Twin-Screw Extrusion Mechanical Pretreatment for Enhancing Biomethane Production from Agro-Industrial, Agricultural and Catch Crop Biomasses
Abstract
1. Introduction
2. Materials and Methods
2.1. Feedstocks and Inoculum
2.2. Extrusion Pretreatment
2.3. Biochemical Methane Potential (BMP)
2.4. Analytical Methods
2.4.1. Sample Preparation for Analysis
2.4.2. Fiber Composition
2.4.3. Granulometry and Tapped Density
2.4.4. Cellulose Crystallinity
2.5. Data Analyses
2.6. Statistical Analyses
3. Results and Discussion
3.1. Description of the Twin-Screw Extrusion Pretreatments
3.2. Effect of the Twin-Screw Extrusion Mechanical Pretreatment on the Chemical Composition of Biomasses
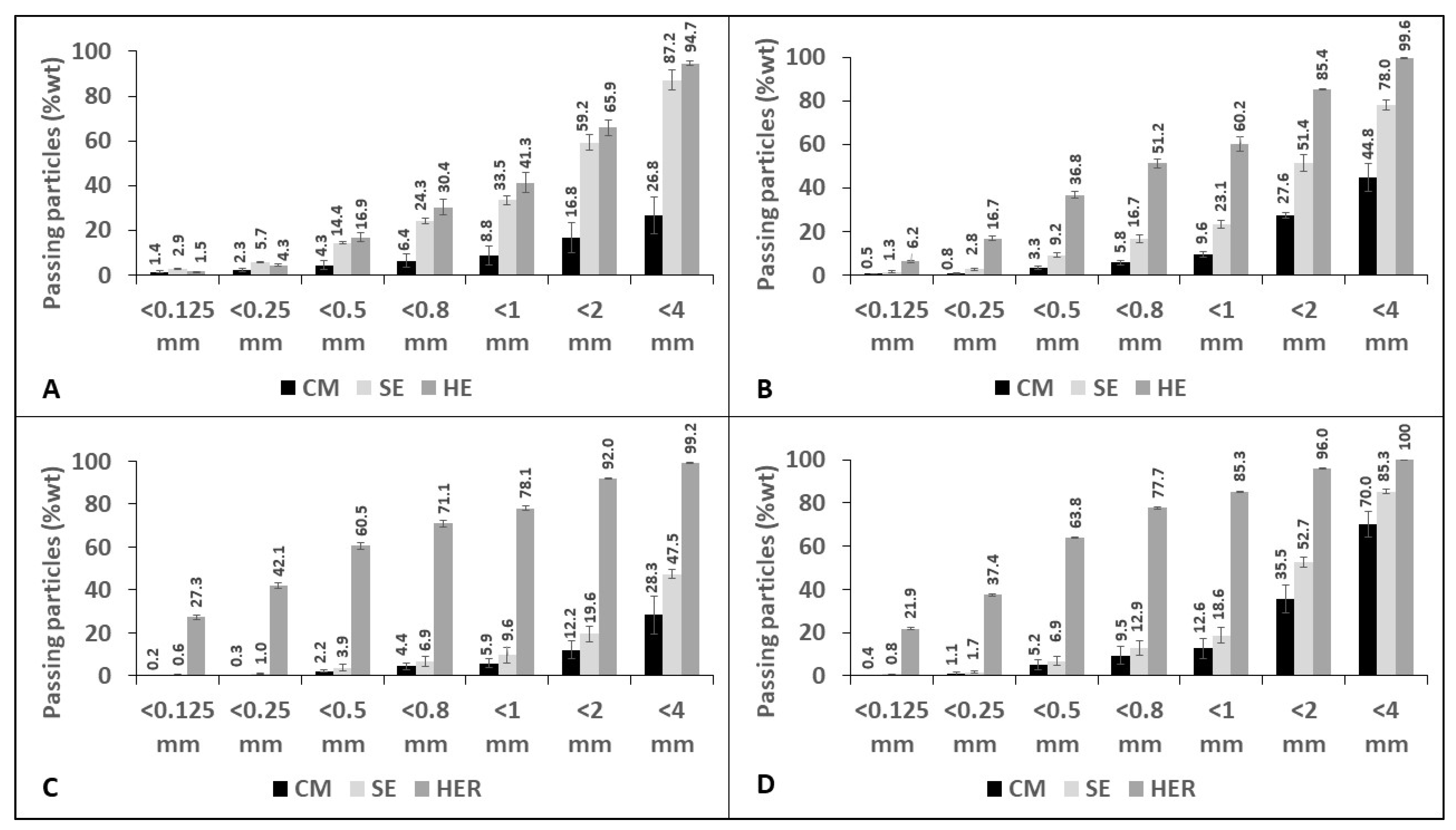
3.3. Effect of the Twin-Screw Extrusion Mechanical Pretreatment on Granulometry
3.4. Effect of the Twin-Screw Extrusion Mechanical Pretreatment on Tapped Density
3.5. Effect of the Twin-Screw Extrusion Mechanical Pretreatment on Cellulose Crystallinity
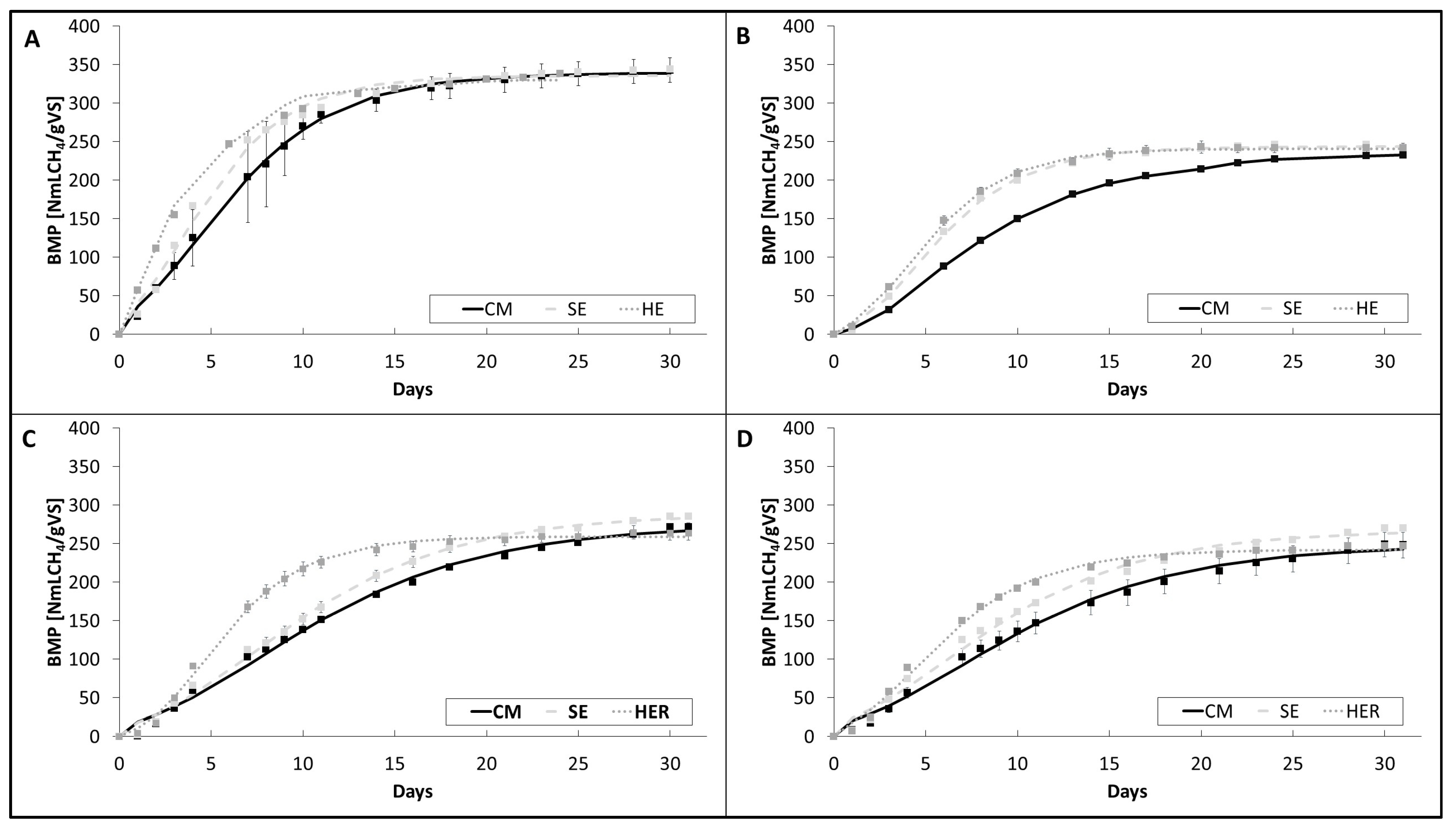
3.6. Effect of the Twin-Screw Extrusion Mechanical Pretreatment on BMP Results
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abas, N.; Kalair, A.; Khan, N. Review of fossil fuels and future energy technologies. Futures 2015, 69, 31–49. [Google Scholar] [CrossRef]
- Li, Y.; Chen, Y.; Wu, J. Enhancement of methane production in anaerobic digestion process: A review. Appl. Energy 2019, 240, 120–137. [Google Scholar] [CrossRef]
- Ampese, L.C.; Sganzerla, W.G.; Di Domenico Ziero, H.; Mudhoo, A.; Martins, G.; Forster-Carneiro, T. Research progress, trends, and updates on anaerobic digestion technology: A bibliometric analysis. J. Clean. Prod. 2022, 331, 130004. [Google Scholar] [CrossRef]
- Konan, D.; Koffi, E.; Ndao, A.; Peterson, E.C.; Rodrigue, D.; Adjallé, K. An Overview of Extrusion as a Pretreatment Method of Lignocellulosic Biomass. Energies 2022, 15, 3002. [Google Scholar] [CrossRef]
- Hendriks, A.T.W.M.; Zeeman, G. Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour. Technol. 2009, 100, 10–18. [Google Scholar] [CrossRef]
- Tye, Y.Y.; Lee, K.T.; Wan Abdullah, W.N.; Leh, C.P. The world availability of non-wood lignocellulosic biomass for the production of cellulosic ethanol and potential pretreatments for the enhancement of enzymatic saccharification. Renew. Sustain. Energy Rev. 2016, 60, 155–172. [Google Scholar] [CrossRef]
- Gissén, C.; Prade, T.; Kreuger, E.; Nges, I.A.; Rosenqvist, H.; Svensson, S.-E.; Lantz, M.; Mattsson, J.E.; Börjesson, P.; Björnsson, L. Comparing energy crops for biogas production—Yields, energy input and costs in cultivation using digestate and mineral fertilisation. Biomass Bioenergy 2014, 64, 199–210. [Google Scholar] [CrossRef]
- Xu, N.; Liu, S.; Xin, F.; Zhou, J.; Jia, H.; Xu, J.; Jiang, M.; Dong, W. Biomethane Production from Lignocellulose: Biomass Recalcitrance and Its Impacts on Anaerobic Digestion. Front. Bioeng. Biotechnol. 2019, 7, 191. [Google Scholar] [CrossRef]
- Atelge, M.R.; Atabani, A.E.; Banu, J.R.; Krisa, D.; Kaya, M.; Eskicioglu, C.; Kumar, G.; Lee, C.; Yildiz, Y.Ş.; Unalan, S.; et al. A critical review of pretreatment technologies to enhance anaerobic digestion and energy recovery. Fuel 2020, 270, 117494. [Google Scholar] [CrossRef]
