Bathyptilones: Terpenoids from an Antarctic Sea Pen, Anthoptilum grandiflorum (Verrill, 1879)
Abstract
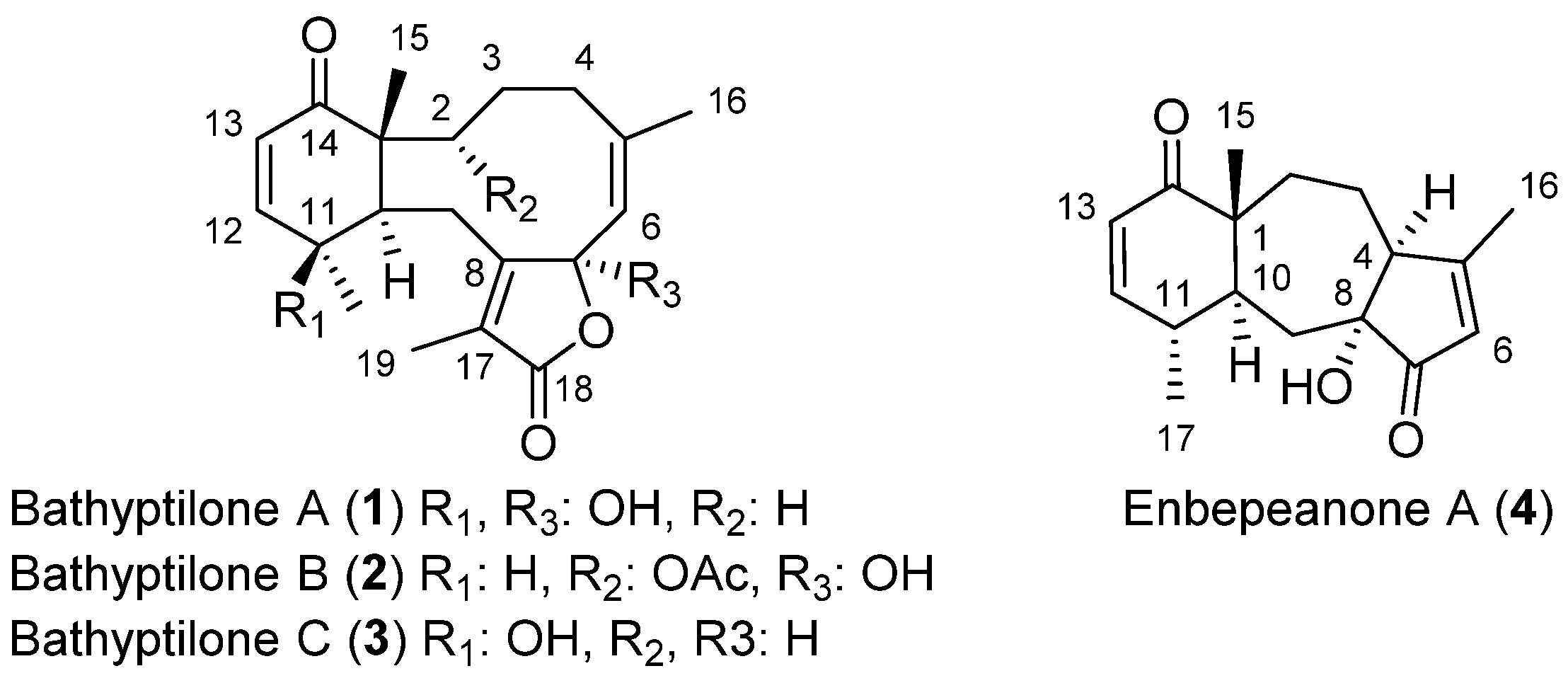
1. Introduction
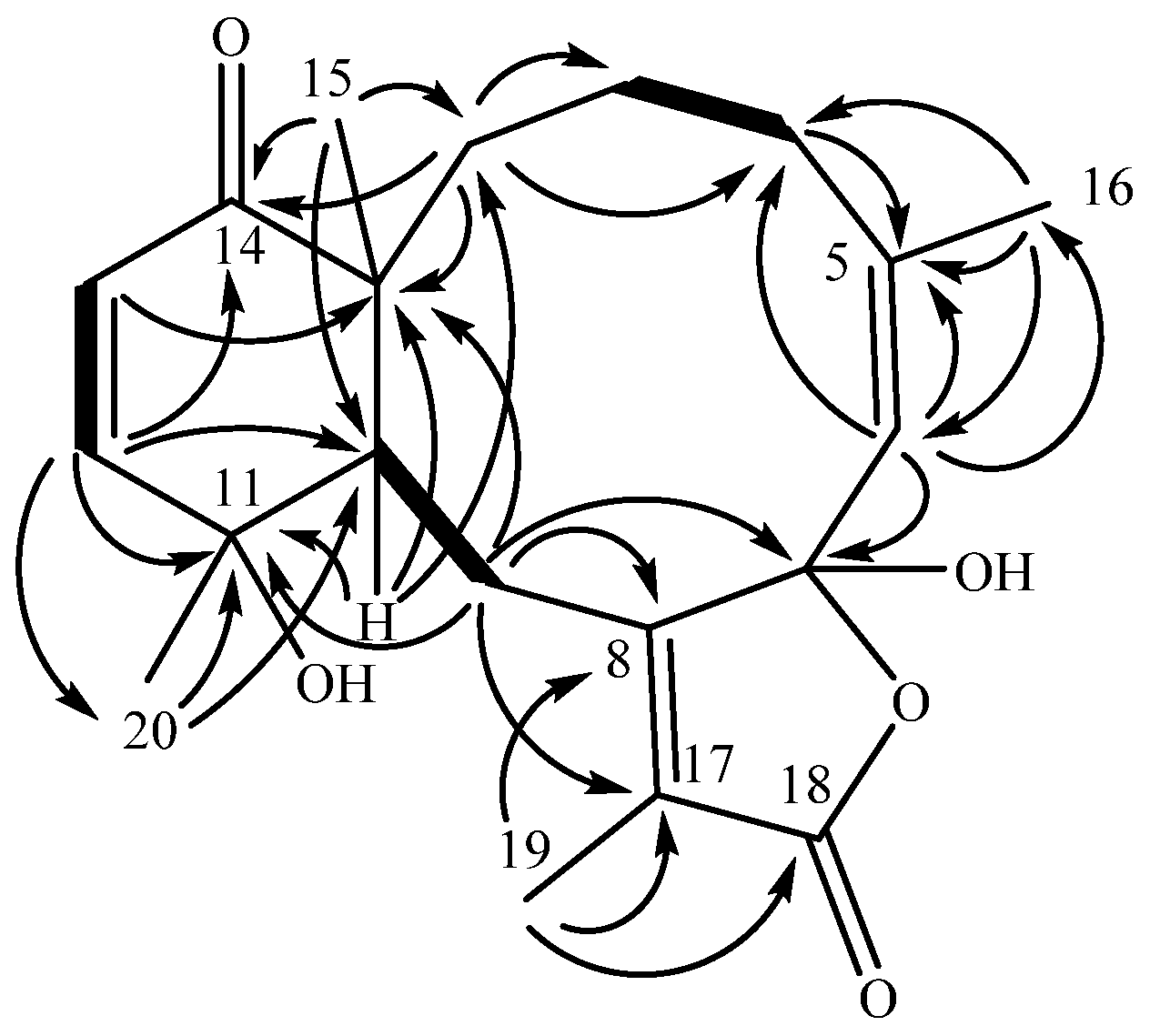
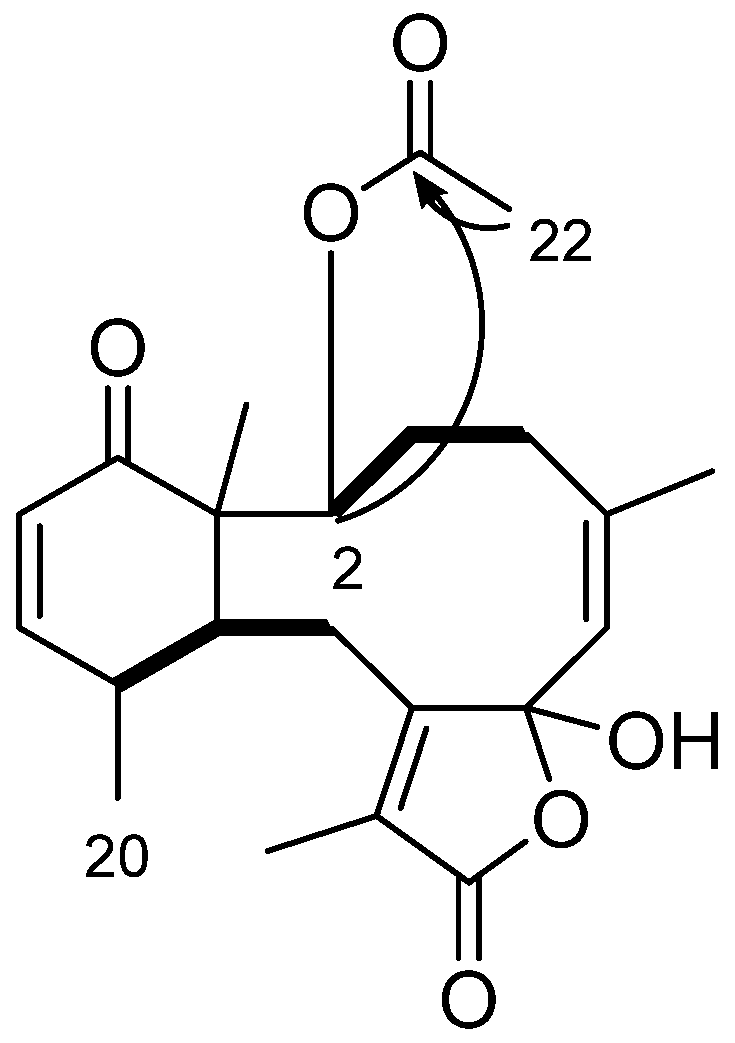
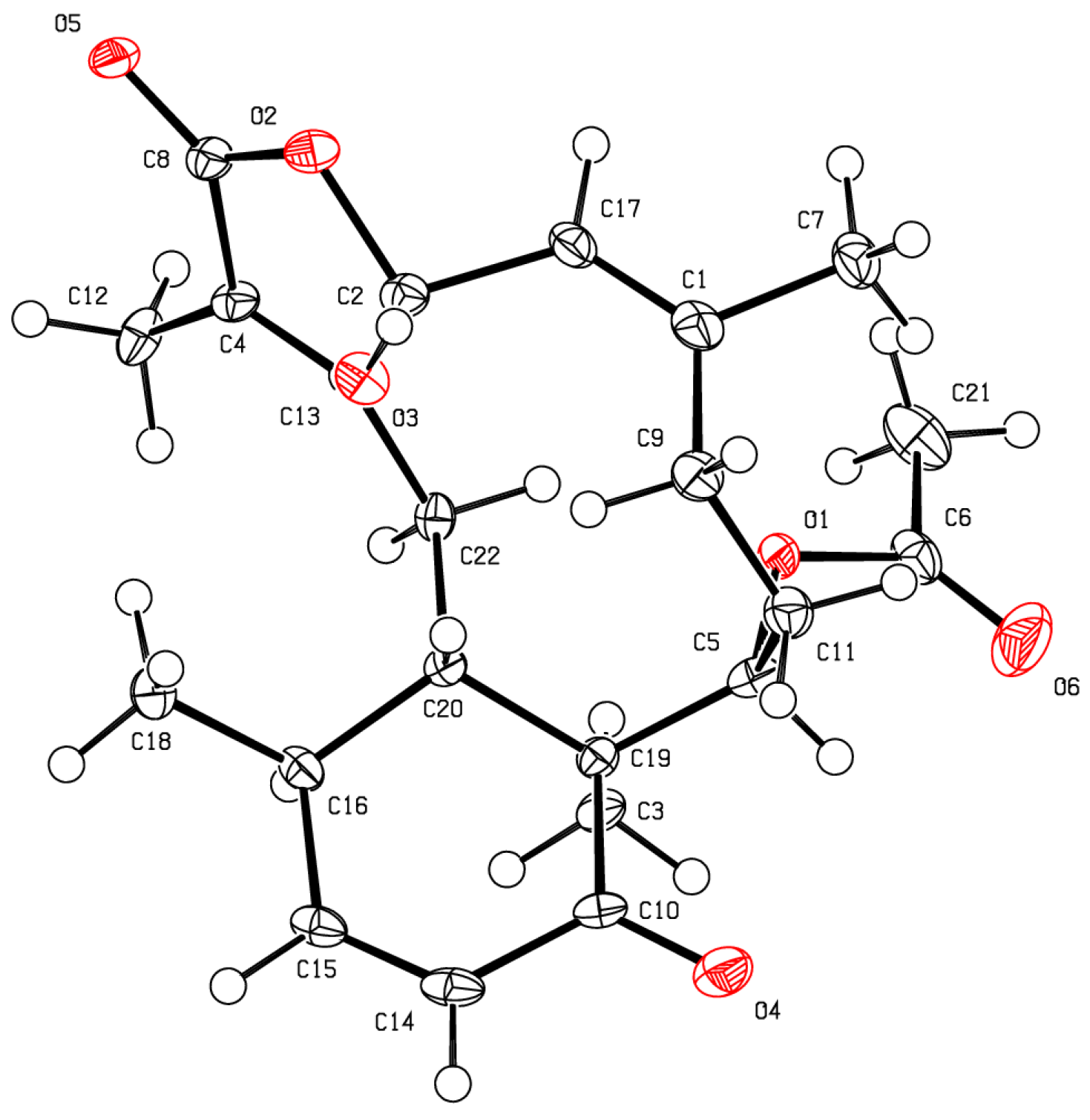
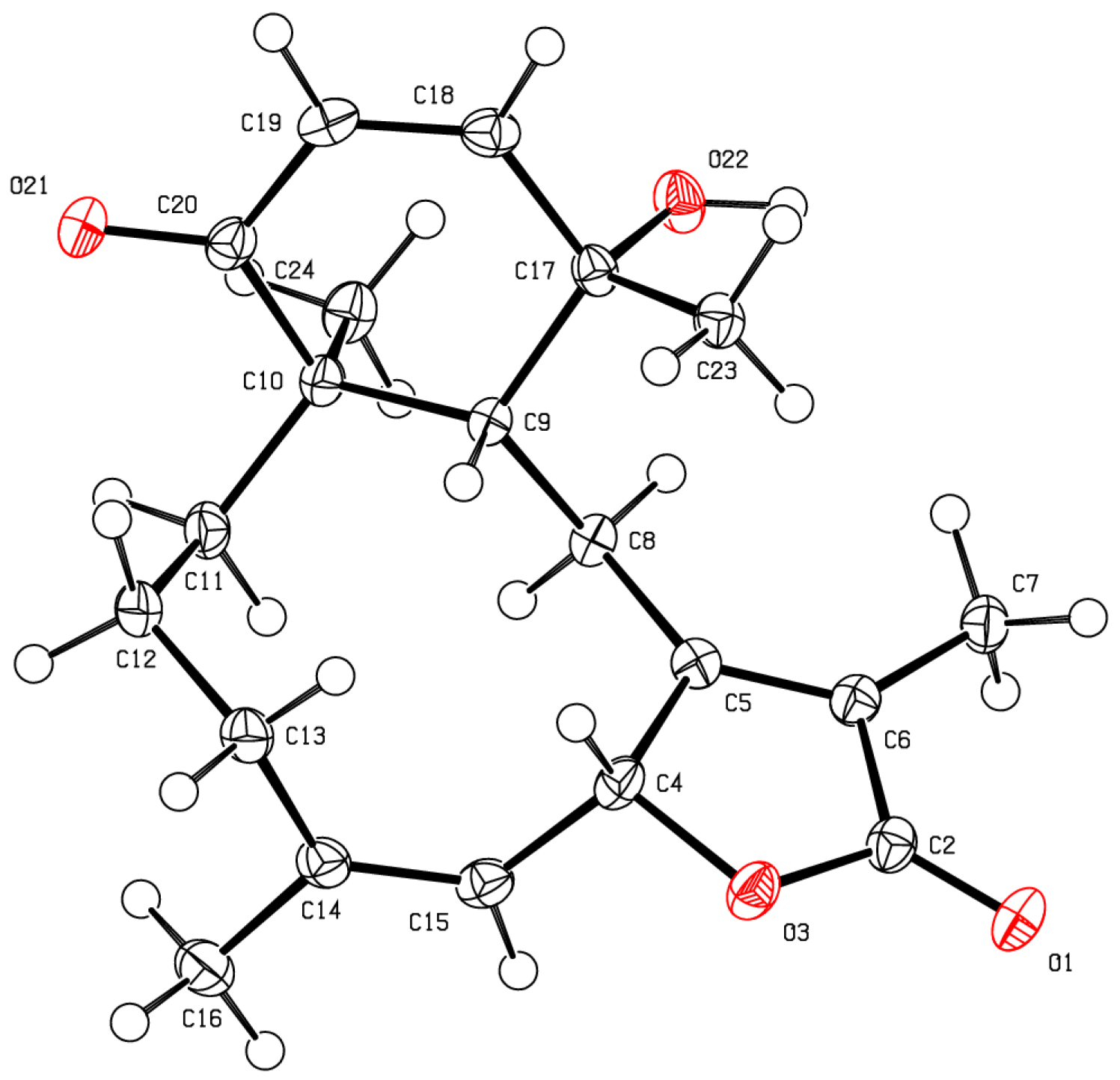
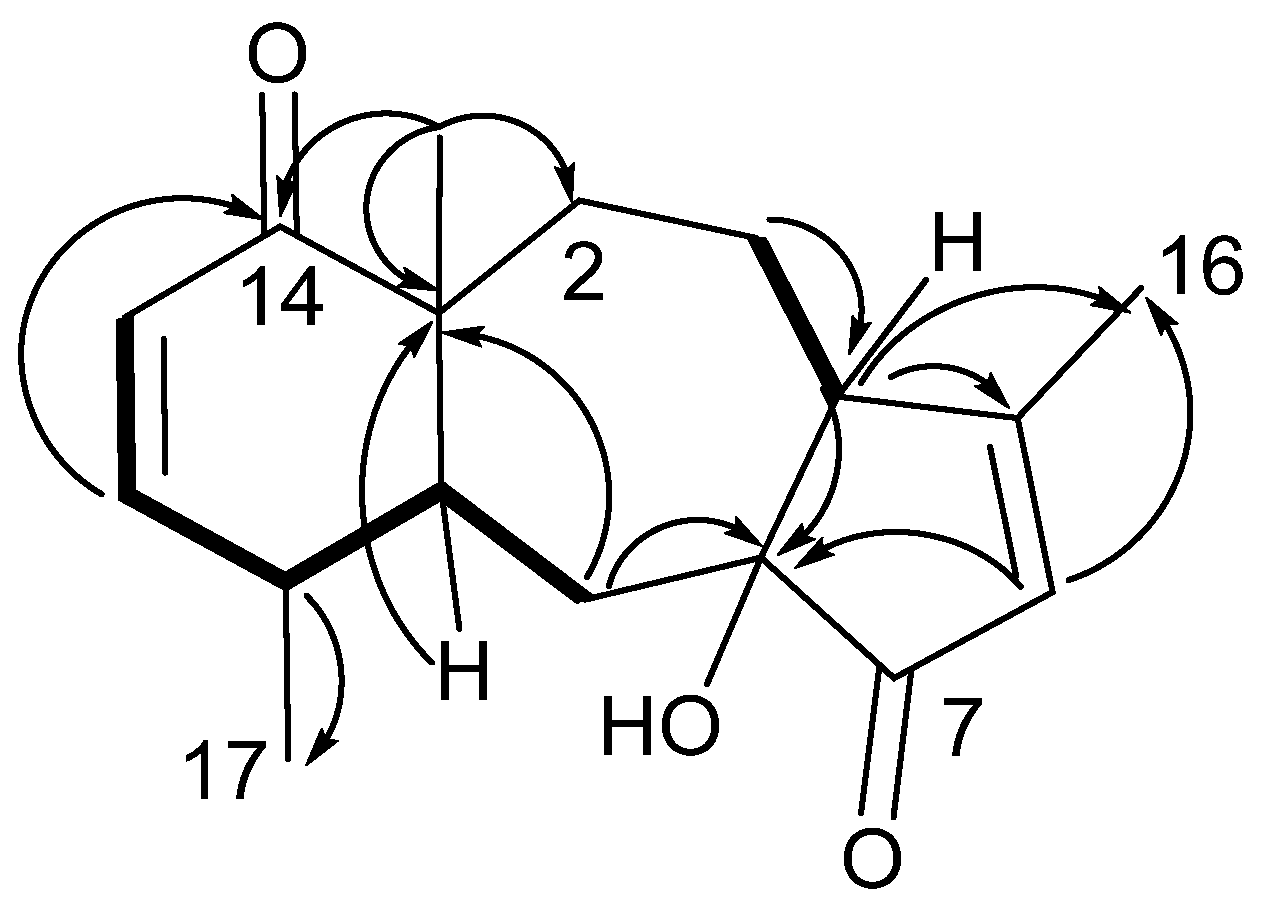
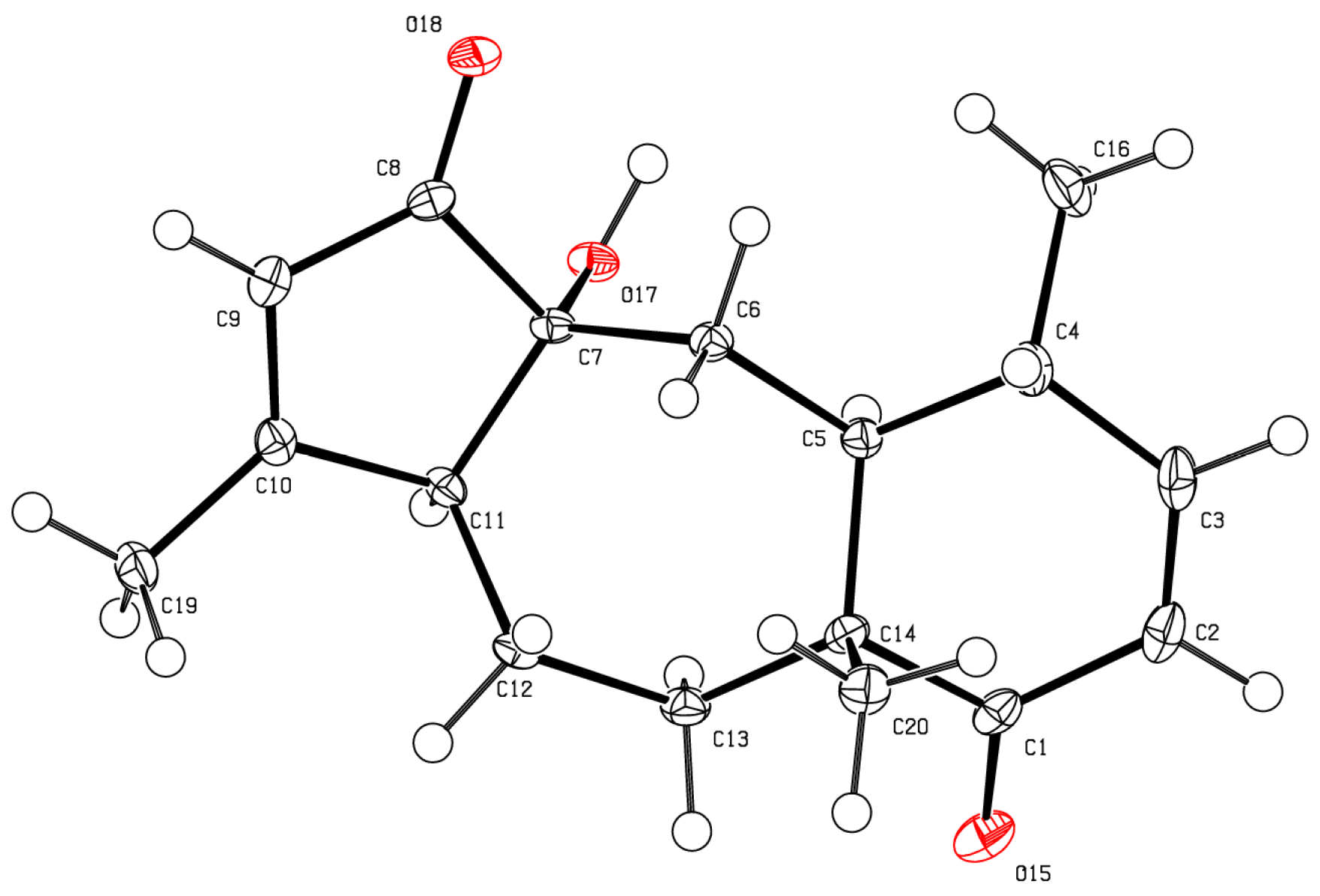
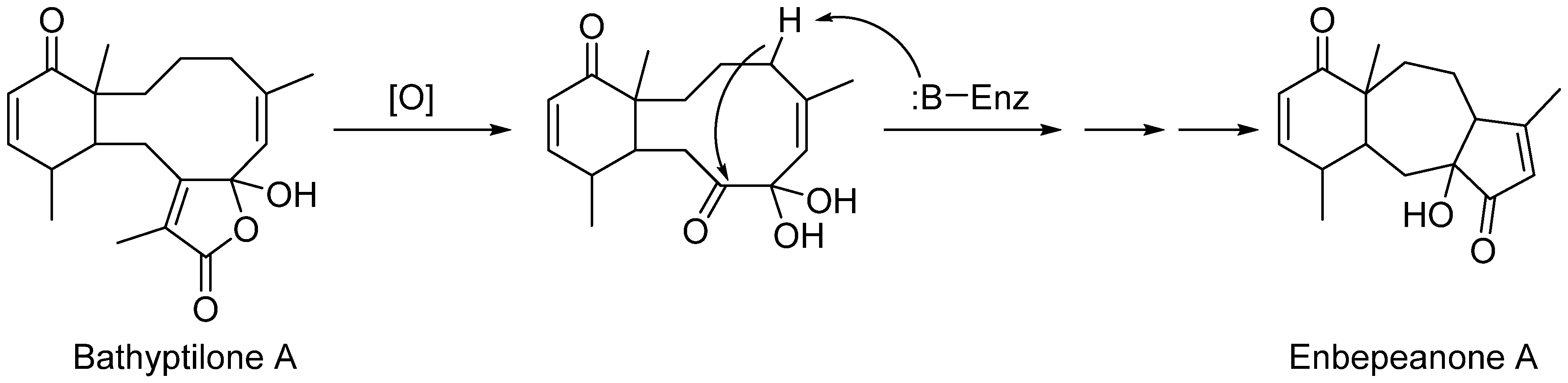
2. Results and Discussion
3. Experimental Section
3.1. General Experimental Procedure
3.2. Animal Material
3.3. Isolation of Bathyptilones and Enbepeanone
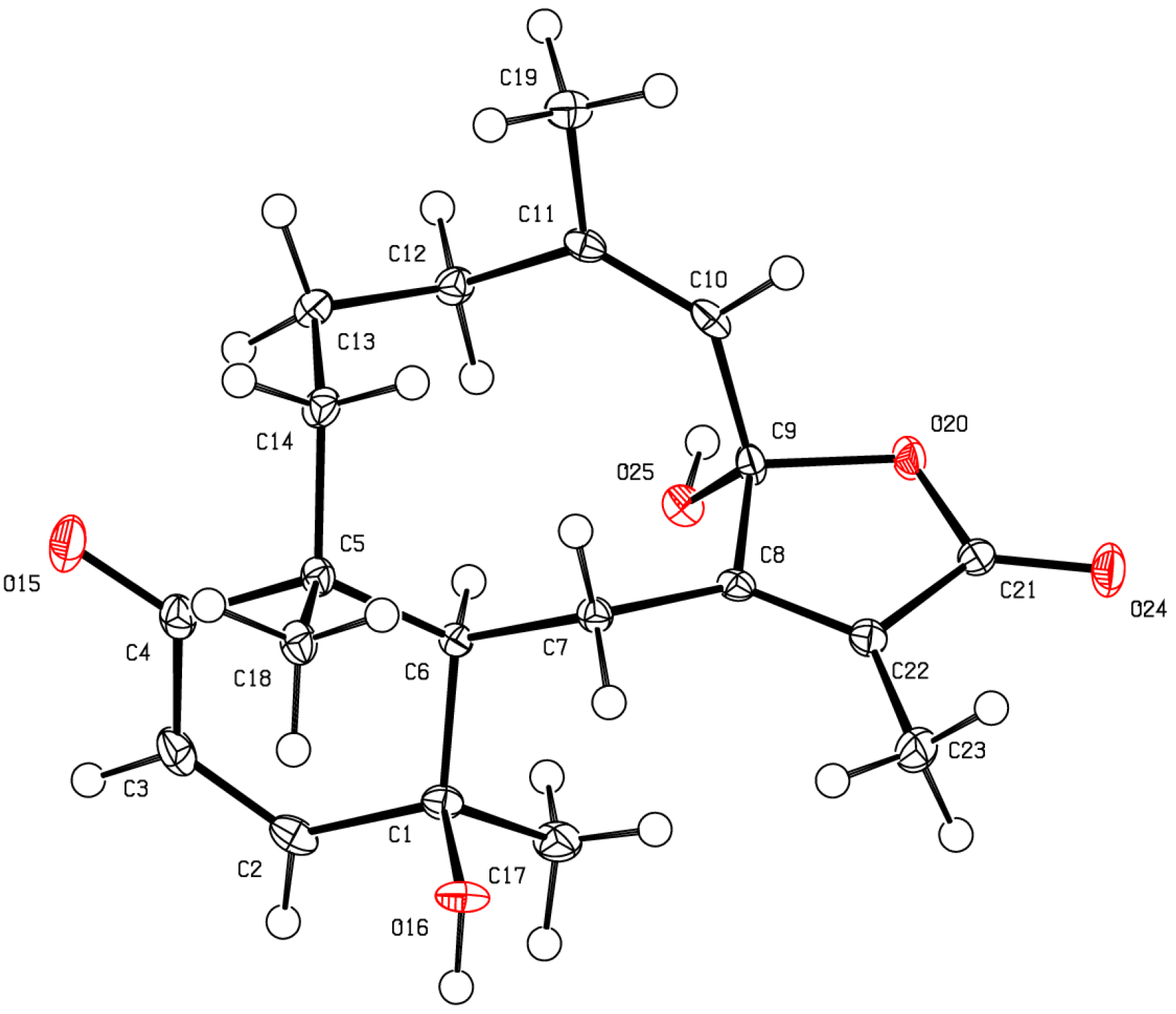
3.4. X-Ray Crystallography
3.5. Biological Assays
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Soldatou, S.; Baker, B.J. Cold-water marine natural products, 2006 to 2016. Nat. Prod. Rep. 2017, 34, 585–626. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2015, 32, 116–211. [Google Scholar] [CrossRef]
- Trimurtulu, G.; Faulkner, D.J.; Perry, N.B.; Ettouati, L.; Litaudon, M.; Blunt, J.W.; Munro, M.H.G.; Jameson, G.B. Alkaloids from the Antarctic sponge Kirkpatrickia varialosa. part 2: variolin A and N(3′)-methyl tetrahydrovariolin B. Tetrahedron 1994, 50, 3993–4000. [Google Scholar] [CrossRef]
- Perry, N.B.; Ettouati, L.; Litaudon, M.; Blunt, J.W.; Munro, M.H.G.; Parkin, S.; Hope, H. Alkaloids from the Antarctic sponge Kirkpatrickia varialosa. Tetrahedron 1994, 50, 3987–3992. [Google Scholar] [CrossRef]
