A Concise Review of Catalytic Synthesis of Methanol from Synthesis Gas
Abstract
1. Research Necessity
2. Research Background and Current Situation
2.1. Gas Phase Processes
2.1.1. Synthesis Methods and Catalysts
High-Pressure Gas-Phase Methanol Synthesis
Low-Pressure Gas-Phase Methanol Synthesis
Medium-Pressure Gas-Phase Methanol Synthesis
Non-Copper Catalyst
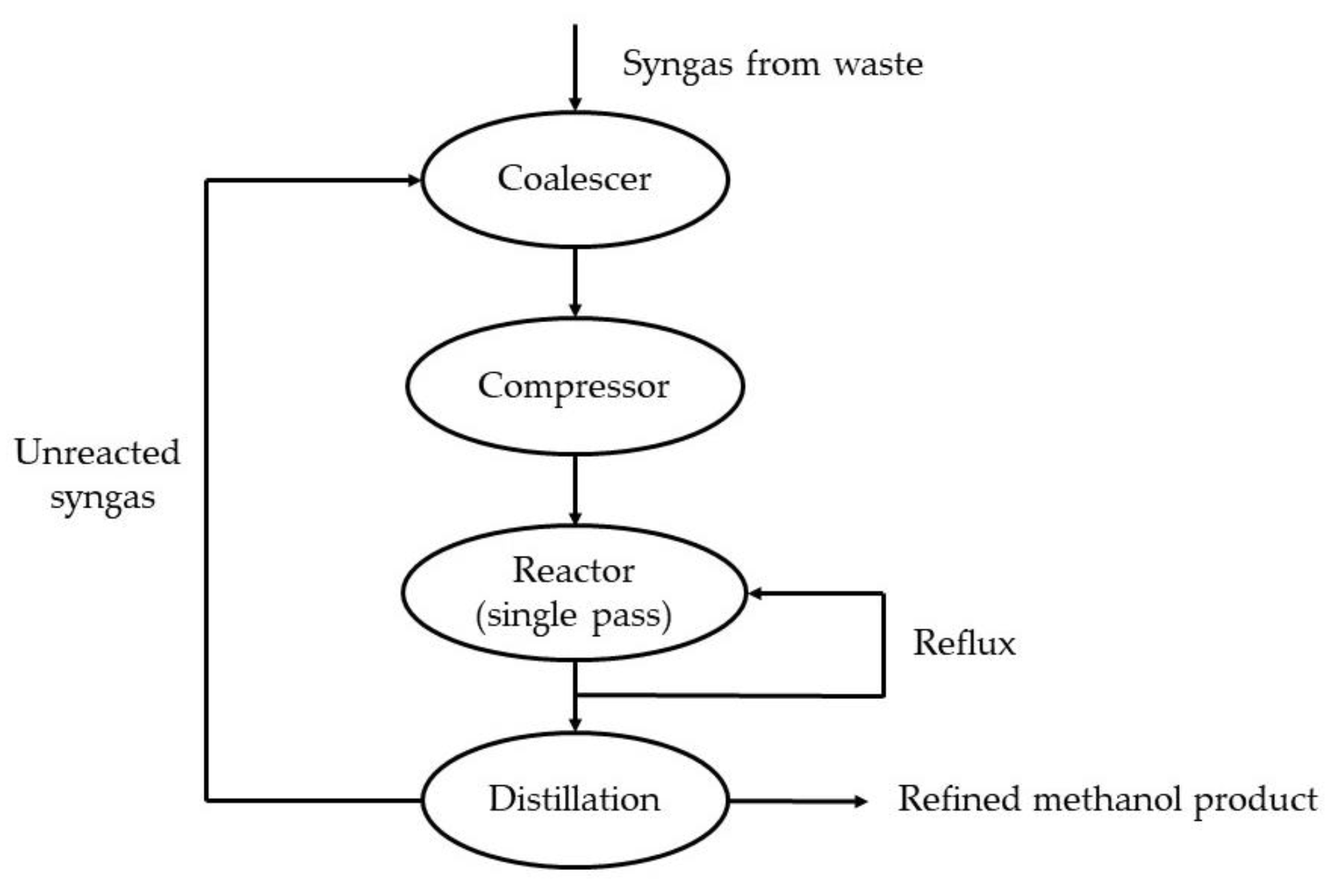
2.1.2. Types of Reactors
ICI Method
Lurgi Method
Other
2.1.3. Reaction Mechanism
2.1.4. The Role of CO2 in Methanol Synthesis
2.1.5. Research on the Active Center of Copper-Based Catalysts
Cu0 Is the Active Center
Cu+ Is the Active Center
Cu0-Cu+ Is the Active Center
2.1.6. Summary
2.2. Liquid Phase Processes
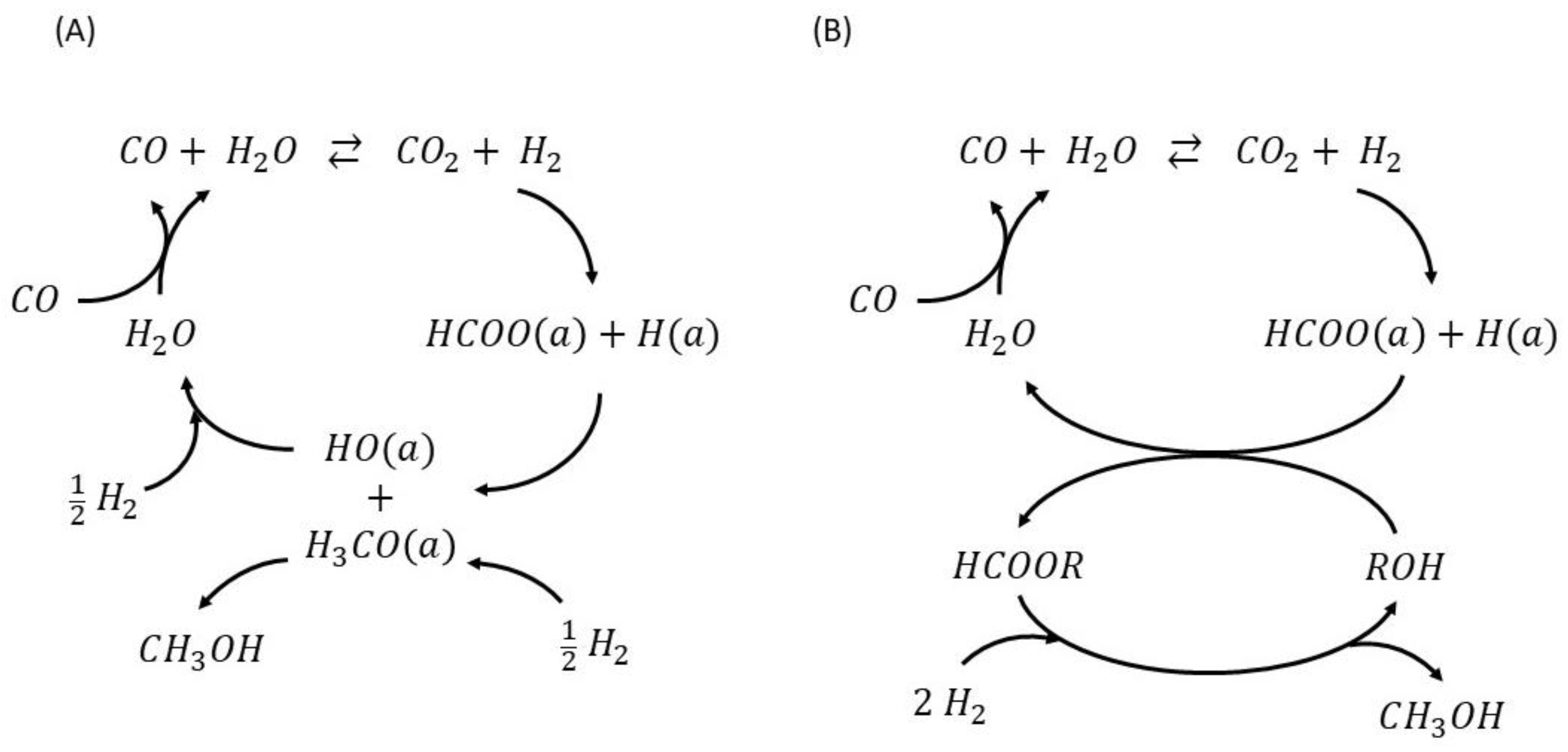
2.2.1. Reaction Mechanism
CO/H2 Reaction System
CO/CO2/H2 Reaction System
2.2.2. Main Catalyst
Carbonylation Catalyst
Hydrogenolysis Catalyst
Nickel-Based Catalyst
Copper-Based Catalyst
2.2.3. Solvent
Inert Solvent
Mixed Solvent
Alcohol Solvent
2.2.4. Additives and Supports
2.2.5. Preparation Method for Catalyst
Co-Precipitation Method
Sol-Gel Method
Other Synthesis Methods
2.2.6. Reactor Types
Slurry Bed Process
Trickle Bed Process
2.3. CO2 and H2 Methanol Synthesis
3. Summary and Prospect
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roode-Gutzmer, Q.I.; Kaiser, D.; Bertau, M. Renewable Methanol Synthesis. ChemBioEng Rev. 2019, 6, 209–236. [Google Scholar] [CrossRef]
- Brunetti, A.; Migliori, M.; Cozza, D.; Catizzone, E.; Giordano, G.; Barbieri, G. Methanol Conversion to Dimethyl Ether in Catalytic Zeolite Membrane Reactors. ACS Sustain. Chem. Eng. 2020, 8, 10471–10479. [Google Scholar] [CrossRef]
- Tian, P.; Wei, Y.; Ye, M.; Liu, Z. Methanol to Olefins (MTO): From Fundamentals to Commercialization. ACS Catal. 2015, 5, 1922–1938. [Google Scholar] [CrossRef]
- Keil, F.J. Methanol-to-hydrocarbons: Process technology. Microporous Mesoporous Mater. 1999, 29, 49–66. [Google Scholar] [CrossRef]
- Wang, T.; Tang, X.; Huang, X.; Qian, W.; Cui, Y.; Hui, X.; Yang, W.; Wei, F. Conversion of methanol to aromatics in fluidized bed reactor. Catal. Today 2014, 233, 8–13. [Google Scholar] [CrossRef]
- Kohli, K.; Sharma, B.K.; Panchal, C.B. Dimethyl Carbonate: Review of Synthesis Routes and Catalysts Used. Energies 2022, 15, 5133. [Google Scholar] [CrossRef]