- Alvira, P.; Tomás-Pejó, E.; Ballesteros, M.; Negro, M.J. Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: A review. Bioresour. Technol. 2010, 101, 4851–4861. [Google Scholar] [CrossRef]
- Carrere, H.; Antonopoulou, G.; Affes, R.; Passos, F.; Battimelli, A.; Lyberatos, G.; Ferrer, I. Review of feedstock pretreatment strategies for improved anaerobic digestion: From lab-scale research to full-scale application. Bioresour. Technol. 2016, 199, 386–397. [Google Scholar] [CrossRef]
- Mankar, A.R.; Pandey, A.; Modak, A.; Pant, K.K. Pretreatment of lignocellulosic biomass: A review on recent advances. Bioresour. Technol. 2021, 334, 125235. [Google Scholar] [CrossRef]
- Kasinath, A.; Fudala-Ksiazek, S.; Szopinska, M.; Bylinski, H.; Artichowicz, W.; Remiszewska-Skwarek, A.; Luczkiewicz, A. Biomass in biogas production: Pretreatment and codigestion. Renew. Sustain. Energy Rev. 2021, 150, 111509. [Google Scholar] [CrossRef]
- Gałązka, A.; Szadkowski, J. Enzymatic hydrolysis of fast-growing poplar wood after pretreatment by steam explosion. Cellul. Chem. Technol. 2021, 55, 637–647. [Google Scholar] [CrossRef]
- Thamizhakaran Stanley, J.; Thanarasu, A.; Senthil Kumar, P.; Periyasamy, K.; Raghunandhakumar, S.; Periyaraman, P.; Devaraj, K.; Dhanasekaran, A.; Subramanian, S. Potential pre-treatment of lignocellulosic biomass for the enhancement of biomethane production through anaerobic digestion—A review. Fuel 2022, 318, 123593. [Google Scholar] [CrossRef]
- Monlau, F.; Sambusiti, C.; Barakat, A.; Quéméneur, M.; Trably, E.; Steyer, J.-P.; Carrère, H. Do furanic and phenolic compounds of lignocellulosic and algae biomass hydrolyzate inhibit anaerobic mixed cultures? A comprehensive review. Biotechnol. Adv. 2014, 32, 934–951. [Google Scholar] [CrossRef]
- Naik, G.P.; Poonia, A.K.; Chaudhari, P.K. Pretreatment of lignocellulosic agricultural waste for delignification, rapid hydrolysis, and enhanced biogas production: A review. J. Indian Chem. Soc. 2021, 98, 100147. [Google Scholar] [CrossRef]
- Hjorth, M.; Gränitz, K.; Adamsen, A.P.S.; Møller, H.B. Extrusion as a pretreatment to increase biogas production. Bioresour. Technol. 2011, 102, 4989–4994. [Google Scholar] [CrossRef]
- Evon, P.; Vandenbossche, V.; Candy, L.; Pontalier, P.-Y.; Rouilly, A. Twin-Screw Extrusion: A Key Technology for the Biorefinery. In Biomass Extrusion and Reaction Technologies: Principles to Practices and Future Potential; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2018; Volume 1304, pp. 17–33. ISBN 978/0/8412/3371/3. [Google Scholar]
- Turick, C.; Peck, M.; Chynoweth, D.; Jerger, D.; White, E.; Zsuffa, L.; Kenney, W. Methane fermentation of woody biomass. Bioresour. Technol. 1991, 37, 141–147. [Google Scholar] [CrossRef]
- Zheng, J.; Rehmann, L. Extrusion pretreatment of lignocellulosic biomass: A review. Int. J. Mol. Sci. 2014, 15, 18967–18984. [Google Scholar] [CrossRef]
- Zhang, S.; Keshwani, D.R.; Xu, Y.; Hanna, M.A. Alkali combined extrusion pretreatment of corn stover to enhance enzyme saccharification. Ind. Crop. Prod. 2012, 37, 352–357. [Google Scholar] [CrossRef]
