Comparative Evaluation of Marine Algae-Based Biostimulants for Enhancing Growth, Physiological Performance, and Essential Oil Yield in Lavender (Lavandula angustifolia) Under Greenhouse Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site and Conditions
2.2. Plant Material and Pot Setup
2.3. Experimental Design
- Treatment A: Ascophyllum nodosum APP053.1 (France, Brittany) (composition: Fucoidan: 350 mg/g dry weight (DW); Alginic acid: 100 mg/g DW; Laminarin: 52 mg/g DW; Mannitol: 61 mg/g DW) (composition: Alginic Acid: 28% of dry weight (DW); Fucoidan: 6% mg/g DW; Laminarin: 12% mg/g DW; Mannitol: 13% mg/g DW; Total carbohydrates: 36% DW; Protein: 8% DW; Ash/Mineral Content: 17% DW; Phlorotannins (polyphenols): 6% DW).
- Treatment B: Laminaria digitata APP054.1 (Iceland) (composition: Alginic acid mg/g 26%; Laminarin mg/g 12%; Fucoidan mg/g 7%; Mannitol mg/g 11%; Total carbohydrates 48% (incl. polysaccharides); Polyphenols (phlorotannins) 6%; Proteins (total nitrogen) 7%; Ash (mineral content) 23%; Pigments (fucoxanthin) <1%; Vitamins (A, C, E, K) trace amounts).
- Treatment C: Ascophyllum nodosum APP055.1 (Greenland) (composition: Alginic acid 32%; Laminarin 11%; Fucoidan 6%; Mannitol 12%; Fucoxanthin 660 mg/Kg DW).
- Treatment D: Sargassum muticum APP031.10 (Italy, Venice) (composition: Proteins 8.3% mg/g DW; Total sugars 28 mg/g DW; Crude fiber (cell wall polysaccharides, mainly alginate): 32% mg/g DW; Macro-minerals (Na + K + Ca + Mg): 135 mg/g DW; Micro-elements (Se, Fe, Zn, Mn, Cu, Ni, Cr): 2 mg/g DW).
- Treatment E (Commercial Control 1): Seaweed-based commercial biostimulant (Ecklonia maxima-derived, Kelpak®, composition: Cytokinins (natural): 0.03–0.06 mg/L; Auxins (natural): 11–14 mg/L; Organic matter: >60%; Carbohydrates: Including alginates and laminarins; Minerals: Potassium, calcium, magnesium, and trace elements).
- Treatment F (Commercial Control 2): Amino acid-based commercial biostimulant (Trainer®, Enzymatic hydrolysate of plant proteins, composition: Total nitrogen (N): 6%; Organic nitrogen: 5.5%; Free amino acids: ≥24% w/w; Carbon from biological origin: >20%).
- Treatment G (Negative Control): Untreated control—plants received only standard basal fertilization, with no foliar or soil-applied biostimulants.
- Relevance: Both products are well-established in the European biostimulant market and commonly used in high-value crops including ornamentals, herbs, and medicinal plants.
- Contrast in biostimulant categories:
- 3.
- Benchmarking: Including these controls ensures that the efficacy of experimental macroalgae treatments (A–D) can be benchmarked against real-world, commercially available alternatives. This allows researchers and growers to assess whether novel algae sources (like Ascophyllum nodosum from Greenland or Sargassum muticum) offer competitive or superior performance.
2.4. Fertilization Protocol
- A slow-release organic fertilizer (NPK 6-5-10) incorporated at transplant (5 g/pot);
- Supplementary watering with a balanced liquid fertilizer (NPK 20-20-20 at 1 g/L) once every 2 weeks to avoid nutrient deficiencies under greenhouse conditions.
2.5. Biostimulant Preparation and Application
- Foliar spray: A quantity of 100 mL of solution was sprayed uniformly over the plant canopy using a handheld mist sprayer, ensuring full leaf coverage.
- Soil drench: An amount of 150 mL of solution was applied directly to the substrate to reach the rhizosphere.
2.6. Abiotic Stress Application
- Control (non-stressed): Plants were watered regularly (every 2–3 days) to maintain substrate moisture at 75% field capacity.
- Drought stress: Irrigation was withheld for 10 days during the flowering stage (week 8), after which plants were re-watered and monitored.
- Salinity stress: Starting from week 6, plants were irrigated with 100 mM NaCl solution twice a week for 4 weeks, simulating gradual salt buildup.
2.7. Measured Parameters
2.7.1. Morphological Growth Parameters
- Plant height (cm) was measured weekly using a digital meter from the base of the stem to the apical meristem.
