Abstract
The chemical composition of the Cynophalla flexuosa hydroethanolic extract (CFHEE) was identified and its antioxidant, antifungal and antipleomorphic activities against C. albicans and C. tropicalis strains were evaluated. Phytochemical prospecting evidenced polyphenolic and triterpenoid compounds while UPLC-MS analysis revealed the presence of Isopropyl/n-propyl-GLS; Methylpropyl-GLS/butyl-GLS; Methylbutyl-GLS; Quercetin O-di-hexoside; Quercetin-pentosyl-hexoside; Rutin; Quercetin O-glucoside; Kaempferol O-rhamnosyl-hexoside; Kaempferol O-pentosyl-hexoside and Lariciresinol hexoside. The CFHEE inhibited the effect of the DPPH● free radical, both when evaluated individually and in combination with the antifungal fluconazole. When associated with 256 μg/mL fluconazole, the extract, at concentrations from 128 μg/mL, reduced the effect of the DPPH● free radical with values ranging from 37.7% to 95.8%. The extract’s antifungal effect was considered clinically irrelevant and its combination with the antifungal triggered an antagonistic effect against the two strains, thus indicating the popular use of bravo beans in the form of teas or infusions should not be combined with the intake of the drug Fluconazole as this may lead to a reduction of its clinical effect. The extract, however, inhibited morphological changes (pleomorphism) in Candida species, preventing the development of hyphae. The CFHEE possesses an antioxidant effect and a potential pharmacological activity for the inhibition of one of the Candida spp. virulence factors.
1. Introduction
Species belonging to the Candida genus are responsible for causing various fungal infections that can range from a simple superficial pathology to more serious ones, such as systemic affections, especially in immunocompromised individuals, where performing different diagnostic methods and therapeutic combinations become necessary for their treatment [1,2]. Candida albicans is the main species associated with hospitalizations due to infections with mortality rates greater than 50% in cases of septicemia [3]. Candida tropicalis also causes candidemia in debilitated individuals, being one of the main pathogens associated with candidiasis dispersion in patients with severe hematological diseases, presenting high virulence and low sensitivity to commonly used antifungals such as azoles [4,5].
In recent decades, infections caused by species from this genus have been increasing; however, their treatment is still limited due to the restricted number of available antifungals and the toxicity that they present [4,6]. Therefore, the search for new therapeutic alternatives, such as the association of antifungals with bioactive molecules, as well as the evaluation of the antifungal activity of natural products, becomes very relevant [7,8,9].
Synergistic interactions between antimicrobial agents offer improvements in the efficacy of clinical treatment, the inhibitory concentration of antibacterial agents, the risk associated with the development of bacterial resistance and the reduction of the toxic effect of antifungals to the host. Therefore, the use of associated substances has been widely studied and used as a treatment for infections [10,11,12].
At the same time, natural products can have an antioxidant effect on biological organisms. This action is linked to the chemical composition of the secondary metabolism of plants, especially phenolic compounds, such as flavonoids. Such action is responsible for eliminating or reducing free radicals that are formed during metabolic reactions. If not eliminated by enzymes or antioxidant substances, they can cause degenerative diseases of aging and cell death overtime [12,13,14].
Cynophallaflexuosa (L.) J. Presl, belonging to the Capparaceaefamily, which has Capparis flexuosa (L.) L. as a synonym, is popularly known as “feijão-bravo” and is closely related to the mustard (Cruciferae) family with an abundance of glucosinolates and flavonoids. Cynophalla flexuosa develops in the Brazilian semiarid region as a shrub or tree reaching up to 4 m in height, with straight, glabrous branches and leaves, varying from approximately 3 to 5 cm in length; the species is used to treat fevers, as a depurative, in the restoration of the menstrual flow to normal levels and against cough, in addition to possessing great nutritional value [15,16,17,18,19,20].
The C. flexuosa entire plant, its leaves, bark and wood are used by the population for producing abortive venom, as a combustible, for veterinary purposes and mainly as fodder, for staying green throughout the year and possessing chemical characteristics such as good palatability, thus being of great importance for animal feeding in drought periods [18,21].
Due to the food and medicinal use of C. flexuosa in the brazilian semi-arid region, the objective of this study was to carry out a phytochemical prospecting, the comprehensive identification of its phytochemicals and to evaluate the in vitro antioxidant and antifungal potential of the C. flexuosa hydroethanolic extract against Candida spp. strains, as well as to evaluate the ability of the extract to reduce the virulence of these fungi by affecting their morphological transition.
2. Materials and Methods
2.1. Plant Material
The C. flexuosa plant material was randomly collected from the municipality of Milagres—CE, Brazil, in October 2017. Botanical identification was carried out by the Herbarium of the Regional University of Cariri and identified by Dr Karina Vieira Alves Linhares. A voucher specimen of the species was deposited in the Herbarium Dárdano deAndrade Lima—HCDAL—URCA under number #13257.
2.2. Preparation of the Cynophalla flexuosa Hydroethanolic Extract (CFHEE)
A total of 414.0 g of freshly picked leaves of C. flexuosa were chopped and extracted at room temperature in a mixture (2000 mL) of co-solvents containing 30% of water and 70% of ethanol (v/v) [22]. The plant material remained in contact with the extractive mixture at room temperature, protected from air and light and being shaken every 12 h, for 72 h, time in which the leaves reached approximately 75% depigmentation. The resulting solution was concentrated under reduced pressure in a rota-evaporator (Q-344B—Quimis—Brazil—40 rpm, 60 °C), and was subsequently filtered and dried in a 65 °C water bath, after which, it was obtained the extract in the form of amorphous paste.
2.3. Chemical Analyses
2.3.1. Qualitative Chemical Prospection
Chemical assays were used for the qualitative analysis for the presence of secondary metabolites. In the evaluation for the presence of phenols, tannins, flavonoids and alkaloids, the method described by Matos [22] was adopted based on the visualization of color changes and the formation of precipitates after the addition of specific reagents. The Liebermann–Burchard test was used for the detection of phytosteroids and/or triterpenoids, using the method adapted from Campbell and Shawn [23]. According to this method, colorimetric changes or precipitate formation in the fraction solution after the addition of specific reagents are indicative of the presence of the corresponding class of secondary metabolites. The screening of phenolic compounds was performed using FeCl3, 1% methanolic solution, and 1 N HCl, 10% (w/v) NaOH, while the screening of alkaloids was performed using 5% (v/v) acetic acid, 10% (v/v) NH4OH, chloroform p.a., and Dragendorff’s reagent. For the screening of phytosteroids and/or triterpenoids the acetic anhydride and sulfuric acid were used as reagents of detection.
2.3.2. Total Phenol Quantification
Evaluation of the phenolic compounds present in the CFHEE was performed using the method developed by Singleton and Rossi [24], modified by Nuutila et al. [25], using the Folin–Ciocalteu reagent colorimetric method, based on the reduction of phosphomolybdic and phosphotungstic acids in alkaline solution.
A total of 200 μL of the extract, 600 μL of ethanol 70% (v/v), 400 μL of 10% (v/v) Folin–Ciocalteu reagent and 2000 μL of 7.5% (w/v) sodium carbonate solution were added to test tubes. The mixture was homogenized and another 800 μL of 7.5% (w/v) sodium carbonate solution was then added to the test tubes and they were incubated for 20 min at 45 °C in the dark. Subsequently, the sample was centrifuged in a Donner CD100 centrifuge at 3500 rpm for 5 min and left to stand for 15 min at room temperature.
The absorbance reading of the blue color produced by the reduction of the Folin–Ciocalteu reagent due to phenolic compounds was performed at 735 nm using the UV-visible T80 spectrophotometer PG Instruments LTD. Calculation of the phenolic content was performed through the preparation of a gallic acid standard curve using triplicates at concentrations ranging from 0.5 to 5 μg/mL. The extract was analyzed in triplicates, and the results obtained were expressed in milligrams of gallic acid per gram of dry extract.
