Thallium Toxicity: Mechanisms of Action, Available Therapies, and Experimental Models
Abstract
1. Introduction
2. Literature Search Strategy
3. Characteristics of Tl
4. Environmental Contamination by Tl
5. Statistics and Reported Cases of Tl Poisoning in Humans
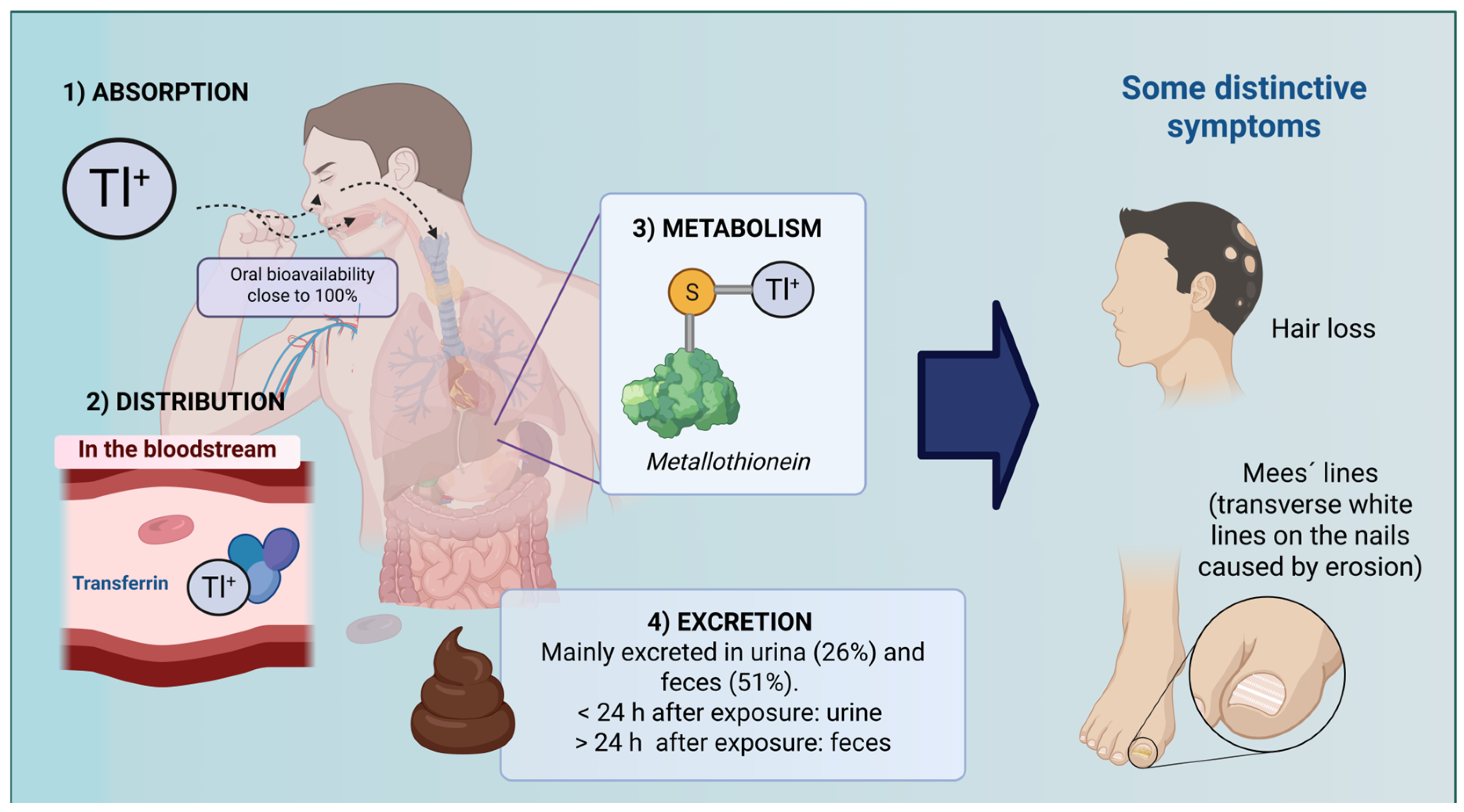
6. Toxicokinetics of Tl
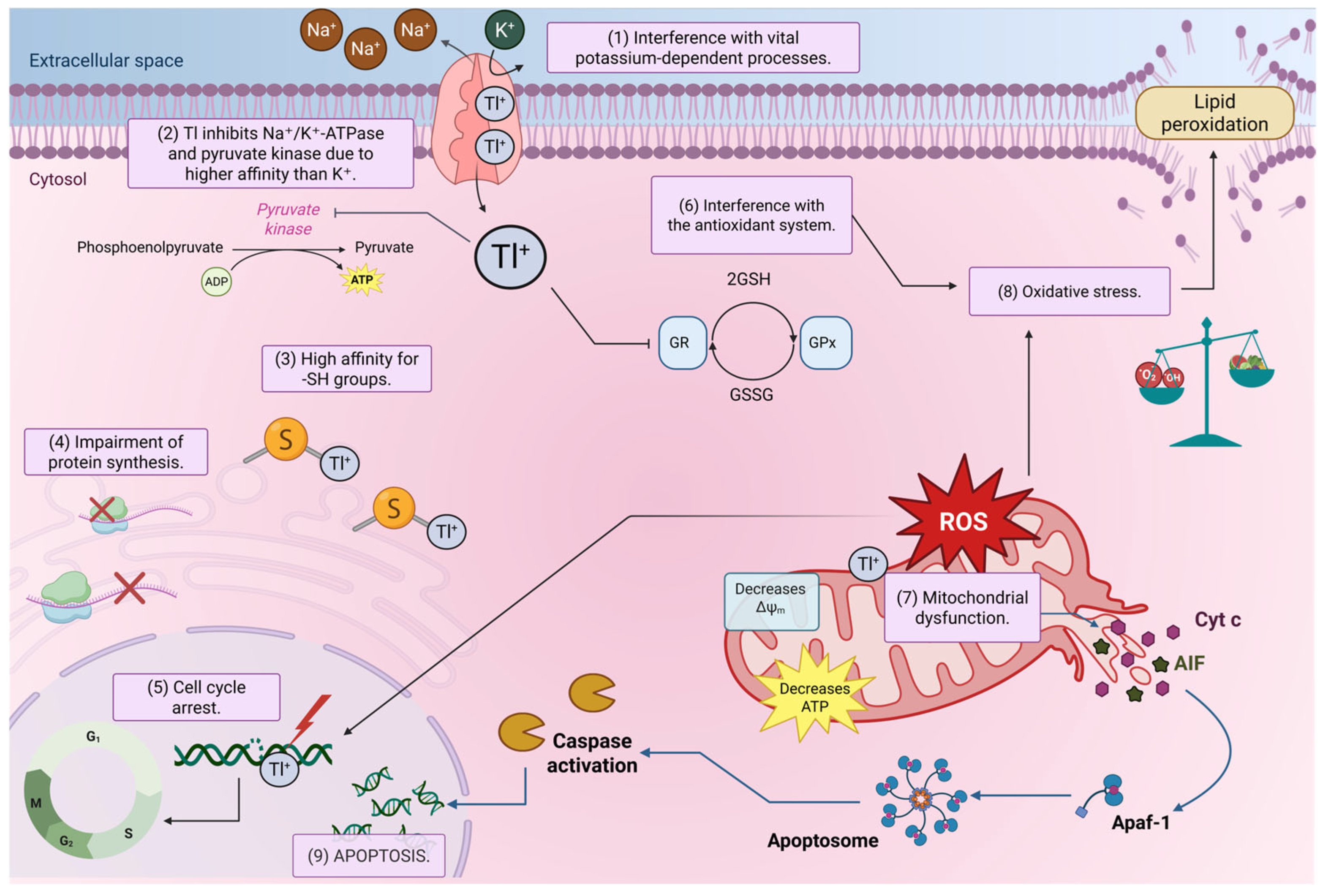
7. Mechanisms of Toxicity
7.1. Cellular Effects
7.2. Effects on Organelles
7.3. Effects on Metabolic Pathways
7.4. Other Mechanisms of Toxic Action
7.4.1. Effect of Tl on Calcium Homeostasis
7.4.2. Effects of Tl on Myelin
7.4.3. Mutagenic and Genotoxic Effects of Tl
8. Treatment of Tl Poisoning in Humans
9. Biological Models for the Study of Tl Poisoning
9.1. Traditional Models: Rodents
9.1.1. Rats
9.1.2. Mice
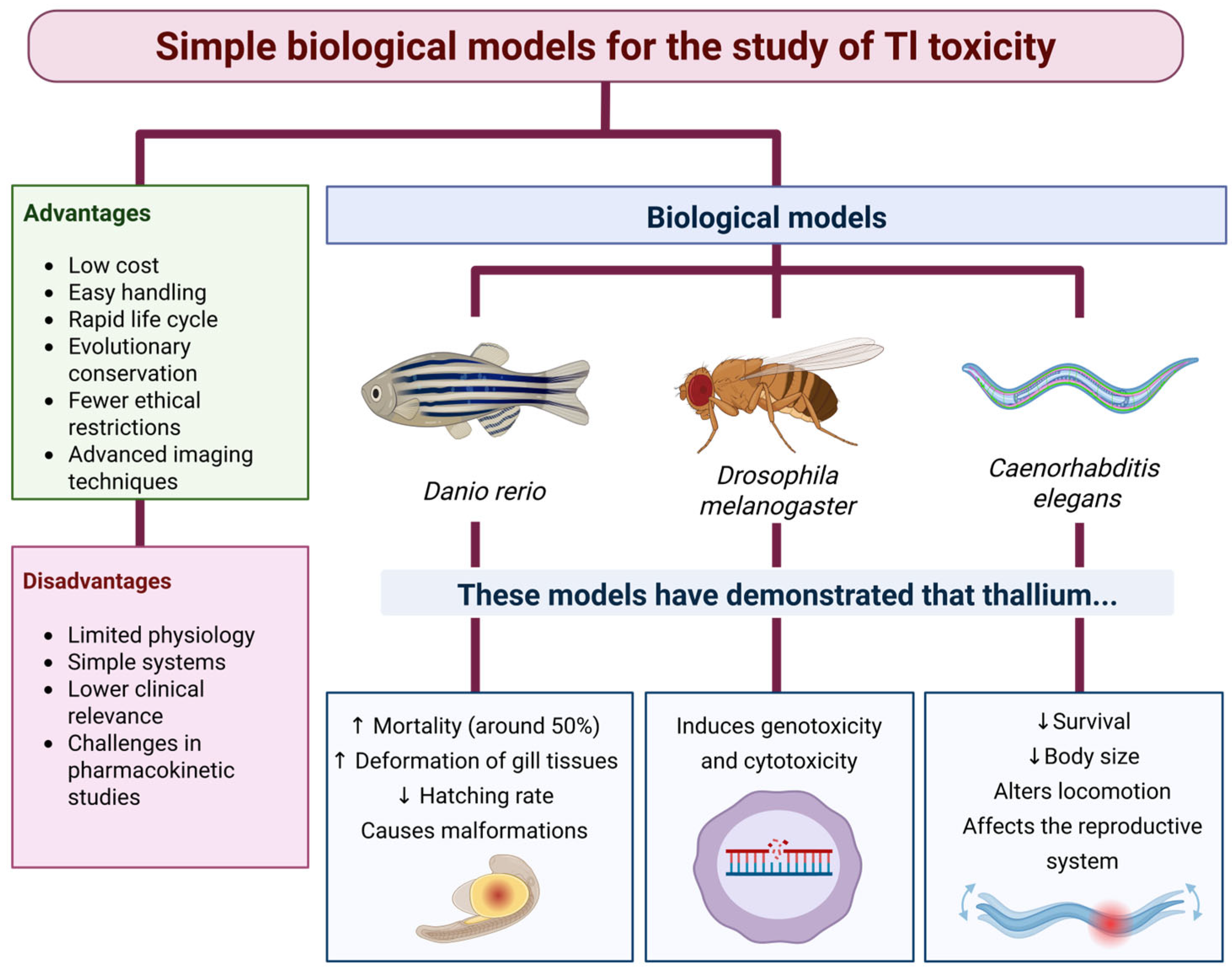
9.2. Simple Biological Models
9.2.1. Zebrafish (D. rerio)
9.2.2. Fruit Fly (D. melanogaster)
9.2.3. Nematode (C. elegans)
9.3. Emerging Models: Organoids and iPSC-Derived Systems
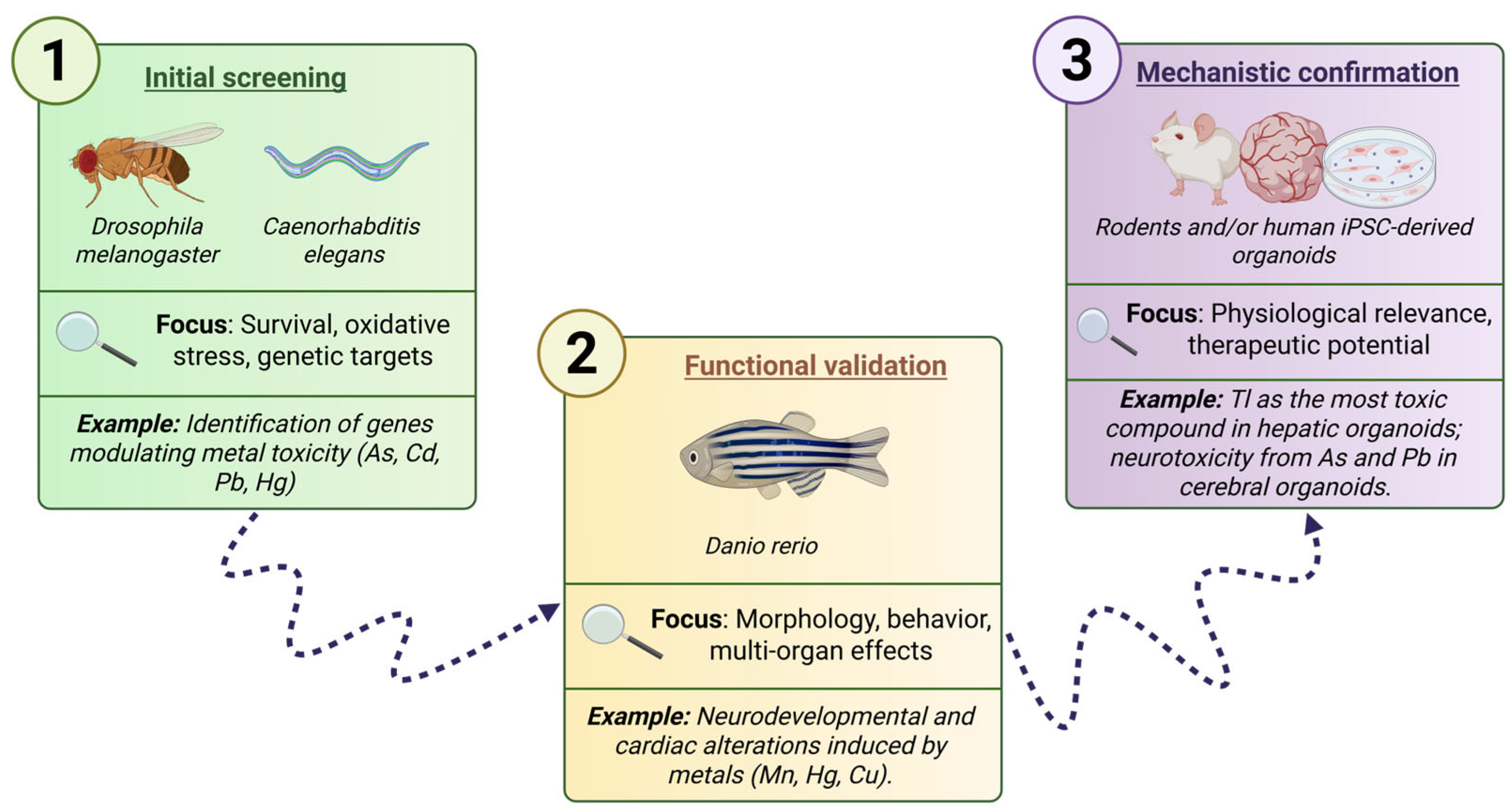
10. Strategic Selection of Biological Models for Tl Toxicity: A Tiered and Complementary Approach
11. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADME | Absorption, Distribution, Metabolism, and Excretion |
| AIF | Apoptosis-Inducing Factor |
| Apaf-1 | Apoptotic Protease Activating Factor 1 |
| ARDS | Acute Respiratory Distress Syndrome |
| ATP | Adenosine Triphosphate |
| CCME | Canadian Council of Ministers of the Environment |
| CDK2 | Cyclin-Dependent Kinase 2 |
| C. elegans | Caenorhabditis elegans |
| CoA | Coenzyme A |
| CVVH | Continuous Veno Venous Hemofiltration |
| Cyt c | Cytochrome c |
| Δψm | Mitochondrial Membrane Potential |
| DEGs | Differentially Expressed Genes |
| DMS | Dimercaptosuccinic Acid |
| EEG | Electroencephalogram |
| Eh | Redox Potential |
| EMG | Electromyography |
| ETC | Electron Transport Chain |
| EPA | Environmental Protection Agency (USA) |
| ER | Endoplasmic Reticulum |
| FDA | Food and Drug Administration (USA) |
| FISH | Fluorescence In Situ Hybridization |
| FRs | Free Radicals |
| GPx | Glutathione Peroxidase |
| GR | Glutathione Reductase |
| GSH | Glutathione |
| GSSG | Oxidized Glutathione (Glutathione Disulfide) |
| GT1-7 | Immortalized Neuronal Cell Line from Mouse Hypothalamus |
| i.p. | Intraperitoneal (route of administration) |
| iPSC | Induced Pluripotent Stem Cell |
| K+ | Potassium Ion |
| LD90–LD100 | Lethal Dose for 90–100% of Animals |
| MDA | Malondialdehyde |
| MDCKs | Madin–Darby Canine Kidney Cells |
| MK-801 | NMDA Receptor Antagonist (Dizocilpine) |
| mPTP | Mitochondrial Permeability Transition Pore |
| MT | Metallothionein |
| NAC | N-acetylcysteine |
| Na+ | Sodium Ion |
| NPDSs | National Poison Data System (USA) |
| NOM | Norma Oficial Mexicana (Mexican Official Standard) |
| PBGS | Porphobilinogen Synthase |
| PDH | Pyruvate Dehydrogenase |
| ROS | Reactive Oxygen Species |
| SOD | Superoxide Dismutase |
| SDH | Succinate Dehydrogenase |
| SMART | Somatic Mutation and Recombination Test |
