, S.H.; Zhang, H.T. DNA-methylation-mediated silencing of miR-7-5p promotes gastric cancer stem cell invasion via increasing Smo and Hes1. J. Cell Physiol. 2020, 235, 2643–2654. [Google Scholar] [CrossRef]
- Al-Batran, S.E.; Homann, N.; Pauligk, C.; Goetze, T.O.; Meiler, J.; Kasper, S.; Kopp, H.G.; Mayer, F.; Haag, G.M.; Luley, K.; et al. Perioperative chemotherapy with fluorouracil plus leucovorin, oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus cisplatin and epirubicin for locally advanced, resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4): A randomised, phase 2/3 trial. Lancet 2019, 393, 1948–1957. [Google Scholar] [CrossRef]
- Cunningham, D.; Allum, W.H.; Stenning, S.P.; Thompson, J.N.; Van de Velde, C.J.; Nicolson, M.; Scarffe, J.H.; Lofts, F.J.; Falk, S.J.; Iveson, T.J.; et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N. Engl. J. Med. 2006, 355, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Bang, Y.J.; Kim, Y.W.; Yang, H.K.; Chung, H.C.; Park, Y.K.; Lee, K.H.; Lee, K.W.; Kim, Y.H.; Noh, S.I.; Cho, J.Y.; et al. Adjuvant capecitabine and oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): A phase 3 open-label, randomised controlled trial. Lancet 2012, 379, 315–321. [Google Scholar] [CrossRef]
- Wachtel, M.S.; Zhang, Y.; Chiriva-Internati, M.; Frezza, E.E. Different regression equations relate age to the incidence of Lauren types 1 and 2 stomach cancer in the SEER database: These equations are unaffected by sex or race. BMC Cancer 2006, 6, 65. [Google Scholar] [CrossRef]
- Yano, S.; Zhang, Y.; Miwa, S.; Tome, Y.; Hiroshima, Y.; Uehara, F.; Yamamoto, M.; Suetsugu, A.; Kishimoto, H.; Tazawa, H.; et al. Spatial-temporal FUCCI imaging of each cell in a tumor demonstrates locational dependence of cell cycle dynamics and chemoresponsiveness. Cell Cycle 2014, 13, 2110–2119. [Google Scholar] [CrossRef] [PubMed]
- Yano, S.; Miwa, S.; Mii, S.; Hiroshima, Y.; Uehara, F.; Yamamoto, M.; Kishimoto, H.; Tazawa, H.; Bouvet, M.; Fujiwara, T.; et al. Invading cancer cells are predominantly in G0/G1 resulting in chemoresistance demonstrated by real-time FUCCI imaging. Cell Cycle 2014, 13, 953–960. [Google Scholar] [CrossRef]
- Kaiser, P. Methionine Dependence of Cancer. Biomolecules 2020, 10, 568. [Google Scholar] [CrossRef]
- Stern, P.H.; Hoffman, R.M. Elevated overall rates of transmethylation in cell lines from diverse human tumors. In Vitro 1984, 20, 663–670. [Google Scholar] [CrossRef]
- Cao, W.X.; Cheng, Q.M.; Fei, X.F.; Li, S.F.; Yin, H.R.; Lin, Y.Z. A study of preoperative methionine-depleting parenteral nutrition plus chemotherapy in gastric cancer patients. World J. Gastroenterol. 2000, 6, 255–258. [Google Scholar]
- Sugisawa, N.; Higuchi, T.; Han, Q.; Hozumi, C.; Yamamoto, J.; Tashiro, Y.; Nishino, H.; Kawaguchi, K.; Bouvet, M.; Murata, T.; et al. Oral recombinant methioninase combined with paclitaxel arrests recalcitrant ovarian clear cell carcinoma growth in a patient-derived orthotopic xenograft (PDOX) nude-mouse model. Cancer Chemother. Pharmacol. 2021, 88, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, R.M.; Stern, P.H. Dietary Methionine Restriction-Based Cancer Chemotherapy in Rodents. Methods Mol. Biol. 2019, 1866, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Yano, S.; Li, S.K.; Han, Q.H.; Tan, Y.Y.; Fujiwara, T.; Hoffman, R.M. Chemotherapy of methioninase-synchronized S/G2 phase-blocked cancer cells identified by cell cycle-specific fluorescent reporters. J. Clin. Oncol. 2012, 30, 1063. [Google Scholar] [CrossRef]
- Bui, N.T.; Livolsi, A.; Peyron, J.F.; Prehn, J.H. Activation of nuclear factor kappaB and Bcl-x survival gene expression by nerve growth factor requires tyrosine phosphorylation of IkappaBalpha. J. Cell Biol. 2001, 152, 753–764. [Google Scholar] [CrossRef]
- Pitti, R.M.; Marsters, S.A.; Ruppert, S.; Donahue, C.J.; Moore, A.; Ashkenazi, A. Induction of apoptosis by Apo-2 ligand, a new member of the tumor necrosis factor cytokine family. J. Biol. Chem. 1996, 271, 12687–12690. [Google Scholar] [CrossRef] [PubMed]
- Thivat, E.; Farges, M.C.; Bacin, F.; D’Incan, M.; Mouret-Reynier, M.A.; Cellarier, E.; Madelmont, J.C.; Vasson, M.P.; Chollet, P.; Durando, X. Phase II trial of the association of a methionine-free diet with cystemustine therapy in melanoma and glioma. Anticancer Res. 2009, 29, 5235–5240. [Google Scholar] [PubMed]
- Kubota, Y.; Han, Q.; Masaki, N.; Hozumi, C.; Hamada, K.; Aoki, Y.; Obara, K.; Tsunoda, T.; Hoffman, R.M. Elimination of Axillary-Lymph-Node Metastases in a Patient With Invasive Lobular Breast Cancer Treated by First-line Neo-adjuvant Chemotherapy Combined With Methionine Restriction. Anticancer Res. 2022, 42, 5819–5823. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zou, Y.; Yuan, Y.; Zhou, Q.; Yue, Z.; Liu, J.; Fan, L.; Xu, H.; Xin, L. The Role of Methionine Restriction in Gastric Cancer: A Summary of Mechanisms and a Discussion on Tumor Heterogeneity. Biomolecules 2024, 14, 161. https://doi.org/10.3390/biom14020161
Zou Y, Yuan Y, Zhou Q, Yue Z, Liu J, Fan L, Xu H, Xin L. The Role of Methionine Restriction in Gastric Cancer: A Summary of Mechanisms and a Discussion on Tumor Heterogeneity. Biomolecules. 2024; 14(2):161. https://doi.org/10.3390/biom14020161
Chicago/Turabian StyleZou, Yonghui, Yiwu Yuan, Qi Zhou, Zhenqi Yue, Jiang Liu, Luojun Fan, Hesong Xu, and Lin Xin. 2024. "The Role of Methionine Restriction in Gastric Cancer: A Summary of Mechanisms and a Discussion on Tumor Heterogeneity" Biomolecules 14, no. 2: 161. https://doi.org/10.3390/biom14020161
APA StyleZou, Y., Yuan, Y., Zhou, Q., Yue, Z., Liu, J., Fan, L., Xu, H., & Xin, L. (2024). The Role of Methionine Restriction in Gastric Cancer: A Summary of Mechanisms and a Discussion on Tumor Heterogeneity. Biomolecules, 14(2), 161. https://doi.org/10.3390/biom14020161






