Microalgae as an Eco-Friendly and Functional Ingredient for Sustainable Aquafeed
Abstract
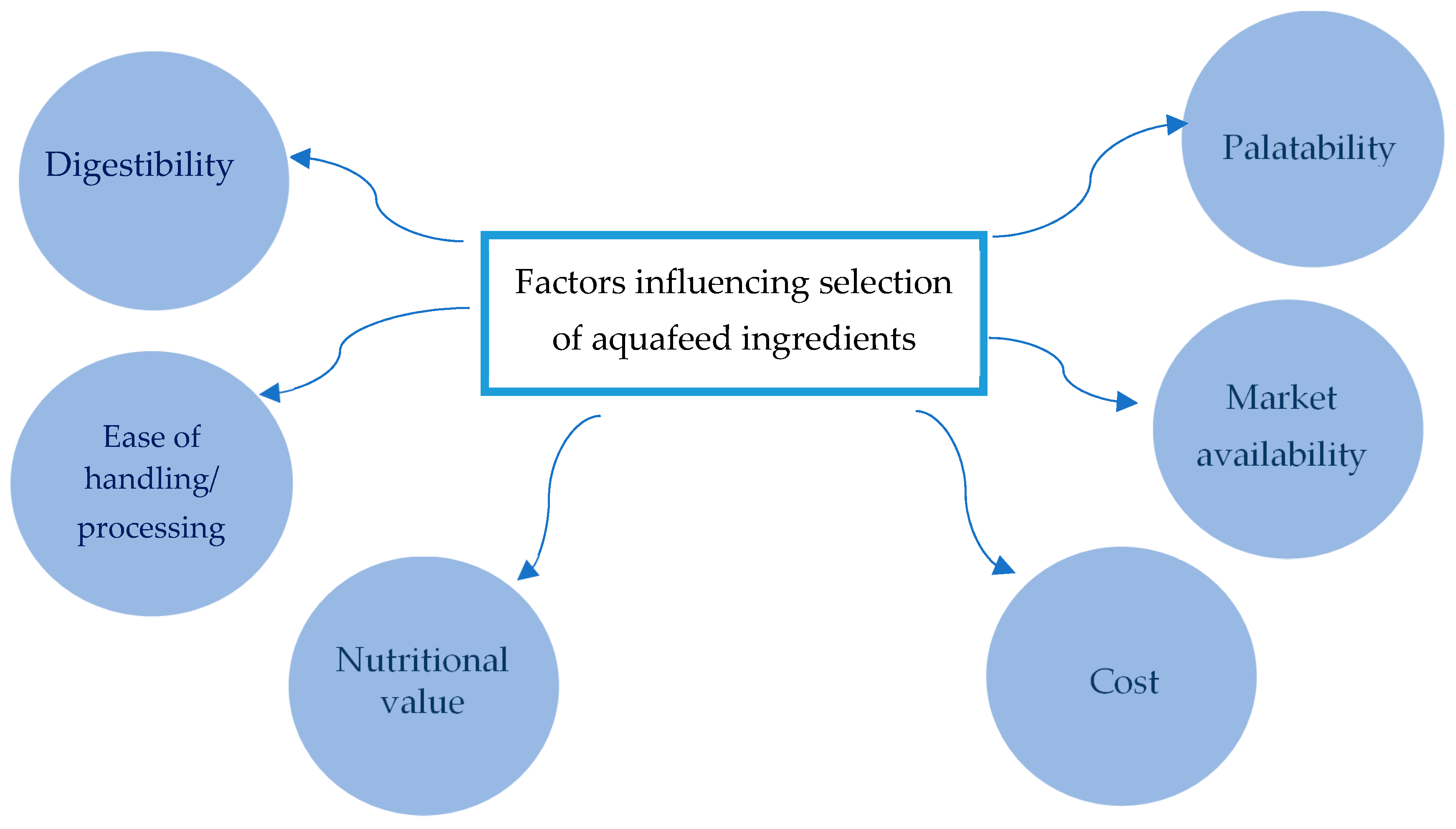
1. Introduction
2. Microalgae as Aquaculture Feed
2.1. Bioactive and Nutritional Components from Microalgae for Functional Aquafeed Applications
2.1.1. Protein
2.1.2. Fatty Acids
2.1.3. Carbohydrates
2.1.4. Pigment Components
2.1.5. Vitamins
2.2. Microalgae in Aquaculture-Current and Future Trends
2.3. Advantages and Limitations of Microalgae Feed
3. Green Technology Application for Feed Development
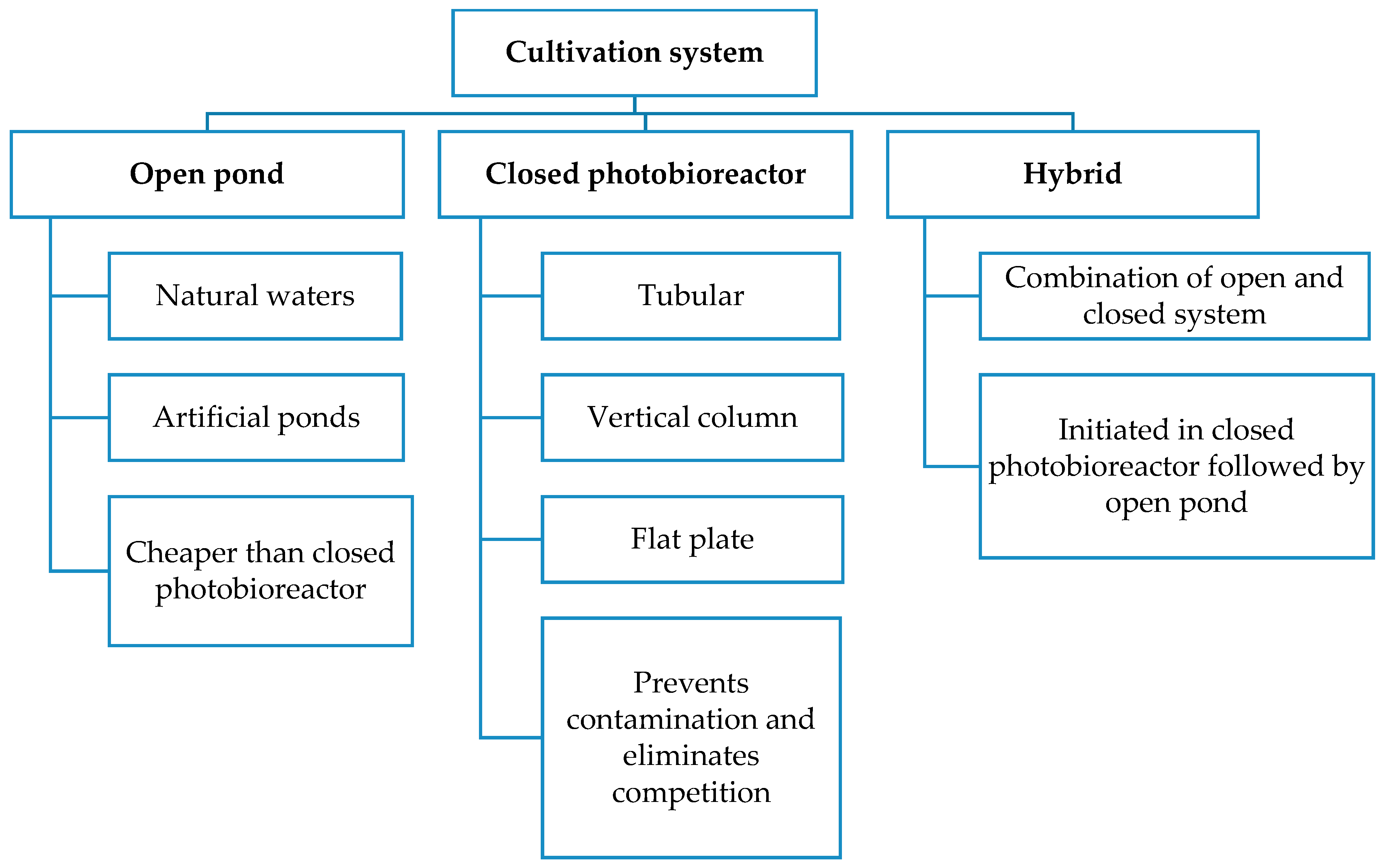
3.1. Biomass Cultivation, Harvesting and Dehydration
| Cultivation System | Advantages and Limitations | Reference |
|---|---|---|
| Raceway pond (RP) A closed circulation channel (depth of 0.2–1.0 m) and one/two paddle wheels that drive the circulation of the water body | Advantages:
| [55,91,92,102] |
Limitations:
| ||
| Revolving algal biofilm (RAB) A system consisting of microalgal biofilm, a drive unit, and an open pond with wastewater. Theoretically, the RAB system has higher land utilisation efficiency and biomass productivity than the RP system | Advantages:
| [55,90,93,103] |
Limitations:
|
3.2. Extraction Process of Bioactive Compound Using Green Techniques
| Extraction Method | Mechanism | Advantages | Limitations | Suitability for Aquafeed | References |
|---|---|---|---|---|---|
| Maceration | Solvent diffusion at room temperature | Simple, low-cost | Long extraction time, low efficiency | Limited due to solvent residue concerns | [126,127] |
| Heat-Assisted Extraction | Uses heat to enhance solubility | Faster than maceration | Thermal degradation of bioactives | Not ideal for heat-sensitive compounds | [128] |
| Soxhlet Extraction | Continuous solvent cycling with heating | Effective for stable compounds | Large solvent volumes, energy-intensive | Risk of solvent contamination in feed | [127] |
| Supercritical Fluid Extraction (SFE) | CO2 under high pressure and low temperature | High purity, preserves heat-sensitive compounds | Expensive equipment, low-polarity CO2 | Highly suitable for lipophilic bioactives (e.g., EPA, DHA) | [129,130] |
| Pressurized Liquid Extraction (PLE) | Solvent under elevated pressure and temperature | Rapid extraction, eco-friendly solvents | Solvent selection critical, high pressure | Good for polar and semi-polar compounds | [109,131,132] |
| Ultrasound-Assisted Extraction (UAE) | Acoustic cavitation disrupts cells | Fast, low solvent use, scalable | Requires optimisation of parameters | Effective for a broad compound range | [132,133] |
| Microwave-Assisted Extraction (MAE) | Microwave heating of intracellular water | High efficiency, short time | Risk of thermal hotspots | Best for polar molecules (e.g., pigments, polyphenols) | [134,135] |
