Molecular Identification and Ecology of Portuguese Wild-Caught Phlebotomine Sandfly Specimens
Abstract
1. Introduction
2. Materials and Methods
3. Results
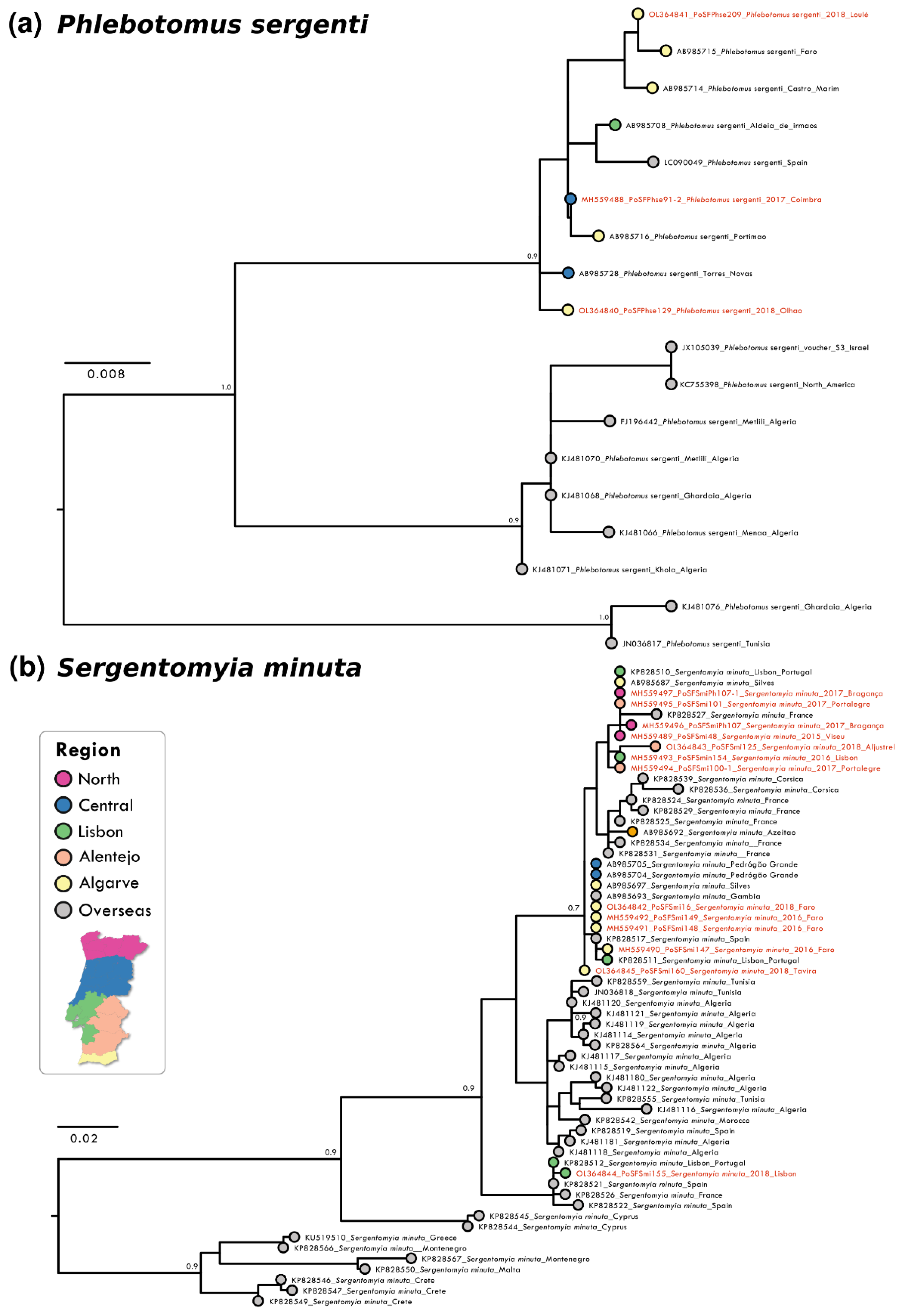
3.1. Genetic Analysis
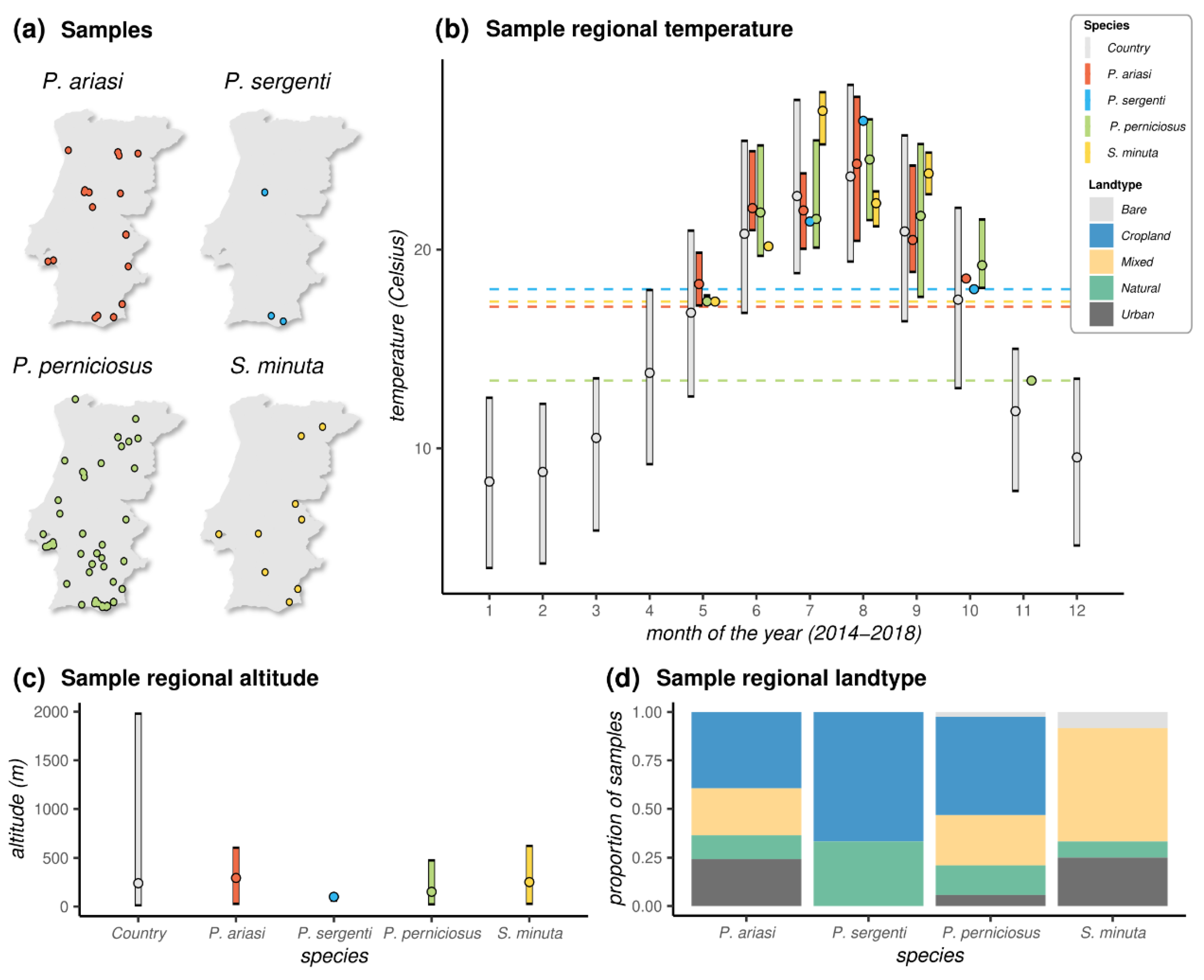
3.2. Distribution of Samples and Ecological Metadata
3.3. Blood Meals
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Svobodová, M.; Sádlová, J.; Chang, K.P.; Volf, P. Short report: Distribution and feeding preference of the sand flies Phlebotomus sergenti and P. papatasi in a cutaneous leishmaniasis focus in Sanliurfa, Turkey. Am. J. Trop. Med. Hyg. 2003, 68, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Maia, C.; Campino, L. Leishmaniasis in Portugal in the beginning of the XXI century. Anais Do IHMT 2018, 13, 25–28. [Google Scholar] [CrossRef]
- Quinnell, R.; Courtenay, O. Transmission, reservoir hosts and control of zoonotic visceral leishmaniasis. Parasitology 2009, 136, 1915–1934. [Google Scholar] [CrossRef] [PubMed]
- WHO Expert Committee on the Control of the Leishmaniases & World Health Organization. Control of the Leishmaniases: Report of a Meeting of the WHO Expert Commitee on the Control of Leishmaniases, Geneva, 22–26 March 2010; World Health Organization: Geneva, Switzerland, 2010; Available online: https://apps.who.int/iris/handle/10665/44412 (accessed on 9 December 2021).
