Spent Nuclear Fuel—Waste or Resource? The Potential of Strategic Materials Recovery during Recycle for Sustainability and Advanced Waste Management
Abstract
1. Introduction
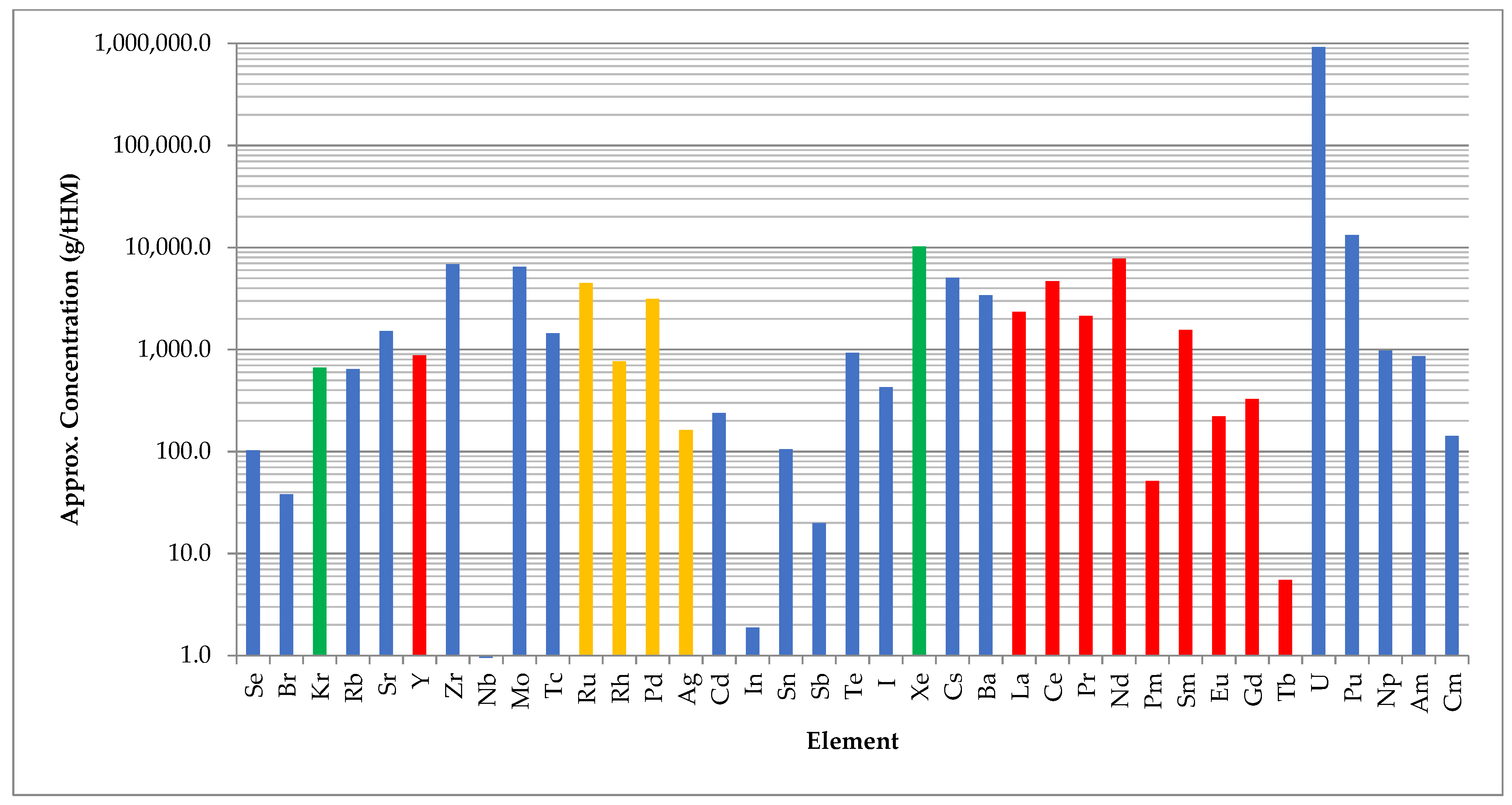
2. Resources within SNF
2.1. Elements
2.1.1. Actinides
2.1.2. Fission Products
- The PGMs do not completely dissolve in aqueous nitric acid media when SNF is processed in the early stages of SNF recycle, meaning that partitioning and full recovery of these metals using aqueous process technology compatible with current SNF recycle processes is likely to be difficult [27] (Ch. 8). The partitioning of Ru specifically is further complicated by the (partial) formation of volatile RuO4 [34,35].
- Several radioisotopes of the PGMs are produced during fission, which may limit the end-uses open to PGMs recovered from SNF, or necessitate decay storage [8]:
- Rh is essentially monoisotopic (as Table 1 03Rh), though trace (<0.1 wt% radioactive) 101Rh (t0.5 = 3.3 y)—which decays by electron capture—and 102Rh (t0.5 = 0.57 y)—a γ emitter—are typically found in SNF [7,8]. These would necessitate decay storage of up to 30 years for the most sensitive applications, such as electronic applications [7,8], but shorter storage times are likely be needed for the most common use for Rh in catalytic converters in the automotive industry and in other industrial processes.
- Ru contains approx 0.2 wt% 106Ru (after 5 years of post-reactor cooling, t0.5 = 1.02 y), which decays to the short-lived but high-decay energy 106Rh (t0.5 = 30 s, decay energy = 3.54 MeV). 106Ru has been proposed for use in brachytherapy [36]. The shorter-lived 103Ru (t0.5 = 39.2 d) will have entirely decayed to stability after the 5 or more years of cooling time needed by HBU SNF before aqueous recycle operations can commence [7].