| Enzyme-Assisted Extraction (EAE) | Enzymes (e.g., cellulase or protease) degrade cell walls to release intracellular contents | Mild conditions, selective, eco-friendly | Enzyme cost, reaction time, and need for optimisation | Suitable for proteins, polysaccharides, and functional peptides | [109,125] |
| Pulsed Electric Field (PEF) Extraction | Applies short bursts of high-voltage electric pulses to create pores in cell membranes | Non-thermal, preserves bioactives, efficient for wet biomass | Limited by biomass conductivity and uneven field distribution | Suitable for lipid and protein recovery from wet microalgae | [114] |
| Ionic Liquid (IL) Extraction | Tailor-made ionic solvents disrupt cell walls and dissolve intracellular compounds | High efficiency, tunable selectivity, minimal volatility | Costly, potential toxicity, purification required for feed safety | Promising for high-value compounds, but detoxification essential for feed use | [115] |
3.3. Benefits of Green Technology in Aquafeed Development
3.4. Toward Scalable and Circular Biorefinery Approaches
4. Recent Innovation in Microalgae-Based Aquafeed Development
4.1. Waste Valorisation and Circular Economy Models
4.2. Mixed-Microalgae Culture for Biomass Production
4.3. Microalgae as Aquafeed in Integrated Multi-Tropic Aquaculture (IMTA) Systems
4.4. Genetic Engineering and Strain Optimisation
4.5. Challenges and AI-Driven Outlook
5. Conclusions and Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
List of Abbreviations
| ARA | arachidonic acid |
| BFT | Biofloc Technologies |
| DHA | docosahexaenoic acid |
| DPA | docosapentaenoic acid |
| EAE | enzyme-assisted extraction |
| EPA | eicosapentaenoic acid |
| FCR | feed conversion ratio |
| FO | fish oil |
| FM | fish meal |
| GAIN | Green Aquaculture Intensification in Europe |
| GM | genetically modified |
| HRAP | high-rate algal ponds |
| ILs | ionic liquids |
| IMTA | integrated multi-tropic aquaculture |
| MAE | microwave-assisted extraction |
| n-3 LC-PUFAs | omega-3 long-chain polyunsaturated fatty acids |
| PEF | pulsed electric field extraction |
| PLE | pressurized liquid extraction |
| PUFAs | polyunsaturated fatty acids |
| RAB | revolving algal biofilm |
| RAS | recirculating aquaculture system |
| RP | raceway pond |
| SFE | supercritical fluid extraction |
| SNIPH | Sustainable New Ingredients to Promote Health |
| UAE | ultrasound-assisted extraction |
References
- FAO. The State of World Fisheries and Aquaculture 2024—Blue Transformation in Action; FAO: Rome, Italy, 2024; p. 264. [Google Scholar] [CrossRef]
- Fry, J.P.; Mailloux, N.A.; Love, D.C.; Milli, M.C.; Cao, L. Feed Conversion Efficiency in Aquaculture: Do We Measure It Correctly? Environ. Res. Lett. 2018, 13, 024017. [Google Scholar] [CrossRef]
- White, P.G. FAO Fisheries and Aquaculture Technical Paper No. 583; FAO: Rome, Italy, 2013; pp. 553–564. Available online: http://www.aquaculture.asia/files/online_03/Environmental%20consequences%20of%20poor%20feed%20quality%20and%20feeding%20management.pdf (accessed on 20 January 2025).
