1. Introduction
Xerostomia, the subjective sensation of oral dryness, is a common adverse effect of chronic pharmacotherapy, particularly among institutionalized psychiatric patients receiving antipsychotics and anticholinergic agents [
1]. The reduction in salivary flow, known as hyposialia, has been shown to impair essential oral functions such as lubrication, antimicrobial defense and remineralization, which can increase the risk of caries, mucosal infections, and reduced quality of life [
2]. This population is especially vulnerable due to polypharmacy and limited access to preventive dental care [
1]. Topical sialogogues present a promising therapeutic alternative by virtue of their capacity to locally stimulate salivary flow with minimal systemic side effects. Nevertheless, the clinical evidence supporting their use in this specific context remains limited [
3,
4]. The objective of this study is to evaluate the effectiveness of a topical sialogogue gel in improving salivary flow and relieving symptoms of xerostomia (dry mouth) in long-term residents of the Lisbon Psychiatric Hospital Centre (C.H.P.L.). The findings of this study will contribute to the development strategies to improve oral health in individuals with complex medical conditions and high social vulnerability.
2. Materials and Methods
A cross-sectional, correlational study was conducted from March to June 2023 over a period of four months at the Lisbon Psychiatric Hospital Centre (C.H.P.L.), involving three closed psychiatric inpatient wards (18, 21B, and Forensic ward). The study was approved by the Scientific and Pedagogical Committee and the Ethics Committee of the C.H.P.L., as well as by the hospital’s Executive Board (approval number: 1197). All participants were informed of the study objectives and procedures and provided written informed consent, in accordance with the Declaration of Helsinki and national legislation.
A total of 81 institutionalized patients with chronic psychiatric conditions who met the inclusion criteria and voluntarily agreed to participate were enrolled. The inclusion criteria comprised the following: (1) age ≥ 18 years; (2) permanent inpatient status at C.H.P.L.; (3) both sexes; and (4) capacity to understand and sign informed consent. Salivary flow was assessed using sialometry at baseline (T0) and after 15 days (T1). A subsequent investigation involved the implementation of a questionnaire regarding sociodemographic variables and the Summated Xerostomia Inventory—Portuguese short version (SXI-PL). The unstimulated salivary flow rate (USFR) was measured via passive drooling; the stimulated salivary flow rate (SSFR) was collected by chewing a 1g sterile paraffin tablet. Buffer capacity and pH were evaluated using Saliva Check-buffer® kits. Hyposialia was defined as USFR < 0.1 mL/min and/or SSFR < 0.7 mL/min. Participants received topical application of Xeros Dentaid Gel® (betaine 1%, aloe vera 0.05%, xylitol 10%, sodium fluoride 0.0033%), administered by trained nurses two times a day, after oral hygiene and before main meals, for 15 consecutive days. The application covered teeth, gingivae, and oral floor without rinsing, ensuring optimal overnight hydration. Data were analyzed using IBM SPSS Statistics® v.28.
3. Results
A total of 81 institutionalized psychiatric patients participated in the study, with a mean age of 47.83 ± 15.39 years. Most participants were male (81.5%, n = 66), with only 18.5% (n = 15) identifying as female. The frequency distributions of psychiatric patients were as follows: schizophrenia and schizotypal and delusional disorders, 59.2% (48); epilepsy, 21% (17); and neurotic, stress-related, and somatoform disorders, 16 (19.8%). The majority of patients in the study were medicated with antipsychotics (98.8% (80)) and antiepileptics (55.6% (45)), followed by anxiolytics (72.8% (59)), antidepressants (43.2% (35)), and hypnotics/sedatives (28.4% (23)).
3.1. Salivary Flow Rates—Before and After Treatment
The unstimulated salivary flow rate (USFR) at baseline (T0) ranged from 0 to 3 mL/min, with a mean of 0.43 ± 0.47 mL/min. The stimulated salivary flow rate (SSFR) at T0 ranged from 0 to 3 mL/min, with a mean of 0.53 ± 0.58 mL/min. Fifteen days after topical sialogogue application (T1), USFR increased to 0.72 ± 0.54 mL/min and SSFR to 0.69 ± 0.68 mL/min. The mean increases of 0.28 mL/min in USFR and 0.15 mL/min in SSFR were statistically significant (p < 0.001), suggesting clear improvement in salivary function following treatment.