- The vast majority of proposed MA separation and recovery processes also partition and separately strip the chemically similar REEs [2,40], thus providing a viable recovery route to these elements. Conventional chromatographic techniques used industrially could then be used to separate the individual REEs from each other [41,42,43].
- The most valuable REEs outlined above exist only as stable isotopes [8], alongside Y, La, and Tb. Ce, Pm, Sm, and Eu all possess radioisotopes, and thus effective decontamination between elements would be required.
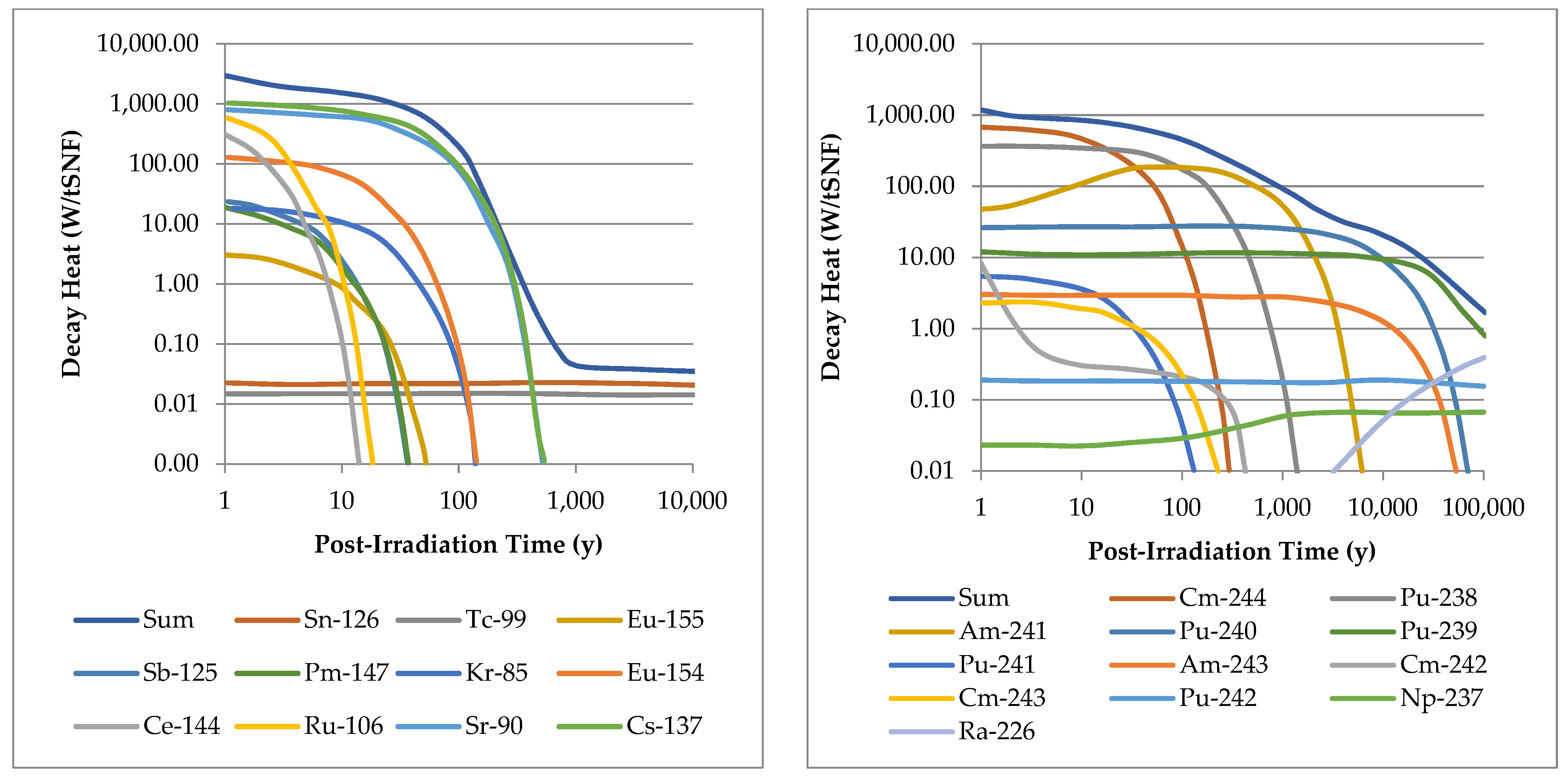
2.2. Isotopes
- 90Y (the daughter of 90Sr) [48] and 106Rh (the daughter of 106Ru) [49] have been proposed as β- and γ-emitting radiopharmaceuticals, respectively. There is also potential for 126Sb (the daughter of 126Sn) [50] and 144Pr (the daughter of 144Ce) to be used in this role [51]. 137Cs, alongside 60Co, is already used for cancer therapy in developing economies [52].