- Vandenbossche, V.; Brault, J.; Hernandez-Melendez, O.; Evon, P.; Barzana, E.; Vilarem, G.; Rigal, L. Suitability assessment of a continuous process combining thermo-mechano-chemical and bio-catalytic action in a single pilot-scale twin-screw extruder for six different biomass sources. Bioresour. Technol. 2016, 211, 146–153. [Google Scholar] [CrossRef]
- Gatt, E.; Rigal, L.; Vandenbossche, V. Biomass pretreatment with reactive extrusion using enzymes: A review. Ind. Crop. Prod. 2018, 122, 329–339. [Google Scholar] [CrossRef]
- Cha, Y.-L.; Yang, J.; Park, Y.; An, G.H.; Ahn, J.-W.; Moon, Y.-H.; Yoon, Y.-M.; Yu, G.-D.; Choi, I.-H. Continuous alkaline pretreatment of Miscanthus sacchariflorus using a bench-scale single screw reactor. Bioresour. Technol. 2015, 181, 338–344. [Google Scholar] [CrossRef]
- Menardo, S.; Cacciatore, V.; Balsari, P. Batch and continuous biogas production arising from feed varying in rice straw volumes following pre-treatment with extrusion. Bioresour. Technol. 2015, 180, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhang, Y.; Gu, Y.; Liu, Z.; Shen, Z.; Chu, H.; Zhou, X. Enhancing methane production from rice straw by extrusion pretreatment. Appl. Energy 2014, 122, 34–41. [Google Scholar] [CrossRef]
- Khor, W.C.; Rabaey, K.; Vervaeren, H. Low temperature calcium hydroxide treatment enhances anaerobic methane production from (extruded) biomass. Bioresour. Technol. 2015, 176, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Frydendal-Nielsen, S.; Hjorth, M.; Baby, S.; Felby, C.; Jørgensen, U.; Gislum, R. The effect of harvest time, dry matter content and mechanical pretreatments on anaerobic digestion and enzymatic hydrolysis of miscanthus. Bioresour. Technol. 2016, 218, 1008–1015. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Rodríguez, N.; García-Bernet, D.; Domínguez, J.M. Faster methane production after sequential extrusion and enzymatic hydrolysis of vine trimming shoots. Environ. Chem. Lett. 2018, 16, 295–299. [Google Scholar] [CrossRef]
- Niu, W.; Huang, G.; Liu, X.; Chen, L.; Han, L. Chemical Composition and Calorific Value Prediction of Wheat Straw at Different Maturity Stages Using Near-Infrared Reflectance Spectroscopy. Energy Fuels 2014, 28, 7474–7482. [Google Scholar] [CrossRef]
- Buswell, A.M.; Mueller, H.F. Mechanism of Methane Fermentation. Ind. Eng. Chem. 1952, 44, 550–552. [Google Scholar] [CrossRef]
- Achinas, S.; Euverink, G.J.W. Theoretical analysis of biogas potential prediction from agricultural waste. Resour. Effic. Technol. 2016, 2, 143–147. [Google Scholar] [CrossRef]
- Khongchamnan, P.; Wanmolee, W.; Laosiripojana, N.; Champreda, V.; Suriyachai, N.; Kreetachat, T.; Sakulthaew, C.; Chokejaroenrat, C.; Imman, S. Solvothermal-Based Lignin Fractionation from Corn Stover: Process Optimization and Product Characteristics. Front. Chem. 2021, 9, 697237. [Google Scholar] [CrossRef]
- Sluiter, A. Determination of Total Solids in Biomass and Total Dissolved Solids in Liquid Process Samples: Laboratory Analytical Procedure (LAP). Tech. Rep. 2008, 9. Available online: https://www.nrel.gov/docs/gen/fy08/42621.pdf (accessed on 2 May 2022).