- Leaf area (cm2) was measured from three representative leaves per plant using a portable digital leaf area meter (Model LI-3000, LI-COR) [41].
2.7.2. Biomass Evaluation
- Vegetative biomass (g) was recorded after drying the aerial part of the plant at 65 °C for 72 h.
- Root biomass (g) was similarly obtained by washing roots free of soil, drying, and weighing.
2.7.3. Chlorophyll Content
2.7.4. Microbial Activity in Substrate
- Substrate samples (5 g) were collected at week 10 from the root zone of each replicate.
- Samples were diluted (1:10) in sterile water and serially plated on nutrient agar and PDA (potato dextrose agar) for bacterial and fungal counts, respectively.
- Colony-forming units (CFU) were counted after 48 h at 28 °C.
2.7.5. Essential Oil Yield Analysis
- Oil yield was expressed as a percentage of dry biomass.
- Oils were stored in amber vials at 4 °C for future chemical profiling.
2.7.6. Antioxidant Enzyme Assays
- At the end of the stress period (week 10), fresh leaf tissue (0.5 g) was collected, frozen in liquid nitrogen, and stored at −80 °C. Enzyme extracts were prepared using phosphate buffer (pH 7.0), and the following assays were performed [45]:
- SOD activity: Based on the photoreduction of NBT (nitroblue tetrazolium);
- CAT activity: Measured by the decomposition rate of H2O2 at 240 nm;
- POD activity: Quantified via guaiacol oxidation at 470 nm.
2.7.7. Osmoprotectant Quantification
- Free proline content: Measured via the Bates method (1973), using sulfosalicylicacid extraction and ninhydrin reaction (absorbance at 520 nm);
- Soluble sugar content: Determined using the phenol-sulfuric acid method (absorbance at 490 nm).
2.8. Statistical Analysis
3. Results
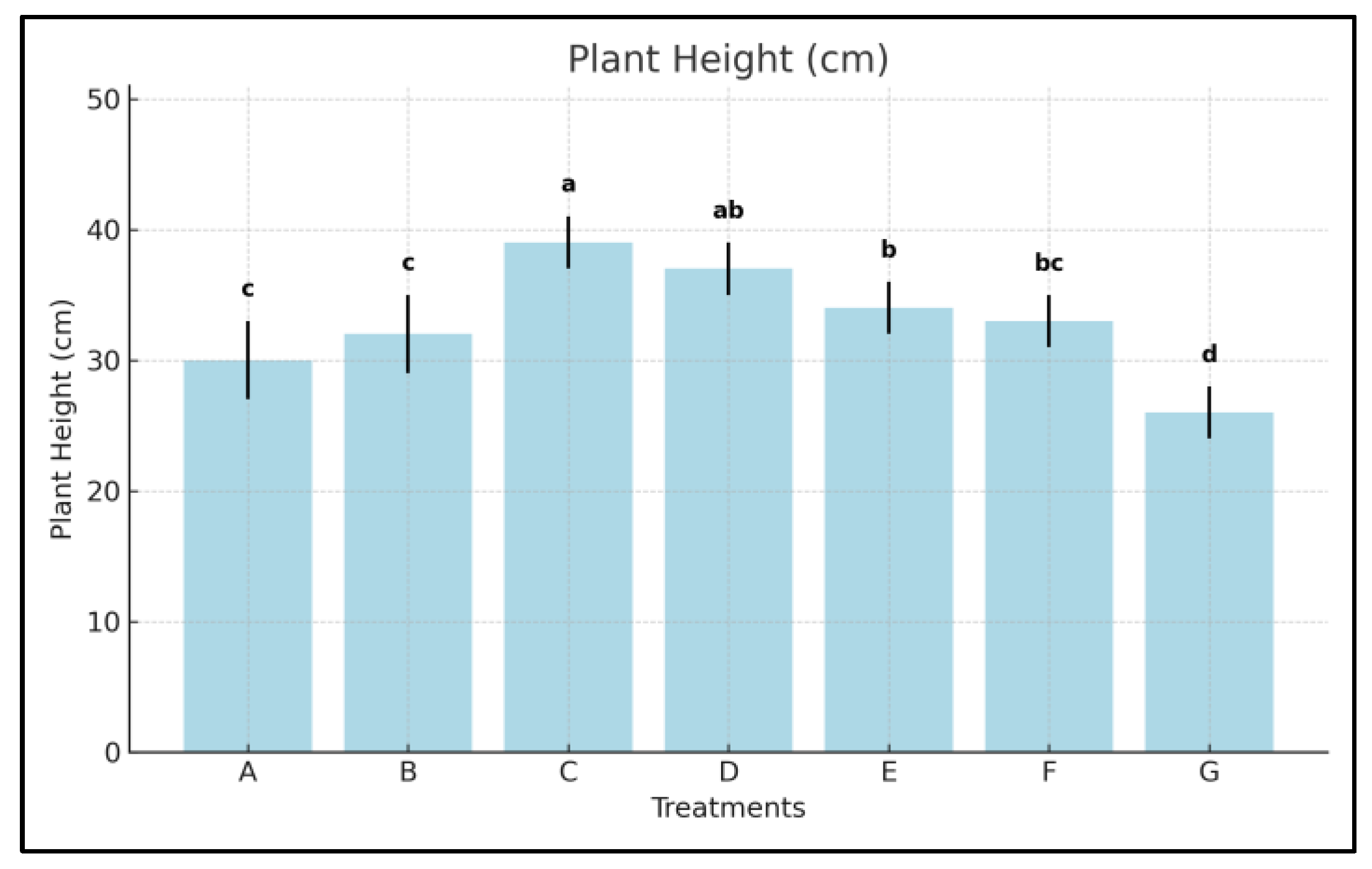
3.1. Plant Height
