Extraction, Characterization, and Chitosan Microencapsulation of Bioactive Compounds from Cannabis sativa L., Cannabis indica L., and Mitragyna speiosa K.
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Materials
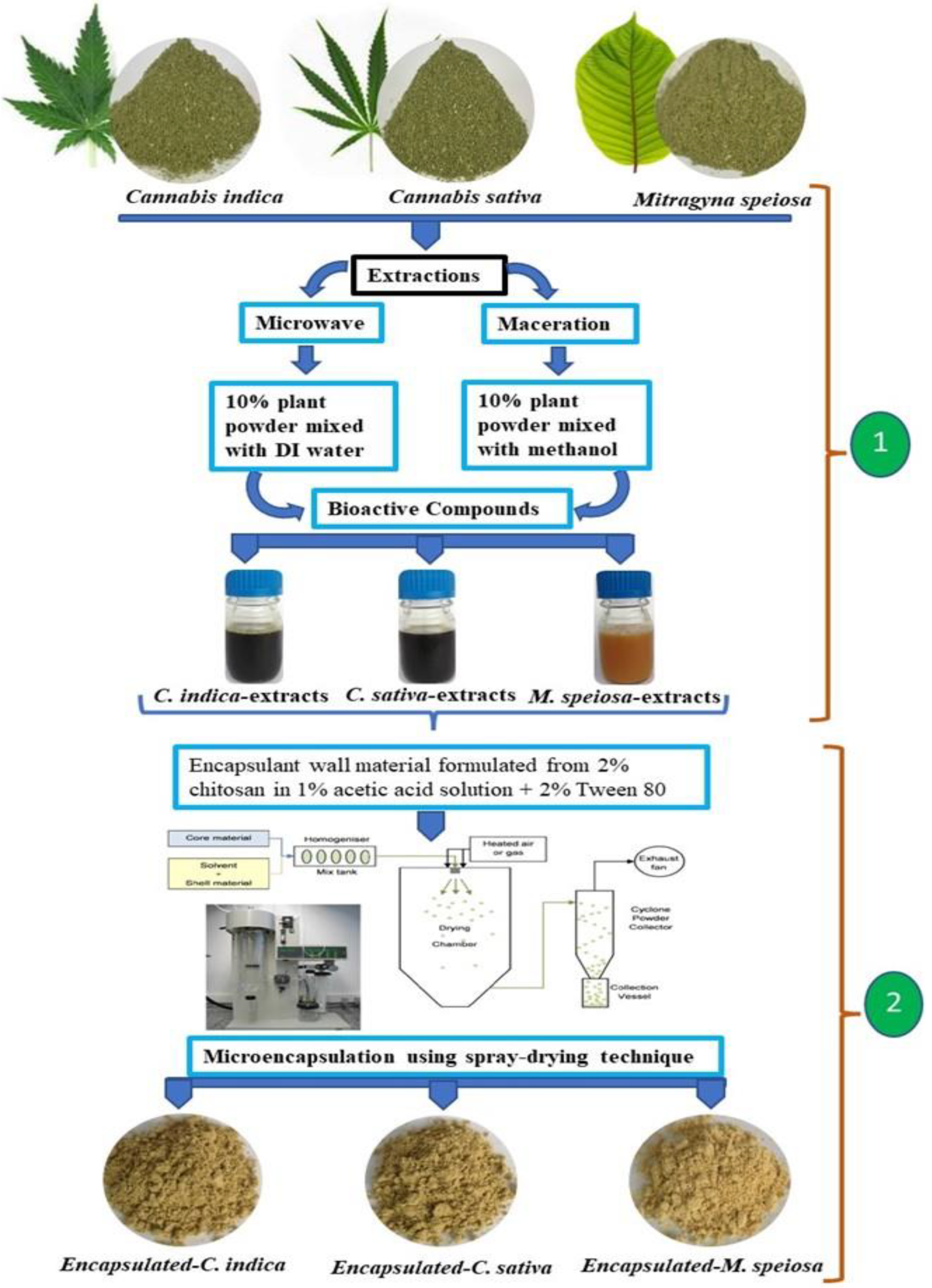
2.3. Extraction of Bioactive Compounds
2.4. Estimation of Total Polyphenolic and Total Flavonoid Contents and Antioxidant Capacity
2.5. Microencapsulation of Bioactive Compound Formulation Using Spray-Drying Technique
2.6. Encapsulation Efficiency
2.7. Morphological Characterization of Microencapsules
2.8. Statistical Analysis
3. Results and Discussion
3.1. Characteristics of Extracted-Bioactive Compounds
3.2. Bioactive Values and Encapsulation Efficiency of Microencapsulation
3.3. Microstructure and Surface Morphology of Dried Extracts and Chitosan Microcapsules
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mohammadalinejhad, S.; Kurek, M.A. Microencapsulation of Anthocyanins—Critical Review of Techniques and Wall Materials. Appl. Sci. 2021, 11, 3936. [Google Scholar] [CrossRef]
- Kurek, M.A.; Pratap-Singh, A. Plant-Based (Hemp, Pea, and Rice) Protein–Maltodextrin Combinations as Wall Material for Spray-Drying Microencapsulation of Hempseed (Cannabis sativa) Oil. Foods 2020, 9, 1707. [Google Scholar] [CrossRef]
- Drosou, C.G.; Krokida, M.K.; Biliaderis, C.G. Encapsulation of Bioactive Compounds through Electrospinning/Electrospraying and Spray Drying: A Comparative Assessment of Food-Related Applications. Dry. Technol. 2017, 35, 139–162. [Google Scholar] [CrossRef]
- Ko, J.A.; Park, H.J.; Hwang, S.J.; Park, J.B.; Lee, J.S. Preparation and Characterization of Chitosan Microparticles Intended for Controlled Drug Delivery. Int. J. Pharm. 2002, 249, 165–174. [Google Scholar] [CrossRef]
- Chaudhary, S.; Kumar, S.; Kumar, V.; Sharma, R. Chitosan Nanoemulsions as Advanced Edible Coatings for Fruits and Vegetables: Composition, Fabrication and Developments in Last Decade. Int. J. Biol. Macromol. 2020, 152, 154–170. [Google Scholar] [CrossRef]
- Nouri, A. Chitosan Nano-Encapsulation Improves the Effects of Mint, Thyme, and Cinnamon Essential Oils in Broiler Chickens. Br. Poult. Sci. 2019, 60, 530–538. [Google Scholar] [CrossRef]
- Drinić, Z.; Vladić, J.; Koren, A.; Zeremski, T.; Stojanov, N.; Kiprovski, B.; Vidović, S. Microwave-Assisted Extraction of Cannabinoids and Antioxidants from Cannabis sativa Aerial Parts and Process Modeling. J. Chem. Technol. Biotechnol. 2020, 95, 831–839. [Google Scholar] [CrossRef]
- Isidore, E.; Karim, H.; Ioannou, I. Extraction of Phenolic Compounds and Terpenes from Cannabis sativa L. By-Products: From Conventional to Intensified Processes. Antioxidants 2021, 10, 942. [Google Scholar] [CrossRef]
