Abstract
Cerebral ischemia represents a particular condition among neurological diseases due to its high frequency, high associated mortality, and the permanent disability in patients that survive it. Numerous studies in animal models have demonstrated the protective properties of resveratrol against cerebral ischemia. Resveratrol is a soluble molecule in polar solvents with high membrane permeability; however, it is rapidly metabolized at the liver and is also a substrate of the ATP binding cassette transporters located at the blood–brain barrier. These circumstances reduced bioavailability of resveratrol to the brain. In this review, we examined nasal resveratrol’s formulations including nanocarriers such as nanostructured lipid carriers, nanoemulsions, nanoparticles, bilosomes, cubosomal, and transferosomes that are directly transported to the brain. An intranasal administration route evades resveratrol transformation due to liver metabolism. Components of nanoformulations increased resveratrol absorption to the brain by enhancing permeation through specific approaches and also maintaining stability during storage. Both characteristics improved the delivery of resveratrol with conserved antioxidant capacity and protective properties for neurological models. Although demonstration that the nanoformulations prevents resveratrol’s blood–brain barrier retention is missing, properties of resveratrol’s nanoformulation encourage testing in clinical trials; however, regulatory approval for a novel nanocarrier in nasal drug delivery is complicated and needs approval.
1. Introduction
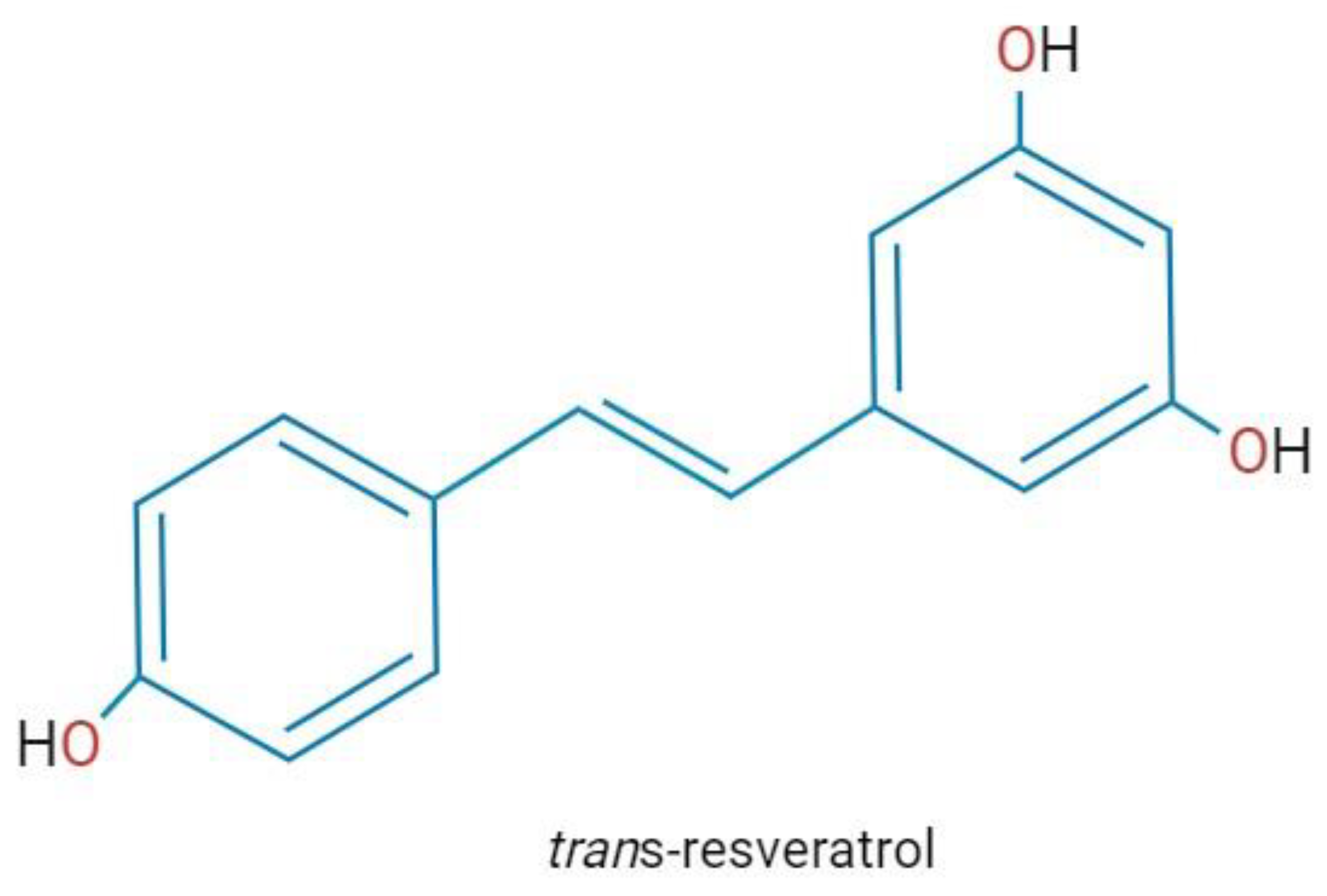
Resveratrol (3, 5, 40-trihydroxystilbene) is a natural polyphenol produced in many plants (e.g., grapes, peanuts, and hops) as a defense molecule against microbial infections and physical injury [1] (Figure 1). In plants, the content of resveratrol is limited, therefore, to achieve commercial purposes, it has been obtained from Japanese knotweed and grape skins and seeds, which has been reported to have a high content (ranging from 2 to 13 mg/L). Also, due to resveratrol’s multiple pharmacological activities found in humans, alternative methods to obtain larger amounts of resveratrol have been implemented, like chemical synthesis and biotechnological approaches, such as tissue culture and genetic engineering [2].
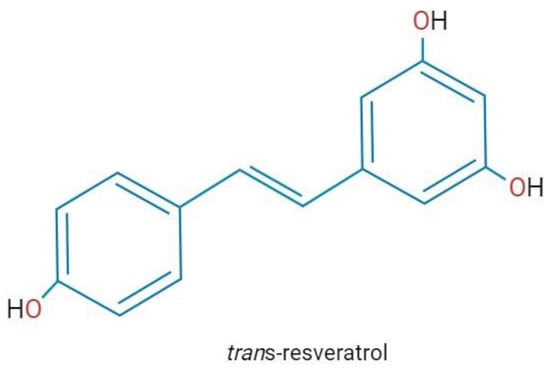
Figure 1.
Chemical structure of resveratrol. Created with BioRender.com; accessed on 25 January 2023.
Stroke is placed as the second leading cause of global death, with a high number of survivors being permanently incapacitated. Interestingly, the statistics have been changing: stroke was considered a disease of the developed world, nevertheless, the burden of stroke seems to be shifting to the developing countries, somewhat due to poor community information of stroke notice signs (which is the main cause of delay in arrival to the hospital). A wide range of stroke risk factors have been recognizably responsible for 90% of all strokes, among them, age is the strongest determinant, since age doubles the risk of stroke every decade above the age of 55 years. Regrettably, individuals who survive stroke live with a profound effect on health-related quality of life [3]. Generally, stroke is classified as an ischemic stroke, caused by interruption of the blood supply to a specific region of the brain (the most common type) and hemorrhagic stroke, attributed to the rupture of a blood vessel. In an ischemic stroke, the reduction of the steady blood flow provokes hypoxia and hypoglycemia that leads to severe affection to brain cell function. Typical treatment for ischemic stroke is the restoration of blood flow/oxygenation to the brain in a timely fashion by thrombolysis, using the intravenous recombinant tissue-type plasminogen activator, or by mechanical thrombectomy [4]. Paradoxically, reperfusion therapy after a prolong period of ischemia (>3 h) often leads to further cerebral damage. Ischemia and reperfusion activates a cascade of events including excitotoxicity, driven by the excessive activation of N-methyl-D-Aspartate glutamate receptors; oxidative stress, associated with mitochondrial dysfunction and the increase of reactive oxygen species production; the inflammatory response, which can cause damage due to high concentrations of inflammatory mediators; autophagy, a type of cell death induced by organelle damage and extracellular injury stimulation; and the blood–brain barrier (BBB) breakdown process, initiated by the deterioration of thigh junction proteins at the endothelial cells and aggravated by inflammation [5,6,7].
Unfortunately, establishing an effective treatment for stroke has been unfavorable; therefore, it is indispensable to continue the study of compounds that alleviate the damage induced in animal models in future clinical studies.
The ineffectiveness of pharmacological therapies in the clinical setting may stem from the omission of significant comorbidities, such as aging, in experimental models. Notably, the limitations of drugs exhibiting protective effects in the preclinical phase involve issues of bioavailability and challenges in crossing the BBB. Considering that the majority of strokes occur in elderly patients [8], it is crucial to recognize that drug absorption in the brain undergoes age-related changes. For instance, a key transport mechanism frequently employed for drug delivery to the brain, transcytosis through the transferrin receptor, is compromised during the aging process [9]. Additionally, in murine models of aging, an observed increase in certain efflux transporters poses a significant obstacle to the efficient delivery of drugs to the brain [10]. Not surprisingly, the combination of these mechanisms leads to dramatically low net delivery fluxes of resveratrol to the brain. This is particularly relevant when attempting to reverse brain damage following cerebral infarction in patients undergoing aging processes.
Therefore, it is imperative to develop a biocompatible formulation that operates autonomously from the expression of endogenous transport systems and can effectively traverse natural barriers, even in the presence of aging processes. For that reason, the design of intranasal formulations of materials that fusion with membranes is currently seen as a promissory alternative for its use in clinical studies; this route allows direct transport to the brain from the nasal cavity along the olfactory and trigeminal nerves, hence, it is a critical step that will facilitate the delivery of the drugs to the brain.
2. Resveratrol’s Protective Effect in Cerebral Ischemia
Strong evidence has suggested that resveratrol has a neuroprotective role in ischemic stroke in experimental models. This effect was confirmed by a meta-analysis performed in 54 studies with high quality scores [11]. The main mechanisms of neuroprotection in the ischemic stroke were found to be six biological activities: promoting angiogenesis and neurogenesis, inhibiting neuroinflammation, reducing oxidative stress, improving metabolic adaptations, and alleviating brain edema [11,12,13,14]. Interestingly, the meta-analysis showed that the factor inducing heterogeneity that impacts the reduction in the neurobehavioral score after the treatment was the dosage of resveratrol (the greatest effect was obtained with 20 and 50 mg/kg) while the influence factor in decline, the infarct volume, was the delivery route. The intraperitoneal treatment presented a superior effect than intravenous and oral; the intravenous treatment accomplished greater efficacy than oral possibly due to the increased bioavailability of resveratrol. Remarkably, no significant differences were found neither between the effect for a single treatment and daily treatment, nor the administration timing (ranging from 30 days before ischemia onset to 3 days after ischemia onset) proposing that resveratrol has an extended therapeutic time window [11].
Importantly, the toxic effects of resveratrol are minimal, showing mild and sporadic symptoms in less of the 30% of the individuals studied. Healthy volunteers that were given oral supplementation of resveratrol in doses from 25 to 150 mg at intervals of 4 h for 48 h reported minimal negative effects (e.g., headaches, dizziness, and ependymitis); other cases with negative effects were postmenopausal women after 1 g for 12 weeks, that reported diarrhea and increased total cholesterol [15]. This resveratrol low toxicity combined with high effectively alleviating and preventing the damage induced by cerebral ischemia reinforces the importance to initiate clinical studies in stroke patients.
3. Resveratrol Bioavailability
Resveratrol is a molecule with low aqueous solubility ranging from 0.05 mg/mL in water to 374 mg/mL in polyethylene glycol 400 (PEG-400), and poor stability dependent on temperature, light, and pH; it is relatively stable above pH 6 and has maximum degradation at pH 9 [16,17]. Resveratrol is a soluble molecule in polar solvents such as ethanol and DMSO; it has a high membrane permeability (estimated by its log P of 3.1) which provides a transport mechanism by transepithelial diffusion.
Resveratrol is defined as a drug class II according to the Biopharmaceutical Classification System; this feature allows for this molecule to be absorbed at the intestine and passed to the bloodstream speedily after oral administration. For instance, over 70% of a 25-mg oral dose is efficiently absorbed, resulting in a peak plasma concentration of 491 ± 90 ng/mL (approximately 2 μM) around 1 h after administration [18].
