1. Introduction
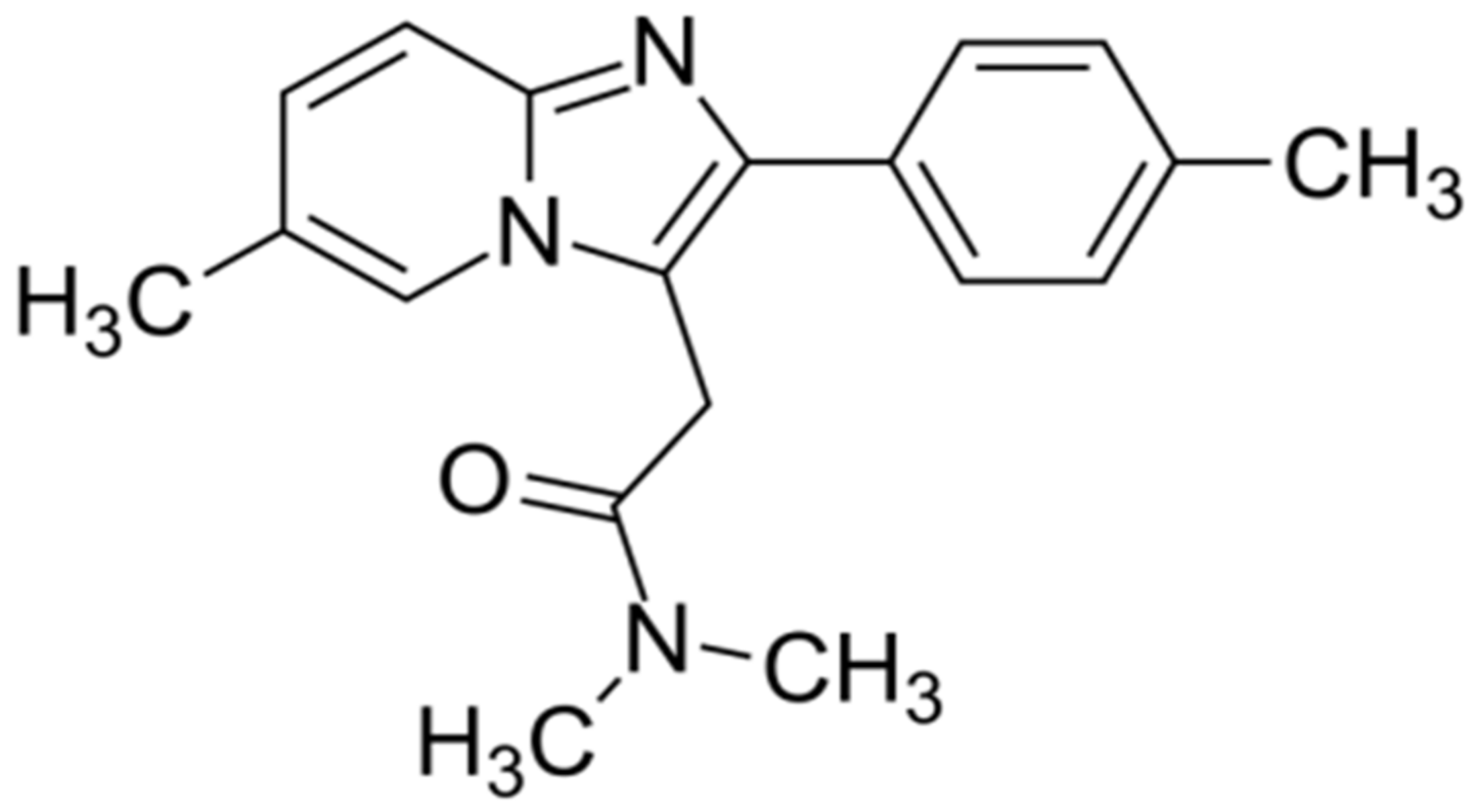
The drug zolpidem is an imidazopyridine (
Figure 1) and is mainly used for the short-term treatment of sleeping problems. In patients with temporary (transient) insomnia, this medication reduces the time it takes to fall asleep (sleep latency), enhances the duration of sleep, and reduces the frequency of awakenings throughout sleep. The hypnotic efficacy of zolpidem is generally equivalent to that of benzodiazepines as well as nonbenzodiazepine hypnotic medications; it works as a GABA (gamma-aminobutyric acid) receptor chloride channel modulator/agonist, increasing GABA inhibitory effects and causing sedation [
1]. In healthy patients, zolpidem is rapidly absorbed by the gastrointestinal tract and has a short half-life. It is available in both immediate and extended-release forms and is also available in the form of an oral spray that is sprayed into the mouth over the tongue and a sublingual tablet. Because of the risk of dependence and tolerance, zolpidem should not be used for an extended time period (no more than 4 weeks); should only be used after cognitive behavioral therapy for insomnia and behavioral changes; and with a doctor’s prescription, which should include the dose and duration of treatment [
2]. Nonetheless, zolpidem is regarded as a reasonable choice of therapy because, when compared with other drugs, it has a lower incidence of residual daytime sleepiness and a lower risk of falls [
3].
