Soybean Seed Coat Cracks and Green Seeds—Predisposing Conditions, Identification and Management
Abstract
1. Introduction
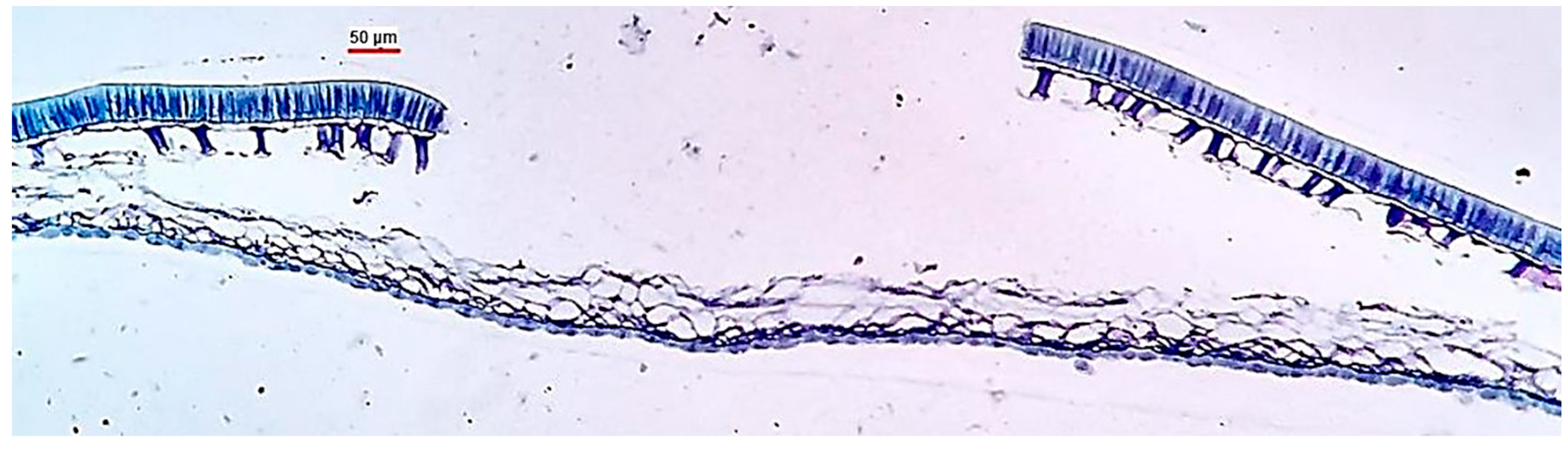
2. Seed Tegument

3. Seed Coat Cracking
4. Green Seeds
5. Mapping of QTLs for Seed Coat Cracking and Color Soybean Seeds
6. Crop Desiccation as a Crop Management Tool
7. Perspectives and Future Research
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yaklich, R.W.; Barla-Szabo, G. Seed Coat Cracking in Soybean. Crop. Sci. 1993, 33, 1016–1019. [Google Scholar] [CrossRef]
- França Neto, J.B.; Pádua, G.P.; Krzyzanowski, F.C.; Carvalho, M.L.M.; Henning, A.A.; Lorini, I. Semente Esverdeada de Soja: Causas e Efeitos Sobre o Desempenho Fisiológico—Série Sementes; Circular Técnica 91; Embrapa: Londrina, Brazil, 2012; 16p. [Google Scholar]
- Zorato, M.d.F.; Peske, S.T.; Takeda, C.; Neto, J.d.B.F. Presença de sementes esverdeadas em soja e seus efeitos sobre seu potencial fisiológico. Rev. Bras. Sement 2007, 29, 11–19. [Google Scholar] [CrossRef]
- Adams, C.A.; Fjerstad, M.C.; Rinne, R.W. Characteristics of Soybean Seed Maturation: Necessity for Slow Dehydration1. Crop. Sci. 1983, 23, 265–267. [Google Scholar] [CrossRef]
- Nedel, J.L. Fundamentos da Qualidade de Sementes. In Sementes Fundamentos Científicos e Tecnológicos; UFPel: Pelotas, Brazil, 2006; pp. 99–109. Available online: http://gsem.weebly.com/uploads/9/3/5/1/9351412/sementes_-_fundamentos_cient%C3%ADficos_e_tecnol%C3%B3gicos_-_silmar_peske_-_2%C2%AA_ed.pdf (accessed on 7 October 2023).