- Din, I.U.; Shaharun, M.S.; Alotaibi, M.A.; Alharthi, A.I.; Naeem, A. Recent developments on heterogeneous catalytic CO2 reduction to methanol. J. CO2 Util. 2019, 34, 20–33. [Google Scholar] [CrossRef]
- Khadzhiev, S.N.; Kolesnichenko, N.V.; Ezhova, N.N. Slurry technology in methanol synthesis (Review). Pet. Chem. 2016, 56, 77–95. [Google Scholar] [CrossRef]
- Marie-Rose, S.; Lemieux, A.; Lavoie, J.-M. Conversion of Non-Homogeneous Biomass to Ultraclean Syngas and Catalytic Conversion to Ethanol. In Biofuel’s Engineering Process Technology; IntechOpen: London, UK, 2011. [Google Scholar] [CrossRef]
- Tijm, P.J.A.; Waller, F.J.; Brown, D.M. Methanol technology developments for the new millennium. Appl. Catal. Gen. 2001, 221, 275–282. [Google Scholar] [CrossRef]
- Bozzano, G.; Manenti, F. Efficient methanol synthesis: Perspectives, technologies and optimization strategies. Prog. Energy Combust. Sci. 2016, 56, 71–105. [Google Scholar] [CrossRef]
- Kim, W.-Y.; Hayashi, H.; Kishida, M.; Nagata, H.; Wakabayashi, K. Methanol synthesis from syngas over supported palladium catalysts prepared using water-in-oil microemulsion. Appl. Catal. A Gen. 1998, 169, 157–164. [Google Scholar] [CrossRef]
- Mériaudeau, P.; Albano, K.; Naccache, C. Promotion of Platinum-based Catalysts for Methanol Synthesis from Syngas. J. Chem. Soc. Faraday Trans. I 1987, 83, 2113–2118. [Google Scholar] [CrossRef]
- Yang, C.; Ma, Z.; Zhao, N.; Wei, W.; Hu, T.; Sun, Y. Methanol synthesis from CO2-rich syngas over a ZrO2 doped CuZnO catalyst. Catal. Today 2006, 115, 222–227. [Google Scholar] [CrossRef]
- Liu, L.; Fan, F.; Bai, M.; Xue, F.; Ma, X.; Jiang, Z.; Fang, T. Mechanistic study of methanol synthesis from CO2 hydrogenation on Rh-doped Cu(111) surfaces. Mol. Catal. 2019, 466, 26–36. [Google Scholar] [CrossRef]
- Song, J.-R.; Wen, L.-X.; Shao, L.; Chen, J.-F. Preparation and characterization of novel Pd/SiO2 and Ca–Pd/SiO2 egg-shell catalysts with porous hollow silica. Appl. Surf. Sci. 2006, 253, 2678–2684. [Google Scholar] [CrossRef]
- Dry, R.J. Possibilities for the Development of Large-Capacity Methanol Synthesis. Ind. Eng. Chem. Res. 1988, 27, 616–624. [Google Scholar] [CrossRef]
