Extracellular Vesicles as Mediators of Intercellular Communication: Implications for Drug Discovery and Targeted Therapies
Abstract
1. Introduction
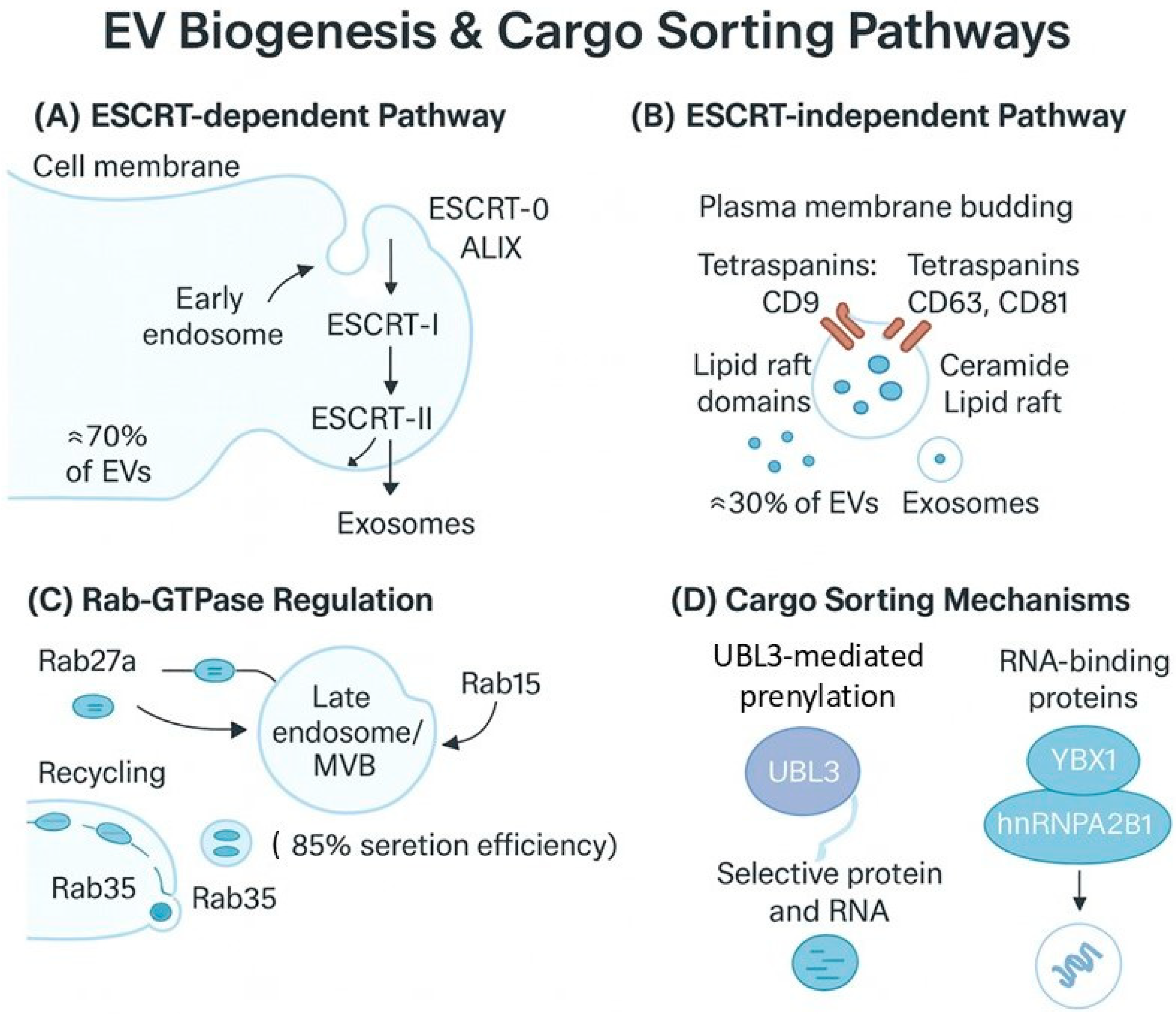
2. Molecular Mechanisms of EV Biogenesis and Cargo Sorting
ESCRT-Dependent and Independent EV Biogenesis Pathways
- 1.
- ESCRT-Dependent Pathway (Primary Route—70% of EVs)
- 2.
- ESCRT-Independent Pathway (Alternative Route—30% of EVs)
- 3.
- Rab GTPases (Trafficking Regulators)
- 4.
- UBL3 (Cargo Customization Factor)
- 5.
- Additional Regulatory Mechanisms
- 6.
- Clinical Relevance
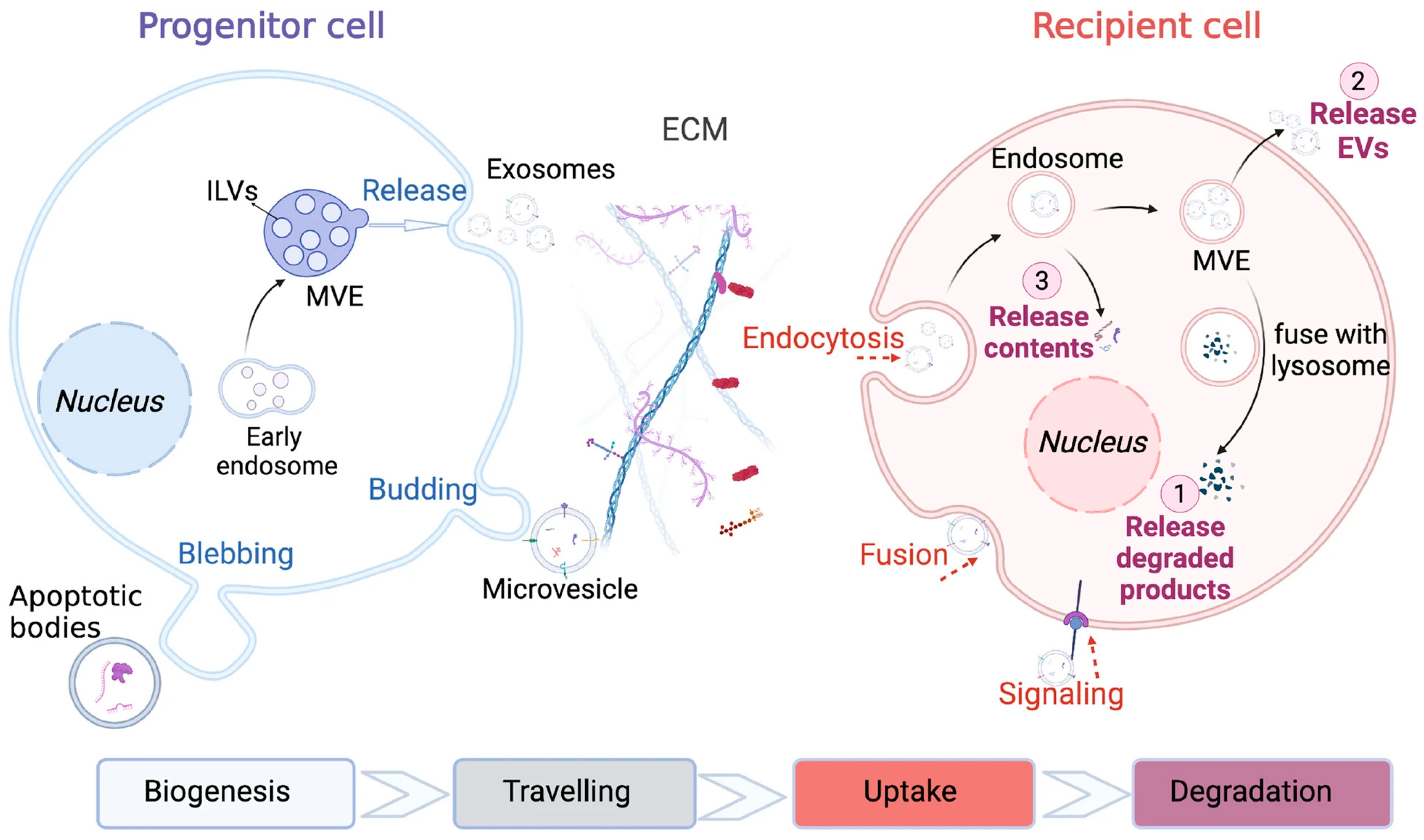
3. EV-Mediated Intercellular Communication in Disease Microenvironments
4. Pharmacological Targeting of EV Pathways
4.1. Inhibitors of EV Release and Uptake
4.2. Enhancers of Therapeutic EV Production
4.3. EV Cargo Modulation Strategies
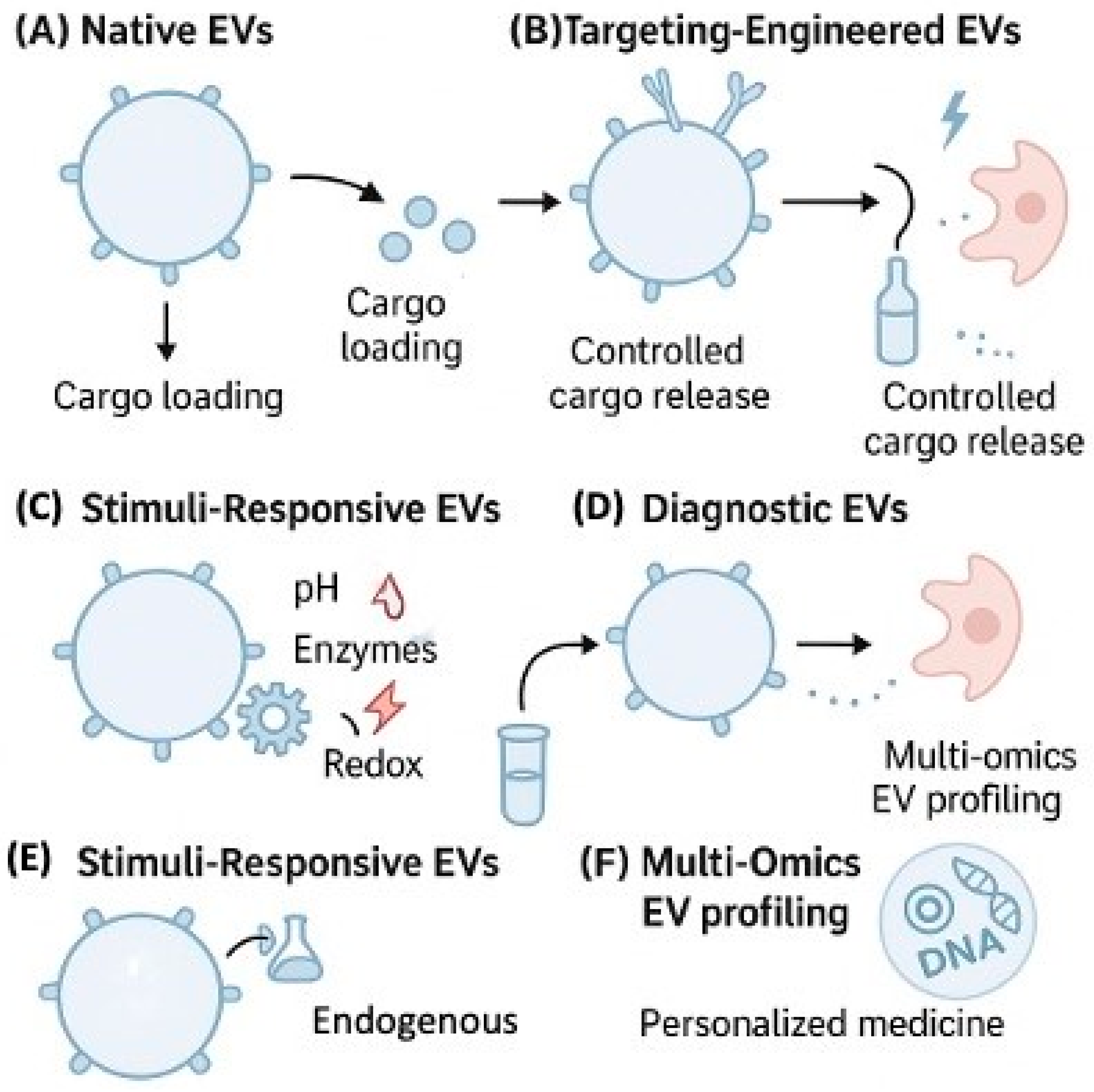
5. EVs as Drug Delivery Vehicles and Biomarker Reservoirs
5.1. EVs for Drug Delivery
5.1.1. Therapeutic Cargo Loading and Delivery
5.1.2. Advanced EV Engineering Approaches
5.2. EVs as Diagnostic Biomarker Reservoirs
5.2.1. Liquid Biopsy Applications
5.2.2. Multi-Omics Profiling and Personalized Medicine
6. EVs in Personalized Medicine and Companion Diagnostics
6.1. EV-Based Biomarker Discovery for Therapeutic Stratification
6.2. Companion Diagnostics and Therapy Selection
6.3. Treatment Response Monitoring and Adaptive Therapy
7. Challenges and Future Perspectives in EV-Based Drug Discovery
7.1. Critical Clinical Translation Failures and Implementation Barriers
7.2. Safety Considerations
7.3. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s Disease |
| ALS | Amyotrophic Lateral Sclerosis |
| Aβ | Amyloid beta |
| BBB | Blood–Brain Barrier |
| CNS | Central Nervous System |
| CM | Conditioned Medium |
| CRC | Colorectal Cancer |
| DC | Dendritic Cell |
| DNA | Deoxyribonucleic Acid |
| EV | Extracellular Vesicle |
| FDA | Food and Drug Administration |
| GSC | Glioma Stem Cell |
| GTPase | Guanosine Triphosphatase |
| HNSCC | Head and Neck Squamous Cell Carcinoma |
| HSP | Heat Shock Protein |
| ILV | Intraluminal Vesicle |
| ISEV | International Society for Extracellular Vesicles |
| KO | Knockout |
| LAMP | Lysosomal-Associated Membrane Protein |
| miRNA | MicroRNA |
| miR | MicroRNA |
| MISEV | Minimal Information for Studies of Extracellular Vesicles |
| MSC | Mesenchymal Stem Cell |
| NSCLC | Non-Small Cell Lung Cancer |
| PD | Parkinson’s Disease |
| PD-L1 | Programmed Death-Ligand 1 |
| RNA | Ribonucleic Acid |
| siRNA | Small Interfering RNA |
| sEV | Small Extracellular Vesicle |
| TME | Tumor Microenvironment |
| TNBC | Triple-Negative Breast Cancer |
| UBL3 | Ubiquitin-Like Protein 3 |
| WT | Wild-Type |
References
- Moghassemi, S.; Dadashzadeh, A.; Sousa, M.J.; Vlieghe, H.; Yang, J.; León-Félix, C.M.; Amorim, C.A. Extracellular Vesicles in Nanomedicine and Regenerative Medicine: A Review over the Last Decade. Bioact. Mater. 2024, 36, 126–156. [Google Scholar] [CrossRef]
- Chronopoulos, A.; Kalluri, R. Emerging Role of Bacterial Extracellular Vesicles in Cancer. Oncogene 2020, 39, 6951–6960. [Google Scholar] [CrossRef]
- El Andaloussi, S.; Mäger, I.; Breakefield, X.O.; Wood, M.J.A. Extracellular Vesicles: Biology and Emerging Therapeutic Opportunities. Nat. Rev. Drug Discov. 2013, 12, 347–357. [Google Scholar] [CrossRef]
- Van Niel, G.; D’Angelo, G.; Raposo, G. Shedding Light on the Cell Biology of Extracellular Vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Anand, S.; Samuel, M.; Kumar, S.; Mathivanan, S. Ticket to a Bubble Ride: Cargo Sorting into Exosomes and Extracellular Vesicles. Biochim. Biophys. Acta Proteins Proteom. 2019, 1867, 140203. [Google Scholar] [CrossRef] [PubMed]
- Yáñez-Mó, M.; Siljander, P.R.M.; Andreu, Z.; Zavec, A.B.; Borràs, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological Properties of Extracellular Vesicles and Their Physiological Functions. J. Extracell. Vesicles 2015, 4, 27066. [Google Scholar] [CrossRef]
- Colombo, M.; Raposo, G.; Théry, C. Biogenesis, Secretion, and Intercellular Interactions of Exosomes and Other Extracellular Vesicles. Annu. Rev. Cell Dev. Biol. 2014, 30, 255–289. [Google Scholar] [CrossRef] [PubMed]
- Raposo, G.; Stoorvogel, W. Extracellular Vesicles: Exosomes, Microvesicles, and Friends. J. Cell Biol. 2013, 200, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Pascucci, L.; Coccè, V.; Bonomi, A.; Ami, D.; Ceccarelli, P.; Ciusani, E.; Viganò, L.; Locatelli, A.; Sisto, F.; Doglia, S.M.; et al. Paclitaxel Is Incorporated by Mesenchymal Stromal Cells and Released in Exosomes That Inhibit in Vitro Tumor Growth: A New Approach for Drug Delivery. J. Control. Release 2014, 192, 262–270. [Google Scholar] [CrossRef]
- Roefs, M.T.; Sluijter, J.P.G.; Vader, P. Extracellular Vesicle-Associated Proteins in Tissue Repair. Trends Cell Biol. 2020, 30, 990–1013. [Google Scholar] [CrossRef]
- Kamerkar, S.; Lebleu, V.S.; Sugimoto, H.; Yang, S.; Ruivo, C.F.; Melo, S.A.; Lee, J.J.; Kalluri, R. Exosomes Facilitate Therapeutic Targeting of Oncogenic KRAS in Pancreatic Cancer. Nature 2017, 546, 498–503. [Google Scholar] [CrossRef]
- Skog, J.; Würdinger, T.; van Rijn, S.; Meijer, D.H.; Gainche, L.; Curry, W.T.; Carter, B.S.; Krichevsky, A.M.; Breakefield, X.O. Glioblastoma Microvesicles Transport RNA and Proteins That Promote Tumour Growth and Provide Diagnostic Biomarkers. Nat. Cell Biol. 2008, 10, 1470–1476. [Google Scholar] [CrossRef] [PubMed]
- Doyle, L.M.; Wang, M.Z. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells 2019, 8, 727. [Google Scholar] [CrossRef] [PubMed]
- Oyama, S.; Zhang, H.; Ferdous, R.; Tomochika, Y.; Chen, B.; Jiang, S.; Islam, M.S.; Hasan, M.M.; Zhai, Q.; Waliullah, A.S.M.; et al. UBL3 Interacts with PolyQ-Expanded Huntingtin Fragments and Modifies Their Intracellular Sorting. Neurol. Int. 2024, 16, 1175–1188. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Erviti, L.; Seow, Y.; Yin, H.; Betts, C.; Lakhal, S.; Wood, M.J.A. Delivery of SiRNA to the Mouse Brain by Systemic Injection of Targeted Exosomes. Nat. Biotechnol. 2011, 29, 341–345. [Google Scholar] [CrossRef]
