Effect of Harvest Age on Forage Production and Silage Quality of Pearl Millet Hybrid in Cerrado Biome
Abstract
1. Introduction
2. Materials and Methods
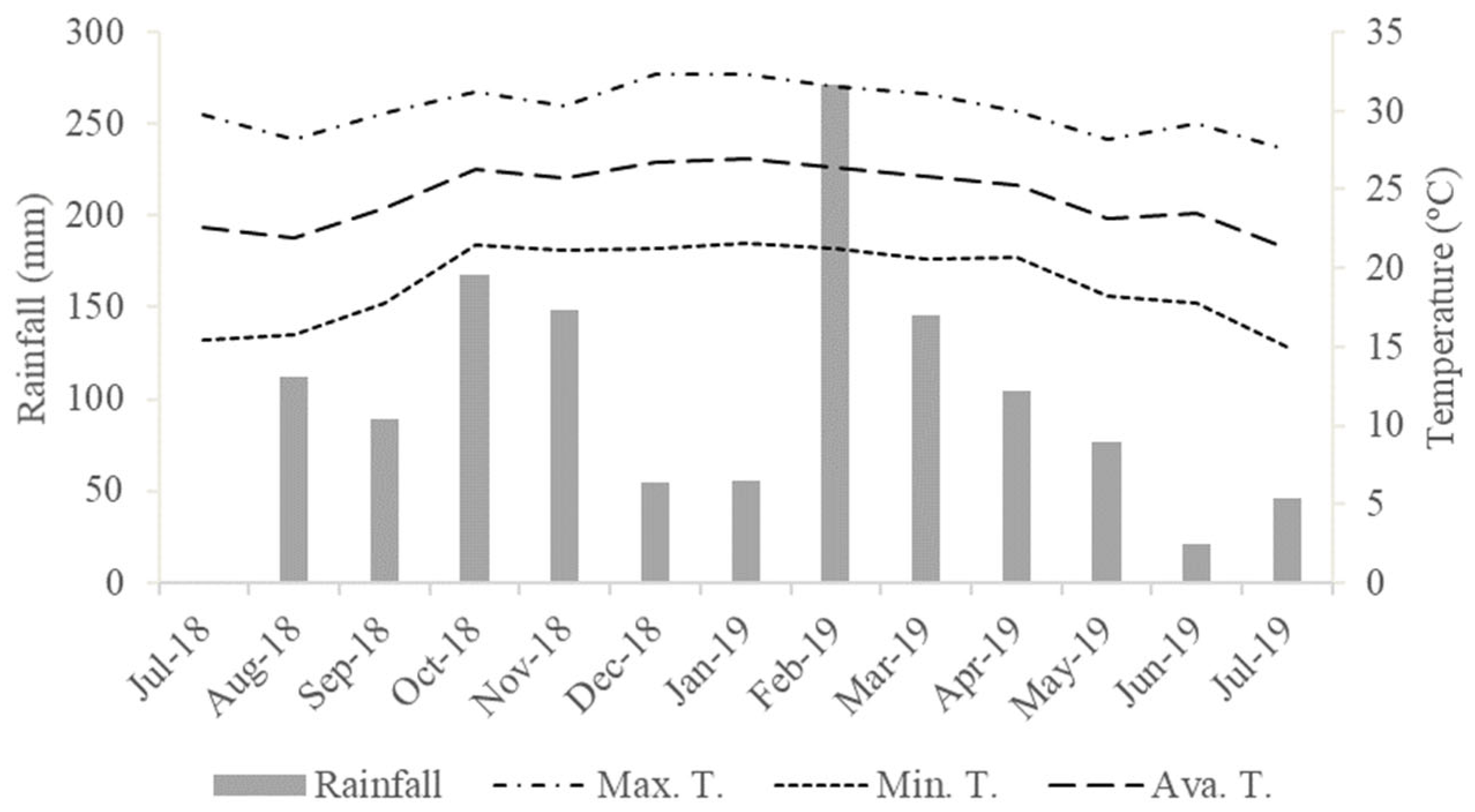
2.1. Location and Experimental Design
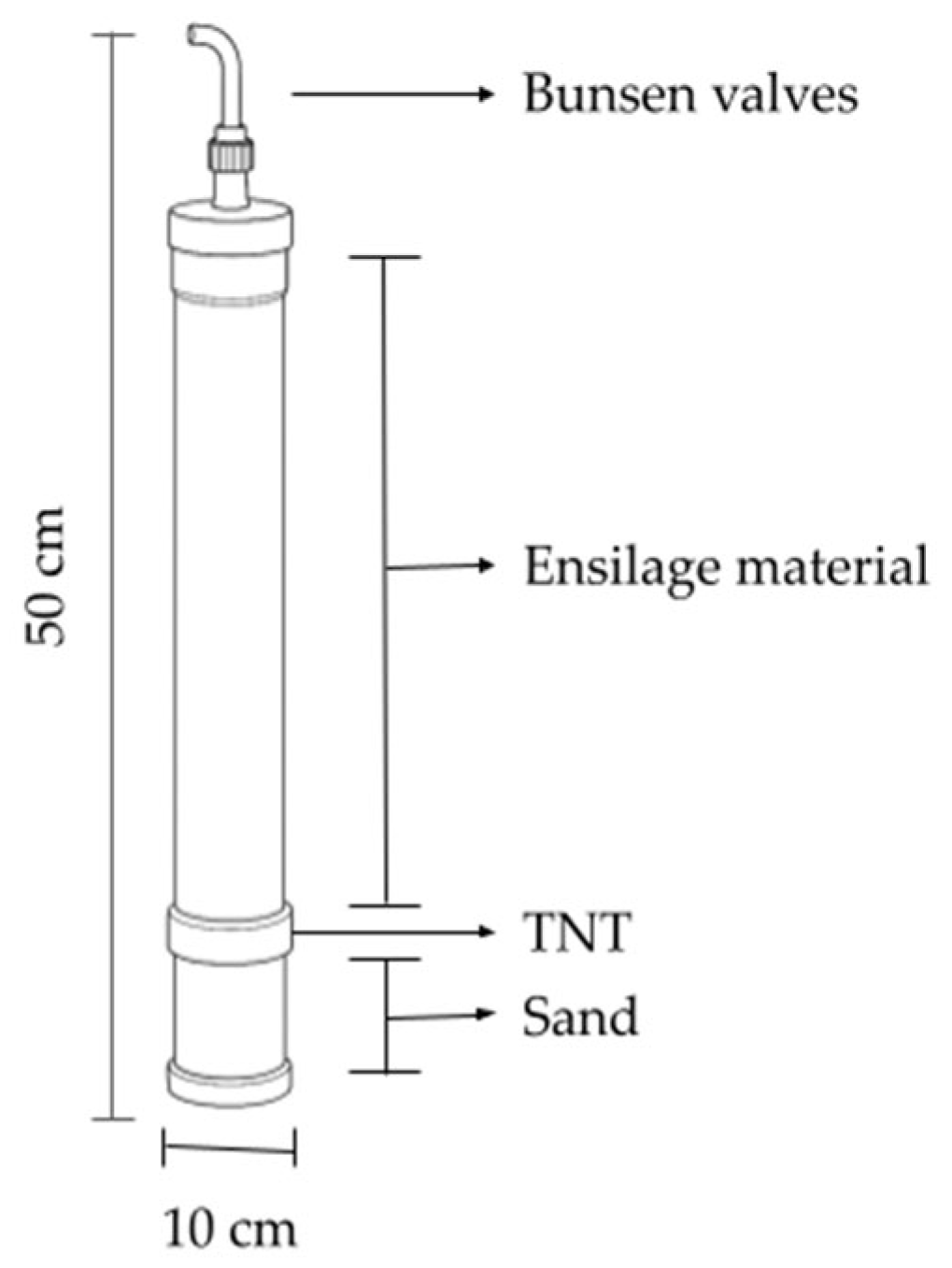
2.2. Forage Mass and Ensilage
2.3. Silage Fermentation Parameters and Density
2.4. Chemical Composition and In Vitro Digestibility
2.5. Statistical Analysis
3. Results
3.1. Forage Mass and Composition Before Ensilage
3.2. Fermentation Parameters of Silage
3.3. Chemical Composition and In Vitro DM Digestibility of Silage
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tarafdar, J.C.; Raliya, R.; Mahawar, H.; Rathore, I. Development of zinc nanofertilizer to enhance crop production in pearl millet (Pennisetum americanum). Agric. Res. 2014, 3, 257–262. [Google Scholar] [CrossRef]
- Basavaraj, G.; Parthasarathy, P.R.; Bhagavatula, S.; Ahmed, W. Availability and utilization of pearl millet in India. J. SAT Agric. Res. 2010, 8, 1–6. [Google Scholar]
- Nambiar, V.S.; Dhaduk, J.J.; Sareen, N.; Shahu, T.; Desai, R. Potential functional implications of pearl millet (Pennisetum glaucum) in health and disease. J. Appl. Pharm. Sci. 2011, 1, 62–67. [Google Scholar] [CrossRef]
- FAO. Unleashing the Potential of Millets—International Year of Millets; FAO: Rome, Italy, 2024. [Google Scholar] [CrossRef]
- Bandyopadhyay, T.; Muthamilarasan, M.; Prasad, M. Millets for next generation climate-smart agriculture. Front. Plant Sci. 2017, 8, 1266. [Google Scholar] [CrossRef]
- Amer, S.; Mustafa, A.F. Effects of feeding pearl millet silage on milk production of lactating dairy cows. J. Dairy Sci. 2010, 93, 5921–5925. [Google Scholar] [CrossRef]
- Guimarães, R., Jr.; Gonçalves, L.C.; Rodrigues, J.A.S.; Pires, D.A.A.; Jayme, D.G.; Rodriguez, N.M.; Saliba, E.O.S. Avaliação agronômica de genótipos de milheto (P. glaucum) plantados em período de safrinha. Arch. Zootec. 2009, 58, 629–632. [Google Scholar]