Regrettably, it is rapidly metabolized by glucuronidation and conjugation of sodium sulphate with the phenolic groups and hydrogenation of the aliphatic double bond at liver, leaving around 1% of free resveratrol in plasma. The recorded plasma half-life of resveratrol is 9.2 ± 0.6 h, marking the duration of its efficacy in the bloodstream [18]. The resveratrol’s free form can bound to blood proteins in a non-covalent manner (the mean plasma protein binding is 98.3%) [17]. Then, resveratrol enters cells reaching sub-micromolar levels depending on the administered dose. In humans, a dose of 25 mg reaches a peak of 10 ng/mL in plasma, which increases 7-fold (72 ng/mL) when the dose is increased 20-fold (500 mg) [19]. Additionally, only approximately 2% of the plasmatic resveratrol crosses the BBB [15]. This situation limits resveratrol health benefits since high doses are required to reach adequate therapeutic effect in the brain.
4. Resveratrol Delivery to the Brain
Despite all the evidence of the neuroprotective effects of resveratrol, its low bioavailability is still a great limitation to its successful use as co-adjuvant in the therapy of neurologic diseases. Thus, it is critical to explore the mechanisms associated with its transport into the central nervous system (CNS). Previous studies have demonstrated that resveratrol reaches the brain of rodents and humans [20,21]. Evidence is based on resveratrol detection in tissue homogenates of the brain, which indeed suggest that resveratrol must be able to cross the BBB, although the low level detection also suggests that it must overcome some obstacles that suffer drugs delivered to the CNS.
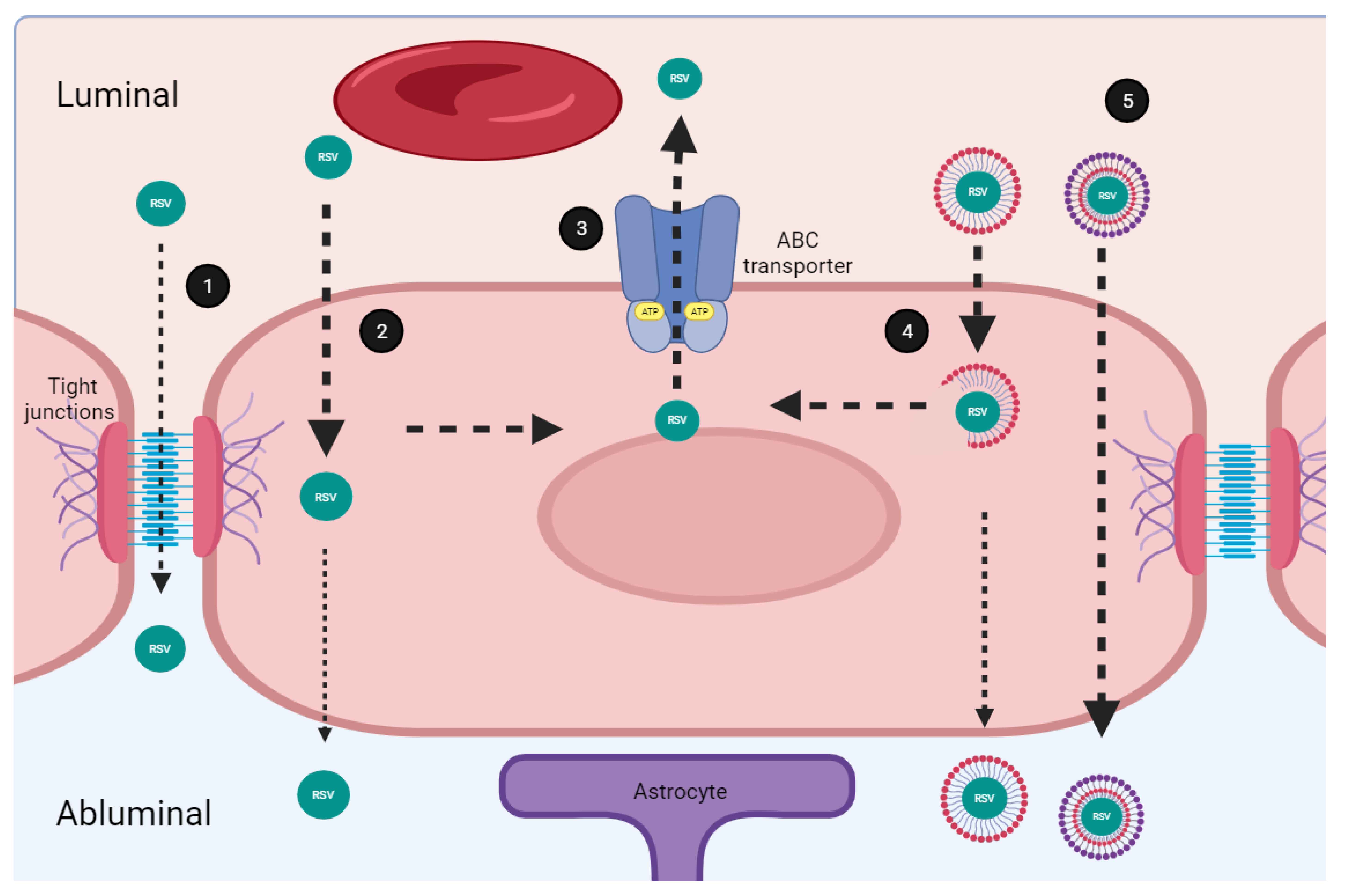
The tight junctions expressed in the brain micro endothelial cells that integrate the BBB are responsible for the severe restriction of the paracellular diffusional pathway even for ions and other polar solutes [22]. Despite this situation, small hydrophobic molecules with a molecular weight of less than 500 Daltons (less than 1 nm) transmigrate paracellularly into the neuroparenchyma. We calculated the resveratrol’s radius of gyration using the Python program; we found that it is around 6.60 Å with a maximum diameter of 13.12 Å (1.31 nm). This data indicates that the molecular size of resveratrol impedes its spread through the paracellular route easily. Nevertheless, molecules with high lipid solubility might use the transcellular route, which allows for the effective penetration of the membrane of the endothelial cells [23], unfortunately, this route has drawbacks that resveratrol can’t overcome (Figure 2).
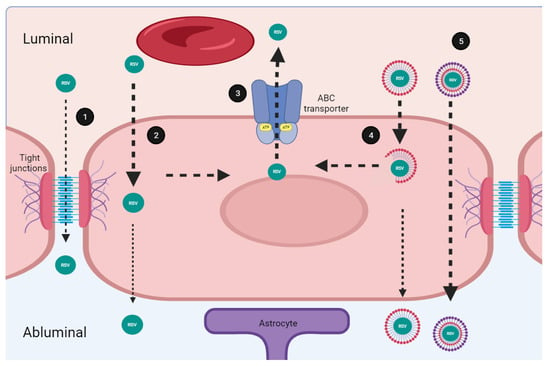
Figure 2.
Transport mechanisms of resveratrol through the brain blood barrier. (1) Small molecules (≤1 nm) cross the blood–brain barrier (BBB) by passive transport, through the paracellular route. Resveratrol (RSV) partially crosses the BBB through this route. (2) Molecules with high lipid solubility might use the transcellular route which allows for the effective passive penetration of endothelial cells membrane. RSV is soluble in polar solvents, and highly permeable in membrane which facilitates its transportation through this route. (3) Micro endothelial cells express on their luminal side several multi drug resistance transporters (ABC, ATP-binding cassette transporters). The ABC transporters mediate the efflux of RSV decreasing its uptake rate. (4) Nano formulations consist of a polymeric system. The molecular composition impacts solubility and improves RSV passage through the BBB. (5) Some components of nanoformulations such as Polyethylene glycol 1000 and the vitamin E succinate inhibit the ABC transporters. Improved formulations overcome restrictions associated to the BBB facilitating RSV transport. Created with BioRender.com; accessed on 25 January 2023.
The micro endothelial cells express on their luminal side (which faces to the bloodstream) several multi drug resistance transporters called ABC energy-dependent efflux transporters (ABC, ATP-Binding Cassette transporters). The ABC proteins are a family of membrane transporters that mediates the efflux of a wide variety of circulating substrates decreasing the uptake rate of potential drugs [24,25]. Importantly, lipophilic substances of low molecular weight tend to be substrates for some of the best-known ABC type transporters such as the P-glycoprotein, the breast cancer resistance protein (BCRP), and the multidrug resistance-associated protein 3 (MRP3) which are expressed in the BBB [26,27]. Interestingly, experiments using a knockout mouse for the P-glycoprotein showed that the oral administration of resveratrol was not affected; in contrast, the absence of the BCRP and the MRP3 modifies the oral bioavailability and distribution of resveratrol in the brain [28]. These results confirm that resveratrol is an important substrate for ABC transporters, so they could frustrate its passage through the BBB, decreasing the uptake rate, and preventing resveratrol from reaching the brain. Notwithstanding, other barriers, such as the blood-cerebrospinal fluid barrier established by the choroid plexus, could facilitate the entry of resveratrol through the BCRP/ABCG2 transporter [29]. ABCG2 localizes at the abluminal membrane of choroid plexus epithelial cells facing the cerebral spinal fluid, and facilitates resveratrol transport from the blood forward the brain tissue [30,31,32]. This is an opposite distribution to that found in the BBB, where the BCRP/ABCG2 localize at the luminal side of endothelial cells and therefore restrain the access of the drugs to the CNS [33,34].
The transcellular pathway is a regular physiological route that seems to be the most promising route in passing resveratrol to reach the brain; however, there are no formulations capable of effectively bypassing the defensive barriers; therefore, other approaches to transport resveratrol should be explored.
5. Nose Structure and Functions
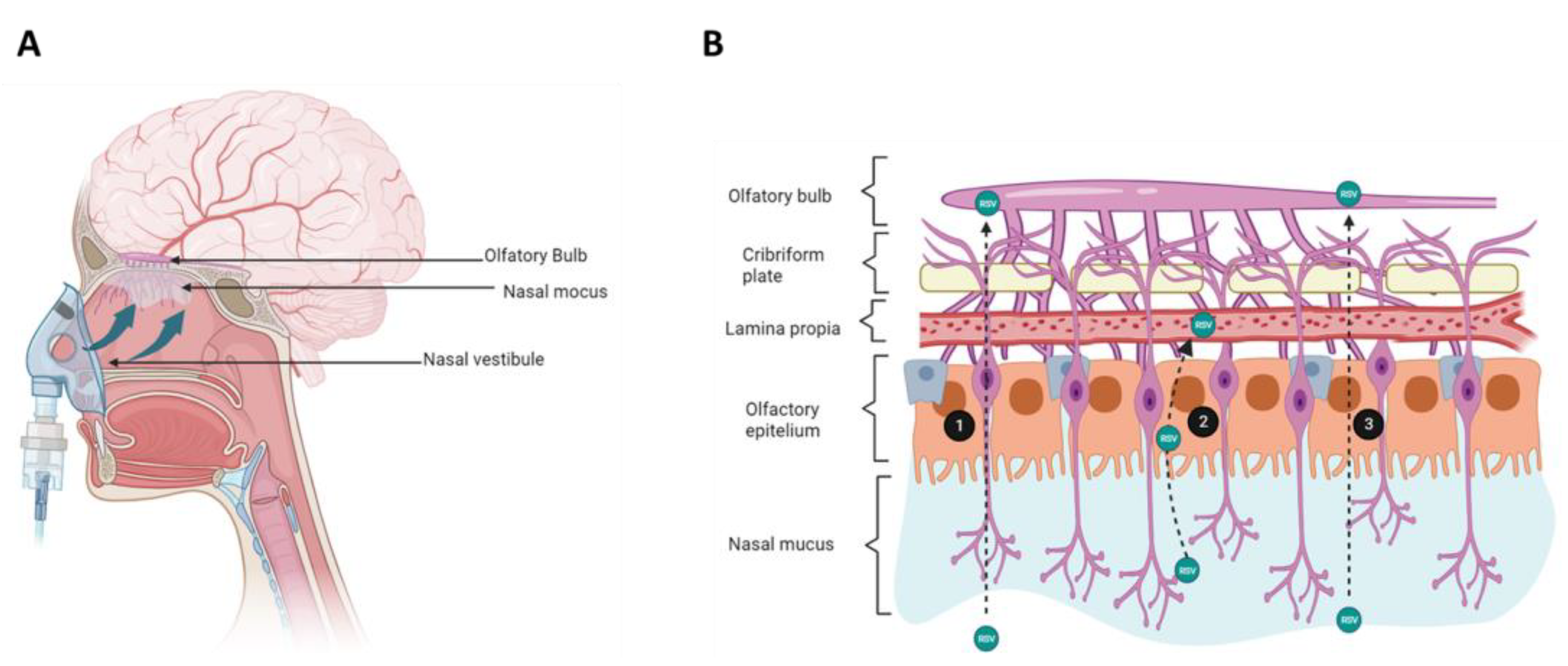
The nose is one of the sensory organs that, through smell, allow animals to relate to the environment. It fulfills the essential function of respiration, through the inhalation of air, activity that also involves a defense against invasion by pathogens, toxins, dust, or gases. The nose also works by humidifying and heating the inspired air to settle body temperature. Furthermore, the nose recognizes more than 10,000 different odors, and fulfills a complementary function for the sound of the voice. In its external part, the nose is made up of bone, cartilage, and skin, components that form an appendage on the face that ends in two holes, through which the air enters. Inside, the nasal cavity is found with a surface area of 150–200 cm2, which represents a volume of 15–20 mL. The nasal septum symmetrically divides the nasal cavity into two chambers (the nostrils) which are completely covered by a mucous layer. The nasal cavities are further divided into three regions. The vestibular region, which is the most anterior region that directly connects the inside of the nose with the environment; in this region, the nasal hairs filter inhaled particles. The vestibular region is composed mainly of squamous cells, occupies a total surface area of 0.6 cm2, and is poorly vascularized. For its part, the respiratory region covers the lateral walls of the nasal cavities. It contains four cell types: globet cells, responsible for producing mucus, basal cells, and ciliated and non-ciliated cells. It has an extension of approximately 130 cm2, so it has the largest area and the most highly vascularized. Likewise, it is also innervated by the maxillary branch of the trigeminal nerve. The last region, known as the olfactory region, is located at the top of the nasal cavity, covering the lower surface of the cribriform plate of the entomoid bone. The extent of this region is variable depending on the species, in humans it can cover 10%, while in rodents it can occupy up to 50%. The cell types found in this area are basal cells, which function as progenitors for other cell types, support cells, and trigeminal and olfactory neurons. Olfactory neurons project axons through the cribriform foramen that synapse with the mitral cells of the olfactory bulb. The subepithelial lamina propria is found on the basolateral aspect of all regions of the nasal cavity, and contains numerous blood vessels, nasal lymphatics, and fibroblasts. There are also two layers of glands that secrete the enzymatic and osmotic components of nasal mucus. Sentinel cells and immunocompetent cells also reside in this region [35,36] (Figure 3).