- Haney, M.J.; Klyachko, N.L.; Zhao, Y.; Gupta, R.; Plotnikova, E.G.; He, Z.; Patel, T.; Piroyan, A.; Sokolsky, M.; Kabanov, A.V.; et al. Exosomes as Drug Delivery Vehicles for Parkinson’s Disease Therapy. J. Control. Release 2015, 207, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Kwok, Z.H.; Wang, C.; Jin, Y. Extracellular Vesicle Transportation and Uptake by Recipient Cells: A Critical Process to Regulate Human Diseases. Processes 2021, 9, 273. [Google Scholar] [CrossRef] [PubMed]
- French, K.C.; Antonyak, M.A.; Cerione, R.A. Extracellular Vesicle Docking at the Cellular Port: Extracellular Vesicle Binding and Uptake. Semin. Cell Dev. Biol. 2017, 67, 48. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, A.; Costa-Silva, B.; Shen, T.L.; Rodrigues, G.; Hashimoto, A.; Tesic Mark, M.; Molina, H.; Kohsaka, S.; Di Giannatale, A.; Ceder, S.; et al. Tumour Exosome Integrins Determine Organotropic Metastasis. Nature 2015, 527, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Vader, P.; Mol, E.A.; Pasterkamp, G.; Schiffelers, R.M. Extracellular Vesicles for Drug Delivery. Adv. Drug Deliv. Rev. 2016, 106, 148–156. [Google Scholar] [CrossRef]
- Du, S.; Guan, Y.; Xie, A.; Yan, Z.; Gao, S.; Li, W.; Rao, L.; Chen, X.; Chen, T. Extracellular Vesicles: A Rising Star for Therapeutics and Drug Delivery. J. Nanobiotechnol. 2023, 21, 231. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, X.; Yang, Z.; Wang, B.; Gong, H.; Zhang, K.; Lin, Y.; Sun, M. Extracellular Vesicles: Biological Mechanisms and Emerging Therapeutic Opportunities in Neurodegenerative Diseases. Transl. Neurodegener. 2024, 13, 60. [Google Scholar] [CrossRef]
- Klyachko, N.L.; Arzt, C.J.; Li, S.M.; Gololobova, O.A.; Batrakova, E.V. Extracellular Vesicle-Based Therapeutics: Preclinical and Clinical Investigations. Pharmaceutics 2020, 12, 1171. [Google Scholar] [CrossRef] [PubMed]
- Marar, C.; Starich, B.; Wirtz, D. Extracellular Vesicles in Immunomodulation and Tumor Progression. Nat. Immunol. 2021, 22, 560–570. [Google Scholar] [CrossRef] [PubMed]
- Fyfe, J.; Casari, I.; Manfredi, M.; Falasca, M. Role of Lipid Signalling in Extracellular Vesicles-Mediated Cell-to-Cell Communication. Cytokine Growth Factor Rev. 2023, 73, 20–26. [Google Scholar] [CrossRef]
- Crewe, C. Energetic Stress-Induced Metabolic Regulation by Extracellular Vesicles. Compr. Physiol. 2023, 13, 5051–5068. [Google Scholar] [CrossRef]
- Beetler, D.J.; Di Florio, D.N.; Bruno, K.A.; Ikezu, T.; March, K.L.; Cooper, L.T.; Wolfram, J.; Fairweather, D.L. Extracellular Vesicles as Personalized Medicine. Mol. Aspects Med. 2023, 91, 101155. [Google Scholar] [CrossRef]
- Fu, E.; Pan, K.; Li, Z. Engineering Extracellular Vesicles for Targeted Therapeutics in Cardiovascular Disease. Front. Cardiovasc. Med. 2024, 11, 1503830. [Google Scholar] [CrossRef]
- Barile, L.; Vassalli, G. Exosomes: Therapy Delivery Tools and Biomarkers of Diseases. Pharmacol. Ther. 2017, 174, 63–78. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, K.; Breyne, K.; Ughetto, S.; Laurent, L.C.; Breakefield, X.O. RNA Delivery by Extracellular Vesicles in Mammalian Cells and Its Applications. Nat. Rev. Mol. Cell Biol. 2020, 21, 585–606. [Google Scholar] [CrossRef] [PubMed]
- Sluijter, J.P.G.; Davidson, S.M.; Boulanger, C.M.; Buzás, E.I.; De Kleijn, D.P.V.; Engel, F.B.; Giricz, Z.; Hausenloy, D.J.; Kishore, R.; Lecour, S.; et al. Extracellular Vesicles in Diagnostics and Therapy of the Ischaemic Heart: Position Paper from the Working Group on Cellular Biology of the Heart of the European Society of Cardiology. Cardiovasc. Res. 2018, 114, 19–34. [Google Scholar] [CrossRef]
- Gong, Z.; Cheng, C.; Sun, C.; Cheng, X. Harnessing Engineered Extracellular Vesicles for Enhanced Therapeutic Efficacy: Advancements in Cancer Immunotherapy. J. Exp. Clin. Cancer Res. 2025, 44, 138. [Google Scholar] [CrossRef] [PubMed]
- Murphy, D.E.; de Jong, O.G.; Brouwer, M.; Wood, M.J.; Lavieu, G.; Schiffelers, R.M.; Vader, P. Extracellular Vesicle-Based Therapeutics: Natural versus Engineered Targeting and Trafficking. Exp. Mol. Med. 2019, 51, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.C.; Jayasinghe, M.K.; Pham, T.T.; Yang, Y.; Wei, L.; Usman, W.M.; Chen, H.; Pirisinu, M.; Gong, J.; Kim, S.; et al. Covalent Conjugation of Extracellular Vesicles with Peptides and Nanobodies for Targeted Therapeutic Delivery. J. Extracell. Vesicles 2021, 10, e12057. [Google Scholar] [CrossRef]
- Doeppner, T.R.; Herz, J.; Görgens, A.; Schlechter, J.; Ludwig, A.-K.; Radtke, S.; de Miroschedji, K.; Horn, P.A.; Giebel, B.; Hermann, D.M. Extracellular Vesicles Improve Post-Stroke Neuroregeneration and Prevent Postischemic Immunosuppression. Stem Cells Transl. Med. 2015, 4, 1131–1143. [Google Scholar] [CrossRef] [PubMed]
- Mimi, M.A.; Hasan, M.M.; Takanashi, Y.; Waliullah, A.S.M.; Mamun, M.A.; Chi, Z.; Kahyo, T.; Aramaki, S.; Takatsuka, D.; Koizumi, K.; et al. UBL3 Overexpression Enhances EV-Mediated Achilles Protein Secretion in Conditioned Media of MDA-MB-231 Cells. Biochem. Biophys. Res. Commun. 2024, 738, 150559. [Google Scholar] [CrossRef] [PubMed]
- You, S.; Barkalifa, R.; Chaney, E.J.; Tu, H.; Park, J.; Sorrells, J.E.; Sun, Y.; Liu, Y.Z.; Yang, L.; Chen, D.Z.; et al. Label-Free Visualization and Characterization of Extracellular Vesicles in Breast Cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 24012–24018. [Google Scholar] [CrossRef]
- Sorrells, J.E.; Park, J.; Aksamitiene, E.; Marjanovic, M.; Martin, E.M.; Chaney, E.J.; Higham, A.M.; Cradock, K.A.; Liu, Z.G.; Boppart, S.A. Label-Free Nonlinear Optical Signatures of Extracellular Vesicles in Liquid and Tissue Biopsies of Human Breast Cancer. Sci. Rep. 2024, 14, 5528. [Google Scholar] [CrossRef]
- Imanbekova, M.; Suarasan, S.; Lu, Y.; Jurchuk, S.; Wachsmann-Hogiu, S. Recent Advances in Optical Label-Free Characterization of Extracellular Vesicles. Nanophotonics 2022, 11, 2827. [Google Scholar] [CrossRef] [PubMed]