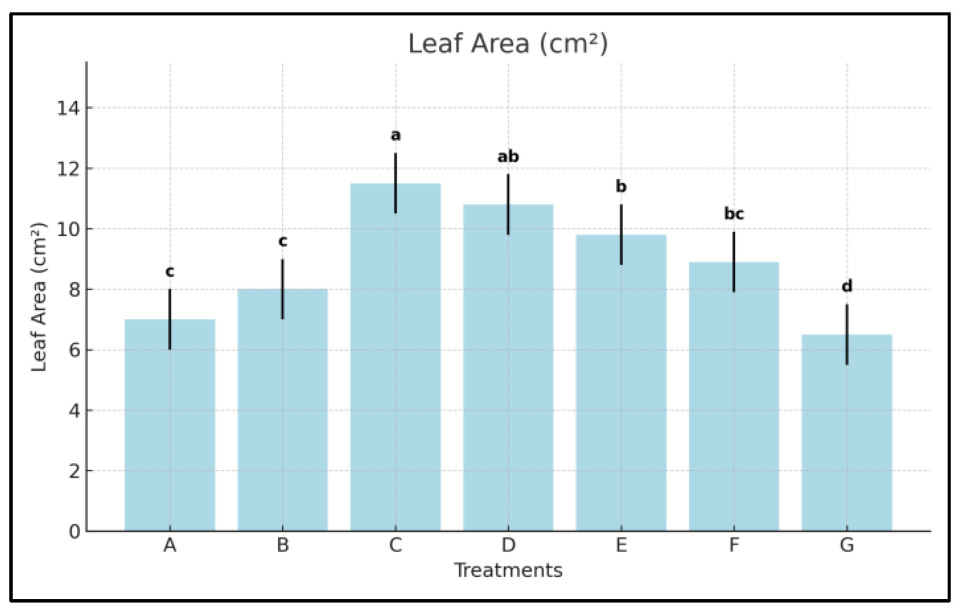
3.2. Leaf Area
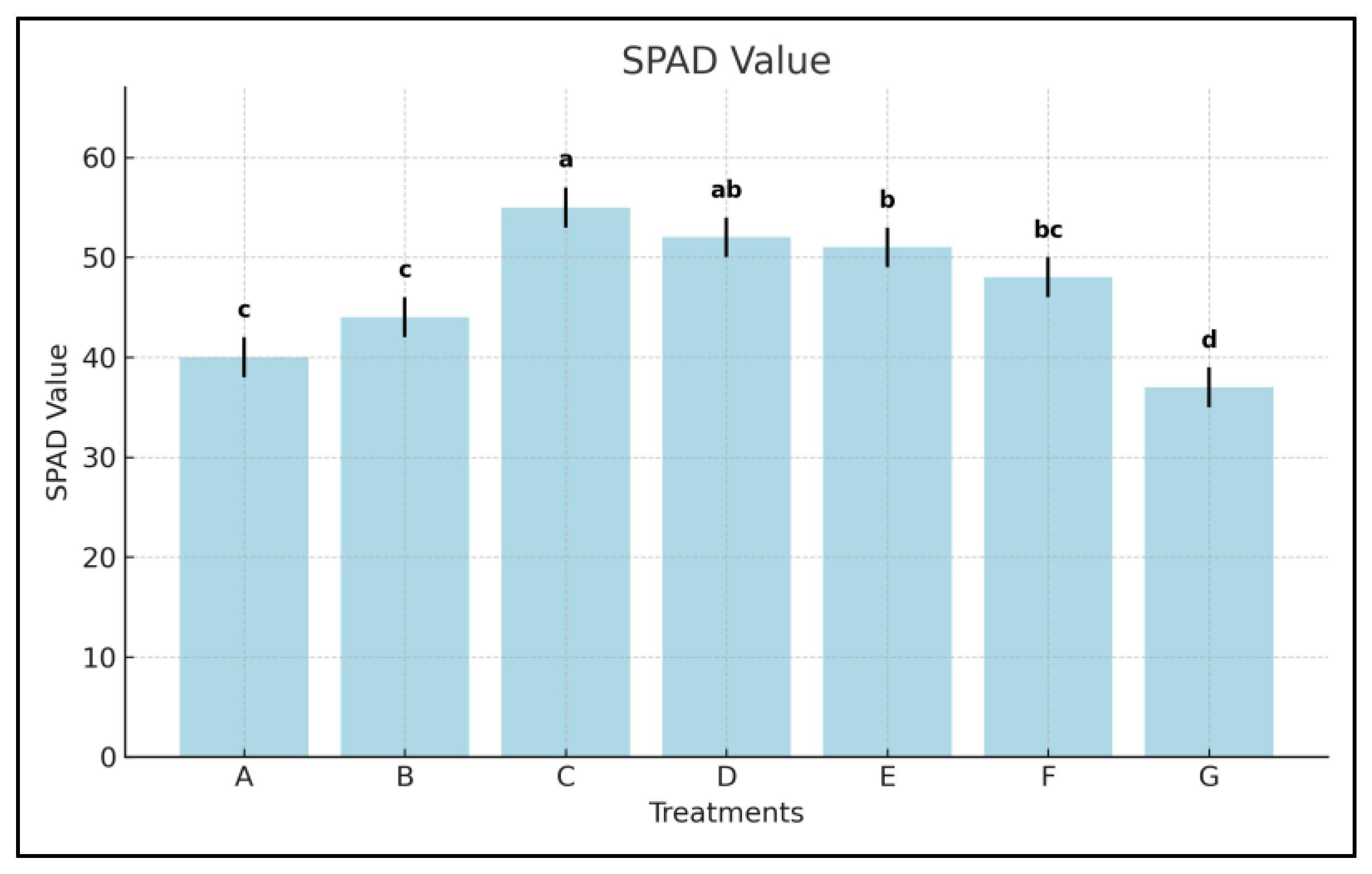
3.3. Chlorophyll Content (SPAD Index)
3.4. Microbial Colony Counts in the Rhizosphere
- The highest bacterial CFU count was observed in Treatment D (Sargassum muticum), averaging 9.1 × 106 CFU g−1, followed closely by Treatment C (A. nodosum—Greenland) at 8.6 × 106 CFU g−1.
- The commercial seaweed product (E) yielded 7.8 × 106 CFU g−1, while the amino acid biostimulant (F) produced 6.9 × 106 CFU g−1.
- The untreated control (G) had the lowest bacterial count at 5.3 × 106 CFU g−1.
- Treatment D recorded the highest fungal CFU value at 4.2 × 106 CFU g−1, with Treatment C close behind at 3.9 × 106 CFU g−1.
- Commercial Treatments E and F showed moderate fungal stimulation (3.4 and 3.1 × 106 CFU g−1, respectively).
- The control group again had the lowest fungal activity (2.2 × 106 CFU g−1).