| Tl | Thallium |
| Tl+ | Thallium Ion in +1 Oxidation State |
| Tl3+ | Thallium Ion in +3 Oxidation State |
| TlCH3COO | Thallium Acetate |
| TID | Ter In Die. |
| TlNO3 | Thallium Nitrate |
| Tl2SO4 | Thallium Sulfate |
| USGS | United States Geological Survey |
| WHO | World Health Organization |
References
- Duan, W.; Wang, Y.; Li, Z.; Fu, G.; Mao, L.; Song, Y.; Qu, Y.; Ye, L.; Zhou, Q.; Yang, F.; et al. Thallium Exposure at Low Concentration Leads to Early Damage on Multiple Organs in Children: A Case Study Followed-up for Four Years. Environ. Pollut. 2020, 258, 113319. [Google Scholar] [CrossRef]
- Peter, A.L.J.; Viraraghavan, T. Thallium: A Review of Public Health and Environmental Concerns. Environ. Int. 2005, 31, 493–501. [Google Scholar] [CrossRef]
- Karbowska, B. Presence of Thallium in the Environment: Sources of Contaminations, Distribution and Monitoring Methods. Environ. Monit. Assess. 2016, 188, 640. [Google Scholar] [CrossRef] [PubMed]
- Soave, P.M.; Chirico, F.; Pallocchi, M.; Magnavita, N. Thallium Poisoning: Case Report and Scoping Review on Diagnostic Delay and Therapeutic Outcome. Appl. Sci. 2025, 15, 1732. [Google Scholar] [CrossRef]
- Fujihara, J.; Nishimoto, N. Thallium—Poisoner’s Poison: An Overview and Review of Current Knowledge on the Toxicological Effects and Mechanisms. Curr. Res. Toxicol. 2024, 6, 100157. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, R.S. Thallium Toxicity and the Role of Prussian Blue in Therapy. Toxicol. Rev. 2003, 22, 29–40. [Google Scholar] [CrossRef]
- Chang, Y.; Tsai, J.-F.; Chen, P.-J.; Huang, Y.-T.; Liu, B.-H. Thallium Exposure Interfered with Heart Development in Embryonic Zebrafish (Danio Rerio): From Phenotype to Genotype. Sci. Total Environ. 2023, 878, 162901. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Li, X.; Liu, P.; Li, L.; Chen, H.; Li, D.; Liu, J.; Xie, L. Integrated Physiological, Biochemical, and Transcriptomic Analysis of Thallium Toxicity in Zebrafish (Danio Rerio) Larvae. Sci. Total Environ. 2023, 859, 160265. [Google Scholar] [CrossRef] [PubMed]
- Pitchakarn, P.; Inthachat, W.; Karinchai, J.; Temviriyanukul, P. Human Hazard Assessment Using Drosophila Wing Spot Test as an Alternative In Vivo Model for Genotoxicity Testing—A Review. Int. J. Mol. Sci. 2021, 22, 9932. [Google Scholar] [CrossRef]
- Hurtado-Díaz, M.E.; Estrada-Valencia, R.; Rangel-López, E.; Maya-López, M.; Colonnello, A.; Galván-Arzate, S.; Verstraeten, S.V.; Karasu, C.; Túnez, I.; Aschner, M.; et al. Thallium Toxicity in Caenorhabditis elegans: Involvement of the SKN-1 Pathway and Protection by S-Allylcysteine. Neurotox. Res. 2020, 38, 287–298. [Google Scholar] [CrossRef]
- Wu, X.; Chen, Y.; Kreutz, A.; Silver, B.; Tokar, E.J. Pluripotent Stem Cells for Target Organ Developmental Toxicity Testing. Toxicol. Sci. 2024, 199, 163–171. [Google Scholar] [CrossRef]
- Genchi, G.; Carocci, A.; Lauria, G.; Sinicropi, M.S.; Catalano, A. Thallium Use, Toxicity, and Detoxification Therapy: An Overview. Appl. Sci. 2021, 11, 8322. [Google Scholar] [CrossRef]