- Duffield, C.; Rey Gomez, L.M.; Tsao, S.C.H.; Wang, Y. Recent Advances in SERS Assays for Detection of Multiple Extracellular Vesicles Biomarkers for Cancer Diagnosis. Nanoscale 2025, 17, 3635–3655. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Hasan, M.M.; Zhang, H.; Zhai, Q.; Waliullah, A.S.M.; Ping, Y.; Zhang, C.; Oyama, S.; Mimi, M.A.; Tomochika, Y.; et al. UBL3 Interacts with Alpha-Synuclein in Cells and the Interaction Is Downregulated by the EGFR Pathway Inhibitor Osimertinib. Biomedicines 2023, 11, 1685. [Google Scholar] [CrossRef]
- Han, J.; Zhang, X.; Kang, L.; Guan, J. Extracellular Vesicles as Therapeutic Modulators of Neuroinflammation in Alzheimer’s Disease: A Focus on Signaling Mechanisms. J. Neuroinflamm. 2025, 22, 120. [Google Scholar] [CrossRef]
- Cabrera-Pastor, A. Extracellular Vesicles as Mediators of Neuroinflammation in Intercellular and Inter-Organ Crosstalk. Int. J. Mol. Sci. 2024, 25, 7041. [Google Scholar] [CrossRef]
- Kumar, M.A.; Baba, S.K.; Sadida, H.Q.; Al Marzooqi, S.; Jerobin, J.; Altemani, F.H.; Algehainy, N.; Alanazi, M.A.; Abou-Samra, A.B.; Kumar, R.; et al. Extracellular Vesicles as Tools and Targets in Therapy for Diseases. Signal Transduct. Target. Ther. 2024, 9, 27. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Liu, X.; Sun, M.; Xiong, S.; Xiao, N.; Li, J.; He, X.; Xie, J. Recent Progress in Extracellular Vesicle-Based Carriers for Targeted Drug Delivery in Cancer Therapy. Pharmaceutics 2023, 15, 1902. [Google Scholar] [CrossRef] [PubMed]
- Lener, T.; Gimona, M.; Aigner, L.; Börger, V.; Buzas, E.; Camussi, G.; Chaput, N.; Chatterjee, D.; Court, F.A.; del Portillo, H.A.; et al. Applying Extracellular Vesicles Based Therapeutics in Clinical Trials-An ISEV Position Paper. J. Extracell. Vesicles 2015, 4, 30087. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lan, M.; Chen, Y. Minimal Information for Studies of Extracellular Vesicles (MISEV): Ten-Year Evolution (2014–2023). Pharmaceutics 2024, 16, 1394. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ren, L.; Li, S.; Li, W.; Zheng, X.; Yang, Y.; Fu, W.; Yi, J.; Wang, J.; Du, G. The Biology, Function, and Applications of Exosomes in Cancer. Acta Pharm. Sin. B 2021, 11, 2783–2797. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, X.; Zhang, W.; Yu, L.; Wang, Y.; Deng, Z.; Liu, M.; Mo, S.; Wang, R.; Zhao, J.; et al. Trends in the Biological Functions and Medical Applications of Extracellular Vesicles and Analogues. Acta Pharm. Sin. B 2021, 11, 2114–2135. [Google Scholar] [CrossRef] [PubMed]
- Takanashi, Y.; Kahyo, T.; Kamamoto, S.; Zhang, H.; Chen, B.; Ping, Y.; Mizuno, K.; Kawase, A.; Koizumi, K.; Satou, M.; et al. Ubiquitin-like 3 as a New Protein-Sorting Factor for Small Extracellular Vesicles. Cell Struct. Funct. 2022, 47, 1–18. [Google Scholar] [CrossRef]
- Ageta, H.; Ageta-Ishihara, N.; Hitachi, K.; Karayel, O.; Onouchi, T.; Yamaguchi, H.; Kahyo, T.; Hatanaka, K.; Ikegami, K.; Yoshioka, Y.; et al. UBL3 Modification Influences Protein Sorting to Small Extracellular Vesicles. Nat. Commun. 2018, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Homma, Y.; Hiragi, S.; Fukuda, M. Rab Family of Small GTPases: An Updated View on Their Regulation and Functions. FEBS J. 2020, 288, 36. [Google Scholar] [CrossRef] [PubMed]
- Villarroya-Beltri, C.; Baixauli, F.; Gutiérrez-Vázquez, C.; Sánchez-Madrid, F.; Mittelbrunn, M. Sorting it out: Regulation of Exosome Loading. Semin. Cancer Biol. 2014, 28, 3. [Google Scholar] [CrossRef]
- Ostrowski, M.; Carmo, N.B.; Krumeich, S.; Fanget, I.; Raposo, G.; Savina, A.; Moita, C.F.; Schauer, K.; Hume, A.N.; Freitas, R.P.; et al. Rab27a and Rab27b Control Different Steps of the Exosome Secretion Pathway. Nat. Cell Biol. 2010, 12, 19–30. [Google Scholar] [CrossRef]
- Han, Q.F.; Li, W.J.; Hu, K.S.; Gao, J.; Zhai, W.L.; Yang, J.H.; Zhang, S.J. Exosome Biogenesis: Machinery, Regulation, and Therapeutic Implications in Cancer. Mol. Cancer 2022, 21, 207. [Google Scholar] [CrossRef]
- Alenquer, M.; Amorim, M.J. Exosome Biogenesis, Regulation, and Function in Viral Infection. Viruses 2015, 7, 5066–5083. [Google Scholar] [CrossRef]
- Lee, Y.J.; Shin, K.J.; Chae, Y.C. Regulation of Cargo Selection in Exosome Biogenesis and Its Biomedical Applications in Cancer. Exp. Mol. Med. 2024, 56, 877–889. [Google Scholar] [CrossRef] [PubMed]
- Ju, Y.; Bai, H.; Ren, L.; Zhang, L. The Role of Exosome and the ESCRT Pathway on Enveloped Virus Infection. Int. J. Mol. Sci. 2021, 22, 9060. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, N.; Kyuuma, M.; Sugamura, K. Endosomal Sorting Complex Required for Transport Proteins in Cancer Pathogenesis, Vesicular Transport, and Non-endosomal Functions. Cancer Sci. 2008, 99, 1293. [Google Scholar] [CrossRef] [PubMed]
- Gurung, S.; Perocheau, D.; Touramanidou, L.; Baruteau, J. The Exosome Journey: From Biogenesis to Uptake and Intracellular Signalling. Cell Commun. Signal. 2021, 19, 47. [Google Scholar] [CrossRef] [PubMed]
- Van Deun, J.; Mestdagh, P.; Sormunen, R.; Cocquyt, V.; Vermaelen, K.; Vandesompele, J.; Bracke, M.; De Wever, O.; Hendrix, A. The Impact of Disparate Isolation Methods for Extracellular Vesicles on Downstream RNA Profiling. J. Extracell. Vesicles 2014, 3, 24858. [Google Scholar] [CrossRef] [PubMed]
- Andreu, Z.; Yáñez-Mó, M. Tetraspanins in Extracellular Vesicle Formation and Function. Front. Immunol. 2014, 5, 109543. [Google Scholar] [CrossRef]
- Trajkovic, K.; Hsu, C.; Chiantia, S.; Rajendran, L.; Wenzel, D.; Wieland, F.; Schwille, P.; Brügger, B.; Simons, M. Ceramide Triggers Budding of Exosome Vesicles into Multivesicular Endosomes. Science 2008, 319, 1244–1247. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Chen, B.; Waliullah, A.S.M.; Aramaki, S.; Ping, Y.; Takanashi, Y.; Zhang, C.; Zhai, Q.; Yan, J.; Oyama, S.; et al. A New Potential Therapeutic Target for Cancer in Ubiquitin-Like Proteins—UBL3. Int. J. Mol. Sci. 2023, 24, 1231. [Google Scholar] [CrossRef] [PubMed]