2.3.3. Ultra-Performance Liquid Chromatography—ElectroSpray Ionization—Quadrupole-Time-of-Flight—Tandem Mass Spectrometry (UPLC-ESI-qTOF-MS/MS)
AnAcquity UPLC system (Waters) coupled to a Quadrupole/Time of Flight system (QtoF, Waters) was performed. Chromatographic runs were performed on a Waters Acquity UPLC BEH column C18 (150 mm × 2.1 mm, 1.7 μm) with a fixed temperature of 40 °C, mobile water phases with 0.1% formic acid (A) and acetonitrile with 0.1% formic acid (B), a gradient ranging from 2% to 95% B (15 min), a flow rate of 0.4 mL/min and injection volume of 5 μL. The ESI− mode was acquired in the 110–1180 Da range, with a fixed source temperature at 120 °C, desolvation temperature at 350 °C, desolvation gas flow of 500 L/h, an extraction cone of 0.5 V and capillary voltage of 2.6 kV. Leucine enkephalin was used as a lock mass. The acquisition mode used was MSE. The instrument was controlled by the Masslynx 4.1 Software (Waters Corporation, Taunton, MA, USA) [26].
2.4. Antioxidant Activity
The DPPH● sequestering activity of Fluconazole and the CFHEE in isolation or in combination with fluconazole was determined using the DPPH● (1,1-diphenyl-2-picrylhydrazyl) photocolorimetric method, according to Choi et al. [27], with modifications, using ascorbic acid as the standard antioxidant. DPPH● inhibition was accompanied by the disappearance of the DPPH● absorption [28], where the degree of discoloration is proportional to the concentration and potency of the present antioxidants. The tests were performed in duplicates.
A total of 20 μL of ascorbic acid, antifungal and extract solutions were resuspended separately in 80 μL of 95% ethanol and 100 μL of a 0.4 mM ethanolic DPPH● radical solution in 96-well ELISA plates; generating the final concentrations of ascorbic acid and the extract (2048, 1024, 512, 256, 128, 64, 32, 16, 8, 4 and 2 μg/mL) as well as DPPH● (0.2 mM).
A protocol was also established, for the first time, to evaluate the association of the extract with a fixed antifungal concentration. A total of 20 μL of the antifungal solution and 20 μL of the extract were resuspended in 60 μL of 95% ethanol and 100 μL of the 0.4 mM ethanolic DPPH● radical solution; generating the final antifungal (256 μg/mL), extract (2048 up to 2 μg/mL) and DPPH● (0.2 mM) concentrations. After 30 min of incubation at room temperature in the dark, absorbance measurements were taken at 518 nm using a UV-Visible TR Reader Shimadzu spectrophotometer. Under the same conditions and separately, absorbance readings for the antifungal, extract, antifungal plus the extract and ascorbic acid blanks were performed by adding 20 μL of each substance’s dissolution plus 95% ethanol up to the volume of 200 μL, generating the same concentrations for each protocol.
DPPH● free radical inhibition percentages (%) were expressed as the percentage (%) compared to the control, consisting of 100 μL of the 0.4 mM ethanolic DPPH● solution and 100 μL of 95% ethanol, calculated using the formula:
where Abs.Control is the absorbance of the DPPH● solution without the extract; Abs.Extract is the absorbance of the extract with DPPH●; Abs.Blank is the absorbance of the extract without DPPH●.
DPPH % inhibition = [Abs.Control − (Abs.Extract − Abs.Blank)] × 100%
2.5. Antifungal Activity
2.5.1. Strains, Culture Media Used and Solution Preparation
Standard strains (Candida albicans—CA INCQS 40006 and Candida tropicalis—CT INCQS 40042 yeasts) were obtained from the Reference Micro-organisms Collection in Sanitary Surveillance-CMRVS, FIOCRUZ-INCQS National Institute of Quality Control in Health, Rio de Janeiro, RJ, Brazil.
The microorganisms were inoculated into Sabouraud Dextrose Agar (SDA, KASVI) and incubated for 24 h at 37 °C. Small yeast aliquots were then transferred to test tubes, each containing 3 mL of sterile saline (0.9%). The concentration of the inoculum was standardized by comparing its turbidity with the 0.5 McFarland scale standard [29].
The prepared inoculums were thus used to determine the Minimum Inhibitory Concentration (MIC) in Dextrose Sabouraud double concentrate (SDB, HIMEDIA) broth. Potato Dextrose Agar (PDA, DIFCO), used for the micromorphology analysis, was prepared by diluting it more than the manufacturer’s recommendation to make it an impoverished medium capable of stimulating the production of hyphae. The diluted agar was added to obtain a solid medium.
Dimethyl sulfoxide (DMSO, Merck, Darmstadt, Germany) was used for the dilution of the extract while Fluconazole (Capsule-Flucomed) was diluted in water and used as reference. The matrix solution of the extract was prepared by weighing 0.05 g of the extract and diluting it in 1 mL of DMSO. To obtain the desired concentration for the assays, the extract was further diluted in sterile distilled water such that the DMSO concentration would not exert an effect on the test cells [30].
2.5.2. Determination of the 50% Fungal Inhibitory Concentration IC50
The broth microdilution method in 96-well plates was performed to determine the 50% Fungal Inhibitory Concentration. Each well was filled with 100 μL of SDB containing 10% fungal inoculum, followed by the addition of 100 μL of the CFHEE (4096 μg/mL) or Fluconazole (antifungal reference, at the same concentration) to the first well followed by serial double dilution. The concentrations in the wells ranged from 2 to 2048 μg/mL. The last well, without the extract or antifungal, served as normal growth control [31].
Controls were prepared for the product’s diluents (using saline instead of the inoculum) as well as the sterile medium. All tests were performed in triplicate. The plates were incubated at 37 °C for 24 h and subsequently read at 630 nm. The reading results were used to construct the cell viability curve and determine the CFHEE IC50 [32].
2.5.3. Determination of the Minimum Fungicidal Concentration (MFC)
A small sterile rod was placed in each well of the MIC test plate (except for the sterility control), which after mixing the medium in each well, was taken to a petri dish containing SDA, transferring the solution (medium + inoculum + CFHEE) for subculture and cell viability verification. After 24 h of incubation, the plates were inspected for any Candida colony formations [33], with modifications. The concentration in which no fungal colony growth was observed was considered the MFC of the CFHEE.
2.5.4. Extract Effect on Fungal Morphology
To determine if the CFHEE caused alterations in fungal morphology by inhibiting the development of hyphae, microculture chambers were prepared. A total of 3 mL of PDA medium depleted by dilution were added to the chambers, containing concentrations of the CFHEE at HCA/4, HCA and HCA × 2, where HCA is the Highest Concentration Assayed (HCA) in microdilution.
Inoculum aliquots were removed from the Petri dish culture to make two parallel striae in the solid medium, which were covered with a sterile coverslip. The chambers were placed in an incubator for 24 h (37 °C) and inspected under a light microscope at 400× magnification. A chamber was attached to the microscope to randomly capture images at 4× zoom. Controls for yeast growth (hyphae stimulated by exhaustion) and Fluconazole, for comparative purposes, were used [34] with modifications.
2.6. Statistical Analysis
The data obtained from each sample and concentration were checked for their normal distribution and analyzed by a one-way ANOVA followed by Bonferroni’s post-hoc test. The IC50 values were obtained by non-linear regression with the objective of interpolating standard curve values (using the Graphpad Prism 6.0 Software, Graphpad Software Inc., San Diego, CA, USA) of the percentage growth as a function of the concentrations, and the IC50 values in μg/mL.