- Pennisi, M.G.; Cardoso, L.; Baneth, G.; Bourdeau, P.; Koutinas, A.; Miró, G.; Oliva, G.; Solano-Gallego, L. LeishVet update and recommendations on feline leishmaniosis. Parasites Vectors 2015, 8, 302. [Google Scholar] [CrossRef] [PubMed]
- Maia, C.; Dantas-Torres, F.; Campino, L. Parasite Biology: The Reservoir Hosts. In The Leishmaniases: Old Neglected Tropical Diseases; Bruschi, F., Gradoni, L., Eds.; Springer: Cham, Switzerland, 2018; pp. 79–106. [Google Scholar] [CrossRef]
- Spada, E.; Perego, R.; Vitale, F.; Bruno, F.; Castelli, G.; Tarantola, G.; Baggiani, L.; Magistrelli, S.; Proverbio, D. Feline Leishmania spp. infection in a Non-Endemic Area of Northern Italy. Animals 2020, 10, 817. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.; Parreira, R.; Cristóvão, J.M.; Vitale, F.; Bastien, P.; Campino, L.; Maia, C. Leishmania infantum strains from cats are similar in biological properties to canine and human strains. Vet. Parasitol. 2021, 298, 109531. [Google Scholar] [CrossRef]
- Pires, C.A. Phlebotomus of Portugal. I-Natural infestation of Phlebotomus ariasi Tonnoir, 1921 and Phlebotomus perniciosus Newstead, 1911, by Leishmania in the zoonotic focus of Arrábida (Portugal). Ann. Parasitol. Hum. Comp. 1984, 59, 521–524. [Google Scholar] [CrossRef]
- International Committee on Taxonomy of Viruses. Virus Taxonomy Release: 2020 Release. Email Ratification March 2021 (MSL #36). Available online: https://talk.ictvonline.org/taxonomy/ (accessed on 9 December 2021).
- Alkan, C.; Bichaud, L.; de Lamballerie, X.; Alten, B.; Gould, E.A.; Charrel, R.N. Sandfly-borne phleboviruses of Eurasia and Africa: Epidemiology, genetic diversity, geographic range, control measures. Antivir. Res. 2013, 100, 54–74. [Google Scholar] [CrossRef]
- Moriconi, M.; Rugna, G.; Calzolari, M.; Bellini, R.; Albieri, A.; Angelini, P.; Cagarelli, R.; Landini, M.P.; Charrel, R.N.; Varani, S. Phlebotomine sand fly-borne pathogens in the Mediterranean Basin: Human leishmaniasis and phlebovirus infections. PLOS Negl. Trop. Dis. 2017, 11, e0005660. [Google Scholar] [CrossRef]
- Amaro, F.; Zé-Zé, L.; Alves, M.J.; Börstler, J.; Clos, J.; Lorenzen, S.; Becker, S.C.; Schmidt-Chanasit, J.; Cadar, D. Co-circulation of a novel phlebovirus and Massilia virus in sandflies, Portugal. Virol. J. 2015, 12, 174. [Google Scholar] [CrossRef]
- Amaro, F.; Hanke, D.; Zé-Zé, L.; Alves, M.J.; Becker, S.C.; Höper, D. Genetic characterization of Arrabida virus, a novel phlebovirus isolated in South Portugal. Virus Res. 2016, 214, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Amaro, F.; Zé-Zé, L.; Lourenço, J.; Giovanetti, M.; Becker, S.C.; Alves, M.J. Phylogenetic Analysis of Massilia phlebovirus in Portugal. Viruses 2021, 13, 1412. [Google Scholar] [CrossRef] [PubMed]
- Amaro, F.; Zé-Zé, L.; Luz, M.T.; Alves, M.J. Toscana Virus: Ten Years of Diagnostics in Portugal. Acta Med. Port. 2021, 34, 677–681. [Google Scholar] [CrossRef] [PubMed]
- Service, M. Phlebotomine sand-flies (Phlebotominae). In Medical Entomology for Students; Cambridge University Press: Cambridge, UK, 2008; pp. 93–102. [Google Scholar] [CrossRef]