In 2013, the Food and Drug Administration (FDA) issued a safety announcement, “Risk of next-morning impairment after use of insomnia drugs; FDA requires lower recommended doses for certain drugs containing zolpidem”, notifying the public with new information about zolpidem, a widely prescribed insomnia drug [
4]. These new data revealed that females were at risk for excessive daytime sedation and impaired driving proficiency following bedtime doses of zolpidem. The reason for this variability in response was the reduced metabolic clearance of zolpidem and higher morning blood concentrations in females compared with males. After verifying these sex differences, the FDA issued a recommendation to reduce the dosage for females down to 50% of the dose for males and required the labeling of zolpidem products to include new information related to the doses in males and females [
5,
6]. In 2017, the FDA issued an update to the Safety Announcement from 2013, claiming that they had approved label changes specifying new dosing recommendations for zolpidem products. Additionally, a warning for patients who take the sleep medication zolpidem extended-release (Ambien CR) - either 6.25 mg or 12.5 mg―was included, claiming that these patients should not drive or engage in other activities that require complete mental alertness the day after taking the drug because zolpidem levels can remain high enough the next day to impair these activities. [
5]. No other regulatory agency worldwide has taken similar action [
7].
In 2014, the European Medicines Agency (EMA), represented by the Pharmacovigilance Risk Assessment Committee (PRAC), issued an assessment report (EMA/418370/2014) for zolpidem-containing medicinal products. Safety and efficacy data related to the risk of impaired driving and somnambulism after treatment with zolpidem were reviewed by the PRAC. Upon PRAC review it was decided that the recommended daily dose of zolpidem should not be changed, claiming the lack of evidence in ensuring that a lower dose would be effective or that would reduce the risk of impaired driving and somnambulism. On the other hand, it was agreed that reducing the daily recommended dose could lead to the use of non-effective doses, which could lead the patients to take additional doses in the middle of the night, thereby increasing the risk of accidents in the next day. Additionally, the PRAC recommended to include some warnings in the Summary of Product Characteristics (SmPC) and the Package Leaflet (PL) of products containing zolpidem. [