- Diyabalanage, T.; Amsler, C.D.; McClintock, J.B.; Baker, B.J. Palmerolide a, a cytotoxic macrolide from the antarctic tunicate Synoicum adareanum. J. Am. Chem. Soc. 2006, 128, 5630–5631. [Google Scholar] [CrossRef] [PubMed]
- von Salm, J.L.; Witowski, C.G.; Fleeman, R.M.; McClintock, J.B.; Amsler, C.D.; Shaw, L.N.; Baker, B.J. Darwinolide, a new diterpene scaffold that inhibits methicillin-resistant Staphylococcus aureus biofilm from the Antarctic sponge Dendrilla membranosa. Org. Lett. 2016, 18, 2596–2599. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.A.L.; von Salm, J.L.; Clark, S.; Ferlita, S.; Nemani, P.; Azhari, A.; Rice, C.A.; Wilson, N.G.; Kyle, D.E.; Baker, B.J. Keikipukalides, furanocembrane diterpenes from the Antarctic deep sea octocoral Plumerella delicatissima. J. Nat. Prod. 2018, 81, 117–123. [Google Scholar] [CrossRef]
- Shilling, A.J.; von Salm, J.L.; Sanchez, A.R.; Kee, Y.; Amsler, C.D.; McClintock, J.B.; Baker, B.J. Anverenes B-E, new polyhalogenated monoterpenes form the Antarctic re alga Plocamium cartilagineum. Mar. Drugs 2019, 17, 230. [Google Scholar] [CrossRef]
- Knestrick, M.A.; Wilson, N.G.; Roth, A.; Adams, J.H.; Baker, B.J. Friomaramide, a highly modified linear hexapeptide from an Antarctic sponge Inflatella coelosphaeroides. J. Nat. Prod. 2019, 82, 2354–2358. [Google Scholar] [CrossRef]