- França, C. Phlebotomus papatasi (Scopoli) et fièvre à Pappataci au Portugal. Bull. Soc. Pathol. Éxotique 1913, 6, 123–124. [Google Scholar]
- França, C. Notes sur les espèces portugaises du genre Phlebotomus. Bull. Soc. Pathol. Éxotique 1918, 11, 730–733. [Google Scholar]
- Alves-Pires, C.; Afonso, M.O.; Janz, J.G.; Semião-Santos, S.J. The phlebotomine sand flies of Portugal. XII. The phlebotomine of Évora Leishmaniasis focus (1999–2000). Acta Parasitológica Port. 2004, 11, 41–45. [Google Scholar]
- European Centre for Disease Prevention and Control. Phlebotomine sand flies—Factsheet for Experts. 2015. Available online: https://www.ecdc.europa.eu/en/disease-vectors/facts/phlebotomine-sand-flies (accessed on 28 December 2021).
- Alten, B.; Maia, C.; Afonso, M.O.; Campino, L.; Jiménez, M.; González, E.; Molina, R.; Bañuls, A.L.; Prudhomme, J.; Vergnes, B.; et al. Seasonal dynamics of phlebotomine sand fly species proven vectors of Mediterranean leishmaniasis caused by Leishmania infantum. PLoS Negl. Trop. Dis. 2016, 10, e0004458. [Google Scholar] [CrossRef]
- Ballart, C.; Baron, S.; Alcover, M.M.; Portus, M.; Gallego, M. Distribution of phlebotomine sand flies (Diptera: Psychodidae) in Andorra: First finding of P. perniciosus and wide distribution of P. ariasi. Acta Trop. 2012, 122, 155–159. [Google Scholar] [CrossRef]
- Maia, C.; Parreira, R.; Cristóvão, J.M.; Afonso, M.O.; Campino, L. Exploring the utility of phylogenetic analysis of cytochrome oxidase gene subunit I as a complementary tool to classical taxonomical identification of phlebotomine sand fly species (Diptera, Psychodidae) from southern Europe. Acta. Trop. 2015, 144, 1–8. [Google Scholar] [CrossRef]
- Pereira, S.; Pita-Pereira, D.; Araujo-Pereira, T.; Britto, C.; Costa-Rego, T.; Ferrolho, J.; Vilhena, M.; Rangel, E.F.; Vilela, M.L.; Afonso, M.O. First molecular detection of Leishmania infantum in Sergentomyia minuta (Diptera, Psychodidae) in Alentejo, southern Portugal. Acta Trop. 2017, 174, 45–48. [Google Scholar] [CrossRef]
- Pires, C. Os Flebótomos (Diptera, Psychodidae) dos Focos Zoonóticos de Leishmanioses em Portugal. Ph.D. Thesis, Universidade Nova de Lisboa, Instituto de Higiene e Medicina Tropical, Lisboa, Portugal, 2000. (In Portuguese). [Google Scholar]
- Alves-Pires, C.; Ribeiro, H. The phlebotomine sandflies of Portugal. V—Observations of the ecology of the vectors of leishmaniasis in the Alto-Douro region. Parasitologia 1991, 33, 63–68. [Google Scholar]
- Afonso, M.O.; Campino, L.; Cortes, S.; Alves-Pires, C. The phlebotomine sandflies of Portugal. XIII--Occurrence of Phlebotomus sergenti Parrot, 1917 in the Arrabida leishmaniasis focus. Parasite 2005, 12, 69–72. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Osório, H.C.; Zé-Zé, L.; Amaro, F.; Alves, M.J. Mosquito surveillance for prevention and control of emerging mosquito-borne diseases in Portugal—2008–2014. Int. J. Environ. Res. Public Health 2014, 11, 11583–11596. [Google Scholar] [CrossRef] [PubMed]
- Topographic-map.com. Available online: https://pt-br.topographic-map.com/maps/zhkm/ (accessed on 29 December 2021).