- Carnino, J.M.; Ni, K.; Jin, Y. Post-Translational Modification Regulates Formation and Cargo-Loading of Extracellular Vesicles. Front. Immunol. 2020, 11, 948. [Google Scholar] [CrossRef]
- Atukorala, I.; Mathivanan, S. The Role of Post-Translational Modifications in Targeting Protein Cargo to Extracellular Vesicles. Subcell. Biochem. 2021, 97, 45–60. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Singh, J.; Schekman, R. Two RNA-Binding Proteins Mediate the Sorting of MiR223 from Mitochondria into Exosomes. Elife 2023, 12, e85878. [Google Scholar] [CrossRef] [PubMed]
- Mir, B.; Goettsch, C. Extracellular Vesicles as Delivery Vehicles of Specific Cellular Cargo. Cells 2020, 9, 1601. [Google Scholar] [CrossRef] [PubMed]
- Booth, A.; Marklew, C.J.; Ciani, B.; Beales, P.A. The influence of phosphatidylserine localisation and lipid phase on membrane remodelling by the ESCRT-II/ESCRT-III complex. Faraday Discussions 2021, 232, 188–202. [Google Scholar] [CrossRef]
- Liu, H.; Wilson, K.R.; Firth, A.M.; Macri, C.; Schriek, P.; Blum, A.B.; Villar, J.; Wormald, S.; Shambrook, M.; Xu, B.; et al. Ubiquitin-like Protein 3 (UBL3) Is Required for MARCH Ubiquitination of Major Histocompatibility Complex Class II and CD86. Nat. Commun. 2022, 13, 1934. [Google Scholar] [CrossRef]
- Terada, Y.; Obara, K.; Yoshioka, Y.; Ochiya, T.; Bito, H.; Tsuchida, K.; Ageta, H.; Ageta-Ishihara, N. Intracellular Dynamics of Ubiquitin-like 3 Visualized Using an Inducible Fluorescent Timer Expression System. Biol. Open 2024, 13, bio060345. [Google Scholar] [CrossRef] [PubMed]
- Elu, N.; Lectez, B.; Ramirez, J.; Osinalde, N.; Mayor, U. Mass Spectrometry-Based Characterization of Ub- and UbL-Modified Proteins. Methods Mol. Biol. 2020, 2051, 265–276. [Google Scholar] [CrossRef]
- Jeram, S.M.; Srikumar, T.; Pedrioli, P.G.A.; Raught, B. Using Mass Spectrometry to Identify Ubiquitin and Ubiquitin-like Protein Conjugation Sites. Proteomics 2009, 9, 922–934. [Google Scholar] [CrossRef] [PubMed]
- Faber, S.; Letteboer, S.J.F.; Junger, K.; Butcher, R.; Tammana, T.V.S.; van Beersum, S.E.C.; Ueffing, M.; Collin, R.W.J.; Liu, Q.; Boldt, K.; et al. PDE6D Mediates Trafficking of Prenylated Proteins NIM1K and UBL3 to Primary Cilia. Cells 2023, 12, 312. [Google Scholar] [CrossRef] [PubMed]
- Menck, K.; Sönmezer, C.; Worst, T.S.; Schulz, M.; Dihazi, G.H.; Streit, F.; Erdmann, G.; Kling, S.; Boutros, M.; Binder, C.; et al. Neutral Sphingomyelinases Control Extracellular Vesicles Budding from the Plasma Membrane. J. Extracell. Vesicles 2017, 6, 1378056. [Google Scholar] [CrossRef]
- Keerthikumar, S.; Gangoda, L.; Liem, M.; Fonseka, P.; Atukorala, I.; Ozcitti, C.; Mechler, A.; Adda, C.G.; Ang, C.S.; Mathivanan, S. Proteogenomic Analysis Reveals Exosomes Are More Oncogenic than Ectosomes. Oncotarget 2015, 6, 15375–15396. [Google Scholar] [CrossRef]
- King, H.W.; Michael, M.Z.; Gleadle, J.M. Hypoxic Enhancement of Exosome Release by Breast Cancer Cells. BMC Cancer 2012, 12, 421. [Google Scholar] [CrossRef]
- Mittelbrunn, M.; Gutiérrez-Vázquez, C.; Villarroya-Beltri, C.; González, S.; Sánchez-Cabo, F.; González, M.Á.; Bernad, A.; Sánchez-Madrid, F. Unidirectional Transfer of MicroRNA-Loaded Exosomes from T Cells to Antigen-Presenting Cells. Nat. Commun. 2011, 2, 282. [Google Scholar] [CrossRef] [PubMed]
- Budnik, V.; Ruiz-Cañada, C.; Wendler, F. Extracellular Vesicles Round off Communication in the Nervous System. Nat. Rev. Neurosci. 2016, 17, 160–172. [Google Scholar] [CrossRef]
- Prieto-Vila, M.; Yoshioka, Y.; Ochiya, T. Biological Functions Driven by MRNAs Carried by Extracellular Vesicles in Cancer. Front. Cell Dev. Biol. 2021, 9, 620498. [Google Scholar] [CrossRef]
- Bao, Q.; Huang, Q.; Chen, Y.; Wang, Q.; Sang, R.; Wang, L.; Xie, Y.; Chen, W. Tumor-Derived Extracellular Vesicles Regulate Cancer Progression in the Tumor Microenvironment. Front. Mol. Biosci. 2022, 8, 796385. [Google Scholar] [CrossRef] [PubMed]
- Jurj, A.; Zanoaga, O.; Braicu, C.; Lazar, V.; Tomuleasa, C.; Irimie, A.; Berindan-neagoe, I. A Comprehensive Picture of Extracellular Vesicles and Their Contents. Molecular Transfer to Cancer Cells. Cancers 2020, 12, 298. [Google Scholar] [CrossRef] [PubMed]
- Jabalee, J.; Towle, R.; Garnis, C. The role of extracellular vesicles in cancer: Cargo, function, and therapeutic implications. Cells 2018, 7, 93. [Google Scholar] [CrossRef]
- Basso, M.; Bonetto, V. Extracellular Vesicles and a Novel Form of Communication in the Brain. Front. Neurosci. 2016, 10, 127. [Google Scholar] [CrossRef] [PubMed]
- Arif, S.; Qazi, T.J.; Quan, Z.; Ni, J.; Li, Z.; Qiu, Y.; Qing, H. Extracellular Vesicle-Packed MicroRNAs Profiling in Alzheimer’s Disease: The Molecular Intermediary between Pathology and Diagnosis. Ageing Res. Rev. 2025, 104, 102614. [Google Scholar] [CrossRef]
- Aulston, B.; Liu, Q.; Mante, M.; Florio, J.; Rissman, R.A.; Yuan, S.H. Extracellular Vesicles Isolated from Familial Alzheimer’s Disease Neuronal Cultures Induce Aberrant Tau Phosphorylation in the Wild-Type Mouse Brain. J. Alzheimers Dis. 2019, 72, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Pérez, M.; Avila, J.; Hernández, F. Propagation of Tau via Extracellular Vesicles. Front. Neurosci. 2019, 13, 698. [Google Scholar] [CrossRef]
- Hodge, A.L.; Baxter, A.A.; Poon, I.K.H. Gift Bags from the Sentinel Cells of the Immune System: The Diverse Role of Dendritic Cell-Derived Extracellular Vesicles. J. Leukoc. Biol. 2022, 111, 903–920. [Google Scholar] [CrossRef]
- Fridman, E.S.; Ginini, L.; Gil, Z. The Role of Extracellular Vesicles in Metabolic Reprogramming of the Tumor Microenvironment. Cells 2022, 11, 1433. [Google Scholar] [CrossRef]
- Zhang, Q.S.; Heng, Y.; Yuan, Y.H.; Chen, N.H. Pathological α-Synuclein Exacerbates the Progression of Parkinson’s Disease through Microglial Activation. Toxicol. Lett. 2017, 265, 30–37. [Google Scholar] [CrossRef]
- Klein, S. Alpha-Synuclein Promotes Dopaminergic Neuron Death in Parkinson’s Disease through Microglial and NLRP3 Activation. Univ. Sask. Undergrad. Res. J. 2020, 6. [Google Scholar] [CrossRef]