- Matešić, N.; Jurina, T.; Benković, M.; Panić, M.; Valinger, D.; Gajdoš Kljusurić, J.; Jurinjak Tušek, A. Microwave-Assisted Extraction of Phenolic Compounds from Cannabis sativa L.: Optimization and Kinetics Study. Sep. Sci. Technol. 2021, 56, 2047–2060. [Google Scholar] [CrossRef]
- Radoiu, M.; Kaur, H.; Bakowska-Barczak, A.; Splinter, S. Microwave-Assisted Industrial Scale Cannabis Extraction. Technologies 2020, 8, 45. [Google Scholar] [CrossRef]
- Fiorini, D.; Scortichini, S.; Bonacucina, G.; Greco, N.G.; Mazzara, E.; Petrelli, R.; Torresi, J.; Maggi, F.; Cespi, M. Cannabidiol-Enriched Hemp Essential Oil Obtained by an Optimized Microwave-Assisted Extraction Using a Central Composite Design. Ind. Crops Prod. 2020, 154, 112688. [Google Scholar] [CrossRef]
- Yiin, C.L.; Yusup, S.; Quitain, A.T.; Sasaki, M.; Uemura, Y.; Kida, T. Microwave-Assisted Hydrothermal Extraction of Natural Malic Acid for the Synthesis of Low Transition Temperature Mixtures. J. Clean. Prod. 2016, 113, 919–924. [Google Scholar] [CrossRef]
- Teh, S.S.; El-Din Bekhit, A.; Birch, J. Antioxidative Polyphenols from Defatted Oilseed Cakes: Effect of Solvents. Antioxidants 2014, 3, 67–80. [Google Scholar] [CrossRef]
- Ahmed, M.; Ji, M.; Qin, P.; Gu, Z.; Liu, Y.; Sikandar, A.; Iqbal, M.F.; Javeed, A. Phytochemical Screening, Total Phenolic and Flavonoids Contents and Antioxidant Activities of Citrullus Colocynthis L. and Cannabis sativa L. Appl. Ecol. Environ. Res. 2019, 17, 6961–6979. [Google Scholar] [CrossRef]
- Lazarjani, M.P.; Young, O.; Kebede, L.; Seyfoddin, A. Processing and Extraction Methods of Medicinal Cannabis: A Narrative Review. J. Cannabis Res. 2021, 3, 32. [Google Scholar] [CrossRef]
- Bailoni, L.; Bacchin, E.; Trocino, A.; Arango, S. Hemp (Cannabis sativa L.) Seed and Co-Products Inclusion in Diets for Dairy Ruminants: A Review. Animals 2021, 11, 856. [Google Scholar] [CrossRef]
- Phesatcha, K.; Phesatcha, B.; Wanapat, M.; Cherdthong, A. Mitragyna Speciosa Korth Leaves Supplementation on Feed Utilization, Rumen Fermentation Efficiency, Microbial Population, and Methane Production in Vitro. Fermentation 2022, 8, 8. [Google Scholar] [CrossRef]
- Chatterjee, P.N.; Kamra, D.N.; Agarwal, N.; Patra, A.K. Influence of Supplementation of Tropical Plant Feed Additives on in Vitro Rumen Fermentation and Methanogenesis. Anim. Prod. Sci. 2014, 54, 1770–1774. [Google Scholar] [CrossRef]
- Patra, A.K.; Saxena, J. The Effect and Mode of Action of Saponins on the Microbial Populations and Fermentation in the Rumen and Ruminant Production. Nutr. Res. Rev. 2009, 22, 204–219. [Google Scholar] [CrossRef]