8]. It is important to note that, within PRAC, the decision was not unanimous. A PRAC member issued a divergent statement that was aligned with the FDA position.
Table 1 and
Table 2 summarize the Label changes that occurred after the FDA recommendation of zolpidem products in the US and the EU.
Following the divergent positions of the two main drug agencies in the world, the FDA and EMA, several studies have been published to assess the sex differences when taking zolpidem products. Zolpidem pharmacokinetics and sex have long since been settled, and there is not much more to describe in a descriptive sense. The question is: what is the mechanism of the sex difference?
Based on our experience about in silico pharmacology [
9,
10,
11,
12,
13,
14,
15,
16], this study aimed to perform an in silico study to assess EMA and FDA positions on dose adjustment based on sex. The results from this study were critically assessed and confronted with the already available published studies and also with the studies used by the agencies to sustain their positions.
2. Materials and Methods
2.1. Clinical Data and Study Design
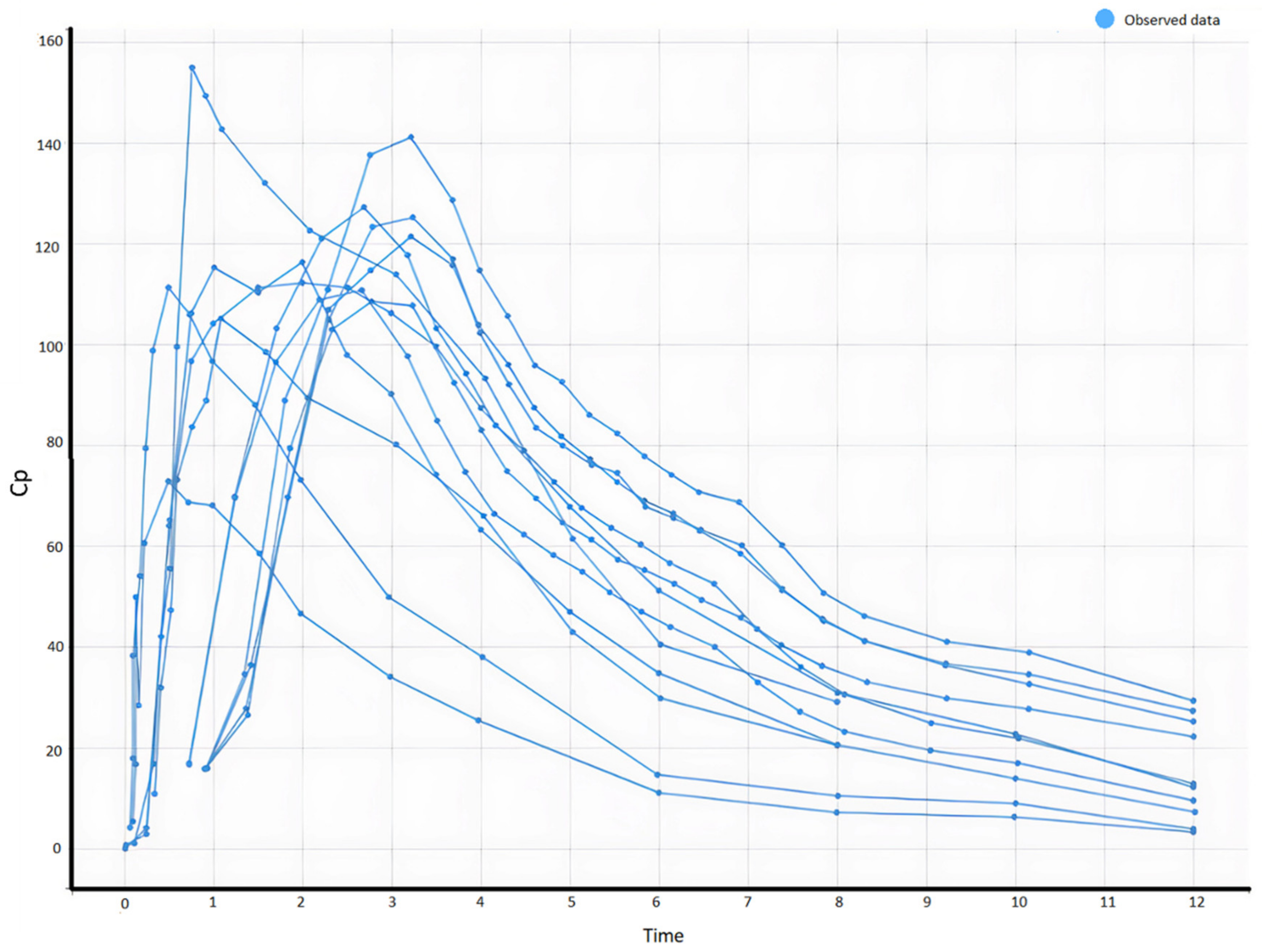
Previous studies evaluated the pharmacokinetics of zolpidem extended-release tablets on healthy volunteers; we gathered those studies and used them for our research. Data analysis for the published study aggregated male and female patients’ intake of 5, 10, and 12.5 mg of zolpidem. In the present report, kinetic and dynamic data from the same study were analyzed separately for male and female subjects, and sex-related differences were assessed to understand the differences between the three dosages in terms of efficacy and safety. Using WebPlotDigitizer, we extracted data points from plasma concentration-time graphs to create a virtual population for our study. The collected profiles were used for model building, evaluation, and simulation of the intake of 5, 10, and 12.5 mg of zolpidem extended-release tablets [