3. Results
3.1. Extraction of Plant Material, Chemical Prospection and Total Phenols
The hydroethanolic extract of Cynophalla flexuosa (CFHEE) was obtained as a dark brown amorphous paste. The extraction showed a yield of 6.01% (w/w) which corresponds to the initial mass of plant material. The chemical characterization revealed that the medicinal species presents chemical heterogeneity arising from its secondary metabolism. In the reaction with FeCl3, the formation of a red color indicated the presence of phenols, and the formation of a green precipitate revealed the presence of condensed tannins. Color changes among red, lilac, blue, yellow and orange of the alcoholic sample, when reacting with HCl and NaOH, revealed the presence of anthocyanidins, flavones, flavonols, xanthones, chalcones, aurones, and flavononols; the same conditions reaction when subjected to heat, the sample exhibited colors ranging from red, orange and yellow, indicating the presence of leucoanthocyanidins, flavonones and catechins. The formation of a deep green color with acetic anhydride, after a few minutes, confirmed the presence of phytosteroids. The non-formation of precipitate with Dragendorff’s reagent indicated the absence of alkaloids. In addition, it was possible to note that the extract does not have pyrogallates tannin in its constitution as shown in (Table 1).

Table 1.
Phytochemical prospection of the Cynophalla flexuosa hydroethanolic extract (CFHEE).
Regarding the total of phenols, it was shown that C. flexuosa hydroethanolic extract (CFHEE) has a total of 1.53 ± 0.042 mg/g of gallic acid.
3.2. Chemical Composition by UPLC-ESI-qTOF-MS/MS
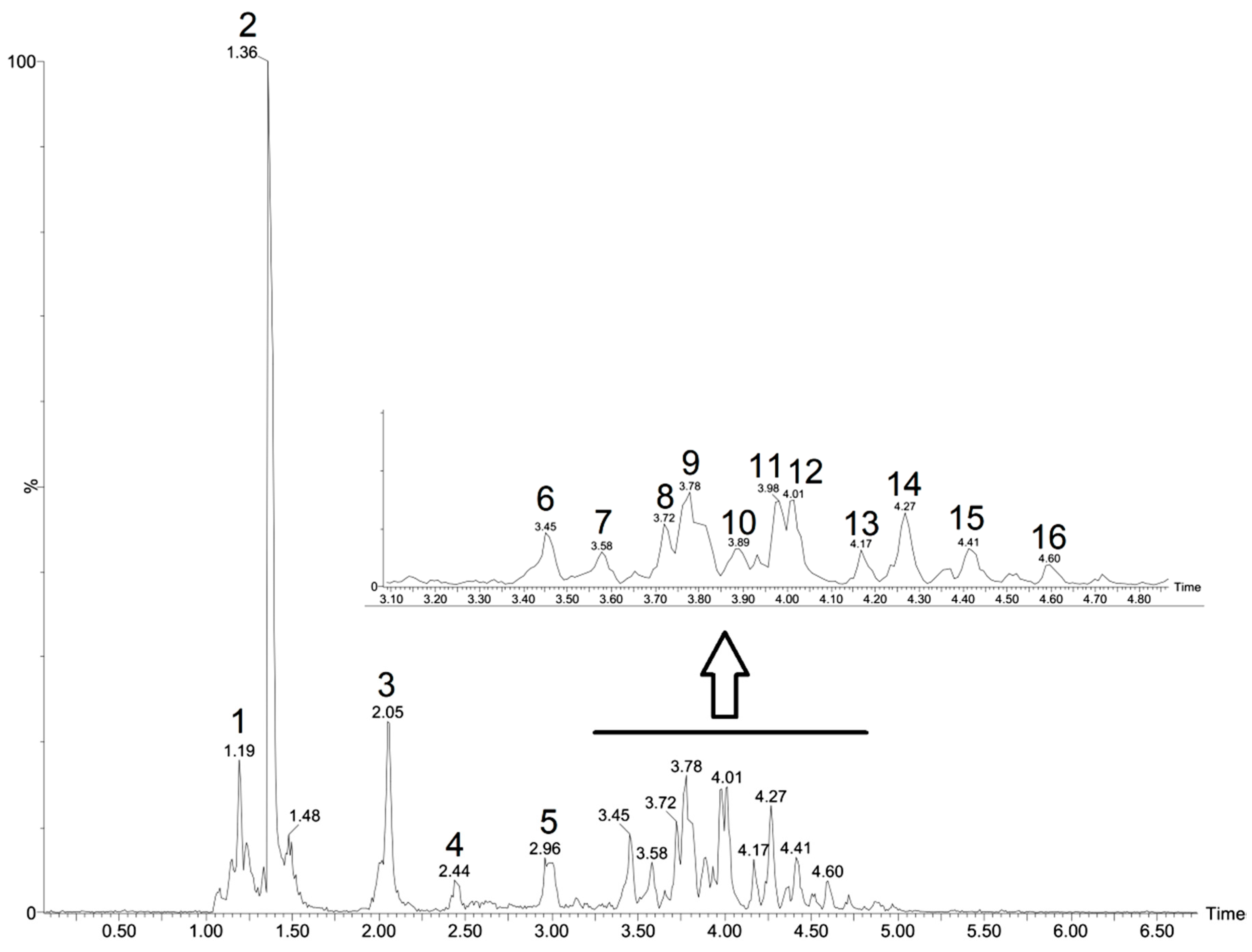
The CFHEE was analyzed using the UPLC-ESI-qTOF-MS/MS in the ESI− mode. The constituents were identified based on their exact masses, fragmentation patterns and comparison with data reported in the literature (Table 2). The chromatogram with the results from the analysis, in which the components are represented by peaks, can be visualized in Figure 1.

Table 2.
Peak assignment of metabolites in the Cynophalla flexuosa hydroethanolic extract (CFHEE) identified as intact deprotonated molecules, [M-H]−, using UPLC-ESI-qTOF-MS/MS in the negative mode. GLS are glucosinolates.
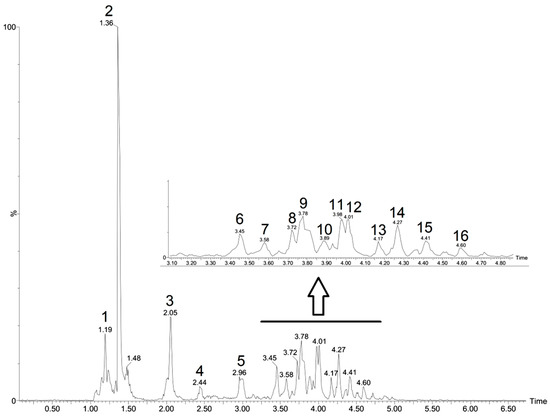
Figure 1.
Peaks attributed to metabolites in the Cynophalla flexuosa hydroethanolic extract (CFHEE) using UPLC-ESI-qTOF-MS/MS in the negative mode.