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial Cytochrome C oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar]
- Osório, H.C.; Zé-Zé, L.; Neto, M.; Silva, S.; Marques, F.; Silva, A.S.; Alves, M.J. Detection of the invasive mosquito species Aedes (Stegomya) albopictus (Diptera: Culicidae) in Portugal. Int. J. Environ. Res. Public Health 2018, 15, E820. [Google Scholar] [CrossRef]
- Molaei, G.; Andreadis, T.G. Identification of avian- and mammalian-derived bloodmeals in Aedes vexans and Culiseta melanura (Diptera: Culicidae) and its implication for West Nile virus transmission in Connecticut, U.S.A. J. Med. Entomol. 2006, 43, 1088–1093. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl. Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Yu, G. Using ggtree to Visualize Data on Tree-Like Structures. Curr. Protoc. Bioinform. 2020, 69, e96. [Google Scholar] [CrossRef]
- Librado, P.; Rozas, J. DnaSP v5: A software for comprehensive analysis of DNA polymorphism data. Bioinformatics 2009, 25, 1451–1452. [Google Scholar] [CrossRef] [PubMed]
- Land Cover Classification Gridded Maps from 1992 to Present Derived from Satellite Observations. Available online: https://cds.climate.copernicus.eu/cdsapp#!/dataset/satellite-land-cover?tab=overview (accessed on 15 December 2021).
- Essential Climate Variables for Assessment of Climate Variability from 1979 to Present. Available online: https://cds.climate.copernicus.eu/cdsapp#!/dataset/ecv-for-climate-change (accessed on 14 December 2021).
- Lourenço, J.; Barros, S.C.; Zé-Zé, L.; Damineli, D.C.S.; Giovanetti, M.; Osório, H.C.; Amaro, F.; Henriques, A.M.; Ramos, F.; Luís, T.; et al. West Nile virus transmission potential in Portugal. Comm. Biol. 2022, 5, 6. [Google Scholar] [CrossRef] [PubMed]
- Depaquit, J.; Hadj-Henni, L.; Bounamous, A.; Strutz, S.; Boussaa, S.; Morillas-Marquez, F.; Pesson, B.; Gállego, M.; Delécolle, J.C.; Afonso, M.O.; et al. Mitochondrial DNA Intraspecific Variability in Sergentomyia minuta (Diptera: Psychodidae). J. Med. Entomol. 2015, 52, 819–828. [Google Scholar] [CrossRef] [PubMed]
- Ballart, C.; Guerrero, I.; Castells, X.; Barón, S.; Castillejo, S.; Alcover, M.M.; Portús, M.; Gállego, M. Importance of individual analysis of environmental and climatic factors affecting the density of Leishmania vectors living in the same geographical area: The example of Phlebotomus ariasi and P. perniciosus in northeast Spain. Geospat. Health 2014, 8, 389–403. [Google Scholar] [CrossRef] [PubMed]
- McCoy, K.D. The population genetic structure of vectors and our understanding of disease epidemiology. Parasite 2008, 15, 444–448. [Google Scholar] [CrossRef] [PubMed]
- De Colmenares, M.; Portús, M.; Botet, J.; Dobaño, C.; Gállego, M.; Wolff, M.; Seguí, G. Identification of blood meals of Phlebotomus perniciosus (Diptera: Psychodidae) in Spain by a competitive enzyme-linked immunosorbent assay biotin/avidin method. J. Med. Entomol. 1995, 32, 229–233. [Google Scholar] [CrossRef]
- Scarpassa, V.M.; Figueiredo, A.S.; Alencar, R.B. Genetic diversity and population structure in the Leishmania guyanensis vector Lutzomyia anduzei (Diptera, Psychodidae) from the Brazilian Amazon. Infect. Genet. Evol. 2015, 31, 312–320. [Google Scholar] [CrossRef]