- Beaufils, N.; Boucher, J.; Peydecastaing, J.; Rigal, L.; Vilarem, G.; Villette, M.-J.; Candy, L.; Pontalier, P.-Y. The effect of time and temperature on the extraction of xylose and total phenolic compounds with pressurized hot water from hardwood species used for pulp and paper production in the South of France. Bioresour. Technol. Rep. 2021, 16, 100832. [Google Scholar] [CrossRef]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of structural carbohydrates and lignin in biomass, in: Laboratory Analytical Procedure (LAP). Natl. Renew. Energy Lab. 2008, 1617. Available online: https://www.nrel.gov/docs/gen/fy13/42618.pdf (accessed on 2 May 2022).
- Segal, L.; Creely, J.J.; Martin, A.E.; Conrad, C.M. An Empirical Method for Estimating the Degree of Crystallinity of Native Cellulose Using the X-ray Diffractometer. Text. Res. J. 1959, 29, 786–794. [Google Scholar] [CrossRef]
- Zwietering, M.H.; Jongenburger, I.; Rombouts, F.M.; van’t Riet, K. Modeling of the Bacterial Growth Curve. Appl. Environ. Microbiol. 1990, 56, 1875–1881. [Google Scholar] [CrossRef]
- Evon, P.; Vandenbossche, V.; Pontalier, P.-Y.; Rigal, L. New thermal insulation fiberboards from cake generated during biorefinery of sunflower whole plant in a twin-screw extruder. Ind. Crop. Prod. 2014, 52, 354–362. [Google Scholar] [CrossRef]
- Heredia-Olea, E.; Pérez-Carrillo, E.; Montoya-Chiw, M.; Serna-Saldívar, S.O. Effects of Extrusion Pretreatment Parameters on Sweet Sorghum Bagasse Enzymatic Hydrolysis and Its Subsequent Conversion into Bioethanol. BioMed Res. Int. 2015, 2015, 325905. [Google Scholar] [CrossRef]
- Mazaheri Tehrani, M.; Ehtiati, A.; Sharifi Azghandi, S. Application of genetic algorithm to optimize extrusion condition for soy-based meat analogue texturization. J. Food Sci. Technol. 2017, 54, 1119–1125. [Google Scholar] [CrossRef] [PubMed]
- Brishti, F.H.; Chay, S.Y.; Muhammad, K.; Ismail-Fitry, M.R.; Zarei, M.; Saari, N. Texturized mung bean protein as a sustainable food source: Effects of extrusion on its physical, textural and protein quality. Innov. Food Sci. Emerg. Technol. 2021, 67, 102591. [Google Scholar] [CrossRef]
- Choudhury, G.S.; Gautam, A. Comparative study of mixing elements during twin-screw extrusion of rice flour. Food Res. Int. 1998, 31, 7–17. [Google Scholar] [CrossRef]
- Lallement, A.; Peyrelasse, C.; Lagnet, C.; Barakat, A.; Schraauwers, B.; Maunas, S.; Monlau, F. A Detailed Database of the Chemical Properties and Methane Potential of Biomasses Covering a Large Range of Common Agricultural Biogas Plant Feedstocks. Waste 2023, 1, 195–227. [Google Scholar] [CrossRef]
- Pronyk, C.; Mazza, G.; Tamaki, Y. Production of Carbohydrates, Lignins, and Minor Components from Triticale Straw by Hydrothermal Treatment. J. Agric. Food Chem. 2011, 59, 3788–3796. [Google Scholar] [CrossRef]
- Tamaki, Y.; Mazza, G. Measurement of structural carbohydrates, lignins, and micro-components of straw and shives: Effects of extractives, particle size and crop species. Ind. Crop. Prod. 2010, 31, 534–541. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Cook, H.; Thomas, B. The lignin and cellulose contents of certain grassland species at different stages of growth. J. Agric. Sci. 1950, 40, 93–99. [Google Scholar] [CrossRef]
- Plant Variety Catalogues, Databases & Information Systems. Available online: https://food.ec.europa.eu/plants/plant-reproductive-material/plant-variety-catalogues-databases-information-systems_en (accessed on 5 March 2023).