17]. Using this method, it was only possible to gather 12 subjects for the study since in some articles the separation between individuals’ sexes and other demographics was missing, which made it difficult to generate more virtual individuals for the study. All the estimations were based on the real data provided by those articles on population pharmacokinetic modelling of zolpidem.
2.2. Pharmacokinetic Analysis
Population pharmacokinetic (PK) analysis of zolpidem was conducted using a nonlinear mixed-effects approach within the Monolix software
® (version 2020R1; Lixoft, Antony, France,
http://lixoft.com/, accessed on 1 December 2020). Monolix uses a stochastic approximation expectation maximization (SAEM) algorithm to perform maximum likelihood estimation of the pharmacokinetic parameters without approximating the statistical model [
18]. The Fisher information matrix was computed using stochastic approximation, and the likelihood was calculated by importance sampling.
2.3. Model Building
With and without delays, one- and two-compartment structural models were fitted to the pooled data. The apparent volume of distribution (
Vd/F) and zolpidem clearance (
CL/F) were estimated using all compartmental models. The goodness of fit was used to evaluate and select models. Unstable models and models that, biologically, did not make sense were excluded from the model-building process. The Akaike information criteria and the Bayesian information criterion [
19] were used to aid in the selection of structural models. The percentage of relative standard error (RSE) was also employed as a precision metric, and it was taken into account while deciding on the optimum error model. The precision of parameter estimations, measures of variability, and the objective function value were all used to compare models (OFV). The base model was developed using an empirical approach that focused on evaluating a variety of structural models. Several absorption models were tested to see which one best described zolpidem absorption in the data set. The effects of lag time and absorption rate constants (
Ka) on zolpidem absorption process estimations were also investigated.
2.4. Covariate Model