- Cherdthong, A.; Khonkhaeng, B.; Foiklang, S.; Wanapat, M.; Gunun, N.; Gunun, P.; Chanjula, P.; Polyorach, S. Effects of Supplementation of Piper Sarmentosum Leaf Powder on Feed Efficiency, Rumen Ecology and Rumen Protozoal Concentration in Thai Native Beef Cattle. Animals 2019, 9, 130. [Google Scholar] [CrossRef]
- Matra, M.; Totakul, P.; Wanapat, M. Utilization of Dragon Fruit Waste By-Products and Non-Protein Nitrogen Source: Effects on in Vitro Rumen Fermentation, Nutrients Degradability and Methane Production. Livest. Sci. 2021, 243, 104386. [Google Scholar] [CrossRef]
- Phupaboon, S.; Punyauppa-Path, S.; Kontongdee, P.; Piyatheerawong, W.; Yunchalard, S. Development and Enhancement of Antioxidant Peptides from Spontaneous Plaa-Som Fermentation Co-Stimulated with Chiangrai Phulae Pineapple Enzymatic Reaction. Int. Food Res. J. 2022, 29, 406–415. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic Phosphotungstic Acid Reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. [Google Scholar]
- Braca, A.; Fico, G.; Morelli, I.; De Simone, F.; Tomè, F.; De Tommasi, N. Antioxidant and Free Radical Scavenging Activity of Flavonol Glycosides from Different Aconitum Species. J. Ethnopharmacol. 2003, 86, 63–67. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. LWT—Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Dudonnédudonn, S.; Vitrac, X.; Couti, P.; Woillez, M.; de Bélis, R.; Sen, L.; des Substances Végétales, E.; Biologique, A. Comparative Study of Antioxidant Properties and Total Phenolic Content of 30 Plant Extracts of Industrial Interest Using DPPH, ABTS, FRAP, SOD, and ORAC Assays. J. Agric. Food Chem. 2009, 57, 1768–1774. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [Green Version]
- Adejoro, F.A.; Hassen, A.; Thantsha, M.S. Characterization of Starch and Gum Arabic-Maltodextrin Microparticles Encapsulating Acacia Tannin Extract and Evaluation of Their Potential Use in Ruminant Nutrition. Asian-Australas. J. Anim. Sci. 2019, 32, 977–987. [Google Scholar] [CrossRef]
- Lehmann, T.; Brenneisen, R. A New Chromatographic Method for the Isolation of (−)-Δ9-(Trans)-tetrahydrocannabinolic Acid A. Phytochem. Anal. 1992, 3, 88–90. [Google Scholar] [CrossRef]
- Brighenti, V.; Pellati, F.; Steinbach, M.; Maran, D.; Benvenuti, S. Development of a New Extraction Technique and HPLC Method for the Analysis of Non-Psychoactive Cannabinoids in Fibre-Type Cannabis sativa L. (Hemp). J. Pharm. Biomed. Anal. 2017, 143, 228–236. [Google Scholar] [CrossRef]
- Al Ubeed, H.M.S.; Bhuyan, D.J.; Alsherbiny, M.A.; Basu, A.; Vuong, Q.V. A Comprehensive Review on the Techniques for Extraction of Bioactive Compounds from Medicinal Cannabis. Molecules 2022, 27, 604. [Google Scholar] [CrossRef]
- Latif, S.; Anwar, F. Physicochemical Studies of Hemp (Cannabis sativa) Seed Oil Using Enzyme-Assisted Cold-Pressing. Eur. J. Lipid Sci. Technol. 2009, 111, 1042–1048. [Google Scholar] [CrossRef]