- Pordesimo, L.O.; Hames, B.R.; Sokhansanj, S.; Edens, W.C. Variation in corn stover composition and energy content with crop maturity. Biomass Bioenergy 2005, 28, 366–374. [Google Scholar] [CrossRef]
- Collins, S.R.; Wellner, N.; Martinez Bordonado, I.; Harper, A.L.; Miller, C.N.; Bancroft, I.; Waldron, K.W. Variation in the chemical composition of wheat straw: The role of tissue ratio and composition. Biotechnol. Biofuels 2014, 7, 121. [Google Scholar] [CrossRef]
- Zillinsky, F.J. The Development of Triticale. In Advances in Agronomy; Brady, N.C., Ed.; Academic Press: Cambridge, MA, USA, 1974; Volume 26, pp. 315–348. [Google Scholar]
- Vandenbossche, V.; Doumeng, C.; Rigal, L. Thermomechanical and Thermo-mechano-chemical Pretreatment of Wheat Straw using a Twin-screw Extruder. BioResources 2014, 9, 1519–1538. [Google Scholar] [CrossRef]
- Zheng, J.; Choo, K.; Rehmann, L. The effects of screw elements on enzymatic digestibility of corncobs after pretreatment in a twin-screw extruder. Biomass Bioenergy 2015, 74, 224–232. [Google Scholar] [CrossRef]
- Wang, Z.; He, X.; Yan, L.; Wang, J.; Hu, X.; Sun, Q.; Zhang, H. Enhancing enzymatic hydrolysis of corn stover by twin-screw extrusion pretreatment. Ind. Crop. Prod. 2020, 143, 111960. [Google Scholar] [CrossRef]
- Elalami, D.; Aouine, M.; Monlau, F.; Guillon, F.; Dumon, C.; Hernandez Raquet, G.; Barakat, A. Enhanced enzymatic hydrolysis of corn stover using twin-screw extrusion under mild conditions. Biofuels Bioprod. Biorefining 2022, 16, 1642–1654. [Google Scholar] [CrossRef]
- Duque, A.; Manzanares, P.; Ballesteros, I.; Negro, M.J.; Oliva, J.M.; González, A.; Ballesteros, M. Sugar production from barley straw biomass pretreated by combined alkali and enzymatic extrusion. Bioresour. Technol. 2014, 158, 262–268. [Google Scholar] [CrossRef]
- Garuti, M.; Sinisgalli, E.; Soldano, M.; Fermoso, F.G.; Rodriguez, A.J.; Carnevale, M.; Gallucci, F. Mechanical pretreatments of different agri-based feedstock in full-scale biogas plants under real operational conditions. Biomass Bioenergy 2022, 158, 106352. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhao, J.; Xu, F.; Li, Y. Pretreatment of lignocellulosic biomass for enhanced biogas production. Prog. Energy Combust. Sci. 2014, 42, 35–53. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, X.; Gu, Y.; Zhou, X. A physicochemical method for increasing methane production from rice straw: Extrusion combined with alkali pretreatment. Appl. Energy 2015, 160, 39–48. [Google Scholar] [CrossRef]
- Monlau, F.; Sambusiti, C.; Barakat, A.; Guo, X.M.; Latrille, E.; Trably, E.; Steyer, J.-P.; Carrere, H. Predictive Models of Biohydrogen and Biomethane Production Based on the Compositional and Structural Features of Lignocellulosic Materials. Environ. Sci. Technol. 2012, 46, 12217–12225. [Google Scholar] [CrossRef]
