Literature Review of Safety Event Reporting in Observational Studies: Challenges Extrapolating across Comparable Products
Abstract
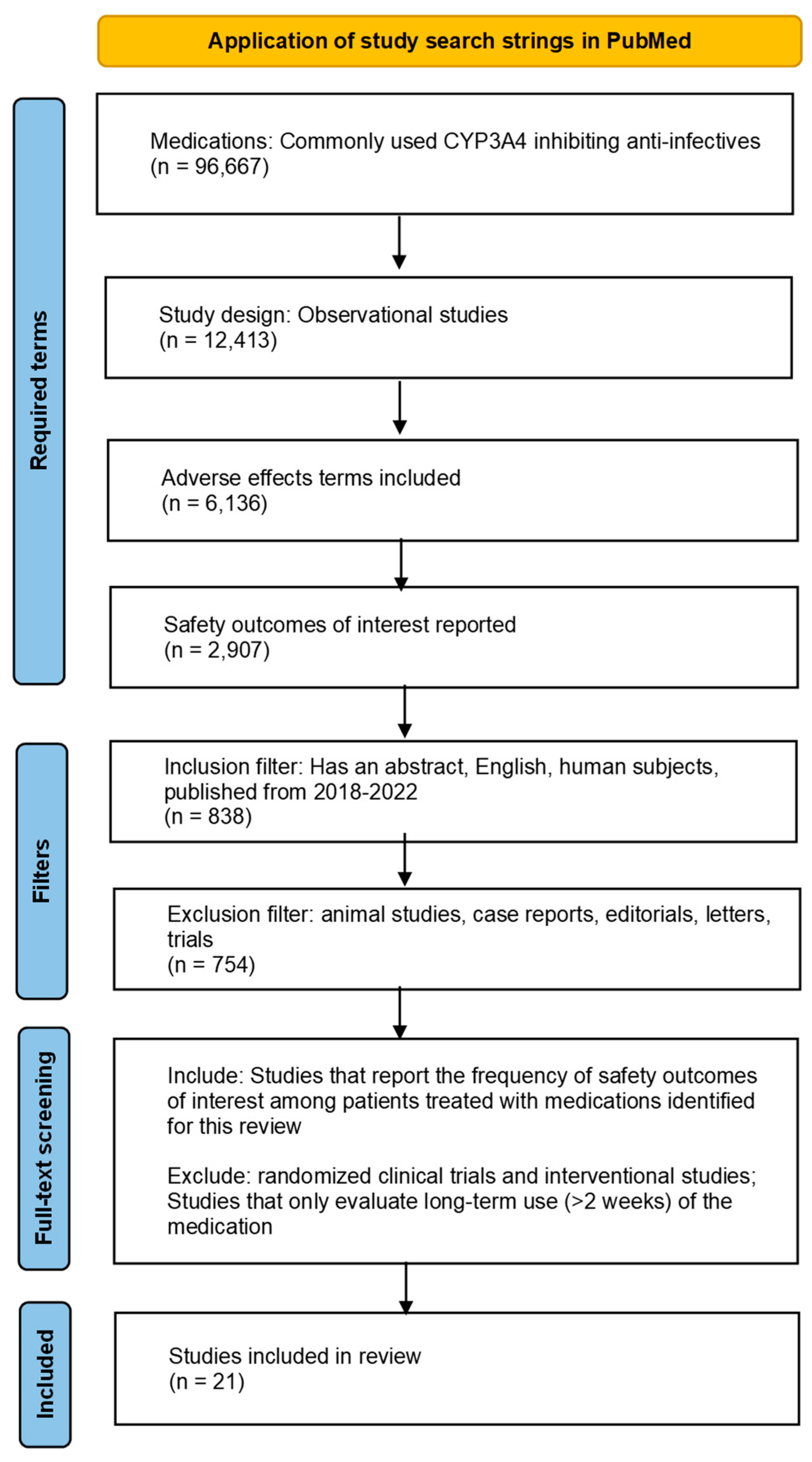
:1. Background and Rationale
2. Results
3. Discussion
3.1. Outcome Generalizability
3.2. Patient Representativeness
3.3. Setting
3.4. Exposure Definition
4. Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Crellin, E.; Mansfield, K.E.; Leyrat, C.; Nitsch, D.; Douglas, I.J.; Root, A.; Williamson, E.; Smeeth, L.; Tomlinson, L.A. Trimethoprim use for urinary tract infection and risk of adverse outcomes in older patients: Cohort study. BMJ 2018, 360, k341. [Google Scholar] [CrossRef] [PubMed]
- Inghammar, M.; Nibell, O.; Pasternak, B.; Melbye, M.; Svanström, H.; Hviid, A. Long-term risk of cardiovascular death with use of clarithromycin and roxithromycin: A nationwide cohort study. Am. J. Epidemiol. 2018, 187, 777–785. [Google Scholar] [CrossRef]
- Jödicke, A.M.; Curkovic, I.; Zellweger, U.; Tomka, I.T.; Neuer, T.; Kullak-Ublick, G.A.; Roos, M.; Egbring, M. Analysis of drug-drug interactions in Swiss claims data using tizanidine and ciprofloxacin as a prototypical contraindicated combination. Ann. Pharmacother. 2018, 52, 983–991. [Google Scholar] [CrossRef]
- Latif, S.; Akther, N.; Amjed, S.; Jafar, J.; Saleem, B.; Usman, M.; Amjad, T.; Maqsood, W.; Waseem, H.; Ashraf, N. Efficacy of standard triple therapy versus levofloxacin based alternate therapy against Helicobacter pylori infection. J. Pak. Med. Assoc. 2018, 68, 1295–1299. [Google Scholar] [PubMed]
- Mosholder, A.D.; Lee, J.Y.; Zhou, E.H.; Kang, E.M.; Ghosh, M.; Izem, R.; Major, J.M.; Graham, D.J. Long-term risk of acute myocardial infarction, stroke, and death with outpatient use of clarithromycin: A retrospective cohort study. Am. J. Epidemiol. 2018, 187, 786–792. [Google Scholar] [CrossRef] [PubMed]
- Pasternak, B.; Inghammar, M.; Svanström, H. Fluoroquinolone use and risk of aortic aneurysm and dissection: Nationwide cohort study. BMJ 2018, 360, k678. [Google Scholar] [CrossRef]
- Pasternak, B.; Wintzell, V.; Furu, K.; Engeland, A.; Neovius, M.; Stephansson, O. Oral fluconazole in pregnancy and risk of stillbirth and neonatal death. JAMA 2018, 319, 2333–2335. [Google Scholar] [CrossRef]
- Bérard, A.; Sheehy, O.; Zhao, J.P.; Gorgui, J.; Bernatsky, S.; de Moura, C.S.; Abrahamowicz, M. Associations between low- and high-dose oral fluconazole and pregnancy outcomes: 3 nested case-control studies. CMAJ 2019, 191, E179–E187. [Google Scholar] [CrossRef]
- Jeong, R.; Quinn, R.R.; Lentine, K.L.; Lloyd, A.; Ravani, P.; Hemmelgarn, B.; Braam, B.; Garg, A.X.; Wen, K.; Wong-Chan, A.; et al. Outcomes following macrolide use in kidney transplant recipients. Can. J. Kidney Health Dis. 2019, 6, 2054358119830706. [Google Scholar] [CrossRef]