- Kitrytė, V.; Bagdonaitė, D.; Rimantas Venskutonis, P. Biorefining of Industrial Hemp (Cannabis sativa L.) Threshing Residues into Cannabinoid and Antioxidant Fractions by Supercritical Carbon Dioxide, Pressurized Liquid and Enzyme-Assisted Extractions. Food Chem. 2018, 267, 420–429. [Google Scholar] [CrossRef]
- Azad, M.O.K.; Ryu, B.R.; Rana, M.S.; Rahman, M.H.; Lim, J.D.; Lim, Y.S. Enhancing the Cannabidiol (CBD) Compound in Formulated Hemp (Cannabis sativa L.) Leaves through the Application of Hot-Melt Extrusion. Processes 2021, 9, 837. [Google Scholar] [CrossRef]
- Fathordoobady, F.; Singh, A.; Kitts, D.D.; Pratap Singh, A. Hemp (Cannabis sativa L.) Extract: Anti-Microbial Properties, Methods of Extraction, and Potential Oral Delivery. Food Rev. Int. 2019, 35, 664–684. [Google Scholar] [CrossRef]
- Mazzara, E.; Torresi, J.; Fico, G.; Papini, A.; Kulbaka, N.; Dall’acqua, S.; Sut, S.; Garzoli, S.; Mustafa, A.M.; Cappellacci, L.; et al. A Comprehensive Phytochemical Analysis of Terpenes, Polyphenols and Cannabinoids, and Micromorphological Characterization of 9 Commercial Varieties of Cannabis sativa L. Plants 2022, 11, 891. [Google Scholar] [CrossRef]
- De Vita, D.; Madia, V.N.; Tudino, V.; Saccoliti, F.; De Leo, A.; Messore, A.; Roscilli, P.; Botto, A.; Pindinello, I.; Santilli, G.; et al. Comparison of Different Methods for the Extraction of Cannabinoids from Cannabis. Nat. Prod. Res. 2020, 34, 2952–2958. [Google Scholar] [CrossRef]
- Lewis-Bakker, M.M.; Yang, Y.; Vyawahare, R.; Kotra, L.P. Extractions of Medical Cannabis Cultivars and the Role of Decarboxylation in Optimal Receptor Responses. Cannabis Cannabinoid Res. 2019, 4, 183–194. [Google Scholar] [CrossRef] [Green Version]
- Belwal, T.; Ezzat, S.M.; Rastrelli, L.; Bhatt, I.D.; Daglia, M.; Baldi, A.; Devkota, H.P.; Orhan, I.E.; Patra, J.K.; Das, G.; et al. A Critical Analysis of Extraction Techniques Used for Botanicals: Trends, Priorities, Industrial Uses and Optimization Strategies. TrAC—Trends Anal. Chem. 2018, 100, 82–102. [Google Scholar] [CrossRef]
- Chemat, F.; Abert Vian, M.; Fabiano-Tixier, A.S.; Nutrizio, M.; Režek Jambrak, A.; Munekata, P.E.S.; Lorenzo, J.M.; Barba, F.J.; Binello, A.; Cravotto, G. A Review of Sustainable and Intensified Techniques for Extraction of Food and Natural Products. Green Chem. 2020, 22, 2325–2353. [Google Scholar] [CrossRef] [Green Version]
- Ibrahim, A.; Rahayu, B.P.; Arifuddin, M.; Nur, Y.; Prabowo, W.C.; Ambarwati, N.S.S.; Ahmad, I. Single Factor Effect of Natural Deep Eutectic Solvent Citric Acid-Glucose Based Microwave-Assisted Extraction on Total Polyphenols Content from Mitragyna speciosa Korth. Havil leaves. Pharmacogn. J. 2021, 13, 1109–1115. [Google Scholar]