- Unites States Environmental Protection Agency (US EPA). Toxic and Priority Pollutants under the Clean Water Act. Available online: https://www.epa.gov/eg/toxic-and-priority-pollutants-under-clean-water-act (accessed on 16 January 2024).
- Clarkson, T.W. Inorganic and Organometal Pesticides. In Handbook of Pesticide Toxicology; Elsevier: Amsterdam, The Netherlands, 2001; pp. 1357–1428. [Google Scholar]
- Ingram, J.T. Thallium Acetate In The Treatment Of Ringworm Of The Scalp. BMJ 1932, 1, 8–26. [Google Scholar] [CrossRef] [PubMed]
- United States Geological Survey Thallium Statistics and Information. Available online: https://www.usgs.gov/centers/national-minerals-information-center/thallium-statistics-and-information (accessed on 10 August 2025).
- Turetta, C.; Barbante, C.; Capodaglio, G.; Gambaro, A.; Cescon, P. The Distribution of Dissolved Thallium in the Different Water Masses of the Western Sector of the Ross Sea (Antarctica) during the Austral Summer. Microchem. J. 2010, 96, 194–202. [Google Scholar] [CrossRef]
- Li, S.; Xue, W.; Gao, N.; Niu, X.; Wang, T.; Zhu, B.; Zhang, X.; Wang, Z. A Multi-Biomarker Approach to Study the Toxic Effects of Thallium on Earthworms (Eisenia Fetida) Fed with Different Food Sources. Appl. Soil. Ecol. 2025, 213, 106246. [Google Scholar] [CrossRef]
- Xiao, T.; Guha, J.; Boyle, D.; Liu, C.-Q.; Chen, J. Environmental Concerns Related to High Thallium Levels in Soils and Thallium Uptake by Plants in Southwest Guizhou, China. Sci. Total Environ. 2004, 318, 223–244. [Google Scholar] [CrossRef]
- Migaszewski, Z.M.; Gałuszka, A. Abundance and Fate of Thallium and Its Stable Isotopes in the Environment. Rev. Environ. Sci. Biotechnol. 2021, 20, 5–30. [Google Scholar] [CrossRef]
- Wierzbicka, M.; Szarek-Łukaszewska, G.; Grodzińska, K. Highly Toxic Thallium in Plants from the Vicinity of Olkusz (Poland). Ecotoxicol. Environ. Saf. 2004, 59, 84–88. [Google Scholar] [CrossRef]
- Liu, J.; Wang, J.; Tsang, D.C.W.; Xiao, T.; Chen, Y.; Hou, L. Emerging Thallium Pollution in China and Source Tracing by Thallium Isotopes. Environ. Sci. Technol. 2018, 52, 11977–11979. [Google Scholar] [CrossRef]
- Daye Municipal People’s Government. Statistical Bulletin on the Economic and Social Development of Agriculture and Rural Areas in Daye in 2024. Available online: https://hbdaye.gov.cn/zfxxgk/dfbmptlj/szbm/dysnyncjjgsz/fdzdgknr_16507/tjxx_20101/202505/t20250506_1219064.html (accessed on 17 June 2025).
- Wang, J.; Zhou, Y.; Dong, X.; Yin, M.; Tsang, D.C.W.; Sun, J.; Liu, J.; Song, G.; Liu, Y. Temporal Sedimentary Record of Thallium Pollution in an Urban Lake: An Emerging Thallium Pollution Source from Copper Metallurgy. Chemosphere 2020, 242, 125172. [Google Scholar] [CrossRef] [PubMed]
- EWG’s Tap Water Database. Thallium. Available online: https://www.ewg.org/tapwater/contaminant.php?contamcode=1085&utm_source=chatgpt.com (accessed on 2 July 2025).
- Canadian Council of Ministers of the Environment. Thallium. Available online: https://ccme.ca/en/chemical/211 (accessed on 2 July 2025).