- Williamson, E.; Denaxas, S.; Morris, S.; Clarke, C.S.; Thomas, M.; Evans, H.; Direk, K.; Gonzalez-Izquierdo, A.; Little, P.; Lund, V.; et al. Risk of mortality and cardiovascular events following macrolide prescription in chronic rhinosinusitis patients: A cohort study using linked primary care electronic health records. Rhinology 2019, 57, 252–260. [Google Scholar] [CrossRef]
- Baik, S.; Lau, J.; Huser, V.; McDonald, C.J. Association between tendon ruptures and use of fluoroquinolone, and other oral antibiotics: A 10-year retrospective study of 1 million US senior Medicare beneficiaries. BMJ Open 2020, 10, e034844. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.; Sucha, E.; Rhodes, E.; Carrier, M.; Garg, A.X.; Harel, Z.; Hundemer, G.L.; Clark, E.G.; Knoll, G.; McArthur, E.; et al. Risk of hospitalization with hemorrhage among older adults taking clarithromycin vs azithromycin and direct oral anticoagulants. JAMA Intern. Med. 2020, 180, 1052–1060. [Google Scholar] [CrossRef] [PubMed]
- Mordi, I.R.; Chan, B.K.; Yanez, N.D.; Palmer, C.N.A.; Lang, C.C.; Chalmers, J.D. Genetic and pharmacological relationship between P-glycoprotein and increased cardiovascular risk associated with clarithromycin prescription: An epidemiological and genomic population-based cohort study in Scotland, UK. PLoS Med. 2020, 17, e1003372. [Google Scholar] [CrossRef]
- Fung, K.W.; Baye, F.; Kapusnik-Uner, J.; McDonald, C.J. Using medicare data to assess the proarrhythmic risk of non-cardiac treatment drugs that prolong the QT interval in older adults: An observational cohort study. Drugs Real. World Outcomes 2021, 8, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Noergaard, M.; Gotfredsen, D.R.; Sørensen, A.M.S.; Andersen, J.T. Ciprofloxacin exposure and adverse pregnancy outcomes: A Danish nationwide cohort study. BJOG 2022, 129, 1503–1511. [Google Scholar] [CrossRef]
- Assimon, M.M.; Pun, P.H.; Wang, L.C.; Al-Khatib, S.M.; Brookhart, M.A.; Weber, D.J.; Winkelmayer, W.C.; Flythe, J.E. Analysis of respiratory fluoroquinolones and the risk of sudden cardiac death among patients receiving hemodialysis. JAMA Cardiol. 2022, 7, 75–83. [Google Scholar] [CrossRef]
- Liao, S.H.; Hu, S.Y.; How, C.K.; Hsieh, V.C.; Chan, C.M.; Chiu, C.S.; Hsieh, M.S. Risk for hypoglycemic emergency with levofloxacin use, a population-based propensity score matched nested case-control study. PLoS ONE 2022, 17, e0266471. [Google Scholar] [CrossRef]
- Muanda, F.T.; Sood, M.M.; Weir, M.A.; Sontrop, J.M.; Ahmadi, F.; Yoo, E.; Kim, R.B.; Silverman, M.S.; Knoll, G.A.; Garg, A.X. Association of higher-dose fluoroquinolone therapy with serious adverse events in older adults with advanced chronic kidney disease. JAMA Netw. Open 2022, 5, e2224892. [Google Scholar] [CrossRef]