- Morales, J.U.; Alatorre, J.A.H.; Nieto, C.A.R.; Becerra, J.F.C. Forage production and nutritional concentration of silage from three varieties of pearl millet (Pennisetum glaucum) harvested at two maturity stages. J. Anim. Plant Sci. 2015, 27, 4161–4169. [Google Scholar]
- Silva, A.G.; Farias, O.L., Jr.; França, A.F.S.; Miyagi, E.S.; Rios, L.C.; Moraes Filho, C.G.; Ferreira, J.L. Rendimento forrageiro e composição bromatológica de milheto sob adubação nitrogenada. Ciênc. Anim. Bras. 2012, 13, 67–75. [Google Scholar] [CrossRef]
- Costa, R.R.G.F.; Costa, K.A.P.; Souza, W.F.; Epifanio, P.S.; Santos, C.B.; Silva, J.T.; Oliveira, S.S. Production and quality of silages pearl millet and paiaguas palisadegrass in monocropping and intercropping in different forage systems. Biosci. J. 2018, 34, 357–367. [Google Scholar] [CrossRef]
- Jobim, C.; Nussio, L.; Reis, A.R.; Schmidt, P. Avanços metodológicos na avaliação da qualidade da forragem conservada. Rev. Bras. Zootec. 2007, 36, 101–119. [Google Scholar] [CrossRef]
- Silva, D.J.; Queiroz, A.C. Análises de Alimentos: Métodos Químicos e Biológicos, 3rd ed.; UFV: Viçosa, Brazil, 2002. [Google Scholar]
- Bolsen, K.K.; Lin, C.; Brent, B.E.; Gadeken, D. Effect of silage additives on the microbial succession and fermentation process of alfalfa and corn silages. J. Dairy Sci. 1992, 75, 3066–3083. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of the Association of Official Analytical Chemists, 19th ed.; AOAC International: Gaithersburg, MD, USA, 2012. [Google Scholar]
- Mertens, D.R. Gravimetric determination of amylase-treated neutral detergent fiber in feeds with refluxing in beakers or crucibles: Collaborative study. J. AOAC Int. 2002, 85, 1217–1240. [Google Scholar] [CrossRef] [PubMed]
- Van Soest, P.J.; Robertson, J. Analysis of Forages and Fibrous Foods: A Laboratory Manual for Animal Science; Cornell University: Ithaca, NY, USA, 1985. [Google Scholar]