After selecting the base model, we looked into potential covariate impacts on pharmacokinetic parameters. Several demographic and clinical factors were studied to see if they had an effect on zolpidem pharmacokinetics. Age, sex, weight, and clearance were all taken into account. An exploratory analysis was carried out to establish a link between zolpidem pharmacokinetic parameters and the aforesaid characteristics. The empirical Bayesian estimates from the base model’s individual parameters were plotted against the covariate values and compared visually. Following the initial analysis, final covariates were chosen for inclusion in the model. Covariates were added to the model until the OFV did not decrease any further.
2.5. Model Evaluation
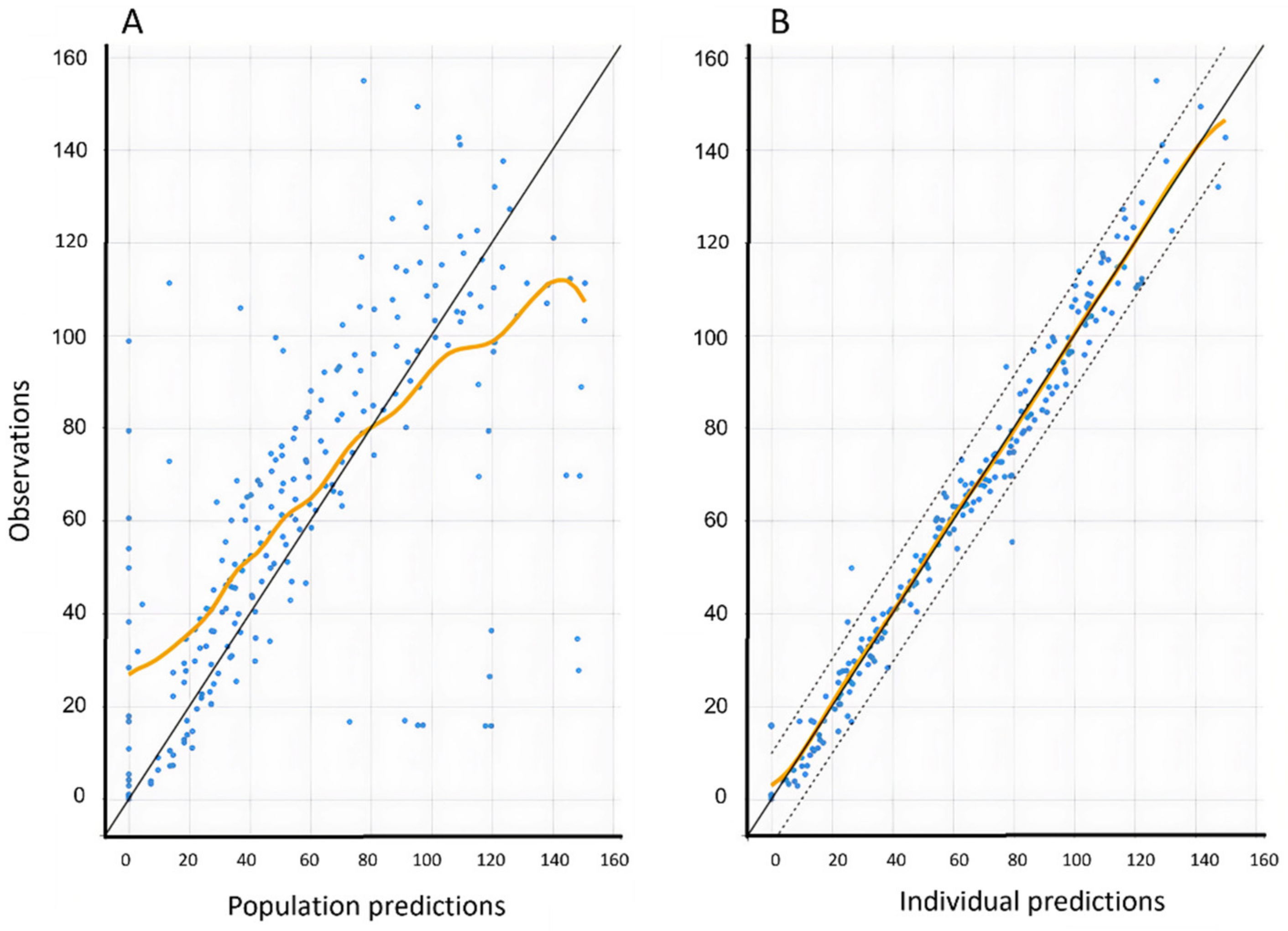
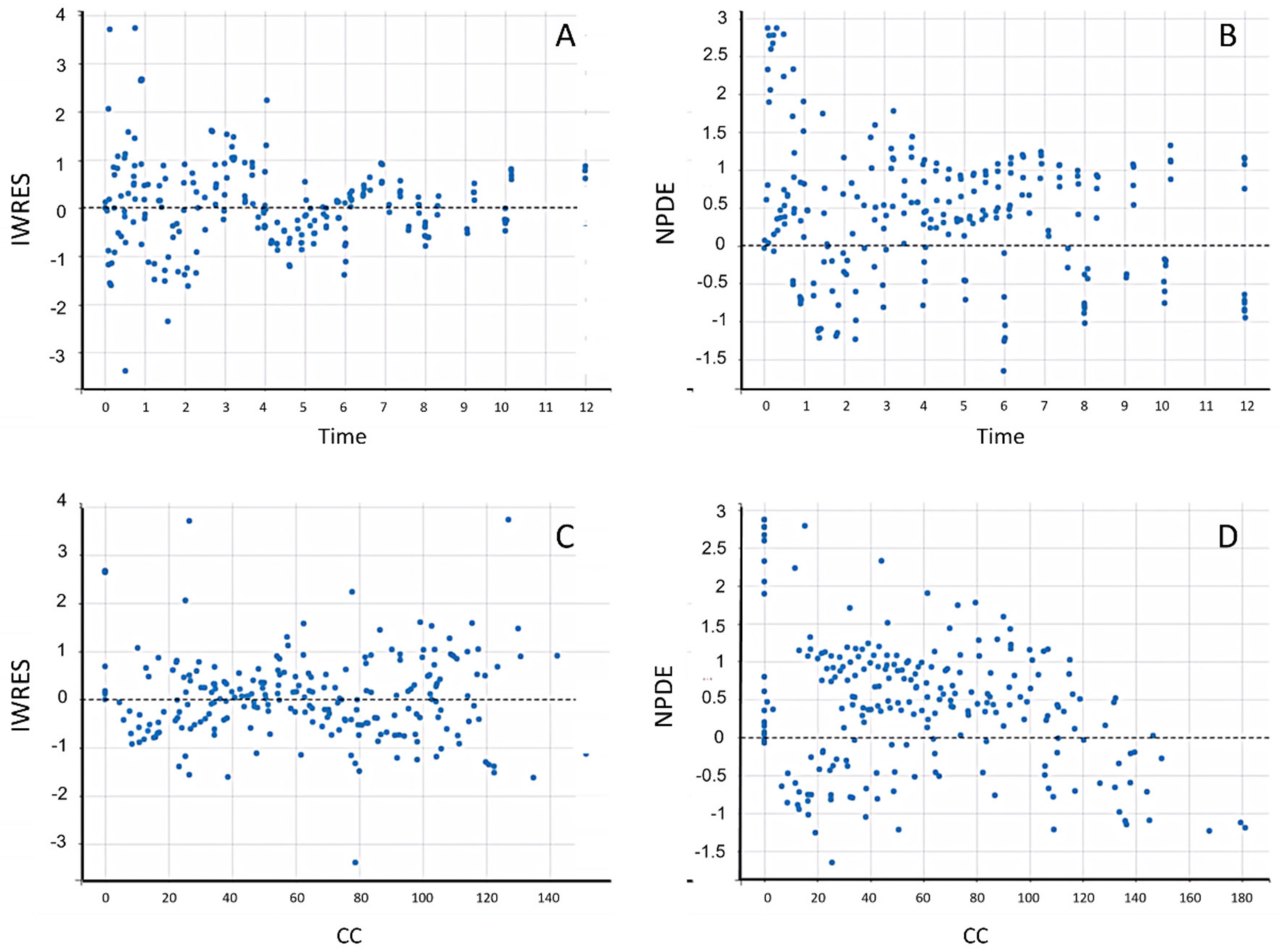
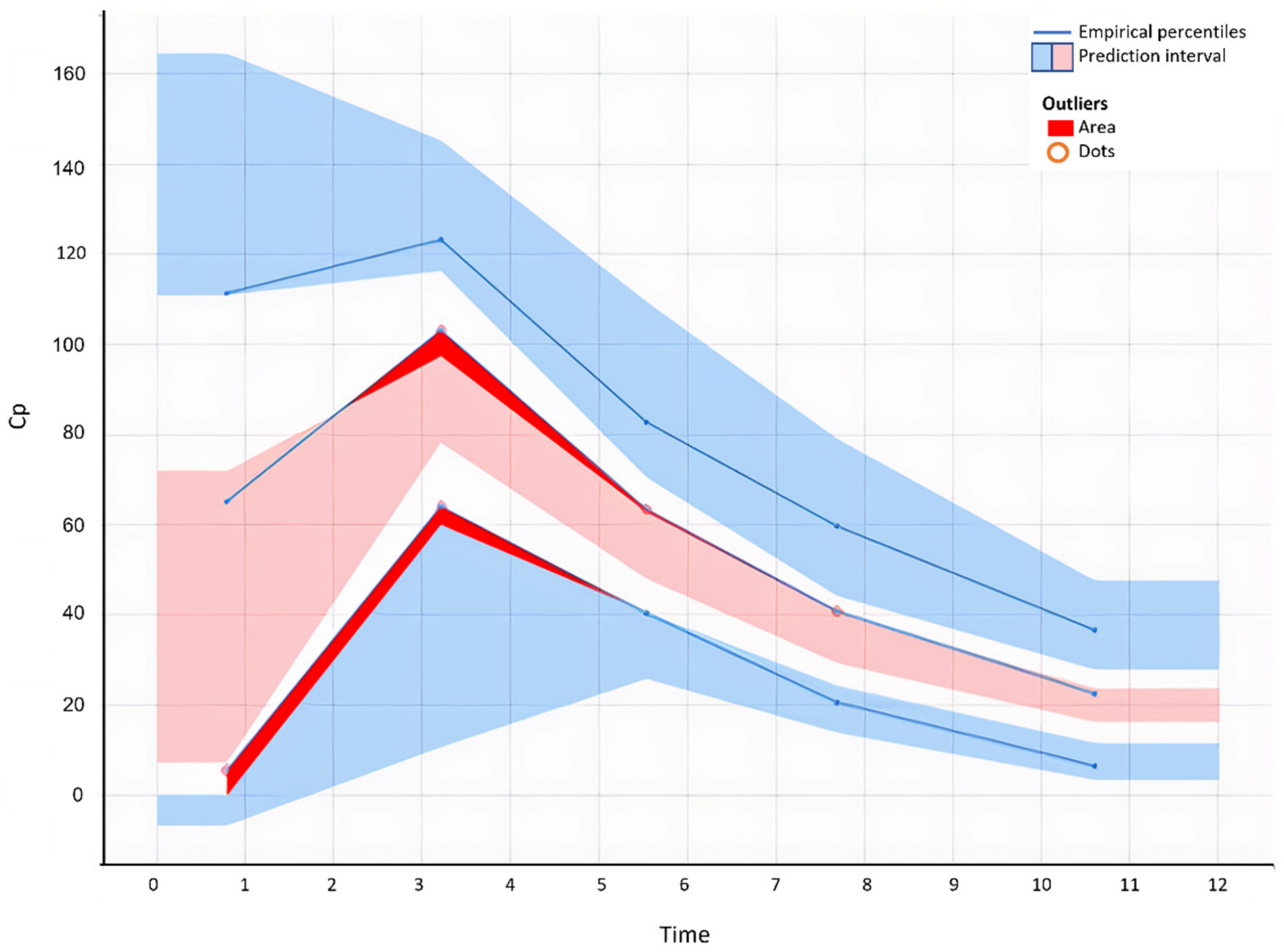
The model’s performance was evaluated numerically and graphically, with observed drug concentrations visually scrutinized for correlation with projected values. The standard errors for both the estimated population parameters and the random effects error models were calculated. Several diagnostic plots, such as observed against population-predicted zolpidem concentrations and observed versus individual-predicted zolpidem concentrations, were utilized to visually test the model’s fit during model development [
20]. Visual inspection was also carried out on plots of residuals and conditional weighted residuals vs. time or population-predicted zolpidem concentrations. A step-by-step technique involving forward inclusion and backward deletion was used to create the covariate model. If a covariate improved the fit while lowering the Bayesian information threshold, it was kept in the model (BIC). The resulting covariate model was then tested numerically and visually for predicting accuracy. The resultant model was assessed further using graphs of normalized prediction distribution error as a function of time and population-predicted zolpidem concentrations. [
21].
2.6. Monte Carlo Simulation Assessment for the Different Dose Regimens and Optimization (FDA vs. EMA)
First, based on the model, we first checked what percentage of the population satisfied the efficacy and safety targets criteria. Monte Carlo simulations were performed by Simulx software
® (version 2020R1; Lixoft, Antony, France,
http://lixoft.com/, accessed on 5 January 2021), based on the final PK model to generate 100 Pk profiles of zolpidem for each candidate regimen. Ambien is usually prescribed for no more than four weeks and recommended for shorter periods, if possible [
2].