- DE Souza, F.H.D.; Marcos-Filho, J. The seed coat as a modulator of seed-environment relationships in Fabaceae. Braz. J. Bot. 2001, 24, 365–375. [Google Scholar] [CrossRef]
- Marcos-Filho, J. Fisiologia de sementes de plantas cultivadas; ABRATES: Londrina, Brazil, 2015; 659p. [Google Scholar]
- Moïse, J.A.; Han, S.; Gudynaitę-Savitch, L.; Johnson, D.A.; Miki, B.L.A. Seed coats: Structure, development, composition, and biotechnology. Vitr. Cell. Dev. Biol. Plant 2005, 41, 620–644. [Google Scholar] [CrossRef]
- Weber, H.; Borisjuk, L.; Wobus, U. Molecular physiology of legume seed development. Annu. Rev. Plant Biol. 2005, 56, 253–279. [Google Scholar] [CrossRef]
- Swanson, B.G.; Hughes, J.S.; Rasmussen, H. Seed Microstructure: Review of Water Imbibition in Legumes. Food Microestruture 1985, 4, 115–153. Available online: https://digitalcommons.usu.edu/cgi/viewcontent.cgi?article=1091&context=foodmicrostructure (accessed on 12 October 2023).
- Teixeira, S.B. Rasgo no Tegumento em Sementes de Soja Suas Causas e Consequências. Thesis Dissertation, Programa de Pós-Graduação em Ciência e Tecnologia de Sementes. Universidade Federal de Pelotas, Pelotas, Brazil, 2021; 103p. Available online: http://guaiaca.ufpel.edu.br/handle/prefix/9079 (accessed on 12 October 2023).
- Caviness, C.E.; Simpson, A.M., Jr. Influence of variety and location on seed coat thickness of mature soybean seed. Proc. Assoc. Off. Seed Anal. 1974, 64, 102–108. [Google Scholar]
- Lowry, J.B.; Conlan, L.L.; Schlink, A.C.; McSweeney, C.S. Acid detergent dispersible lignin in tropical grasses. J. Sci. Food Agric. 1994, 65, 41–49. [Google Scholar] [CrossRef]
- Krzyzanowski, F.C.; França-Neto, J.d.B.; Henning, F.A. Importance of the lignin content in the pod wall and seed coat on soybean seed physiological and health performances. J. Seed Sci. 2023, 45, e202345006. [Google Scholar] [CrossRef]
- Ramsay, G. Inheritance and linkage of a gene for testa-imposed seed dormancy in faba bean (Vicia faba L.). Plant Breed. 1997, 116, 287–289. [Google Scholar] [CrossRef]
- Liu, H.-L. Inheritance of defective seed coat in soybeans. J. Hered. 1949, 40, 317–322. [Google Scholar] [CrossRef]
- Wolf, W.J.; Baker, F.L.; Bernard, R.L. Soybean Seed Coat Structural Features: Pits, Deposits and Cracks. Scanning Electron Microscopy III: 531–544. 1981. Available online: https://naldc.nal.usda.gov/download/26138/PDF (accessed on 10 October 2023).
- Barrionuevo, F. Influência da Ruptura do Tegumento na Qualidade de Sementes de Soja. Master’s Thesis. Universidade Tecnológica Federal do Paraná. 72p. Available online: http://repositorio.utfpr.edu.br/jspui/handle/1/31111 (accessed on 12 October 2023).
- Zorato, M.F. Laboratório de Análise de Sementes—Engrenagem Fundamental na Produção de Sementes Contemporânea. Seed News, XXI(2): Mar/apr. 2017. Available online: https://seednews.com.br/artigos/41-laboratorio-de-analise-de-sementes-edicao-marco-2017 (accessed on 29 September 2023).
- Teixeira, S.B.; Xavier, F.M.; Gonçalves, D.W.M.; Zorato, M.F.; Meneghello, G.E. Rasgo no Tegumento de Soja: Implicações na Qualidade de Sementes. XXI Encontro de Pós-Graduação, 5ª Semana Integrada, UFPEL. 2019. Available online: http://cti.ufpel.edu.br/siepe/arquivos/2019/CA_02163.pdf (accessed on 19 October 2023).