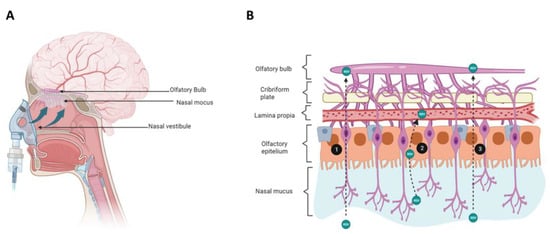
Figure 3.
Resveratrol delivery to the brain via the nasal route administration. (A) Schematic diagram of nose anatomy and the route involved in resveratrol (RSV) delivery from the nasal cavity to the brain. (B) Histology of the nasal cavity, epithelium, and routes used by RSV to reach the central nervous system. RSV administered to the nasal cavity moves along the olfactory sensory neuron through the cribriform place (1). RSV can also cross the olfactory epithelium through the transcellular route and get the lamina propria; there, RSV can be lost to systemic absorption via lymphatic or vasculature (2). RSV is transported to the central nervous system along the olfactory epithelium to reach the olfactory bulb (3). Created with BioRender.com accessed on 25 January 2023.
6. The Intranasal Route of Administration
Different routes of drug delivery have specific benefits but also disadvantages that should be considered when a formulation is designed. Resveratrol is a drug traditionally administrated through the intravenous and intraperitoneal routes; regrettably, these routes induce a trap of nanoparticles in the liver and spleen allowing the delivery of minor amounts in other organs including the brain. Therefore, alternative methods should be considered. Although oral delivery is a non-invasive route of administration preferred for patients, it has the difficulty of achieving the therapeutic concentration due to the solubility of drugs, its degradation in the gastrointestinal tract, and low delivery to a specific organ.
The intranasal route of administration has been used for a long time. Nevertheless, it has been traditionally used for topical administration of drugs for respiratory diseases or allergies [37]. Later, the intranasal route was used as a safe alternative to the oral and parietal route for the delivery of some systemic drugs, such as hormone replacement therapy or insulin, among others [38]. In 1991, William H. Frey II patented the intranasal route for administration of drugs with action into the CNS [39], and very soon, the nose-to-brain route received a great deal of attention as a drug delivery route [40]. Hence, the intranasal route appears as ideal for therapeutics in the CNS because it also is a non-invasive route and represent a direct route to reach the brain. Significantly, due to the size of the surface of the olfactory region and the extensive vascularization of the respiratory region, these are suitable regions for drug absorption, unlike the vestibular region [36].
The transport of a drug from the nasal cavity to the brain can occur through both the olfactory and trigeminal nerves, through intracellular and extracellular routes. In the intracellular route, an olfactory neuron internalizes the molecule through the formation of an endocytic vesicle, which travels to the neuron’s projection site where it will be released by exocytosis in the olfactory bulb; this process will be replicated by the olfactory neurons carrying the drug to other brain regions [41]. In the extracellular route, the molecule first crosses the nasal epithelium and reaches the lamina propria. Second, the molecule passes through the tight junctions that connect the various cell types that make the lamina propria up before being transported externally; third, the molecule travels along the neuronal axon through the flow of cerebrospinal fluid, through the paracellular, and perineural spaces, to lastly reach the subarachnoid space of the brain [36]. Although some authors agree that afferent lymphatic vessels and blood vessels can also participate in the nose-to-brain transport of drugs, it is considered that the main route is through the olfactory nerve, since the neuroepithelium of the olfactory region is the only part of the CNS that is directly exposed to the environment [36] (Figure 3).
Studies have shown that a drug administered intranasally is as effective as one performed by intravenous administration or rectally. Additionally, it has a better duration of action and efficacy than one administered orally or sublingually [42]. This superior efficiency also allows a reduction in the concentration of the effective dose, as has been documented in the case of corticosteroids or antihistamines [43,44]. However, not all drugs are candidates to be administered by this route, particularly those that have molecular masses greater than 1 KDa or drugs that are harmful to the nasal mucosa [45]. In addition, the nose’s own defense mechanisms, such as the production of mucus and the beating of cilia, reduce the effectiveness of this route, since they clean up the mucosa every 15 to 20 min, limiting the contact time of molecules with the mucosa [36]. Likewise, there is a series of proteolytic enzymes, which are intended to destroy pathogens and foreign bodies, which can also destroy therapeutic molecules. There are also efflux transporter pumps, which pump drugs out the nose [46]. Furthermore, this pathway can be affected by climatic or physiological conditions, for example, high temperatures can promote dryness of the mucosa or nasal bleeding, and low temperatures or the presence of environmental allergens promote mucus secretion. Likewise, smoking and nasal polyps are pathological conditions that alter the absorption of drugs [47]. However, perhaps one of the main factors influencing the extent to which drugs can be absorbed by the nasal lining are the physicochemical characteristics of the molecules, such as the aqueous and lipid drug partition coefficient, pKa, the molecular weight of the agent, the infusion rate, the volume of perfusate, the acidity of the solution, and the concentration [48]. The volume that can be administered through the nostril (~150 to 200 µL) is also a limiting factor of this route, which cannot always be achieved in the case of hydrophobic molecules [42].
Currently, multiple strategies are being developed to expand the use of intranasal technology for the greatest possible number of drugs, especially for their use as CNS therapeutic agents. For example, using absorption enhancers, enzyme inhibitors, mucoadhesives, or gelling agents to increase the viscosity of the formulations. Recently, nanoparticles, microspheres, and liposomes have begun to be an essential part of nasal drug preparations, because they improve the solubility of drugs, and in addition incorporate several components simultaneously [45,49]. This strategy has particularly benefited pharmacologically active compounds with poor stability in gastrointestinal fluids, poor intestinal absorption and/or extensive first-pass hepatic elimination such as peptides, proteins, and polar or poorly soluble molecules, as is the case of resveratrol [49].
7. Formulations for the Release of Resveratrol to the Brain
In recent years, nanotechnology have revolutionized drug delivery and modified nanoformulations to improve targeting, enhance the pharmacokinetic parameters, along with achieving the desired therapeutic levels of drugs in different tissues (including the brain) and notably, the evolution of strategies continues to this day. Essentially, a nanoformulation consist in a nanoparticulate system that include the use of natural or synthetic materials, from which multiple arrangements can emerge [50].
The components of the formulation contained an oil phase (e.g., medium chain triglyceride, lipoid-E80, eutanol, triglycerides, octyldodecanol, paraffin oil, labrafil, sefsol-218, lipoid-S75, benzyl alcohol, cholesterols, 2-butanone, isopropyl myristate, etc.). Some of the lipids are from different natural sources (e.g., canola, eucalyptus, coconut, sesame, rice bran, safflower, soybean, and cottonseed) but also synthetic lipids are used (Caproyl 90, triacetin, isopropyl myristate, oleic acid, palm oil esters, corn oil, olive oil, isopropyl palmitate, LabrafilMM44CS, Maisine 35-1, Miglyol 812, Captex 200, Captex 355, and Captex 8000). Also, requiring a surfactant, compounds that modified the surface properties and stabilized the particle by reducing the tension, promoting dispersion (inhibiting droplet aggregation), and acting as foaming agents. Frequently used surfactans are plantacare-810, polyoxyethylene sorbitan monolaurate 20, 40, 60 and 80 (Tweens), dicaprylic carbonate, soybean lecithin, poloxamer-407, epikuron-170, polyoxyethylenated castor oil (Cremophor EL), ethanol, n-butanol, polysorbate-80, sucrose esters, sorbitan monolaurate 20, 40, 60 and 80 (Spans) amongst others. The formulation also needs a cosurfactant, which should be introduced in small amounts. The majority of cosurfactants are made of short and medium-chain alcohols (C3–C8). They reduce interfacial tension and increase an interface’s fluidity and permeability. Some of these compounds used are Span-80, sorbitanoleate, ethanol, potassium sorbate, propylene glycol monolaurate, poloxamer-180, cetrimide, transcutol-P, solutol, 1-O-alkylglycerol, plurol oleique CC 497 caprol, diethylene glycol mono ethylether, emulphor E1-620, etc. [51,52].
The progress in resveratrol nanoformulations have attracted attention due to their simple preparation process, the scalability of synthesis, and the controllability of shape and size of the particle. In this manner, resveratrol’s formulations with improved properties have been designed and some of them used for the treatment of different diseases (Table 1). Importantly, there are also formulations designed for intranasal delivery (Table 2) [53]. The BBB has represented a special and difficult issue that has been overcome employing different strategies. For example, a promising approach is to delivery drugs using as a target BBB-associated carrier and receptor proteins such as the transferrin receptor that is expressed by the brain capillary endothelial cells; these proteins improve transport and facilitate adhesion and endocytosis [54,55]. Likewise, some components enhance properties of formulations: polyethylene glycol 1000 (PEG) is a hydrophilic polymer that stabilizes the nanoparticle and acts as a mucus diffusion enhancer by reducing interaction with mucin; vitamin E succinate, is an inhibitor of the P-glycoprotein transporter; and chitosan, is a polysaccharide of D-glucosamine and N-acetyl -D-glucosamine obtained from the deacetylation of chitin which positive charged permit particles stay longer at the negatively charged olfactory and respiratory epithelium [55,56,57]. The bio-incompatibility is another important challenge to overcome, as interaction of nanoparticles with cells implicates complex factors to attend such as toxicity and activation of the immune system (e.g., platelet activation, inflammation, and antibody production that might cause of thrombosis and hemolysis). In this case, the use of lipid-based carriers made from biocompatible and biodegradable lipids like those constituting the cell membrane represent a solution. To characterize nanoparticles, they should be measured different properties, due to the limitations of each technique a complete knowledge of the formulation is necessary. The most common evaluation include: visual appearance (analysis of homogeneity and color at equilibrium), color, odor and taste (for those oral formulations), density (associated to the trapped air inside the vesicle), pH, osmolarity, phase behavior, viscosity (to evaluated flow behavior), conductivity (detects the phase inversion phenomenon), structural and dynamic features (performed by dielectric measurements and nuclear magnetic resonance studies), emulsion droplet polarity, dispersibility, interfacial tension, thermal characterization and other measurements like refractive index and zeta potential [51,52].