- Tan, M.S.; Gomez-Lumbreras, A.; Villa-Zapata, L.; Malone, D.C. Colchicine and macrolides: A cohort study of the risk of adverse outcomes associated with concomitant exposure. Rheumatol. Int. 2022, 42, 2253–2259. [Google Scholar] [CrossRef]
- Wang, J.; Gagne, J.J.; Kattinakere-Sreedhara, S.; Fischer, M.A.; Bykov, K. Association between initiation of fluoroquinolones and hospital admission or emergency department visit for suicidality: Population based cohort study. BMJ 2022, 379, e069931. [Google Scholar] [CrossRef]
- Yu, B.H.; Chen, Y.C.; Li, Y.D.; Chiou, W.Y.; Chen, Y.C. No dose-response relationship of clarithromycin utilization on cardiovascular outcomes in patients with stable coronary heart disease: Analysis of Taiwan’s national health insurance claims data. Front. Cardiovasc. Med. 2022, 9, 1018194. [Google Scholar] [CrossRef] [PubMed]
| Author Year; Location; Study Years; Study Design. | Population | Exposure (Drug) | Data Source | Safety Events and Outcomes of Interest | Frequency of Safety Outcomes of Interest: % Exposed Experiencing Event and/or Comparative Analysis If Provided |
|---|---|---|---|---|---|
| Crellin 2018 [1]; UK; 1997–2015; Retrospective cohort | Patients aged 65+ y with UTI in UK CPRD and HES database | CIP, n = 15,594; AMX, n = 17,536 | Rx for an antibiotic for a UTI after the latest of the following: 65th birthday; date of medical practice was data certified; or 1 y after practice registration date | Events with 14 d of therapy: 1. Acute kidney injury 2. Hyperkalemia 3. Death | Event frequency: CIP; AMX 1. Acute kidney injury 0.4%; 0.3% 2. Hyperkalemia 0.1%; 0.1% 3. Deaths 0.6%; 0.7% OR (95% CI), CIP vs. AMX: 1. Acute kidney injury 1.48 (1.03–2.13) 2. Hyperkalemia 1.17 (0.68–2.02) 3. Deaths 0.92 (0.73–1.15) |
| Inghammar 2018 [2]; Denmark; 1997–2011; Retrospective cohort | Patients aged 40–74 y in Denmark with infections eligible for study antibiotics | CLR, n = 187,887; Roxithromycin, n = 698,899; PEN controls, n = 3,473,081 | Rx data from the Danish National Prescription Registry. Patients can contribute >1 course of therapy | CV death | Safety event within 0–7 d of initiation: CLR 5.0/1000 p-y, RR 1.66 vs. PEN Roxithromycin 1.9/1000 p-y, RR 0.88 vs. PEN |
| Jödicke 2018 [3]; Switzerland; 2014–2015; Retrospective cohort | Patients in Swiss claims database | CIP + tizanidine (coprescribed within a 7-d period), n = 199; Other antibiotic + tizanidine, n = 960 | Rx claims data from large insurance company | Hospitalization event at 7, 14, and 30 d after co-Rx of interacting drugs | % experiencing safety event; OR (95% CI): 7 d: 4.0%; 2.19 (0.88–5.02) 14 d: 4.0%; 1.52 (0.63–3.33) 30 d: 6.5%; 1.68 (0.84–3.17) |