- Fehr, W.R.; Caviness, C.E.; Burmood, D.T.; Pennington, J.S. Stage of Development Descriptions for Soybeans, Glycine max (L.) Merrill1. Crop. Sci. 1971, 11, 929–931. [Google Scholar] [CrossRef]
- Agrawal, V.K.; Menon, S.K. Lignin content and seed coat thickness in relation to seed coat cracking in soybean. Seed Res. 1974, 2, 64–66. [Google Scholar]
- Bahry, C.A.; Acunha, T.D.S.; Fernando, J.A.; Chaves, F.C.; Nardino, M.; Zimmer, P.D. Chemical composition and structural characterization of contrasting colors of soybean seed coats. Semin. Ciênc. Agrár. 2015, 36, 1913. [Google Scholar] [CrossRef]
- Senda, M.; Kawasaki, M.; Hiraoka, M.; Yamashita, K.; Maeda, H.; Yamaguchi, N. Occurrence and tolerance mechanisms of seed cracking under low temperatures in soybean (Glycine max). Planta 2018, 248, 369–379. [Google Scholar] [CrossRef]
- Qutob, D.; Ma, F.; Peterson, C.A.; Bernards, M.A.; Gijzen, M. Structural and permeability properties of the soybean seed coat. Botany 2008, 86, 219–227. [Google Scholar] [CrossRef]
- Vieira, B.G.T.L.; Barbosa, G.F.; Barbosa, R.M.; Vieira, R.D. Structural Changes in Soybean Seed Coat Due to Harvest Time and Storage. J. Food Agric. Environ. 2013, 11, 625–628. Available online: http://acervodigital.unesp.br/handle/11449/74810 (accessed on 2 April 2023).
- Naflath, T.V.; Rajendraprasad, S.; Ravikumar, R.L. Evaluation of diverse soybean genotypes for seed longevity and its association with seed coat colour. Sci. Rep. 2023, 13, 4313. [Google Scholar] [CrossRef]
- Rao, P.J.M.; Pallavi, M.; Bharathi, Y.; Priya, P.B.; Sujatha, P.; Prabhavathi, K. Insights into mechanisms of seed longevity in soybean: A review. Front. Plant Sci. 2023, 14, 1206318. [Google Scholar] [CrossRef]
- Capeleti, I.; Bonini, E.A.; Ferrarese, M.d.L.L.; Teixeira, A.C.N.; Krzyzanowski, F.C.; Ferrarese-Filho, O. Lignin content and peroxidase activity in soybean seed coat susceptible and resistant to mechanical damage. Acta Physiol. Plant. 2005, 27, 103–108. [Google Scholar] [CrossRef]
- Menino, T.V.R.; Joia, B.M.; Almeida, A.M.; Krzyzanowski, F.C.; Marchiosi, R.; Ferrarese-Filho, O. Lignin monomeric composition in soybean seed coats and resistance to mechanical damage. J. Seed Sci. 2023, 45, e202345035. [Google Scholar] [CrossRef]
- Ma, F.; Cholewa, E.; Mohamed, T.; Peterson, C.A.; Gijzen, M. Cracks in the Palisade Cuticle of Soybean Seed Coats Correlate with their Permeability to Water. Ann. Bot. 2004, 94, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Lazarevic, J.; Zoric, L.; Karagic, D.; Milosevic, B.; Karanovic, D.; Milic, D.; Tepic, A.; Lukovic, J. Anatomical and micromorphological characteristics of the seed coat of field pea (Pisum sativum L.) genotypes in relation to cracks and damage of seeds. Arch. Biol. Sci. 2017, 69, 503–512. [Google Scholar] [CrossRef]
- Khan, F.A.; Bhat, S.A.; Narayan, S.; Maqbool, R.; Imtiyaz, M.; Khan, F.U. Seed Deterioration and Priming—An Overview. SKUAST J. Res. 2017, 19, 12–21. Available online: https://indianjournals.com/ijor.aspx?target=ijor%3Askuastjr&volume=19&issue=1&article=002&utm_source=TrendMD&utm_medium=cpc&utm_campaign=SKUAST_Journal_of_Research_TrendMD_0 (accessed on 6 April 2023).