- Tilley, J.M.A.; Terry, R.A. A two-stage technique for the in vitro digestion of forage crops. J. Br. Grassl. Soc. 1963, 18, 104–111. [Google Scholar] [CrossRef]
- Holden, L.A. Comparison of methods of in vitro dry matter digestibility for ten feeds. J. Dairy Sci. 1999, 82, 1791–1794. [Google Scholar] [CrossRef] [PubMed]
- Simão, E.P.; Gontijo Neto, M.M.; Santos, E.A.; Wendling, I.J. Produção de biomassa e composição bromatológica de duas cultivares de milheto semeadas em diferentes épocas. Rev. Bras. Milho Sorgo 2015, 14, 196–206. [Google Scholar] [CrossRef]
- Muck, R.E. Factores influencing silage quality and their implications for management. J. Dairy Sci. 1988, 71, 2992–3002. [Google Scholar] [CrossRef]
- McDonald, P.; Henderson, A.R.; Heron, S.J.E. The Biochemistry of Silage, 2nd, ed.; Kingston: Chalcombe, UK, 1991. [Google Scholar]
- Van Soest, P.J. Nutritional Ecology of the Ruminant; Cornell University Press: Ithaca, Greece, 1994. [Google Scholar]
- Brunette, T.; Baurhoo, B.; Mustafa, A.F. Effects of replacing grass silage with forage pearl millet silage on milk yield, nutrient digestion, and ruminal fermentation of lactating dairy cows. J. Dairy Sci. 2016, 99, 269–279. [Google Scholar] [CrossRef]
- Tolentino, D.C.; Rodrigues, J.A.S.; Pires, D.A.A.; Veriato, F.T.; Lima, L.O.B.; Moura, M.M.A. The quality of silage of different sorghum genotypes. Acta Sci. Anim. Sci. 2016, 38, 143–149. [Google Scholar] [CrossRef]
- Dias, A.M.; Ítavo, L.C.V.; Ítavo, C.C.B.F.; Blan, L.R.; Gomes, E.N.O.; Soares, C.M.; Leal, E.S.; Nogueira, E.; Coelho, E.M. Ureia e glicerina bruta como aditivos na ensilagem de cana-de-açúcar. Arq. Bras. Med. Vet. Zootec. 2014, 66, 1874–1882. [Google Scholar] [CrossRef]
- Jacovetti, R.; França, A.F.S.; Carnevalli, R.A.; Miyagi, E.S.; Brunes, L.C.; Corrêa, D.S. Milheto como silagem comparado a gramíneas tradicionais: Aspectos quantitativos, qualitativos e econômicos. Ciênc. Anim. Bras. 2018, 19, e-26539. [Google Scholar] [CrossRef]