3. Separation, Recovery, and Purification Methods
3.1. Rare Earth Elements
3.2. Platinum Group Metals
3.3. Noble Gases
3.4. Radioisotopes
3.5. Operational Considerations
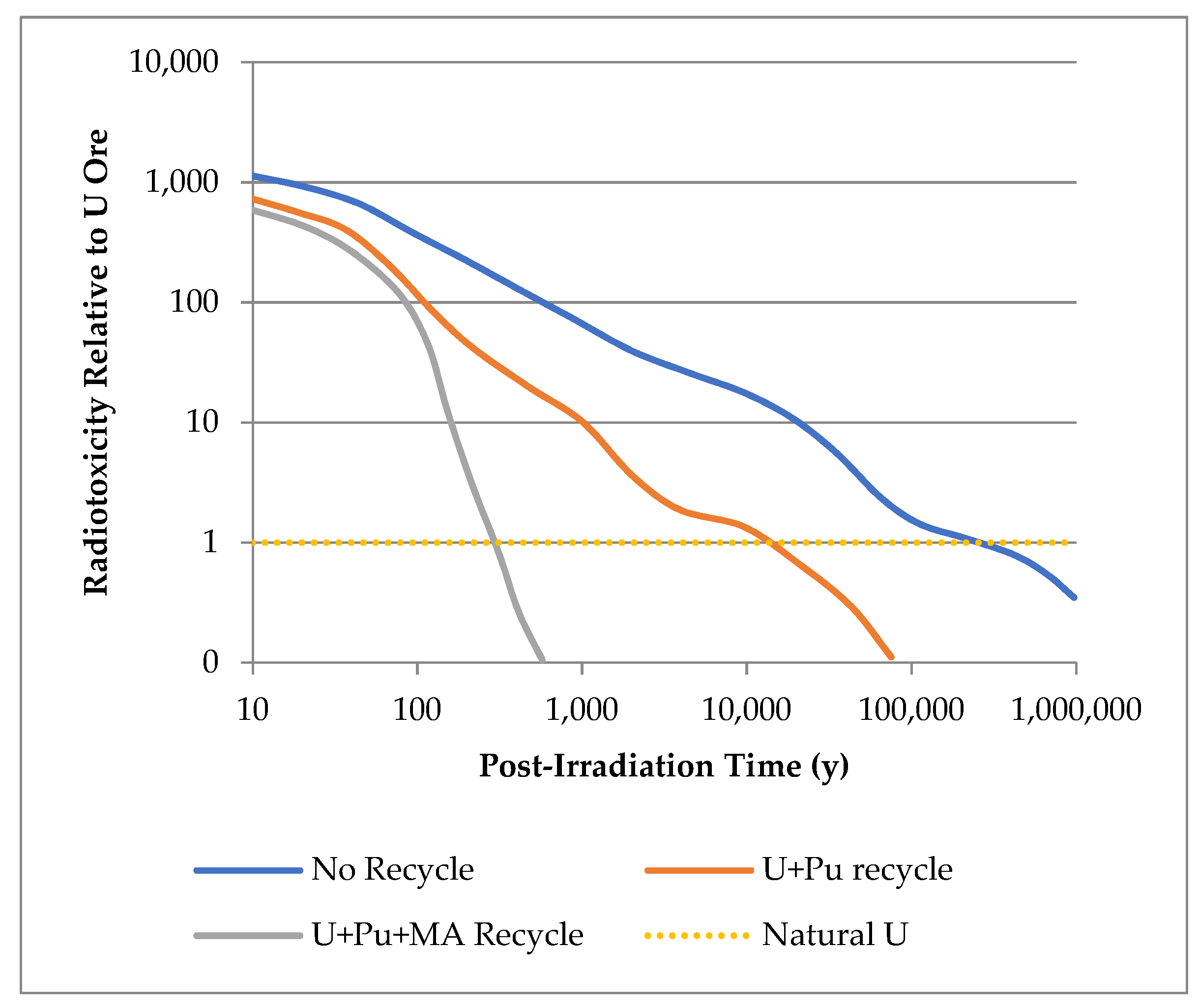
4. Challenges
- A thorough assessment to identify all elemental and isotopic resources present in SNF, both actinide and FP, and their potential values and acceptable end-use cases accounting for radioactivity while assessing the necessity for decay storage.
- The technologies necessary to selectively (where possible) separate these resources during SNF recycle using minimally disruptive processes (i.e., with minimal feed adjustments and arising effects on downstream operations).
- A complete safety case with a technical and economic assessment of these processes and recoveries on SNF recycle operations once technological means for the recovery of economically viable resources are developed.
- A cradle-to-grave life cycle assessment of these processes on the NFC and effects on supporting and competing industries, including comparisons against the incumbent methods of sourcing the recovered element, i.e., mining operations [12].
- Compliance with national and international regulatory factors, or development of appropriate frameworks where these are insufficient for emerging technologies.
- Consideration of emerging concepts in the NFC, including new reactor types (e.g., Gen IV reactors), new SNF types (e.g., advanced technology fuels—ATFs), recycle technologies (e.g., voloxidation), etc.
5. Conclusions
- The PGMs represent the most valuable FP component in SNF (up to USD 500,000 /tHM in HBU SNF) and thus the most appealing to recover. However, given the chemical and radiological complexities involved, these are also likely the most challenging to isolate. Decay storage may be required for certain end-use applications.
- Xe represents a valuable component of SNF (up to USD 30,000/tHM in HBU SNF) which could be readily recovered using current technologies, though decontamination of Kr will be essential to avoid trace radioactivity. He levels present in SNF are likely to be too small to be of consideration for recovery, though this could be reconsidered if global shortages increase in severity and prices increase further.
- The REEs will be isolated if MA partitioning is employed in future SNF recycle, and, as such, are already separated from the bulk raffinate and other FPs. Conventional chromatography can separate these elements, though sufficient decontamination between elements will be required for radiological safety. This would likely require much higher REE prices to be economically viable given the value present in SNF (~USD 1000 s/tHM in HBU SNF), but if shortages increase, this prospect could become more attractive.
- The direct economic values of isotopes are much harder to discern, but increased availability and supply would lower the cost of implementing advanced radioisotope technologies for any species recovered from SNF. The complexities in recovering and handling the range of elements with radioactive isotopes present in SNF represent an additional layer of complexity compared to the simple recovery of the elements.