- Mazibuko, T.G. Alleviation of Cotyledonal Cracking in Green Beans (Phaseolus vulgaris L.) by Calcium Seed Treatment. Master’s Thesis. University of Natal. 122p. 2003. Available online: http://hdl.handle.net/10413/11718 (accessed on 19 April 2023).
- Chen, J.; Shao, F.; Igbokwe, C.J.; Duan, Y.; Cai, M.; Ma, H.; Zhang, H. Ultrasound treatments improve germinability of soybean seeds: The key role of working frequency. Ultrason. Sonochem. 2023, 96, 106434. [Google Scholar] [CrossRef] [PubMed]
- Shelar, V.R.; Shaikh, R.S.; Nikam, A.S. Soybean Seed Quality during Storage a Review. Agric. Rev. 2008, 29, 125–131. Available online: https://arccarticles.s3.amazonaws.com/webArticle/articles/ar292006.pdf (accessed on 19 April 2023).
- Machado, B.R.; Pereira, C.C.D.O.; Carvalho, P.B.; Da Silva, J.O.; Mendes, L.M.D.O.; Lima, M.L.D.P.; Silva, J.M.; De Menezes, I.P.P. Effect of tear/crack on soybean (Glycine max) seed coat, physiological quality and pathology of the seed. Aust. J. Crop Sci. 2019, 13, 988–994. [Google Scholar] [CrossRef]
- de Pádua, G.P.; De Carvalho, M.L.M.; França-Neto, J.D.B.; Guerreiro, M.C.; Guimarães, R.M. Response of soybean genotypes to the expression of green seed under temperature and water stresses. Rev. Bras. Sement 2009, 31, 140–149. [Google Scholar] [CrossRef]
- Mandarino, J.M.G. Grãos Verdes: Influência na Qualidade dos Produtos à Base de Soja; EMBRAPA: Londrina, Brazil, 2012; Available online: https://ainfo.cnptia.embrapa.br/digital/bitstream/item/53614/1/CT90-OL1.pdf (accessed on 2 April 2023).
- Palmer, R.G.; Kilen, T.C. Qualitative genetics and cytogenetics. In Soybeans: Improvement, Production, and Uses; Wilcox, J.R., Ed.; American Society of Agronomy: Madison, WI, USA, 1987; pp. 135–209. [Google Scholar]
- Pelissari, F.; Coimbra, R.d.A. Sementes de soja esverdeadas: Causas e consequências na qualidade fisiológica. Sci. Electron. Arch. 2023, 16, 4. [Google Scholar] [CrossRef]
- Ajala-Luccas, D.; Ribeiro-Oliveira, J.P.; Teixeira, R.N.; Ducatti, K.R.; França-Neto, J.B.; Hilhorst, H.W.M.; da Silva, E.A.A. The Seed–Seedling Transition in Commercial Soybean Cultivars with the Presence of Greenish Seeds in the Sample: A Perspective from Classical Genetic Parameters. Agronomy 2023, 13, 1966. [Google Scholar] [CrossRef]
- Harbach, C.J.; Allen, T.W.; Bowen, C.R.; Davis, J.A.; Hill, C.B.; Leitman, M.; Leonard, B.R.; Mueller, D.S.; Padgett, G.B.; Phillips, X.A.; et al. Delayed Senescence in Soybean: Terminology, Research Update, and Survey Results from Growers. Plant Health Prog. 2016, 17, 76–83. [Google Scholar] [CrossRef]
- Heaton, J.W.; Marangoni, A.G. Chlorophyll degradation in processed foods and senescent plant tissues. Trends Food Sci. Technol. 1996, 7, 8–15. [Google Scholar] [CrossRef]
- Mangos, T.J.; Berger, R.G. Determination of major chlorophyll degradation products. Z. Leb. Forsch. A 1997, 204, 345–350. [Google Scholar] [CrossRef]
- Suzuki, Y.; Tanabe, K.; Shioi, Y. Determination of chemical oxidation products of chlorophyll and porphyrin by high-performance liquid chromatography. J. Chromatogr. A 1999, 839, 85–91. [Google Scholar] [CrossRef]