Three standard concentrations of 5, 10, and 12.5 mg were compared across simulation groups. The simulation group of 5 mg was defined as the reference group and all other groups were compared to this reference group. Moreover, we simulated three different dose regimens for each dose treatment: (A) 1 day, 1 pill; (B) 1 week, 1 pill per day, every day for 7 days; and (C) 1 month, 1 pill per day, every day for 4 weeks. Lastly, we selected individuals with very high Cmax concentrations to see if they met the efficacy and safety targets for drug individualization.
2.7. EMA and FDA Data Collection of Positions on Zolpidem Dosage for Males and Females
Since this study aimed to confront the data behind EMA and FDA positions, a summary of the data collection is represented below. EMA and FDA websites were explored with search using the terms “zolpidem” and “dose” or “dosage”. It was possible to verify that EMA and FDA issued their decisions based on several studies.
The FDA provides a data summary that mentions that driving simulation and laboratory studies demonstrated that blood levels of zolpidem above 50 ng/mL could impair driving by increasing the risk of an accident. Pharmacokinetic trials that included 250 males and 250 females, with the dosage of 10 mg of Ambien (or bioequivalent zolpidem products), have shown that 15% of females and 3% of males had zolpidem concentrations exceeding 50 ng/mL 8 h after dosing. Furthermore, some measurements were even superior to this value: one measurement in males and three in females were were ≥90 ng/mL at about 8 h after dose.
The extended-release zolpidem products (Ambien CR or generic equivalents) represent an even higher percentage of population (males and females) that experience impairing morning zolpidem levels. For zolpidem extended-release 12.5 mg, pharmacokinetic trials have shown that 33% of females and 25% of males had zolpidem blood concentrations exceeding 50 ng/mL 8 h after dosing and 5% of patients had blood levels ≥100 ng/mL.
Studies for zolpidem extended-release 6.25 mg, 8 h after dosing, have described that 15% of adult females and 5% of adult males had a zolpidem level of ≥50 ng/mL, while for elderly males and females, 10% had this amount of zolpidem concentration.
The FDA is still evaluating the risk of next-morning impairment not only for zolpidem products, but also with other insomnia drugs [
4,
22].
EMA through the PRAC Assessment Report from 2014, EMA/418370/2014, provided a very extensive and completed assessment for which a different kind of data was taken into account. Clinical Safety was assessed through reporting rates, individual case safety reports, literature, and an EMA drug utilization study (DUS). After assessing all the data, an overall discussion on safety was conducted. To assess the Pharmacokinetic-Pharmacodynamic relationship, the PRAC requested the MAHs to present any relevant evidence on the PK-PD relationship across dose levels (5 mg and 10 mg); special populations and other available publications were also taken into account. Clinical efficacy was assessed through the only existing three dose-finding studies starting with 5 mg of zolpidem (all other studies included doses only of 10 mg or higher and were not analyzed). Literature data were also considered to review the efficacy data. After the overall discussion on efficacy, and overall benefit-risk assessment was issued.
As a consequence, it was concluded that the benefit-risk balance of the medicinal products containing zolpidem remains favorable, subject to the agreed changes to the related warning and precautions sections on product information as described in
Table 2 [
8].
Table 1 mentions proposed labelling changes related to dosing by agencies.
Taking into account the provided data by the EMA and the FDA, an in-silico study was performed to better clarify the mechanism behind the difference in the PK of males and females and also to understand what rationale is behind each position and determine if another unknown fact was impacting the FDA or EMA positions.
4. Discussion
In 2007, a study supported by SanofiAventis, the pharmaceutical company that launched zolpidem, was released comparing the pharmacokinetics and safety of 12.5 mg zolpidem extended-release and 10 mg original zolpidem in healthy volunteers to evaluate the potential for an improved clinical profile. The time to maximum concentration (tmax) and terminal elimination half-life (t1/2) of 12.5 mg zolpidem extended-release were comparable to those of 10 mg zolpidem, suggesting an elimination profile that reduced the risk of next-day performance impairment and a similar rapid onset of action. The ideal dose formulation for extending the duration of effect while keeping a safety profile similar to original zolpidem and placebo was discovered to be zolpidem extended-release, significantly reducing the number of awakenings [
23]. Indications for zolpidem use recommend that patients use the lowest effective dose. The recommended starting dose for females is 6.25 mg, while for males it is either 6.25 or 12.5 mg, and it should be used just once each night, right before bedtime, at least 7–8 h before the desired time of awakening. If the 6.25 mg dose is not working, one can increase the amount to 12.5 mg (according to FDA recommendations). Higher morning blood levels following administration of the 12.5 mg dose enhance the risk of next-day impairment of driving and other activities requiring complete alertness in some patients. Therefore, in the European Union (EU) member states, zolpidem is available only as 5 mg and 10 mg immediate-release (IR) formulations to be both more effective and safer.
An optimum dose schedule should increase the chances of most patients attaining the target pharmacodynamic index while having no or few patients fall into the pharmacokinetic dangerous range [
24]. Most studies regarding zolpidem dose regimens agree that a 10 mg dose in the non-elderly is linked with clinically adequate efficacy and a low incidence of side effects [
25]. Lower doses produce inadequate efficacy, whereas greater doses produce more frequent side effects being only marginally more effective. Therefore, this study aimed to understand if the FDA and EMA dose regimen differences are reasonable.