- Technological, regulatory, environmental, and socioeconomic assessments of all aspects of this concept are required before implementation can be considered.
- The potential to offset the presently high costs of the NFC and reduce current, large waste volumes and thus the potential load on geological repositories by maximising recovery of resources in SNF is one of many drivers behind such research, especially with the necessity to decarbonise power grids as soon as is possible.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Element | Conc. (g/tHM) | Elem. Value (USD/kg) | Radioisotope(s) (t0.5 – y) | Notes/Applications of Isotopes | |
|---|---|---|---|---|---|
| Noble Gases | He | ~10 | 50 (g) | Stable | |
| Kr | 665 | 330 (g) | 85Kr (10.8) | ||
| Xe | 10,244 | 3000 (g) | Stable | ||
| Rare Earth Elements | Y | 874 | 4 (ox) | Stable | |
| La | 2332 | 2 (ox) | Stable | ||
| Ce | 4665 | 2 (ox) | 144Ce (0.78) | 144Pr (17.3 m) daughter potential medical isotope | |
| Pr | 2138 | 136 (ox) | Stable | ||
| Nd | 7792 | 136 (ox) | Stable | ||
| Pm | 52 | - | 147Pm (2.62) | Potential betavoltaic source [87] | |
| Sm | 1560 | 4 (ox) | 151Sr (90) | ||
| Eu | 221 | 29 (ox) | 154Eu (8.6), 155Eu (4.8) | ||
| Gd | 326 | 69 (ox) | Stable | ||
| Tb | 6 | 2 (ox) | Stable | ||
| Plat. Grp. Metals | Ru | 4470 | 19,000 (m) | 106Ru (1.02) | 106Ru/106Rh (30 s) potential medical isotope |
| Rh | 767 | 451,000 (m) | 101Rh (3.3), 102Rh (0.56) | ||
| Pd | 3126 | 61,000 (m) | 107Pd (6.5M) | ||
| Ag | 162 | 1,000 (m) | 110mAg (0.68) | ||
| Actinides | U | 917,823 | - | Various | Fissile/fertile |
| Np | 974 | - | 237Np (2.1 M) | Precursor to 238Pu for RTGs [29] | |
| Pu | 13,700 | - | Various | MOX fuel | |
| Am | 860 | - | 241Am | RTG isotope [52], precursor to 238Pu for RTGs (via 242Cm) [29] | |
| Cm | 142 | - | Various | Transmutation targets | |
| Useful Isotopes | Cs | 5035 | - | 134Cs (2.1), 135Cs (2.3 M) 137Cs (30.1) | HHR, irradiation |
| Sr | 1518 | - | 90Sr (28.9) | HHR, RTG isotope, 90Y (64 h) daughter medical isotope | |
| Sn | 105 | - | 126Sn (218 k) | 126Sb (12.4 d) daughter potential medical isotope |
References
- Murray, R. Understanding Radioactive Waste; Report PNL-3570; Pacific Northwest Lab.: Richland, WA, USA, 1981. [Google Scholar] [CrossRef]
- Baron, P.; Cornet, S.M.; Collins, E.D.; DeAngelis, G.; Del Cul, G.; Fedorov, Y.; Glatz, J.P.; Ignatiev, V.; Inoue, T.; Khaperskaya, A.; et al. A review of separation processes proposed for advanced fuel cycles based on technology readiness level assessments. Prog. Nucl. Energy 2019, 117, 24. [Google Scholar] [CrossRef]
- Bunn, M.; Holdren, J.P.; Fetter, S.; van der Zwaan, B. The Economics of Reprocessing versus Direct Disposal of Spent Nuclear Fuel. Nucl. Technol. 2005, 150, 209–230. [Google Scholar] [CrossRef]
- Kim, S.; Ko, W.; Nam, H.; Kim, C.; Chung, Y.; Bang, S. Statistical model for forecasting uranium prices to estimate the nuclear fuel cycle cost. Nucl. Eng. Technol. 2017, 49, 1063–1070. [Google Scholar] [CrossRef]
- Woo, S.M.; Chirayath, S.S.; Fuhrmann, M. Nuclear fuel reprocessing: Can pyro-processing reduce nuclear proliferation risk? Energy Policy 2020, 144, 111601. [Google Scholar] [CrossRef]
- Ho, S.S.; Leong, A.D.; Looi, J.; Chen, L.; Pang, N.; Tandoc, E. Science Literacy or Value Predisposition? A Meta-Analysis of Factors Predicting Public Perceptions of Benefits, Risks, and Acceptance of Nuclear Energy. Environ. Commun. 2018, 13, 457–471. [Google Scholar] [CrossRef]
- Bush, R.P. Recovery of Platinum Group Metals from High Level Radioactive Waste. Plat. Group Rev. 1991, 35, 202–208. [Google Scholar]
- Bourg, S.; Poinssot, C. Could spent nuclear fuel be considered as a non-conventional mine of critical raw materials? Prog. Nucl. Energy 2017, 94, 222–228. [Google Scholar] [CrossRef]
- Koch, H.; Schober, A. On the possibilities of the extraction and application of low-activity fission products from high-activity nuclear fuel wastes. Isotopenpraxis 1976, 12, 49–56. (In German) [Google Scholar]
- Lang, L.W.; Deonigi, D.E.; Rohrmann, C.A. Power Cost Reduction from Isotope Revenues. Nucl. Appl. 1967, 3, 665–678. [Google Scholar] [CrossRef]
- European Chemical Society. Available online: https://www.euchems.eu/euchems-periodic-table/ (accessed on 20 October 2022).