- Woolford, M.K. The Silage Fermentation; Marcel Dekker: New York, NY, USA, 1984. [Google Scholar]
- Sucu, E.; Kalkan, H.; Canbolat, O.; Filya, I. Effects of ensiling density on nutritive value of maize and sorghum silages. Rev. Bras. Zootec. 2016, 45, 596–603. [Google Scholar] [CrossRef]
- Silva, Y.A.; Orrico Júnior, M.A.P.; Retore, M.; Ceccon, G.; Fernandes, T.; Orrico, A.C.A. Chemical Composition, Fermentation Parameters and Losses of Silages From Different Hybrids of Biomass Sorghum. Grass Forage Sci. 2025, 80, e12706. [Google Scholar] [CrossRef]
- Muck, R.E.; Nadeau, E.M.G.; McAllister, T.A.; Contreras-Govea, F.E.; Santos, M.C.; Kung, L., Jr. Silage review: Recent advances and future uses of silage additives. J. Dairy Sci. 2018, 101, 3980–4000. [Google Scholar] [CrossRef] [PubMed]
- Machado, S.M.; Sales, E.C.J.; Rigueria, J.P.S.; Pires, D.A.A.; Silva, A.F.; Monção, F.P. Glicerina bruta melhora o valor nutricional de silagem de milheto colhido em duas idades. Agrarian 2019, 12, 204–213. [Google Scholar] [CrossRef]
- Adesogan, A.T.; Arriola, K.G.; Jiang, Y.; Oyebade, A.; Paula, E.M.; Pech-Cervantes, A.A.; Romero, J.J.; Ferraretto, L.F.; Vyas, D. Symposium review: Technologies for improving fiber utilization. J. Dairy Sci. 2019, 102, 5726–5755. [Google Scholar] [CrossRef]
| Item | Harvest Age (Days) | SEM | p-Value | Equation (R2) | ||||
|---|---|---|---|---|---|---|---|---|
| 75 | 85 | 95 | 105 | L | Q | |||
| Forage mass (kg/ha) | ||||||||
| Green mass | 33,114 | 48,574 | 52,581 | 65,980 | 881.65 | <0.001 | 0.792 | Y = −42280.677 + 1026.0273x (R2 = 0.73) |
| Dry mass | 6666 | 9851 | 11,372 | 15,569 | 402.72 | <0.001 | 0.544 | Y = −14544.065 + 282.3157x (R2 = 0.81) |
| CP concentration | 1071 | 1284 | 1340 | 1650 | 102.0 | <0.001 | 0.235 | Y = −989.845 + 25.5632x (R2 = 0.86) |
| Chemical composition | ||||||||
| Dry matter (%) | 16.70 | 19.85 | 21.80 | 23.50 | 0.31 | <0.001 | 0.565 | Y = 0.601 + 0.2056x (R2 = 0.94) |