- Putra, M.Y.; Wibowo, J.T.; Murniasih, T. A review of chemistry and biological activities of the Indonesian octocorallia. J. Appl. Pharm. Sci. 2017, 7, 219–227. [Google Scholar]
- Su, Y.D.; Su, J.H.; Hwang, T.L.; Wen, Z.H.; Sheu, J.H.; Wu, Y.C.; Sung, P.J. Briarane diterpenoids isolated from octocorals between 2014 and 2016. Mar. Drugs 2017, 15, 44. [Google Scholar] [CrossRef] [PubMed]
- Liaw, C.C.; Shen, Y.C.; Lin, Y.S.; Hwang, T.L.; Kuo, Y.H.; Khalil, A.T. Frajunolides E-K, briarane diterpenes from Junceella fragilis. J. Nat. Prod. 2008, 71, 1551–1556. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.F.; Di Su, Y.; Hwang, T.L.; Liao, Z.J.; Tsui, K.H.; Wen, Z.H.; Wu, Y.C.; Sung, P.J.; Schmidt, T.J. Briarenols C-E, new polyoxygenated briaranes from the octocoral Briareum excavatum. Molecules 2017, 22, 475. [Google Scholar] [CrossRef] [PubMed]
- Lei, H. Diterpenoids of gorgonian corals: chemistry and bioactivity. Chem. Biodivers. 2016, 13, 345–365. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; La, M.P.; Tang, H.; Sun, P.; Liu, B.S.; Zhuang, C.L.; Yi, Y.H.; Zhang, W. Chemistry and bioactivity of briaranes from the South China Sea gorgonian Dichotella gemmacea. Mar. Drugs 2016, 14, 201. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; La, M.-P.; Li, L.; Li, X.-B.; Tang, H.; Liu, B.-S.; Krohn, K.; Sun, P.; Yi, Y.-H.; Zhang, W. Bioactive 11,20-epoxy-3,5(16)-diene briarane diterpenoids from the South China Sea gorgonian Dichotella gemmacea. J. Nat. Prod. 2011, 74, 1658–1662. [Google Scholar] [CrossRef] [PubMed]