- Encarnação, C.C.; Faria, G.M.; Franco, V.A.; Botelho, L.G.X.; Moraes, J.A.; Renovato-Martins, M. Interconnections within the Tumor Microenvironment: Extracellular Vesicles as Critical Players of Metabolic Reprogramming in Tumor Cells. J. Cancer Metastasis Treat. 2024, 10, 28. [Google Scholar] [CrossRef]
- Poggio, M.; Hu, T.; Pai, C.C.; Chu, B.; Belair, C.D.; Chang, A.; Montabana, E.; Lang, U.E.; Fu, Q.; Fong, L.; et al. Suppression of Exosomal PD-L1 Induces Systemic Anti-Tumor Immunity and Memory. Cell 2019, 177, 414–427.e13. [Google Scholar] [CrossRef] [PubMed]
- Czernek, L.; Düchler, M. Functions of Cancer-Derived Extracellular Vesicles in Immunosuppression. Arch. Immunol. Ther. Exp. 2017, 65, 311–323. [Google Scholar] [CrossRef]
- Robbins, P.D.; Morelli, A.E. Regulation of Immune Responses by Extracellular Vesicles. Nat. Rev. Immunol. 2014, 14, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Patras, L.; Banciu, M. Intercellular Crosstalk Via Extracellular Vesicles in Tumor Milieu as Emerging Therapies for Cancer Progression. Curr. Pharm. Des. 2019, 25, 1980–2006. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Hu, Y.; Yan, W. Extracellular Vesicle-Mediated Interorgan Communication in Metabolic Diseases. Trends Endocrinol. Metab. 2023, 34, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, W.; Hu, D.; Liang, Y.; Liu, Z.; Zhong, T.; Wang, X. Tumor-Derived Extracellular Vesicles Regulate Macrophage Polarization: Role and Therapeutic Perspectives. Front. Immunol. 2024, 15, 1346587. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Wang, F.; Xia, Y.; Wang, L.; Lin, H.; Zhong, T.; Wang, X. Research Progress on Astrocyte-Derived Extracellular Vesicles in the Pathogenesis and Treatment of Neurodegenerative Diseases. Rev. Neurosci. 2024, 35, 855–875. [Google Scholar] [CrossRef]
- Liu, Y.J.; Wang, C. A Review of the Regulatory Mechanisms of Extracellular Vesicles-Mediated Intercellular Communication. Cell Commun. Signal. 2023, 21, 77. [Google Scholar] [CrossRef]
- Mulcahy, L.A.; Pink, R.C.; Raul, D.; Carter, F.; David, D.; Carter, R.F. Routes and Mechanisms of Extracellular Vesicle Uptake. J. Extracell. Vesicles 2014, 3, 24641. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Ru, Z.; Xiao, W.; Xiong, Z.; Wang, C.; Yuan, C.; Zhang, X.; Yang, H. Adipose Tissue Browning in Cancer-Associated Cachexia Can Be Attenuated by Inhibition of Exosome Generation. Biochem. Biophys. Res. Commun. 2018, 506, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Kumar, P.; Sharma, M.; Kim, S.; Singh, S.; Kridel, S.J.; Deep, G. Role of Extracellular Vesicles Secretion in Paclitaxel Resistance of Prostate Cancer Cells. Cancer Drug Resist. 2022, 5, 612. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wang, K.; Lu, Y.; Sun, B.; Sun, J.; Liang, W. Monensin Enhanced Generation of Extracellular Vesicles as Transfersomes for Promoting Tumor Penetration of Pyropheophorbide-a from Fusogenic Liposome. Nano Lett. 2022, 22, 1415–1424. [Google Scholar] [CrossRef]
- Erwin, N.; Serafim, M.F.; He, M. Enhancing the Cellular Production of Extracellular Vesicles for Developing Therapeutic Applications. Pharm. Res. 2022, 40, 833. [Google Scholar] [CrossRef]
- Tu, C.; Du, Z.; Zhang, H.; Feng, Y.; Qi, Y.; Zheng, Y.; Liu, J.; Wang, J. Endocytic Pathway Inhibition Attenuates Extracellular Vesicle-Induced Reduction of Chemosensitivity to Bortezomib in Multiple Myeloma Cells. Theranostics 2021, 11, 2364. [Google Scholar] [CrossRef] [PubMed]
- Essandoh, K.; Yang, L.; Wang, X.; Huang, W.; Qin, D.; Hao, J.; Wang, Y.; Zingarelli, B.; Peng, T.; Fan, G.C. Blockade of Exosome Generation with GW4869 Dampens the Sepsis-Induced Inflammation and Cardiac Dysfunction. Biochim. Biophys. Acta 2015, 1852, 2362. [Google Scholar] [CrossRef] [PubMed]
- Catalano, M.; O’Driscoll, L. Inhibiting Extracellular Vesicles Formation and Release: A Review of EV Inhibitors. J. Extracell. Vesicles 2019, 9, 1703244. [Google Scholar] [CrossRef] [PubMed]
- Kariminejad-Farsangi, H.; Mirzaee Khalilabadi, R.; Afgar, A.; Mirzaie, M. and Mardani Valandani, H. Microvesicle inhibition enhances the therapeutic effects of ATRA in acute promyelocytic leukemia cells via changes in miRNAs: The promising antileukemic potential of imipramine. Clin. Exp. Med. 2025, 25, 217. [Google Scholar] [CrossRef] [PubMed]
- Devulder, J.; Baker, J.; Cao, S.; Donnelly, L.; Barnes, P. Extracellular vesicles are taken up by airway epithelial cells through a dynamin-dependent mechanism. Eur. Respir. J. 2023, 62 (Suppl. 67), 562. [Google Scholar] [CrossRef]
- Jackson Cullison, S.R.; Flemming, J.P.; Karagoz, K.; Wermuth, P.J.; Mahoney, M.G. Mechanisms of Extracellular Vesicle Uptake and Implications for the Design of Cancer Therapeutics. J. Extracell. Biol. 2024, 3, e70017. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Song, H.; Zhou, Z.; Chen, X.; Li, H.; Zhang, Y.; Wang, J.; Ren, X.; Wang, X. Promotion or Inhibition of Extracellular Vesicle Release: Emerging Therapeutic Opportunities. J. Control. Release 2021, 340, 136–148. [Google Scholar] [CrossRef]
- Man, K.; Brunet, M.Y.; Fernandez-Rhodes, M.; Williams, S.; Heaney, L.M.; Gethings, L.A.; Federici, A.; Davies, O.G.; Hoey, D.; Cox, S.C. Epigenetic Reprogramming Enhances the Therapeutic Efficacy of Osteoblast-Derived Extracellular Vesicles to Promote Human Bone Marrow Stem Cell Osteogenic Differentiation. J. Extracell. Vesicles 2021, 10, e12118. [Google Scholar] [CrossRef] [PubMed]
- Dixson, A.C.; Dawson, T.R.; Di Vizio, D.; Weaver, A.M. Context-Specific Regulation of Extracellular Vesicle Biogenesis and Cargo Selection. Nat. Rev. Mol. Cell Biol. 2023, 24, 454–476. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, P.; Vardaki, I.; Occhionero, A.; Panaretakis, T. Metabolic and Signaling Functions of Cancer Cell-Derived Extracellular Vesicles. Int. Rev. Cell Mol. Biol. 2016, 326, 175–199. [Google Scholar] [CrossRef]
- Beaumont, J.E.J.; Barbeau, L.M.O.; Ju, J.; Savelkouls, K.G.; Bouwman, F.G.; Zonneveld, M.I.; Bronckaers, A.; Kampen, K.R.; Keulers, T.G.H.; Rouschop, K.M.A. Cancer EV Stimulate Endothelial Glycolysis to Fuel Protein Synthesis via MTOR and AMPKα Activation. J. Extracell. Vesicles 2024, 13, e12449. [Google Scholar] [CrossRef]