- Bobbio, F.O.; Bobbio, P.A. Introdução à Química de Alimentos, 2nd ed.; São Paulo: Varella, Brazil, 1989; 223p. [Google Scholar]
- Bobbio, F.O.; Bobbio, P.A. Química do Processamento de Alimentos, 2nd ed.; São Paulo: Varella, Brazil, 1992; 151p. [Google Scholar]
- Shimokawa, k.; Shimada, s.; Yaeo, K. Ethyleneenhanced chlorophylate activity during degreening of Citrus unshiu Marc. Sci. Hortic. 1978, 8, 129–135. [Google Scholar] [CrossRef]
- Yamauchi, N.; Akiyama, Y.; Kako, S.; Hashinaga, F. Chlorophyll degradation in Wase satsuma mandarin (Citrus unshiu Marc.) fruit with on-tree maturation and ethylene treatment. Sci. Hortic. 1997, 71, 35–42. [Google Scholar] [CrossRef]
- Stewart, I.; Wheaton, T.A. Carotenoids in citrus. Their accumulation induced by ethylene. J. Agric. Food Chem. 1972, 20, 448–449. [Google Scholar] [CrossRef]
- Takamiya, K.-I.; Tsuchiya, T.; Ohta, H. Degradation pathway(s) of chlorophyll: What has gene cloning revealed? Trends Plant Sci. 2000, 5, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, R.N.; Ligterink, W.; França-Neto, J.d.B.; Hilhorst, H.W.; da Silva, E.A.A. Gene expression profiling of the green seed problem in Soybean. BMC Plant Biol. 2016, 16, 37. [Google Scholar] [CrossRef] [PubMed]
- Kohzuma, K.; Sato, Y.; Ito, H.; Okuzaki, A.; Watanabe, M.; Kobayashi, H.; Nakano, M.; Yamatani, H.; Masuda, Y.; Nagashima, Y.; et al. The Non-Mendelian Green Cotyledon Gene in Soybean Encodes a Small Subunit of Photosystem II. Plant Physiol. 2017, 173, 2138–2147. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, S.B.; da Silva, J.G.; Meneguzzo, M.R.R.; Martins, A.B.N.; Meneghello, G.E.; de Tunes, L.V.M. Green soybean seeds: Effect on physiological quality. Ciência Rural. 2020, 50, e20180631. [Google Scholar] [CrossRef]
- Tonello, C.Z.; Machado, B.O.; Maurer, T.R.; Lângaro, N.C. Physiological performance of green soybean seeds: A systematic review. Conjecturas 2022, 22, 749–758. [Google Scholar] [CrossRef]
- de Pádua, G.P.; França-Neto, J.D.B.; de Carvalho, M.L.M.; Costa, O.; Krzyzanowski, F.C.; Da Costa, N.P. Tolerance level of green seed in soybean seed lots after storage. Rev. Bras. Sement 2007, 29, 128–138. [Google Scholar] [CrossRef]
- Daltro, E.M.F.; Albuquerque, M.C.d.F.e.; Neto, J.d.B.F.; Guimarães, S.C.; Gazziero, D.L.P.; Henning, A.A. Aplicação de dessecantes em pré-colheita: Efeito na qualidade fisiológica de sementes de soja. Rev. Bras. Sement 2010, 32, 111–122. [Google Scholar] [CrossRef]
- Sant’Anna, J.C., Jr. Efeito da Dessecação na Qualidade de Sementes de Cultivares de Soja, na Região dos Cerrados. Master’s Thesis. Universidade de Brasília. 2006. 67p. Available online: http://icts.unb.br/jspui/bitstream/10482/13232/1/2006_JersondeCastroSantAnnaJunior.pdf (accessed on 7 April 2023).
- Bezerra, A.R.G. Efeitos da Dessecação Pré-Colheita Nas Características Agronômicas e Qualidade de Sementes de Soja de Tipos de Crescimento Determinado e Indeterminado. Master’s Thesis. Universidade Federal de Viçosa. 2013. 59p. Available online: https://www.locus.ufv.br/bitstream/123456789/4630/1/texto%20completo.pdf (accessed on 2 October 2023).