- Thomsen, S.T.; Spliid, H.; Østergård, H. Statistical prediction of biomethane potentials based on the composition of lignocellulosic biomass. Bioresour. Technol. 2014, 154, 80–86. [Google Scholar] [CrossRef]
- Wahid, R.; Hjorth, M.; Kristensen, S.; Møller, H.B. Extrusion as Pretreatment for Boosting Methane Production: Effect of Screw Configurations. Energy Fuels 2015, 29, 4030–4037. [Google Scholar] [CrossRef]
- Victorin, M.; Davidsson, Å.; Wallberg, O. Characterization of Mechanically Pretreated Wheat Straw for Biogas Production. BioEnergy Res. 2020, 13, 833–844. [Google Scholar] [CrossRef]
- Khan, M.U.; Ahring, B.K. Lignin degradation under anaerobic digestion: Influence of lignin modifications—A review. Biomass Bioenergy 2019, 128, 105325. [Google Scholar] [CrossRef]
- Velázquez-Martí, B.; Meneses Quelal, O.; Gaibor, J.; Niño, Z. Review of Mathematical Models for the Anaerobic Digestion Process. In Anaerobic Digestion; IntechOpen: London, UK, 2018; ISBN 978/1/83881/849/4. [Google Scholar]
- Mancini, G.; Papirio, S.; Lens, P.N.L.; Esposito, G. Increased biogas production from wheat straw by chemical pretreatments. Renew. Energy 2018, 119, 608–614. [Google Scholar] [CrossRef]
- Lee, J.; Park, K.Y. Impact of hydrothermal pretreatment on anaerobic digestion efficiency for lignocellulosic biomass: Influence of pretreatment temperature on the formation of biomass-degrading byproducts. Chemosphere 2020, 256, 127116. [Google Scholar] [CrossRef]
- Sieborg, M.U.; Jønson, B.D.; Larsen, S.U.; Vazifehkhoran, A.H.; Triolo, J.M. Co-Ensiling of Wheat Straw as an Alternative Pre-Treatment to Chemical, Hydrothermal and Mechanical Methods for Methane Production. Energies 2020, 13, 4047. [Google Scholar] [CrossRef]
- Tsapekos, P.; Kougias, P.G.; Angelidaki, I. Mechanical pretreatment for increased biogas production from lignocellulosic biomass; predicting the methane yield from structural plant components. Waste Manag. 2018, 78, 903–910. [Google Scholar] [CrossRef]
- Pérez-Rodríguez, N.; García-Bernet, D.; Domínguez, J.M. Extrusion and enzymatic hydrolysis as pretreatments on corn cob for biogas production. Renew. Energy 2017, 107, 597–603. [Google Scholar] [CrossRef]
- Meegoda, J.N.; Li, B.; Patel, K.; Wang, L.B. A Review of the Processes, Parameters, and Optimization of Anaerobic Digestion. Int. J. Environ. Res. Public. Health 2018, 15, 2224. [Google Scholar] [CrossRef]
- Janke, L.; Weinrich, S.; Leite, A.F.; Terzariol, F.K.; Nikolausz, M.; Nelles, M.; Stinner, W. Improving anaerobic digestion of sugarcane straw for methane production: Combined benefits of mechanical and sodium hydroxide pretreatment for process designing. Energy Convers. Manag. 2017, 141, 378–389. [Google Scholar] [CrossRef]

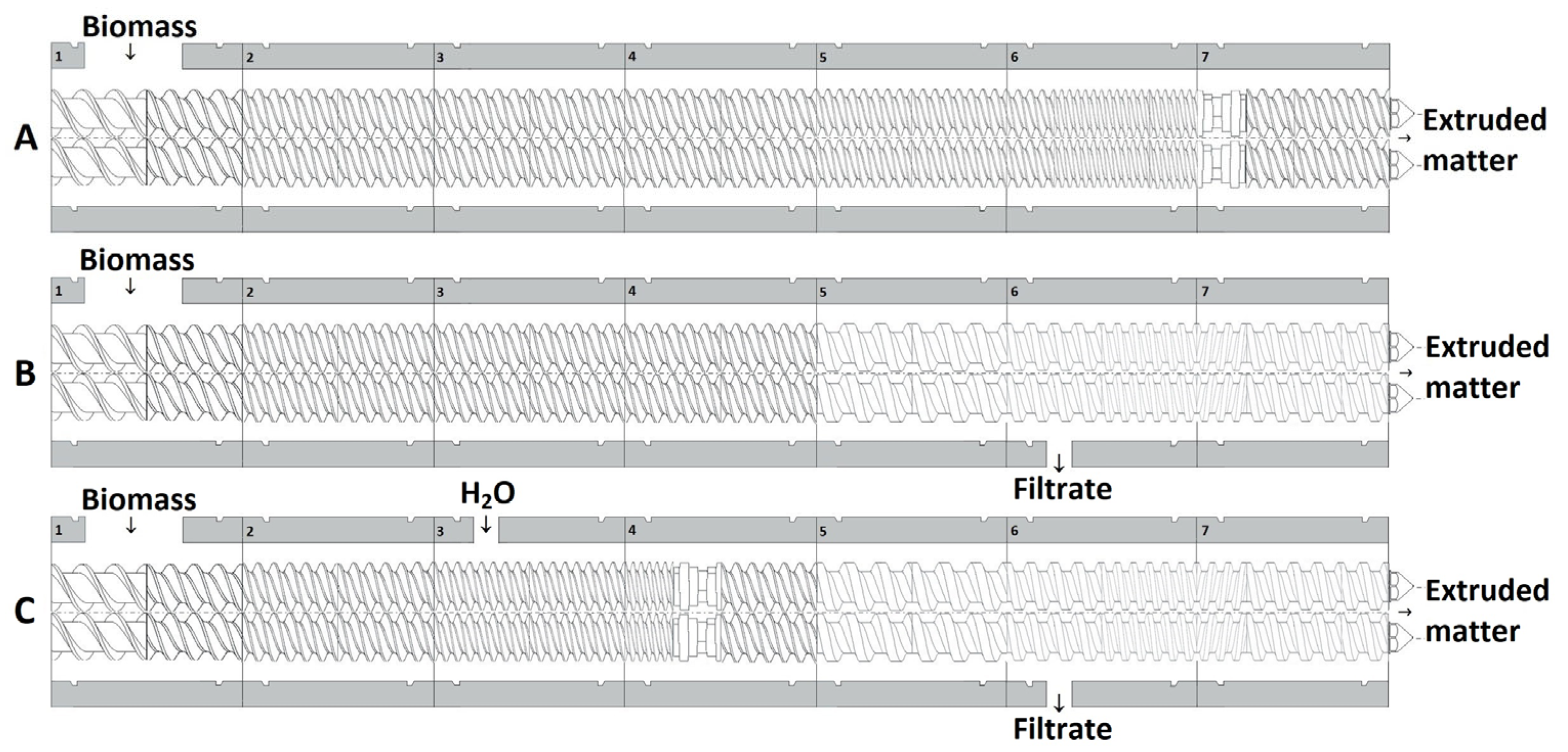
| Raw Material | SB | WT | CS | WS | |||||
|---|---|---|---|---|---|---|---|---|---|
| Screw Profile | SE | HE | SE | HE | SE | HER | SE | HER | |