| Organic matter (%DM) | 94.00 | 94.20 | 94.30 | 94.50 | 0.17 | 0.387 | 0.899 | - |
| Crude protein (%DM) | 15.10 | 14.00 | 11.80 | 10.90 | 0.20 | <0.001 | 0.487 | Y = 31.027 − 0.1923x (R2 = 0.93) |
| Ether extract (%DM) | 3.1 | 3.2 | 3.5 | 3.3 | 0.04 | 0.425 | 0.252 | - |
| NDF (%DM) | 58.90 | 60.80 | 62.30 | 63.20 | 0.13 | <0.001 | 0.367 | Y = 52.524 + 0.2092x (R2 = 0.91) |
| NFC (%DM) | 16.90 | 16.30 | 16.70 | 17.20 | 0.10 | 0.275 | 0.325 | - |
| IVDMD (%DM) | 72.30 | 70.10 | 67.30 | 65.60 | 0.37 | <0.001 | 0.207 | Y = 91.027 − 0.2375x (R2 = 0.90) |
| Item | Harvest Age (Days) | SEM | p-Value | Equation (R2) | ||||
|---|---|---|---|---|---|---|---|---|
| 75 | 85 | 95 | 105 | L | Q | |||
| pH | 4.25 | 3.92 | 3.76 | 3.60 | 0.009 | <0.001 | 0.384 | Y = 6.159 − 0.0252x (R2 = 0.95) |
| NH3-N (% TN) | 2.05 | 1.75 | 1.49 | 1.22 | 0.090 | 0.003 | 0.987 | Y = 3.449 + 0.0197x (R2 = 0.86) |
| Total DM loss (% DM) | 15.52 | 12.54 | 10.31 | 5.82 | 0.415 | <0.001 | 0.563 | Y = 40.034 − 0.3133x (R2 = 0.98) |
| Effluent loss (kg/ton) | 43.25 | 39.90 | 34.80 | 31.05 | 2.140 | <0.001 | 0.464 | Y = 72.627 − 0.3967x (R2= 0.95) |
| Gas loss (% DM) | 8.50 | 7.04 | 5.25 | 2.82 | 0.234 | <0.001 | 0.056 | Y = 24.063 − 0.2023x (R2 = 0.96) |
| DM recovery (%) | 80.20 | 85.62 | 87.00 | 95.50 | 0.281 | <0.001 | 0.185 | Y = 38.537 + 0.5339x (R2 = 0.93) |
| Density fresh mass (kg/m3) | 770.00 | 695.80 | 656.50 | 620.10 | 7.896 | <0.001 | 0.116 | Y = 1135.951 − 5.0623x (R2 = 0.89) |
| Density dry mass (kg/m3) | 145.00 | 143.00 | 143.80 | 145.70 | 2.652 | 0.356 | 0.265 | - |
| Item | Harvest Age (Days) | SEM | p-Value | Equation (R2) | ||||
|---|---|---|---|---|---|---|---|---|
| 75 | 85 | 95 | 105 | L | Q | |||
| Dry matter (%) | 18.83 | 20.56 | 21.90 | 23.50 | 0.200 | 0.002 | 0.181 | Y = 4.157 + 0.1925x (R2 = 0.87) |
| Organic matter (% DM) | 94.40 | 94.60 | 94.00 | 94.70 | 0.109 | 0.052 | 0.194 | - |
| Crude protein (%DM) | 14.00 | 13.30 | 11.10 | 10.20 | 0.118 | <0.001 | 0.716 | Y = 27.744 − 0.1793x (R2 = 0.95) |