- Pham, N.B.; Butler, M.S.; Healy, P.C.; Quinn, R.J. Anthoptilides A-E, new briarane diterpenes from the Australian sea pen Anthoptilum cf. kukenthali. 2000, 318–321. [Google Scholar]
- Garcia-Fernàndez, J. The genesis and evolution of homeobox gene clusters. Nat. Rev. Genet. 2005, 6, 881–892. [Google Scholar] [CrossRef]
- Stern, M.; Gierse, A.; Tan, S.; Bicker, G. Human ntera2 cells as a predictive in vitro test system for developmental neurotoxicity. Arch. Toxicol. 2014, 88, 127–136. [Google Scholar] [CrossRef]
- Tharmarajah, L.; Samarakoon, S.R.; Ediriweera, M.K.; Piyathilaka, P.; Tennekoon, K.H.; Senathilake, K.S.; Rajagopalan, U.; Galhena, P.B.; Thabrew, I. In vitro anticancer effect of gedunin on human teratocarcinomal (Ntera-2) cancer stem-like cells. Biomed Res. Int. 2017, 2413197. [Google Scholar] [CrossRef]
- France, S.C.; Hoover, L.L. Analysis of variation in mitochondrial DNA sequences (ND3, ND4L, MSH) among Octocorallia (= Alcyonaria)(Cnidaria: Anthozoa). Bull. Biol. Soc. Wash. 2001, 10, 110–118. [Google Scholar]
- France, S.C.; Hoover, L.L. DNA sequences of the mitochondrial COI gene have low levels of divergence among deep-sea octocorals (Cnidaria: Anthozoa). Hydrobiologia 2002, 471, 149–155. [Google Scholar] [CrossRef]