3.2. Prevalence of Xerostomia/Hyposialia
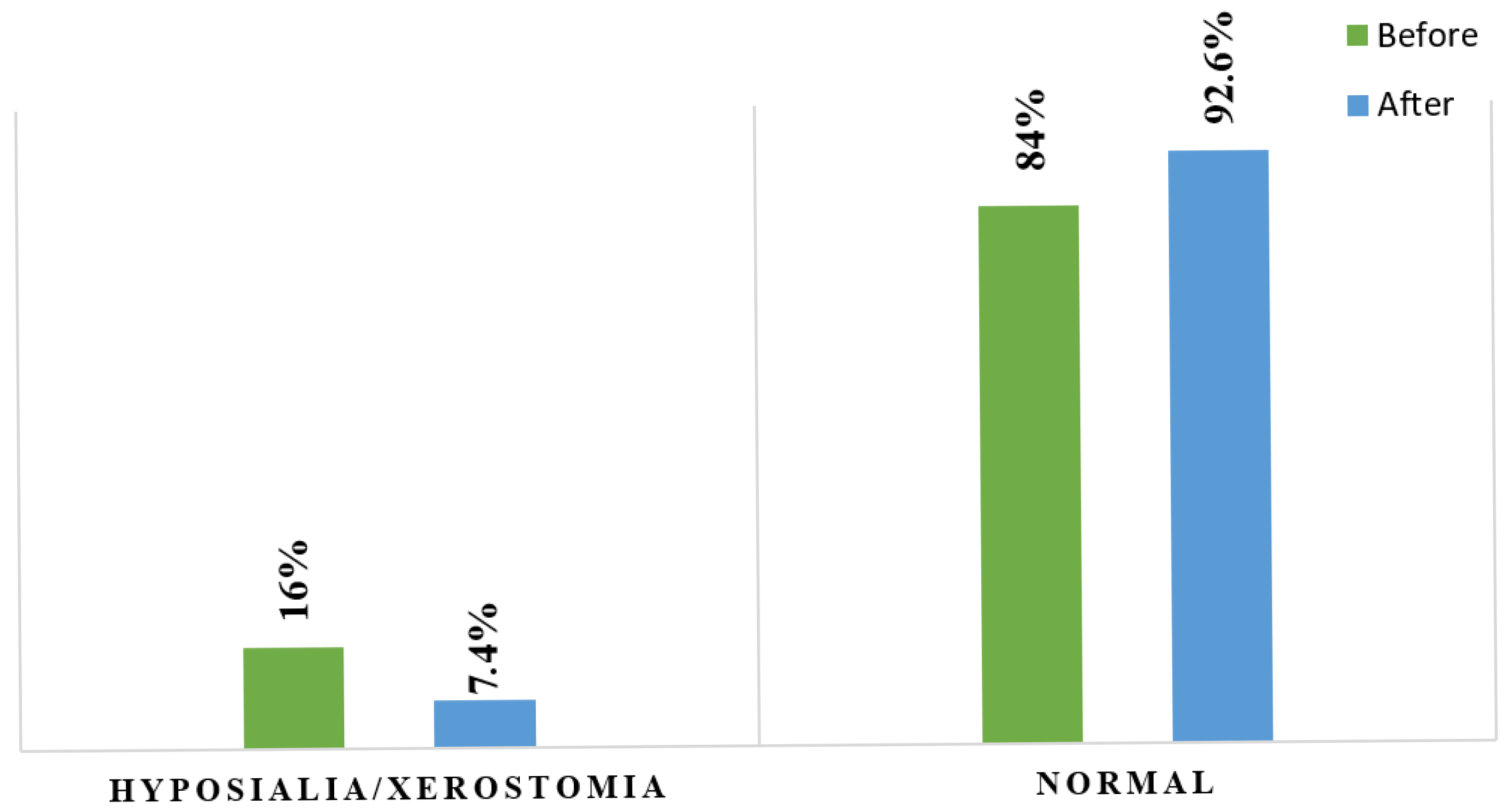
At baseline (T0), 13 patients (16%) were classified as having hyposialia (USFR < 0.1 mL/min and/or SSFR < 0.7 mL/min), while 68 (84%) presented normal salivary flow (
Table 1). After 15 days of treatment (T1), the prevalence of xerostomia dropped to 7.4% (
n = 6), while 92.6% (
n = 75) exhibited normal salivary flow (
Table 2). Fisher’s Exact Test confirmed a statistically significant difference in xerostomia prevalence before and after treatment (
p < 0.001), demonstrating the efficacy of the topical sialogogue in reducing drug-induced xerostomia among psychiatric inpatients (
Figure 1).
3.3. Salivary pH and Buffer Capacity
Although a significant increase in salivary pH was observed post-intervention (
Table 3 and
Table 4), no significant alterations were noted in buffer capacity. These findings suggest that while the sialogogue improves hydration and flow, it does not modify the intrinsic buffering properties of saliva.
3.4. SXI-PL
The SXI-PL questionnaire revealed total scores ranging from 5 to 15, with a mean of 7.16 ± 2.46. A strong positive correlation was observed between total SXI-PL scores and responses to the standard dry mouth question (
p < 0.001), confirming the internal consistency and validity of the instrument. The most frequently reported symptom was “My lips feel dry” (mean = 1.50 ± 0.65), indicating it as the most impactful in participants’ perception of xerostomia (
Table 5). Over half of the sample (53.1%) reported experiencing dry mouth, and these individuals had higher SXI-PL scores (mean = 8.84 ± 2.28), suggesting greater symptom severity. A statistically significant association (
p > 0.05) was observed between antidepressant use and the presence of xerostomia, suggesting a potential contributory role of this pharmacological group in salivary flow reduction.
