Antioxidant and Compositional HPLC Analysis of Three Common Bamboo Leaves
Abstract
1. Introduction
2. Results
2.1. Validation of the HPLC Method
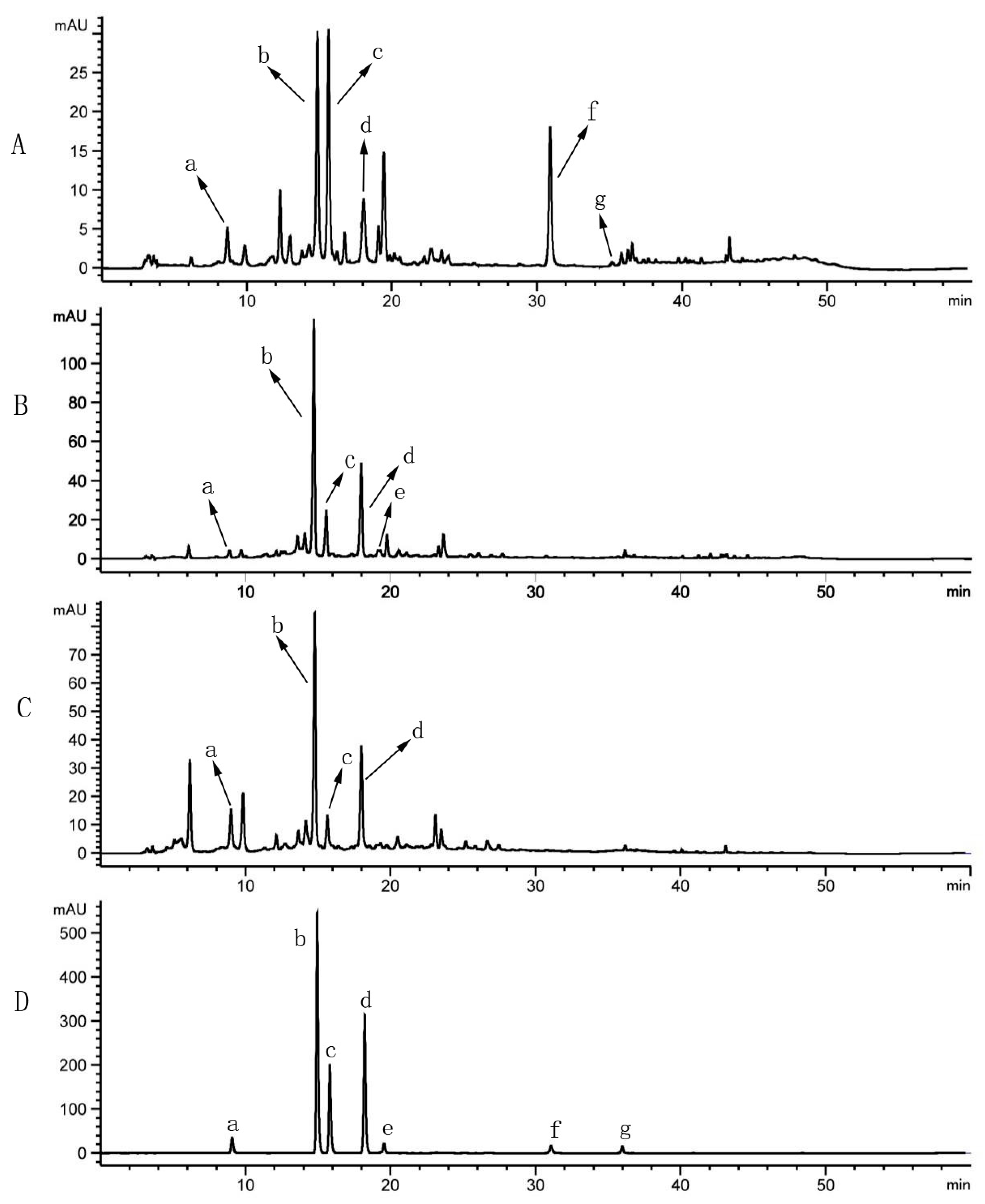
2.2. HPLC Analysis of Sample
2.3. DPPH Assay and Statistical Analysis
3. Discussion
4. Materials and Methods
4.1. Reagents and Chemicals
4.2. Plant Materials
4.3. Instruments and Chromatographic Conditions
4.4. Preparation of Solutions for HPLC Analysis
4.4.1. Standard Solutions and Calibration Curves
4.4.2. Sample Solutions
4.5. Preparation of Solutions for DPPH Assay
4.6. Statistical Analysis of Content and Antioxidant Activity
Author Contributions
Funding
Conflicts of Interest
References
- Peng, Z.; Lu, Y.; Li, L.; Zhao, Q.; Feng, Q.; Gao, Z.; Lu, H.; Hu, T.; Yao, N.; Liu, K.; et al. The draft genome of the fast-growing non-timber forest species moso bamboo (Phyllostachys heterocycla). Nat. Genet. 2013, 45, 456–461. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Tang, F.; Yue, Y.D. Research Progress of Chemical Constituents and Their Pharmacological Activities of Bamboo Plants. Chem. Ind. Forest Prod. 2013, 33, 149–156. [Google Scholar] [CrossRef]
- Geng, B.J.; Wang, Z.P. Flora Reipublicae Popularis Sinicae; Beijing Science Press: Beijing, China, 1996; Volume 35, p. 260. [Google Scholar]
- Cai, H.Q.; Zhan, Z.L.; Zheng, L.X.; Huang, Z.H. Overview of Quality Criteria of Lophatheri Herba. ACTA Chin. Med. 2017, 12, 2430–2434. [Google Scholar] [CrossRef]
- Huang, Z.H.; Cai, H.Q.; Zheng, L.X.; Guo, Y.L.; Zhan, Z.L. Textual research on the text and text of Chinese herbal medicine light bamboo leaf. J. Chin. Med. Mater. 2017, 40, 973–977. [Google Scholar] [CrossRef]
- Pharmacopoeia Committee of the people’s Republic of China. Pharmacopoeia of the People’s Republic of China; Beijing Chemical Industry Press: Beijing, China, 2015. [Google Scholar]
- Ren, Y.; Ma, Y.S.; Zhang, Z.D.; Qiu, L.Y.; Zhai, H.H.; Gu, R.M.; Xie, Y.P. Total Alkaloids from Bamboo Shoots and Bamboo Shoot Shells of Pleioblastus amarus (Keng) Keng f. and Their Anti-Inflammatory Activities. Molecules 2019, 24, 2699. [Google Scholar] [CrossRef]
- Ren, Y.; Dang, Y.H.; Zhang, Z.D.; Wan, D.G. A study on the leaves of Phyllostachys pubescens. J. Chin. Med. Mater. 2016, 39, 1430–1432. [Google Scholar] [CrossRef]
- Kyung, L.P.; Sung, T.K.; Min, J.K.; Hee, K.O. Antioxidative Components and Anti-Oralmicrobial Effect of Bamboo (Phyllostachys nigra var. henonis Stapf) Leaves. J. Korean Soc. Food Sci. Nutr. 2016, 45, 1265–1272. [Google Scholar] [CrossRef]
- Moon-Hee, C.; Han-Gyo, J.; Ji, H.Y.; Sung, H.K.; Hyun-Jae, S. Antioxidative and Anti-Melanogenic Activities of Bamboo Stems (Phyllostachys nigra variety henosis) via PKA/CREB-Mediated MITF Downregulation in B16F10 Melanoma Cells. Int. J. Mol. Sci. 2018, 19, 409. [Google Scholar] [CrossRef]
- Ji, H.Y.; Moon-Hee, C.; Chang-Su, N.; Sam, S.C.; Jae, H.K.; Sae, K.K.; Il, J.C.; Hyun-Jae, S.; Sung, H.K. Bamboo Stems (Phyllostachys nigra variety henosis) Containing Polyphenol Mixtures Activate Nrf2 and Attenuate Phenylhydrazine-Induced Oxidative Stress and Liver Injury. J. Nutrients 2019, 11, 114. [Google Scholar] [CrossRef]