3.3. Antioxidant Activity
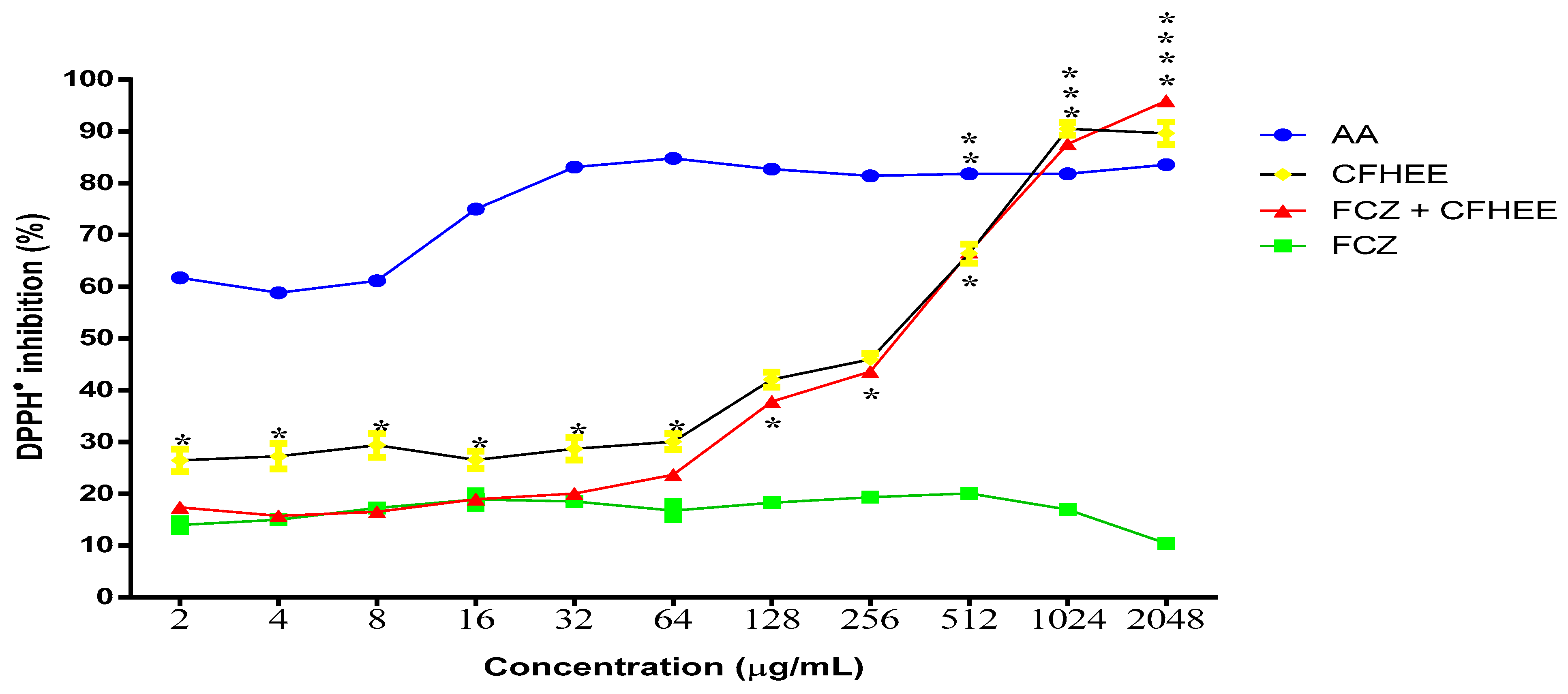
The evaluation of the CFHEE antioxidant activity and the positive control of ascorbic acid in the different reaction systems are represented in Figure 2. The extract alone possessed a DPPH● free radical sequestering activity with an IC50 of 124 μg/mL and when in combination with the antifungal Fluconazole it presented an IC50 of 196.6 μg/mL (Table 3). On the other hand, the antifungal Fluconazole did not exhibit significant antioxidant activity compared to the extract and the ascorbic acid control. AA obtained a superior antioxidant activity, obtaining an IC50 value lower than 2 μg/mL. At the two highest concentrations, the extract alone and in association with the antifungal showed a greater inhibitory potential than AA.
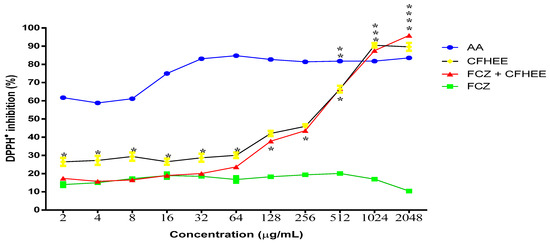
Figure 2.
Free radical scavenging activity of the Cynophalla flexuosa hydroethanolic extract (CFHEE) as measured by the DPPH• 1,1-Diphenyl—2 picryhydrazyl (0.2 Mm) free radical assay, with ascorbic acid as a positive control. The values from the free radical scavenging activity were expressed as mean ± S.E.M. (n = 8). Non-linear regression of the curve transformations, and analyzed by one-way ANOVA followed by Tukey’s multiple comparison test, with a simple pooled variance. * Statistical significance with p < 0.05; ** Statistical significance with p < 0.01; *** Statistical significance with p < 0.001; **** Statistical significance with p < 0.

Table 3.
Inhibitory concentration 50% (IC50) of several products against the DPPH● free radical.
The extract in combination with Fluconazole exhibited a significantly higher antioxidant activity from the concentration of 64 μg/mL compared to the antifungal alone. This may have occurred because the antifungal possesses a pro-oxidant effect at the tested concentration (256 μg/mL), which was more expressive than the antioxidant effect of the extract up until the 64 μg/mL concentration. From this concentration onwards until reaching the 2048 μg/mL concentration, the extract exhibited an antioxidant activity that significantly reversed the antifungal effect, thus inhibiting the DPPH● radical.
The effect of Fluconazole on reducing the antioxidant potential of the extract is confirmed by observing that, in the absence of an association, Fluconazole showed a significantly higher antioxidant activity up to the 64 μg/mL concentration. At the 128 μg/mL concentration, the extract associated with Fluconazole inhibited DPPH● by 37.79% and when evaluated insolation it inhibited DPPH● by 42%, with no significant differences in percentage inhibition between these two systems being observed until the 1024 μg/mL concentration. This demonstrates that the extract reversed the effect of Fluconazole and still managed to significantly inhibit the DPPH● free radical. When associated with the antifungal at a concentration of 2048 μg/mL, the extract inhibited DPPH● by 95.84%, compared to 89.62% DPPH● inhibition with the isolated extract.
3.4. Antifungal Activity
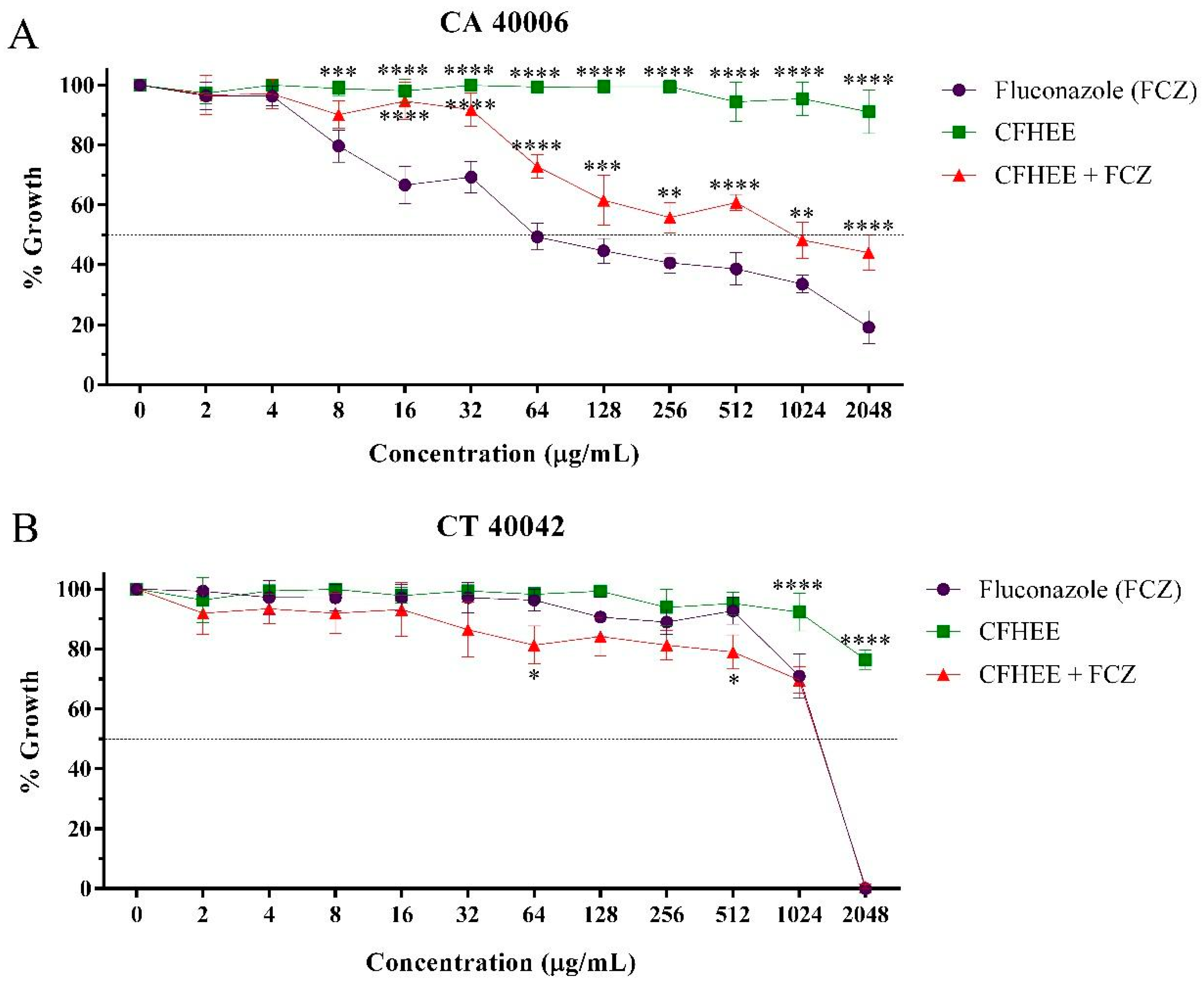
The CFHEE showed an inhibitory effect on the growth of resistant Candida strains when used at high concentrations with a MIC value equal to or greater than 4096 μg/mL. The evaluation of the potential modulatory activity of the extract on the Fluconazole antifungal effect against different Candida strains revealed the combination of this drug with the hydroethanolic extract, at subinhibitory concentrations, causes the inhibition of microorganismal growth at concentrations greater than when it was tested in isolation. This effect was also verified in the cell viability curve (Figure 3).