- Joo, O.S.; Jung, K.D.; Moon, I.; Rozovskii, A.Y.; Lin, G.I.; Han, S.H.; Uhm, S.J. Carbon Dioxide Hydrogenation To Form Methanol via a Reverse-Water-Gas-Shift Reaction (the CAMERE Process). Ind. Eng. Chem. Res. 1999, 38, 1808–1812. [Google Scholar] [CrossRef]
- Dybkjaer, I. Topsoe Methanol Technology. Chem. Econ. Eng. Rev. 1981, 6, 17–25. [Google Scholar]
- Klier, K. Methanol Synthesis. Adv. Catal. 1982, 31, 243–313. [Google Scholar]
- Denise, B.; Sneeden, R.P.A.; Hamon, C. Hamon Hydrocondensation of carbon dioxide: IV. J. Mol. Catal. 1982, 17, 359–366. [Google Scholar] [CrossRef]
- Chinchen, G.C.; Denny, P.J.; Parker, D.G.; Spencer, M.S.; Whan, D.A. Mechanism of methanol synthesis from CO2/CO/H2 mixtures over copper/zinc oxide/alumina catalysts: Use of14C-labelled reactants. Appl. Catal. 1987, 30, 333–338. [Google Scholar] [CrossRef]
- Zhang, Y.; Sun, Q.; Deng, J.; Wu, D.; Chen, S. A high activity Cu/ZnO/A1203 catalyst for methanol synthesis: Preparation and catalytic properties. Appl. Catal. A Gen. 1997, 158, 105–120. [Google Scholar] [CrossRef]
- Laudenschleger, D.; Ruland, H.; Muhler, M. Identifying the nature of the active sites in methanol synthesis over Cu/ZnO/Al2O3 catalysts. Nat. Commun. 2020, 11, 3898. [Google Scholar] [CrossRef]
- Sehested, J. Industrial and scientific directions of methanol catalyst development. J. Catal. 2019, 371, 368–375. [Google Scholar] [CrossRef]
- Waugh, K.C. Methanol Synthesis. Catal. Today 1992, 15, 51–75. [Google Scholar] [CrossRef]
- Chinchen, G.C.; Waugh, K.C.; Whan, D.A. Whan The activity and state of the copper surface in methanol synthesis catalysts. Appl. Catal. 1986, 25, 101–107. [Google Scholar] [CrossRef]
- Robbins, J.L. Methanol synthesis over Cu/SiO2 catalysts. Catal. Lett. 1991, 10, 1–10. [Google Scholar] [CrossRef]
- Fleisch, T.H.; Fleisch, R.L.M. Studies on the chemical state of Cu during methanol synthesis. J. Catal. 1984, 90, 165–172. [Google Scholar] [CrossRef]
- Herman, R.G.; Herman, K.K.; Simmons, G.; Finn, B. Catalytic synthesis of methanol from COH2: I. Phase composition, electronic properties, and activities of the Cu/ZnO/M2O3 catalysts. J. Catal. 1979, 56, 407–429. [Google Scholar] [CrossRef]