- Kouloumpis, V.; Stamford, L.; Azapagic, A. Decarbonising electricity supply: Is climate change mitigation going to be carried out at the expense of other environmental impacts? Sustain. Prod. Consum. 2015, 1, 1–21. [Google Scholar] [CrossRef]
- Bodel, W.; Hesketh, K.; McGlynn, G.; Matthews, J.; Butler, G. Generic Feasibility Assessment: Helping to Choose the Nuclear Piece of the Net Zero Jigsaw. Energies 2021, 14, 1229. [Google Scholar] [CrossRef]
- Poinssot, C.; Rostaing, C.; Greandjean, S.; Boullis, B. Recycling the Actinides, The Cornerstone of Any Sustainable Nuclear Fuel Cycles. Procedia Chem. 2012, 7, 349–357. [Google Scholar] [CrossRef]
- Eccles, H. Nuclear fuel cycle technologies -sustainable in the twenty first century? Solvent Extr. Ion Exch. 2000, 18, 633–654. [Google Scholar] [CrossRef]
- Rodríguez-Penalonga, L.; Soria, B.Y.M. A Review of the Nuclear Fuel Cycle Strategies and the Spent Nuclear Fuel Management Technologies. Energies 2017, 10, 1235. [Google Scholar] [CrossRef]
- Taylor, R.; Bodel, W.; Stamford, L.; Butler, G. A Review of Environmental and Economic Implications of Closing the Nuclear Fuel Cycle—Part One: Wastes and Environmental Impacts. Energies 2022, 15, 1433. [Google Scholar] [CrossRef]
- Taylor, R.; Bodel, W.; Butler, G. A Review of Environmental and Economic Implications of Closing the Nuclear Fuel Cycle—Part Two: Economic Impacts. Energies 2022, 15, 2472. [Google Scholar] [CrossRef]
- Baker, A.; Fells, A.; Carrott, M.J.; Maher, C.J.; Hanson, B.C. Process intensification of element extraction using centrifugal contactors in the nuclear fuel cycle. Chem. Soc. Rev. 2022, 51, 3964–3999. [Google Scholar] [CrossRef]
- Fernández-Arias, P.; Vergara, D.; Orosa, J.A. A Global Review of PWR Nuclear Power Plants. Appl. Sci. 2020, 10, 4434. [Google Scholar] [CrossRef]
- Forsberg, C.W. Rethinking High-Level Waste Disposal: Separate Disposal of High-Heat Radionuclides (90Sr and 137Cs). Nucl. Technol. 2000, 131, 252–268. [Google Scholar] [CrossRef]
- Krall, L.; Macfarlane, A. Burning waste or playing with fire? Waste management considerations for non-traditional reactors. Bull. At. Sci. 2018, 74, 326–334. [Google Scholar] [CrossRef]
- Mallants, D.; Travis, K.; Chapman, N.; Brady, P.V.; Griffiths, H. The State of the Science and Technology in Deep Borehole Disposal of Nuclear Waste. Energies 2020, 13, 833. [Google Scholar] [CrossRef]
- Holdsworth, A.F.; George, K.; Adams, S.J.; Sharrad, C.A. An accessible statistical regression approach for the estimation of spent nuclear fuel compositions and decay heats to support the development of nuclear fuel management strategies. Prog. Nucl. Energy 2021, 141, 103935. [Google Scholar] [CrossRef]
- Francis, M.W.; Weber, C.F.; Pigni, M.T.; Gauld, I.C. Reactor Fuel Isotopics and Code Validation for Nuclear Applications; National Technical Information: Springfield, VA, USA, 2015. [Google Scholar] [CrossRef]
- Ando, Y.; Takano, H. Estimation of LWR Spent Fuel Composition, Report: JAERI-Research-99-004; Japanese Atomic Energy Research Agency: Tokai, Japan, 1999. [Google Scholar]
- Nash, K.L.; Lumetta, G.J. (Eds.) Advanced Separation Techniques for Nuclear Fuel Reprocessing and Radioactive Waste Treatment; Woodhead: Oxford, UK, 2011. [Google Scholar] [CrossRef]
- Salvatores, M.; Palmiotti, G. Radioactive waste partitioning and transmutation within advanced fuel cycles: Achievements and challenges. Prog. Part. Nucl. Phys. 2011, 66, 144–166. [Google Scholar] [CrossRef]
- Dustin, J.S.; Borrelli, R. Assessment of alternative radionuclides for use in a radioisotope thermoelectric generator. Nucl. Eng. Des. 2021, 385, 111475. [Google Scholar] [CrossRef]
- Vesnovskii, S.P. RFNC-VNIIEF capabilities to production high pure isotopes for scientific and medical applications. J. Radioanal. Nucl. Chem. 2003, 257, 27–31. [Google Scholar] [CrossRef]
- Rirnbaum., E.R.; Fassbender, M.E.; Ferrier, M.G.; John, K.D.; Mastren, T. Actindes in Medicine. In The Heaviest Metals: Science and Technology of The Actinides and Beyond; Evans, W.J., Hanusa, T.P., Eds.; Wiley: Oxford, UK, 2007; pp. 445–466. [Google Scholar]
- Ternovykh, M.; Tikhomirov, G.; Saldikov, I.; Gerasimov, A. Decay heat power of spent nuclear fuel of power reactors with high burnup at long-term storage. EPJ Web Conf. 2017, 153, 7035. [Google Scholar] [CrossRef]
- Johnson Matthey. Available online: https://matthey.com/products-and-markets/pgms-and-circularity/pgm-management (accessed on 29 October 2022).