- Lamego, F.; Gallon, M.; Basso, C.; Kulczynski, S.; Ruchel, Q.; Kaspary, T.; Santi, A. Dessecação pré-colheita e efeitos sobre a produtividade e qualidade fisiológica de sementes de soja. Planta Daninha 2013, 31, 929–938. [Google Scholar] [CrossRef]
- Finoto, E.L.; Sediyama, T.; Albuquerque, J.d.A.A.d.; Soares, M.B.B.; Galli, J.A.; Junior, P.S.C.; de Menezes, P.H.S. Anticipation and harvest delay in oil and protein contents of soybean seeds, grow crops Valiosa RR. Sci. Agropecu. 2017, 8, 99–107. [Google Scholar] [CrossRef][Green Version]
- Santos, L.T.S.; Holtz, V.; Massola, M.P.; Santana, G.C.; Schiehl, M.T.; Nunes, F.C. Aplicação de Paraquat na Pré-Colheita da Soja em Diferentes Estádios Reprodutivos. In Anais of Proceedings of the XXX Congresso Brasileiro de Agronomia Fortaleza; CBA: São Paulo, Brazil, 2017; p. 5. [Google Scholar]
- Toledo, M.Z.; Ishizuka, M.S.; Cavariani, C.; França-Neto, J.D.B.; Picoli, L.B. Pre-harvest desiccation with glyphosate and quality of stored soybean seeds. Semin. Ciênc. Agrár. 2014, 35, 765–773. [Google Scholar] [CrossRef]
- Souza, F.L.G. Dessecação Com Glyphosate em Pré-Colheita e Qualidade Fisiológica de Sementes de Soja. Master’s Thesis. Universidade Estadual Paulista “Júlio de Mesquita Filho”. 2009. 49p. Available online: http://acervodigital.unesp.br/handle/11449/86350 (accessed on 7 April 2023).
- Albrecht, L.; Albrecht, A.; Braccini, A.; Oliveira, R.S., Jr.; Zobiole, L.; Ávila, M. The role of glyphosate in RR soybean production and seed quality. Planta Daninha 2014, 32, 401–407. [Google Scholar] [CrossRef]
- Albrecht, L.P.; Barbosa, A.P.; Silva, A.F.M.; Mendes, M.A.; Albrecht, A.J.P.; Ávila, M.R. RR soybean seed quality after application of glyphosate in different stages of crop development. Rev. Bras. Sement 2012, 34, 373–381. [Google Scholar] [CrossRef][Green Version]
- Castro, D.G.; Bruzi, A.T.; Zambiazzi, E.V.; de Rezende, P.M.; Zuffo, A.M.; de Sales, A.P.; Soares, I.O.; Borges, I.M.M.; Bianchi, M.C. Qualidade fisiológica e expressão enzimática de sementes de soja RR. Rev. Ciênc. Agrár. 2017, 40, 222–235. [Google Scholar] [CrossRef]
- Cakmak, I.; Yazici, A.; Tutus, Y.; Ozturk, L. Glyphosate reduced seed and leaf concentrations of calcium, manganese, magnesium, and iron in non-glyphosate resistant soybean. Eur. J. Agron. 2009, 31, 114–119. [Google Scholar] [CrossRef]
- Albrecht, L.P.; Albrecht, A.J.P.; Yokoyama, A.S.; Silva, A.F.M.; Bottcher, A.A.; de Carvalho, M.G.; Battiston, F.M. Formulações de glufosinate na dessecação pré-colheita da soja. Nativa 2023, 11, 96–100. [Google Scholar] [CrossRef]
- Azevedo, M.; Pagnoncelli, C.A.; Coltro-Roncato, S.; Matte, S.C.S.; Gonçalves, E.D.V.; Dildey, O.D.F.; Heling, A.L. Aplicação de diferentes herbicidas para dessecação em pré-colheita de soja. Agrarian 2015, 8, 246–252. Available online: https://ojs.ufgd.edu.br/index.php/agrarian/article/view/3325 (accessed on 2 April 2023).