| Latif 2018 [4]; Pakistan; 2016–2017; Prospective cohort | Patients with H. pylori infection | CLR + AMX + omeprazole, n = 150; LVX + AMX + omeprazole, n = 150 | Study drugs were provided to participants [1] | Signs or symptoms observed or reported at d 7 and 14 (no safety outcome type specified) | 0% No safety outcomes reported in either group |
| Mosholder 2018 [5]; UK; 2000–2013; Retrospective cohort | Patients aged 40–85 y, enrolled in a primary care database, no stroke or MI up to 90 d before Rx date | CLR, n = 287,748; DOX, n = 267,729; ERY, n = 442,999 | Rx for study drug the UK CPRD from 2000 to 2013; patients needed to have a 1-y minimum washout for Rx of any study drugs | 1. ACM 2. AMI 3. Stroke | 1. ACM: CLR 7.9%, DOX 4.2%, ERY 7.3%; HR (95% CI): CLR vs. DOX 1.23 (1.20–1.25); CLR vs. ERY 1.13 (1.11–1.15); ERY vs. DOX 1.09 (1.06–1.11) 2. AMI (% not reported); HR (95% CI): CLR vs. DOX 1.13 (1.06–1.20); CLR vs. ERY 1.03 (0.98–1.07); ERY vs. DOX 1.10 (1.04–1.17) 3. Stroke (% not reported); HR (95% CI): CLR vs. DOX 1.15 (1.08–1.22); CLR vs. ERY 1.04 (1.00–1.09); ERY vs. DOX 1.10 (1.04–1.17) |
| Pasternak 2018 [6]; Sweden, 2006–2013; Retrospective cohort | Patients aged ≥ 50 y in Swedish registry | Oral FLQ, n = 360,088; AMX, n = 360,088 | Outpatient Rx dispensing | Aortic aneurysm or dissection | FLQ: 0.02%; incidence 1.2/1000 p-y; AMX: 0.01%; incidence 0.7/1000 p-y |
| Pasternak 2018 [7]; Sweden and Norway; 2006–2015; Retrospective cohort | Pregnancies with singleton live births and stillbirths from national registry data | FLZ, n = 10,669; Non-FLZ, n = 106,690 | FLZ Rx within 28 d before conception | Stillbirth or neonatal death | FLZ: 0.27% Non-FLZ: 0.35% |
| Berard 2019 [8]; Canada; 1998–2015; Case-control, nested | Pregnant women with outcome of SA (n = 320,868) or MCM (n = 226,599) | FLZ low dose ≤ 150 mg (SA cases n = 345, controls n = 1356; MCM cases n = 92, controls n = 821) FLZ high dose > 150 mg (SA cases n = 249, controls n = 642; MCM cases n = 50, controls n = 350) | Rx for oral FLZ filled from the Quebec Prescription Drug Insurance database, using dispensing date and duration of treatment For MCM, exposure was in the first trimester only | 1. SA 2. MCM in the first 6 mo | 1. SA: FLZ low dose: 1.2%, OR 2.23 (1.96–2.54) FLZ high dose: 0.9%, OR 3.20 (2.73–3.75) 2. MCM: FLZ low dose: 0.5%, OR 1.08 (0.87–1.34) FLZ high dose: 0.3%, OR 1.30 (0.97–1.75) |