| Measured parameters | Material temperature range in module 7 (°C) | 18–22 | 31–36 | 26–27 | 30–34 | 32–34 | 28–42 | 27–32 | 58–63 |
| Motor current range (A) | 14–15 | 16–25 | 27–35 | 41–47 | 11–19 | 27–37 | 13–21 | 27–30 | |
| Substrate | Dry matter content (%) | 27.8 ± 0.2 | 28.2 ± 1.0 | 21.1 ± 0.2 | 21.1 ± 0.2 | 69.3 ± 1.0 | 89.6 ± 0.1 | 77.0 ± 1.1 | 88.5 ± 0.1 |
| Inlet flow rate (kg/h) | 19.2 ± 3.1 | 26.0 ± 3.3 | 26.5 ± 3.8 | 17.5 ± 2.8 | 5.3 ± 0.5 | 1.0 ± 0.1 | 6.6 ± 0.3 | 2.0 ± 0.1 | |
| Dry inlet flow rate (kg/h) | 5.3 ± 0.9 | 7.2 ± 0.9 | 5.6 ± 0.8 | 3.7 ± 0.6 | 3.6 ± 0.3 | 0.9 ± 0.1 | 5.1 ± 0.2 | 1.8 ± 0.1 | |
| Liquid | Water inlet flow rate (kg/h) | - | - | - | - | - | 5.0 | - | 8.5 |
| Water-to-dry matter ratio | - | - | - | - | - | 85/15 | - | 82/18 | |
| Extrudate | Dry matter content (%) | 30.6 ± 0.3 | 43.6 ± 1.2 | 23.6 ± 0.3 | 63.4 ± 0.3 | 75.3 ± 0.6 | 82.3 ± 0.1 | 81.6 ± 0.4 | 76.3 ± 0.3 |
| Outlet flow rate (kg/h) | 17.4 ± 2.8 | 13.4 ± 1.8 | 23.6 ± 3.2 | 4.9 ± 0.2 | 4.9 ± 0.4 | 1.0 ± 0.1 | 6.3 ± 0.3 | 2.2 ± 0.1 | |
| Dry outlet flow rate (kg/h) | 5.3 ± 0.9 | 5.8 ± 0.8 | 5.6 ± 0.8 | 3.1 ± 0.1 | 3.6 ± 0.3 | 0.8 ± 0.1 | 5.1 ± 0.2 | 1.7 ± 0.1 | |
| Filtrate | Dry matter content (%) | - | 12.7 ± 0.8 | - | 7.3 ± 0.0 | - | 1.9 ± 0.0 | - | 1.9 ± 0.0 |
| Outlet flow rate (kg/h) | - | 11.0 ± 2.5 | - | 8.1 ± 0.6 | - | 4.7 ± 0.2 | - | 7.6 ± 0.3 | |
| Dry outlet flow rate (kg/h) | - | 1.4 ± 0.3 | - | 0.6 ± 0.0 | - | 0.1 ± 0.0 | - | 0.1 ± 0.0 | |
| SB | WT | CS | WS | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CM | SE | HE | CM | SE | HE | CM | SE | HER | CM | SE | HER | |
| TS 2 (%FM 1) | 28.2 ± 0.3 a | 28.8 ± 0.3 a | 43.6 ± 1.1 b | 21.1 ± 0.2 a | 23.6 ± 0.3 b | 63.4 ± 0.3 c | 89.6 ± 0.1 a | 75.3 ± 0.6 b | 82.3 ± 0.1 c | 88.5 ± 0.1 a | 81.6 ± 0.4 b | 76.3 ± 0.3 c |
| VS 3 (%FM 1) | 27.2 ± 0.3 a | 27.8 ± 0.3 a | 42.5 ± 1.1 b | 18.9 ± 0.2 a | 21.5 ± 0.2 b | 59.6 ± 0.3 c | 86.5 ± 0.1 a | 72.7 ± 0.6 b | 80.8 ± 0.1 c | 85.0 ± 0.1 a | 78.4 ± 0.4 b | 75.1 ± 0.3 c |
| Ash (%FM 1) | 1.0 ± 0.0 a | 1.0 ± 0.0 a | 1.2 ± 0.0 b | 2.2 ± 0.0 a | 2.1 ± 0.0 b | 3.8 ± 0.0 c | 3.1 ± 0.0 a | 2.6 ± 0.0 b | 1.5 ± 0.2 c | 3.5 ± 0.1 a | 3.2 ± 0.1 b | 1.2 ± 0.0 c |
| Extractables (%TS 2) | 28.4 ± 0.7 a | 25.8 ± 0.3 b | 22.3 ± 0.2 c | 25.2 ± 0.0 a | 20.5 ± 0.1 b | 10.0 ± 0.1 c | 18.6 ± 0.1 a | 18.1 ± 0.3 a | 5.3 ± 0.0 b | 10.7 ± 0.0 a | 9.9 ± 0.1 b | 5.2 ± 0.3 c |
| Cellulose (%TS 2) | 29.9 ± 0.7 a | 29.6 ± 0.5 a | 32.0 ± 0.4 b | 27.1 ± 0.1 a | 31.6 ± 0.2 b | 34.9 ± 1.8 c | 31.7 ± 1.6 a | 33.3 ± 0.0 a | 40.7 ± 1.0 b | 34.3 ± 0.2 a | 33.6 ± 0.1 b | 36.2 ± 0.1 c |
| Hemicelluloses (%TS 2) | 17.0 ± 0.7 a | 19.7 ± 0.6 b | 18.0 ± 0.4 c | 24.6 ± 0.1 a | 21.3 ± 0.1 b | 23.4 ± 1.1 c | 18.8 ± 0.5 a | 18.6 ± 0.0 a | 22.3 ± 0.6 b | 19.3 ± 0.1 a | 19.2 ± 0.0 a | 20.9 ± 0.1 b |