- Wang, S.H.; Ji, Z.X.; Li, J.Y. Syndrome analysis and clinical application of Lophatherum and Gypsum Decoction. China’s Naturop. 2018, 26, 47–49. [Google Scholar] [CrossRef]
- Wei, Q.; Wang, S.; Tang, F.; Zhang, H.; Yu, J.; Yue, Y. Simultaneous Determination of 13 Flavonoids in Bamboo Leaves by HPLC. Sci. Silvae Sin. 2015, 8, 81–87. [Google Scholar] [CrossRef]
- Tang, Q.; Shao, M.; Wang, Y.; Zhao, H.; Fan, C.; Huang, X.; Li, Y.; Ye, W. Simultaneous Determination of 10 Bioactive Components of Lophatherum gracile Brongn by HPLC-DAD. Chromatogr. Sci. 2015, 53, 963–967. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.N.; Chen, M.; Fan, C.L.; Wang, Y.; Li, Y.L.; Ye, W.C. A new flavone C-glycoside from leaves of Lophatherum gracile. China, J. Chin. Mater. Med. 2014, 39, 247–249. [Google Scholar] [CrossRef]
- Sun, J.; Tang, F.; Yue, Y.D.; Xun, H.; Guo, X.F. Two new compounds from the dry leaves of Pleioblastus amarus (Keng) keng f. Asian Nat. Prod. Res. 2014, 16, 930–935. [Google Scholar] [CrossRef]
- Gong, J.; Xia, D.; Huang, J.; Ge, Q.; Mao, J.; Liu, S.; Zhang, Y. Functional components of bamboo shavings and bamboo leaf extracts and their antioxidant activities in vitro. Med. Food 2014, 18, 453–459. [Google Scholar] [CrossRef]
- Wang, L.P.; Guo, D.; Wang, G.; Zhou, H.H. Advancement of Chlorogenic Acid in Traditional Chinese Medicine. Lishizhen Med. Mater. Med. Res. 2011, 22, 961–963. [Google Scholar] [CrossRef]
- Luo, C.; Li, Y.; Long, J.G.; Zhang, Y.Q. Pharmacological Effects of Isoorientin. Space Med. Med. Eng. 2016, 29, 381–384. [Google Scholar] [CrossRef]
- Wan, S.Q.; Liu, L.Y.; Liu, M.S.; Huang, X.L. Study on Pharmacological Action Mechanism of Orientin. J. Med. Res. 2018, 47, 183–185. [Google Scholar] [CrossRef]
- Ying, Z.; Min, Z.; Ting, R.Z.; Jun, W.W.; Jun, W.; Ke, H.L.; Nan, N.Z. Antioxidant and Anti-inflammatory Activities of Cynaroside from Elsholtiza bodinieri. Nat. Prod. Commun. 2018, 13, 1501–1504. [Google Scholar] [CrossRef]
- Al-Megrin, W.A.; Alkhuriji, A.F.; Yousef, A.O.S.; Metwally, D.M.; Habotta, O.A.; Kassab, R.B.; Abdel Moneim, A.E.; El-Khadragy, M.F. Antagonistic Efficacy of Luteolin against Lead Acetate Exposure-Associated with Hepatotoxicity is Mediated via Antioxidant, Anti-Inflammatory, and Anti-Apoptotic Activities. Antioxidants (Basel) 2020, 9, 10. [Google Scholar] [CrossRef]
- Miao, Y.Z.; Li, Y.H.; Jing, S.L.; Lin, Z. Luteolin Attenuates Diabetic Nephropathy through Suppressing Inflammatory Response and Oxidative Stress by Inhibiting STAT3 Pathway. Exp. Clin. Endocr. Diab. 2020. [Google Scholar] [CrossRef]
- Sun, X.Q.; Li, R.H.; Tang, T. Research Progress of Apigenin Antioxidant Effect. China Mod. Dr. 2009, 47, 34–35. Available online: http://www.cnki.net (accessed on 1 September 2019).
- Zeng, M.F. Application Research of Several Natural Antioxidants in Cosmetics. Synth. Mater. Aging Appl. 2014, 43, 73–77. Available online: https://wenku.baidu.com/view/c112f3d2fd0a79563d1e723f.html (accessed on 31 August 2019).