| Jeong 2019 [9]; Canada; 2008–2015; Retrospective cohort | Outpatient kidney transplant recipients (≥1 y after transplant) | Among patients, coRx calcineurin inhibitors: CLR or ERY, n = 112; AZM, n = 181 | Pharmaceutical Information Network containing Rx data in Alberta, Canada | Within 30 d of coRx: 1. Composite of all-cause hospitalization, acute kidney injury, or ACM 2. All-cause hospitalization 3. Acute kidney injury (as ≥0.3 mg/dL serum creatinine increase or 1.5 times baseline) | 1. Composite outcome: CLR/ERY 17%, AZM 10.5%; OR 1.74 (95% CI, 0.88–3.46) 2. Hospitalization: CLR/ERY 9.8%, AZM 3.3%; OR 3.18 (95% CI, 1.14–8.84) 3. Acute kidney injury: CLR/ERY 14.3%, AZM 9.4%; OR 1.61 (95% CI, 0.78–3.33) |
| Williamson 2019 [10]; UK; 1997–2016; Retrospective cohort | Patients with chronic rhinosinusitis, ≥1 Rx of MAC or PEN in CPRD, HES databases | MAC, n = 12,833; CLR, n = 5299; PEN, n = 53,498 | Pharmacy dispensing data | 1. ACM 2. Cardiac death 3. MI 4. Stroke 5. PVD 6. CA | 1. ACM: MAC 4.7%; CLR 3.9%; PEN 5.0%. 2. Cardiac death: MAC 0.9%; CLR 0.6%; PEN 1.3% 3. MI: MAC 1.4%; CLR 1.0%; PEN 1.4% 4. Stroke: MAC 1.2%; CLR 0.9%; PEN 1.3% 5. PVD: MAC 1.0%; CLR 0.7%; PEN 1.0% 6. CA: MAC 2.2%; CLR 1.6%; PEN 2.1% |
| Baik 2020 [11]; USA; 2007–2016; Retrospective cohort | Medicare patients with inpatient and outpatient claims | CIP, n = 234,994; LVX, n = 155,991; MOXI, n = 14,728 | Medicare Part D outpatient Rx drug records 2007–2016, exposure from dispensing date to the end of days of supply | Tendon rupture; 10-y rates post-Rx | CIP 3.7% LVX 3.8% MOXI 5.2% No antibiotic use 2.9% |
| Hill 2020 [12]; Canada; 2009–2016; Retrospective cohort | Outpatients taking an anticoagulant in linked databases for Ontario region | CLR, n = 6592; AZM, n =18,351 | Outpatient Rx dispensing records for all adults >65 y, with an error rate of <1% | Major bleeding events leading to hospitalization within 30 d of antibiotic dispensing | CLR: 0.77% AZM: 0.43% Adjusted HR (95% CI): 1.71 (1.20–2.45) |
| Mordi 2020 [13]; Scotland; 2004–2014; Retrospective cohort | Patients participating in the Genetics of Diabetes Audit and Research Tayside Scotland study | Total unique patients: CLR, n = 11,489 AMX, n = 36,537 Total prescriptions: CLR, n = 34,074 AMX, n = 171,153 | Database of community prescribing data | 1. CV hospitalization 2. Hospitalization for MI 3. CV mortality 4. ACM | At 0–14 d, 15–30 d, and 30 d–1 y, respectively: 1. CV Hospitalization: CLR, 1.6%, 1.3%, 5.4%; AMX, 1.4%, 1.2%, 6.6% 2. Hospitalization for MI: CLR, 0.1%, 0.07%, 0.5%; AMX, 0.1%, 0.08%, 0.6% 3. CV mortality: CLR, 0.2%, 0.2%, 1.5%; AMX, 0.3%, 2.3%, 1.0% 4. ACM: CLR, 0.8%, 0.8%, 4.5%; AMX, 1.0%, 1.0%, 5.8% |
| Fung 2021 [14]; USA; 2007–2016; Retrospective cohort | Medicare patients with inpatient and outpatient claims | CIP n = 343,320; LVX n = 239,083; MOXI n = 26,528; AZM n = 446,943; CLR n = 54,805; ERY n = 11,695; FLZ n = 116,539 | Medicare Part D Rx claim with dispensing date, with short-term use cutoff at median 30 d supply | VA or SD | HR (95% CI) for VA/SD vs. nonusers: LVX: 1.51 (1.44–1.57) MOXI: 1.23 (1.03–1.45) ERY: 1.63 (1.32–2.02) FLZ: 2.23 (2.15–2.32) |