- Trifinopoulos, J.; Nguyen, L.T.; von Haeseler, A.; Minh, B.Q. W-IQ-TREE: A fast online phylogenetic tool for maximum likelihood analysis. Nucl. Acids Res. 2016, 44, W232–W235. [Google Scholar] [CrossRef] [PubMed]
- Kalyaanamoorthy, S.; Minh, B.; Wong, T.K.; Von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nature Methods 2017, 14, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Hogan, R.I.; Hopkins, K.; Wheeler, A.J.; Allcock, A.L.; Yesson, C. Novel diversity in mitochondrial genomes of deep-sea Pennatulacea (Cnidaria: Anthozoa: Octocorallia). Mitochondrial DNA Part A 2019, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.C. The global diversity of sea pens (Cnidaria: Octocorallia: Pennatulacea). PLoS one 2011, 6, e22747. [Google Scholar] [CrossRef] [PubMed]
- Bruker. APEX3 User Manual; Bruker AXS Inc.: Madison, WI, USA, 2016. [Google Scholar]
- Bruker. SAINT v8.35a. Data Reduction Software; Bruker AXS Inc.: Madison, WI, USA, 2017. [Google Scholar]
- Sheldrick, G.M. SADABS program for empirical absorption correction. Ph.D. Thesis, University of Gottingen, Gottingen, Germany, 1996. [Google Scholar]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A 2015, A71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Phase annealing in shelx-90: direct methods for larger structures. Acta Crystallogr. Sect. A 1990, 46, 467–473. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of shelx. Acta Crystallogr. Sect. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal structure refinement with shelxl. Acta Crystallogr. Sect. C 2015, C71, 3–8. [Google Scholar]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Spek, A.L. Structure validation in chemical crystallography. Acta Crystallogr. Sect. D Biol. Crystallogr. 2009, 65, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Hooft, R.W.W.; Straver, L.H.; Spek, A.L. Determination of absolute structure using Bayesian statistics on Bijvoet differences. J. Appl. Crystallogr. 2008, 41, 96–103. [Google Scholar] [CrossRef] [PubMed]
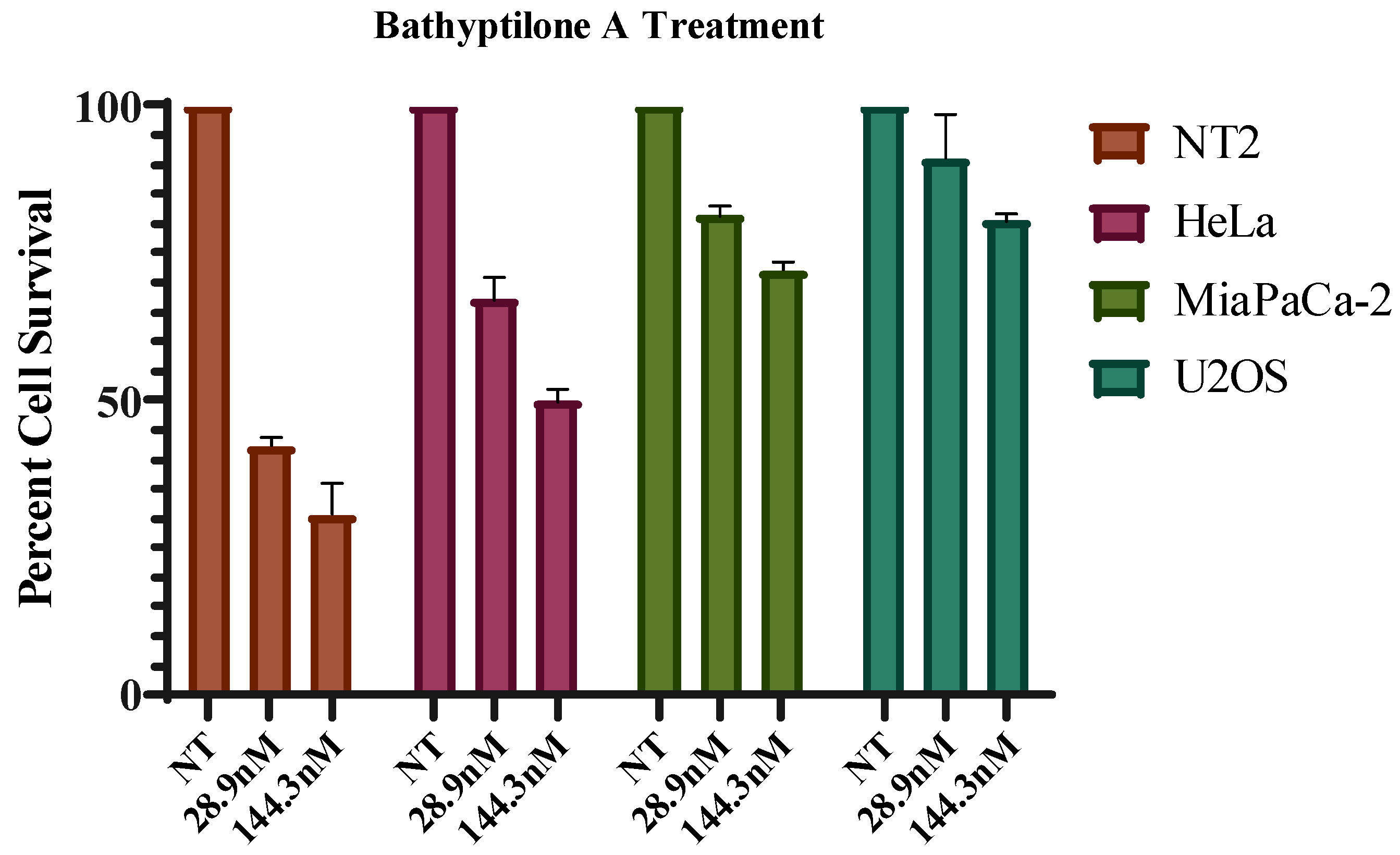
| Position | 13C, type | 1H (J in Hz) | HMBC |
|---|---|---|---|
| 1 | 50.2, C | ||
| 2 | 21.8, CH2 | a 1.54, m b 1.84, m | 14 1, 4 |
| 3 | 31.6, CH2 | a 1.52, m b 1.98, m | 2 1, 2, 4, 10, 15 |
| 4 | 29.5, CH2 | a 1.85, m b 3.52, br t | 5, 6 |
| 5 | 145.3, C | ||
| 6 | 124.1, CH | 5.35, s | 4, 5, 7, 16 |
| 7 | 106.2, C | ||
| 8 | 160.7, C | ||
| 9 | 22.7, CH2 | a 2.47, d (15.8) b 3.25, dd (9.5, 15.3) | 1, 8, 10, 11, 17 7, 8, 10, 11, 17 |
| 10 | 41.4, CH | 3.14, d (9.5) | 1, 8, 9, 11, 14, 15 |
| 11 | 69.3, C | ||
| 12 | 151.7, CH | 6.63, d (10.0) | 10, 11, 14, 20 |
| 13 | 127.4, CH | 5.97, d (10.0) | 1, 11 |
| 14 | 203.7, C | ||
| 15 | 24.2, CH3 | 1.16, s | 1, 2, 3, 10, 14 |
| 16 | 23.5, CH3 | 1.78, s | 4, 5, 6 |
| 17 | 125.8, C | ||
| 18 | 171.7, C | ||
| 19 | 9.1, CH3 | 1.95, s | 8, 17, 18 |
| 20 | 30.4, CH3 | 1.35, s | 10, 11, 12 |
| 7, 11-OH | 3.52, br |