- Pan, J.J.; She, X.Y.; Hu, L.F.; Shi, L.W.; Zhang, H. The Application of Bamboo Leaf Extracts in Foods. J. Bamboo Res. 2019, 38, 57–61. Available online: http://www.cqvip.com/QK/97864A/201902/7100180029.html (accessed on 31 June 2019).
- Suzuki, H.; Tabata, T.; Koizumi, H.; Hohchi, N.; Takeuchi, S.; Kitamura, T.; Fujino, Y.; Ohbuchi, T. Prediction of hearing outcomes by multiple regression analysis in patients with idiopathic sudden sensorineural hearing loss. Ann. Oto. Rhinol. Laryn. 2014, 123, 821–825. [Google Scholar] [CrossRef]
- Cao, H.H.; Du, R.F.; Yang, J.N.; Feng, Y. Correlation of dry granulation process parameters and granule quality based on multiple regression analysis. Acta Pharm. Sin. 2014, 49, 406–410. [Google Scholar] [CrossRef]
- Iizuka, A.; Iijima, O.T.; Kondo, K.; Itakura, H.; Yoshie, F.; Miyamoto, H.; Kubo, M.; Higuchi, M.; Takeda, H.; Matsumiya, T. Evaluation of Rhubarb using antioxidative activity as an index of pharmacological usefulness. J. Ethnopharmacol. 2004, 91, 89–94. [Google Scholar] [CrossRef]
- Wang, C.Z.; Zhang, H.Y.; Li, W.J.; Ye, J.Z. Chemical constituents and structural characterization of polysaccharides from four typical bamboo species leaves. Molecules 2015, 20, 4162–4179. [Google Scholar] [CrossRef]
- Gong, J.; Huang, J.; Xiao, G.; Chen, F.; Lee, B.; Ge, Q.; You, Y.; Liu, S.; Zhang, Y. Antioxidant Capacities of Fractions of Bamboo Shaving Extract and Their Antioxidant Components. Molecules 2016, 21, 996. [Google Scholar] [CrossRef]
- Mao, J.W.; Yin, J.; Ge, Q.; Jiang, Z.L.; Gong, J.Y. In vitro antioxidant activities of polysaccharides extracted from Moso Bamboo-Leaf. Int. J. Biol. Macromol. 2013, 55, 1–5. [Google Scholar] [CrossRef]
- Hamdi, A.; Viane, J.; Mahjoub, M.A.; Majouli, K.; Gad, M.; Kharbach, M.; Demeyer, K.; Marzouk, Z.; Heyden, Y.V. Polyphenolic contents, antioxidant activities and UPLC-ESI-MS analysis of Haplophyllum tuberculatum A. Juss leaves extracts. Int. J. Biol. Macromol. 2017, 106, 1071–1079. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
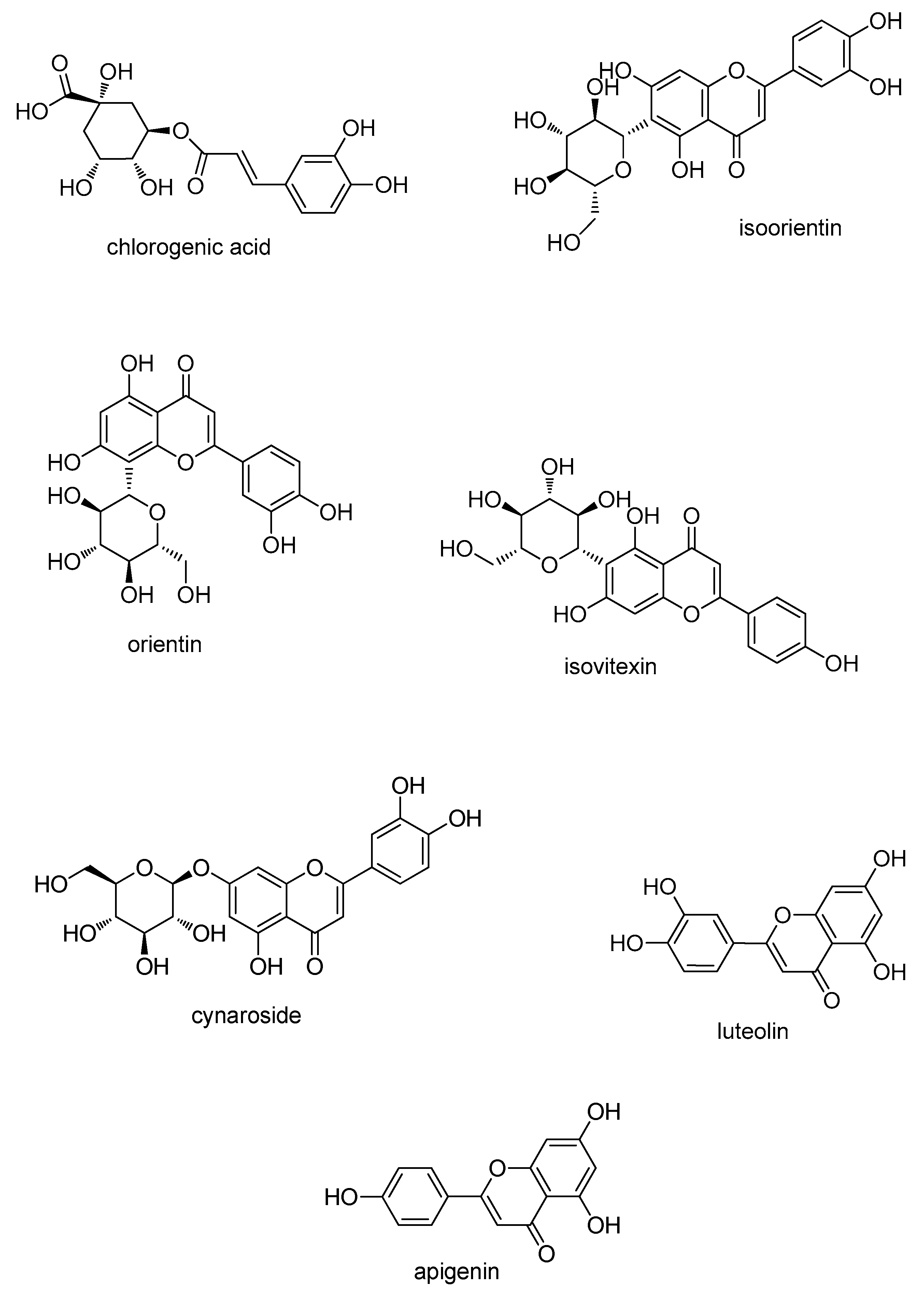