- Baki, B.; Yücel, S. Feed Cost/Production Income Analysis of Seabass (Dicentrarchus labrax) Aquaculture. Int. J. Ecosyst. Ecol. Sci. 2017, 7, 859–864. [Google Scholar]
- Tibbetts, S.M. The Potential for ‘Next-Generation’, Microalgae-Based Feed Ingredients for Salmonid Aquaculture in Context of the Blue Revolution. In Microalgal Biotechnology; InTech: London, UK, 2018. [Google Scholar]
- White, P.G. Environmental Consequences of Poor Feed Quality and Feed Management. In On-Farm Feeding and Feed Management in Aquaculture; Hasan, M.R., New, M.B., Eds.; FAO: Rome, Italy, 2013; pp. 553–564. [Google Scholar]
- FAO. The State of World Fisheries and Aquaculture 2020; Food and Agriculture Organization of the United Nations: Rome, Italy, 2020. [Google Scholar]
- Oliva-Teles, A.; Enes, P.; Peres, H. Replacing Fishmeal and Fish Oil in Industrial Aquafeeds for Carnivorous Fish. In Feed and Feeding Practices in Aquaculture; Davis, D.A., Ed.; Woodhead Publishing: Oxford, UK, 2015; pp. 203–233. [Google Scholar] [CrossRef]
- Shah, M.R.; Lutzu, G.A.; Alam, A.; Sarker, P.; Kabir Chowdhury, M.A.; Parsaeimehr, A.; Liang, Y.; Daroch, M. Microalgae in Aquafeeds for a Sustainable Aquaculture Industry. J. Appl. Phycol. 2018, 30, 197–213. [Google Scholar] [CrossRef]
- Shahin, S.; Okomoda, V.T.; Ma, H.; Abdullah, M.I. Sustainable Alternative Feed for Aquaculture: State of the Art and Future Perspective. Planet Sustain. 2023, 1, 62–96. [Google Scholar] [CrossRef]
- Ahmad, A.; Hassan, S.W.; Banat, F.; Hassan, S.W. An Overview of Microalgae Biomass as a Sustainable Aquaculture Feed Ingredient: Food Security and Circular Economy. Bioengineered 2022, 13, 9521–9547. [Google Scholar] [CrossRef]
- SNIPH. Sustainable New Ingredients to Promote Health: Our Mission; SNIPH: Stockholm, Sweden, 2020. [Google Scholar]
- Sarker, P.K.; Kapuscinski, A.R.; Bae, A.Y.; Donaldson, E.; Sitek, A.J.; Fitzgerald, D.S.; Edelson, O.F. Towards Sustainable Aquafeeds: Evaluating Substitution of Fishmeal with Lipid-Extracted Microalgal Co-Product (Nannochloropsis oculata) in Diets of Juvenile Nile Tilapia (Oreochromis niloticus). PLoS ONE 2018, 13, e0201315. [Google Scholar] [CrossRef]
- Cottrell, R.S.; Blanchard, J.L.; Halpern, B.S.; Metian, M.; Froehlich, H.E. Global Adoption of Novel Aquaculture Feeds Could Substantially Reduce Forage Fish Demand by 2030. Nat. Food 2020, 1, 301–308. [Google Scholar] [CrossRef]
- Tibbetts, S.M.; Scaife, M.A.; Armenta, R.E. Apparent Digestibility of Proximate Nutrients, Energy and Fatty Acids in Nutritionally-Balanced Diets with Partial or Complete Replacement of Dietary Fish Oil with Microbial Oil from a Novel Schizochytrium sp. (T18) by Juvenile Atlantic Salmon (Salmo salar). Aquaculture 2020, 520, 735003. [Google Scholar] [CrossRef]
- Manam, V.K. Fish Feed Nutrition and Its Management in Aquaculture. Int. J. Fish. Aquat. Stud. 2023, 11, 58–61. [Google Scholar] [CrossRef]
- Sutter, D.A.H.; Nuraini, R.; Chen, I.J.; Verreth, J.A.J.; Elesho, F.E.; Kr, S. Effect of Feeding Level on the Digestibility of Alternative Protein-Rich Ingredients for African Catfish (Clarias gariepinus). Aquaculture 2021, 544, 737067. [Google Scholar] [CrossRef]
- Apandi, N.M.; Mohamed, R.; Al-Gheethi, A.; Kassim, A.H.M. Microalgal Biomass Production through Phycoremediation of Fresh Market Wastewater and Potential Applications as Aquaculture Feeds. Environ. Sci. Pollut. Res. 2019, 26, 3226–3242. [Google Scholar] [CrossRef]
- Hemaiswarya, S.; Raja, R.; Kumar, R.R.; Ganesan, V.; Anbazhagan, C. Microalgae: A Sustainable Feed Source for Aquaculture. World J. Microbiol. Biotechnol. 2011, 27, 1737–1746. [Google Scholar] [CrossRef]
- Skifa, I.; Chauchat, N.; Cocquet, P.H.; Le Guer, Y. Microalgae Cultivation in Raceway Ponds: Advances, Challenges, and Hydrodynamic Considerations. EFB Bioecon. J. 2025, 5, 100073. [Google Scholar] [CrossRef]
- Ende, S.; Henjes, J.; Spiller, M.; Elshobary, M.; Hanelt, D. Recent Advances in Recirculating Aquaculture Systems and Role of Microalgae to Close System Loop. Bioresour. Technol. 2024, 407, 131107. [Google Scholar] [CrossRef] [PubMed]
- Chauton, M.S.; Reitan, K.I.; Norsker, N.H.; Tveterås, R.; Kleivdal, H.T. A techno-economic analysis of industrial production of marine microalgae as a source of EPA and DHA-rich raw material for aquafeed: Research challenges and possibilities. Aquaculture 2015, 436, 95–103. [Google Scholar] [CrossRef]
- Venkata Subhash, G.; Chugh, N.; Iyer, S.; Waghmare, A.; Musale, A.S.; Nandru, R.; Dixit, R.B.; Gaikwad, M.S.; Menon, D.; Thorat, R.; et al. Application of in vitro protein solubility for selection of microalgae biomass as protein ingredient in animal and aquafeed. J. Appl. Phycol. 2020, 32, 3955–3970. [Google Scholar] [CrossRef]
- García-Encinas, J.P.; Ruiz-Cruz, S.; Juárez, J.; Ornelas-Paz, J.J.; Del Toro-Sánchez, C.L.; Márquez-Ríos, E. Proteins from microalgae: Nutritional, functional and bioactive properties. Foods 2025, 14, 921. [Google Scholar] [CrossRef]
- Islam, S.M.; Willora, F.P.; Sørensen, M.; Rbbani, G.; Siddik, A.B.; Zatti, K.; Gupta, S.; Carr, I.; Santigosa, E.; Brinchmann, M.F.; et al. Mucosal barrier status in Atlantic salmon fed rapeseed oil and Schizochytrium oil partly or fully replacing fish oil through winter depression. Fish Shellfish Immunol. 2024, 149, 108126. [Google Scholar] [CrossRef]
- Dineshbabu, G.; Goswami, G.; Kumar, R.; Sinha, A.; Das, D. Microalgae–nutritious, sustainable aqua- and animal feed source. J. Funct. Foods 2019, 62, 103545. [Google Scholar] [CrossRef]
- Tibbetts, S.M.; Yasumaru, F.; Lemos, D. In vitro prediction of digestible protein content of marine microalgae (Nannochloropsis granulata) meals for Pacific white shrimp (Litopenaeus vannamei) and rainbow trout (Oncorhynchus mykiss). Algal Res. 2017, 21, 76–80. [Google Scholar] [CrossRef]
- Qiao, H.; Hu, D.; Ma, J.; Wang, X.; Wu, H.; Wang, J. Feeding effects of the microalga Nannochloropsis sp. on juvenile turbot (Scophthalmus maximus L.). Algal Res. 2019, 41, 101574. [Google Scholar] [CrossRef]
- Twibell, R.; Johnson, R.; Hyde, N.; Gannam, A. Evaluation of Spirulina and plant oil in diets for juvenile steelhead (Oncorhynchus mykiss). Aquaculture 2020, 528, 735598. [Google Scholar] [CrossRef]
- Pereira, H.; Sardinha, M.; Santos, T.; Gouveia, L.; Barreira, L.; Dias, J.; Varela, J. Incorporation of defatted microalgal biomass (Tetraselmis sp. CTP4) at the expense of soybean meal as a feed ingredient for juvenile gilthead seabream (Sparus aurata). Algal Res. 2020, 47, 101869. [Google Scholar] [CrossRef]