4. Discussion
This study demonstrated significant improvement in both unstimulated and stimulated salivary flow rates following the application of a topical sialogogue over a 15-day period in institutionalized psychiatric patients. The mean increase in USFR and in SSFR was statistically significant (
p < 0.001), aligning with the findings of Gómez-Moreno et al. (2013), Marín et al. (2021), and Niklander et al. (2018), who reported similar benefits from topical agents [
3,
4,
5]. The prevalence of xerostomia and hyposialia decreased from 16% at baseline to 7.4% post-treatment, suggesting that even short-term use of a topical sialogogue may effectively reduce symptoms. These results reinforce evidence from Dalodom et al. (2016), although their study applied the intervention for a longer period (1 month), indicating that treatment duration may influence therapeutic outcomes [
6]. The SXI-PL results showed a mean score of 7.16 ± 2.46. The item “my lips feel dry” had the highest impact (1.50 ± 0.65), indicating greater perceived severity. This finding aligns with that of Farsi (2007), who identified dry lips as the most common symptom of oral dryness, reported by 37.5% of participants [
7]. Salivary pH showed a significant shift towards normal physiological values, indicating a less acidic oral environment after treatment. However, no substantial change was observed in buffer capacity, diverging from some previous studies. This difference may be attributed to the relatively short duration of the current intervention. It is evident that no adverse events were reported by the patients. Following the administration of the treatment, no clinically significant alterations were identified, and no adverse events were documented.
5. Conclusions
This study’s limitations include a convenience sample, limited follow-up, and indirect administration of the gel by nursing staff. Nonetheless, the significant improvements observed highlight the potential role of topical sialogogues in xerostomia management for vulnerable populations. Future research should aim for longer-term evaluations, the integration of patient-reported outcomes, and broader population samples to consolidate these preliminary findings and guide clinical protocols in psychiatric settings.
Author Contributions
Conceptualization, E.V. and C.R.; methodology, E.V. and C.R.; software, E.V.; validation, E.V., C.R. and A.C.M.; formal analysis, E.V. and C.R.; investigation, E.V. and C.R.; resources, E.V.; data curation, E.V. and C.R.; writing—original draft preparation, E.V.; writing—review and editing, E.V., C.R. and A.C.M.; visualization, E.V., C.R. and A.C.M.; supervision, E.V. and C.R.; project administration, E.V., C.R. and A.C.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Egas Moniz School of Health and Science (protocol code 1197, approved in 2 March 2023).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available as they are part of an ongoing study.
Acknowledgments
The authors thank the outpatients who participated in the study. We are indebted to the occupational therapy and nursing staff at the CHPL.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Carmona-Huerta, J.; Castiello-de Obeso, S.; Ramírez-Palomino, J.; Duran-Gutiérrez, R.; Cardona-Muller, D.; Grover-Paez, F.; Fernández-Dorantes, P.; Medina-Dávalos, R. Polypharmacy in a hospitalized psychiatric population: Risk estimation and damage quantification. BMC Psychiatry 2019, 19, 78. [Google Scholar] [CrossRef] [PubMed]
- Niklander, S.; Veas, L.; Barrera, C.; Fuentes, F.; Chiappini, G.; Marshall, M. Risk factors, hyposalivation and impact of xerostomia on oral health-related quality of life. Braz. Oral Res. 2017, 31, e14. [Google Scholar] [CrossRef]
- Gómez-Moreno, G.; Guardia, J.; Aguilar-Salvatierra, A.; Cabrera-Ayala, M.; de-Val, J.E.M.S.; Calvo-Guirado, J.L. Effectiveness of malic acid 1% in patients with xerostomia induced by antihypertensive drugs. Med. Oral Patol. Oral Cir. Bucal 2013, 18, e49. [Google Scholar] [CrossRef] [PubMed]
- Marín, C.; Díaz-de-Valdés, L.; Conejeros, C.; Martínez, R.; Niklander, S. Interventions for the treatment of xerostomia: A randomized controlled clinical trial. J. Clin. Exp. Dent. 2021, 13, e104–e111. [Google Scholar] [CrossRef] [PubMed]
- Niklander, S.; Fuentes, F.; Sanchez, D.; Araya, V.; Chiappini, G.; Martinez, R.; Marshall, M. Impact of 1% malic acid spray on the oral health-related quality of life of patients with xerostomia. J. Oral Sci. 2018, 60, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Dalodom, S.; Lam-Ubol, A.; Jeanmaneechotechai, S.; Takamfoo, L.; Intachai, W.; Duangchada, K.; Hongsachum, B.; Kanjanatiwat, P.; Vacharotayangul, P.; Trachootham, D. Influence of oral moisturizing jelly as a saliva substitute for the relief of xerostomia in elderly patients with hypertension and diabetes mellitus. Geriatr. Nurs. 2016, 37, 101–109. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Farsi, N.M. Signs of oral dryness in relation to salivary flow rate, pH, buffering capacity and dry mouth complaints. BMC Oral Health 2007, 7, 15. [Google Scholar] [CrossRef] [PubMed]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).