The model presented in this study was developed with the guidance of Stockman et al.’s article, a preliminary assessment of zolpidem pharmacokinetics in pediatric burn patients [
26]. Using this article as a basis for our study, we developed a model with the clinical data that was digitized from previous studies. Zolpidem Pk parameters were estimated using non-linear mixed-effects modeling. We sought to understand the variability in drug concentrations among individuals in a group of interest to predict different levels of plasma concentrations, using virtual individuals and building mathematical models to describe those data.
In this study, a two-compartment model, with lag time, first-order absorption and linear elimination best described the Pk of zolpidem. However, our model has some limitations. First, the creation of the model to frame mechanisms and hypotheses is empirical and depends on the decisions of the model developer. Second, the model would have benefited from non-compartmental analysis to define elementary pharmacokinetic information of zolpidem, characterize new drug products, and help guide development at each stage. Moreover, the sample size was small (only 12 subjects) and from a homogenous population. Hence, the likelihood of finding significant covariates on the pharmacokinetic parameters was low and the extrapolation of results to therapeutic practice may be more difficult. Age, clearance, weight, and sex all influenced zolpidem pharmacokinetics; however, according to Salvà, P. et al., these factors do not represent statistically significant differences [
27].
Keeping in mind the limitations, the obtained results should be interpreted with a clear understanding of the model’s assumptions. Simulations evaluated what percentage of a typical population met the efficacy and safety criteria for zolpidem tablets of 10 and 12.5 mg. At first, there was only a 57% chance of being efficient for an individual of this population. When we simulated the different dose regimens among the same groups, based on Monte Carlo simulation, we obtained different percentages of chances to be effective and safe, according to the different regimens (
Table 5). For the treatment of insomnia, it is recommended that zolpidem be taken orally right before bedtime. In adults, the maximum daily dose of zolpidem is presently 10 mg for EMA and 12.5 mg for FDA and duration of each zolpidem course should not exceed four weeks. According to our study, the zolpidem optimal dose regimen is 10 mg.
The study brings a related finding and efficacy of 12 mg tablets, thus explaining why in the EU, Zolpidem is available only as 5 mg and 10 mg immediate-release (IR) formulations. The data obtained suggested that 12.5 mg are dispensable since they did not meet the efficacy and safety criteria established. Zolpidem’s acute toxicity is milder than that of other short-acting benzodiazepines like triazolam and midazolam. We focused on extended-releases because the existing literature already demonstrates sufficient evidence for effectiveness/safety immediate release. Additionally, the proposed regimen is unlikely to result in Ctroughs of >20 ng/mL. The Ctrough is the concentration reached by a drug immediately before the next dose is administered. When zolpidem is administered in multiple doses, it is necessary to pay attention to the cumulative effects of this drug, thereby minimizing the likelihood of overexposure and toxicity. According to our data analysis, we concluded that, for achieving patient safety, Ctroguhs lower than 20 ng/mL were sufficient. For the simulation, we selected the option “same individuals among groups” to ensure that the differences are due to the different treatments and not from sampling different individuals. To be more confident about our results, we should have also simulated the treatment with different individuals to mimic a parallel clinical trial design so we could test if bigger or smaller group sizes would give different results.
Not only ours, but also previous model-based simulations suggest the recommended dose regimen from the EMA agency to be both more effective and safer, justifying the reason not to commercialize the 12.5 mg tablets due to the small percentage of effectiveness (<50%) and low percentage of safety (36%). The 10 mg treatment, according to our model, had only a 69% chance of being efficient and a 36% chance of being safe for an individual of this population. Although these values are not ideal, they are better than the values obtained for the 12.5 mg. According to the literature, in healthy participants, zolpidem at a dose of 10 mg/day was demonstrated to reduce the time to onset and lengthen the duration of sleep [
1,
25]. When considering the uncertainty of population pk models, it is important to remember that these results are subject to error, which can be reduced with new clinical data and/or therapeutic drug monitoring.
Despite the fact that sex has no significant effect on zolpidem pharmacokinetics, our findings showed that females’ plasma concentrations were consistently greater than men’s at equivalent doses and times following the treatment, which is consistent with previous research. Adult non-elderly females had lower zolpidem clearance than adult non-elderly men, according to several clinical PK analyses. As a result, females obtain higher plasma concentrations and a larger area under the plasma concentration-time curve (AUC) than males at any given dose. Body size and clearance only explain a portion of these differences [
28].