Nanovehicles can be classified as polymeric nanoparticles, lipid-based nanoparticles, metallic nanoparticles, etc. Most used lipid-based nanoparticles are liposomes, nanoemulsions formed with micelles, microemulsions, solid lipid nanoparticles (SLN), and nanostructured lipid carriers (NLC). These nanoparticles are often modified on its surface with polymers such as PEG or poloxamers to improve the colloidal stability through increasing the surface hydrophilicity and reducing the surface negative potential [58].
Liposomes are spherical-shaped vesicular structures made essentially from phospholipids, which contain a hydrophilic head and two hydrophobic tails. To augment structure stability and drug loading, liposomes can contain cholesterol, ethanol, or non-ionic surfactants. The alcohol included a (40–45%) increase in lipid fluidity and cell membrane permeability; additionally, provides a negative charge that avoids aggregation. Depending on its composition, different types of liposomes are classified as: niosomes, systems formed by the hydration of synthetic non-ionic surfactants with or without the addition of cholesterol or other lipids; transferosomes, phospholipids with additional single-chain non-ionic surfactants, also called edge activator, are extremely flexible and self-regulating membranes; ethosomes, vesicular structures with a high amount of ethanol; bilosomes, sotf lipid nanocarriers that contain bile salts as an edge activator; cubosomes, are three-dimensional cubic lattice formed by the self-assembly of lipid molecules with a wide variety of morphologies (spheres, cylinders, vesicles, films, fibers, tubules, etc.). The liquid-crystalline phase encloses two separate regions of water divided by surfactant-controlled bilayer. The lipids used include nonionic surfactants (e.g., monoolein, which possess both hydrophilic and hydrophobic regions) given the ability to encapsulate and deliver hydrophobic and hydrophilic drugs [59,60].
Nanoemulsions are systems composed of nanosized oil droplets dispersed in an aqueous phase and an emulsifier, which gives stability on heterogeneous systems and potential thermodynamic stability (does not aggregate or flocculate), have high oral bioavailability because they avoid hepatic first-pass metabolism, and a large surface area that increases penetration in the emulsion system. It is classified in three types: oil-in-water, water-in oil (reversed micelles), and bi-continuous. Nanoemulsions are super solvents because they include both hydrophilic and lipophilic drugs. In oil-in-water emulsions, the formulation typically consists of 5 to 20% of lipid [51].
Microemulsions are transparent isotropic colloidal dispersions of oil and water stabilized by an interfacial film of surfactant and cosurfactant. Nanoemulsions share some characteristics with microemulsions, however, microemulsions form spontaneously and involve a higher surfactant to cosurfactant ratio, need a low value of interfacial tension, have a spherical or nonspherical shape, and have a longer shelf life [52].
SLN are colloidal particles composed of a biocompatible and biodegradable lipid matrix that is solid (crystalized) at body temperature (physiological lipids that replaced the liquid lipid), with a surfactant at the outer shell. The drug has been located between fatty acid chains, the lipid layers, and the crystal imperfections which reduced the capacity to accommodate large amounts of the drug [61].
NLC were developed to overcome the disadvantages of SLN, with the objective of increase drug loading and prevent drug expulsion. The NLC are a mixture of solid and liquid lipids that allow high drug loading of hydrophobic molecules [58,62].
7.1. Nose to Brain Delivery of Resveratrol Formulations
Limitations of drug to brain delivery can be reduced by the proper election of the drug carrier and the route of administration. Some authors have chosen among the multiple arrangements to test resveratrol delivery to the brain using the intranasal route, showing promising results that could be easily tested in models of cerebral ischemia (Table 2).

Table 1.
Nanotechnology-based formulations for resveratrol delivery.
Table 1.
Nanotechnology-based formulations for resveratrol delivery.
| Drug Delivery Nanosystem | Structural Characteristics | Drug Encapsulation Efficiency (%) | Spherical Shape (nm) | Observations | Reference |
|---|---|---|---|---|---|
| Vesicles | |||||
| NIOSOMES Formed by an aqueous core enclosed within a non-ionic surfactant bilayer. | Gelot 64 (SF) oleic or linoleic acids (ST) | Resveratrol 35 ± 2 | 299–402 | Ex vivo skin permeation experiments indicated that niosomes prepared with the ethanol injection are more effective. | [63] |
| Span 60 (SF) Dodecanol (ST) | Resveratrol 64 ± 8 | 168 ± 4 | Niosomes exhibited a small mean size, narrow size distribution, high entrapment efficiency, and good stability | [64] | |
| Maisine 35–1 (SF) Dodecanol (ST) | Resveratrol 53 ± 8 | 175 ± 13 | Niosomes exhibited a small mean size, narrow size distribution, high encapsulation efficiency and good stability | [64] | |
| Span 60 (SF) Cetyl alcoho (ST) | Resveratrol 81 ± 2 | 108 ± 3 | Nanoencapsulation of resveratrol using non-ionic surfactant and fatty alcohol dramatically improved its solubility and functionality | [65] | |
| LIPOSOMES Formed by phospholipids, which can include several other constituents like cholesterol to improve the stability of the bilayer. | OramixTM CG110 (SF) LauroglycolTM FCC (SF) Soybean phosphatidylcholine (ST) Oleico acid (ST) | Resveratrol >70 | 90 | Toxicity of the vesicular formulations evaluated in vitro, showed no alteration on cell viability after incubation with RSV loaded vesicles. | [66] |
| Soybean lecithin (ST) Oleico acid (ST) N-succinyl chitosan as protective shell of liposomes | Resveratrol and Quercetin 70 ± 5 | 82 ± 3 | The succinyl-chitosan shell increased physical stability of the vesicular system and release of the polyphenols to a greater extent at pH 7.0 (mimicking the intestinal environment). | [67] | |
| Tri-block polymer L64 D-α-tocopheryl polyethylene glycol 1000 succinate (SF) Phospholipon 90G (ST) | Resveratrol 95 | 86 ± 3 | Biocompatibility was demonstrated in an ex vivo model of hemolysis in human erythrocytes. Liposomes maintained the antioxidant properties of resveratrol. | [16] | |
| Emulsions | |||||
| SIMPLE EMULSIONS Formed by emulsifiers dispersed in oil, and water. | Soybean oil (OP) Soy lecithinas (SF) Labrasol (ST) | Resveratrol 92.5 ± 2.2 | 132 ± 9 | The improved oral bioavailability of resveratrol was attributable to the inhibition of intestinal glucuronidation by the presence of labrasol. | [68] |
| Labrafac (OP) Cremophor® 40 (SF) Hyaluronic acid (SF) | Resveratrol and curcumin N. A | 115.2 ± 0.2 | The mucoadhesive nanoemulsion was safe to nasal mucosa and increase amount of both polypehnols in the brain. | [69] | |
| Soybean phospholipiddipropylene glycol (OP) 2-hydroxypropyl-β-cyclodextrin (SF) Phosphatidyl choline 60 (SF) | Resveratrol N. A | 122 ± 08 | Nanoformulation presented good compatibility and skin permeation. | [70] | |
| Olive oil (OP) Polyethylenglycol Pluronic® P105 (SF) Cremophor® EL (SF) | Resveratrol N. A | 155 ± 33 | Perfluoropolyether was used as a nano emulsion tracer. Resveratrol-loaded nano emulsions was internalized by macrophages | [71] | |
| Capryol™ 90 (OP) Cremophor® EL (SF) Tween 20 (SF) | Resveratrol N. A | 41.3 ± 4 | The formulation increased oral bioavailability 3.2-fold compared to resveratrol alone. Anti-fatigue properties of resveratrol were improved. | [72] | |
| Labrafil (OP) Gelucire® 44–14 (SF) Labrasol (SF) | Resveratrol N. A | 21 ± 5 | Nanoemulsions significantly increased intestinal permeation across rat jejunum. | [73] | |
| Labrafil (OP) Miglyol® 812 (SF) Tween 80 (SF) | Resveratrol N. A | 103 ± 14 | Nanoemulsion significantly increased intestinal permeation across rat jejunum. | [73] | |
| Labrafil (OP) Labrasol (SF) Cremophor RH 40 (SF) | Resveratrol N. A | 26 ± 1 | Formulation components inhibited the UDP-glucuronosyltransferase and consequently increases the bioavailability of resveratrol. | [74] | |
| Miglyol® 812 (OP) orange oil (OP) Quinoa starch particles (SF) Octenyl succinic anhydride (SF) | Resveratrol 98 ± 2 | 50,000 | Appropriate resveratrol carrier system for use in oral formulations. | [75] | |
| Miglyol® 812 (OP) orange oil (OP) Quinoa starch particles (SF) Tween 20 (SF) | Resveratrol 63 ± 1 | 50,000 | Appropriate resveratrol carrier system for use in oral formulations. | [75] | |
| Olive oil (OP) Sodium lignin sulfonate (SF) Tween 80 (SF) PEG-400 (SF) | Resveratrol N. A | 119 ± 5 | Excellent biocompatibility, improved solubility, and showed antiradical efficiency compared to free trans-RSV | [76] | |
| MULTIPLE EMULSIONS Formed by multiple emulsions of water-in-oil-in-water. | Medium-chain Triacylglycerols (OP) Tween 80 (SF) For double layer nanoemulsions: chitosan, alginate or β-cyclodextrin | Resveratrol N. A | 27 ± 55 | The stability of resveratrol in the double-layer nanoemulsions complexed with chitosan or β-cyclodextrin was higher, compared with single-layer nanoemulsions. | [77] |
| Miglyol® 812 (OP) The inner aqueous phase: ethanol, RSV, and 0.1 M NaCl solution The external aqueous phase: carboxymethylcellulose, Tween 20, and 0.1 M NaCl. | Resveratrol 55 | 59,800 ± 0.002 | High encapsulation efficiency and slow storage release. | [78] | |
| Single-layer emulsion: lactoferrin Multilayer emulsion: lactoferrin/alginate lactoferrin/alginate/ε-poly-L-lysine | Resveratrol N. A | 249 | The antioxidant activity of resveratrol-loaded emulsion did not significantly change during storage, whereas it decreased in nonencapsulated resveratrol oil from the third week onwards. | [79] | |
| The single layer: ethanol/water RSV solution Miglyol 812 Polyglycerol polyricinoleate (ST) Multilayer emulsion: Tween 20 solution with and without sodium carboximethylcellulose as thickening agent. | Resveratrol 77.50 | 10,000–30,000 | The emulsion showed high encapsulation efficiency, good storage stability, shear-thinning behavior, and dominant elastic character. These double emulsions may be suitable for food applications. | [80] | |
| Particles | |||||
| LIPIDIC NANOPARTICLES Formed by a solid hydrophobic core stabilized by a hydrophilic layer of surfactant molecules or polymers. | Tristearin (OP) Chitosan (ST) Hydrogenated phosphatidylcholine (SF) | Resveratrol 67 | 6000 | The cream containing the chitosan-coated lipidic nano particles produced a significant enhancement in the in vivo permeation of resveratrol | [81] |
Abbreviations: SF, surfactant; ST, stabilizers; OP, oily phase; N. A, not applicable.

Table 2.
Formulations for resveratrol intranasal delivery to the brain.
Table 2.