- Macias-Sancho, J.; Poersch, L.H.; Bauer, W.; Romano, L.A.; Wasielesky, W.; Tesser, M.B. Fishmeal substitution with Arthrospira (Spirulina platensis) in a practical diet for Litopenaeus vannamei: Effects on growth and immunological parameters. Aquaculture 2014, 426–427, 120–125. [Google Scholar] [CrossRef]
- Abdelghany, M.F.; El-Sawy, H.B.; Abd El-Hameed, S.A.A.; Khames, M.K.; Abdel-Latif, H.M.R.; Naiel, M.A.E. Effects of dietary Nannochloropsis oculata on growth performance, serum biochemical parameters, immune responses, and resistance against Aeromonas veronii challenge in Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol. 2020, 107, 277–288. [Google Scholar] [CrossRef]
- Orozco Colonia, B.S.; Pereira, G.V.M.; Soccol, C.R. Omega-3 microbial oils from marine thraustochytrids as a sustainable and technological solution: A review and patent landscape. Trends Food Sci. Technol. 2020, 99, 244–256. [Google Scholar] [CrossRef]
- Zakaria, M.F.; Mat Zaidi, F.N.A.; Ahmad Kamal, A.H.; Aleng, N.A.; Abd Wahid, M.E.; Katayama, T.; Jusoh, M. PUFA from microalgae: Challenges, factors affecting high production and industrial application. Aquac. Fish 2025, 10, 545–555. [Google Scholar] [CrossRef]
- Lutfi, E.; Berge, G.M.; Bæverfjord, G.; Sigholt, T.; Bou, M.; Larsson, T.; Mørkøre, T.; Evensen, Ø.; Sissener, N.H.; Rosenlund, G.; et al. Increasing dietary levels of the n-3 long-chain PUFA, EPA and DHA, improves the growth, welfare, robustness and fillet quality of Atlantic salmon in sea cages. Br. J. Nutr. 2023, 129, 10–28. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Miar, Y.; Huyben, D.; Colombo, S.M. Omega-3 long-chain polyunsaturated fatty acids in Atlantic salmon: Functions, requirements, sources, de novo biosynthesis and selective breeding strategies. Aquaculture 2024, 573, 103041. [Google Scholar] [CrossRef]
- Huyben, D.; Cronin, T.; Bartie, K.L.; Matthew, C.; Sissener, N.H.; Hundal, B.K.; Homer, N.Z.M.; Ruyter, B.; Glencross, B. Steroidogenic and innate immune responses in Atlantic salmon are influenced by dietary total lipid, long chain polyunsaturated fatty acids and dissolved oxygen. Aquaculture 2023, 564, 739028. [Google Scholar] [CrossRef]
- Carvalho, M.; Montero, D.; Rosenlund, G.; Fontanillas, R.; Ginés, R.; Izquierdo, M. Effective complete replacement of fish oil by combining poultry and microalgae oils in practical diets for gilthead sea bream (Sparus aurata) fingerlings. Aquaculture 2020, 529, 735696. [Google Scholar] [CrossRef]
- Galasso, C.; Gentile, A.; Orefice, I.; Ianora, A.; Bruno, A.; Noonan, D.M.; Sansone, C.; Albini, A.; Brunet, C. Microalgal derivatives as potential nutraceutical and food supplements for human health: A focus on cancer prevention and interception. Nutrients 2019, 11, 1226. [Google Scholar] [CrossRef]
- Dragone, G.; Fernandes, B.D.; Abreu, A.P.; Vicente, A.A.; Teixeira, J.A. Nutrient limitation as a strategy for increasing starch accumulation in microalgae. Appl. Energy 2011, 88, 3331–3335. [Google Scholar] [CrossRef]
- Matos, Â.P.; Feller, R.; Moecke, E.H.S.; de Oliveira, J.V.; Junior, A.F.; Derner, R.B.; Sant’Anna, E.S. Chemical characterization of six microalgae with potential utility for food application. J. Am. Oil Chem. Soc. 2016, 93, 963–972. [Google Scholar] [CrossRef]
- Niccolai, A.; Chini Zittelli, G.; Rodolfi, L.; Biondi, N.; Tredici, M.R. Microalgae of interest as food source: Biochemical composition and digestibility. Algal Res. 2019, 42, 101617. [Google Scholar] [CrossRef]
- Raposo, M.F.; de Morais, R.M.; de Morais, A.M.B. Bioactivity and applications of sulphated polysaccharides from marine microalgae. Mar. Drugs 2013, 11, 233–252. [Google Scholar] [CrossRef]
- Pandeirada, C.O.; Maricato, É.; Ferreira, S.S.; Correia, V.G.; Pinheiro, B.A.; Evtuguin, D.V.; Palma, A.S.; Correia, A.; Vilanova, M.; Coimbra, M.A.; et al. Structural analysis and potential immunostimulatory activity of Nannochloropsis oculata polysaccharides. Carbohydr. Polym. 2019, 222, 114962. [Google Scholar] [CrossRef]
- Silva, S.C.; Ferreira, I.; Dias, M.M.; Barreiro, M.F. Microalgae-derived pigments: A 10-year bibliometric review and industry and market trend analysis. Molecules 2020, 25, 3406. [Google Scholar] [CrossRef]
- Shah, M.M.; Liang, Y.; Cheng, J.J.; Daroch, M. Astaxanthin-producing green microalga Haematococcus pluvialis: From single cell to high value commercial products. Front. Plant Sci. 2016, 7, 531. [Google Scholar] [CrossRef] [PubMed]
- Hoang, T.H.; Qin, J.G.; Stone, D.A.J.; Harris, J.O.; Duong, D.N.; Bansemer, M.S. Colour changes of greenlip abalone (Haliotis laevigata Donovan) fed fresh macroalgae and dried algal supplement. Aquaculture 2016, 456, 16–23. [Google Scholar] [CrossRef]
- Cardinaletti, G.; Messina, M.; Bruno, M.; Tulli, F.; Poli, B.M.; Giorgi, G.; Chini-Zittelli, G.; Tredici, M.; Tibaldi, E. Effects of graded levels of a blend of Tisochrysis lutea and Tetraselmis suecica dried biomass on growth and muscle tissue composition of European sea bass (Dicentrarchus labrax) fed diets low in fish meal and oil. Aquaculture 2018, 485, 173–182. [Google Scholar] [CrossRef]
- Ju, Z.Y.; Deng, D.F.; Dominy, W. A defatted microalgae (Haematococcus pluvialis) meal as a protein ingredient to partially replace fishmeal in diets of Pacific white shrimp (Litopenaeus vannamei, Boone, 1931). Aquaculture 2012, 354–355, 50–55. [Google Scholar] [CrossRef]
- Hekimoglu, M.A.; Firat, K.; Saka, S.; Süzer, C.; Kop, A.; Durmaz, Y. Effect of supplemented algal carotenoid diets on skin color of tomato clownfish, Amphiprion frenatus. Pak. J. Zool. 2017, 49, 2. [Google Scholar] [CrossRef]
- Ambati, R.R.; Phang, S.M.; Ravi, S.; Aswathanarayana, R.G. Astaxanthin: Sources, extraction, stability, biological activities and its commercial applications—A review. Mar. Drugs 2014, 12, 128–152. [Google Scholar] [CrossRef]
- Levasseur, W.; Perré, P.; Pozzobon, V. A review of high value-added molecules production by microalgae in light of the classification. Biotechnol. Adv. 2020, 41, 107545. [Google Scholar] [CrossRef]
- Islam, M.N.; Alsenani, F.; Schenk, P.M. Microalgae as a sustainable source of nutraceuticals. In Microbial Functional Foods and Nutraceuticals; Gupta, V.K., Treichel, H., Shapaval, V., Oliveira, L.A., Tuohy, M.G., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2018; pp. 1–19. [Google Scholar]
- Yarnold, J.; Karan, H.; Oey, M.; Hankamer, B. Microalgal aquafeeds as part of a circular bioeconomy. Trends Plant Sci. 2019, 24, 959–970. [Google Scholar] [CrossRef]
- Han, P.; Lu, Q.; Fan, L.; Zhou, W. A review on the use of microalgae for sustainable aquaculture. Appl. Sci. 2019, 9, 2377. [Google Scholar] [CrossRef]
- Siddik, M.A.B.; Sørensen, M.; Islam, S.M.M.; Saha, N.; Rahman, M.A.; Francis, D.S. Expanded utilisation of microalgae in global aquafeeds. Rev. Aquac. 2024, 16, 6–33. [Google Scholar] [CrossRef]