- Killick-Kendrick, R.; Killick-Kendrick, M. Biology of sand fly vectors of Mediterranean canine leishmaniasis. In Canine Leishmaniasis: An Update. Proceedings of the International Canine Leishmaniasis Forum, Barcelona, Spain, 20–24 May 1999; Hoechst Roussel Vet: Wiesbaden, Germany, 1999; pp. 26–31. [Google Scholar]
- Benkova, I.; Volf, P. Effect of temperature on metabolism of Phlebotomus papatasi (Diptera: Psychodidae). J. Med. Entomol. 2007, 44, 150–154. [Google Scholar] [CrossRef]
- Trájer, A.J.; Bede-Fazekas, A.; Hufnagel, l.; Horvath, L.; Bovbos, J. The effect of climate change on the potential distribution of the European Phlebotomus species. Appl. Ecol. Env. Res. 2013, 11, 189–208. [Google Scholar] [CrossRef]
- Risueño, J.; Muñoz, C.; Pérez-Cutillas, P.; Goyena, E.; Gonzálvez, M.; Ortuño, M.; Bernal, L.J.; Ortiz, J.; Alten, B.; Berriatua, E. Understanding Phlebotomus perniciosus abundance in south-east Spain: Assessing the role of environmental and anthropic factors. Parasit. Vectors 2017, 10, 189. [Google Scholar] [CrossRef]
- Obolski, U.; Perez, P.N.; Villabona-Arenas, C.J.; Thézé, J.; Faria, N.R.; Lourenço, J. MVSE: An R-package that estimates a climate-driven mosquito-borne viral suitability index. Methods Ecol. Evol. 2019, 10, 1357–1370. [Google Scholar] [CrossRef] [PubMed]
- Bellone, R.; Failloux, A.B. The Role of Temperature in Shaping Mosquito-Borne Viruses Transmission. Front. Microbiol. 2020, 11, 584846. [Google Scholar] [CrossRef] [PubMed]
- Chamaillé, L.; Tran, A.; Meunier, A.; Bourdoiseau, G.; Ready, P.; Dedet, J.P. Environmental risk mapping of canine leishmaniasis in France. Parasit. Vectors 2010, 3, 31. [Google Scholar] [CrossRef] [PubMed]
- Merino-Espinosa, G.; Corpas-Lopez, V.; Callejon-Fernandez, R.; Porcel-Rodriguez, L.; Diaz-Saez, V.; Gallego, M.; Ballart, C.; Molina, R.; Jiménez, M.; Morillas-Márquez, F.; et al. Differential ecological traits of two Phlebotomus sergenti mitochondrial lineages in southwestern Europe and their epidemiological implications. Trop. Med. Int. Health 2016, 21, 630–641. [Google Scholar] [CrossRef] [PubMed]
- Casimiro, E.; Calheiros, J.; Santos, F.D.; Kovats, S. National assessment of human health effects of climate change in Portugal: Approach and key findings. Environ. Health Perspect. 2006, 114, 1950–1956. [Google Scholar] [CrossRef] [PubMed]
- Colacicco-Mayhugh, M.G.; Masuoka, P.M.; Grieco, J.P. Ecological niche model of Phlebotomus alexandri and P. papatasi (Diptera: Psychodidae) in the Middle East. Int. J. Health Geogr. 2010, 9, 2. [Google Scholar] [CrossRef]
- Trájer, A.J.; Sebestyén, V. The changing distribution of Leishmania infantum Nicolle, 1908 and its Mediterranean sandfly vectors in the last 140 kys. Sci. Rep. 2019, 9, 11820. [Google Scholar] [CrossRef]
- Killick-Kendrick, R.; Rioux, J.A.; Bailly, M.; Guy, M.W.; Wilkes, T.J.; Guy, F.M.; Davidson, I.; Knechtli, R.; Ward, R.D.; Guilvard, E.; et al. Ecology of leishmaniasis in the south of France. 20. Dispersal of Phlebotomus ariasi Tonnoir, 1921 as a factor in the spread of visceral leishmaniasis in the Cévennes. Ann. Parasitol. Hum. Comp. 1984, 59, 555–572. [Google Scholar] [CrossRef]