- Duprez, D.; Ferhat-Hamida, Z.; Bettahar, M.M. Surface mobility and reactivity of oxygen species on a copper-zinc catalyst in methanol synthesis. J. Catal. 1990, 124, 1–11. [Google Scholar] [CrossRef]
- Chu, P.J.; Gerstein, B.C.; Sheffer, G.R.; King, T.S. NMR studies of 65Cu and 133Cs in alkali-metal-promoted copper catalysts. J. Catal. 1989, 115, 194–204. [Google Scholar] [CrossRef]
- Denise, B.; Sneeden, R.P.A.; Beguin, B.; Cherifi, O. Supported copper catalysts in the synthesis of methanol: N2O-titrations. Appl. Catal. 1987, 30, 353–363. [Google Scholar] [CrossRef]
- Cybulski, A.; Edvinsson, R.; Irandoust, S.; Andersson, B. Liquid-phase methanol synthesis: Modelling of a monolithic reactor. Chem. Eng. Sci. 1993, 48, 3463–3478. [Google Scholar] [CrossRef]
- Hu, B.; Fujimoto, K. Low Temperature Methanol Synthesis in Slurry Phase With a Hybrid Copper-Formate System. Catal. Lett. 2009, 129, 416–421. [Google Scholar] [CrossRef]
- Zeng, J.-Q.; Tsubaki, N.; Fujimoto, K. The promoting effect of alcohols in a new process of low-temperature synthesis of methanol from CO/CO2/H2. Fuel 2002, 81, 125–127. [Google Scholar] [CrossRef]
- Yang, R.; Fu, Y.; Zhang, Y.; Tsubaki, N. In situ DRIFT study of low-temperature methanol synthesis mechanism on Cu/ZnO catalysts from CO2-containing syngas using ethanol promoter. J. Catal. 2004, 228, 23–35. [Google Scholar] [CrossRef]
- Yang, R.; Zhang, Y.; Iwama, Y.; Tsubaki, N. Mechanistic study of a new low-temperature methanol synthesis on Cu/MgO catalysts. Appl. Catal. A Gen. 2005, 288, 126–133. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, R.; Tsubaki, N. A new low-temperature methanol synthesis method: Mechanistic and kinetics study of catalytic process. Catal. Today 2008, 132, 93–100. [Google Scholar] [CrossRef]
- Yang, R.; Zhang, Y.; Tsubaki, N. Rideal-type reaction of formate species with alcohol: A key step in new low-temperature methanol synthesis method. Catal. Commun. 2007, 8, 1829–1833. [Google Scholar] [CrossRef]
- Hu, B.; Fujimoto, K. High-performance Cu/MgO–Na catalyst for methanol synthesis via ethyl formate. Appl. Catal. A Gen. 2008, 346, 174–178. [Google Scholar] [CrossRef]
- EP 0104875; Green, M.J. Process for the Production of Formates. European Patent Office: Munich, Germany, 1984.
- EP 0115387; Green, M.J. Decarbonylation Process. European Patent Office: Munich, Germany, 1984.