- Rusco, G.; Roncarati, A.; Di Iorio, M.; Cariglia, M.; Longo, C.; Iaffaldano, N. Can IMTA system improve the productivity and quality traits of aquatic organisms produced at different trophic levels? The benefits of IMTA—Not only for the ecosystem. Biology 2024, 13, 946. [Google Scholar] [CrossRef] [PubMed]
- Vijayaram, S.; Ringø, E.; Ghafarifarsani, H.; Hoseinifar, S.H. Use of Algae in Aquaculture: A Review. Fishes 2024, 9, 63. [Google Scholar] [CrossRef]
- Kaparapu, J. Application of Microalgae in Aquaculture. Phykos 2018, 48, 21–26. [Google Scholar]
- Adibah, W.; Mahari, W.; Waiho, K.; Fazhan, H.; Azwar, E. Emerging paradigms in sustainable shellfish aquaculture: Microalgae and biofloc technologies for wastewater treatment. Aquaculture 2024, 587, 740835. [Google Scholar] [CrossRef]
- FAO. Thematic Background Study No. 2: Genetic Resources for Microorganisms of Current and Potential Use in Aquaculture; Committee on Fisheries, Sub-Committee on Aquaculture: Rome, Italy, 2017; pp. 1–43. [Google Scholar]
- Chithambaran, S.; Harbi, M.; Broom, M.; Khobrani, K.; Ahmad, O.; Fattani, H.; Sofyani, A.; Ayaril, N. Green water technology for the production of Pacific white shrimp Penaeus vannamei (Boone, 1931). Indian J. Fish. 2017, 64, 43–49. [Google Scholar] [CrossRef]
- Bossier, P.; Ekasari, J. Biofloc technology application in aquaculture to support sustainable development goals. Microb. Biotechnol. 2017, 10, 1012–1016. [Google Scholar] [CrossRef] [PubMed]
- Kaya, D.; Genc, E.; Genc, M.A.; Aktas, M.; Eroldogan, O.T.; Guroy, D. Biofloc technology in recirculating aquaculture system as a culture model for green tiger shrimp, Penaeus semisulcatus: Effects of different feeding rates and stocking densities. Aquaculture 2020, 528, 735526. [Google Scholar] [CrossRef]
- Sajali, U.S.B.A.; Atkinson, N.L.; Desbois, A.P.; Little, D.C.; Murray, F.J.; Shinn, A.P. Prophylactic properties of biofloc- or Nile tilapia-conditioned water against Vibrio parahaemolyticus infection of whiteleg shrimp (Penaeus vannamei). Aquaculture 2019, 498, 496–502. [Google Scholar] [CrossRef]
- Shao, J.; Liu, M.; Wang, B.; Jiang, K.; Wang, M.; Wang, L. Evaluation of biofloc meal as an ingredient in diets for white shrimp Litopenaeus vannamei under practical conditions: Effect on growth performance, digestive enzymes and TOR signaling pathway. Aquaculture 2017, 479, 516–521. [Google Scholar] [CrossRef]
- Jung, J.Y.; Damusaru, J.H.; Park, Y.; Kim, K.; Seong, M.; Je, H.W.; Kim, S.; Bai, S.C. Autotrophic biofloc technology system (ABFT) using Chlorella vulgaris and Scenedesmus obliquus positively affects performance of Nile tilapia (Oreochromis niloticus). Algal Res. 2017, 27, 259–264. [Google Scholar] [CrossRef]
- Boyd, C.E.; D’Abramo, L.R.; Glencross, B.D.; Huyben, D.C.; Juarez, L.M.; Lockwood, G.S.; McNevin, A.A.; Tacon, A.G.J.; Teletchea, F.; Tomasso, J.R., Jr.; et al. Achieving sustainable aquaculture: Historical and current perspectives and future needs and challenges. J. World Aquac. Soc. 2020, 51, 578–633. [Google Scholar] [CrossRef]
- Senroy, S.; Pal, R. Microalgae in Aquaculture: A Review with Special References to Nutritional Value and Fish Dietetics. Proc. Zool. Soc. 2014, 68, 1–8. [Google Scholar]
- Tolve, R.; Tchuenbou-Magaia, F.; Di Cairano, M.; Caruso, M.C.; Scarpa, T.; Galgano, F. Encapsulation of bioactive compounds for the formulation of functional animal feeds: The biofortification of derivative foods. Anim. Feed Sci. Technol. 2021, 274, 115036. [Google Scholar] [CrossRef]
- da Silva, A.F.; Moreira, A.F.; Miguel, S.P.; Coutinho, P. Recent advances in microalgae encapsulation techniques for biomedical applications. Adv. Colloid Interface Sci. 2024, 333, 103297. [Google Scholar] [CrossRef]
- Yanes-Roca, C.; Štěrbová, K.; Mráz, J.; Veselý, L.; Malinovskyi, O.; Pěnka, T.; Masojídek, J.; Policar, T. Live feed enrichments using microalgae for pikeperch (Sander lucioperca) larval culture. J. World Aquac. Soc. 2024, 55, e13059. [Google Scholar] [CrossRef]
- Nagappan, S.; Das, P.; AbdulQuadir, M.; Thaher, M.; Khan, S.; Mahata, C.; Al-Jabri, H.; Vatland, A.K.; Kumar, G. Potential of microalgae as a sustainable feed ingredient for aquaculture. J. Biotechnol. 2021, 341, 1–20. [Google Scholar] [CrossRef]
- Li, Y.P.; Ahmadi, F.; Kariman, K.; Lackner, M. Recent advances and challenges in single cell protein (SCP) technologies for food and feed production. NPJ Sci. Food 2024, 8, 66. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Shan, L.; Zhao, W.; Lu, X. Harnessing artificial intelligence to revolutionize microalgae biotechnology: Unlocking sustainable solutions for high-value compounds and carbon neutrality. Mar. Drugs 2025, 23, 184. [Google Scholar] [CrossRef]
- Lim, H.R.; Khoo, K.S.; Chia, W.Y.; Wayne, C.K.; Ho, S.H.; Show, P.L. Smart microalgae farming with internet-of-things for sustainable agriculture. Biotechnol. Adv. 2022, 57, 107931. [Google Scholar] [CrossRef]
- Alghamdi, M.; Haraz, Y.G. Smart Biofloc Systems: Leveraging Artificial Intelligence (AI) and Internet of Things (IoT) for Sustainable Aquaculture Practices. Processes 2025, 13, 2204. [Google Scholar] [CrossRef]
- Hamed, I. The evolution and versatility of microalgal biotechnology: A review. Compr. Rev. Food Sci. Food Saf. 2016, 15, 1104–1123. [Google Scholar] [CrossRef]
- Salin, K.R.; Arun, V.V.; Nair, C.M.; Tidwell, J.H. Sustainable aquafeed. In Sustainable Aquaculture; Hai, F., Visvanathan, C., Boopathy, R., Eds.; Springer: Cham, Switzerland, 2018; pp. 123–151. [Google Scholar]
- Origin Clear. Algae Harvesting: Use of Algae as Aquafeed to Improve Production in Aquaculture Operations; White Paper: Los Angeles, CA, USA, 2015. [Google Scholar]
- Enzing, C.; Ploeg, M.; Barbosa, M.; Sijtsma, L. Microalgae-based products for the food and feed sector: An outlook for Europe. In JRC Scientific and Policy Reports; Vigani, M., Parisi, C., Cerezo, E.R., Eds.; Publications Office of the European Union: Luxembourg, 2014; pp. 7–78. [Google Scholar]
- Fu, W.; Nelson, D.R.; Yi, Z.; Xu, M.; Khraiwesh, B.; Jijakli, K.; Chaiboonchoe, A.; Alzahmi, A.; Al-Khairy, D.; Brynjolfsson, S.; et al. Bioactive compounds from microalgae: Current development and prospects. In Studies in Natural Products Chemistry; Atta, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 199–225. [Google Scholar]
- Linares, L.C.F.; Falfán, K.Á.G.; Ramírez-López, C. Microalgal biomass: A biorefinery approach. In Biomass Volume Estimation and Valorization for Energy; Tumuluru, J.S., Ed.; IntechOpen: London, UK, 2017; p. 293. [Google Scholar]