- Azara, A.; Castiglia, P.; Piana, A.; Masia, M.D.; Palmieri, A.; Arru, B.; Maida, G.; Dettori, M. Derogation from Drinking Water Quality Standards in Italy According to the European Directive 98/83/EC and the Legislative Decree 31/2001—a Look at the Recent Past. Ann. Ig. 2018, 30, 517–526. [Google Scholar] [CrossRef]
- Xia, Y.; Liao, G.; Wang, Z.; Wei, Y.; Liu, H.; Tang, Y.; Tang, H.; Liu, X.; Shi, J.; Liu, C. Efficient and Deep Adsorption of Thallium(I) from Complex Water Based on Hard-Soft Acid-Base Theory. Sep. Purif. Technol. 2025, 360, 131221. [Google Scholar] [CrossRef]
- SEMARNAT NORMA Oficial Mexicana NOM-147-SEMARNAT/SSA1-2004. Available online: https://www.dof.gob.mx/nota_detalle.php?codigo=4964569&fecha=02/03/2007#gsc.tab=0 (accessed on 26 August 2025).
- Liu, J.; Yuan, W.; Lin, K.; Wang, J.; Sonne, C.; Rinklebe, J. Thallium Pollution from the Lithium Industry Calls for Urgent International Action on Regulations. Environ. Sci. Technol. 2023, 57, 19099–19101. [Google Scholar] [CrossRef]
- Antić-Mladenović, S.; Frohne, T.; Kresović, M.; Stärk, H.-J.; Savić, D.; Ličina, V.; Rinklebe, J. Redox-Controlled Release Dynamics of Thallium in Periodically Flooded Arable Soil. Chemosphere 2017, 178, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.; House, I.; Dixon, A. Thallium Poisoning. Diagnosis May Be Elusive but Alopecia Is the Clue. BMJ 1993, 306, 1527–1529. [Google Scholar] [CrossRef]
- Kemnic, T.R.; Coleman, M. Thallium Toxicity. Available online: https://www.ncbi.nlm.nih.gov/books/NBK513240/ (accessed on 6 August 2025).
- Gummin, D.D.; Mowry, J.B.; Beuhler, M.C.; Spyker, D.A.; Rivers, L.J.; Feldman, R.; Brown, K.; Pham, N.P.T.; Bronstein, A.C.; DesLauriers, C. 2023 Annual Report of the National Poison Data System® (NPDS) from America’s Poison Centers®: 41st Annual Report. Clin. Toxicol. 2024, 62, 793–1027. [Google Scholar] [CrossRef] [PubMed]
- Gummin, D.D.; Mowry, J.B.; Beuhler, M.C.; Spyker, D.A.; Brooks, D.E.; Dibert, K.W.; Rivers, L.J.; Pham, N.P.T.; Ryan, M.L. 2019 Annual Report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 37th Annual Report. Clin. Toxicol. 2020, 58, 1360–1541. [Google Scholar] [CrossRef]
- Jimenez, O.; Cáceres, H.; Gimenez, L.; Soto, L.; Montenegro, M.; Rueda, J.A.A. Thallium Poisoning: A Case Report. J. Yeungnam Med. Sci. 2022, 40, 311–314. [Google Scholar] [CrossRef]
- Li, J.M.; Wang, W.; Lei, S.; Zhao, L.L.; Zhou, D.; Xiong, H. Misdiagnosis and Long-Term Outcome of 13 Patients with Acute Thallium Poisoning in China. Clin. Toxicol. 2014, 52, 181–186. [Google Scholar] [CrossRef]
- Sun, T.-W.; Xu, Q.-Y.; Zhang, X.-J.; Wu, Q.; Liu, Z.-S.; Kan, Q.-C.; Sun, C.-Y.; Wang, L. Management of Thallium Poisoning in Patients with Delayed Hospital Admission. Clin. Toxicol. 2012, 50, 65–69. [Google Scholar] [CrossRef]
- Ash, R.D.; He, M. Details of a Thallium Poisoning Case Revealed by Single Hair Analysis Using Laser Ablation Inductively Coupled Plasma Mass Spectrometry. Forensic Sci. Int. 2018, 292, 224–231. [Google Scholar] [CrossRef]
- Rangel- Guerra, R.; Martínez, H.R.; Villarreal, H.J.; Montoya Cabrera, M.A. Intoxicación Por Talio. Experiencia Con 50 Pacientes. Gac. Med. Mex. 1990, 126, 487–495. [Google Scholar]
- Yang, G.; Li, C.; Long, Y.; Sheng, L. Hair Loss: Evidence to Thallium Poisoning. Case Rep. Emerg. Med. 2018, 2018, 1–3. [Google Scholar] [CrossRef]
- Tsai, Y.-T.; Huang, C.-C.; Kuo, H.-C.; Wang, H.-M.; Shen, W.-S.; Shih, T.-S.; Chu, N.-S. Central Nervous System Effects in Acute Thallium Poisoning. Neurotoxicology 2006, 27, 291–295. [Google Scholar] [CrossRef]
- Munch, J.C.; Olden, G.; Ginsburg, H.M.; Nixon, C. The 1932 Thallotoxicosis Outbreak in California. JAMA J. Am. Med. Assoc. 1933, 100, 1315–1319. [Google Scholar] [CrossRef]
- Huang, C.; Zhang, X.; Li, G.; Jiang, Y.; Wang, Q.; Tian, R. A Case of Severe Thallium Poisoning Successfully Treated with Hemoperfusion and Continuous Veno-Venous Hemofiltration. Hum. Exp. Toxicol. 2014, 33, 554–558. [Google Scholar] [CrossRef]
- Spadaro, A.; Lee, A.S.Y.; Pineda, H.; Ruck, B.; Calello, D.P.; Greller, H.A.; Nelson, L.S.; Parris, M.A. Attempted Self-Harm with Elemental Thallium Purchased Online: Case Report with Analytical Confirmation. J. Med. Toxicol. 2024, 20, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Hirata, M.; Taoda, A.; Ono-Ogasawara, M.; Takaya, M.; Hisanaga, N. A Probable Case of Chronic Occupational Thallium Poisoning in a Glass Factory. Ind. Health 1998, 36, 300–303. [Google Scholar] [CrossRef]
- Villanueva, E.; Hernandez-Cueto, C.; Lachica, E.; Rodrigo, M.D.; Ramos, V. Poisoning by Thallium A Study of Five Cases. Drug Saf. 1990, 5, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Salas Pérez, G. Intoxicación Por Talio: Reporte de 5 Casos. Trabajo de Grado de Especialización; Universidad Nacional Autónoma de México: Mexico, Mexico, 1993. [Google Scholar]
- Rodríguez-Mercado, J.J.; Altamirano-Lozano, M.A. Genetic Toxicology of Thallium: A Review. Drug Chem. Toxicol. 2013, 36, 369–383. [Google Scholar] [CrossRef] [PubMed]
- Mulkey, J.P.; Oehme, F.W. A Review of Thallium Toxicity. Vet. Hum. Toxicol. 1993, 35, 445–453. [Google Scholar]
- Ren, X.; Feng, H.; Zhao, M.; Zhou, X.; Zhu, X.; Ouyang, X.; Tang, J.; Li, C.; Wang, J.; Tang, W.; et al. Recent Advances in Thallium Removal from Water Environment by Metal Oxide Material. Int. J. Environ. Res. Public. Health 2023, 20, 3829. [Google Scholar] [CrossRef]
- Liu, Y.; Wei, L.; Luo, D.; Xiao, T.; Lekhov, A.; Xie, X.; Huang, X.; Su, X. Geochemical Distribution and Speciation of Thallium in Groundwater Impacted by Acid Mine Drainage (Southern China). Chemosphere 2021, 280, 130743. [Google Scholar] [CrossRef]
- Asadoulahi, T.; Dadfarnia, S.; Shabani, A.M.H. Determination of Thallium Traces by ETAAS after On-Line Matrix Separation and Preconcentration in a Flow Injection System. J. Braz. Chem. Soc. 2007, 18, 1353–1359. [Google Scholar] [CrossRef]
- De Groot, G.; Van Heijst, A.N.P. Toxicokinetic Aspects of Thallium Poisoning. Methods of Treatment by Toxin Elimination. Sci. Total Environ. 1988, 71, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Ríos, C.; Galván-Arzate, S.; Tapia, R. Brain Regional Thallium Distribution in Rats Acutely Intoxicated with Tl2SO4. Arch. Toxicol. 1989, 63, 34–37. [Google Scholar] [CrossRef]
- Galván-Arzate, S.; Martínez, A.; Medina, E.; Santamaría, A.; Ríos, C. Subchronic Administration of Sublethal Doses of Thallium to Rats: Effects on Distribution and Lipid Peroxidation in Brain Regions. Toxicol. Lett. 2000, 116, 37–43. [Google Scholar] [CrossRef]
- Hoffman, R.S.; Hoffman, R. Thallium Poisoning During Pregnancy: A Case Report and Comprehensive Literature Review. J. Toxicol. Clin. Toxicol. 2000, 38, 767–775. [Google Scholar] [CrossRef]
- Nordberg, M.; Nordberg, G.F. Toxicological Aspects of Metallothionein. Cell Mol. Biol. (Noisy-Le-Grand) 2000, 46, 451–463. [Google Scholar]
- Sugiura, T.; Yamashita, U. B Cell Stimulating Activity of Metallothionein in Vitro. Int. J. Immunopharmacol. 2000, 22, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Kılıç, G.A.; Kutlu, M. Effects of Exogenous Metallothionein against Thallium-Induced Oxidative Stress in Rat Liver. Food Chem. Toxicol. 2010, 48, 980–987. [Google Scholar] [CrossRef]
- Favari, L.; Mourelle, M. Thallium Replaces Potassium in Activation of the (Na +, K +)-atpase of Rat Liver Plasma Membranes. J. Appl. Toxicol. 1985, 5, 32–34. [Google Scholar] [CrossRef]
- Britten, J.S.; Blank, M. Thallium Activation of the (Na+-K+)-Activated ATPase of Rabbit Kidney. Biochim. Et. Biophys. Acta (BBA) Enzymol. 1968, 159, 160–166. [Google Scholar] [CrossRef]