- Charrel, R.N.; Izri, A.; Temmam, S.; de Lamballerie, X.; Parola, P. Toscana virus RNA in Sergentomyia minuta flies. Emer. Infect. Dis. 2006, 12, 1299–1300. [Google Scholar] [CrossRef]
- González, E.; Molina, R.; Aldea, I.; Iriso, A.; Tello, A.; Jiménez, M. Leishmania sp. detection and blood-feeding behaviour of Sergentomyia minuta collected in the human leishmaniasis focus of southwestern Madrid, Spain (2012–2017). Transbound. Emerg. Dis. 2020, 67, 1393–1400. [Google Scholar] [CrossRef]
- Aransay, A.; Ready, P.; Morillas-Marquez, F. Population differentiation of Phlebotomus perniciosus in Spain following postglacial dispersal. Heredity 2003, 90, 316–325. [Google Scholar] [CrossRef] [PubMed]
- Perrotey, S.; Mahamdallie, S.S.; Pesson, B.; Richardson, K.J.; Gállego, M.; Ready, P. Postglacial dispersal of Phlebotomus perniciosus into France. Parasite 2006, 12, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Arrivillaga, J.C.; Norris, D.E.; Feliciangeli, M.D.; Lanzaro, G.C. Phylogeography of the neotropical sand fly Lutzomyia longipalpis inferred from mitochondrial DNA sequences. Infect. Genet. Evol. 2002, 2, 83–95. [Google Scholar] [CrossRef]
- Scarpassa, V.M.; Alencar, R.B. Lutzomyia umbratilis, the Main Vector of Leishmania guyanensis, Represents a Novel Species Complex? PLoS ONE 2012, 7, e37341. [Google Scholar] [CrossRef]
- Pech-May, A.; Marina, C.F.; Vázquez-Domínguez, E.; Berzunza-Cruz, M.; Rebollar-Téllez, E.A.; Narváez-Zapata, J.A.; Moo-Llanes, D.; Ibáñez-Bernal, S.; Ramsey, J.M.; Becker, I. Genetic structure and divergence in populations of Lutzomyia cruciata, a phlebotomine sand fly (Diptera: Psychodidae) vector of Leishmania mexicana in southeastern Mexico. Infect. Genet. Evol. 2013, 16, 254–262. [Google Scholar] [CrossRef]
- Yamamoto, K.; Cáceres, A.G.; Gomez, E.A.; Mimori, T.; Iwata, H.; Korenaga, M.; Sakurai, T.; Katakura, K.; Hashiguchi, Y.; Kato, H. Genetic diversity of the mitochondrial cytochrome b gene in Lutzomyia spp., with special reference to Lutzomyia peruensis, a main vector of Leishmania (Viannia) peruviana in the Peruvian Andes. Acta Trop. 2013, 126, 156–163. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zé-Zé, L.; Amaro, F.; Osório, H.C.; Giovanetti, M.; Lourenço, J.; Alves, M.J. Molecular Identification and Ecology of Portuguese Wild-Caught Phlebotomine Sandfly Specimens. Zoonotic Dis. 2022, 2, 19-31. https://doi.org/10.3390/zoonoticdis2010003
Zé-Zé L, Amaro F, Osório HC, Giovanetti M, Lourenço J, Alves MJ. Molecular Identification and Ecology of Portuguese Wild-Caught Phlebotomine Sandfly Specimens. Zoonotic Diseases. 2022; 2(1):19-31. https://doi.org/10.3390/zoonoticdis2010003
Chicago/Turabian StyleZé-Zé, Líbia, Fátima Amaro, Hugo Costa Osório, Marta Giovanetti, José Lourenço, and Maria João Alves. 2022. "Molecular Identification and Ecology of Portuguese Wild-Caught Phlebotomine Sandfly Specimens" Zoonotic Diseases 2, no. 1: 19-31. https://doi.org/10.3390/zoonoticdis2010003
APA StyleZé-Zé, L., Amaro, F., Osório, H. C., Giovanetti, M., Lourenço, J., & Alves, M. J. (2022). Molecular Identification and Ecology of Portuguese Wild-Caught Phlebotomine Sandfly Specimens. Zoonotic Diseases, 2(1), 19-31. https://doi.org/10.3390/zoonoticdis2010003