| Klason lignin (%TS 2) | 15.5 ± 0.2 a | 16.2 ± 0.1 b | 16.7 ± 0.2 c | 17.8 ± 0.5 a | 19.1 ± 0.2 b | 24.2 ± 0.0 c | 17.3 ± 0.3 a | 17.5 ± 0.1 a | 20.5 ± 0.3 b | 22.6 ± 0.1 a | 22.9 ± 0.1 b | 24.6 ± 0.2 c |
| Residual chemicals (%TS 2) | 9.2 ± 0.2 a | 8.8 ± 0.1 b | 10.9 ± 0.2 c | 5.4 ± 0.4 a | 7.5 ± 0.2 b | 7.4 ± 0.0 b | 13.7 ± 0.3 a | 12.5 ± 0.1 b | 11.3 ± 0.3 c | 13.1 ± 0.1 a | 14.3 ± 0.1 b | 13.3 ± 0.2 a |
| SB | WT | CS | WS | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CM | SE | HE | CM | SE | HE | CM | SE | HER | CM | SE | HER | |
| Tapped density (kg/m3) | 21.0 ± 0.9 a | 57.5 ± 0.8 b | 110.6 ± 1.1 c | 11.6 ± 1.4 a | 55.3 ± 1.4 b | 62.9 ± 1.3 c | 28.7 ± 0.8 a | 69.2 ± 1.1 b | 200.3 ± 1.7 c | 14.7 ± 0.2 a | 38.7 ± 0.0 b | 124.0 ± 0.5 c |
| Cellulose crystallinity (%) | 29.2 ± 1.2 a | 27.3 ± 0. 5 a | 28.0 ± 0.1 a | 52.4 ± 0.4 a | 52.3 ± 1.6 a | 53.9 ± 0.4 a | 67.5 ± 2.1 a | 64.2 ± 0.8 ab | 58.9 ± 1.4 b | 55.5 ± 0.6 a | 53.7 ± 1.1 a | 56.4 ± 1.3 a |
| Biomass | SB | WT | CS | WS | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pretreament | CM | SE | HE | CM | SE | HE | CM | SE | HER | CM | SE | HER |
| Experimental BMP (NmL/gVS) | 345 ± 16 a | 345 ± 7 a | 338 ± 2 a | 233 ± 4 a | 247 ± 2 a | 242 ± 6 a | 272 ± 5 ab | 286 ± 3 a | 264 ± 9 b | 249 ± 17 ab | 270 ± 2 a | 247 ± 0 b |
| BMPth adjusted (NmL/gVS) | 395 | 388 | 375 | 356 | 346 | 362 | 366 | 369 | 350 | 352 | 343 | 346 |
| BI (%) | 87 | 89 | 90 | 66 | 66 | 67 | 74 | 78 | 75 | 71 | 79 | 71 |
| B (NmL/gVS) | 339 | 336 | 330 | 229 | 244 | 241 | 268 | 284 | 259 | 244 | 265 | 242 |
| Rm (NmL/gVS.day) | 31 | 39 | 56 | 18 | 28 | 30 | 15 | 17 | 31 | 14 | 17 | 24 |
| λ (day) | 0.19 | 0.20 | 0.01 | 1.28 | 1.33 | 1.03 | 0.83 | 1.08 | 1.43 | 0.46 | 0.29 | 0.80 |
| R2 | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chevalier, A.; Evon, P.; Monlau, F.; Vandenbossche, V.; Sambusiti, C. Twin-Screw Extrusion Mechanical Pretreatment for Enhancing Biomethane Production from Agro-Industrial, Agricultural and Catch Crop Biomasses. Waste 2023, 1, 497-514. https://doi.org/10.3390/waste1020030
Chevalier A, Evon P, Monlau F, Vandenbossche V, Sambusiti C. Twin-Screw Extrusion Mechanical Pretreatment for Enhancing Biomethane Production from Agro-Industrial, Agricultural and Catch Crop Biomasses. Waste. 2023; 1(2):497-514. https://doi.org/10.3390/waste1020030
Chicago/Turabian StyleChevalier, Arthur, Philippe Evon, Florian Monlau, Virginie Vandenbossche, and Cecilia Sambusiti. 2023. "Twin-Screw Extrusion Mechanical Pretreatment for Enhancing Biomethane Production from Agro-Industrial, Agricultural and Catch Crop Biomasses" Waste 1, no. 2: 497-514. https://doi.org/10.3390/waste1020030
APA StyleChevalier, A., Evon, P., Monlau, F., Vandenbossche, V., & Sambusiti, C. (2023). Twin-Screw Extrusion Mechanical Pretreatment for Enhancing Biomethane Production from Agro-Industrial, Agricultural and Catch Crop Biomasses. Waste, 1(2), 497-514. https://doi.org/10.3390/waste1020030