- Swain, P.; Mallika, C.; Srinivasan, R.; Mudali, U.K.; Natarajan, R. Separation and recovery of ruthenium: A review. J. Radioanal. Nucl. Chem. 2013, 298, 781–796. [Google Scholar] [CrossRef]
- Moeyaert, P.; Miguirditchian, M.; Masson, M.; Dinh, B.; Hérès, X.; De Sio, S.; Sorel, C. Experimental and modelling study of ruthenium extraction with tri-n-butylphosphate in the purex process. Chem. Eng. Sci. 2017, 158, 580–586. [Google Scholar] [CrossRef]
- Pe’er, J. Ruthenium-106 brachytherapy. In Current Concepts in Uveal Melanoma; Jager, M.J., Desjardins, L., Kivela, T., Damato, B.E., Eds.; Karger: Basel, Switzerland, 2012; Volume 49, pp. 27–40. [Google Scholar] [CrossRef]
- Allison, W. We Should Stop Running Away from Radiation. Philos. Technol. 2011, 24, 193–195. [Google Scholar] [CrossRef]
- Adams, B.D.; Chen, A. The role of palladium in a hydrogen economy. Mater. Today 2011, 14, 282–289. [Google Scholar] [CrossRef]
- SMM. Available online: https://www.metal.com/Rare-Earth-Oxides (accessed on 29 October 2022).
- Natrajan, L.S.; Langford Paden, M.H. F-block Elements Recovery, in RSC Green Chemistry No. 22—Element Recovery and Sustainability; Hunt, A.J., Ed.; Royal Society of Chemistry: Cambridge, UK, 2013; Chapter 6. [Google Scholar]
- Asadollahzadeh, M.; Torkaman, R.; Torab-Mostaedi, M. Extraction and Separation of Rare Earth Elements by Adsorption Approaches: Current Status and Future Trends. Sep. Purif. Rev. 2020, 50, 417–444. [Google Scholar] [CrossRef]
- Chen, Z.; Li, Z.; Chen, J.; Kallem, P.; Banat, F.; Qiu, H. Recent advances in selective separation technologies of rare earth elements: A review. J. Environ. Chem. Eng. 2021, 10, 107104. [Google Scholar] [CrossRef]
- Opare, E.O.; Struhs, E.; Mirkouei, A. A comparative state-of-technology review and future directions for rare earth element separation. Renew. Sustain. Energy Rev. 2021, 143, 110917. [Google Scholar] [CrossRef]
- Tingey, G.L.; McClanahan, E.D.; Bayne, M.A.; Gray, W.J.; Hinman, C.A. Krypton-85 Storage in Solid Matrices in Scientific Basis for Nuclear Waste Management; Northrup, C.J.M., Ed.; Springer: New York, NY, USA, 1980; Chapter 18. [Google Scholar]
- Chemistry Learner. Available online: https://www.chemistrylearner.com/krypton-85.html (accessed on 29 October 2022).
- Aisen, E.M.; Borisevich, V.D.; Levin, E.V.; Yupatov, S.V.; Tikhomirov, A.V.; Popov, G.E. Separation of stable isotopes of low, middle, and heavy masses by a gas centrifuge. In Proceedings of the Workshop on Separation Phenomena in Liquids and Gases, Foz do Iguacu, Brazil, 22–26 September 1996. [Google Scholar]
- Marty, B. On the noble gas isotopic fractionation in naturally occurring gases. Geochem. J. 1984, 18, 157–162. [Google Scholar] [CrossRef]
- Williams, S.J.; Rilling, W.S.; White, S.B. Quality of Life and Cost Considerations: Y-90 Radioembolization. Semin. Interv. Radiol. 2021, 38, 482–487. [Google Scholar] [CrossRef]
- Katsaros, N.; Anagnostopoulou, A. Rhodium and its compounds as potential agents in cancer treatment. Crit. Rev. Oncol. 2002, 42, 297–308. [Google Scholar] [CrossRef]
- Robertson, A.K.H.; Kunz, P.; Hoehr, C.; Schaffer, P. Nuclide production cross sections from irradiation of thorium by 438 MeV protons and a comparison to fluka and geant4 simulations. Phys. Rev. C 2020, 102, 044613. [Google Scholar] [CrossRef]
- Amoroso, A.J.; Fallis, I.A.; Pope, S.J. Chelating agents for radiolanthanides: Applications to imaging and therapy. Coord. Chem. Rev. 2017, 340, 198–219. [Google Scholar] [CrossRef]
- Murayama, Y.; Feloa, J.M.; Beach, J.L. HeLa cell tumor response to 60Co, Cs-137, Cf-252 radiations and cisplatin chemotherapy in nude mice. Cancer 1984, 52, 247–252. [Google Scholar] [CrossRef]
- Mikhalev, A.V.; Chernov, D.O.; Korobeinikov, V.V. Use of Am-241 in RTGs. J. Phys. Conf. Ser. 2020, 1689, 012063. [Google Scholar] [CrossRef]
- Bykov, A.S.; Malinkovich, M.D.; Kubasov, I.V.; Kislyuk, A.M.; Kiselev, D.A.; Ksenich, S.V.; Zhukov, R.N.; Temirov, A.A.; Chichkov, M.V.; Polisan, A.A.; et al. Application of Radioactive Isotopes for Beta-Voltaic Generators. Russ. Microelectron. 2017, 46, 527–539. [Google Scholar] [CrossRef]
- Boisseau, P. Food Preservation by Irradiation: A Brief Introduction. Solid State Phenom. 1992, 30–31, 433–450. [Google Scholar] [CrossRef]
- Cao, Y.; Zhou, L.; Ren, H.; Zou, H. Determination, Separation and Application of 137Cs: A Review. Int. J. Enviro. Res. Pub. Health 2022, 19, 10183. [Google Scholar] [CrossRef] [PubMed]