| NDF (% DM) | 59.80 | 62.00 | 64.40 | 66.30 | 0.338 | <0.001 | 0.458 | Y = 25.702 + 0.4085x (R2 = 0.90) |
| ADF (%DM) | 34.00 | 36.70 | 39.60 | 41.20 | 0.383 | <0.001 | 0.164 | Y = 09.032 + 0.3383x (R2 = 0.90) |
| Ether extract (%DM) | 3.20 | 3.15 | 3.10 | 3.10 | 0.080 | 0.409 | 0.926 | - |
| NFC (%DM) | 16.90 | 15.70 | 15.40 | 14.90 | 0.196 | <0.001 | 0.739 | Y = 31.019 − 0.2167x (R2 = 0.92) |
| IVDMD (%DM) | 77.30 | 75.20 | 72.60 | 70.50 | 0.384 | <0.001 | 0.375 | Y = 115.801 − 0.4337x (R2 = 0.92) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Silva, A.P.; Dias, A.M.; Candido, A.R.; Gomes, E.N.O.; Batistoti, J.O.; Ítavo, L.C.V.; Santos, G.T.d.; dos Santos Difante, G.; Coelho, E.M. Effect of Harvest Age on Forage Production and Silage Quality of Pearl Millet Hybrid in Cerrado Biome. Grasses 2025, 4, 34. https://doi.org/10.3390/grasses4030034
da Silva AP, Dias AM, Candido AR, Gomes ENO, Batistoti JO, Ítavo LCV, Santos GTd, dos Santos Difante G, Coelho EM. Effect of Harvest Age on Forage Production and Silage Quality of Pearl Millet Hybrid in Cerrado Biome. Grasses. 2025; 4(3):34. https://doi.org/10.3390/grasses4030034
Chicago/Turabian Styleda Silva, Amarildo Pedro, Alexandre Menezes Dias, Anderson Ramires Candido, Eva Nara Oliveira Gomes, Juliana Oliveira Batistoti, Luís Carlos Vinhas Ítavo, Geraldo Tadeu dos Santos, Gelson dos Santos Difante, and Elson Martins Coelho. 2025. "Effect of Harvest Age on Forage Production and Silage Quality of Pearl Millet Hybrid in Cerrado Biome" Grasses 4, no. 3: 34. https://doi.org/10.3390/grasses4030034
APA Styleda Silva, A. P., Dias, A. M., Candido, A. R., Gomes, E. N. O., Batistoti, J. O., Ítavo, L. C. V., Santos, G. T. d., dos Santos Difante, G., & Coelho, E. M. (2025). Effect of Harvest Age on Forage Production and Silage Quality of Pearl Millet Hybrid in Cerrado Biome. Grasses, 4(3), 34. https://doi.org/10.3390/grasses4030034