- EP 0107441; Green, M.J. Process for the Preparation of Formamides. European Patent Office: Munich, Germany, 1984.
- EP 0168167; Green, M.J. Catalysis Using Supported Strong Base Catalysts. European Patent Office: Munich, Germany, 1986.
- Kudo, K. Kinetic Study of Methyl Formate Synthesis from Methanol and Carbon Monoxide Catalyzed by 1, 8-Diazabicyclo [5. 4. 0] undec-7-ene. Nippon Kagaku Kaishi 1976, 4, 457–465. [Google Scholar]
- He, L.; Liu, H.; Xiao, C.; Kou, Y. Liquid-phase synthesis of methyl formate via heterogeneous carbonylation of methanol over a soluble copper nanocluster catalyst. Green Chem. 2008, 10, 619–622. [Google Scholar] [CrossRef]
- US 4619946; Sapienza, R.S.; Slegeir, W.A.; O’Hare, T.E.; Mahajan, D. Low Temperature Catalysts for Methanol Production. U.S. Patent and Trademark Office: Alexandria, Egypt, 1986.
- Mahajan, D. Atom-economical reduction of carbon monoxide to methanol catalyzed by soluble transition metal complexes at low temperatures. Top. Catal. 2005, 32, 209–214. [Google Scholar] [CrossRef]
- US 4992480; Mahajan, D.; Sapienza, R.S.; Slegeir, W.A.; O’Hare, T.E. Homogeneous Catalyst Formulations for Methanol Production. U.S. Patent and Trademark Office: Alexandria, Egypt, 1990.
- Ohyama, S. Low-temperature methanol synthesis in catalytic systems composed of nickel compounds and alkali alkoxides in liquid phases. Appl. Catal. A Gen. 1999, 180, 217–225. [Google Scholar] [CrossRef]
- Ohyama, S. In situ FTIR study on reaction pathways in Ni(CO)4/CH3OK catalytic system for low-temperature methanol synthesis in a liquid medium. Appl. Catal. A Gen. 2001, 220, 235–242. [Google Scholar] [CrossRef]
- Ohyama, S. Transformation of the nickel precursor in catalytic systems for low-temperature methanol synthesis in liquid phase. Appl. Catal. A Gen. 1999, 181, 87–93. [Google Scholar] [CrossRef]
- Lee, E.S.; Aika, K.I. Low-temperature methanol synthesis in liquid-phase with a Raney Nickel–alkoxide system: Effect of Raney Nickel pretreatment and reaction conditions. J. Mol. Catal. A Chem. 1999, 141, 241–248. [Google Scholar] [CrossRef]
- EP 0504981; Marchionna, M.; Lami, M. Catalyst System and Process for the Liquid-Phase Production of Methanol from Synthesis Gas. European Patent Office: Munich, Germany, 1992.