- Siddique, M.M.; Bikman, B.T.; Wang, L.; Ying, L.; Reinhardt, E.; Shui, G.; Wenk, M.R.; Summers, S.A. Ablation of Dihydroceramide Desaturase Confers Resistance to Etoposide-Induced Apoptosis In Vitro. PLoS ONE 2012, 7, e44042. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jiang, H.; Kumarasamy, R.V.; Pei, J.J.; Raju, K.; Kanniappan, G.V.; Palanisamy, C.P.; Mironescu, I.D. Integrating Engineered Nanomaterials with Extracellular Vesicles: Advancing Targeted Drug Delivery and Biomedical Applications. Front. Nanotechnol. 2024, 6, 1513683. [Google Scholar] [CrossRef]
- Armstrong, J.P.K.; Holme, M.N.; Stevens, M.M. Re-Engineering Extracellular Vesicles as Smart Nanoscale Therapeutics. ACS Nano 2017, 11, 69–83. [Google Scholar] [CrossRef]
- Kodam, S.P.; Ullah, M. Diagnostic and therapeutic potential of extracellular vesicles. Technol. Cancer Res. Treat. 2021, 20, 15330338211041203. [Google Scholar] [CrossRef]
- Tenchov, R.; Sasso, J.M.; Wang, X.; Liaw, W.S.; Chen, C.A.; and Zhou, Q.A. Exosomes─ nature’s lipid nanoparticles, a rising star in drug delivery and diagnostics. ACS Nano 2022, 16, 17802–17846. [Google Scholar] [CrossRef]
- Han, Y.; Jones, T.W.; Dutta, S.; Zhu, Y.; Wang, X.; Narayanan, S.P.; Fagan, S.C.; Zhang, D. Overview and Update on Methods for Cargo Loading into Extracellular Vesicles. Processes 2021, 9, 356. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Godbout, K.; Lamothe, G.; Tremblay, J.P. CRISPR-Cas9 Delivery Strategies with Engineered Extracellular Vesicles. Mol. Ther. Nucleic Acids 2023, 34, 102040. [Google Scholar] [CrossRef]
- Sutaria, D.S.; Badawi, M.; Phelps, M.A.; Schmittgen, T.D. Achieving the Promise of Therapeutic Extracellular Vesicles: The Devil Is in Details of Therapeutic Loading. Pharm. Res. 2017, 34, 1053–1066. [Google Scholar] [CrossRef] [PubMed]
- Dooley, K.; McConnell, R.E.; Xu, K.; Lewis, N.D.; Haupt, S.; Youniss, M.R.; Martin, S.; Sia, C.L.; McCoy, C.; Moniz, R.J.; et al. A Versatile Platform for Generating Engineered Extracellular Vesicles with Defined Therapeutic Properties. Mol. Ther. 2021, 29, 1729–1743. [Google Scholar] [CrossRef]
- Wang, L.; Wang, D.; Ye, Z.; Xu, J. Engineering Extracellular Vesicles as Delivery Systems in Therapeutic Applications. Adv. Sci. 2023, 10, 2300552. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.; Busatto, S.; Pham, A.; Tian, M.; Suh, A.; Carson, K.; Quintero, A.; Lafrence, M.; Malik, H.; Santana, M.X.; et al. Extracellular Vesicle-Based Drug Delivery Systems for Cancer Treatment. Theranostics 2019, 9, 8001. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, D.A.; Vader, P. Extracellular Vesicle-Based Hybrid Systems for Advanced Drug Delivery. Pharmaceutics 2022, 14, 267. [Google Scholar] [CrossRef] [PubMed]
- Ducrot, C.; Loiseau, S.; Wong, C.; Madec, E.; Volatron, J.; Piffoux, M. Hybrid Extracellular Vesicles for Drug Delivery. Cancer Lett. 2023, 558, 216107. [Google Scholar] [CrossRef]
- Frolova, L.; Li, I.T.S. Targeting Capabilities of Native and Bioengineered Extracellular Vesicles for Drug Delivery. Bioengineering 2022, 9, 496. [Google Scholar] [CrossRef]
- Sheng, Y.; Hu, J.; Shi, J.; Lee, L.J. Stimuli-Responsive Carriers for Controlled Intracellular Drug Release. Curr. Med. Chem. 2019, 26, 2377–2388. [Google Scholar] [CrossRef]
- Liu, J.; Chen, Y.; Pei, F.; Zeng, C.; Yao, Y.; Liao, W.; Zhao, Z. Extracellular Vesicles in Liquid Biopsies: Potential for Disease Diagnosis. Biomed. Res. Int. 2021, 2021, 6611244. [Google Scholar] [CrossRef]
- Revenfeld, A.L.S.; Bæk, R.; Nielsen, M.H.; Stensballe, A.; Varming, K.; Jørgensen, M. Diagnostic and Prognostic Potential of Extracellular Vesicles in Peripheral Blood. Clin. Ther. 2014, 36, 830–846. [Google Scholar] [CrossRef] [PubMed]
- Boukouris, S.; Mathivanan, S. Exosomes in Bodily Fluids Are a Highly Stable Resource of Disease Biomarkers. Proteom. Clin. Appl. 2015, 9, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Jia, E.; Ren, N.; Shi, X.; Zhang, R.; Yu, H.; Yu, F.; Qin, S.; Xue, J. Extracellular Vesicle Biomarkers for Pancreatic Cancer Diagnosis: A Systematic Review and Meta-Analysis. BMC Cancer 2022, 22, 573. [Google Scholar] [CrossRef]
- Yang, K.S.; Im, H.; Hong, S.; Pergolini, I.; Del Castillo, A.F.; Wang, R.; Clardy, S.; Huang, C.H.; Pille, C.; Ferrone, S.; et al. Multiparametric Plasma EV Profiling Facilitates Diagnosis of Pancreatic Malignancy. Sci. Transl. Med. 2017, 9, eaal3226. [Google Scholar] [CrossRef] [PubMed]
- Yap, S.A.; Münster-Wandowski, A.; Nonnenmacher, A.; Keilholz, U.; Liebs, S. Analysis of Cancer-Related Mutations in Extracellular Vesicles RNA by Droplet DigitalTMPCR. Biotechniques 2020, 69, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Butler, T.M.; Spellman, P.T.; Gray, J. Circulating-Tumor DNA as an Early Detection and Diagnostic Tool. Curr. Opin. Genet. Dev. 2017, 42, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, M.H.; Caires, H.R.; Ābols, A.; Xavier, C.P.R.; Linē, A. Extracellular Vesicles as a Novel Source of Biomarkers in Liquid Biopsies for Monitoring Cancer Progression and Drug Resistance. Drug Resist. Updates 2019, 47, 100647. [Google Scholar] [CrossRef]
- Rayamajhi, S.; Sipes, J.; Tetlow, A.L.; Saha, S.; Bansal, A.; Godwin, A.K. Extracellular Vesicles as Liquid Biopsy Biomarkers across the Cancer Journey: From Early Detection to Recurrence. Clin. Chem. 2024, 70, 206–219. [Google Scholar] [CrossRef] [PubMed]
- Paproski, R.J.; Pink, D.; Sosnowski, D.L.; Vasquez, C.; Lewis, J.D. Building Predictive Disease Models Using Extracellular Vesicle Microscale Flow Cytometry and Machine Learning. Mol. Oncol. 2023, 17, 407–421. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Fu, Y.; Liu, G.; Shu, B.; Davis, J.; Tofaris, G.K. Multiplexed Profiling of Extracellular Vesicles for Biomarker Development. Nanomicro Lett. 2022, 14, 3. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.E. Extracellular Vesicles in Cancer Therapy. Semin. Cancer Biol. 2022, 86, 296. [Google Scholar] [CrossRef] [PubMed]