| Com. | Linear Range (µg·mL−1) | Linear Regression Equation (Y = a X + b) 1 | r | LOD (µg·mL−1) | LOQ (µg·mL−1) | Repeatability (RSD, %, n = 5) |
|---|---|---|---|---|---|---|
| CA | 1.15–73.80 | Y = 13.39 X + 8.37 | 0.9999 | 0.14 | 0.42 | 3.04 |
| iso-ORI | 1.85‒118.40 | Y = 29.14 X + 23.96 | 0.9999 | 0.08 | 0.17 | 2.86 |
| ORI | 0.85‒54.20 | Y = 17.18 X + 5.86 | 0.9999 | 0.08 | 0.12 | 2.91 |
| iso-VIT | 1.74–111.40 | Y = 26.87 X + 22.16 | 0.9999 | 0.06 | 0.11 | 2.71 |
| CYN | 0.32–20.24 | Y = 28.32 X + 2.35 | 1.0000 | 0.09 | 0.18 | 2.09 |
| LUT | 1.54–24.72 | Y = 39.79 X − 43.71 | 0.9998 | 0.19 | 0.58 | 0.14 |
| API | 1.28–10.22 | Y = 47.77 X − 16.63 | 0.9996 | 0.35 | 1.04 | 0.19 |
| Com. | Concentration (µg·mL−1) | Intraday Precision (RSD, %) | Stability (RSD, %) | Recovery Test | ||||
|---|---|---|---|---|---|---|---|---|
| Initial (µg) | Added (µg) | Detected (µg) | Recovery (%) | RSD (%) | ||||
| CA | 4.612 | 0.14 | 0.73 | 5.45 | 2.72 | 8.37 | 107.02 | 0.86 |
| 18.45 | 0.28 | 5.45 | 11.22 | 105.79 | 2.30 | |||
| 73.80 | 1.13 | 8.18 | 14.19 | 106.88 | 0.53 | |||
| iso-ORI | 7.400 | 0.21 | 1.72 | 41.10 | 20.54 | 60.63 | 95.03 | 5.17 |
| 29.60 | 0.21 | 41.09 | 79.28 | 92.90 | 2.21 | |||
| 118.4 | 0.26 | 61.64 | 98.58 | 93.26 | 0.87 | |||
| ORI | 3.388 | 0.14 | 1.01 | 14.16 | 7.08 | 21.54 | 104.16 | 1.16 |
| 13.55 | 0.19 | 14.16 | 29.12 | 105.64 | 0.84 | |||
| 54.20 | 0.28 | 21.24 | 36.86 | 106.86 | 0.25 | |||
| iso-VIT | 6.962 | 0.10 | 2.18 | 20.77 | 10.38 | 31.12 | 99.71 | 2.59 |
| 26.85 | 0.21 | 20.75 | 41.92 | 101.92 | 1.01 | |||
| 111.4 | 0.28 | 31.11 | 53.55 | 105.30 | 0.05 | |||
| CYN | 1.265 | 0.22 | 2.01 | 1.26 | 0.63 | 1.92 | 103.30 | 2.61 |
| 5.060 | 0.20 | 1.26 | 2.52 | 99.24 | 1.10 | |||
| 20.24 | 0.39 | 1.89 | 3.15 | 99.59 | 0.92 | |||
| LUT | 1.545 | 1.07 | 2.58 | 0.45 | 0.22 | 0.69 | 107.40 | 1.56 |
| 6.180 | 0.41 | 0.44 | 0.92 | 107.39 | 0.54 | |||
| 24.72 | 0.28 | 0.67 | 1.18 | 109.26 | 0.41 | |||
| API | 0.6388 | 0.87 | 3.35 | 0.22 | 0.11 | 0.36 | 122.32 | 0.10 |
| 2.555 | 0.15 | 0.22 | 0.48 | 112.14 | 0.39 | |||
| 10.22 | 0.22 | 0.34 | 0.59 | 109.74 | 0.66 | |||