- Hanzel, C.E.; Villaverde, M.S.; Verstraeten, S.V. Glutathione Metabolism Is Impaired in Vitro by Thallium(III) Hydroxide. Toxicology 2005, 207, 501–510. [Google Scholar] [CrossRef]
- Villaverde, M.S.; Hanzel, C.E.; Verstraeten, S.V. In Vitro Interactions of Thallium with Components of the Glutathione-Dependent Antioxidant Defence System. Free Radic. Res. 2004, 38, 977–984. [Google Scholar] [CrossRef]
- Verstraeten, S.V. Relationship between Thallium(I)-Mediated Plasma Membrane Fluidification and Cell Oxidants Production in Jurkat T Cells. Toxicology 2006, 222, 95–102. [Google Scholar] [CrossRef]
- Villaverde, M.S.; Verstraeten, S. V Effects of Thallium(I) and Thallium(III) on Liposome Membrane Physical Properties. Arch. Biochem. Biophys. 2003, 417, 235–243. [Google Scholar] [CrossRef]
- Chia, C.; Chen, S.; Chen, C.; Shih, C.; Lee, H.; Wu, C. Thallium Acetate Induces C6 Glioma Cell Apoptosis. Ann. N. Y. Acad. Sci. 2005, 1042, 523–530. [Google Scholar] [CrossRef] [PubMed]
- López-Lanuza, A.; Álvarez-Barrera, L.; López-Muñoz, H.; Mateos-Nava, R.A.; Rodríguez-Mercado, J.J. Thallium (III) Disrupts the Cell Cycle and Induces Oxidative DNA Damage in Human Lymphocytes in Vitro. Curr. Res. Toxicol. 2025, 8, 100240. [Google Scholar] [CrossRef] [PubMed]
- Nikitina, E.R.; Glazunov, V.V. Involvement of K+–ATP-Dependent Channel in Transport of Monovalent Thallium (Tl+) across the Inner Membrane of Rat Liver Mitochondria. Dokl. Biochem. Biophys. 2003, 392, 244–246. [Google Scholar] [CrossRef]
- Pourahmad, J.; Eskandari, M.R.; Daraei, B. A Comparison of Hepatocyte Cytotoxic Mechanisms for Thallium (I) and Thallium (III). Environ. Toxicol. 2010, 25, 456–467. [Google Scholar] [CrossRef] [PubMed]
- Hanzel, C.; Verstraeten, S. Thallium Induces Hydrogen Peroxide Generation by Impairing Mitochondrial Function. Toxicol. Appl. Pharmacol. 2006, 216, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Eskandari, M.R.; Mashayekhi, V.; Aslani, M.; Hosseini, M. Toxicity of Thallium on Isolated Rat Liver Mitochondria: The Role of Oxidative Stress and MPT Pore Opening. Environ. Toxicol. 2015, 30, 232–241. [Google Scholar] [CrossRef]
- Korotkov, S.M.; Nesterov, V.P.; Sobol, K.V. The Effects of Thallium on the Spontaneous Contraction of the Heart Muscle and the Energetic Processes in Cardiomyocyte Mitochondria. Biophysics 2019, 64, 777–785. [Google Scholar] [CrossRef]
- Hultin, T.; Näslund, P.H. Effects of Thallium (I) on the Structure and Functions of Mammalian Ribosomes. Chem. Biol. Interact. 1974, 8, 315–328. [Google Scholar] [CrossRef]
- Chou, Y.-T.; Lo, K.-Y. Thallium(I) Treatment Induces Nucleolar Stress to Stop Protein Synthesis and Cell Growth. Sci. Rep. 2019, 9, 6905. [Google Scholar] [CrossRef]
- Morel Gómez, E.; Casali, C.I.; Fernández, M.d.C.; Verstraeten, S.V. Tl(I) and Tl(III) Induce Reticulum Stress in MDCK Cells. Environ. Toxicol. Pharmacol. 2023, 101, 104192. [Google Scholar] [CrossRef]
- Woods, J.S.; Fowler, B.A. Alteration of Hepatocellular Structure and Function by Thallium Chloride: Ultrastructural, Morphometric, and Biochemical Studies. Toxicol. Appl. Pharmacol. 1986, 83, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Ozgul Ozalp, F.; Kutlu, M.; Iscan, A. The Effects of Thallium Acetate on Hepatopancreatic Cells of Gammarus Pulex (Crustacea: Amphipoda). Ekoloji 2011, 20, 15–20. [Google Scholar] [CrossRef]
- Kayne, F.J. Thallium (I) Activation of Pyruvate Kinase. Arch. Biochem. Biophys. 1971, 143, 232–239. [Google Scholar] [CrossRef]
- Rocha, J. Effect of Group 13 Metals on Porphobilinogen Synthase in Vitro. Toxicol. Appl. Pharmacol. 2004, 200, 169–176. [Google Scholar] [CrossRef]
- Nava- Ruiz, C.; Méndez- Armenta, M. Efectos Neurotóxicos de Metales Pesados (Cadmio, Plomo, Arsénico y Talio). Arch. De. Neurocienc. 2011, 16, 140–147. [Google Scholar]
- Bramanti, E.; Onor, M.; Colombaioni, L. Neurotoxicity Induced by Low Thallium Doses in Living Hippocampal Neurons: Evidence of Early Onset Mitochondrial Dysfunction and Correlation with Ethanol Production. ACS Chem. Neurosci. 2019, 10, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Korotkov, S.M. Mitochondrial Oxidative Stress Is the General Reason for Apoptosis Induced by Different-Valence Heavy Metals in Cells and Mitochondria. Int. J. Mol. Sci. 2023, 24, 14459. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Guan, A.; Huo, Z.; Li, X.; Zhu, Y.; Liang, H.; Liu, W.; Zhou, H.; Lin, Z.; Yan, B. Distinct Neurotoxic Mechanisms of Thallium and Lead: Calcium-Mediated Apoptosis and Iron-Induced Ferroptosis in Zebrafish at Environmental Concentrations. J. Hazard. Mater. 2025, 492, 138288. [Google Scholar] [CrossRef]
- Unuma, K.; Wen, S.; Sugahara, S.; Nagano, S.; Aki, T.; Ogawa, T.; Takeda-Homma, S.; Oikawa, M.; Tojo, A. Thallium Reabsorption via NKCC2 Causes Severe Acute Kidney Injury with Outer Medulla-Specific Calcium Crystal Casts in Rats. Arch. Toxicol. 2024, 98, 3973–3986. [Google Scholar] [CrossRef]
- Davis, L.E.; Standefer, J.C.; Kornfeld, M.; Abercrombie, D.M.; Butler, C. Acute Thallium Poisoning: Toxicological and Morphological Studies of the Nervous System. Ann. Neurol. 1981, 10, 38–44. [Google Scholar] [CrossRef]
- Misra, U.K.; Kalita, J.; Yadav, R.K.; Ranjan, P. Thallium Poisoning: Emphasis on Early Diagnosis and Response to Haemodialysis. Postgrad. Med. J. 2003, 79, 103–105. [Google Scholar] [CrossRef]
- Spencer, P.S.; Peterson, E.R.; Madrid, A.R.; Raine, C.S. Effects of Thallium Salts on Neuronal Mitochondria in Organotypic Cord-Ganglia-Muscle Combination Cultures. J. Cell Biol. 1973, 58, 79–95. [Google Scholar] [CrossRef]
- Windebank, A.J. Specific Inhibition of Myelination by Lead in Vitro; Comparison with Arsenic, Thallium, and Mercury. Exp. Neurol. 1986, 94, 203–212. [Google Scholar] [CrossRef]
- Barroso-Moguel, R.; Mendez-Armenta, M.; Villeda-Hernandez, J.; Rios, C.; Galvan-Arzate, S. Experimental Neuromyopathy Induced by Thallium in Rats. J. Appl. Toxicol. 1996, 16, 385–389. [Google Scholar] [CrossRef]
- Kanematsu, N.; Hara, M.; Kada, T. Rec Assay and Mutagenicity Studies on Metal Compounds. Mutat. Res. /Genet. Toxicol. 1980, 77, 109–116. [Google Scholar] [CrossRef]
- Zasukhina, G.D.; Vasilyeva, I.M.; Sdirkova, N.I.; Krasovsky, G.N.; Vasyukovich, L.Y.; Kenesariev, U.I.; Butenko, P.G. Mutagenic Effect of Thallium and Mercury Salts on Rodent Cells with Different Repair Activities. Mutat. Res. /Genet. Toxicol. 1983, 124, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Mercado, J.J.; Hernández-de la Cruz, H.; Felipe-Reyes, M.; Jaramillo-Cruz, E.; Altamirano-Lozano, M.A. Evaluation of Cytogenetic and DNA Damage Caused by Thallium(I) Acetate in Human Blood Cells. Environ. Toxicol. 2015, 30, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Nikiforov Alexey, S.N. Cytogenetic Investigation of Thallium-Poisoned People: Pilot Study. J. Toxicol. Environ. Health A 1999, 58, 465–468. [Google Scholar] [CrossRef] [PubMed]
- Migliore, L.; Cocchi, L.; Nesti, C.; Sabbioni, E. Micronuclei Assay and FISH Analysis in Human Lymphocytes Treated with Six Metal Salts. Environ. Mol. Mutagen. 1999, 34, 279–284. [Google Scholar] [CrossRef]
- Vrij, A. Successful Recovery of a Patient with Thallium Poisoning. Neth. J. Med. 1995, 47, 121–126. [Google Scholar] [CrossRef]
- Thompson, D.F.; Callen, E.D. Soluble or Insoluble Prussian Blue for Radiocesium and Thallium Poisoning? Ann. Pharmacother. 2004, 38, 1509–1514. [Google Scholar] [CrossRef]
- Silberman, J.; Galuska, M.A.; Taylor, A. Activated Charcoal. Available online: https://www.ncbi.nlm.nih.gov/books/NBK482294/ (accessed on 26 August 2025).