- US 6028119; Kokubu, Y. Process for Methanol Production. U.S. Patent and Trademark Office: Alexandria, Egypt, 2000.
- Vishwesh, M.; Palekar, H.J.; John, W. Tiemey and Irving Wender Slurry phase synthesis of methanol with a potassium methoxide/copper chromite catalytic system. Appl. Catal. A Gen. 1993, 102, 13–34. [Google Scholar]
- Zhang, X.; Zhong, L.; Guo, Q.; Fan, H.; Zheng, H.; Xie, K. Influence of the calcination on the activity and stability of the Cu/ZnO/Al2O3 catalyst in liquid phase methanol synthesis. Fuel 2010, 89, 1348–1352. [Google Scholar] [CrossRef]
- Huang, W.; Li, W.; Sun, J.; Yin, L. Effect of Carbon Promoters on the Performance of Cu-Zn-Al Catalyst Prepared by Complete Liquid-Phase Technology. Chin. J. Catal. (Chin. Version) 2011, 31, 1393–1398. [Google Scholar] [CrossRef]
- Li, Y. Study on Cu/MgO/ZnO Catalyst for Low-temperature Synthesis of Methanol. Chin. J. Process Eng. 2010, 10, 781–787. [Google Scholar]
- Chu, W.; Zhang, T.; He, C.; Wu, Y. Low-temperature methanol synthesis (LTMS) in liquid phase on novel copper-based catalysts. Catal. Lett. 2002, 79, 129–132. [Google Scholar] [CrossRef]
- Huang, L.; Chu, W.; Long, Y.; Ci, Z.; Luo, S. Influence of Zirconia Promoter on Catalytic Properties of Cu–Cr–Si Catalysts for Methanol Synthesis at High CO Conversion in Slurry Phase. Catal. Lett. 2006, 108, 113–118. [Google Scholar] [CrossRef]
- Palekar, V.M.; Tierney, J.W.; Wender, I. Alkali compounds and copper chromite as low-temperature slurry phase methanol catalysts. Appl. Catal. A Gen. 1993, 103, 105–122. [Google Scholar] [CrossRef]
- Zhao, T.-S.; Yoneyama, Y.; Fujimoto, K.; Yamane, N.; Fujimoto, K.; Tsubaki, N. Promotional Effect of Potassium Salt in Low-temperature Formate and Methanol Synthesis from CO/CO2/H2 on Copper Catalyst. Chem. Lett. 2007, 36, 734–735. [Google Scholar] [CrossRef]
- Zhao, T.-S.; Zhang, K.; Chen, X.; Ma, Q.; Tsubaki, N. A novel low-temperature methanol synthesis method from CO/H2/CO2 based on the synergistic effect between solid catalyst and homogeneous catalyst. Catal. Today 2010, 149, 98–104. [Google Scholar] [CrossRef]
- Hu, B.; Fujimoto, K. Promoting behaviors of alkali compounds in low temperature methanol synthesis over copper-based catalyst. Appl. Catal. B Environ. 2010, 95, 208–216. [Google Scholar] [CrossRef]
- Tsubaki, N.; Ito, M.; Fujimoto, K. A New Method of Low-Temperature Methanol Synthesis. J. Catal. 2001, 197, 224–227. [Google Scholar] [CrossRef]
- Zeng, J.; Fujimoto, K.; Tsubaki, N. A New Low-Temperature Synthesis Route of Methanol: Catalytic Effect of the Alcoholic Solvent. Energy Fuels 2002, 16, 83–86. [Google Scholar] [CrossRef]
- Reubroycharoen, P.; Yamagami, T.; Vitidsant, T.; Yoneyama, Y.; Ito, M.; Tsubaki, N. Continuous Low-Temperature Methanol Synthesis from Syngas Using Alcohol Promoters. Energy Fuels 2003, 17, 817–821. [Google Scholar] [CrossRef]
- Reubroycharoen, P.; Vitidsant, T.; Yoneyama, Y.; Tsubaki, N. Development of a new low-temperature methanol synthesis process. Catal. Today 2004, 89, 447–454. [Google Scholar] [CrossRef]
- Bao, J.; Liu, Z.; Zhang, Y.; Tsubaki, N. Preparation of mesoporous Cu/ZnO catalyst and its application in low-temperature methanol synthesis. Catal. Commun. 2008, 9, 913–918. [Google Scholar] [CrossRef]