- McKibben, J.M. Chemistry of the Purex Process. Radiocim. Acta 1984, 36, 3–15. [Google Scholar] [CrossRef]
- Bond, G.; Eccles, H.; Kavi, P.C.; Holdsworth, A.F.; Rowbotham, D.; Mao, R. Removal of Cesium from Simulated Spent Fuel Dissolver Liquor. J. Chromatog. Separ. Tech. 2019, 10, 417. [Google Scholar]
- Holdsworth, A.F.; Eccles, H.; Rowbotham, D.; Bond, G.; Kavi, P.C.; Edge, R. The Effect of Gamma Irradiation on the Ion Exchange Properties of Caesium-Selective Ammonium Phosphomolybdate-Polyacrylonitrile (AMP-PAN) Composites under Spent Fuel Recycling Conditions. Separations 2019, 6, 23. [Google Scholar] [CrossRef]
- Holdsworth, A.F.; Eccles, H.; Rowbotham, D.; Brookfield, A.; Collison, D.; Bond, G.; Kavi, P.C.; Edge, R. The Effect of Gamma Irradiation on the Physiochemical Properties of Caesium-Selective Ammonium Phosphomolybdate–Polyacrylonitrile (AMP–PAN) Composites. Clean Technol. 2019, 11, 20. [Google Scholar] [CrossRef]
- Holdsworth, A.F.; Eccles, H.; Bond, G.; Rowbotham, D.; Brookfield, A.; Collison, D.; Kavi, P.C.; Natrajan, L.S.; Spencer, B.; Muryn, C.; et al. Heterogeneous Separations of Highly Active Radionuclides for Advanced Decay Heat & Waste Management in Next-Generation Spent Fuel Recycling/Reprocessing. In Proceedings of the 4th Cloud Conference on Nuclear Waste Management and Disposal, Online, UK, 9 July 2020. [Google Scholar]
- Venkatesan, K.A.; Sukumaran, V.; Antony, M.P.; Srinivasan, T.G. Studies on the feasibility of using crystalline silicotitanates for the separation of cesium-137 from fast reactor high-level liquid waste. J. Radioanal. Nucl. Chem. 2009, 280, 129–136. [Google Scholar] [CrossRef]
- Bibler, N.E. Results of Scoping Studies for Determining Radiolytic Hydrogen Production from Moist CST and CST Slurries; Report WRSC-RP-98-01143; Savannah River Site: Jackson, SC, USA, 1998. [Google Scholar] [CrossRef]
- Banks, H.O., Jr. The Cesium-137 Power Program; Quarterly Report No. 3, Report TID-17004; Royal Research Corp.: Hayward, CA, USA, 1961. [Google Scholar] [CrossRef]
- Amphlett, J.; Ogden, M.; Foster, R.; Syna, N.; Soldenhoff, K.H.; Sharrad, C.A. The effect of contaminants on the application of polyamine functionalised ion exchange resins for uranium extraction from sulfate based mining process waters. Chem. Eng. J. 2018, 354, 633–640. [Google Scholar] [CrossRef]
- Sole, K.C.; Cole, P.M.; Feather, A.M.; Kotze, M.H. Solvent Extraction and Ion Exchange Applications in Africa’s Resurging Uranium Industry: A Review. Solvent Extr. Ion Exch. 2011, 29, 868–899. [Google Scholar] [CrossRef]
- Rahman, R.O.A.; Metwally, S.S.; El-Kamash, A.M. Life Cycle of Ion Exchangers in Nuclear Industry: Application and Management of Spent Exchangers. In Handbook of Ecomater; Martinez, L., Kharissova, O., Kharisov, B., Eds.; Springer: New York, NY, USA, 2019. [Google Scholar] [CrossRef]
- Choi, E.-Y.; Jeong, S.M. Electrochemical processing of spent nuclear fuels: An overview of oxide reduction in pyroprocessing technology. Prog. Nat. Sci. 2015, 25, 572–582. [Google Scholar] [CrossRef]
- Carlson, K.; Gardner, L.; Moon, J.; Riley, B.; Amoroso, J.; Chidambaram, D. Molten salt reactors and electrochemical reprocessing: Synthesis and chemical durability of potential waste forms for metal and salt waste streams. Int. Mater. Rev. 2020, 66, 339–363. [Google Scholar] [CrossRef]
- Yoshida, Z.; Aoyagi, H.; Mutoh, H.; Takeishi, H.; Sasaki, Y.; Uno, S.; Tachikawa, E. Spent fuel reprocessing based on electrochemical extraction process (SREEP). J. Alloys Compd. 1994, 213–214, 453–455. [Google Scholar] [CrossRef]
- Shadrin, A.Y.; Dvoeglazov, K.N.; Mochalov, Y.S.; Vidanov, V.V.; Kashcheev, V.A.; Terentiev, A.G.; Gerasimenko, M.N.; Cheshuyakov, S.A. Hydrometallurgical and combined technologies fast reactor MNUP and MOX UNF reprocessing. J. Phys. Conf. Ser. 2020, 1475, 012021. [Google Scholar] [CrossRef]
- Authen, T.L.; Adnet, J.-M.; Bourg, S.; Carrott, M.; Ekberg, C.; Galán, H.; Geist, A.; Guilbaud, P.; Miguirditchian, M.; Modolo, G.; et al. An overview of solvent extraction processes developed in Europe for advanced nuclear fuel recycling, Part 2—Homogeneous recycling. Sep. Sci. Technol. 2021, 57, 1724–1744. [Google Scholar] [CrossRef]
- Geist, A.; Adnet, J.-M.; Bourg, S.; Ekberg, C.; Galán, H.; Guilbaud, P.; Miguirditchian, M.; Modolo, G.; Rhodes, C.; Taylor, R. An overview of solvent extraction processes developed in Europe for advanced nuclear fuel recycling, part 1—Heterogeneous recycling. Sep. Sci. Technol. 2020, 56, 1866–1881. [Google Scholar] [CrossRef]