- Wallbridge, T.; James, S.; Lee, R.; Khan, A.; Bradberry, S.; Elamin, M.E.M.O. Successful Treatment of Potentially Lethal Dose Thallium Sulfate Poisoning with Sequential Use of Prussian Blue and Multiple-Dose Activated Charcoal. Clin. Toxicol. 2023, 61, 200–201. [Google Scholar] [CrossRef]
- Zimmerman, J.L.; Rudis, M. Poisonings. In Critical Care Medicine; Elsevier: Amsterdam, The Netherlands, 2008; pp. 1453–1474. [Google Scholar]
- Ghannoum, M.; Gosselin, S. Enhanced Poison Elimination in Critical Care. Adv. Chronic Kidney Dis. 2013, 20, 94–101. [Google Scholar] [CrossRef]
- Nogué, S.; Mas, A.; Parés, A.; Nadal, P.; Bertrán, A.; Millá, J.; Carrera, M.; To, J.; Pazos, M.R.; Corbella, J. Acute Thallium Poisoning: An Evaluation of Different Forms of Treatment. J. Toxicol. Clin. Toxicol. 1982, 19, 1015–1021. [Google Scholar] [CrossRef]
- Pedersen, R.S.; Olesen, A.S.; Freund, L.G.; Solgaard, P.; Larsen, E. Thallium Intoxication Treated with Long-term Hemodialysis, Forced Diuresis and Prussian Blue. Acta Med. Scand. 1978, 204, 429–432. [Google Scholar] [CrossRef]
- Maya-López, M.; Mireles-García, M.V.; Ramírez-Toledo, M.; Colín-González, A.L.; Galván-Arzate, S.; Túnez, I.; Santamaría, A. Thallium-Induced Toxicity in Rat Brain Crude Synaptosomal/Mitochondrial Fractions Is Sensitive to Anti-Excitatory and Antioxidant Agents. Neurotox. Res. 2018, 33, 634–640. [Google Scholar] [CrossRef]
- Mizuno, D.; Kawahara, M.; Konoha-Mizuno, K.; Ogawara, T.; Hama, R.; Yamazaki, K. Toxic Effects of Two Redox States of Thallium on Immortalised Hypothalamic GT1-7 Neuronal Cells. Int. J. Mol. Sci. 2023, 24, 11583. [Google Scholar] [CrossRef]
- Meggs, W.J.; Cahill-Morasco, R.; Shih, R.D.; Goldfrank, L.R.; Hoffman, R.S. Effects of Prussian Blue and N-Acetylcysteine on Thallium Toxicity in Mice. J. Toxicol. Clin. Toxicol. 1997, 35, 163–166. [Google Scholar] [CrossRef]
- Osorio-Rico, L.; Villeda-Hernández, J.; Santamaría, A.; Königsberg, M.; Galván-Arzate, S. The N-Methyl-d-Aspartate Receptor Antagonist MK-801 Prevents Thallium-Induced Behavioral and Biochemical Alterations in the Rat Brain. Int. J. Toxicol. 2015, 34, 505–513. [Google Scholar] [CrossRef]
- Abdel-Daim, M.M.; Abdou, R.H. Protective Effects of Diallyl Sulfide and Curcumin Separately against Thal-lium-Induced Toxicity in Rats. Cell J 2015, 17, 379–388. [Google Scholar] [CrossRef]
- Lie, R.; Thomas, R.G.; Scott, J.K. The Distribution and Excretion of Thallium-204 in the Rat, With Suggested MPCʼs and a Bio-Assay Procedure. Health Phys. 1960, 2, 334–340. [Google Scholar] [CrossRef]
- Aoyama, H. Distribution and Excretion of Thallium after Oral and Intraperitoneal Administration of Thallous Malonate and Thallous Sulfate in Hamsters. Bull. Environ. Contam. Toxicol. 1989, 42, 456–463. [Google Scholar] [CrossRef]
- Galván-Arzate, S.; Rios, C. Thallium Distribution in Organs and Brain Regions of Developing Rats. Toxicology 1994, 90, 63–69. [Google Scholar] [CrossRef]
- Leung, K.M.; Ooi, V.E.C. Studies on Thallium Toxicity, Its Tissue Distribution and Histopathological Effects in Rats. Chemosphere 2000, 41, 155–159. [Google Scholar] [CrossRef]
- Salehi, S.; Saljooghi, A.S.; Badiee, S.; Moqadam, M.M. Chelation of Thallium (III) in Rats Using Combined Deferasirox and Deferiprone Therapy. Toxicol. Res. 2017, 33, 299–304. [Google Scholar] [CrossRef]
- Szpirer, C. Rat Models of Human Diseases and Related Phenotypes: A Systematic Inventory of the Causative Genes. J. Biomed. Sci. 2020, 27, 84. [Google Scholar] [CrossRef]
- Modlinska, K.; Pisula, W. The Norway Rat, from an Obnoxious Pest to a Laboratory Pet. Elife 2020, 9, e50651. [Google Scholar] [CrossRef]
- Vandamme, T. Use of Rodents as Models of Human Diseases. J. Pharm. Bioallied Sci. 2014, 6, 2. [Google Scholar] [CrossRef]
- Li, D.; Yao, H.; Du, L.; Zeng, X.; Xiao, Q. Thallium (I and III) Exposure Leads to Liver Damage and Disorders of Fatty Acid Metabolism in Mice. Chemosphere 2022, 307, 135618. [Google Scholar] [CrossRef]
- Alonzo, R.R.; Núñez Tapia, F.A.; Rodríguez Mercado, J.J.; González Calixto, C.; Ortuño Pineda, C.; Urióstegui-Acosta, M. Genotoxic and Cytotoxic Effect of Exposure to Thallium on Mature Spermatozoa of Mice. Cienc. Lat. Rev. Científica Multidiscip. 2022, 6, 5538–5555. [Google Scholar] [CrossRef]
- Li, D.; Li, L.; Yao, H.; Su, Q.; Ye, J. Thallium Exposure Induces Changes in B and T Cell Generation in Mice. Toxicology 2023, 492, 153532. [Google Scholar] [CrossRef]
- Sandner, G.; König, A.; Wallner, M.; Weghuber, J. Alternative Model Organisms for Toxicological Fingerprinting of Relevant Parameters in Food and Nutrition. Crit. Rev. Food Sci. Nutr. 2022, 62, 5965–5982. [Google Scholar] [CrossRef]
- Spitsbergen, J.M.; Kent, M.L. The State of the Art of the Zebrafish Model for Toxicology and Toxicologic Pathology Research—Advantages and Current Limitations. Toxicol. Pathol. 2003, 31, 62–87. [Google Scholar] [CrossRef]
- Alper, S.R.; Dorsky, R.I. Unique Advantages of Zebrafish Larvae as a Model for Spinal Cord Regeneration. Front. Mol. Neurosci. 2022, 15, 983336. [Google Scholar] [CrossRef]
- Zhao, R.; Hu, Y.; Li, B.; Chen, M.; Ren, Z. Potential Effects of Internal Physio-Ecological Changes on the Online Biomonitoring of Water Quality: The Behavior Responses with Circadian Rhythms of Zebrafish (Danio Rerio) to Different Chemicals. Chemosphere 2020, 239, 124752. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, Y.; Wei, X.; Chen, Y.; Beiyuan, J.; She, J.; Wang, L.; Liu, J.; Liu, Y.; Wang, J.; et al. Effects of Thallium Exposure on Intestinal Microbial Community and Organ Functions in Zebrafish (Danio Rerio). Elementa 2021, 9, 92. [Google Scholar] [CrossRef]
- Adhish, M.; Manjubala, I. Effectiveness of Zebrafish Models in Understanding Human Diseases—A Review of Models. Heliyon 2023, 9, e14557. [Google Scholar] [CrossRef]
- Fernández-Moreno, M.A.; Farr, C.L.; Kaguni, L.S.; Garesse, R. Drosophila Melanogaster as a Model System to Study Mitochondrial Biology. Methods Mol Biol. 2007, 372, 33–49. [Google Scholar]
- Calap-Quintana, P.; González-Fernández, J.; Sebastiá-Ortega, N.; Llorens, J.; Moltó, M. Drosophila Melanogaster Models of Metal-Related Human Diseases and Metal Toxicity. Int. J. Mol. Sci. 2017, 18, 1456. [Google Scholar] [CrossRef]
- Reyes-Rodríguez, M.Á.; Santos-Cruz, L.F.; García-Castro, C.; Durán-Díaz, Á.; Castañeda-Partida, L.; Dueñas-García, I.E.; Heres-Pulido, M.E.; Rodríguez-Mercado, J.J. Genotoxicity and Cytotoxicity Evaluation of Two Thallium Compounds Using the Drosophila Wing Somatic Mutation and Recombination Test. Heliyon 2021, 7, e07087. [Google Scholar] [CrossRef]
- Wittkowski, P.; Marx-Stoelting, P.; Violet, N.; Fetz, V.; Schwarz, F.; Oelgeschläger, M.; Schönfelder, G.; Vogl, S. Caenorhabditis elegans As a Promising Alternative Model for Environmental Chemical Mixture Effect Assessment—A Comparative Study. Environ. Sci. Technol. 2019, 53, 12725–12733. [Google Scholar] [CrossRef]