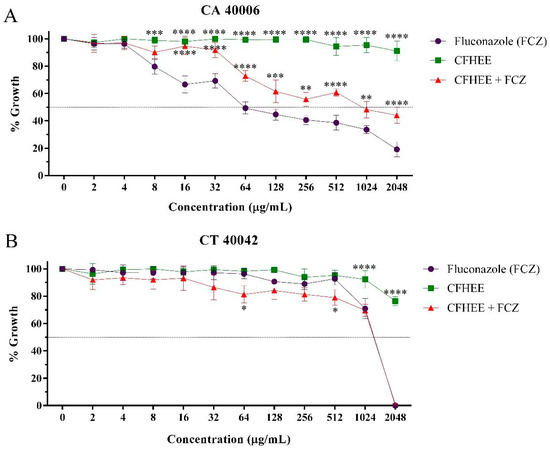
Figure 3.
Antifungal effect of fluconazole and its association with the Cynophalla flexuosa hydroethanolic extract (CFHEE) (MIC/16 μg/mL). FCZ: Fluconazole. (A) CA: Candida albicans; (B) CT: Candida tropicalis. These values were expressed as the means ± SEM (n = 3) and analyzed by a one-way ANOVA followed by Bonferroni post-hoc test multiple comparisons. * Significant difference (p < 0.05) at equal concentrations. * Statistical significance with p < 0.05; ** Statistical significance with p < 0.01; *** Statistical significance with p < 0.001; **** Statistical significance with p < 0.
The IC50 values (Table 4) obtained in the evaluation showed that CFHEE possesses a high inhibitory concentration compared to Fluconazole and, when in combination with this drug, the extract significantly increased its IC50 against C. albicans, revealing an antagonism. A small reduction in the IC50 of Fluconazole, however without significance, was observed when this was associated with the extract against C. tropicalis.

Table 4.
50% Candida spp. Inhibitory Concentration (IC50—μg/mL) by the Cynophalla flexuosa hydroethanolic extract (CFHEE).
Concentrations ranging from 2 to 2048 μg/mL were used to determine the fungicidal effect of the CFHEE. After 24 h of incubation, fungal growth was observed at all concentrations, showing the MFC was greater than 4096 μg/mL.
3.5. Control of Virulence of Candida Strains
In experiments evaluating the action of the extract on fungal morphology, two controls were used: microorganismal growth, in order to evaluate if the medium is ideal for the induction of pleomorphism; and the control for comparative verification of the effect of the antifungal Fluconazole, as shown in Figure 4. The growth controls for both strains were shown to be effective.

Figure 4.
Controls used for the Candida albicans and Candida tropicalis strains in fungal pleomorphism assays with the Cynophalla flexuosa hydroethanolic extract. HCA: Highest Concentration Assayed (2048 µg/mL); HCAx2 (4096 µg/mL) and HCA/4 (512 µg/mL); CA: Candida albicans; CT: Candida tropicalis; INCQS: National Institute of Health Quality Control.
In the Fluconazole and Extract Assays, the Highest Concentration Assayed—HCA of the microdilution (2048 μg/mL), HCAx2 (4096 μg/mL) and HCA/4 (512 μg/mL) were tested. Fluconazole significantly reduced the formation of hyphae and pseudohyphae as the concentration increased, especially against the C. albicans strain (INCQS 40006).
The images from the microcultures using the extract are shown in Figure 5, where the effective action of the CFHEE on C. albicans and C. tropicalis strains can be observed as the concentration increases. For C. tropicalis, the action can be observed from lower concentrations.

Figure 5.
Effect of the Cynophalla flexuosa hydroethanolic extract (CFHEE) on C. albicans and C. tropicalis fungal dimorphism. HCA: Highest Concentration Assayed (2048 µg/mL); HCAx2 (4096 µg/mL) and HCA/4 (512 µg/mL); CA: Candida albicans; CT: Candida tropicalis; INCQS: National Institute of Health Quality Control.
4. Discussion
The presence of isopropyl/propyl-GLS in the CFHEE, established a chemical relationship with the Cynophalla ovata and Cynophalla spinosa species, in which glucosinolates (GLS) were also reported [36,44,45]. These compounds demonstrated high potent antifungal effect [46]; however, the CFHEE did not exhibit antifungal activity; on the contrary, it was antagonistic in association with fluconazole, which could be due to the low isopropyl/propyl-GLS concentration. However, the antifungal effect of these compounds will be further evaluated.
The total polyphenolic content of the CFHEE was relatively low at 1.53 mg GA/g. DE, however, when associated with fluconazole, values of up to 95.8% of DPPH● radical scavenging were observed. Such values may mitigate the possible pro-oxidant effect of fluconazole against fungal strains, which could also be a component of the antibiotic properties of the antibiotic, giving rise to its antagonistic behavior in the antifungal action modulation, therefore, such result could be associated with the presence of antioxidant compounds such as the flavonoids, which possess antioxidant activities widely reported in the literature and associated with the dihydroxy (catechol) structure in ring B, which acts to form o-quinones when reacting with free radicals, the 3-hydroxyl group on the C-ring and the 2,3-double bond in conjunction with the oxo ring C function [46,47,48]. Such a structural arrangement makes them labile in transferring electrons and donating protons, which characterizes them as standard antioxidants through their radical elimination activities in innumerable in vitro and in vivo studies [49,50,51].
Previous studies have reported the resistance of Candida species to fluconazole [5,6,52,53]. The resistance of this genus to azoles is shown by the decrease in expression of the enzyme lanosterol-14α-azol-demethylase or the presence of specific mutations that affect its structure and function, preventing the destabilization of the fungal membrane [54]. In another study, β-sitosterol is reported to exhibit a rapid activity against fungal cells, indicating that it may be a good antimicrobial agent [55]. Cowan, [56] reported the antimicrobial potency of β-sitosterol may be associated with the membrane rupture of susceptible organisms. However, the presence of β-sitosterol in the CFHEE did not contribute to an antifungal activity, leading to the assumption that it did not modify the cell membrane structure to favor the entry of fluconazole, flavonoids and GLS or antifungal constituents to the interior of fungal cells, and thus can be possible to trigger effective antifungal effects.
A high concentration of flavonoids in the C. spinosa hydroethanolic extract were found [57], and it has been considered a very important source of antifungal medications [58]. The aforementioned species presents some secondary metabolites in common with C. flexuosa. However, despite the similarities, the CFHEE presented no direct or synergistic antifungal effect with fluconazole. One reason for these results may be that flavonoid levels in the extract were not elevated (1.53 mg GA/g. DE).