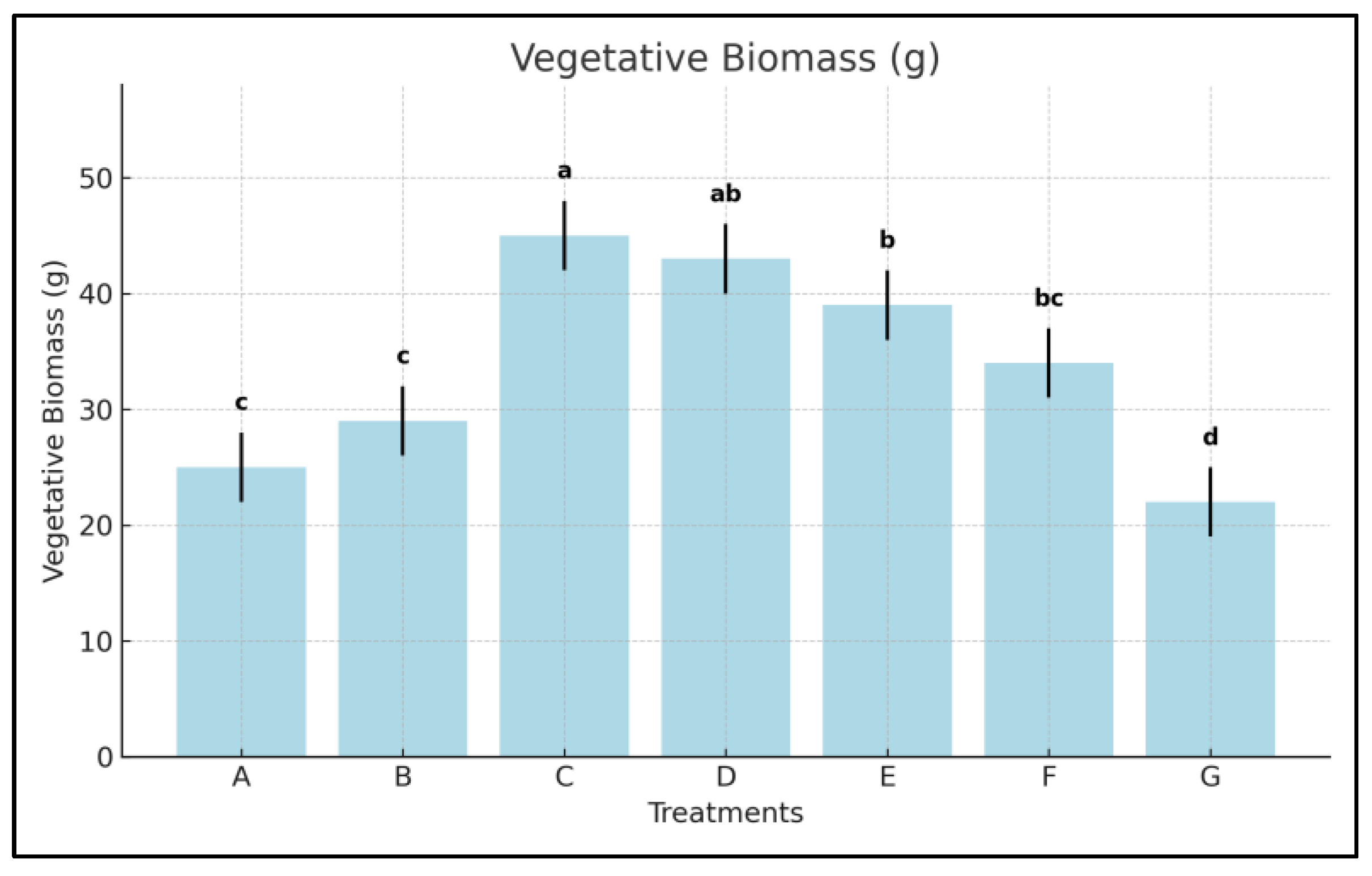
3.5. Vegetative Biomass
3.6. Improvement Root Biomass
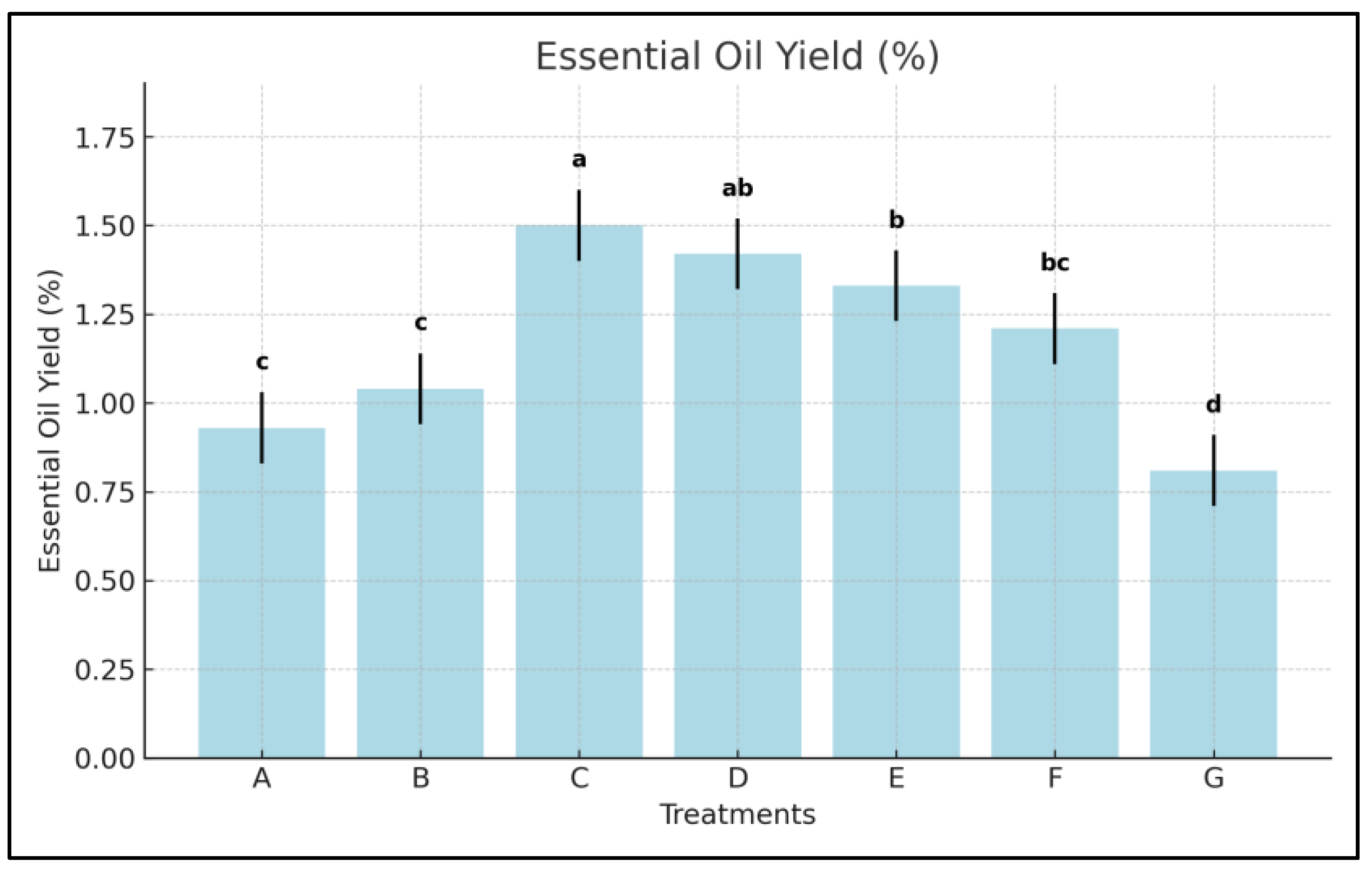
3.7. Essential Oil Yield
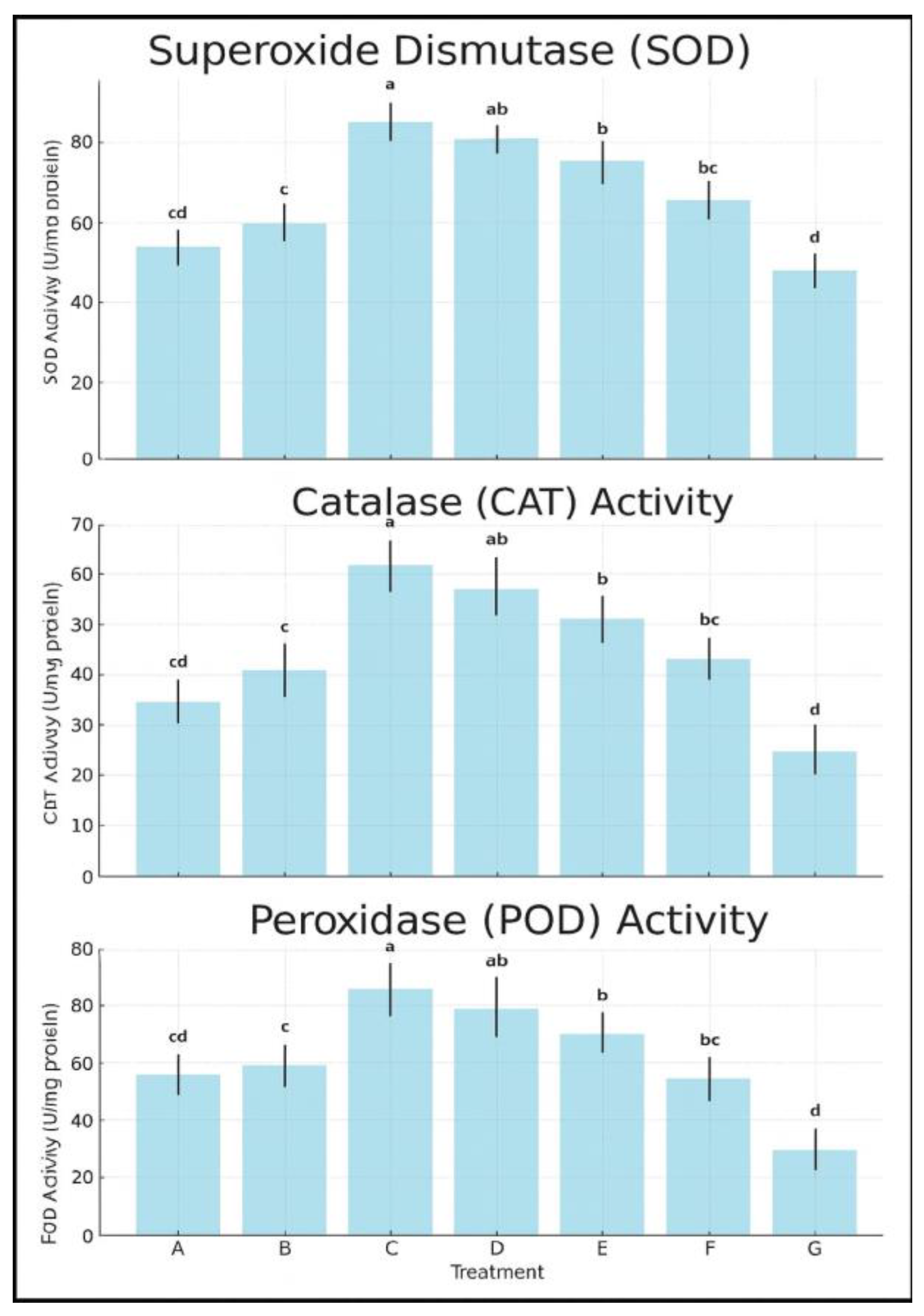
3.8. Improvement Antioxidant Enzyme Activity
- SOD activity peaked in Treatment C at 85.2 U/mg protein, followed closely by D (80.6 U/mg) and E (75.1 U/mg), while the untreated control recorded only 47.3 U/mg (Figure 8).
- CAT activity in Treatment C reached 61.4 U/mg, significantly higher than all other treatments; the control group recorded only 29.7 U/mg (Figure 8).
- POD activity was similarly enhanced in C and D (102.5 and 98.3 U/mg, respectively), compared to 58.9 U/mg in the control (Figure 8).
- Trends were comparable, with SOD activity again highest in Treatment C (88.7 U/mg), followed by D (84.2 U/mg), while the control remained significantly lower (50.1 U/mg).
- CAT and POD levels followed similar patterns, with Treatments C and D consistently forming the highest statistical groupings (p < 0.05).