Formulations for resveratrol intranasal delivery to the brain.
| Formulation | Components | Physical Characteristics | EE (%) PDI ZP (mV) | Observations | Reference |
|---|---|---|---|---|---|
| Vitamin E nanoemulsion | Vitamin E:sefsol (1:1) (ST) Tween 80 (SF) Transcutol P (co-SF) | Transparent and monophasic. Spherical globules (102 ± 1.46 nm). | N.D. 0.158 ± 0.02 −35 ± 0.02 | Histological studies showed decreased degenerative changes | [82] |
| F6SPION/W3 SPION-loaded with chitosan coated bilosomes | Cholesterol:sorbitan monosterate (Span 60) (1:1), sodium deoxycholate (10 mg), glycerin. Chitosan (0.1%). SPION (magnetite, Fe3O4, 10.3 mg/mL). Waffers: sodium alginate: poly vinyl pyrrolidone K-25 (1:2) + glycerol (10%). | Black spheres surrounded by a layer of less intense black color (chitosan coating layer). Porosity 88% (243 ± 1.87 nm). | 85 ± 1.08 0.14 ± 0.06 35 ± 2.05 | Resveratrol (20 mg/Kg/day, i.n.) improved induced memory and cognitive functions in mice with induced Alzheimer model with lipopolysaccharide. | [83] |
| Cubosomal in situ nasal gel | 4% w/v glicerol monooleate and 1.5% Lutrol F127. 12% w/v Poloxamer 407 (cubosomal gel, permeation enhancer) | Cubical in shape, uniform with smooth surface (161.5 ± 0.12 nm). | 83.08 ± 0.21 0.279 ± 0.15 −20.9 ± 0.11 | The formulation showed higher trans-nasal permeation and brain’s distribution compared to resveratrol solution. | [84] |
| Transferosomes capped with gold nanoparticles [gold (III) chloride HauCl4] | Soy lecithin (glycerophopholipids), ethanol (permeation enhancers), and Cremophor RH 40 (SF) in a ratio of 46.7:20:33.3 (w/w%). | Gold- nanoparticles (GNP), resveratrol-transferosomes-GNP, and resveratrol-nanoemulsion-GNP showed uniform spherical shape with size of 10.30 + 2.4, 94.93 + 5.6, and 20.36 + 4.3 nm | 69.53 ± 3.82 0.194 ± 0.08 −28.7 ± 4.7 | Transferosomes penetrated through nasal mucosal layers. Resveratrol-trans-GNP gel enhanced the spatial memory recovery in amnesic rats compared to i.n. pure resveratrol. | [85] |
| Nanoemulsion capped with gold nanoparticles [gold (III) chloride HAuCl4] | Capryol 90 (ST) Tween 20 (SF), and transcutol (co-SF) at a ratio of 30:60:10 v/v%. | 95.72 ± 5.34 0.26 ± 0.04 −18 ± 2.6 | |||
| Nonoestructured lipid carrier loaded in a situ gel | Cetyl palmitate (solid lipid), Capmul MCM (ST) 1:1 ratio. Acrysol (nonionic, solvent-free and hydrophobically modified ethylene). Poloxamer 188 (SF), Tween 80 (SF). Gellan gum 0.5% and xanthan gum 0.15% (in situ gel formulation) | 132 ± 11.9 nm | 74.05 ± 11.40 0.209 ± 0.005 −23 ± 3.79 | The scopolamine-induced amnesia was reduced with the optimized formulation. | [86] |
| Chitosan-coated lipid microparticles | Phosphatidylcholine (SF) 1.75%, w/v, Stearic acid (ST) Chitosan (bioadhesive excipient) | 68.5 ± 3.1 µm | N.D. N.D. −12.7 ± 2.1 | Resveratrol was found in the rat cerebrospinal fluid with no distribution in blood after nasal administration. | [87] |
| 76.3 ± 5.2 µm | 76.50% N.D. 24.0 ± 4.7 | ||||
| 84.5 ± 8.1 µm | 81.00% N.D. 44.6 ± 3.1 | ||||
| Ionic-sensitive in situ gel loaded with resveratrol nanosuspensions | 0.6% w/v gellan gum | Spherical shape in gellan gum matrix and dispersed uniformly without adhesions. | N.D. 0.234 −8.8 | The increase in the bioavailability of resveratrol in the brain showed in the pharmacokinetics studies suggested the direct nose-to brain delivery. | [88] |
| Nanosized transferosome based intranasal in situ gel | Transferosome Soya lecithin/PE (ratio 7:3) + (Cremophor RH 40 (SF) Gel: Poloxamer 407 (18%) and Carbopol 934 (0.4%) | Spherical shape (83.79 ± 2.54 nm) | 72.58 ± 4.51% N.D. N.D. | Resveratrol bioavailability was enhanced through the nasal route of administration. | [89] |
| Hyaluronic acid based lipidic nanoemulsion | Labrafac Lipophile/cremophor 40 10% (SF); hyaluronic acid 1.5% w/v | Spherical morphology and nanometer size (115 ± 0.15 nm). | N.D. 0.235 ± 0.01 −23.9 ±1.7 | The pharmacokinetic analysis showed that the intranasally administered mucoadhesive formula allowed the absorption of resveratrol and curcumin reaching the brain. | [69] |
Abbreviations: N.D., not defined.
7.1.1. Lipid Microparticles
Lipid microparticles consist of a natural lipid-based solid core stabilized by a layer of surfactant at the outer shell that exhibit high biocompatibility and biodegradability and low production cost. Trotta et al. (2008) designed a formulation testing various lipids (tristearin, glyceryl behenate, and stearic acid) in combination with phosphatidylcholine as a surfactant [87]. Combinations showed an entrapment efficiency that varied from 65.9 to 78.6% and resveratrol loading capacity from 5.5 to 6.8%. The stearic acid was selected as the lipid material because it reached the highest rates of release after 15 and 120 min (50 and 100%, respectively). A Chitosan coating was added to prolong the residence time of the formulation on the nasal mucosa, and, because of its reversible capability to open the tight junctions between cells, increased the permeability. In vitro studies showed a constant permeation of the formulation for 40 min that was increased two-fold in the coated vesicles with a high concentration of Chitosan (plus formulation). Chitosan hydrochloride solution increased resveratrol concentration in the cerebrospinal fluid of rats, 60 min after nasal administration, reaching similar values observed with the non-coated lipid microparticles, whereas a radical increase was observed with chitosan-coated microparticles plus formulation (Table 3). These results showed the superior ability of the chitosan coated microparticles to induce the resveratrol uptake in cerebrospinal fluid. The short time to reach the CNS suggests that the resveratrol uses the paracellular diffusion across the olfactory mucosa rather than by the trans-neuronal pathway which is known to require at least 24 h. Interestingly, resveratrol was undetectable in the bloodstream suggesting that most of resveratrol was delivery to the brain.

Table 3.
Pharmacokinetics parameters of different formulations of resveratrol.
7.1.2. Transferosomes
Transferosomes are ultradeformable and very flexible vesicles composed mainly of phospholipids and a permeation enhancer that soften the lipid bilayers. Transferosomes also show a high permeability coefficient and flux attributed to a marked elasticity since phospholipids increase the affinity of the particles to the biological membranes. These vesicles appear to be a better option than nanoemulsions. Regardless, nanoemulsions are systems with high surfactant content which increases drug solubilization and a big external area that favors cellular uptake, meanwhile transferosomes squeeze and penetrate small spaces to cross nasal mucosa, the rigid mixed micelles of nanoemulsions are less deformable and less intranasal permeable.
In agreement, transferosomes showed a permeation of up to 81.29 ± 2.64% while the nanoemulsion only 54.64 ± 2.75% between 6 and 8 h after administration. When formulations were compared in an experimental model, the intranasal resveratrol-transferosomes –GNP gel enhanced the spatial memory recovery in amnesic rats compared to both resveratrol nanoemulsion gel and the pure resveratrol suspension. This result was attributed to improved intranasal permeation, although it could also associated to micelle formation and decrease entrapment [85].
Similarly, transferosomes obtained using oleic acid as permeation enhancer and cremophor RH 40 as surfactant showed improved vesicular bilayer fluidity and resveratrol solubility compared to resveratrol suspension gel. The nasal nanosized transferosome based gel duplicated resveratrol’s concentration in plasma compared to oral administration of resveratrol suspension (Table 3). The Tmax for nasal transferosomes was increased by 7 h compared with oral resveratrol suspension, response that was associated to a continued release of resveratrol for around 12 h. Importantly, a sustained plasma concentration of resveratrol was observed up to 24 h after intranasal administration of the optimized resveratrol transferosome gel [89]. This last property (sustained and prolonged drug release) is important for a continued activation of pathways involved in protective mechanisms of drugs.
7.1.3. Bilosomes
Bilosomes are a type of liposomes that contain bile salts (e.g., deoxycholic acid, sodium cholate, deoxycholate, or sorbitan tristearate) and exhibit a flexible structure that facilitate penetration in epithelium. Chitosan coating delayed drug leakage from the vesicles by decreasing membrane fluidity. In addition, a biocompatible triblock polymeric compound (Poloxamer) enhanced stability of the vesicle and low the immune response. Abas et al. (2021) designed the formula F6SPION/W3, which is a chitosan coated non- ionic surfactant vesicle composed of superparamagnetic iron oxide nanoparticles [83]. This type of particles reaches high drug targeting effectiveness among carriers since the external magnetic field is applied to the target organ. F6SPION/W3 was administrated for 14 days into the right nostril of the rats. Its mucoadhesive nature allowed that upon contact with mucous entered in close contact with the nasal tissue, favoring extension of the residence time and release of resveratrol up to 8 h after administration. Furthermore, treatment with the formulation showed improved results in the Alzheimer model (memory and cognitive functions in mice) compared to resveratrol suspension. Undoubtedly, bilosomes increased resveratrol’s solubility, bioavailability, and delivery to the brain; nevertheless, authors didn’t measure the concentration of resveratrol reached to the brain.
7.1.4. Cubosomes
Cubosomes are composed of amphiphilic polar lipid that at high concentrations form micelles arranged as cubic structures. Its unique structure offers stability and sustained drug release that can be modulated by varying the lipid composition and modifying the surface; additionally, Poloxamer 407 augmented viscosity of formulation favoring the retention time in the nasal cavity. The drug release mechanism is based on diffusion but many factors influence the rate (drug solubility, cubic-liquid-crystalline geometry, pore size, release medium, etc.). The preparation method is simple but has the drawback that large-scale production is challenging due to their high viscosity and potential to leak throughout the storage or in vivo transmission [59].
Fitty µL of resveratrol loaded cubosome were administrated at the nostril of the rats, showing 67.04% release in 12 h; this value was higher compared to plain resveratrol (up to 20%). The increased discharge was associated to glycerol monooleate, a main component of the cubosomes, which helps controlling the release of the drug. Ex vivo permeability studies indicated that after 8 h resveratrol permeation from cubosomal in situ gel and resveratrol plain in situ gel was 25.29 and 34.06%, respectively, this behavior suggest that the passage of resveratrol loaded cubosomes from solvent to water was slower compared to resveratrol solution. Furthermore, the presence of a lipid matrix over the cubosomes as well as Poloxamer 407 which acts as permeation enhancer, favored passing of cubosomes through BBB [84]. All these characteristics explain the increased bioavailability formulation and the maximal concentration reached compared to resveratrol solution (Table 3).
7.1.5. Nanoemulsions
Nanoemulsions are systems that increase drug solubility of lipophilic drugs providing a large surface area for drug absorption but are thermodynamically unstable and can suffer efflux of the encapsulated drugs during long storage [58]. Nanoemulsions co-encapsulating resveratrol and curcumin were successfully prepared at 10% of the surfactant Cremophor RH 40 with the combination of oily phase (Labrafac and lipophile). Hyaluronic acid was also added to increase the viscosity of the external phase to reduce mucociliary clearance and increase the residence time in the nasal cavity. In total, 40% of both drugs were released from the formula after 0.5 and 60% after 6 h following a diffusion-controlled mechanism. To demonstrate that the polyphenols preserved the antioxidant potential in the nanoemulsions, a DPPH antioxidant assay was used. The DPPH inhibition assay showed that the lipidic nanoemulsion preserved resveratrol and curcumin properties after 3 months [69]. This study showed that intranasally administration allowed the absorption of both drugs by passive diffusion and represent a good option for administration of resveratrol combined with curcumine.
Pangeni et al. (2014) also prepared a nanoemulsion but supplemented with vitamin E [82]. The optimized formulation showed to provide high ex vivo trans-nasal mucosal flux. The in vivo pharmacokinetic studies showed that the brain/blood ratio after 30 min for resveratrol solution intravenous, resveratrol solution intranasal, and the nanoemulsion intranasal was 0.30 ± 0.01, 1.19 ± 0.39, and 2.86 ± 0.70. The percentage of resveratrol transported to the brain was 69.94%, indicating the superiority of intranasal administration and the nanoemulsion for brain delivery through the olfactory route. Additionally, the antioxidant activity of the nanoemulsion was higher (96.48%) compared to ascorbic acid and resveratrol alone (94.45 and 76.97%, respectively) given a property extra to the function of the system.
Comparison between pharmacokinetic parameters of both nanoemulsions showed the superior capacity on delivery to the brain with the particle designed by Nasr et al. [69]. The observed maximum concentration of resveratrol in the brain after administration of the nanoemulsion was 592% higher than after the solution, while the formulation of Pangeni et al. (2014) showed only a 132% of superiority. These data show the importance of testing different preparations [82].