In Figure 3, the graphs present the antagonistic effect observed by the union of the antifungal with the extract against C. albicans (color: red). The combination of the substances caused a decrease in the effect of the drug, this being an indication that the popular use of bravo beans, in the form of teas or infusions, should not be combined with the intake of the drug Fluconazole, due to the reduction of its clinical effect.
One of the virulence factors observed in species from the Candida genus are their capacity for morphological transition or pleomorphism. This feature endows the microorganism with the ability to change its shape from a yeast-like state to a filamentous state (hyphae and pseudohyphae) when subjected to environmental stress conditions with the possibility of becoming a tissue invasive organism in the host [59]. This change in state favors the formation of systemic diseases termed candidemias that affect people who present compromises in their immunological responses [60] and the search for less toxic natural substances to control or prevent morphological transition without the death of the commensal fungal population, which is essential for balance, has increased. The CFHEE inhibited pleomorphic transition of the tested strains with C. tropicalis obtaining the best effect when compared to C. albicans and the antifungal Fluconazole. Although C. albicans is responsible for more than 50% of superficial and systemic infections caused by yeasts [61], infections by non-albicans species have increased and C. tropicalis stands out for being characterized as an agent capable of causing invasive infections [62].
The CFHEE did not present significant intrinsic antifungal activity; however, it inhibited morphological alterations in Candida species, indicating it possesses potential pharmacological activity against fungal biology, potentially revealing an alternative for the treatment of fungal infections [63].
5. Conclusions
The medicinal species C. flexuosa presents chemical heterogeneity in its composition, presenting mainly phenolic compounds. These being the possible responsible for the antioxidant activity. Despite the lack of antifungal activity, the leaves are sources of phytochemicals capable of inhibiting the formation of hyphae of Candida strains. To advance the improvement of its use as an antifungal medicinal plant, complete elucidation of the mechanisms of action require more in-depth study. We emphasize that the species under study possesses potential for future research associated with antioxidant and antifungal activity, using new methodologies, different microorganisms in new assays and in combination with other drugs, in order to evaluate their use for the treatment of infections and reversal of fungal resistance.
Author Contributions
Conceptualization, J.N.P.C. and G.J.T.S.; methodology, A.C.A.d.S., B.G.C., R.O.M.d.S. and R.P.d.C.; investigation, J.C.P.d.S. and A.T.L.d.S.; writing—review and editing, D.L.S., M.H.F. and V.B.d.S.; resources, J.G.M.d.C., K.V.L. and H.D.M.C.; software, J.W.A.-B.; data curation, J.C.A., E.S.d.B. and P.R.V.R.; supervision, M.F.B.M.-B.; project administration, H.D.M.C. and M.F.B.M.-B.; funding acquisition, H.D.M.C. and M.F.B.M.-B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
This work was carried out by the Laboratory of Applied Mycology of Cariri, Laboratory of Microbiology and Molecular Biology and Laboratory of Natural Products Research of the Regional University of Cariri—URCA. We thank the Brazilian Agency for the Coordination of Improvement of Higher Education Personnel (CAPES) and the National Council for Scientific and Technological Development (CNPq).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Arendrup, M.C.; Patterson, T.F. Multidrug-Resistant Candida: Epidemiology, Molecular Mechanisms, and Treatment. J. Infect. Dis. 2017, 216, S445–S451. [Google Scholar] [CrossRef] [PubMed]
- Romo, J.A.; Kumamoto, C.A. On Commensalism of Candida. J. Fungi 2020, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- Sustr, V.; Foessleitner, P.; Kiss, H.; Farr, A. Vulvovaginal Candidosis: Current Concepts, Challenges and Perspectives. J. Fungi. 2020, 6, 267. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.-Y.; Chuang, Y.-C.; Wu, U.-I.; Sun, H.-Y.; Wang, J.-T.; Sheng, W.-H.; Chen, Y.-C.; Chang, S.-C. Mechanisms of Azole Resistance and Trailing in Candida Tropicalis Bloodstream Isolates. J. Fungi. 2021, 7, 612. [Google Scholar] [CrossRef]
- Liu, W.-L.; Huang, Y.-T.; Hsieh, M.-H.; Hii, M.; Lee, Y.-L.; Ho, M.-W.; Liu, C.-E.; Chen, Y.-H.; Wang, F.-D. Clinical Characteristics of Candida tropicalis Fungaemia with Reduced Triazole Susceptibility in Taiwan: A Multicentre Study. Int. J. Antimicrob. Agents 2019, 53, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Nami, S.; Aghebati-Maleki, A.; Morovati, H.; Aghebati-Maleki, L. Current Antifungal Drugs and Immunotherapeutic Approaches as Promising Strategies to Treatment of Fungal Diseases. Biomed. Pharmacother. 2019, 110, 857–868. [Google Scholar] [CrossRef]
- Bezerra, J.W.A.; Costa, A.R.; de Freitas, M.A.; Rodrigues, F.C.; de Souza, M.A.; da Silva, A.R.P.; Dos Santos, A.T.L.; Linhares, K.V.; Coutinho, H.D.M.; de Lima, J.R.S.; et al. Chemical Composition, Antimicrobial, Modulator and Antioxidant Activity of Essential Oil of Dysphania Ambrosioides (L.) Mosyakin & Clemants. Comp. Immunol. Microb. 2019, 65, 58–64. [Google Scholar]
- Costa, A.R.; de Lima Silva, J.R.; de Oliveira, T.J.S.; da Silva, T.G.; Pereira, P.S.; de Oliveira Borba, E.F.; de Brito, E.S.; Ribeiro, P.R.V.; Almeida-Bezerra, J.W.; Júnior, J.T.C.; et al. Phytochemical Profile of Anacardium Occidentale L. (Cashew Tree) and the Cytotoxic and Toxicological Evaluation of Its Bark and Leaf Extracts. S. Afr. J. Bot. 2020, 135, 355–364. [Google Scholar] [CrossRef]
- Zida, A.; Bamba, S.; Yacouba, A.; Ouedraogo-Traore, R.; Guiguemdé, R.T. Anti-Candida Albicans Natural Products, Sources of New Antifungal Drugs: A Review. J. Mycol. Med. 2017, 27, 1–19. [Google Scholar] [CrossRef]
- Bezerra, C.F.; de Alencar Júnior, J.G.; Honorato, R.L.; dos Santos, A.T.L.; da Silva, J.C.P.; da Silva, T.G.; de Freitas, T.S.; Vieira, T.A.T.; Bezerra, M.C.F.; Sales, D.L.; et al. Antifungal Effect of Liposomal α-Bisabolol and When Associated with Fluconazole. Cosmetics 2021, 8, 28. [Google Scholar] [CrossRef]