- Tan, J.S.; Lee, S.Y.; Chew, K.W.; Lam, M.K.; Lim, J.W.; Ho, S.H.; Show, P.L. A review on microalgae cultivation and harvesting, and their biomass extraction processing using ionic liquids. Bioengineered 2020, 11, 116–129. [Google Scholar] [CrossRef]
- Lu, Q.; Yang, L.; Deng, X. Critical thoughts on the application of microalgae in aquaculture industry. Aquaculture 2020, 528, 735538. [Google Scholar] [CrossRef]
- Adarme-Vega, T.C.; Lim, D.K.; Timmins, M.; Vernen, F.; Li, Y.; Schenk, P.M. Microalgal biofactories: A promising approach towards sustainable omega-3 fatty acid production. Microb. Cell Fact. 2012, 11, 96. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Bhatia, R.K.; Yang, Y.H. An overview of microdiesel—A sustainable future source of renewable energy. Renew. Sust. Energy Rev. 2017, 79, 1078–1090. [Google Scholar] [CrossRef]
- Nobre, B.P.; Villalobos, F.; Barragán, B.E.; Oliveira, A.C.; Batista, A.P.; Marques, P.A.; Mendes, R.L.; Sovová, H.; Palavra, A.F.; Gouveia, L. A biorefinery from Nannochloropsis sp. microalga: Extraction of oils and pigments and production of biohydrogen from the leftover biomass. Bioresour. Technol. 2013, 135, 128–136. [Google Scholar] [CrossRef]
- Pacheco, M.M.; Hoeltz, M.; Moraes, M.S.A.; Schneider, R.C.S. Microalgae: Cultivation techniques and wastewater phycoremediation. J. Environ. Sci. Health A 2015, 50, 585–601. [Google Scholar]
- Gross, M.; Wen, Z. Yearlong evaluation of performance and durability of a pilot-scale Revolving Algal Biofilm (RAB) cultivation system. Bioresour. Technol. 2014, 171, 50–58. [Google Scholar] [CrossRef]
- Sfez, S.; Van Den Hende, S.; Taelman, S.E.; De Meester, S.; Dewulf, J. Environmental sustainability assessment of a microalgae raceway pond treating aquaculture wastewater: From up-scaling to system integration. Bioresour. Technol. 2015, 190, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Van Den Hende, S.; Beelen, V.; Bore, G.; Boon, N.; Vervaeren, H. Up-scaling aquaculture wastewater treatment by microalgal bacterial flocs: From lab reactors to an outdoor raceway pond. Bioresour. Technol. 2014, 159, 342–354. [Google Scholar] [CrossRef] [PubMed]
- Gross, M.; Henry, W.; Michael, C.; Wen, Z. Development of a rotating algal biofilm growth system for attached microalgae growth with in situ biomass harvest. Bioresour. Technol. 2013, 150, 195–201. [Google Scholar] [CrossRef]
- Wood, J.; Takemoto, J.; Sims, R. Rotating algae biofilm reactor for management and valorization of produced wastewater. Front. Energy Res. 2022, 10, 774760. [Google Scholar] [CrossRef]
- Laamanen, C.A.; Ross, G.M.; Scott, J.A. Flotation harvesting of microalgae. Renew. Sust. Energy Rev. 2016, 58, 75–86. [Google Scholar] [CrossRef]
- Show, K.Y.; Lee, D.J.; Chang, J.S. Algal biomass dehydration. Bioresour. Technol. 2013, 135, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Mujumdar, A.S. Classification and selection of industrial dryers. In Mujumdar’s Practical Guide to Industrial Drying: Principles, Equipment and New Developments; Devahastin, S., Ed.; Exergex Corporation: Montreal, QC, Canada, 2000; pp. 23–77. [Google Scholar]
- Nappa, M.; Teir, S.; Sorsamäki, L.; Karinen, P.; Oy, C.I. Energy Requirements of Microalgae Biomass Production. In CCSP Deliverable D606; Espoo: Helsinki, Finland, 2016. [Google Scholar]
- Grima, E.M.; Belarbi, E.H.; Fernández, F.G.A.; Medina, A.R.; Chisti, Y. Recovery of Microalgal Biomass and Metabolites: Process Options and Economics. Biotechnol. Adv. 2003, 20, 491–515. [Google Scholar] [CrossRef]
- Oyinloye, T.M.; Yoon, W.B. Effect of Freeze-Drying on Quality and Grinding Process of Food Produce: A Review. Processes 2020, 8, 354. [Google Scholar] [CrossRef]
- Zhou, W.; Min, M.; Hu, B.; Ma, X.; Liu, Y.; Wang, Q.; Shi, J.; Chen, P.; Ruan, R. Filamentous Fungi Assisted Bio-Flocculation: A Novel Alternative Technique for Harvesting Heterotrophic and Autotrophic Microalgal Cells. Sep. Purif. Technol. 2013, 107, 158–165. [Google Scholar] [CrossRef]
- Norsker, N.H.; Barbosa, M.J.; Vermuë, M.H.; Wijffels, R.H. Microalgal Production—A Close Look at the Economics. Biotechnol. Adv. 2011, 29, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Christenson, L.B.; Sims, R.C. Rotating Algal Biofilm Reactor and Spool Harvester for Wastewater Treatment with Biofuels By-Products. Biotechnol. Bioeng. 2012, 109, 1674–1684. [Google Scholar] [CrossRef] [PubMed]
- Ercolano, G.; De Cicco, P.; Ianaro, A. New Drugs from the Sea: Pro-Apoptotic Activity of Sponges and Algae Derived Compounds. Mar. Drugs 2019, 17, 31. [Google Scholar] [CrossRef]
- Molino, A.; Iovine, A.; Casella, P.; Mehariya, S.; Chianese, S.; Cerbone, A.; Rimauro, J.; Musmarra, D. Microalgae Characterization for Consolidated and New Application in Human Food, Animal Feed and Nutraceuticals. Int. J. Environ. Res. Public Health 2018, 15, 2436. [Google Scholar] [CrossRef]
- Srinivasan, R.; Chaitanyakumar, A.; Mageswari, A.; Gomathi, A.; Pavan Kumar, J.G.S.; Jayasindu, M.; Bharath, G.; Shravan, J.S.; Gothandam, K.M. Oral Administration of Lyophilized Dunaliella salina, a Carotenoid-Rich Marine Alga, Reduces Tumor Progression in Mammary Cancer Induced Rats. Food Funct. 2017, 8, 4517–4527. [Google Scholar] [CrossRef]
- Azmir, J.; Zaidul, I.S.M.; Rahman, M.M.; Sharif, K.M.; Mohamed, A.; Sahena, F.; Jahurul, M.H.A.; Ghafoor, K.; Norulaini, N.A.N.; Omar, A.K.M. Techniques for Extraction of Bioactive Compounds from Plant Materials: A Review. J. Food Eng. 2013, 117, 426–436. [Google Scholar] [CrossRef]
- Klejdus, B.; Kopecký, J.; Benešová, L.; Vacek, J. Solid-Phase/Supercritical-Fluid Extraction for Liquid Chromatography of Phenolic Compounds in Freshwater Microalgae and Selected Cyanobacterial Species. J. Chromatogr. A 2009, 1216, 763–771. [Google Scholar] [CrossRef]
- Sánchez-Camargo, A.D.P.; Montero, L.; Stiger-Pouvreau, V.; Tanniou, A.; Cifuentes, A.; Herrero, M. Considerations on the Use of Enzyme-Assisted Extraction in Combination with Pressurized Liquids to Recover Bioactive Compounds from Algae. Food Chem. 2016, 192, 67–74. [Google Scholar] [CrossRef]
- Nisya, A.F.; Rochmadi, R.; Budiman, A. Ultrasound Assisted Extraction of Microalgae Spirulina sp. AIP Conf. Proc. 2023, 2667, 020002. [Google Scholar]
- Martins, R.; Barbosa, A.; Advinha, B.; Sales, H.; Pontes, R.; Nunes, J. Técnicas de Extracción Ecológica de Compuestos Bioactivos: Una Revisión del Estado del Arte. Processes 2023, 11, 1123. [Google Scholar]
- Fernandes, A.S.; Caetano, P.A.; Jacob-Lopes, E.; Zepka, L.Q.; de Rosso, V.V. Alternative Green Solvents Associated with Ultrasound-Assisted Extraction: A Green Chemistry Approach for the Extraction of Carotenoids and Chlorophylls from Microalgae. Food Chem. 2024, 455, 139939. [Google Scholar] [CrossRef]