7.1.6. Nanostructured Lipid Carriers (NLC)
Optimized NLC were prepared testing short, medium, and long-chain glycerides. Resveratrol showed higher solubility in propylene glycol stearate, glyceryl stereate, and gelucire but the maximum value was found with cetyl palmitate. Capmul MCM (caprylic and capric mono-diglyceride esters) was added to the mixture because addition of oil to solid lipid increases drug loading; this oil also provides bacteriostatic activity that protects gel throughout storage. Additionally, the surfactants Poloxamer 188 and Tween 80 which showed the higher entrapment and drug loading were used for emulsification and stabilization of the system. Although surfactants like Tween 40 and Span (20, 60, and 80) showed high resveratrol solubility, they were not used since the resveratrol’s poor solubility promoted its entrapment in the oil-lipid matrix. The 1:1 oil ratio resulted in NLC of ~200 nm, but the particle size was reduced to 112 ± 8.50 nm by the addition of the stabilizer Acrysol K150. The increment of lipid and oil resulted in formation of larger particles that were reduced with the surfactant concentration. The authors also evaluated various gels for the viscosity of sol-gel formation upon adding nasal fluid and measured the minimum time required for gel formation. The tested compounds were gellant gum, xanthan gum, Carbopol 934, Carbopol 971P, Carbopol 980, Carbopol ultrez 10, Ploxamer 188, and Poloxamer 407, alone and combined. The optimal time for gel formation (33 ± 7.02 s) was obtained with the mixture of gellan gum and xanthan gum, this short time was essential for quicker onset of resveratrol delivery. The optimal concentration of the compounds yielded a transparent gel with sufficient gel strength (43 ± 6.24 s) and viscosity after gelation (41.13 ± 5.48 cp) with no significant expansion in gel volume (<3%). In vitro permeation indicated that the selected formulation had 5-fold higher permeation compared to resveratrol-based gel due to the prolonged residence time of the formulation in the nasal cavity. The stability of the mixture was maintained for 6 months, time after which the particle size and zeta potential was maintained (Table 2). The efficiency of resveratrol delivery was tested in an Alzheimer disease model induced by scopolamine (1 mg/kg; i.p.). Comparing administration of the optimized in situ gel and resveratrol in suspension showed the capacity of the formulation to reduce the scopolamine-induced amnesia [86].
7.1.7. Nanosuspensions
Nanocrystals is a term used to refer to nanoparticles of a pure drug, lacking any sort of matrix material, but formulated as colloidal suspensions (nanosuspensions) in water or non-water media and stabilized with surfactants or polymers. Hao et al. (2016) prepared resveratrol nanosuspensions optimized for nasal administration using different amounts of deacetylated gellant gum (0.2–1% w/v), an anionic deacetylated exocellular polysaccharide with linear repeating tetrasaccharide units that can form transparent gels on exposure to certain concentrations of cations (e.g., sodium, potassium, and calcium) that are present in the nasal mucosa mucus [88]. Since the volume of the formed gel increased after suffering the solution-gel phase transition, to avoid clog of the small nasal cavity and cause discomfort, it was determined the expansion coefficient of the in situ gel. The optimal deacetylated gellant gum concentration was below 0.7% because the higher concentration augmented viscosity rendered it problematic for administration. Using less than 0.4% the in situ gel was so easily diluted by mucus of the nasal cavity. The drug release rate from resveratrol nanosuspension in situ gels exhibited faster profiles than the resveratrol suspensions in situ gels, approaching nearly 50% at 2 h and nearly 100% at 6 h compared to 20% at 2 and 50% at 6 h, respectively. As expected, resveratrol concentration in plasma following intravenous injection of nanosuspensions exhibited rapid elimination with a short biological half-life (<0.5 h). On the contrary, following intranasal in situ gel administration the resveratrol concentration was higher at all-time points when compared with the intravenous treatment. Also, there was a higher bioavailability of 2.88 times in the brain obtained by in situ gel nanosuspensions. Evaluation of the organ/plasma distribution following the intranasal and the intravenous administration showed that 78% of resveratrol was absorbed into the brain directly from the nasal cavity. Also, distribution and localization studies using coumarin 6 revealed that resveratrol can be directly transported into the brain via a direct nose-to-brain route.
7.1.8. Cell Penetrating Peptides
Cell penetrating peptides are used in the intracellular delivery of molecules with low penetrability. A formulation for resveratrol was designed using this technology. One phenol group of resveratrol was conjugated with 4-maleimidobutyric acid and then with multimeric leucine (L)- and lysine (K)-rich sequences. These peptides exhibit intracellular penetration by rapid induction of heparin sulfate proteoglycan receptor -mediated endocytosis. The conjugate penetrated the cytoplasm of nasal epithelial cells and subsequently, free resveratrol (~90%) was released within 4 h in the cytosolic by endogenous esterase. Interestingly, 14% RSV-LK or free resveratrol was located in the nucleus and 86% in the cytoplasm; however, in vivo penetration assays showed that the LK peptides are retained in the epithelial cells and only a small portion penetrated deeper into the tissue [90].
8. Disadvantages of the Nanocarriers Systems
Certainly, while nanocarriers in nasal drug delivery hold promise, there are several disadvantages associated with their use. The potential drawbacks of nanocarriers in this context are related to several factors but some authors have overcome many of them. First, the mucociliary clearance, since the rapid removal of nanoparticles by the mucociliary system limits the residence time, hindering sustained drug release and reducing overall bioavailability. Also, there are limitations in the material selection because of the biocompatibility and toxicity concerns, since interaction with nasal mucosa or other biological barriers may trigger immune responses or long-term adverse effects, necessitating rigorous safety assessments before to the clinical transference. There is also a limited understanding of long-term effects due to the prolonged exposure to nanoformulations in the nasal cavity, more research is needed to assess the potential cumulative effects and ensure the safety of chronic administration since nanocarriers might induce nasal irritation, inflammation, discomfort, or other adverse reactions, affecting patient compliance and acceptability.
It is well known that nanocarriers may face challenges in maintaining stability, particularly during storage and transportation, phenomena such as aggregation, degradation, or changes in physicochemical properties over time can compromise the reliability of the drug delivery system. On the other hand, many nanocarriers have a limited capacity for drug loading, restricting the amount of therapeutic agent that can be delivered. To some of these nanosystems, the manufacture is still a challenge, such as the transition from laboratory-scale production to the large-scale, since consistent quality and reproducibility are complex tasks, impacting the feasibility and cost-effectiveness of mass production. Finally, regulatory approval processes for novel nanocarriers in nasal drug delivery is intricate, stringent requirements for safety and efficacy data, coupled with a lack of standardized testing protocols, can prolong the approval timeline and increase development costs, therefore, researchers need to work closely with regulatory bodies to meet the necessary requirements for the approval of nasal nanoformulations of resveratrol.
9. Future Directions
Resveratrol nanoformulations investigated drug bioavailability, where strategies such as the use of permeation enhancers and/or mucoadhesive formulations were explored to enhance drug absorption through the nasal mucosa for achieving therapeutic effects. Some of them also performed toxicity studies that overcome potential adverse effects of nano-sized materials inhaled through the nasal route. However, the transference of nasal nanoformulations into clinical applications requires further investigation by conducting well-designed clinical trials to validate the safety and efficacy of these formulations in humans for their widespread adoption. On the other hand, targeted drug delivery to specific cells of the brain remains an area of interest, that has been aborded by designing nanocarriers that reaches selective targets. With respect to its manufacture, in pharmaceutical research efforts should be made to design and evaluate efficient devices to create aerosols with adequate technological properties for the nasal administration. The considerable variation in nasal anatomy among individuals poses a challenge for the uniform and efficient drug delivery; variations in nasal geometry and mucosal thickness can result in inconsistent drug absorption and therapeutic outcomes.
10. Conclusions
Resveratrol’s intranasal route of administration combined with the use of a nanoformulation, among others benefits, avoid the hepatic metabolism and increase delivery of larger amounts of the drug directly to the brain. Formulation’s composition defines the strategy that enhance permeation (e.g., reducing expression of drug transporters, increasing membrane fluidity or opening the tight functions), but all of them are attractive alternatives to improve the resveratrol’s neuroprotective effects for stroke. However, studies that quantify the amount of resveratrol that reach the brain and showed its specific intracellular location that demonstrated that the blood–brain barrier does not retain the resveratrol, is missing. Additionally, the olfactory region of rodents is much larger than that of humans, so caution will have to be taken with the interpretation of the results of pharmacological and pharmacokinetic studies. Therefore, use of intranasal nanoformulations of resveratrol is encouraged, but future clinical stroke studies will be necessary to evaluate the effectiveness in reducing the damage.
Author Contributions
Conceptualization, I.A.-B. and P.A.; investigation, I.A.-B., I.G.G.-H., S.A.-A. and P.A.; writing—original draft preparation, I.A.-B., I.G.G.-H. and P.A.; writing—review and editing, I.A.-B., S.A.-A. and P.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
We thank the Postdoctoral Fellowship awarded to A-BI by CONACHYT-México (CVU 445084).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Burns, J.; Yokota, T.; Ashihara, H.; Lean, M.E.J.; Crozier, A. Plant Foods and Herbal Sources of Resveratrol. J. Agric. Food Chem. 2002, 50, 3337–3340. [Google Scholar] [CrossRef] [PubMed]