- Botelho, F.J.E.; Oliveira, J.A.; Pinho, V.d.R.V.; Carvalho, E.R.; Figueiredo, B.D.; Andrade, V. Qualidade de sementes de soja obtidas de diferentes cultivares submetidas à dessecação com diferentes herbicidas e épocas de aplicação. Rev. Agro@mbiente On-Line 2016, 10, 137–144. [Google Scholar] [CrossRef][Green Version]
- Delgado, C.M.L.; de Coelho, C.M.M.; Buba, G.P. Mobilization of reserves and vigor of soybean seeds under desiccation with glufosinate ammonium. J. Seed Sci. 2015, 37, 154–161. [Google Scholar] [CrossRef]
- Pereira, T.; Coelho, C.M.M.; Sobiecki, M.; Souza, C.A. Physiological quality of soybean seeds depending on the preharvest desiccation. Planta Daninha 2015, 33, 441–450. [Google Scholar] [CrossRef]
- Silva, G.F.; Chamma, L.; Luperini, B.C.O.; Chaves, P.P.N.; Calonego, J.C.; Nakagawa, J.; da Silva, E.A.A. Physiological Quality of Soybean Seeds as a Function of Soil Management Systems and Pre-Harvest Desiccation. Agronomy 2023, 13, 847. [Google Scholar] [CrossRef]
- Castoldi, C.T. Época de Aplicação de Herbicidas Dessecantes Para Antecipação da Colheita de Sementes de Feijão Carioca. Master’s Thesis. Universidade Federal da Fronteira Sul. 2017. 56p. Available online: https://rd.uffs.edu.br/handle/prefix/3998 (accessed on 7 October 2023).
- Assis, M.O. Dessecação em Pré-Colheita na Produtividade e Qualidade Fisiológica de Sementes de Feijão-Caupi. Master’s Thesis. Universidade Federal de Viçosa. 2016. 44p. Available online: https://locus.ufv.br//handle/123456789/9280 (accessed on 7 October 2023).
- Coelho, C.M.M.; Souza, C.A.; Zilio, M.; Michels, A.F. Ação de dessecante na pré-colheita sobre a produtividade e a qualidade fisiológica de sementes crioulas de feijoeiro. Semin. Ciências Agrárias 2012, 33, 2973–2980. [Google Scholar] [CrossRef]
- Franco, M.H.R.; Nery, M.C.; França, A.C.; Oliveira, M.C.; Franco, G.N.; Lemos, V.T. Produção e qualidade fisiológica de semente de feijão após aplicação do herbicida Diquat. Semin. Ciências Agrárias 2013, 34, 1707–1714. [Google Scholar] [CrossRef]
- Penckowski, L.H.; Podolan, M.J.; Lopez-Ovejero, R.F. Efeito de herbicidas aplicados na pré-colheita na qualidade fisiológica das sementes de feijão. Rev. Bras. Herbic. 2005, 4, 102. [Google Scholar] [CrossRef]
- Cechinel, M.H. Dessecação Química em Pré-Colheita do Trigo. Master’s Thesis. Universidade do Estado de Santa Catarina. 2014. 107p. Available online: https://www.udesc.br/arquivos/cav/id_cpmenu/1346/Disserta__o_Mariana_Cechinel_15676030630189_1346.pdf (accessed on 6 October 2023).
- Lunkes, A. Maturação Antecipada de Trigo: Produtividade de Grãos e Qualidade das Sementes. Master’s Thesis. Universidade de Cruz Alta (UNICRUZ). 2014. 52p. Available online: https://home.unicruz.edu.br/wp-content/uploads/2017/01/Adilson-Lunkes-MATURACAO-ANTECIPADA-DE-TRIGO-PRODUTIVIDADE-E-QUALIDADE-DOS-GRAOS.pdf (accessed on 6 October 2023).
- Kehl, K. Dessecação Pré-Colheita na Produção de Sementes de Trigo. Universidade Federal de Pelotas. Master Thesis. 41 p. 2015. Available online: https://guaiaca.ufpel.edu.br/handle/prefix/3300 (accessed on 23 September 2023).