- Saleem, T.; Sumrin, A.; Bilal, M.; Bashir, H.; Khawar, M.B. Tumor-Derived Extracellular Vesicles: Potential Tool for Cancer Diagnosis, Prognosis, and Therapy. Saudi J. Biol. Sci. 2022, 29, 2063. [Google Scholar] [CrossRef]
- Welsh, J.A.; Goberdhan, D.C.I.; O’Driscoll, L.; Buzas, E.I.; Blenkiron, C.; Bussolati, B.; Cai, H.; Di Vizio, D.; Driedonks, T.A.P.; Erdbrügger, U.; et al. Minimal Information for Studies of Extracellular Vesicles (MISEV2023): From Basic to Advanced Approaches. J. Extracell. Vesicles 2024, 13, e12404. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, Y.; Kosaka, N.; Konishi, Y.; Ohta, H.; Okamoto, H.; Sonoda, H.; Nonaka, R.; Yamamoto, H.; Ishii, H.; Mori, M.; et al. Ultra-Sensitive Liquid Biopsy of Circulating Extracellular Vesicles Using ExoScreen. Nat. Commun. 2014, 5, 3591. [Google Scholar] [CrossRef]
- Holtzman, J.; Lee, H. Emerging Role of Extracellular Vesicles in the Respiratory System. Exp. Mol. Med. 2020, 52, 887–895. [Google Scholar] [CrossRef]
- Li, P.; Kaslan, M.; Lee, S.H.; Yao, J.; Gao, Z. Progress in Exosome Isolation Techniques. Theranostics 2017, 7, 789–804. [Google Scholar] [CrossRef] [PubMed]
- Goričar, K.; Dolžan, V.; Lenassi, M. Extracellular Vesicles: A Novel Tool Facilitating Personalized Medicine and Pharmacogenomics in Oncology. Front. Pharmacol. 2021, 12, 671298. [Google Scholar] [CrossRef] [PubMed]
- Che Shaffi, S.; Hairuddin, O.N.; Mansor, S.F.; Syafiq, T.M.F.; Yahaya, B.H. Unlocking the potential of extracellular vesicles as the next generation therapy: Challenges and opportunities. Tissue Eng. Regen. Med. 2024, 21, 513–527. [Google Scholar] [CrossRef]
- Stremersch, S.; De Smedt, S.C.; Raemdonck, K. Therapeutic and Diagnostic Applications of Extracellular Vesicles. J. Control. Release 2016, 244, 167–183. [Google Scholar] [CrossRef] [PubMed]
- de Miguel-Perez, D.; Russo, A.; Arrieta, O.; Ak, M.; Barron, F.; Gunasekaran, M.; Mamindla, P.; Lara-Mejia, L.; Peterson, C.B.; Er, M.E.; et al. Extracellular Vesicle PD-L1 Dynamics Predict Durable Response to Immune-Checkpoint Inhibitors and Survival in Patients with Non-Small Cell Lung Cancer. J. Exp. Clin. Cancer Res. 2022, 41, 186. [Google Scholar] [CrossRef] [PubMed]
- Scheerens, H.; Malong, A.; Bassett, K.; Boyd, Z.; Gupta, V.; Harris, J.; Mesick, C.; Simnett, S.; Stevens, H.; Gilbert, H.; et al. Current status of companion and complementary diagnostics: Strategic considerations for development and launch. Clin. Transl. Sci. 2017, 10, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Herrero, E.; Campos-Silva, C.; Cáceres-Martell, Y.; Robado De Lope, L.; Sanz-Moreno, S.; Serna-Blasco, R.; Rodríguez-Festa, A.; Ares Trotta, D.; Martín-Acosta, P.; Patiño, C.; et al. ALK-Fusion Transcripts Can Be Detected in Extracellular Vesicles (EVs) from Nonsmall Cell Lung Cancer Cell Lines and Patient Plasma: Toward EV-Based Noninvasive Testing. Clin. Chem. 2022, 68, 668–679. [Google Scholar] [CrossRef] [PubMed]
- Purcell, E.; Owen, S.; Prantzalos, E.; Radomski, A.; Carman, N.; Lo, T.W.; Zeinali, M.; Subramanian, C.; Ramnath, N.; Nagrath, S. Epidermal Growth Factor Receptor Mutations Carried in Extracellular Vesicle-Derived Cargo Mirror Disease Status in Metastatic Non-Small Cell Lung Cancer. Front. Cell Dev. Biol. 2021, 9, 724389. [Google Scholar] [CrossRef] [PubMed]
- Riazifar, M.; Mohammadi, M.R.; Pone, E.J.; Yeri, A.; Lasser, C.; Segaliny, A.I.; McIntyre, L.L.; Shelke, G.V.; Hutchins, E.; Hamamoto, A.; et al. Stem Cell-Derived Exosomes as Nanotherapeutics for Autoimmune and Neurodegenerative Disorders. ACS Nano 2019, 13, 6670–6688. [Google Scholar] [CrossRef]
- Buschmann, D.; Mussack, V.; Byrd, J.B. Separation, Characterization, and Standardization of Extracellular Vesicles for Drug Delivery Applications. Adv. Drug Deliv. Rev. 2021, 174, 348–368. [Google Scholar] [CrossRef]
- Sódar, B.W.; Kittel, Á.; Pálóczi, K.; Vukman, K.V.; Osteikoetxea, X.; Szabó-Taylor, K.; Németh, A.; Sperlágh, B.; Baranyai, T.; Giricz, Z.; et al. Low-Density Lipoprotein Mimics Blood Plasma-Derived Exosomes and Microvesicles during Isolation and Detection. Sci. Rep. 2016, 6, 24316. [Google Scholar] [CrossRef]
- Casanova-Salas, I.; Aguilar, D.; Cordoba-Terreros, S.; Agundez, L.; Brandariz, J.; Herranz, N.; Mas, A.; Gonzalez, M.; Morales-Barrera, R.; Sierra, A.; et al. Circulating Tumor Extracellular Vesicles to Monitor Metastatic Prostate Cancer Genomics and Transcriptomic Evolution. Cancer Cell 2024, 42, 1301–1312.e7. [Google Scholar] [CrossRef]
- Ciani, Y.; Nardella, C.; Demichelis, F. Casting a Wider Net: The Clinical Potential of EV Transcriptomics in Multi-Analyte Liquid Biopsy. Cancer Cell 2024, 42, 1160–1162. [Google Scholar] [CrossRef]
- Carney, R.P.; Mizenko, R.R.; Bozkurt, B.T.; Lowe, N.; Henson, T.; Arizzi, A.; Wang, A.; Tan, C.; George, S.C. Harnessing extracellular vesicle heterogeneity for diagnostic and therapeutic applications. Nat. Nanotechnol. 2025, 20, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Bacon, K.; Lavoie, A.; Rao, B.M.; Daniele, M.; Menegatti, S. Past, Present, and Future of Affinity-Based Cell Separation Technologies. Acta Biomater. 2020, 112, 29–51. [Google Scholar] [CrossRef]
- Wiest, E.F.; Zubair, A.C. Challenges of Manufacturing Mesenchymal Stromal Cell–Derived Extracellular Vesicles in Regenerative Medicine. Cytotherapy 2020, 22, 606–612. [Google Scholar] [CrossRef]
- Mateescu, B.; Kowal, E.J.K.; van Balkom, B.W.M.; Bartel, S.; Bhattacharyya, S.N.; Buzás, E.I.; Buck, A.H.; de Candia, P.; Chow, F.W.N.; Das, S.; et al. Obstacles and Opportunities in the Functional Analysis of Extracellular Vesicle RNA-An ISEV Position Paper. J. Extracell. Vesicles 2017, 6, 1286095. [Google Scholar] [CrossRef]
- Gupta, D.; Zickler, A.M.; El Andaloussi, S. Dosing Extracellular Vesicles. Adv. Drug Deliv. Rev. 2021, 178, 113961. [Google Scholar] [CrossRef]
- Armstrong, J.P.K.; Stevens, M.M. Strategic Design of Extracellular Vesicle Drug Delivery Systems. Adv. Drug Deliv. Rev. 2018, 130, 12–16. [Google Scholar] [CrossRef]