- Leung, C.K.; Wang, Y.; Malany, S.; Deonarine, A.; Nguyen, K.; Vasile, S.; Choe, K.P. An Ultra High-Throughput, Whole-Animal Screen for Small Molecule Modulators of a Specific Genetic Pathway in Caenorhabditis elegans. PLoS ONE 2013, 8, e62166. [Google Scholar] [CrossRef]
- Hernández-Cruz, E.Y.; Aparicio-Trejo, O.E.; Eugenio-Pérez, D.; Juárez-Peredo, E.; Zurita-León, M.; Valdés, V.J.; Pedraza-Chaverri, J. Sulforaphane Exposure Prevents Cadmium-Induced Toxicity and Mitochondrial Dysfunction in the Nematode Caenorhabditis elegans by Regulating the Insulin/Insulin-like Growth Factor Signaling (IIS) Pathway. Antioxidants 2024, 13, 584. [Google Scholar] [CrossRef]
- Morton, K.S.; Wahl, A.K.; Meyer, J.N. The Effect of Common Paralytic Agents Used for Fluorescence Imaging on Redox Tone and ATP Levels in Caenorhabditis elegans. bioRxiv 2023. [Google Scholar] [CrossRef]
- Hartman, J.H.; Widmayer, S.J.; Bergemann, C.M.; King, D.E.; Morton, K.S.; Romersi, R.F.; Jameson, L.E.; Leung, M.C.K.; Andersen, E.C.; Taubert, S.; et al. Xenobiotic Metabolism and Transport in Caenorhabditis elegans. J. Toxicol. Environ. Health Part. B 2021, 24, 51–94. [Google Scholar] [CrossRef]
- Varão, A.M.; Silva, J.D.S.; Amaral, L.O.; Aleixo, L.L.P.; Onduras, A.; Santos, C.S.; Silva, L.P.D.; Ribeiro, D.E.; Filho, J.L.L.; Bornhorst, J.; et al. Toxic Effects of Thallium Acetate by Acute Exposure to the Nematode C. Elegans. J. Trace Elem. Med. Biol. 2021, 68, 126848. [Google Scholar] [CrossRef]
- Hernández-Cruz, E.Y.; Eugenio-Pérez, D.; Ramírez-Magaña, K.J.; Pedraza-Chaverri, J. Effects of Vegetal Extracts and Metabolites against Oxidative Stress and Associated Diseases: Studies in Caenorhabditis elegans. ACS Omega 2023, 8, 8936–8959. [Google Scholar] [CrossRef]
- Gershon, H.; Gershon, D. Caenorhabditis elegans—A Paradigm for Aging Research: Advantages and Limitations. Mech. Ageing Dev. 2002, 123, 261–274. [Google Scholar] [CrossRef]
- Queirós, L.; Pereira, J.L.; Gonçalves, F.J.M.; Pacheco, M.; Aschner, M.; Pereira, P. Caenorhabditis elegans as a Tool for Environmental Risk Assessment: Emerging and Promising Applications for a “Nobelized Worm”. Crit. Rev. Toxicol. 2019, 49, 411–429. [Google Scholar] [CrossRef]
- Alnasser, S.M. From Gut to Liver: Organoids as Platforms for next-Generation Toxicology Assessment Vehicles for Xenobiotics. Stem Cell Res. Ther. 2025, 16, 150. [Google Scholar] [CrossRef]
- Forsythe, S.D.; Devarasetty, M.; Shupe, T.; Bishop, C.; Atala, A.; Soker, S.; Skardal, A. Environmental Toxin Screening Using Human-Derived 3D Bioengineered Liver and Cardiac Organoids. Front. Public. Health 2018, 6, 103. [Google Scholar] [CrossRef]
- Caipa Garcia, A.L.; Arlt, V.M.; Phillips, D.H. Organoids for Toxicology and Genetic Toxicology: Applications with Drugs and Prospects for Environmental Carcinogenesis. Mutagenesis 2022, 37, 143–154. [Google Scholar] [CrossRef]
- Chen, S.; Abdulla, A.; Yan, H.; Mi, Q.; Ding, X.; He, J.; Yan, C. Proteome Signatures of Joint Toxicity to Arsenic (As) and Lead (Pb) in Human Brain Organoids with Optic Vesicles. Environ. Res. 2024, 243, 117875. [Google Scholar] [CrossRef]
- Manna, S.; Firdous, S.M. Unravelling the Developmental Toxicity of Heavy Metals Using Zebrafish as a Model: A Narrative Review. BioMetals 2025, 38, 419–463. [Google Scholar] [CrossRef] [PubMed]
| Case (Location/Year) | Source of Exposure | Detected Doses | Diagnosis (Methodology) | Main Symptoms | Treatment | Outcome | Ref. |
|---|---|---|---|---|---|---|---|
| Zhu Ling (Beijing, China/1994–1995) | Unknown (criminal suspicion) | Urine: 275 µg/L; blood: 31 µg/L; hair: 531 µg/kg; nails: 22,824 µg/kg | ICP-MS in blood, urine, hair, nails, and CSF; lumbar puncture; epidemiological tracing via internet; time reconstruction in hair using laser ablation ICP-MS | Abdominal pain, alopecia, intense leg pain, paresthesia, paralysis, coma | Plasmapheresis (seven sessions, ≈10,000 mL total), ventilatory support, mechanical ventilation, Prussian blue, chelators, and metabolic support | Chronic lower limb paralysis, optic atrophy, generalized brain atrophy, permanent intellectual and physical impairment | [39] |
| 14 family members (mean age 36) with late hospitalization (China/2008–2018) | Ingestion of contaminated food (accidental/criminal) | At 2 weeks: Blood: 219–1414 µg/L (median 535); urine: 956–11,285 µg/L (median 7460) | Atomic absorption or ICP in blood and urine; EMG, brain imaging/EEG in neurological cases | Vomiting, pain, painful neuropathy, alopecia, fatigue, hyperpigmentation, mild hepatitis, confusion or coma | First: chelator sodium dimercaptosulfonate (5 mg/kg IM 1–4x/day); second: oral Prussian blue (250 mg/kg/day in 50 mL 20% mannitol, divided in four doses); daily hemodialysis (4–6 h/day) | One died due to previous pulmonary fibrosis/ARDS, one developed deep vein thrombosis in left leg, and one had residual paresthesia and generalized anxiety. Most recovered without sequelae | [38] |
| 40-year-old male (China, 2014) | Acute unknown ingestion (possible criminal exposure) | Blood: “Supralethal” levels > estimated lethal range of 10–15 mg/kg in humans | Tl monitoring in blood and urine; neurological and metabolic clinical assessment | Severe abdominal pain, intense vomiting, ascending dystrophic neuropathy, confusion | Hemoperfusion (three rounds; reduced Tl in blood by 20–35%), followed by continuous CVVH (5 rounds; 40–64% reduction each round), chelation with 2,3-DMS (250 mg/day IM), nutritional and metabolic support (including Prussian blue 250 mg/kg/day orally) | Fully recovered without significant neurological sequelae | [44] |
| 51-year-old female (Buenos Aires, 2018) | Unidentified accidental exposure | Urine: 540 µg/g creatinine (healthy range: 0.410 µg/g) | Normal heavy-metal screening (Pb, As, Hg, Cu); thallium measured in urine | Myalgia, vertigo, asthenia, abdominal pain, jaundice, confusion, asterixis, thrombocytopenia, alopecia (eyebrows, scalp, armpits), plantar ulcers | Oral D-penicillamine 1000 mg/day (preferred over Prussian blue due to contraindication); supportive care and annual follow-up | Full resolution after one year, normalized urinary excretion, no apparent sequelae | [36] |
| 18-year-old male (USA, 2024) | Ingested elemental thallium bar (~90% pure–100 g) purchased online, suicide attempt | Peak serum: 423.5 µg/L (normal <5.1); urine: 1850.5 µg/g creatinine (normal <0.4); metal fragment ~100 g | Serial ICP-MS in serum and urine; abdominal X-ray; colonoscopy for fragment extraction | No classical symptoms (no vomiting, diarrhea, alopecia, neuropathic pain, limb weakness, or systemic signs) | Activated charcoal, oral Prussian blue (3 g TID), endoscopic fragment removal, intestinal lavage with polyethylene glycol | Completely asymptomatic; negative serum and urine at 44 days; no alopecia or neurological deficits | [45] |