- Parthasarathy, S.; Azizi, J.B.; Ramanathan, S.; Ismail, S.; Sasidharan, S.; Said, M.I.M.; Mansor, S.M. Evaluation of Antioxidant and Antibacterial Activities of Aqueous, Methanolic and Alkaloid Extracts from Mitragyna speciosa (Rubiaceae family) Leaves. Molecules 2009, 14, 3964–3974. [Google Scholar] [CrossRef] [PubMed]
- Rezvankhah, A.; Emam-Djomeh, Z.; Safari, M.; Askari, G.; Salami, M. Microwave-Assisted Extraction of Hempseed Oil: Studying and Comparing of Fatty Acid Composition, Antioxidant Activity, Physiochemical and Thermal Properties with Soxhlet Extraction. J. Food Sci. Technol. 2019, 56, 4198–4210. [Google Scholar] [CrossRef] [PubMed]
- Isahq, M.S.; Afridi, M.S.; Ali, J.; Hussain, M.M.; Ahmad, S.; Kanwal, F. Proximate Composition, Phytochemical Screening, GC-MS Studies of Biologically Active Cannabinoids and Antimicrobial Activities of Cannabis indica. Asian Pac. J. Trop. Dis. 2015, 5, 897–902. [Google Scholar] [CrossRef]
- De Moura, S.C.; Berling, C.L.; Garcia, A.O.; Queiroz, M.B.; Alvim, I.D.; Hubinger, M.D. Release of Anthocyanins from the Hibiscus extract Encapsulated by Ionic Gelation and Application of Microparticles in Jelly Candy. Food Res. Int. 2019, 121, 542–552. [Google Scholar] [CrossRef]
- Ezhilarasi, P.N.; Indrani, D.; Jena, B.S.; Anandharamakrishnan, C. Freeze Drying Technique for Microencapsulation of Garcinia Fruit Extract and Its Effect on Bread Quality. J. Food Eng. 2013, 117, 513–520. [Google Scholar] [CrossRef]
- Kaushik, V.; Roos, Y.H. Limonene Encapsulation in Freeze-Drying of Gum Arabic-Sucrose-Gelatin Systems. LWT—Food Sci. Technol. 2007, 40, 1381–1391. [Google Scholar] [CrossRef]
- Yadav, K.; Bajaj, R.K.; Mandal, S.; Saha, P.; Mann, B. Evaluation of Total Phenol Content and Antioxidant Properties of Encapsulated Grape Seed Extract in Yoghurt. Int. J. Dairy Technol. 2018, 71, 96–104. [Google Scholar] [CrossRef]
- Gruskiene, R.; Bockuviene, A.; Sereikaite, J. Microencapsulation of Bioactive Ingredients for Their Delivery into Fermented Milk Products: A Review. Molecules 2021, 26, 4601. [Google Scholar] [CrossRef]
- Aberkane, L.; Roudaut, G.; Saurel, R. Encapsulation and Oxidative Stability of PUFA-Rich Oil Microencapsulated by Spray Drying Using Pea Protein and Pectin. Food Bioprocess Technol. 2013, 7, 1505–1517. [Google Scholar] [CrossRef]
- Abraham, R.E.; Barrow, C.J.; Puri, M. Relationship to Reducing Sugar Production and Scanning Electron Microscope Structure to Pretreated Hemp Hurd Biomass (Cannabis sativa). Biomass Bioenergy 2013, 58, 180–187. [Google Scholar] [CrossRef]
- Liew, S.Q.; Chin, N.L.; Yusof, Y.A. Extraction and Characterization of Pectin from Passion Fruit Peels. Agric. Agric. Sci. Procedia 2014, 2, 231–236. [Google Scholar] [CrossRef]