- Shang, Y.F.; Kim, S.M.; Lee, W.J.; Um, B.H. Pressurized Liquid Method for Fucoxanthin Extraction from Eisenia bicyclis (Kjellman) Setchell. J. Biosci. Bioeng. 2011, 111, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Buchmann, L.; Brändle, I.; Haberkorn, I.; Hiestand, M.; Mathys, A. Pulsed electric field based cyclic protein extraction of microalgae towards closed-loop biorefinery concepts. Bioresour. Technol. 2019, 291, 121870. [Google Scholar] [CrossRef]
- Orr, V.C.A.; Rehmann, L. Ionic liquids for the fractionation of microalgae biomass. Curr. Opin. Green Sustain. Chem. 2016, 2, 22–27. [Google Scholar] [CrossRef]
- Poojary, M.M.; Barba, F.J.; Aliakbarian, B.; Donsì, F.; Pataro, G.; Dias, D.A.; Juliano, P. Innovative alternative technologies to extract carotenoids from microalgae and seaweeds. Mar. Drugs 2016, 14, 214. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S.K.; Kim, S.H.; Yoon, J.J.; Yang, Y.H. Current status and strategies for second generation biofuel production using microbial systems. Energy Convers. Manag. 2017, 148, 1142–1156. [Google Scholar] [CrossRef]
- Kim, D.Y.; Vijayan, D.; Praveenkumar, R.; Han, J.I.; Lee, K.; Park, J.Y.; Chang, W.S.; Lee, J.S.; Oh, Y.K. Cell-wall disruption and lipid/astaxanthin extraction from microalgae: Chlorella and Haematococcus. Bioresour. Technol. 2016, 199, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Lorente, E.; Farriol, X.; Salvadó, J. Steam explosion as a fractionation step in biofuel production from microalgae. Fuel Process. Technol. 2015, 131, 93–98. [Google Scholar] [CrossRef]
- Molino, A.; Larocca, V.; Chianese, S.; Musmarra, D. Biofuels production by biomass gasification: A review. Energies 2018, 11, 811. [Google Scholar] [CrossRef]
- Praveenkumar, R.; Lee, K.; Lee, J.; Oh, Y.K. Breaking dormancy: An energy-efficient means of recovering astaxanthin from microalgae. Green Chem. 2015, 17, 1226–1234. [Google Scholar] [CrossRef]
- Molino, A.; Mehariya, S.; Di Sanzo, G.; Larocca, V.; Martino, M.; Leone, G.P.; Marino, T.; Chianese, S.; Balducchi, R.; Musmarra, D. Recent developments in supercritical fluid extraction of bioactive compounds from microalgae: Role of key parameters, technological achievements and challenges. J. CO2 Util. 2020, 36, 196–209. [Google Scholar] [CrossRef]
- Herrero, M.; Cifuentes, A.; Ibáñez, E. Sub- and supercritical fluid extraction of functional ingredients from different natural sources: Plants, food-by-products, algae and microalgae: A review. Food Chem. 2006, 98, 136–148. [Google Scholar] [CrossRef]
- Herrero, M.; Mendiola, J.A.; Cifuentes, A.; Ibáñez, E. Supercritical fluid extraction: Recent advances and applications. J. Chromatogr. A 2010, 1217, 2495–2511. [Google Scholar] [CrossRef]
- Kadam, S.U.; Tiwari, B.K.; O’Donnell, C.P. Application of novel extraction technologies for bioactives from marine algae. J. Agric. Food Chem. 2013, 61, 4667–4675. [Google Scholar] [CrossRef]
- Bitwell, C.; Singh Sen, I.; Chimuka, L.; Maseka Kakoma, K. A review of modern and conventional extraction techniques and their applications for extracting phytochemicals from plants. Sci. Afr. 2023, 19, e01585. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, M.; Saraiva, J.A.; Martins, A.P.; Pinto, C.A.; Prieto, M.A.; Simal-Gandara, J.; Cao, H.; Xiao, J.; Barba, F.J. Extraction of lipids from microalgae using classical and innovative approaches. Food Chem. 2022, 384, 132236. [Google Scholar] [CrossRef]
- Rodríguez, M.; García Oliveira, P.; González Pereira, A.; Jiménez-López, C.; Lourenço-Lopes, C.; Carreira-Casais, A.; Echave, J.; Corral, M.F.; Barros, L.; Ferreira, I.C.F.R.; et al. Obtaining healthier and safer food additives from red algae: Comparison of high pressure and heat assisted extraction. In Proceedings of the 58th European High Pressure Research Group International Conference, Tenerife, Spain, 6–11 September 2020. [Google Scholar]
- Saini, R.K.; Keum, Y.S. Carotenoid extraction methods: A review of recent developments. Food Chem. 2018, 240, 90–103. [Google Scholar] [CrossRef]
- Taj-Liad, S.T.; Raviadaran, R.; Chandran, D.; Lam, M.K. Cell disruption and extraction for microalgae biorefinery: Focus on deep eutectic solvents moving towards aqueous biphasic system. Results Eng. 2025, 26, 105066. [Google Scholar] [CrossRef]
- Castillo, A.; Pereira, S.; Otero, A.; Fiol, S.; Garcia-Jares, C.; Lores, M. Matrix solid-phase dispersion as a greener alternative to obtain bioactive extracts from Haematococcus pluvialis: Characterization by UHPLC-QToF. RSC Adv. 2020, 10, 27995–28006. [Google Scholar] [CrossRef]
- Bleakley, S.; Hayes, M. Algal proteins: Extraction, application, and challenges concerning production. Foods 2017, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Mienis, E.; Vandamme, D.; Foubert, I. Ultrasound assisted extraction of Nannochloropsis: Effects on lipid extraction efficiency and lipid stability. Algal Res. 2024, 80, 103520. [Google Scholar] [CrossRef]
- Yagi, S.; Uba, A.I.; Sinan, K.I.; Piatti, D.; Sagratini, G.; Caprioli, G.; Eltigani, S.M.; Lazarova, I.; Zengin, G. Comparative study on the chemical profile, antioxidant activity, and enzyme inhibition capacity of red and white Hibiscus sabdariffa variety calyces. Plants 2023, 8, 42511–42521. [Google Scholar] [CrossRef] [PubMed]
- Georgiopoulou, I.; Tzima, S.; Louli, V.; Magoulas, K. Process optimization of microwave-assisted extraction of chlorophyll, carotenoid and phenolic compounds from Chlorella vulgaris and comparison with conventional and supercritical fluid extraction. Appl. Sci. 2023, 13, 2740. [Google Scholar] [CrossRef]
- Kubrakova, I.V.; Toropchenova, E.S. Microwave heating for enhancing efficiency of analytical operations (Review). Inorg. Mater. 2008, 44, 1509–1519. [Google Scholar] [CrossRef]
- Routray, W.; Orsat, V. Microwave-assisted extraction of flavonoids: A review. Food Bioprocess Technol. 2012, 5, 409–424. [Google Scholar] [CrossRef]
- Chemat, F.; Zille, H.; Khan, M.K. Applications of ultrasound in food technology: Processing, preservation and extraction. Ultrason. Sonochem. 2011, 18, 813–835. [Google Scholar] [CrossRef] [PubMed]
- Pignolet, O.; Jubeau, S.; Vaca-Garcia, C.; Michaud, P. Highly valuable microalgae: Biochemical and topological aspects. J. Ind. Microbiol. Biotechnol. 2013, 40, 781–796. [Google Scholar] [CrossRef]
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef]
- Sanzo, G.D.; Mehariya, S.; Martino, M.; Larocca, V.; Casella, P.; Chianese, S.; Musmarra, D.; Balducchi, R.; Molino, A. Supercritical carbon dioxide extraction of astaxanthin, lutein, and fatty acids from Haematococcus pluvialis microalgae. Mar. Drugs 2018, 16, 334. [Google Scholar] [CrossRef]