- Thapa, S.B.; Pandey, R.P.; Il Park, Y.; Sohng, J.K. Biotechnological Advances in Resveratrol Production and Its Chemical Diversity. Molecules 2019, 24, 2571. [Google Scholar] [CrossRef]
- Donkor, E.S. Stroke in the 21st Century: A Snapshot of the Burden, Epidemiology, and Quality of Life. Stroke Res. Treat. 2018, 2018, 3238165. [Google Scholar] [CrossRef] [PubMed]
- Marko, M.; Posekany, A.; Szabo, S.; Scharer, S.; Kiechl, S.; Knoflach, M.; Serles, W.; Ferrari, J.; Lang, W.; Sommer, P.; et al. Trends of R-TPA (Recombinant Tissue-Type Plasminogen Activator) Treatment and Treatment-Influencing Factors in Acute Ischemic Stroke. Stroke 2020, 51, 1240–1247. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Tuo, Q.-Z.; Lei, P. Iron, Ferroptosis, and Ischemic Stroke. J. Neurochem. 2023, 165, 487–520. [Google Scholar] [CrossRef]
- Neves, D.; Salazar, I.L.; Almeida, R.D.; Silva, R.M. Molecular Mechanisms of Ischemia and Glutamate Excitotoxicity. Life Sci. 2023, 328, 121814. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.-Q.; Zhang, L.-M.; Li, X.; Huang, Z.-H. Crosstalk between Autophagy and Inflammation in Chronic Cerebral Ischaemia. Cell. Mol. Neurobiol. 2023, 43, 2557–2566. [Google Scholar] [CrossRef]
- Yousufuddin, M.; Young, N. Aging and Ischemic Stroke. Aging 2019, 11, 2542–2544. [Google Scholar] [CrossRef]
- Yang, A.C.; Stevens, M.Y.; Chen, M.B.; Lee, D.P.; Stähli, D.; Gate, D.; Contrepois, K.; Chen, W.; Iram, T.; Zhang, L.; et al. Physiological Blood-Brain Transport Is Impaired with Age by a Shift in Transcytosis. Nature 2020, 583, 425–430. [Google Scholar] [CrossRef]
- Do, T.M.; Dodacki, A.; Alata, W.; Calon, F.; Nicolic, S.; Scherrmann, J.-M.; Farinotti, R.; Bourasset, F. Age-Dependent Regulation of the Blood-Brain Barrier Influx/Efflux Equilibrium of Amyloid-β Peptide in a Mouse Model of Alzheimer’s Disease (3xTg-AD). J. Alzheimers. Dis. 2016, 49, 287–300. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; He, J.; Huang, Y.; Hu, Z. Resveratrol Has an Overall Neuroprotective Role in Ischemic Stroke: A Meta-Analysis in Rodents. Front. Pharmacol. 2021, 12, 795409. [Google Scholar] [CrossRef] [PubMed]
- Pineda-Ramírez, N.; Gutiérrez Aguilar, G.F.; Espinoza-Rojo, M.; Aguilera, P. Current Evidence for AMPK Activation Involvement on Resveratrol-Induced Neuroprotection in Cerebral Ischemia. Nutr. Neurosci. 2018, 21, 229–247. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yu, Q.; Wu, M. Antioxidant and Neuroprotective Actions of Resveratrol in Cerebrovascular Diseases. Front. Pharmacol. 2022, 13, 948889. [Google Scholar] [CrossRef]
- Abdelsalam, S.A.; Renu, K.; Zahra, H.A.; Abdallah, B.M.; Ali, E.M.; Veeraraghavan, V.P.; Sivalingam, K.; Ronsard, L.; Ben Ammar, R.; Vidya, D.S.; et al. Polyphenols Mediate Neuroprotection in Cerebral Ischemic Stroke-An Update. Nutrients 2023, 15, 1107. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Garza, S.L.; Laveriano-Santos, E.P.; Marhuenda-Muñoz, M.; Storniolo, C.E.; Tresserra-Rimbau, A.; Vallverdú-Queralt, A.; Lamuela-Raventós, R.M. Health Effects of Resveratrol: Results from Human Intervention Trials. Nutrients 2018, 10, 1892. [Google Scholar] [CrossRef] [PubMed]
- Caddeo, C.; Pucci, L.; Gabriele, M.; Carbone, C.; Fernàndez-Busquets, X.; Valenti, D.; Pons, R.; Vassallo, A.; Fadda, A.M.; Manconi, M. Stability, Biocompatibility and Antioxidant Activity of PEG-Modified Liposomes Containing Resveratrol. Int. J. Pharm. 2018, 538, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Robinson, K.; Mock, C.; Liang, D. Pre-Formulation Studies of Resveratrol. Drug Dev. Ind. Pharm. 2015, 41, 1464–1469. [Google Scholar] [CrossRef] [PubMed]
- Walle, T.; Hsieh, F.; DeLegge, M.H.; Oatis, J.E.J.; Walle, U.K. High Absorption but Very Low Bioavailability of Oral Resveratrol in Humans. Drug Metab. Dispos. 2004, 32, 1377–1382. [Google Scholar] [CrossRef]
- Boocock, D.J.; Faust, G.E.S.; Patel, K.R.; Schinas, A.M.; Brown, V.A.; Ducharme, M.P.; Booth, T.D.; Crowell, J.A.; Perloff, M.; Gescher, A.J.; et al. Phase I Dose Escalation Pharmacokinetic Study in Healthy Volunteers of Resveratrol, a Potential Cancer Chemopreventive Agent. Cancer Epidemiol. Biomark. Prev. 2007, 16, 1246–1252. [Google Scholar] [CrossRef]
- Wang, Q.; Xu, J.; Rottinghaus, G.E.; Simonyi, A.; Lubahn, D.; Sun, G.Y.; Sun, A.Y. Resveratrol Protects against Global Cerebral Ischemic Injury in Gerbils. Brain Res. 2002, 958, 439–447. [Google Scholar] [CrossRef]
- Turner, R.S.; Thomas, R.G.; Craft, S.; Van Dyck, C.H.; Mintzer, J.; Reynolds, B.A.; Brewer, J.B.; Rissman, R.A.; Raman, R.; Aisen, P.S. A Randomized, Double-Blind, Placebo-Controlled Trial of Resveratrol for Alzheimer Disease. Neurology 2015, 85, 1383–1391. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N.J.; Patabendige, A.A.K.; Dolman, D.E.M.; Yusof, S.R.; Begley, D.J. Structure and Function of the Blood-Brain Barrier. Neurobiol. Dis. 2010, 37, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Azarmi, M.; Maleki, H.; Nikkam, N.; Malekinejad, H. Transcellular Brain Drug Delivery: A Review on Recent Advancements. Int. J. Pharm. 2020, 586, 119582. [Google Scholar] [CrossRef]
- Taylor, N.M.I.; Manolaridis, I.; Jackson, S.M.; Kowal, J.; Stahlberg, H.; Locher, K.P. Structure of the Human Multidrug Transporter ABCG2. Nature 2017, 546, 504–509. [Google Scholar] [CrossRef]
- Arvanitis, C.D.; Ferraro, G.B.; Jain, R.K. The Blood-Brain Barrier and Blood-Tumour Barrier in Brain Tumours and Metastases. Nat. Rev. Cancer 2020, 20, 26–41. [Google Scholar] [CrossRef]
- Begley, D.J. ABC Transporters and the Blood-Brain Barrier. Curr. Pharm. Des. 2004, 10, 1295–1312. [Google Scholar] [CrossRef]
- Engdahl, E.; van Schijndel, M.D.M.; Voulgaris, D.; Di Criscio, M.; Ramsbottom, K.A.; Rigden, D.J.; Herland, A.; Rüegg, J. Bisphenol A Inhibits the Transporter Function of the Blood-Brain Barrier by Directly Interacting with the ABC Transporter Breast Cancer Resistance Protein (BCRP). Int. J. Mol. Sci. 2021, 22, 5534. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, A.I.; Vallejo, F.; Barrera, B.; Merino, G.; Prieto, J.G.; Tomás-Barberán, F.; Espín, J.C. Bioavailability of the Glucuronide and Sulfate Conjugates of Genistein and Daidzein in Breast Cancer Resistance Protein 1 Knockout Mice. Drug Metab. Dispos. 2011, 39, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.C.; Rosado, T.; Costa, A.R.; Santos, J.; Gallardo, E.; Quintela, T.; Ishikawa, H.; Schwerk, C.; Schroten, H.; Gonçalves, I.; et al. The Bitter Taste Receptor TAS2R14 Regulates Resveratrol Transport across the Human Blood-Cerebrospinal Fluid Barrier. Biochem. Pharmacol. 2020, 177, 113953. [Google Scholar] [CrossRef]
- Halwachs, S.; Lakoma, C.; Schäfer, I.; Seibel, P.; Honscha, W. The Antiepileptic Drugs Phenobarbital and Carbamazepine Reduce Transport of Methotrexate in Rat Choroid Plexus by Down-Regulation of the Reduced Folate Carrier. Mol. Pharmacol. 2011, 80, 621–629. [Google Scholar] [CrossRef]
- Roberts, L.M.; Black, D.S.; Raman, C.; Woodford, K.; Zhou, M.; Haggerty, J.E.; Yan, A.T.; Cwirla, S.E.; Grindstaff, K.K. Subcellular Localization of Transporters along the Rat Blood-Brain Barrier and Blood-Cerebral-Spinal Fluid Barrier by in Vivo Biotinylation. Neuroscience 2008, 155, 423–438. [Google Scholar] [CrossRef]
- Grube, M.; Hagen, P.; Jedlitschky, G. Neurosteroid Transport in the Brain: Role of ABC and SLC Transporters. Front. Pharmacol. 2018, 9, 354. [Google Scholar] [CrossRef]
- Basseville, A.; Hall, M.D.; Chau, C.H.; Robey, R.W.; Gottesman, M.; Figg, W.D.; Bates, S.E. The ABCG2 Multidrug Transporter. In ABC Transporters—40 Years on; Springer: Berlin/Heidelberg, Germany, 2016; pp. 195–226. [Google Scholar]
- Cooray, H.C.; Blackmore, C.G.; Maskell, L.; Barrand, M.A. Localisation of Breast Cancer Resistance Protein in Microvessel Endothelium of Human Brain. Neuroreport 2002, 13, 2059–2063. [Google Scholar] [CrossRef]
- Sobiesk, J.; Munakomi, S. Anatomy, Head and Neck, Nasal Cavity; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Crowe, T.P.; Greenlee, M.H.W.; Kanthasamy, A.G.; Hsu, W.H. Mechanism of Intranasal Drug Delivery Directly to the Brain. Life Sci. 2018, 195, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Graf, P.; Eccles, R.; Chen, S. Efficacy and Safety of Intranasal Xylometazoline and Ipratropium in Patients with Common Cold. Expert Opin. Pharmacother. 2009, 10, 889–908. [Google Scholar] [CrossRef] [PubMed]
- Wattanakumtornkul, S.; Pinto, A.B.; Williams, D.B. Intranasal Hormone Replacement Therapy. Menopause 2003, 10, 88–98. [Google Scholar] [CrossRef]
- Frey William, H.I. Neurologic Agents for Nasal Administration to the Brain. WO1991007947, 13 June 1991. [Google Scholar]
- Dhuria, S.V.; Hanson, L.R.; Frey, W.H. 2nd Intranasal Delivery to the Central Nervous System: Mechanisms and Experimental Considerations. J. Pharm. Sci. 2010, 99, 1654–1673. [Google Scholar] [CrossRef] [PubMed]
- Keller, L.-A.; Merkel, O.; Popp, A. Intranasal Drug Delivery: Opportunities and Toxicologic Challenges during Drug Development. Drug Deliv. Transl. Res. 2022, 12, 735–757. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Cloyd, J.C.; Siegel, R.A. A Review of Intranasal Formulations for the Treatment of Seizure Emergencies. J. Control. release Off. J. Control. Release Soc. 2016, 237, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Juel-Berg, N.; Darling, P.; Bolvig, J.; Foss-Skiftesvik, M.H.; Halken, S.; Winther, L.; Hansen, K.S.; Askjaer, N.; Heegaard, S.; Madsen, A.R.; et al. Intranasal Corticosteroids Compared with Oral Antihistamines in Allergic Rhinitis: A Systematic Review and Meta-Analysis. Am. J. Rhinol. Allergy 2017, 31, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Ridolo, E.; Barone, A.; Nicoletta, F.; Paoletti, G.; Heffler, E.; Malvezzi, L.; Canonica, G.W. Intranasal Corticosteroid and Antihistamine Combinations in the Treatment of Allergic Rhinitis: The Role of the Novel Formulation Olopatadine/Mometasone Furoate. Expert Rev. Clin. Immunol. 2023, 19, 575–584. [Google Scholar] [CrossRef]
- Arora, P.; Sharma, S.; Garg, S. Permeability Issues in Nasal Drug Delivery. Drug Discov. Today 2002, 7, 967–975. [Google Scholar] [CrossRef]
- Oliveira, P.; Fortuna, A.; Alves, G.; Falcao, A. Drug-Metabolizing Enzymes and Efflux Transporters in Nasal Epithelium: Influence on the Bioavailability of Intranasally Administered Drugs. Curr. Drug Metab. 2016, 17, 628–647. [Google Scholar] [CrossRef] [PubMed]
- Mogollo, A.P. Importancia de La Vía Nasal Para La Administración de Opioides. Med. Paliativa 2015, 22, 33–45. [Google Scholar] [CrossRef]
- Ulusoy, S.; Muluk, N.B.; Karpischenko, S.; Passali, G.C.; Negm, H.; Passali, D.; Milkov, M.; Kopacheva-Barsova, G.; Konstantinidis, I.; Dilber, M. Mechanisms and Solutions for Nasal Drug Delivery—A Narrative Review. Eur. Rev. Med. Pharmacol. Sci. 2022, 26, 72–81. [Google Scholar] [PubMed]