- Ncube, B.; Finnie, J.F.; Van Staden, J. In Vitro Antimicrobial Synergism within Plant Extract Combinations from Three South African Medicinal Bulbs. J. Ethnopharmacol. 2012, 139, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.L.; Pereira, P.S.; Oliveira, C.V.B.; Freitas, M.A.; Silva, J.R.L.; Costa, A.R.; Oliveira-Tintino, C.D.M.; Morais-Braga, F.B.M.; Duarte, A.E.; Coutinho, H.D.M.; et al. Study of the Capacity of the Essential Oil of Lantana Montevidensis to Modulate the Action of Fluconazole on Candida Albicans and Candida Tropicalis Strains. J. Med. Mycol. 2021, 31, 101171. [Google Scholar] [CrossRef]
- Laxa, M.; Liebthal, M.; Telman, W.; Chibani, K.; Dietz, K.-J. The Role of the Plant Antioxidant System in Drought Tolerance. Antioxidants 2019, 8, 94. [Google Scholar] [CrossRef] [PubMed]
- Snezhkina, A.V.; Kudryavtseva, A.V.; Kardymon, O.L.; Savvateeva, M.V.; Melnikova, N.V.; Krasnov, G.S.; Dmitriev, A.A. ROS Generation and Antioxidant Defense Systems in Normal and Malignant Cells. Oxid. Med. Cell. Longev. 2019, 2019, e6175804. [Google Scholar] [CrossRef] [PubMed]
- Agra, M.F.; Baracho, G.S.; Nurit, K.; Basílio, I.J.L.D.; Coelho, V.P.M. Medicinal and Poisonous Diversity of the Flora of “Cariri Paraibano”, Brazil. J. Ethnopharmacol. 2007, 111, 383–395. [Google Scholar] [CrossRef]
- Inocencio, C.; Rivera, D.; Alcaraz, F.; Tomás-Barberán, F.A. Flavonoid Content of Commercial Capers (Capparis Spinosa, C. Sicula and C. Orientalis) Produced in Mediterranean Countries. Eur. Food. Res. Technol. 2000, 212, 70–74. [Google Scholar] [CrossRef]
- Kiddle, G.; Bennett, R.N.; Botting, N.P.; Davidson, N.E.; Robertson, A.A.B.; Wallsgrove, R.M. High-Performance Liquid Chromatographic Separation of Natural and Synthetic Desulphoglucosinolates and Their Chemical Validation by UV, NMR and Chemical Ionisation-MS Methods. Phytochem. Analysis. 2001, 12, 226–242. [Google Scholar] [CrossRef]
- Almeida Neto, J.X.; Andrade, A.P.; Lacerda, A.V.; Félix, L.P.; Silva, D.S. Crescimento e Bromatologia Do Feijão-Bravo (Capparis flexuosa L.) Em Área de Caatinga No Curimataú Paraibano, Brasil. Rev. Cienc. Agron. 2011, 42, 488–494. [Google Scholar] [CrossRef]
- Ribeiro, D.A.; Macêdo, D.G.; Oliveira, L.G.S.; Saraiva, M.E.; Oliveira, S.F.; Souza, M.M.A.; Menezes, I.R.A. Potencial Terapêutico e Uso de Plantas Medicinais Em Uma Área de Caatinga No Estado Do Ceará, Nordeste Do Brasil. Rev. Bras. Plantas Med. 2014, 16, 912–930. [Google Scholar] [CrossRef]
- Yazbek, P.B.; Tezoto, J.; Cassas, F.; Rodrigues, E. Plants Used during Maternity, Menstrual Cycle and Other Women’s Health Conditions among Brazilian Cultures. J. Ethnopharmacol. 2016, 179, 310–331. [Google Scholar] [CrossRef] [PubMed]
- Leite, A.P.; Pedrosa, K.M.; Lucena, C.M.; Kelly, T.; Félix, L.P.; Lucena, R.F.P. Uso e conhecimento de espécies vegetais úteis em uma comunidade rural no Vale do Piancó (Paraíba, Nordeste, Brasil). Rev. Biol. Farm. 2012, 5, 133–157. [Google Scholar]
- Matos, F.J.A. Introduction to Experimental Phytochemistry, 3rd ed.; UFC: Fortaleza, Brasil, 2009. [Google Scholar]
- Campbell, M.K.; Farrell, S.O. Biochemistry, 4th ed.; Thomson Asia Pte Ltd.: Singapore, 2005. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1996, 16, 144–158. [Google Scholar]
- Nuutila, A.M.; Puupponen-Pimiä, R.; Aarni, M.; Oksman-Caldentey, K.-M. Comparison of Antioxidant Activities of Onion and Garlic Extracts by Inhibition of Lipid Peroxidation and Radical Scavenging Activity. Food Chem. 2003, 81, 485–493. [Google Scholar] [CrossRef]
- Silva, V.B.; Almeida-Bezerra, J.W.; Brito, E.S.; Ribeiro, P.R.V.; Cordeiro, L.S.; Júnior, J.T.C.; Costa, J.G.M.; Silva, M.A.P. Effect of Decomposition of Leaves of Azadirachta Indica A. Juss. on Germination and Growth of Myracrodruon Urundeuva Allemão. S. Afr. J. Bot. 2021, 142, 42–52. [Google Scholar] [CrossRef]
- Choi, J.S.; Lee, H.J.; Kang, S.S. Alatemin, Cassiaside and Rubrofusarin Gentiobioside, Radical Scavenging Principles from the Seeds of Cassia tora on 1,1-Diphenyl-2-Picrylhydrazyl (DPPH) Radical. Arch. Pharm. Res. 1994, 17, 462–466. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.-E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Aprovada, N. Método de Referência para Testes de Diluicão em Caldo para Determinacão da Sensibilidade à Terapia Antifúngica das leveduras; Norma Aprovada; NCCLS Norma M27-A2, 2ª ed.; NCCLS: Wayne, PA, USA, 2002; pp. 19087–19898. ISBN 1-56238-469-4. [Google Scholar]
- Stoppa, M.A.; Casemiro, L.A.; Vinholis, A.H.C.; Cunha, W.R.; Silva, M.L.A.; Martins, C.H.G.; Furtado, N.A.J.C. Comparative study of the recommended CLSI and EUCAST methodologies for evaluation of antifungal activity. Quím. Nova 2009, 32, 498–502. [Google Scholar] [CrossRef]
- Javadpour, M.M.; Juban, M.M.; Lo, W.-C.J.; Bishop, S.M.; Alberty, J.B.; Cowell, S.M.; Becker, C.L.; McLaughlin, M.L. De Novo Antimicrobial Peptides with Low Mammalian Cell Toxicity. J. Med. Chem. 1996, 39, 3107–3113. [Google Scholar] [CrossRef]
- Morais-Braga, M.F.B.; Carneiro, J.N.; Machado, A.J.; Sales, D.L.; Brito, D.I.; Albuquerque, R.S.; Boligon, A.A.; Athayde, M.L.; Calixto Junior, J.T.; Souza, D.S.; et al. High-Performance Liquid Chromatography-Diodic Array Detector, Fungistatic, and Anti-Morphogenical Analysis of Extracts from Psidium Brownianum Mart. Ex DC. against Yeasts of the Genus Candida. Int. J. Food. Prop. 2016, 19, 1837–1851. [Google Scholar] [CrossRef]
- Ernst, E.J.; Klepser, M.E.; Ernst, M.E.; Messer, S.A.; Pfaller, M.A. In Vitro Pharmacodynamic Properties of MK-0991 Determined by Time-Kill Methods. Diagn. Microbiol. Infect. Dis. 1999, 33, 75–80. [Google Scholar] [CrossRef]
- Sidrin, J.J.C.; Rocha, M.F.G. Medical Mycology in the Light of Contemporary Authors, 1st ed.; Guanabara Koogan: Rio de Janeiro, Brasil, 2010. [Google Scholar]
- Silva, G.S.; Canuto, K.M.; Ribeiro, P.R.V.; de Brito, E.S.; Nascimento, M.M.; Zocolo, G.J.; Coutinho, J.P.; de Jesus, R.M. Chemical Profiling of Guarana Seeds (Paullinia cupana) from Different Geographical Origins Using UPLC-QTOF-MS Combined with Chemometrics. Food Res. Int. 2017, 102, 700–709. [Google Scholar] [CrossRef] [PubMed]
- Bianco, G.; Lelario, F.; Battista, F.G.; Bufo, S.A.; Cataldi, T.R.I. Identification of Glucosinolates in Capers by LC-ESI-Hybrid Linear Ion Trap with Fourier Transform Ion Cyclotron Resonance Mass Spectrometry (LC-ESI-LTQ-FTICR MS) and Infrared Multiphoton Dissociation. J. Mass Spectrom. 2012, 47, 1160–1169. [Google Scholar] [CrossRef] [PubMed]