- Safi, C.; Charton, M.; Ursu, A.V.; Laroche, C.; Zebib, B.; Pontalier, P.Y.; Vaca-Garcia, C. Release of hydro-soluble microalgal proteins using mechanical and chemical treatments. Algal Res. 2014, 3, 55–60. [Google Scholar] [CrossRef]
- Herrero, M.; Ibáñez, E.; Cifuentes, A.; Reglero, G.; Santoyo, S. Dunaliella salina microalga pressurized liquid extracts as potential antimicrobials. J. Food Prot. 2006, 69, 2471–2477. [Google Scholar] [CrossRef] [PubMed]
- Neori, A. “Green water” microalgae: The leading sector in world aquaculture. J. Appl. Phycol. 2011, 23, 143–149. [Google Scholar] [CrossRef]
- Crab, R.; Defoirdt, T.; Bossier, P.; Verstraete, W. Biofloc technology in aquaculture: Beneficial effects and future challenges. Aquaculture 2012, 356–357, 351–356. [Google Scholar] [CrossRef]
- Chatzimaliakas, P.F.; Malamis, D.; Mai, S. Biorefinery-based energy recovery from algae: Comparative evaluation of liquid and gaseous biofuels. Fermentation 2025, 11, 448. [Google Scholar] [CrossRef]
- Santos, B.; Freitas, F.; Sobral, A.J.F.N.; Encarnação, T. Microalgae and circular economy: Unlocking waste to resource pathways for sustainable development. Int. J. Sustain. Eng. 2025, 18, 2501488. [Google Scholar] [CrossRef]
- Ahmad, A.; Ashraf, S.S. Efficient cultivation and scale-up of marine microalgae Fistulifera peliculosa and Nannochloropsis oculata for sustainable aquaculture applications. Chem. Eng. J. Adv. 2025, 22, 100720. [Google Scholar] [CrossRef]
- Sarker, N.K.; Kaparaju, P. Microalgal Bioeconomy: A Green Economy Approach Towards Achieving Sustainable Development Goals. Sustainability 2024, 16, 11218. [Google Scholar] [CrossRef]
- Tham, P.E.; Lim, H.R.; Khoo, K.S.; Chew, K.W.; Yap, Y.J.; Munawaroh, H.S.H.; Ma, Z.; Rajendran, S.; Gnanasekaran, L.; Show, P.L. Insights of microalgae-based aquaculture feed: A review on circular bioeconomy and perspectives. Algal Res. 2023, 74, 103186. [Google Scholar] [CrossRef]
- Okeke, E.S.; Ejeromedoghene, O.; Okoye, C.O.; Ezeorba, T.P.C.; Nyaruaba, R.; Ikechukwu, C.K.; Oladipo, A.; Orege, J.I. Microalgae biorefinery: An integrated route for the sustainable production of high-value-added products. Energy Convers. Manag. X 2022, 16, 100323. [Google Scholar] [CrossRef]
- Liu, J. Interspecific biodiversity enhances biomass and lipid productivity of microalgae as biofuel feedstock. J. Appl. Phycol. 2016, 28, 25–33. [Google Scholar] [CrossRef]
- Stockenreiter, M.; Haupt, F.; Seppälä, J.; Tamminen, T.; Spilling, K. Nutrient uptake and lipid yield in diverse microalgal communities grown in wastewater. Algal Res. 2016, 15, 77–82. [Google Scholar] [CrossRef]
- Tossavainen, M.; Katyal Chopra, N.; Kostia, S.; Valkonen, K.; Sharma, A.K.; Sharma, S.; Ojala, A.; Romantschuk, M. Conversion of biowaste leachate to valuable biomass and lipids in mixed cultures of Euglena gracilis and chlorophytes. Algal Res. 2018, 35, 76–84. [Google Scholar] [CrossRef]
- Tossavainen, M.; Lahti, K.; Edelmann, M.; Eskola, R.; Lampi, A.M.; Piironen, V.; Korvonen, P.; Ojala, A.; Romantschuk, M. Integrated utilization of microalgae cultured in aquaculture wastewater: Wastewater treatment and production of valuable fatty acids and tocopherols. J. Appl. Phycol. 2019, 31, 1753–1763. [Google Scholar] [CrossRef]
- Milhazes-Cunha, H.; Otero, A. Valorisation of aquaculture effluents with microalgae: The Integrated Multi-Trophic Aquaculture concept. Algal Res. 2017, 24, 416–424. [Google Scholar] [CrossRef]
- Kim, K.; Jung, J.Y.; Han, H.S. Utilization of microalgae in aquaculture system: Biological wastewater treatment. Emerg. Sci. J. 2019, 3, 209–221. [Google Scholar] [CrossRef]
- Li, M.; Callier, M.D.; Blancheton, J.P.; Galès, A.; Nahon, S.; Triplet, S.; Geoffroy, T.; Menniti, C.; Fouilland, E.; D’ORbcastel, E.R. Bioremediation of fishpond effluent and production of microalgae for an oyster farm in an innovative recirculating integrated multi-trophic aquaculture system. Aquaculture 2019, 504, 314–325. [Google Scholar] [CrossRef]
- Dhokane, D.; Shaikh, A.; Yadav, A.; Giri, N.; Bandyopadhyay, A.; Dasgupta, S.; Bhadra, B. CRISPR-based bioengineering in microalgae for production of industrially important biomolecules. Front. Bioeng. Biotechnol. 2023, 11, 1267826. [Google Scholar] [CrossRef]
- Ahmad Kamal, A.H.; Mohd Hamidi, N.F.; Zakaria, M.F.; Ahmad, A.; Harun, M.R.; Chandra Segaran, T.; Jusoh, M. Genetically engineered microalgae for enhanced bioactive compounds. Discov. Appl. Sci. 2024, 6, 482. [Google Scholar] [CrossRef]
- Lin, W.R.; Ng, I.S. Development of CRISPR/Cas9 system in Chlorella vulgaris FSP-E to enhance lipid accumulation. Enzyme Microb. Technol. 2019, 133, 109458. [Google Scholar] [CrossRef]
- Sarker, P.K.; Schoffstall, B.V.; Kapuscinski, A.R.; McKuin, B.; Fitzgerald, D.; Greenwood, C.; O’Shelski, K.; Pasion, E.N.; Gwynne, D.; Gonzalez Orcajo, D.; et al. Towards sustainable aquafeeds: Microalgal (Nannochloropsis sp. QH25) co-product biomass can fully replace fishmeal in the feeds for rainbow trout (Oncorhynchus mykiss). Sustainability 2025, 14, 781. [Google Scholar] [CrossRef] [PubMed]
- Tacon, A.G.J.; Hasan, M.R.; El-Sayed, A.; Kaushik, S. Aquaculture feeds—Addressing the long-term sustainability of the sector. In FAO Fisheries and Aquaculture Technical Paper No. 958; FAO: Rome, Italy, 2012. [Google Scholar]
- Yun, J.H.; Cho, D.H.; Lee, S.; Heo, J.; Tran, Q.G.; Chang, Y.K.; Kim, H.-S. Hybrid operation of photobioreactor and wastewater-fed open raceway ponds enhances the dominance of target algal species and algal biomass production. Algal Res. 2018, 29, 319–329. [Google Scholar] [CrossRef]
| Metabolic Ability | Microalgae Cultivation | Reference |
|---|---|---|
| Photoautotrophic |
| [89] |
| Heterotrophic |
| |
| Mixotrophic |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balasubramaniam, V.; Rathi, D.-N.G.; Mustar, S.; Lee, J.C. Microalgae as an Eco-Friendly and Functional Ingredient for Sustainable Aquafeed. Aquac. J. 2025, 5, 14. https://doi.org/10.3390/aquacj5030014
Balasubramaniam V, Rathi D-NG, Mustar S, Lee JC. Microalgae as an Eco-Friendly and Functional Ingredient for Sustainable Aquafeed. Aquaculture Journal. 2025; 5(3):14. https://doi.org/10.3390/aquacj5030014
Chicago/Turabian StyleBalasubramaniam, Vimala, Devi-Nair Gunasegavan Rathi, Suraiami Mustar, and June Chelyn Lee. 2025. "Microalgae as an Eco-Friendly and Functional Ingredient for Sustainable Aquafeed" Aquaculture Journal 5, no. 3: 14. https://doi.org/10.3390/aquacj5030014
APA StyleBalasubramaniam, V., Rathi, D.-N. G., Mustar, S., & Lee, J. C. (2025). Microalgae as an Eco-Friendly and Functional Ingredient for Sustainable Aquafeed. Aquaculture Journal, 5(3), 14. https://doi.org/10.3390/aquacj5030014