- Jansen-Alves, C.; Fernandes, K.F.; Crizel-Cardozo, M.M.; Krumreich, F.D.; Borges, C.D.; Zambiazi, R.C. Microencapsulation of Propolis in Protein Matrix Using Spray Drying for Application in Food Systems. Food Bioprocess Technol. 2018, 11, 1422–1436. [Google Scholar] [CrossRef]

| Extraction Method | Bioactive Compounds | Antioxidant Capacity | |||
|---|---|---|---|---|---|
| TPC (mg GAE/g DM) | TFC (mg QUE/g DM) | DPPH Inhibition (%) | ABTS Inhibition (%) | FRAP Capacity (mg TROE/g DM) | |
| Microwave extraction | |||||
| C. sativa | 171.7 ± 0.4 c | 66.2 ± 4.7 c | 35.0 ± 6.4 b | 43.1 ± 2.3 b | 14.0 ± 0.7 c |
| C. indica | 218.9 ± 0.9 b | 88.6 ± 3.4 b | 39.3 ± 6.1 b | 94.6 ± 1.0 a | 23.7 ± 0.5 b |
| M. speiosa | 306.9 ± 0.3 a | 119.2 ± 5.2 a | 91.4 ± 0.5 a | 95.3 ± 0.4 a | 39.0 ± 0.1 a |
| Maceration extraction | |||||
| C. sativa | 11.3 ± 0.6 c | 7.1 ± 2.4 b | 24.7 ± 0.3 c | 3.7 ± 0.4 c | 7.7 ± 1.7 c |
| C. indica | 17.7 ± 1.1 b | 10.5 ± 1.9 a, b | 38.0 ± 0.5 b | 20.6 ± 0.6 b | 14.4 ± 0.3 b |
| M. speiosa | 21.7 ± 0.6 a | 13.7 ± 2.7 a | 91.3 ± 0.0 a | 88.9 ± 0.1 a | 39.4 ± 0.1 a |
| Formulations | Bioactive Compound | Antioxidant Capacity | Encapsulation Efficiency (%) | |||
|---|---|---|---|---|---|---|
| TPC (mg GAE/g DM) | TFC (mg QUE/g DM) | DPPH Inhibition (%) | ABTS Inhibition (%) | FRAP Capacity (mg TROE/g DM) | ||
| Encapsulated-C. sativa | 240.0 ± 6.6 c | 22.6 ± 2.7 c | 69.9 ± 0.7 c | 53.0 ± 0.4 a | 17.2 ± 1.1 b | 54.6 ± 10.7 c |
| Encapsulated-C. indica | 266.1 ± 6.8 b | 69.8 ± 3.0 b | 83.5 ± 1.0 b | 84.3 ± 0.9 b | 30.9 ± 0.7 a, b | 82.3 ± 7.6 b |
| Encapsulated-M. speiosa | 307.8 ± 6.4 a | 105.3 ± 3.6 a | 94.8 ± 0.4 a | 90.3 ± 1.2 a | 34.4 ± 0.7 a | 99.7 ± 4.1 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Phupaboon, S.; Matra, M.; Prommachart, R.; Totakul, P.; Supapong, C.; Wanapat, M. Extraction, Characterization, and Chitosan Microencapsulation of Bioactive Compounds from Cannabis sativa L., Cannabis indica L., and Mitragyna speiosa K. Antioxidants 2022, 11, 2103. https://doi.org/10.3390/antiox11112103
Phupaboon S, Matra M, Prommachart R, Totakul P, Supapong C, Wanapat M. Extraction, Characterization, and Chitosan Microencapsulation of Bioactive Compounds from Cannabis sativa L., Cannabis indica L., and Mitragyna speiosa K. Antioxidants. 2022; 11(11):2103. https://doi.org/10.3390/antiox11112103
Chicago/Turabian StylePhupaboon, Srisan, Maharach Matra, Ronnachai Prommachart, Pajaree Totakul, Chanadol Supapong, and Metha Wanapat. 2022. "Extraction, Characterization, and Chitosan Microencapsulation of Bioactive Compounds from Cannabis sativa L., Cannabis indica L., and Mitragyna speiosa K." Antioxidants 11, no. 11: 2103. https://doi.org/10.3390/antiox11112103