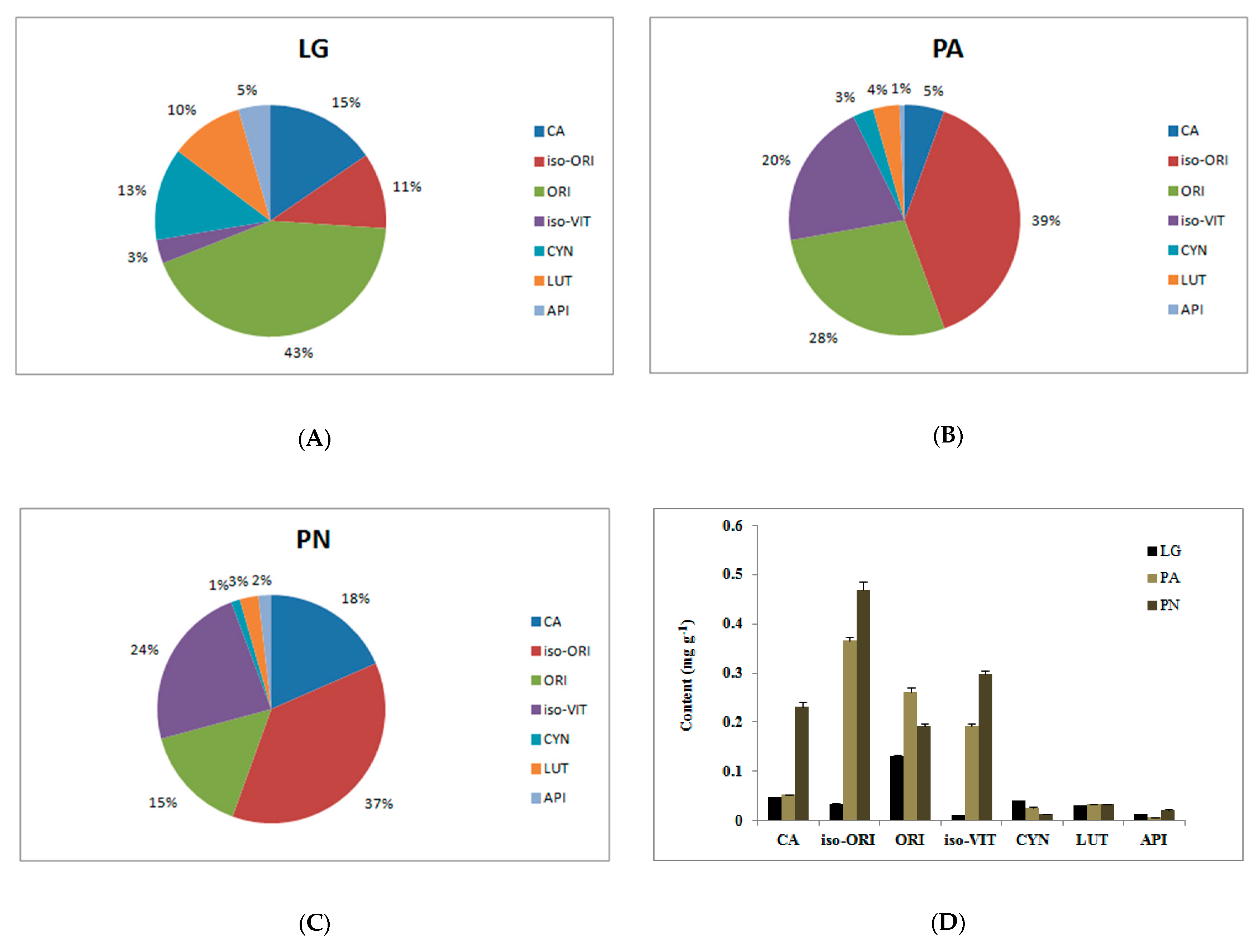
| No. | Botanical Name | Source | CA (µg·g−1) | iso-ORI (µg·g−1) | ORI (µg·g−1) | iso-VIT (µg·g−1) | CYN (µg·g−1) | LUT (µg·g−1) | API (µg·g−1) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | L. gracile | Anhui | 16.9 ± 0.9 | 1.4 ± 0 | Nd | 2.2 ± 0 | Nd | 13 ± 0.1 | 10.8 ± 0 |
| 2 | L. gracile | Sichuan | 7.5 ± 0.2 | 7.1 ± 0.1 | 326 ± 3.8 | 3.9 ± 0 | 92.6 ± 0.4 | 3.3 ± 0 | 11.4 ± 0.2 |
| 3 | L. gracile | Zhejiang | 8.1 ± 0.3 | 3.7 ± 0.1 | 214.1 ± 5 | 0.3 ± 0 | 21.8 ± 1.5 | 0.6 ± 0 | 6.5 ± 0.1 |
| 4 | L. gracile | Guangdong | 61.5 ± 0.6 | 7.6 ± 0.2 | 242.2 ± 6.8 | 4.6 ± 0.2 | 20.3 ± 1.2 | 0.4 ± 0 | 7.1 ± 0.1 |
| 5 | L. gracile | Zhejiang | 26.3 ± 1 | 22.4 ± 0.7 | 2.6 ± 0 | 5.6 ± 0.1 | 17.7 ± 0.8 | 54.1 ± 0.3 | Nd |
| 6 | L. gracile | Zhejiang | 175.5 ± 3.4 | 83.3 ± 7.3 | 6 ± 0.2 | 4.2 ± 0.1 | 48.1 ± 2.9 | 60.5 ± 0.3 | 23.1 ± 0 |
| 7 | L. gracile | Hebei | 35.6 ± 0.7 | 103.1 ± 10.1 | 1.5 ± 0.1 | 51.3 ± 3.7 | 38.5 ± 2.6 | 87.7 ± 1.6 | 23.6 ± 0 |
| 8 | P. amarus | Hebei | 175.9 ± 16.4 | 1923 ± 32.1 | 579.6 ± 10.6 | 897.1 ± 14.8 | 62.6 ± 1.1 | 21.6 ± 0.2 | 13.3 ± 0.1 |
| 9 | P. amarus | Zhejiang | Nd | 61.9 ± 2 | 17.4 ± 0.4 | Nd | Nd | 17.8 ± 0.2 | 6.6 ± 0.6 |
| 10 | P. amarus | Guangdong | 195.1 ± 2.9 | 440.3 ± 17.3 | 168.4 ± 3.4 | 98.9 ± 3.4 | 13.2 ± 0.4 | 16.7 ± 0.2 | 7.5 ± 0.3 |
| 11 | P. amarus | Sichuan | Nd | 52.5 ± 1.1 | 326.5 ± 2.8 | 0.4 ± 0 | 17 ± 0.4 | 49 ± 0 | Nd |
| 12 | P. amarus | Sichuan | Nd | 340.7 ± 8 | 107.3 ± 0.6 | 26.7 ± 0.6 | 101.7 ± 2.4 | 49.5 ± 0.1 | 30 ± 0.1 |
| 13 | P. amarus | Jiangsu | 12.2 ± 1.5 | 310.6 ± 12 | Nd | 46.3 ± 1.2 | 8.6 ± 0.5 | 18.6 ± 0.2 | Nd |
| 14 | P. amarus | Sichuan | Nd | 78.8 ± 3.1 | 473.7 ± 57.1 | 16 ± 0.5 | Nd | Nd | Nd |
| 15 | P. amarus | Sichuan | 30.4 ± 0.1 | 102.8 ± 1.7 | 571.4 ± 12.1 | 1.4 ± 0.1 | 18.8 ± 0.3 | 53.8 ± 0.2 | Nd |
| 16 | P. amarus | Jiangxi | Nd | 307.8 ± 8.6 | 136.8 ± 4.9 | 838 ± 17.6 | 21.2 ± 0.2 | 13.5 ± 0.1 | 11.8 ± 0.1 |
| 17 | P. amarus | Sichuan | Nd | 35.2 ± 1.8 | 226.7 ± 7.5 | 2.5 ± 0 | 11.3 ± 0.3 | 49 ± 0 | Nd |