- Višnjevec, A.M.; Tamayo Tenorio, A.; Steenkjær Hastrup, A.C.; Hansen, N.M.L.; Peeters, K.; Schwarzkopf, M. Glucosinolates and Isothiocyantes in Processed Rapeseed Determined by HPLC-DAD-QTOF. Plants 2021, 10, 2548. [Google Scholar] [CrossRef] [PubMed]
- Kruszka, D.; Sawikowska, A.; Selvakesavan, R.K.; Krajewski, P.; Kachlicki, P.; Franklin, G. Silver Nanoparticles Affect Phenolic and Phytoalexin Composition of Arabidopsis thaliana. Sci. Total Environ. 2020, 716, 135361. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Hoyos, M.; Arnáez-Serrano, E.; Quesada-Mora, S.; Azofeifa-Cordero, G.; Wilhelm-Romero, K.; Quirós-Fallas, M.I.; Alvarado-Corella, D.; Vargas-Huertas, F.; Sánchez-Kopper, A. HRMS Characterization, Antioxidant and Cytotoxic Activities of Polyphenols in Malus domestica Cultivars from Costa Rica. Molecules 2021, 26, 7367. [Google Scholar] [CrossRef]
- Choucry, M.A.; Khalil, M.N.; El Awdan, S.A. Protective Action of Crateva nurvala Buch. Ham Extracts against Renal Ischaemia Reperfusion Injury in Rats via Antioxidant and Anti-Inflammatory Activities. J. Ethnopharmacol. 2018, 214, 47–57. [Google Scholar] [CrossRef]
- Pandey, R.; Chandra, P.; Arya, K.R.; Kumar, B. Development and Validation of an Ultra High Performance Liquid Chromatography Electrospray Ionization Tandem Mass Spectrometry Method for the Simultaneous Determination of Selected Flavonoids in Ginkgo Biloba. J. Sep. Sci. 2014, 37, 3610–3618. [Google Scholar] [CrossRef]
- Mosić, M.; Trifković, J.; Vovk, I.; Gašić, U.; Tešić, Ž.; Šikoparija, B.; Milojković-Opsenica, D. Phenolic Composition Influences the Health-Promoting Potential of Bee-Pollen. Biomolecules 2019, 9, 783. [Google Scholar] [CrossRef]
- Medeiros, J.L.; de Almeida, T.S.; Lopes Neto, J.J.; Almeida Filho, L.C.P.; Ribeiro, P.R.V.; Brito, E.S.; Morgano, M.A.; da Silva, M.G.; Farias, D.F.; Carvalho, A.F.U. Chemical Composition, Nutritional Properties, and Antioxidant Activity of Licania tomentosa (Benth.) Fruit. Food Chem. 2020, 313, 126117. [Google Scholar] [CrossRef]
- Bor, M.; Ozkur, O.; Ozdemir, F.; Turkan, I. Identification and Characterization of the Glucosinolate–Myrosinase System in Caper (Capparis Ovata Desf.). Plant Mol. Biol. Rep. 2009, 27, 518–525. [Google Scholar] [CrossRef]
- Matthäus, B.; Özcan, M. Glucosinolate Composition of Young Shoots and Flower Buds of Capers (Capparis Species) Growing Wild in Turkey. J. Agric. Food Chem. 2002, 50, 7323–7325. [Google Scholar] [CrossRef]
- Montoro, P.; Braca, A.; Pizza, C.; De Tommasi, N. Structure–Antioxidant Activity Relationships of Flavonoids Isolated from Different Plant Species. Food Chem. 2005, 92, 349–355. [Google Scholar] [CrossRef]
- Rösch, D.; Bergmann, M.; Knorr, D.; Kroh, L.W. Structure—Antioxidant Efficiency Relationships of Phenolic Compounds and Their Contribution to the Antioxidant Activity of Sea Buckthorn Juice. J. Agric. Food Chem. 2003, 51, 4233–4239. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-Antioxidant Activity Relationships of Flavonoids and Phenolic Acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Sato, Y.; Itagaki, S.; Kurokawa, T.; Ogura, J.; Kobayashi, M.; Hirano, T.; Sugawara, M.; Iseki, K. In Vitro and in Vivo Antioxidant Properties of Chlorogenic Acid and Caffeic Acid. Int. J. Pharmaceut. 2011, 403, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Heleno, S.A.; Martins, A.; Queiroz, M.J.R.P.; Ferreira, I.C.F.R. Bioactivity of Phenolic Acids: Metabolites versus Parent Compounds: A Review. Food Chem. 2015, 173, 501–513. [Google Scholar] [CrossRef]
- Embuscado, M.E. Spices and Herbs: Natural Sources of Antioxidants—A Mini Review. J. Funct. Foods 2015, 18, 811–819. [Google Scholar] [CrossRef]
- Fica, A. Tratamiento de Infecciones Fúngicas Sistémicas Primera Parte: Fluconazol, Itraconazol y Voriconazol. Rev. Chil. Infectol. 2004, 21, 26–38. [Google Scholar] [CrossRef][Green Version]
- Goel, N.; Gagneja, D.; Chaudhary, U.; Agarwal, R. Fluconazole Resistance among Candida Species Causing Vulvovaginitis. Res. Rev. A J. Microbiol. Virol. 2012, 2, 1–12. [Google Scholar]
- Costa, L.C.; Alves, S.F.; Nogueira, S.A.; Carvalho, G.K. Determination of fluconazole content of magistral and industrial capsules. Rev. Eletron. Fac. Montes Belos 2014, 7, 47–56. [Google Scholar]
- Odiba, J.; Musa, A.; Hassan, H.; Yahay, S.; Okolo, E. Antimicrobial Activity of Isolated Stigmast-5-En-3-β-Ol (β-Sitosterol) from Honeybee Propolis from North-Western, Nigeria. Int. J. Pharm. Sci. Res. 2014, 5, 908–918. [Google Scholar]
- Cowan, M.M. Plant Products as Antimicrobial Agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef] [PubMed]
- Bakr, R.O.; Bishbishy, E.; Helmy, M. Profile of Bioactive Compounds of Capparis Spinosa Var. Aegyptiaca Growing in Egypt. Rev. Bras. Farmacogn. 2016, 26, 514–520. [Google Scholar] [CrossRef]
- Ali-Shtayeh, M.S.; Abu Ghdeib, S.I. Antifungal Activity of Plant Extracts against Dermatophytes. Mycoses 1999, 42, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Su, C.; Liu, H. Candida albicans Hyphal Initiation and Elongation. Trends. Microbiol. 2014, 22, 707–714. [Google Scholar] [CrossRef]
- Morais-Braga, M.F.; Carneiro, J.N.; Machado, A.J.; Sales, D.L.; Dos Santos, A.T.; Boligon, A.A.; Athayde, M.L.; Menezes, I.R.; Souza, D.S.; Costa, J.G.; et al. Phenolic Composition and Medicinal Usage of Psidium Guajava Linn.: Antifungal Activity or Inhibition of Virulence? Saudi J. Biol. Sci. 2017, 24, 302–313. [Google Scholar] [CrossRef]
- Silva-Dias, A.; Miranda, I.M.; Branco, J.; Monteiro-Soares, M.; Pina-Vaz, C.; Rodrigues, A.G. Adhesion, Biofilm Formation, Cell Surface Hydrophobicity, and Antifungal Planktonic Susceptibility: Relationship among Candida spp. Front. Microbiol. 2015, 6, 205. [Google Scholar] [CrossRef]
- Deorukhkar, S.C.; Saini, S.; Mathew, S. Virulence Factors Contributing to Pathogenicity of Candida Tropicalis and Its Antifungal Susceptibility Profile. Int. J. Food Microbiol. 2014, 2014, 456878. [Google Scholar]
- Freitas, M.A.; Santos, A.T.; Machado, A.J.; Silva, A.R.P.; Campina, F.F.; Costa, M.S.; Martins, G.M.; Morais-Braga, M.F.B.; Tintino, S.R.; Menezes, I.R.; et al. Fern Extracts Potentiate Fluconazole Activity and Inhibit Morphological Changes in Candida Species. Asian Pac. J. Trop. Biomed. 2017, 7, 1025–1030. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).