- Yang, R.; Yu, X.; Zhang, Y.; Li, W.; Tsubaki, N. A new method of low-temperature methanol synthesis on Cu/ZnO/Al2O3 catalysts from CO/CO2/H2. Fuel 2008, 87, 443–450. [Google Scholar] [CrossRef]
- Xu, B.; Yang, R.; Meng, F.; Reubroycharoen, P.; Vitidsant, T.; Zhang, Y.; Yoneyama, Y.; Tsubaki, N. A New Method of Low Temperature Methanol Synthesis. Catal. Surv. Asia 2009, 13, 147–163. [Google Scholar] [CrossRef]
- Qi, G.X.; Zheng, X.M.; Fei, J.H.; Hou, Z.Y. Low-temperature methanol synthesis catalyzed over Cu/γ-Al2O3–TiO2 for CO2 hydrogenation. Catal. Lett. 2001, 72, 191–196. [Google Scholar] [CrossRef]
- Melián-Cabrera, I.; López Granados, M.; Fierro, J.L.G. Reverse Topotactic Transformation of a Cu–Zn–Al Catalyst during Wet Pd Impregnation: Relevance for the Performance in Methanol Synthesis from CO2/H2 Mixtures. J. Catal. 2002, 210, 273–284. [Google Scholar] [CrossRef]
- Chen, H.Y.; Lin, J.; Tan, K.L.; Li, J. Comparative studies of manganese-doped copper-based catalysts: The promoter effect of Mn on methanol synthesis. Appl. Surf. Sci. 1998, 126, 323–331. [Google Scholar] [CrossRef]
- Chen, H.-B.; Liao, D.-W.; Yu, L.-J.; Lin, Y.-J.; Yi, J.; Zhang, H.-B.; Tsai, K.-R. Influence of trivalent metal ions on the surface structure of a copper-based catalyst for methanol synthesis. Appl. Surf. Sci. 1999, 147, 85–93. [Google Scholar] [CrossRef]
- Pokrovski, K.A.; Rhodes, M.D.; Bell, A.T. Effects of cerium incorporation into zirconia on the activity of Cu/ZrO2 for methanol synthesis via CO hydrogenation. J. Catal. 2005, 235, 368–377. [Google Scholar] [CrossRef]
- Fujita, S.-i.; Moribe, S.; Kanamori, Y.; Kakudate, M.; Takezawa, N. Preparation of a coprecipitated Cu/ZnO catalyst for the methanol synthesis from CO2—effects of the calcination and reduction conditions on the catalytic performance. Appl. Catal. A Gen. 2001, 207, 121–128. [Google Scholar] [CrossRef]
- Behrens, M.; Brennecke, D.; Girgsdies, F.; Kißner, S.; Trunschke, A.; Nasrudin, N.; Zakaria, S.; Idris, N.F.; Hamid, S.B.A.; Kniep, B. Understanding the complexity of a catalyst synthesis: Co-precipitation of mixed Cu,Zn,Al hydroxycarbonate precursors for Cu/ZnO/Al2O3 catalysts investigated by titration experiments. Appl. Catal. A Gen. 2011, 392, 93–102. [Google Scholar] [CrossRef]
- Jeong, Y.; Kim, I.; Kang, J.Y.; Jeong, H.; Park, J.K.; Park, J.H.; Jung, J.C. Alcohol-assisted low temperature methanol synthesis from syngas over Cu/ZnO catalysts: Effect of pH value in the co-precipitation step. J. Mol. Catal. A Chem. 2015, 400, 132–138. [Google Scholar] [CrossRef]
- Smith, P.J.; Kondrat, S.A.; Chater, P.A.; Yeo, B.R.; Shaw, G.M.; Lu, L.; Bartley, J.K.; Taylor, S.H.; Spencer, M.S.; Kiely, C.J. A new class of Cu/ZnO catalysts derived from zincian georgeite precursors prepared by co-precipitation. Chem. Sci. 2017, 8, 2436–2447. [Google Scholar] [CrossRef] [PubMed]
- Mashayekh-Salehi, A.; Moussavi, G.; Yaghmaeian, K. Preparation, characterization and catalytic activity of a novel mesoporous nanocrystalline MgO nanoparticle for ozonation of acetaminophen as an emerging water contaminant. Chem. Eng. J. 2017, 310, 157–169. [Google Scholar] [CrossRef]