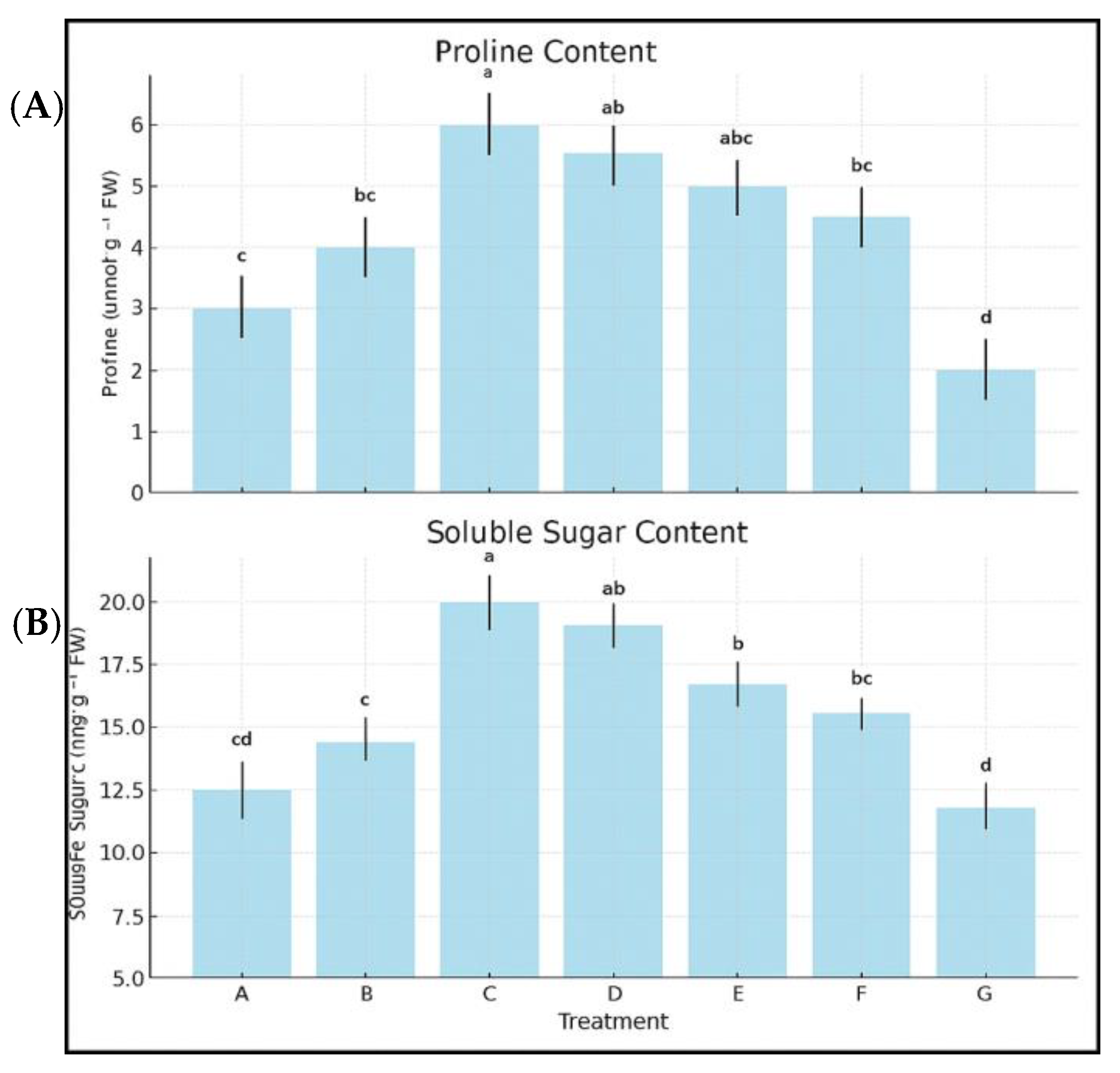
3.9. Proline and Soluble Sugar Accumulation
3.10. Comparative Efficacy of Macroalgal and Commercial Biostimulants
3.10.1. Plant Height
- Treatment C increased plant height by +51.2% over the control, while Treatment D showed a +43.7% gain.
- The commercial seaweed product E (Kelpak®) led to a +34.8% increase, whereas the amino acid-based F (Trainer®) improved height by +29.7%.
- Thus, Treatment C was 16.4% more effective than Kelpak® and 21.5% better than Trainer® in promoting vertical growth.
3.10.2. Leaf Area
- C-treated plants exhibited a +81.0% increase in leaf area compared to control; Treatments D and E achieved +69.8% and +57.1%, respectively.
- The amino acid product (F) provided only a +39.7% improvement.
- Treatment C outperformed the best commercial comparator (E) by 15.2%, confirming its superior capacity for foliar expansion.
3.10.3. Chlorophyll Content (SPAD Index)
- SPAD values rose by +47.3% with Treatment C and +41.6% with D, while commercial Treatments E and F showed increases of +37.2% and +29.9%, respectively.