| 18 | P. nigra | Anhui | 339.6 ± 11.7 | 488.6 ± 2.5 | 180 ± 3 | 390.8 ± 3.3 | 0.5 ± 0 | 15.3 ± 0 | 18.4 ± 0.1 |
| 19 | P. nigra | Jiangxi | 442.1 ± 8.5 | 304 ± 7.2 | 119.1 ± 4.6 | 404.2 ± 5.8 | 5.1 ± 0.5 | 15.5 ± 0 | 14.1 ± 0.3 |
| 20 | P. nigra | Anhui | 449 ± 41.4 | 185.1 ± 6.1 | 49.5 ± 3.9 | 347.2 ± 19.9 | 15.3 ± 1.4 | 14.3 ± 0.6 | 12.9 ± 0.3 |
| 21 | P. nigra | Zhejiang | 195 ± 1.4 | 607.3 ± 6.4 | 288.3 ± 4.4 | 317.5 ± 2.3 | 9.4 ± 0.4 | 49.2 ± 0.1 | 26.4 ± 0 |
| 22 | P. nigra | Zhejiang | 116.7 ± 5.1 | 863.3 ± 37.6 | 224.1 ± 9.7 | 386.1 ± 8.1 | 12.1 ± 0.5 | 50.8 ± 0.1 | 32 ± 0.4 |
| 23 | P. nigra | Zhejiang | 122.6 ± 3.8 | 1065.4 ± 50.1 | 377.5 ± 7.9 | 480.8 ± 14.7 | 11.7 ± 0.1 | 49.8 ± 0.1 | 33.6 ± 0.9 |
| 24 | P. nigra | Sichuan | 32 ± 0.2 | 269.2 ± 26.2 | 15.2 ± 1.4 | 33.2 ± 2.9 | 5.7 ± 0.1 | 52 ± 0.2 | 25.5 ± 0.1 |
| 25 | P. nigra | Zhejiang | 255.7 ± 2.4 | 414.3 ± 13.1 | 269.3 ± 8.4 | 301.6 ± 10.6 | 65.6 ± 1 | 51.6 ± 0.1 | 28 ± 0.2 |
| 26 | P. nigra | Zhejiang | 134.7 ± 3.2 | 12.5 ± 0.2 | 202.3 ± 9.3 | 12.4 ± 0.2 | 2.5 ± 0.2 | 0.4 ± 0 | 7.6 ± 0.1 |
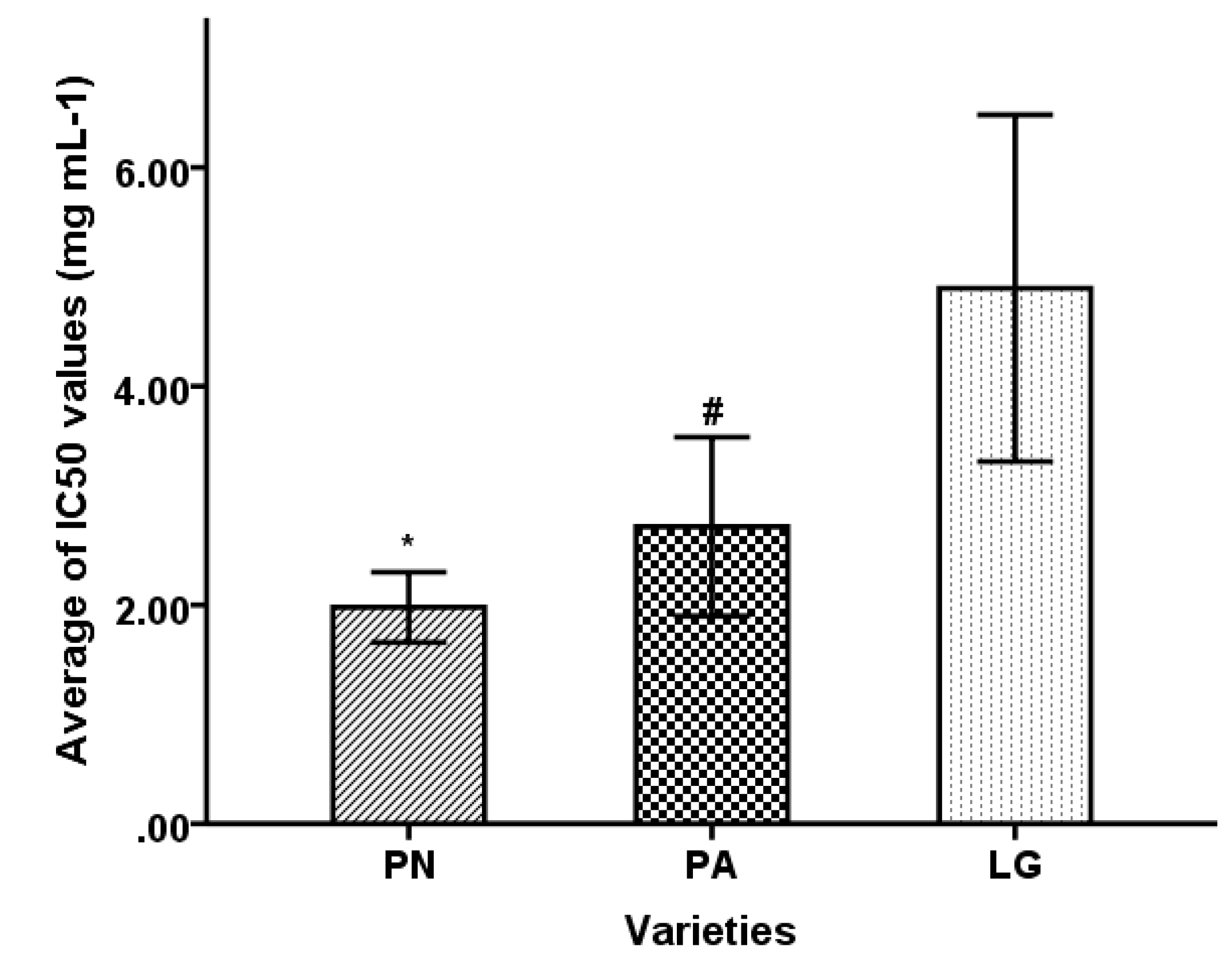
| Sample No. | Botanical Name | Source | Dry Ointment Yield of Extracts (%) | IC50 Value (mg·mL−1) |
|---|---|---|---|---|
| 1 | L. gracile | Anhui | 5.15 ± 0.11 | 4.09 ± 0.84 |
| 2 | L. gracile | Sichuan | 6.80 ± 0.09 | 6.69 ± 0.21 |
| 3 | L. gracile | Zhejiang | 5.51 ± 0.07 | 4.51 ± 0.03 |
| 4 | L. gracile | Guangdong | 5.74 ± 0.03 | 2.10 ± 0.06 |
| 5 | L. gracile | Zhejiang | 6.13 ± 0.07 | 10.17 ± 0.06 |
| 6 | L. gracile | Zhejiang | 8.06 ± 0.10 | 4.89 ± 0.58 |
| 7 | L. gracile | Hebei | 5.25 ± 0.04 | 3.06 ± 0.07 |
| 8 | P. amarus | Hebei | 8.93 ± 0.04 | 1.25 ± 0.12 |
| 9 | P. amarus | Zhejiang | 3.02 ± 0.05 | 2.11 ± 0.06 |
| 10 | P. amarus | Guangdong | 8.64 ± 0.04 | 2.18 ± 0.01 |
| 11 | P. amarus | Sichuan | 6.08 ± 0.03 | 3.09 ± 0.18 |
| 12 | P. amarus | Sichuan | 10.74 ± 1.17 | 3.25 ± 0.56 |
| 13 | P. amarus | Jiangsu | 6.67 ± 0.18 | 3.46 ± 0.39 |
| 14 | P. amarus | Sichuan | 8.04 ± 0.64 | 3.67 ± 0.25 |
| 15 | P. amarus | Sichuan | 8.36 ± 0.05 | 3.89 ± 0.07 |