| Position | Bathyptilone B (2) | Bathyptilone C (3) | ||
|---|---|---|---|---|
| C, type a,c | 1H (J in Hz) b | C, type | 1H (J in Hz) | |
| 1 | 52.5, C | 50.5, C | ||
| 2 | 77.9, CH | 5.89, dd (4.8, 12.9) | 31.5, CH2 | a 1.90, m b 1.56, m |
| 3 | 27.6, CH2 | a 3.93, dt (3.2, 14.7) b 1.89, m | 21.8, CH2 | a 1.95, m b 1.52, m |
| 4 | 26.3, CH2 | a 1.99, m b 2.32, m | 30.3, CH2 | a 2.60, td (13.5, 5.3) b 2.03, m |
| 5 | 146.0, C | 141.2, C | ||
| 6 | 122.8, CH | 5.26, s | 122.4, CH | 5.00, br d (8.6) |
| 7 | 106.3, C | 79.2, CH | 5.66, br d (8.2) | |
| 8 | 161.3, C | 159.4, C | ||
| 9 | 28.4, CH2 | a 3.64, m b 2.40, m | 22.4, CH2 | a 3.28, br dd (15.6, 9.7) b 2.34, br d (15.6) |
| 10 | 41.4, CH | 3.34, t (8.5) | 44.8, CH | 2.42, br d (9.3) |
| 11 | 38.0, CH | 2.45, m | 68.9, C | |
| 12 | 154.8, CH | 6.64, br d (10.3) | 151.2, CH | 6.62, br d (9.7) |
| 13 | 127.0, CH | 5.94, d (10.3) | 127.6, CH | 6.00, br d (9.7) |
| 14 | 201.4, C | 203.2, C | ||
| 15 | 16.7, CH3 | 1.11, s | 24.0, CH3 | 1.14, s |
| 16 | 23.2, CH3 | 1.77, s | 22.7, CH3 | 1.74, s |
| 17 | 124.3, C | 122.9, C | ||
| 18 | 171.7, C | 1.91, s | 163.9, C | |
| 19 | 9.1, CH3 | 9.1, CH3 | 1.94, s | |
| 20 | 18.8, CH3 | 1.14, d (6.5) | 30.8, CH3 | 1.27, s |
| 21 | 169.5, C | |||
| 22 | 21.2, CH3 | 2.03, s | ||
| Position | 13C, type | 1H (J in Hz) |
|---|---|---|
| 1 | 52.8, C | |
| 2 | 31.9, CH2 | 1.14, m 1.39, m |
| 3 | 19.5, CH2 | 1.20, m 1.30, m |