- Nash, K.L. The Chemistry of TALSPEAK: A Review of the Science. Solvent Extr. Ion Exch. 2014, 33, 1–55. [Google Scholar] [CrossRef]
- Taylor, R.; Mathers, G. Developments in Advanced Recycle and Sustainability for Future Fuel Cycle Options. In Proceedings of the SNETP Forum, Online, 3 February 2021. [Google Scholar]
- Modolo, G.; Wilden, A.; Geist, A.; Magnusson, D.; Malmbeck, R. A review of the demonstration of innovative solvent extraction processes for the recovery of trivalent minor actinides from PUREX raffinate. Radiochim. Acta 2012, 100, 715–725. [Google Scholar] [CrossRef]
- Wu, H.; Kim, S.-Y.; Takahashi, T.; Oosugi, H.; Ito, T.; Kanie, K. Extraction behaviors of platinum group metals in simulated high-level liquid waste by a hydrophobic ionic liquid bearing an amino moiety. Nucl. Eng. Technol. 2021, 53, 1218–1223. [Google Scholar] [CrossRef]
- Malmbeck, R.; Magnusson, D.; Bourg, S.; Carrott, M.; Geist, A.; Heres, X.; Miguirditchian, M.; Modolo, G.; Mullich, U.; Sorel, C.; et al. Homogenous recycling of transuranium elements from irradiated fast reactor fuel by the EURO-GANEX solvent extraction process. Radiochim. Acta. 2019, 107, 917–929. [Google Scholar] [CrossRef]
- Van Loon, J.C. Accurate determination of the noble metals I. Sample decomposition and methods of separation. TrAC Trends Anal. Chem. 1984, 3, 272–275. [Google Scholar] [CrossRef]
- Holdsworth, A.F.; Eccles, H.; Bond, G.; Sharrad, C.A. Selective Separation of Resources in Used Nuclear Fuel (UNF) Recycle for Fuel Cycle Sustainability. In Proceedings of the 51st Journées des Actinides Conference, Santa Margharita Ligure, Italy, 9–14 April 2022. [Google Scholar]
- Xu, C.; Wang, J.; Chen, J. Solvent Extraction of Strontium and Cesium: A Review of Recent Progress. Solvent Extr. Ion Exch. 2012, 30, 623–650. [Google Scholar] [CrossRef]
- Vincent, T.; Vincent, C.; Guibal, E. Immobilization of Metal Hexacyanoferrate Ion-Exchangers for the Synthesis of Metal Ion Sorbents—A Mini-Review. Molecules 2015, 20, 20582–20613. [Google Scholar] [CrossRef] [PubMed]
- Baetsle, L.; Huys, D. Structure and ion-exchange characteristics of polyantimonic acid. J. Inorg. Nucl. Chem. 1968, 30, 639–649. [Google Scholar] [CrossRef]
- Nilchi, A.; Maragheh, M.G.; Khanchi, A.; Farajzadeh, M.A.; Aghaei, A.A. Synthesis and ion-exchange properties of crystalline titanium and zirconium phosphates. J. Radioanal. Nucl. Chem. Artic. 2004, 261, 393–400. [Google Scholar] [CrossRef]
- Natarajan, R.; Raj, B. Fast Reactor Fuel Reprocessing Technology in India. J. Nucl. Sci. Technol. 2007, 44, 393–397. [Google Scholar] [CrossRef]
- Spent Fuel Reprocessing Options; Report: IAEA-TECDOC-1587; IAEA (International Atomic Energy Agency): Vienna, Austria, 2008.
- Sachenko, A.; Shkrebtii, A.; Korkishko, R.; Kostylyov, V.; Kulish, M.; Sokolovskyi, I. Efficiency analysis of betavoltaic elements. Solid-State Electron. 2015, 111, 147–152. [Google Scholar] [CrossRef]

| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | Ln * | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | An * | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| Ln * | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||
| An * | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Holdsworth, A.F.; Eccles, H.; Sharrad, C.A.; George, K. Spent Nuclear Fuel—Waste or Resource? The Potential of Strategic Materials Recovery during Recycle for Sustainability and Advanced Waste Management. Waste 2023, 1, 249-263. https://doi.org/10.3390/waste1010016
Holdsworth AF, Eccles H, Sharrad CA, George K. Spent Nuclear Fuel—Waste or Resource? The Potential of Strategic Materials Recovery during Recycle for Sustainability and Advanced Waste Management. Waste. 2023; 1(1):249-263. https://doi.org/10.3390/waste1010016
Chicago/Turabian StyleHoldsworth, Alistair F., Harry Eccles, Clint A. Sharrad, and Kathryn George. 2023. "Spent Nuclear Fuel—Waste or Resource? The Potential of Strategic Materials Recovery during Recycle for Sustainability and Advanced Waste Management" Waste 1, no. 1: 249-263. https://doi.org/10.3390/waste1010016
APA StyleHoldsworth, A. F., Eccles, H., Sharrad, C. A., & George, K. (2023). Spent Nuclear Fuel—Waste or Resource? The Potential of Strategic Materials Recovery during Recycle for Sustainability and Advanced Waste Management. Waste, 1(1), 249-263. https://doi.org/10.3390/waste1010016