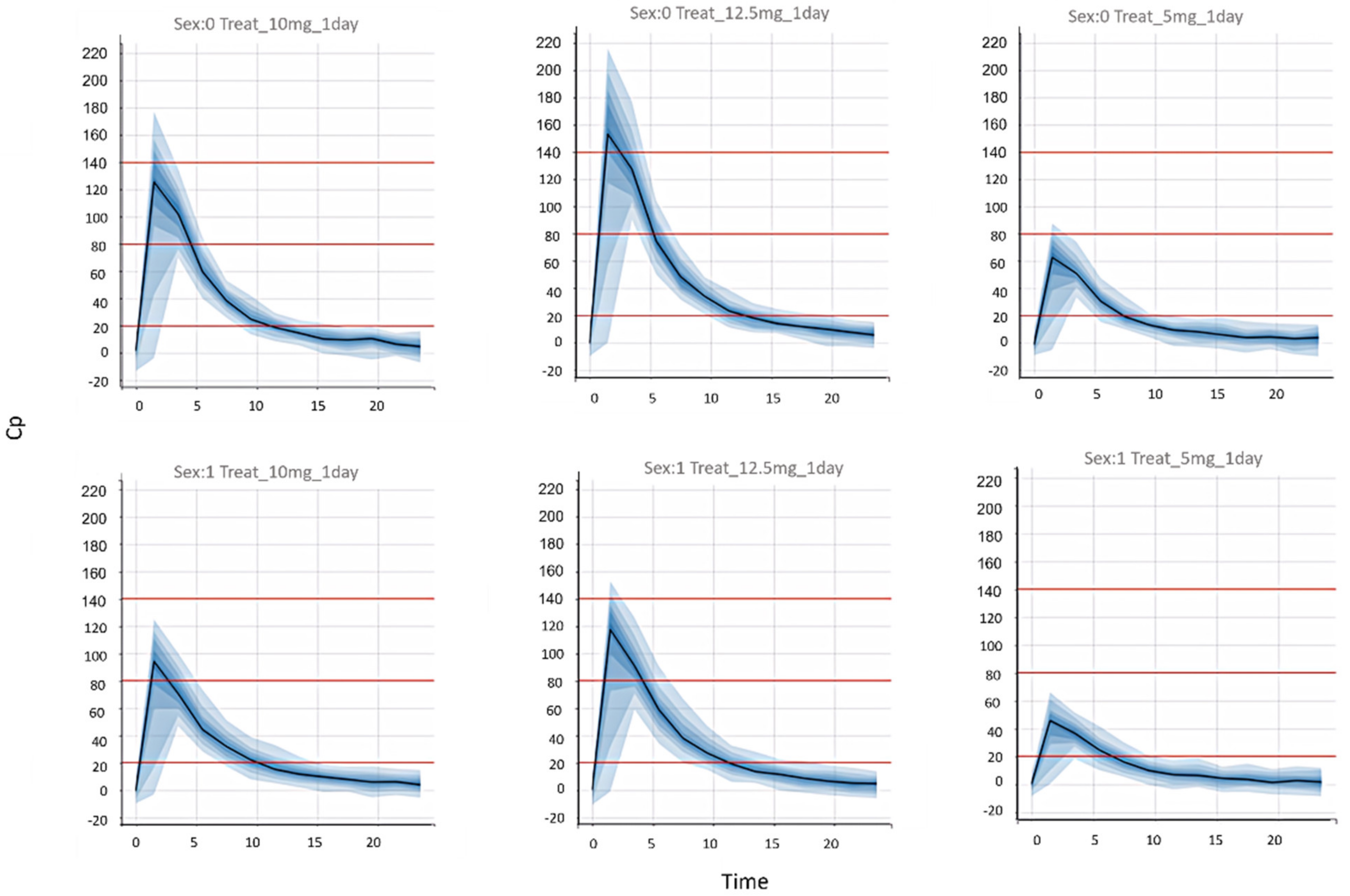
For people with reduced liver function and elderly people, zolpidem tablets are effective and well tolerated at 5 mg. The EMA agency does not distinguish between starting doses for males and females. They only mention that the lowest effective daily dose of zolpidem should be used and it must not be exceeded the 10 mg dose. However, the FDA agency considers that females should start by taking a 5 mg dose and males a 10 mg dose. EMA’s position may be due to the low efficacy that 5 mg zolpidem tablets show. In
Figure 5, we can see that for the modeled population, the 5 mg dose did not meet the efficacy criteria. Furthermore, in
Figure 7, we can see that only a small portion of the female population met the efficacy targets, and the majority would require a higher dose so the drug can be effective. However, we consider that the FDA’s initial dose recommendations for zolpidem prescription are a good reference for a safer use of this drug. Zolpidem has a similar efficacy to other hypnotics as benzodiazepines and zopiclone. When given at bedtime, zolpidem appears to have a minor effect on cognition and psychomotor performance the next day when the dose is correctly prescribed with all the precaution measures. Furthermore, when given as prescribed or for longer periods, there is little indication of tolerance to the hypnotic effects of zolpidem, or of rebound insomnia or withdrawal symptoms after termination of the medicine [
1,
25].
5. Conclusions
The study characterized the pharmacokinetic parameters of zolpidem to assess the EMA and FDA positions regarding the recommended dosage for males and females for zolpidem. The results from the in-silico studies support the EMA decision that does not distinguish between starting doses for males and females claiming that the effective daily dosage for both sexes is 10 mg. FDA considers that females should start by taking a 5 mg dose and males a 10 mg dose, but our findings in the modelled population showed that the 5 mg dose did not meet the efficacy criteria and that the majority of females would require a higher dose to reach efficacy. The study brings an additional finding related to the safety and efficacy of 12.5 mg tablets, thus explaining why in the EU member states, zolpidem is available only as 5 mg and 10 mg immediate-release (IR) formulations. The data obtained suggested that 12.5 mg are dispensable since they did not meet the efficacy and safety criteria established. Zolpidem’s acute toxicity is milder than that of other short-acting benzodiazepines like triazolam and midazolam. However, even at low concentrations, zolpidem can cause coma in patients when taken in combination with other CNS depressants. Although single-drug poisoning is usually harmless and does not require treatment, we believe that FDA recommendations should be updated in order to make zolpidem’s use safer and a reassessment of the extended-release products is advisable. At last, the authors would like to acknowledge that they are aware of the low sample of participants in this study. Efforts were made to increase the number of patients; however, because we conducted our research employing a virtual population, we were limited to the 12 participants identified using literature graphs. To solve this challenge, the authors attempted to thoroughly evaluate the data, confirm the results using higher confidence intervals, and employ simpler models. Moreover, it may be assumed that our results are inconclusive, but we believe them to have enough value to set trends. Undoubtedly, more pharmacodynamic studies and new simulations with a bigger data size may be helpful to further confirm our results and develop appropriate design regimens for dose individualization.