- Pires, A.; Fortuna, A.; Alves, G.; Falcão, A. Intranasal Drug Delivery: How, Why and What For? J. Pharm. Pharm. Sci. 2009, 12, 288–311. [Google Scholar] [CrossRef] [PubMed]
- Machado, N.D.; Fernández, M.A.; Díaz, D.D. Recent Strategies in Resveratrol Delivery Systems. Chempluschem 2019, 84, 951–973. [Google Scholar] [CrossRef]
- Preeti; Sambhakar, S.; Malik, R.; Bhatia, S.; Al Harrasi, A.; Rani, C.; Saharan, R.; Kumar, S.; Geeta; Sehrawat, R. Nanoemulsion: An Emerging Novel Technology for Improving the Bioavailability of Drugs. Scientifica 2023, 2023, 6640103. [Google Scholar] [CrossRef]
- Shakeel, F.; Shafiq, S.; Haq, N.; Alanazi, F.K.; Alsarra, I.A. Nanoemulsions as Potential Vehicles for Transdermal and Dermal Delivery of Hydrophobic Compounds: An Overview. Expert Opin. Drug Deliv. 2012, 9, 953–974. [Google Scholar] [CrossRef]
- Robertson, I.; Wai Hau, T.; Sami, F.; Sajid Ali, M.; Badgujar, V.; Murtuja, S.; Saquib Hasnain, M.; Khan, A.; Majeed, S.; Tahir Ansari, M. The Science of Resveratrol, Formulation, Pharmacokinetic Barriers and Its Chemotherapeutic Potential. Int. J. Pharm. 2022, 618, 121605. [Google Scholar] [CrossRef]
- Johnsen, K.B.; Moos, T. Revisiting Nanoparticle Technology for Blood–Brain Barrier Transport: Unfolding at the Endothelial Gate Improves the Fate of Transferrin Receptor-Targeted Liposomes. J. Control. Release 2016, 222, 32–46. [Google Scholar] [CrossRef]
- Monsalve, Y.; Tosi, G.; Ruozi, B.; Belletti, D.; Vilella, A.; Zoli, M.; Vandelli, M.A.; Forni, F.; Lopez, B.L.; Sierra, L. PEG-g-Chitosan Nanoparticles Functionalized with the Monoclonal Antibody OX26 for Brain Drug Targeting. Nanomedicine 2015, 10, 1735–1750. [Google Scholar] [CrossRef]
- Vllasaliu, D.; Casettari, L.; Fowler, R.; Exposito-Harris, R.; Garnett, M.; Illum, L.; Stolnik, S. Absorption-Promoting Effects of Chitosan in Airway and Intestinal Cell Lines: A Comparative Study. Int. J. Pharm. 2012, 430, 151–160. [Google Scholar] [CrossRef]
- Peng, Y.; Chen, L.; Ye, S.; Kang, Y.; Liu, J.; Zeng, S.; Yu, L. Research and Development of Drug Delivery Systems Based on Drug Transporter and Nano-Formulation. Asian J. Pharm. Sci. 2020, 15, 220–236. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Minko, T. Nanotherapeutics for Nose-to-Brain Drug Delivery: An Approach to Bypass the Blood Brain Barrier. Pharmaceutics 2021, 13, 2049. [Google Scholar] [CrossRef]
- Sivadasan, D.; Sultan, M.H.; Alqahtani, S.S.; Javed, S. Cubosomes in Drug Delivery—A Comprehensive Review on Its Structural Components, Preparation Techniques and Therapeutic Applications. Biomedicines 2023, 11, 1114. [Google Scholar] [CrossRef]
- Waglewska, E.; Pucek-Kaczmarek, A.; Bazylińska, U. Novel Surface-Modified Bilosomes as Functional and Biocompatible Nanocarriers of Hybrid Compounds. Nanomaterials 2020, 10, 2472. [Google Scholar] [CrossRef]
- Mukherjee, S.; Ray, S.; Thakur, R.S. Solid Lipid Nanoparticles: A Modern Formulation Approach in Drug Delivery System. Indian J. Pharm. Sci. 2009, 71, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, M.; Saraf, S.; Saraf, S.; Dubey, S.K.; Puri, A.; Patel, R.J.; Ravichandiran, V.; Murty, U.S.; Alexander, A. Recent Strategies and Advances in the Fabrication of Nano Lipid Carriers and Their Application towards Brain Targeting. J. Control. Release 2020, 321, 372–415. [Google Scholar] [CrossRef]
- Pando, D.; Matos, M.; Gutiérrez, G.; Pazos, C. Formulation of Resveratrol Entrapped Niosomes for Topical Use. Colloids Surfaces B Biointerfaces 2015, 128, 398–404. [Google Scholar] [CrossRef]
- Pando, D.; Beltrán, M.; Gerone, I.; Matos, M.; Pazos, C. Resveratrol Entrapped Niosomes as Yoghurt Additive. Food Chem. 2015, 170, 281–287. [Google Scholar] [CrossRef]
- Vankayala, J.S.; Battula, S.N.; Kandasamy, R.; Mariya, G.A.; Franklin, M.E.E.; Pushpadass, H.A.; Naik, L.N. Surfactants and Fatty Alcohol Based Novel Nanovesicles for Resveratrol: Process Optimization, Characterization and Evaluation of Functional Properties in RAW 264.7 Macrophage Cells. J. Mol. Liq. 2018, 261, 387–396. [Google Scholar] [CrossRef]
- Caddeo, C.; Manconi, M.; Cardia, M.C.; Díez-Sales, O.; Fadda, A.M.; Sinico, C. Investigating the Interactions of Resveratrol with Phospholipid Vesicle Bilayer and the Skin: NMR Studies and Confocal Imaging. Int. J. Pharm. 2015, 484, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Caddeo, C.; Pons, R.; Carbone, C.; Fernàndez-Busquets, X.; Cardia, M.C.; Maccioni, A.M.; Fadda, A.M.; Manconi, M. Physico-Chemical Characterization of Succinyl Chitosan-Stabilized Liposomes for the Oral Co-Delivery of Quercetin and Resveratrol. Carbohydr. Polym. 2017, 157, 1853–1861. [Google Scholar] [CrossRef]
- Zhou, J.; Zhou, M.; Yang, F.F.; Liu, C.Y.; Le Pan, R.; Chang, Q.; Liu, X.M.; Liao, Y.H. Involvement of the Inhibition of Intestinal Glucuronidation in Enhancing the Oral Bioavailability of Resveratrol by Labrasol Containing Nanoemulsions. Mol. Pharm. 2015, 12, 1084–1095. [Google Scholar] [CrossRef]
- Nasr, M. Development of an Optimized Hyaluronic Acid-Based Lipidic Nanoemulsion Co-Encapsulating Two Polyphenols for Nose to Brain Delivery. Drug Deliv. 2016, 23, 1444–1452. [Google Scholar] [CrossRef]
- Fang, Q.; Ma, C.; Xia, Q. Preparation and Evaluation of Trans-Resveratrol NanoSolve. Integr. Ferroelectr. 2016, 169, 7–14. [Google Scholar] [CrossRef]
- Herneisey, M.; Williams, J.; Mirtic, J.; Liu, L.; Potdar, S.; Bagia, C.; Cavanaugh, J.E.; Janjic, J.M. Development and Characterization of Resveratrol Nanoemulsions Carrying Dual-Imaging Agents. Ther. Deliv. 2016, 7, 795–808. [Google Scholar] [CrossRef]
- Yen, C.C.; Chang, C.W.; Hsu, M.C.; Wu, Y.T. Self-Nanoemulsifying Drug Delivery System for Resveratrol: Enhanced Oral Bioavailability and Reduced Physical Fatigue in Rats. Int. J. Mol. Sci. 2017, 18, 1853. [Google Scholar] [CrossRef]
- Mamadou, G.; Charrueau, C.; Dairou, J.; Limas Nzouzi, N.; Eto, B.; Ponchel, G. Increased Intestinal Permeation and Modulation of Presystemic Metabolism of Resveratrol Formulated into Self-Emulsifying Drug Delivery Systems. Int. J. Pharm. 2017, 521, 150–155. [Google Scholar] [CrossRef]
- Yang, F.F.; Zhou, J.; Hu, X.; Cong, Z.Q.; Liu, C.Y.; Le Pan, R.; Chang, Q.; Liu, X.M.; Liao, Y.H. Improving Oral Bioavailability of Resveratrol by a UDP-Glucuronosyltransferase Inhibitory Excipient-Based Self-Microemulsion. Eur. J. Pharm. Sci. 2018, 114, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Matos, M.; Laca, A.; Rea, F.; Iglesias, O.; Rayner, M.; Gutiérrez, G. O/W Emulsions Stabilized by OSA-Modified Starch Granules versus Non-Ionic Surfactant: Stability, Rheological Behaviour and Resveratrol Encapsulation. J. Food Eng. 2018, 222, 207–217. [Google Scholar] [CrossRef]
- Dai, L.; Li, Y.; Kong, F.; Liu, K.; Si, C.; Ni, Y. Lignin-Based Nanoparticles Stabilized Pickering Emulsion for Stability Improvement and Thermal-Controlled Release of Trans-Resveratrol. ACS Sustain. Chem. Eng. 2019, 7, 13497–13504. [Google Scholar] [CrossRef]
- Choi, A.-J.; Jo, Y.; Cho, Y.-J.; Kim, T.-E.; Kim, C.-T. Preparation and Characterization of Resveratrol Nanoemulsions Stabilized by Self-Assembly and Complex Coacervation Consisting of Sodium Alginate, Chitosan, and β-Cyclodextrin. Food Eng. Prog. 2017, 21, 215–224. [Google Scholar] [CrossRef]
- Matos, M.; Gutiérrez, G.; Iglesias, O.; Coca, J.; Pazos, C. Enhancing Encapsulation Efficiency of Food-Grade Double Emulsions Containing Resveratrol or Vitamin B12 by Membrane Emulsification. J. Food Eng. 2015, 166, 212–220. [Google Scholar] [CrossRef]
- Acevedo-Fani, A.; Silva, H.D.; Soliva-Fortuny, R.; Martín-Belloso, O.; Vicente, A.A. Formation, Stability and Antioxidant Activity of Food-Grade Multilayer Emulsions Containing Resveratrol. Food Hydrocoll. 2017, 71, 207–215. [Google Scholar] [CrossRef]
- Matos, M.; Gutiérrez, G.; Martínez-Rey, L.; Iglesias, O.; Pazos, C. Encapsulation of Resveratrol Using Food-Grade Concentrated Double Emulsions: Emulsion Characterization and Rheological Behaviour. J. Food Eng. 2018, 226, 73–81. [Google Scholar] [CrossRef]
- Scalia, S.; Trotta, V.; Iannuccelli, V.; Bianchi, A. Enhancement of in Vivo Human Skin Penetration of Resveratrol by Chitosan-Coated Lipid Microparticles. Colloids Surfaces B Biointerfaces 2015, 135, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Pangeni, R.; Sharma, S.; Mustafa, G.; Ali, J.; Baboota, S. Vitamin e Loaded Resveratrol Nanoemulsion for Brain Targeting for the Treatment of Parkinson’s Disease by Reducing Oxidative Stress. Nanotechnology 2014, 25, 485102. [Google Scholar] [CrossRef]
- Abbas, H.; Refai, H.; El Sayed, N.; Rashed, L.A.; Mousa, M.R.; Zewail, M. Superparamagnetic Iron Oxide Loaded Chitosan Coated Bilosomes for Magnetic Nose to Brain Targeting of Resveratrol. Int. J. Pharm. 2021, 610, 121244. [Google Scholar] [CrossRef]
- Ahirrao, M.; Shrotriya, S. In Vitro and in Vivo Evaluation of Cubosomal in Situ Nasal Gel Containing Resveratrol for Brain Targeting. Drug Dev. Ind. Pharm. 2017, 43, 1686–1693. [Google Scholar] [CrossRef] [PubMed]
- Salem, H.F.; Kharshoum, R.M.; Abou-Taleb, H.A.; Naguib, D.M. Brain Targeting of Resveratrol through Intranasal Lipid Vesicles Labelled with Gold Nanoparticles: In Vivo Evaluation and Bioaccumulation Investigation Using Computed Tomography and Histopathological Examination. J. Drug Target. 2019, 27, 1127–1134. [Google Scholar] [CrossRef]
- Rajput, A.; Bariya, A.; Allam, A.; Othman, S.; Butani, S.B. In Situ Nanostructured Hydrogel of Resveratrol for Brain Targeting: In Vitro-in Vivo Characterization. Drug Deliv. Transl. Res. 2018, 8, 1460–1470. [Google Scholar] [CrossRef]
- Trotta, V.; Pavan, B.; Ferraro, L.; Beggiato, S.; Traini, D.; Des Reis, L.G.; Scalia, S.; Dalpiaz, A. Brain Targeting of Resveratrol by Nasal Administration of Chitosan-Coated Lipid Microparticles. Eur. J. Pharm. Biopharm. 2018, 127, 250–259. [Google Scholar] [CrossRef]
- Hao, J.; Zhao, J.; Zhang, S.; Tong, T.; Zhuang, Q.; Jin, K.; Chen, W.; Tang, H. Fabrication of an Ionic-Sensitive in Situ Gel Loaded with Resveratrol Nanosuspensions Intended for Direct Nose-to-Brain Delivery. Colloids Surfaces B Biointerfaces 2016, 147, 376–386. [Google Scholar] [CrossRef]
- Salem, H.F.; Kharshoum, R.M.; Abou-Taleb, H.A.; Naguib, D.M. Nanosized Transferosome-Based Intranasal In Situ Gel for Brain Targeting of Resveratrol: Formulation, Optimization, In Vitro Evaluation, and In Vivo Pharmacokinetic Study. AAPS PharmSciTech 2019, 20, 181. [Google Scholar] [CrossRef]
- Kim, Y.; Hwang, S.; Khalmuratova, R.; Kang, S.; Lee, M.; Song, Y.; Park, J.W.; Yu, J.; Shin, H.W.; Lee, Y. α-Helical Cell-Penetrating Peptide-Mediated Nasal Delivery of Resveratrol for Inhibition of Epithelial-to-Mesenchymal Transition. J. Control. Release 2020, 317, 181–194. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).