| Glass factory worker (Japan, 1998) | Daily exposure to raw materials containing Tl at glass plant (chronic occupational exposure, ~4 years) | Hair: ~20 ng/g (above healthy range ~5–10 ng/g) | ICP-MS in hair sample; neurological exam; nerve conduction velocity; work history | Mild alopecia, chronic diarrhea, colicky abdominal pain, paresthesia in hands/feet, glove-and-stocking neuropathy | Immediate removal from exposure and symptomatic treatment; no chelators reported | Persistent mild neuropathy; no progression or severe sequelae | [46] |
| Adulterated herbal infusion, family (Granada, Spain/1985–1987) | Infusion consumed by all; adulterated with thallium thiosulfate (presumed criminal act) | Not quantified (clinical information based on symptoms and family evolution) | Clinical exam + family history; diagnosis confirmed by thallium lab analysis (unspecified method) | Vomiting, abdominal pain, GI bleeding, severe headache, paresthesia, sensory–motor polyneuritis | Symptomatic treatment (decontamination, fluids, neuromuscular support); Prussian blue use not always documented | Gradual recovery in 3–9 weeks without lasting sequelae in most survivors | [47] |
| Five patients (Mexico, 1989) | Three suicide attempts; two unknown accidental ingestions | Detectable Tl in blood and urine (not publicly quantified) | Blood and urine analysis; scalp skin biopsy (telogen phase, pigment in keratin, hypotrophic follicles) | Diffuse alopecia (all cases); GI and neurological symptoms in accidental cases. Radiopacities in tibial metaphyses in children | Oral D-penicillamine (dosage not specified); dermatological follow-up | Partial restoration of neuropathy in some cases | [48] |
| 13 cases (Sichuan, China/2000–2010) | Oral ingestion of thallium (criminal or accidental, including contaminated water, food, and mushrooms from Tl-rich soil) | Elevated Tl levels in urine and blood (specific values not reported) | Clinical–toxicological diagnosis; majority initially misdiagnosed (e.g., Guillain–Barré syndrome); later confirmed via toxicological analysis | Peripheral neuropathy, cognitive impairment, alopecia, anxiety, depression, digestive symptoms | Supportive care, Prussian blue, hemodialysis in some cases | One death during hospitalization; follow-up up to 12 years: persistent neurological sequelae in several patients (33% cognitive decline, 50% anxiety, 42% depression) | [37] |
| Model | Tl Compound | Antioxidant (Dose) | Key Findings | Ref. |
|---|---|---|---|---|
| Female Swiss albino mice | Subcutaneous Tl acetate 70–85 mg/kg (LD90–LD100) | Prussian blue (50 mg/kg oral); NAC (200 mg/kg i.p.) | Prussian blue increased survival from 10% to 50% (p = 0.014); NAC to 35% (p = 0.13); NAC + Prussian blue showed no additional benefit over Prussian blue alone | [106] |
| GT1-7 neuronal cells | Tl(I), Tl(III) | Mannitol, ascorbic acid, α-tocopherol (dose not specified) | Protection: ↑ viability, ↓ apoptosis, ↓ MDA, SOD stabilization | [105] |
| Adult rats (i.p.) | Tl+ acetate 32 mg/kg | MK-801 (1 mg/kg) | Reduced lipid peroxidation and GSH depletion in brain | [107] |
| Rat synaptosomes/mitochondria (ex vivo) | Tl+ 5–250 µM | S-allyl-L-cysteine (100 µM) | Mitochondrial and lipid peroxidation protection; less effective than neuroexcitatory antagonists | [104] |
| Rats | Tl (dose not specified) | Diallyl sulfide, curcumin | Hepatoprotective effect: ↓ inflammatory cytokines and liver enzymes | [108] |
| Isolated hepatocytes/mitochondria | Tl(I) | α-tocopherol, deferoxamine, carnitine, L-glutamine, fructose, xylitol | Inhibition of caspase-3 and apoptosis | [70] |
| Aspect | Prussian Blue | Antioxidants |
|---|---|---|
| Main mechanism | Reduces Tl absorption in the intestine by increasing excretion | Cellular protection by reducing oxidative stress |
| Preclinical evidence | Strong: animal models and human case reports | Limited: in vitro and cell-based studies only |
| Clinical evidence | Yes: supported by multiple clinical applications | No clinical evidence in humans to date |
| Potential benefit | Removes Tl from the body | May reduce residual Tl-induced organ damage |
| Substitute or complement? | Clinically validated substitute | Complementary therapy |
| Study Models | Tl Compound | Route of Administration | Administered Dose | Time | Tl Accumulation in the Organism | Ref. |
|---|---|---|---|---|---|---|
| Rats | Tl 204 | Oral | 0.76 mg/kg | Approximately 7 days | Highest amount in the kidneys, followed by the salivary glands, testes, muscles, bones, lymph nodes, gastrointestinal tract, spleen, and liver. | [109] |
| Rats (Wistar) | Tl2SO4 | Intraperitoneal | 16 mg/kg | 24 h after | Greater accumulation of Tl in the kidneys, followed by the testes, spleen, lungs, heart, and liver. | [55] |
| Hamster | C3H2O4Tl2 and Tl2SO4 | Oral (single dose) | C3H2O4Tl2: 12.5 mg/kg Tl2SO4: 12.35 mg | 12, 24, and 72 h after | The Tl content was higher in the kidneys regardless of the post-treatment time, followed by the testes (starting at 24 h post-treatment), the heart, and the liver. | [110] |
| Rats (Wistar) (newborn) | C2H6O2Tl | Intraperitoneal | 16 mg/kg | 24 h after | Highest content in the testes, heart, and kidneys. | [111] |
| Albino rats | Tl2SO4 | Intraperitoneal | 30 mg/kg | 4 days | The Tl content was highest in the kidney, followed by the ileum, stomach, and liver. | [112] |
| Rats (Wistar) | Tl 3+ | Oral | 20 mg/kg/día | 60 days | Greater amount in the kidney, followed by the liver, spleen, intestines, and heart. | [113] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Avendaño-Briseño, K.A.; Escutia-Martínez, J.; Pedraza-Chaverri, J.; Hernández-Cruz, E.Y. Thallium Toxicity: Mechanisms of Action, Available Therapies, and Experimental Models. Future Pharmacol. 2025, 5, 49. https://doi.org/10.3390/futurepharmacol5030049
Avendaño-Briseño KA, Escutia-Martínez J, Pedraza-Chaverri J, Hernández-Cruz EY. Thallium Toxicity: Mechanisms of Action, Available Therapies, and Experimental Models. Future Pharmacology. 2025; 5(3):49. https://doi.org/10.3390/futurepharmacol5030049
Chicago/Turabian StyleAvendaño-Briseño, Karla Alejandra, Jorge Escutia-Martínez, José Pedraza-Chaverri, and Estefani Yaquelin Hernández-Cruz. 2025. "Thallium Toxicity: Mechanisms of Action, Available Therapies, and Experimental Models" Future Pharmacology 5, no. 3: 49. https://doi.org/10.3390/futurepharmacol5030049
APA StyleAvendaño-Briseño, K. A., Escutia-Martínez, J., Pedraza-Chaverri, J., & Hernández-Cruz, E. Y. (2025). Thallium Toxicity: Mechanisms of Action, Available Therapies, and Experimental Models. Future Pharmacology, 5(3), 49. https://doi.org/10.3390/futurepharmacol5030049