- Shi, L.; Tao, K.; Yang, R.; Meng, F.; Xing, C.; Tsubaki, N. Study on the preparation of Cu/ZnO catalyst by sol–gel auto-combustion method and its application for low-temperature methanol synthesis. Appl. Catal. A Gen. 2011, 401, 46–55. [Google Scholar] [CrossRef]
- Huang, L.; Kramer, G.J.; Wieldraaijer, W.; Brands, D.S.; Poels, E.K.; Castricum, H.L.; Bakker, H. Methanol synthesis over Cu/ZnO catalysts prepared by ball milling. Catal. Lett. 1997, 48, 55–59. [Google Scholar] [CrossRef]
- Burch, R.; Chappell, R.J. Support and additive effects in the synthesis of methanol over copper catalysts. Appl. Catal. 1988, 45, 131–150. [Google Scholar] [CrossRef]
- Jensen, J.R.; Johannessen, T.; Wedel, S.; Livbjerg, H. A study of Cu/ZnO/Al2O3 methanol catalysts prepared by flame combustion synthesis. J. Catal. 2003, 218, 67–77. [Google Scholar] [CrossRef]
- Pass, G.; Holzhauser, C.; Akgerman, A.; Anthony, R.G. Methanol synthesis in a trickle-bed reactor. AIChE J. 1990, 36, 1054–1060. [Google Scholar] [CrossRef]
- Biswal, T.; Shadangi, K.P.; Sarangi, P.K.; Srivastava, R.K. Conversion of carbon dioxide to methanol: A comprehensive review. Chemosphere 2022, 298, 134299. [Google Scholar] [CrossRef]
- Bowker, M. Methanol Synthesis from CO2 Hydrogenation. ChemCatChem 2019, 11, 4238–4246. [Google Scholar] [CrossRef] [PubMed]
- Guil-López, R.; Mota, N.; Llorente, J.; Millán, E.; Pawelec, B.; Fierro, J.L.G.; Navarro, R.M. Methanol Synthesis from CO2: A Review of the Latest Developments in Heterogeneous Catalysis. Materials 2019, 12, 3902. [Google Scholar] [CrossRef] [PubMed]
- Dang, S.; Yang, H.; Gao, P.; Wang, H.; Li, X.; Wei, W.; Sun, Y. A review of research progress on heterogeneous catalysts for methanol synthesis from carbon dioxide hydrogenation. Catal. Today 2019, 330, 61–75. [Google Scholar] [CrossRef]
- Studt, F.; Sharafutdinov, I.; Abild-Pedersen, F.; Elkjaer, C.F.; Hummelshoj, J.S.; Dahl, S.; Chorkendorff, I.; Norskov, J.K. Discovery of a Ni-Ga catalyst for carbon dioxide reduction to methanol. Nat. Chem. 2014, 6, 320–324. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, G.; Zhu, J.; Zhang, X.; Ding, F.; Zhang, A.; Guo, X.; Song, C. CO2 Hydrogenation to Methanol over In2O3-Based Catalysts: From Mechanism to Catalyst Development. ACS Catal. 2021, 11, 1406–1423. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, G.; Hagelin-Weaver, H.; Welt, B. A Concise Review of Catalytic Synthesis of Methanol from Synthesis Gas. Waste 2023, 1, 228-248. https://doi.org/10.3390/waste1010015
Liu G, Hagelin-Weaver H, Welt B. A Concise Review of Catalytic Synthesis of Methanol from Synthesis Gas. Waste. 2023; 1(1):228-248. https://doi.org/10.3390/waste1010015
Chicago/Turabian StyleLiu, Guanfu, Helena Hagelin-Weaver, and Bruce Welt. 2023. "A Concise Review of Catalytic Synthesis of Methanol from Synthesis Gas" Waste 1, no. 1: 228-248. https://doi.org/10.3390/waste1010015
APA StyleLiu, G., Hagelin-Weaver, H., & Welt, B. (2023). A Concise Review of Catalytic Synthesis of Methanol from Synthesis Gas. Waste, 1(1), 228-248. https://doi.org/10.3390/waste1010015