| 16 | P. amarus | Jiangxi | 16.20 ± 0.84 | 4.48 ± 0.08 |
| 17 | P. amarus | Sichuan | 7.15 ± 0.07 | 5.07 ± 0.58 |
| 18 | P. nigra | Anhui | 15.10 ± 0.19 | 2.72 ± 0.06 |
| 19 | P. nigra | Jiangxi | 13.04 ± 0.05 | 2.02 ± 0.08 |
| 20 | P. nigra | Anhui | 12.42 ± 0.04 | 1.80 ± 0.02 |
| 21 | P. nigra | Zhejiang | 8.90 ± 0.45 | 1.59 ± 0.09 |
| 22 | P. nigra | Zhejiang | 8.90 ± 0.45 | 1.65 ± 0.08 |
| 23 | P. nigra | Zhejiang | 8.90 ± 0.45 | 1.85 ± 0.03 |
| 24 | P. nigra | Sichuan | 8.90 ± 0.45 | 2.63 ± 0.07 |
| 25 | P. nigra | Zhejiang | 9.43 ± 0.11 | 1.90 ± 0.14 |
| 26 | P. nigra | Zhejiang | 9.37 ± 0.16 | 1.65 ± 0.03 |
| No. | Compound | IC50 Value (mM) | Correlation Coefficient r | ||
|---|---|---|---|---|---|
| PN | PA | LG | |||
| 1 | CA | 1.04 ± 0.04 | −0.023 | −0.508 ** | −0.169 |
| 2 | Iso-ORI | 0.81 ± 0.01 | −0.171 | −0.643 ** | −0.181 |
| 3 | ORI | 0.84 ± 0.02 | −0.501 ** | −0.120 | −0.133 |
| 4 | Iso-VIT | 14.5 ± 0.04 | −0.193 | −0.226 | −0.293 |
| 5 | CYN | 0.43 ± 0.00 | −0.164 | −0.228 | 0.169 |
| 6 | LUT | 0.42 ± 0.01 | 0.021 | 0.431 * | 0.139 |
| 7 | API | Nd | −0.026 | −0.264 | −0.491 * |
| 8 | Trolox | 0.97 ± 0.04 | -- | -- | -- |
| 9 | VC | 0.93 ± 0.02 | -- | -- | -- |
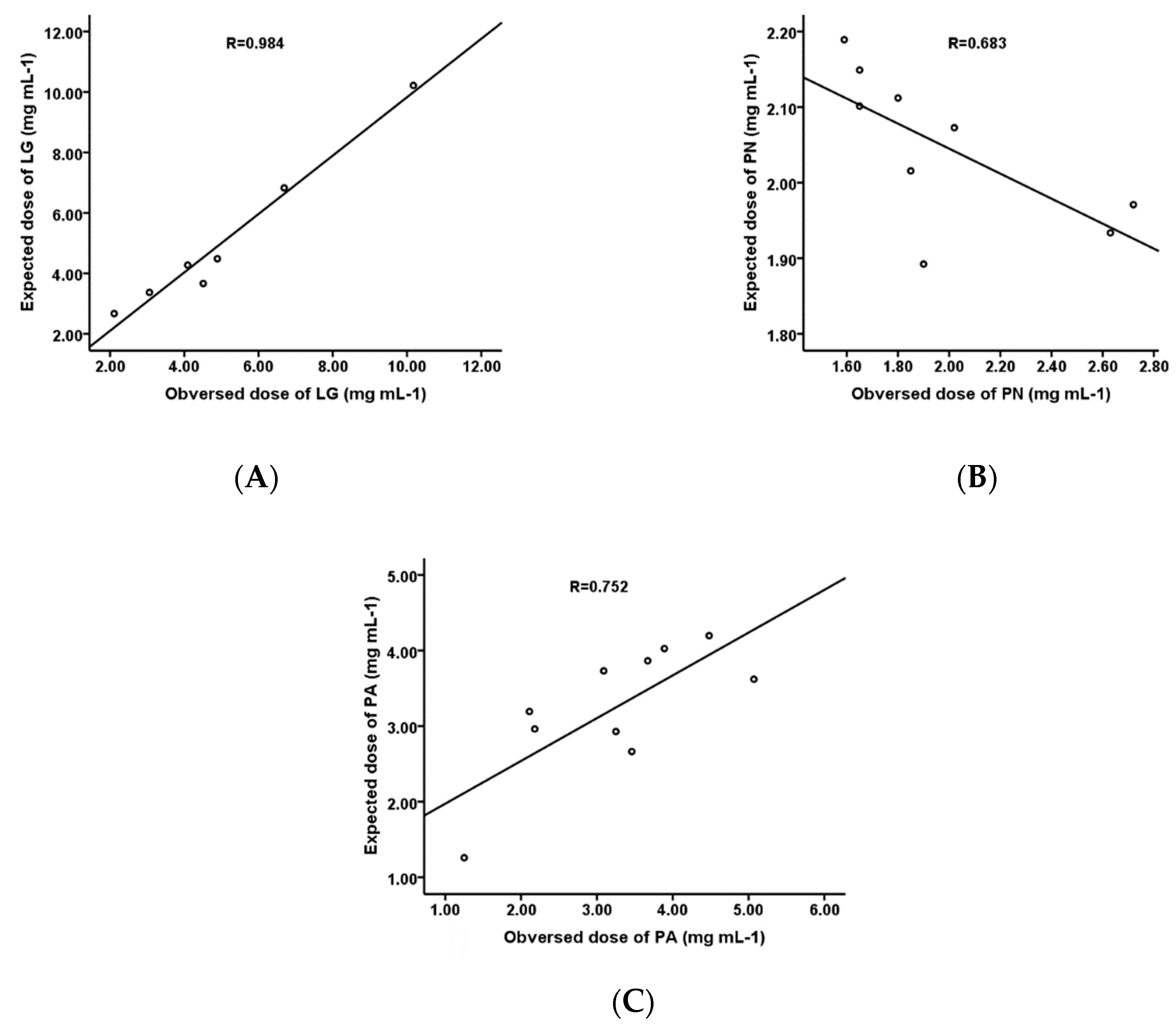
| Materials | Multiple Regression Equations 2 | IC50 Value(mM) |
|---|---|---|
| Lophatherum gracile | Y = 8.413 − 21.269 XORI + 104.946 XCYN − 383.105 XAPI | 5.07 ± 0.26 |
| Phyllostachys nigra | Y = 2.272 − 1.673 XORI | 3.25 ± 0.23 |
| Pleioblastus amarus | Y = 3.279 − 3.855 Xiso-ORI + 1.468 XTotal | 1.98 ± 0.06 |
| Botanical Name | Number | Source | Lot Number | Collection Date |
|---|---|---|---|---|
| P. amarus | 1 | Anguo Yikang Traditional Chinese Medicine Church, Hebei | XY170027 | 14 May 2017 |
| 2 | Anji, Zhejiang | XY170307 | 23 June 2017 | |
| 3 | Meizhou, Guangdong | XY170308 | 23 June 2017 | |
| 4 | Gaoxian, Sichuan | XY181247 | 26 March 2018 | |
| 5 | Yibin, Sichuan | XY181235 | 16 March 2018 | |
| 6 | Suqian, Jiangsu | XY181236 | 16 March 2018 | |
| 7 | Shuanghe, Sichuan | XY181248 | 26 March 2018 | |