| 4 | 46.3, CH | 2.20, m |
| 5 | 177.5, C | |
| 6 | 133.2, CH | 5.85, s |
| 7 | 205.4, C | |
| 8 | 99.5, C | |
| 9 | 28.3, CH2 | 1.36, m 1.46, dd (12.4, 7.0) |
| 10 | 32.2, CH | 1.35, m |
| 11 | 36.1, CH | 2.08, m |
| 12 | 153.6, CH | 6.57, dd (10.9, 6.2) |
| 13 | 128.0, CH | 6.07, d (10.9) |
| 14 | 202.9, C | |
| 15 | 19.3, CH3 | 1.24, s |
| 16 | 25.7, CH3 | 2.09, s |
| 17 | 19.6, CH3 | 1.09, d (6.8) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thomas, S.A.L.; Sanchez, A.; Kee, Y.; Wilson, N.G.; Baker, B.J. Bathyptilones: Terpenoids from an Antarctic Sea Pen, Anthoptilum grandiflorum (Verrill, 1879). Mar. Drugs 2019, 17, 513. https://doi.org/10.3390/md17090513
Thomas SAL, Sanchez A, Kee Y, Wilson NG, Baker BJ. Bathyptilones: Terpenoids from an Antarctic Sea Pen, Anthoptilum grandiflorum (Verrill, 1879). Marine Drugs. 2019; 17(9):513. https://doi.org/10.3390/md17090513
Chicago/Turabian StyleThomas, Santana A.L., Anthony Sanchez, Younghoon Kee, Nerida G. Wilson, and Bill J. Baker. 2019. "Bathyptilones: Terpenoids from an Antarctic Sea Pen, Anthoptilum grandiflorum (Verrill, 1879)" Marine Drugs 17, no. 9: 513. https://doi.org/10.3390/md17090513
APA StyleThomas, S. A. L., Sanchez, A., Kee, Y., Wilson, N. G., & Baker, B. J. (2019). Bathyptilones: Terpenoids from an Antarctic Sea Pen, Anthoptilum grandiflorum (Verrill, 1879). Marine Drugs, 17(9), 513. https://doi.org/10.3390/md17090513