- Darwent, A.L.; Kirkland, K.J.; Townley-Smith, L.; Harker, K.N.; Cessna, A.J. Effect of preharvest applications of glyphosate on the drying, yield and quality of canola. Can. J. Plant Sci. 2000, 80, 433–439. [Google Scholar] [CrossRef]

| Seed Coat Cracking 1 | ||||
| GWAS QTL Name | Original Name | LOD | Chromosome | Linkage Group |
| Seed coat cracking 1-g1 | qSC2-3 | 12.9 | Gm02 | D1b |
| Seed coat cracking 1-g2 | qSC6 | 6.8 | Gm06 | C2 |
| Seed coat cracking 1-g3 | qSC10-2 | 10.7 | Gm10 | O |
| Seed coat cracking 1-g4 | qSC12 | 5.1 | Gm12 | H |
| Seed coat cracking 1-g5 | qSC19-2 | 4.1 | Gm19 | L |
| Seed coat cracking 1-g6 | qSC20 | 6.3 | Gm20 | I |
| Seed Coat Cracking 2 | ||||
| GWAS QTL name | Original name | LOD | Chromosome | Linkage Group |
| Seed coat cracking 2-g1 | qSCD15 | 5 | Gm15 | E |
| Seed coat cracking 2-g2 | qSCD20 | 6.1 | Gm20 | I |
| Seed Coat Color 1 | ||||
| GWAS QTL name | Original name | Associated locus | p-value | Chromosome |
| Seed coat color 1-g1.1 | Seed coat color | Chr01_3568116 | 1.12 × 10−7 | Gm01 |
| Seed coat color 1-g1.2 | Seed coat color | Chr01_3570619 | 1.12 × 10−7 | Gm01 |
| Seed coat color 1-g1.3 | Seed coat color | Chr01_3576803 | 1.12 × 10−7 | Gm01 |
| Seed coat color 1-g2 | Seed coat color | Chr01_53229579 | 4.17 × 10−12 | Gm01 |
| Seed coat color 1-g3.1 | Seed coat color | Chr08_7800853 | 2.63 × 10−35 | Gm08 |
| Seed coat color 1-g3.2 | Seed coat color | Chr08_8607374 | 2.63 × 10−35 | Gm08 |
| Seed coat color 1-g3.3 | Seed coat color | Chr08_9079037 | 2.63 × 10−35 | Gm08 |
| Seed Coat Color 3 | ||||
| GWAS QTL name | Original name | Associated locus | R2 | Chromosome |
| Seed coat color 3-g1 | Black | ss715602762 | 0.62 | Gm08 |
| Seed coat color 3-g2 | Green | ss715580343 | 0.31 | Gm01 |
| Seed coat color 3-g3 | Yellow | ss715620481 | 0.41 | Gm15 |
| Seed coat color 3-g4 | Yellow | ss715580343 | 0.58 | Gm01 |
| Seed coat color 3-g5 | Yellow | ss715602763 | 0.56 | Gm08 |
| Seed coat color 3-g6 | Yellow | ss715593807 | 0.55 | Gm06 |
| Seed Coat Color 4 | ||||
| GWAS QTL name | Original name | Associated locus | p-value | Chromosome |
| Seed coat color 4-g1 | Gm08:8236728 | Gm08:8241052 | 1.20 × 1017 | Gm08 |
| Seed coat color 4-g2 | Gm08:8439795 | Gm08:8422490 | 1.31 × 1015 | Gm08 |
| Seed coat color 4-g3 | Gm08:20745554 | Gm08:20702756 | 1.86 × 109 | Gm08 |
| Seed Coat Color 5 | ||||
| GWAS QTL name | Original name | LOD | Chromosome | Linkage Group |
| Seed coat color 5-g1 | qSC08 | 14.251 | Gm08 | D1b |
| Seed coat color 5-g2 | qSC11 | 8.112 | Gm11 | F |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lemes, E.M.; Catão, H.C.R.M. Soybean Seed Coat Cracks and Green Seeds—Predisposing Conditions, Identification and Management. Seeds 2024, 3, 133-148. https://doi.org/10.3390/seeds3010011
Lemes EM, Catão HCRM. Soybean Seed Coat Cracks and Green Seeds—Predisposing Conditions, Identification and Management. Seeds. 2024; 3(1):133-148. https://doi.org/10.3390/seeds3010011
Chicago/Turabian StyleLemes, Ernane Miranda, and Hugo César Rodrigues Moreira Catão. 2024. "Soybean Seed Coat Cracks and Green Seeds—Predisposing Conditions, Identification and Management" Seeds 3, no. 1: 133-148. https://doi.org/10.3390/seeds3010011
APA StyleLemes, E. M., & Catão, H. C. R. M. (2024). Soybean Seed Coat Cracks and Green Seeds—Predisposing Conditions, Identification and Management. Seeds, 3(1), 133-148. https://doi.org/10.3390/seeds3010011