- Liu, J.; Nordin, J.Z.; McLachlan, A.J.; Chrzanowski, W. Extracellular Vesicles as the Next-Generation Modulators of Pharmacokinetics and Pharmacodynamics of Medications and Their Potential as Adjuvant Therapeutics. Clin. Transl. Med. 2024, 14, e70002. [Google Scholar] [CrossRef]
- Takakura, Y.; Matsumoto, A.; Takahashi, Y. Therapeutic Application of Small Extracellular Vesicles (SEVs): Pharmaceutical and Pharmacokinetic Challenges. Biol. Pharm. Bull. 2020, 43, 576–583. [Google Scholar] [CrossRef]
- Estes, S.; Konstantinov, K.; Young, J.D. Manufactured Extracellular Vesicles as Human Therapeutics: Challenges, Advances, and Opportunities. Curr. Opin. Biotechnol. 2022, 77, 102776. [Google Scholar] [CrossRef] [PubMed]
- Villata, S.; Canta, M.; Cauda, V. Evs and Bioengineering: From Cellular Products to Engineered Nanomachines. Int. J. Mol. Sci. 2020, 21, 6048. [Google Scholar] [CrossRef] [PubMed]
- Claridge, B.; Lozano, J.; Poh, Q.H.; Greening, D.W. Development of Extracellular Vesicle Therapeutics: Challenges, Considerations, and Opportunities. Front. Cell Dev. Biol. 2021, 9, 734720. [Google Scholar] [CrossRef] [PubMed]
- de Almeida Fuzeta, M.; Gonçalves, P.P.; Fernandes-Platzgummer, A.; Cabral, J.M.S.; Bernardes, N.; da Silva, C.L. From Promise to Reality: Bioengineering Strategies to Enhance the Therapeutic Potential of Extracellular Vesicles. Bioengineering 2022, 9, 675. [Google Scholar] [CrossRef] [PubMed]
- Nelson, B.C.; Maragh, S.; Ghiran, I.C.; Jones, J.C.; Derose, P.C.; Elsheikh, E.; Vreeland, W.N.; Wang, L. Measurement and Standardization Challenges for Extracellular Vesicle Therapeutic Delivery Vectors. Nanomedicine 2020, 15, 2149–2170. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Naranjo, J.C.; Wu, H.J.; Ugaz, V.M. Microfluidics for Exosome Isolation and Analysis: Enabling Liquid Biopsy for Personalized Medicine. Lab. Chip 2017, 17, 3558. [Google Scholar] [CrossRef] [PubMed]
- Piffoux, M.; Volatron, J.; Cherukula, K.; Aubertin, K.; Wilhelm, C.; Silva, A.K.; Gazeau, F. Engineering and loading therapeutic extracellular vesicles for clinical translation: A data reporting frame for comparability. Adv. Drug Deliv. Rev. 2021, 178, 113972. [Google Scholar] [CrossRef] [PubMed]
| Agent | Category | Target/Mechanism | Preclinical Efficacy | Toxicity Profile | Delivery Efficiency | Major Knowledge Gaps | Citation |
|---|---|---|---|---|---|---|---|
| GW4869 | Inhibitor | Neutral sphingomyelinase inhibitor | IC50: 1–5 μM; 60–80% EV reduction | MTD: 25 mg/kg; hepatotoxicity at high doses | Limited BBB penetration (<10%) | Long-term effects unknown; EV subtype specificity unclear | [102] |
| Imipramine | Inhibitor | Endolysosomal disruption | EC50: 10–20 μM; 70% exosome inhibition | Established safety profile (FDA-approved) | Moderate CNS penetration (40%) | Mechanism selectivity questioned; off-target effects | [103] |
| Monensin | Enhancer | Golgi pH alteration | 2–4-fold EV release increase | Narrow therapeutic window; cardiotoxicity risk | Variable tissue distribution | Optimal dosing protocols are undefined; reversibility is unknown | [104] |
| Forskolin | Enhancer | cAMP pathway activation | 3–6-fold EV secretion enhancement | Generally well-tolerated; hypotension risk | Good systemic bioavailability | EV cargo quality effects are unstudied; duration of action variable | [105] |
| Dynasore | Inhibitor | Dynamin inhibition | 80–90% uptake blockade | Cytotoxicity at >50 μM; cell viability concerns | Poor in vivo stability | Non-specific endocytosis effects; delivery challenges | [106] |
| UBL3 modulators | Modulator | S-prenylation control | 2–5-fold cargo enrichment | Preliminary safety data only | Targeted EV loading | Early-stage development; clinical translation unclear | [50,64] |
| Trial Name | Phase | Indication | EV Source | Primary Outcome | Status/Results | Identifier |
|---|---|---|---|---|---|---|
| AGLE-102 | II | Diabetic Macular Edema | Allogeneic MSCs | Visual acuity improvement | 65% patients showed ≥15 letter improvement | NCT04213248 |
| ExoFlo | II | COVID-19 ARDS | Bone marrow MSCs | Mortality reduction | Mixed results; 15% mortality reduction (not significant) | NCT04493242 |
| EXOSTEM | I/II | Spinal Cord Injury | Autologous MSCs | Safety/neurological recovery | Safe profile; 40% functional improvement | NCT04202770 |
| MDSTEMCELL | I | Macular Degeneration | Retinal pigment epithelium | Visual function | Ongoing; preliminary safety confirmed | NCT04173650 |
| ARISE-2 | II | Acute Kidney Injury | Allogeneic MSCs | Renal function recovery | 58% reduction in dialysis requirement | NCT04134676 |
| NEUROSTEM | I/II | Alzheimer’s Disease | Neural stem cells | Cognitive assessment | Ongoing; early biomarker improvements | NCT04528641 |
| CARDIO-EXO | I | Heart Failure | Cardiac progenitor cells | Cardiac function | 32% improvement in ejection fraction | NCT04327063 |
| MSC-EXO-COVID | II | COVID-19 | Umbilical cord MSCs | Inflammatory markers | 70% reduction in IL-6 levels | NCT04384445 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mimi, M.A.; Hasan, M.M. Extracellular Vesicles as Mediators of Intercellular Communication: Implications for Drug Discovery and Targeted Therapies. Future Pharmacol. 2025, 5, 48. https://doi.org/10.3390/futurepharmacol5030048
Mimi MA, Hasan MM. Extracellular Vesicles as Mediators of Intercellular Communication: Implications for Drug Discovery and Targeted Therapies. Future Pharmacology. 2025; 5(3):48. https://doi.org/10.3390/futurepharmacol5030048
Chicago/Turabian StyleMimi, Mst. Afsana, and Md. Mahmudul Hasan. 2025. "Extracellular Vesicles as Mediators of Intercellular Communication: Implications for Drug Discovery and Targeted Therapies" Future Pharmacology 5, no. 3: 48. https://doi.org/10.3390/futurepharmacol5030048
APA StyleMimi, M. A., & Hasan, M. M. (2025). Extracellular Vesicles as Mediators of Intercellular Communication: Implications for Drug Discovery and Targeted Therapies. Future Pharmacology, 5(3), 48. https://doi.org/10.3390/futurepharmacol5030048