| Noergaard 2021 [15]; Denmark; 1997–2016; Retrospective cohort | All registered pregnancies in a nationwide register, 1 January 1997, to 31 December 2016 | CIP: ≥1 Rx for systemic CIP within the first 22 wk (miscarriage) or first 12 wk (MCM) of pregnancy, n = 2050 Non-CIP: no Rx of any quinolones up to 3 mo before the LMP date to the pregnancy end date, n = 8200 | Medical Birth Registry, the National Hospital Registry, the Danish National Prescription Registry and Statistics Denmark | 1. Miscarriage 2. MCM | 1. CIP: 8.3%, non-CIP: 8.7% 2. CIP: 3.4%, non-CIP, 3.4% |
| Assimon 2022 [16]; USA; 2007–2017; Retrospective cohort | Patients with hemodialysis-dependent kidney failure | AZM, n = 188,871; LVX, n = 110,230 | Outpatient study antibiotic Rx fills in the US Renal Data System. Treatment episodes = 180-d baseline, 30-d washout, and 10-d follow-up period | Sudden cardiac death: death due to CA or CA listed as the primary cause | Treatment d 1–5 AZM: 0.13%, 12.8/100K p-d LVX: 0.16%, 20.1/100K p-d Days 6–10 AZM: 0.11%, 10.4/100K p-d LVX: 0.13%, 17.8 100K p-d |
| Liao 2022 [17]; Taiwan; 1999–2013; Retrospective cohort (nested case–control) | Patients with T2D | Cephalosporins, n = 2212; PEN, n = 220; FLQ, n = 909; MAC, n = 131; Sulfonamide, n = 514; Tetracycline, n = 128; Metronidazole, n = 82 | Taiwan NHIRD; Nationwide database includes all prescribed antibiotics | Up to 7 d after oral antibiotic use: Hypoglycemic emergency (hypoglycemia diagnosed in the emergency department) | Adjusted OR (95% CI): Cephalosporins 6.12 (5.74–6.52), PEN 3.10 (2.61–3.69), FLQ 12.05 (10.66–13.61), MAC 6.85 (5.91–7.96), Sulfonamide 7.20 (6.29–8.24), Tetracycline 2.13 (1.71–2.64), Metronidazole 3.64 (2.68–4.94) |
| Muanda 2022 [18]; Canada; 2008–2020; Retrospective cohort | Adults aged ≥66 y with advanced CKD | New Rx for a single oral FLQ (CIP, LVX, or norfloxacin) dispensed from an outpatient pharmacy; High-dose FLQ, n = 5482; Low-dose FLQ, n = 5516 | Linked administrative healthcare databases in Ontario, Canada; excluded patients discharged from the hospital or emergency department within 2 d before the index date of Rx | Composite of a hospital visit with nervous system and/or psychiatric disorders, hypoglycemia, or collagen-associated events within 14 d of starting a new fluoroquinolone Rx | High dose: 1.2%; Low dose: 0.9%; High dose vs. low dose fluoroquinolone, weighted RR 1.45 (1.01–2.08) |