| 8 | Meidong, Sichuan | XY181249 | 26 March 2018 | |
| 9 | Yudu, Jiangxi | XY170502 | 14 December 2017 | |
| 10 | Jiangan, Sichuan | XY181250 | 26 March 2018 | |
| L. gracile | 1 | Huoshan, Anhui | XY170499 | 10 December 2017 |
| 2 | Chengdu, Sichuan | XY170402 | 1 September 2017 | |
| 3 | Shanghai kangqiao traditional Chinese medicine decoction pieces co. LTD | XY170487 | 21 September 2017 | |
| 4 | Guangzhou, Guangdong | XY170489 | 5 November 2017 | |
| 5 | Wenzhou, Zhejiang | XY181230 | 12 March 2018 | |
| 6 | Lecheng, Zhejiang | XY181237 | 16 March 2018 | |
| 7 | Anguo Oriental Medicine City, Hebei | XY170311 | 18 June 2017 | |
| P. amarus | 1 | Yueshan, Anhui | XY170524 | 23 December 2017 |
| 2 | Jiujiang, Jiangxi | XY170526 | 25 December 2017 | |
| 3 | Anqing, Anhui | XY1700521 | 21 December 2017 | |
| 4 | Changxing, Zhejiang | XY170492 | 6 November 2017 | |
| 5 | Changxing, Zhejiang | XY181224 | 10 March 2018 | |
| 6 | Zhejiang | XY181233 | 15 March 2018 | |
| 7 | Qvzhou, Zhejiang (drying) | XY181242 | 18 March 2018 | |
| 8 | Qvzhou, Zhejiang (air drying) | XY170027 | 18 March 2018 | |
| 9 | Yibin, Sichuan | XY170307 | 16 March 2018 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, N.-H.; Guo, J.; Xu Chen, S.-H.; Yuan, X.-R.; Zhang, T.; Ding, Y. Antioxidant and Compositional HPLC Analysis of Three Common Bamboo Leaves. Molecules 2020, 25, 409. https://doi.org/10.3390/molecules25020409
Ma N-H, Guo J, Xu Chen S-H, Yuan X-R, Zhang T, Ding Y. Antioxidant and Compositional HPLC Analysis of Three Common Bamboo Leaves. Molecules. 2020; 25(2):409. https://doi.org/10.3390/molecules25020409
Chicago/Turabian StyleMa, Ning-Hui, Jing Guo, Si-Han Xu Chen, Xiu-Rong Yuan, Tong Zhang, and Yue Ding. 2020. "Antioxidant and Compositional HPLC Analysis of Three Common Bamboo Leaves" Molecules 25, no. 2: 409. https://doi.org/10.3390/molecules25020409
APA StyleMa, N.-H., Guo, J., Xu Chen, S.-H., Yuan, X.-R., Zhang, T., & Ding, Y. (2020). Antioxidant and Compositional HPLC Analysis of Three Common Bamboo Leaves. Molecules, 25(2), 409. https://doi.org/10.3390/molecules25020409