| Tan 2022 [19]; USA; 2002–2018; Retrospective cohort | Patients aged ≥ 18 y, receiving colchicine and either a macrolide or nonmacrolide antibiotic | cMAC, n = 2199; Non-cMAC, n = 12,670 | CERNER Health Facts EMR database | 1. Rhabdomyolysis 2. Pancytopenia 3. Muscular weakness 4. Heart failure 5. Acute hepatic failure 6. ACM | 1. Rhabdomyolysis: cMAC 0.8%; Non-cMAC 0.7% 2. Pancytopenia: cMAC 4.7%; Non-cMAC 5% 3. Muscle weakness: cMAC 0.3%; Non-cMAC 0.3% 4. Heart failure: cMAC 18.3%; Non-cMAC 9.1% 5. Acute hepatic failure: cMAC 0.9%; Non-cMAC 0.7% 6. ACM: cMAC 3.9%; Non-cMAC 2.3% |
| Wang 2022 [20] USA; 2003–2015; Retrospective cohort | Adults aged ≥18 y, initiated an oral FLQ or comparator antibiotic, and diagnosed with pneumonia or UTI 3 d before drug initiation | Pneumonia cohort (matched): FLQ (CIP, LVX, MOXI, other), n = 275,521; AZM comparator, n = 275,521 UTI cohort (matched): FLQ (CIP, LVX, MOXI, other), n = 1,102,613; TMP-SMX comparator, n = 1,102,613 | Pharmacy dispensing data; claims and encounters from US health insurance claims database (IBM Marketscan) | Hospital admission or emergency department visit for suicidal ideation or self-harm within 60 d after treatment initiation | Pneumonia cohort: FLQ: 0.03%; AZM: 0.03% HR (95% CI) 1.01 (0.76–1.36) UTI cohort: FLQ 0.04%; TMP-SMX 0.04% HR (95% CI) 1.03 (0.91–1.17) |
| Yu 2022 [21]; Taiwan; 2000–2016; Retrospective cohort | Patients diagnosed with coronary heart disease | CLR, n = 9631; non-CLR, n = 9631 | Taiwan Longitudinal Generation Tracking Database; new prescription for CLR propensity matched by date with nonusers | 1. ACM; 2. CV mortality; 3. Non-CV mortality | 1. ACM: CLR 8.9%; non-CLR 7.0% 2. CV mortality: CLR 2.0%; non-CLR 1.7% 3. Non-CV mortality: CLR 6.9%; Non-CLR 5.3% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ward, H.A.; Nguyen-Khoa, B.-A.; Massouh, R. Literature Review of Safety Event Reporting in Observational Studies: Challenges Extrapolating across Comparable Products. Pharmacoepidemiology 2023, 2, 338-349. https://doi.org/10.3390/pharma2040029
Ward HA, Nguyen-Khoa B-A, Massouh R. Literature Review of Safety Event Reporting in Observational Studies: Challenges Extrapolating across Comparable Products. Pharmacoepidemiology. 2023; 2(4):338-349. https://doi.org/10.3390/pharma2040029
Chicago/Turabian StyleWard, Heather A., Bao-Anh Nguyen-Khoa, and Robert Massouh. 2023. "Literature Review of Safety Event Reporting in Observational Studies: Challenges Extrapolating across Comparable Products" Pharmacoepidemiology 2, no. 4: 338-349. https://doi.org/10.3390/pharma2040029
APA StyleWard, H. A., Nguyen-Khoa, B.-A., & Massouh, R. (2023). Literature Review of Safety Event Reporting in Observational Studies: Challenges Extrapolating across Comparable Products. Pharmacoepidemiology, 2(4), 338-349. https://doi.org/10.3390/pharma2040029