- Algal extracts from Greenland and Venice were 10–18% more effective than commercial formulations in enhancing chlorophyll concentration, reflecting improved photosynthetic efficiency.
3.10.4. Vegetative Biomass
- Treatment C led to a +109.3% increase in shoot biomass relative to the control, D followed with +97.7%, and E achieved +83.2%.
- The amino acid biostimulant (F) contributed to a +57.0% increase.
- Compared to F, Treatment C nearly doubled biomass accumulation (+92.3% over F), highlighting the superior growth-promoting effect of cold-climate algae.
3.10.5. Root Biomass
- The most effective root biomass stimulation was recorded in D (+113.0%) and C (+101.9%) versus control.
- Commercial products showed +70.4% (F) and +70.3% (E) increases.
- These results indicate that whole-algae extracts foster better root architecture than either amino acids or standard seaweed formulations.
3.10.6. Essential Oil Yield
- Oil-yield improvements reached +83.9% in Treatment C and +75.3% in D, compared to +64.2% in E and +49.4% in F.
- In practical terms, Treatment C produced 12.0% more oil than Kelpak®, and 34.5% more than Trainer®, which is a substantial commercial advantage in aromatic crop production.
3.10.7. Antioxidant Enzyme Activity
- Under drought stress, SOD activity in Treatment C was +80.1% higher than in control, compared to +58.8% in E and +38.1% in F.
- Similarly, CAT and POD activities followed this trend, reinforcing the superior biochemical resilience of algae-based treatments from cold or dynamic environments.
3.10.8. Osmoprotectant Accumulation
- Proline levels in C-treated plants were +203% higher than the control, while Kelpak® and Trainer® improved proline by +145% and +119%, respectively.
- Soluble sugar content increased by +126.4% in Treatment C vs. +79.3% in Kelpak® and +64.3% in Trainer®.
- These results suggest a clear biochemical advantage of the experimental macroalgal treatments in promoting osmotic stress tolerance.
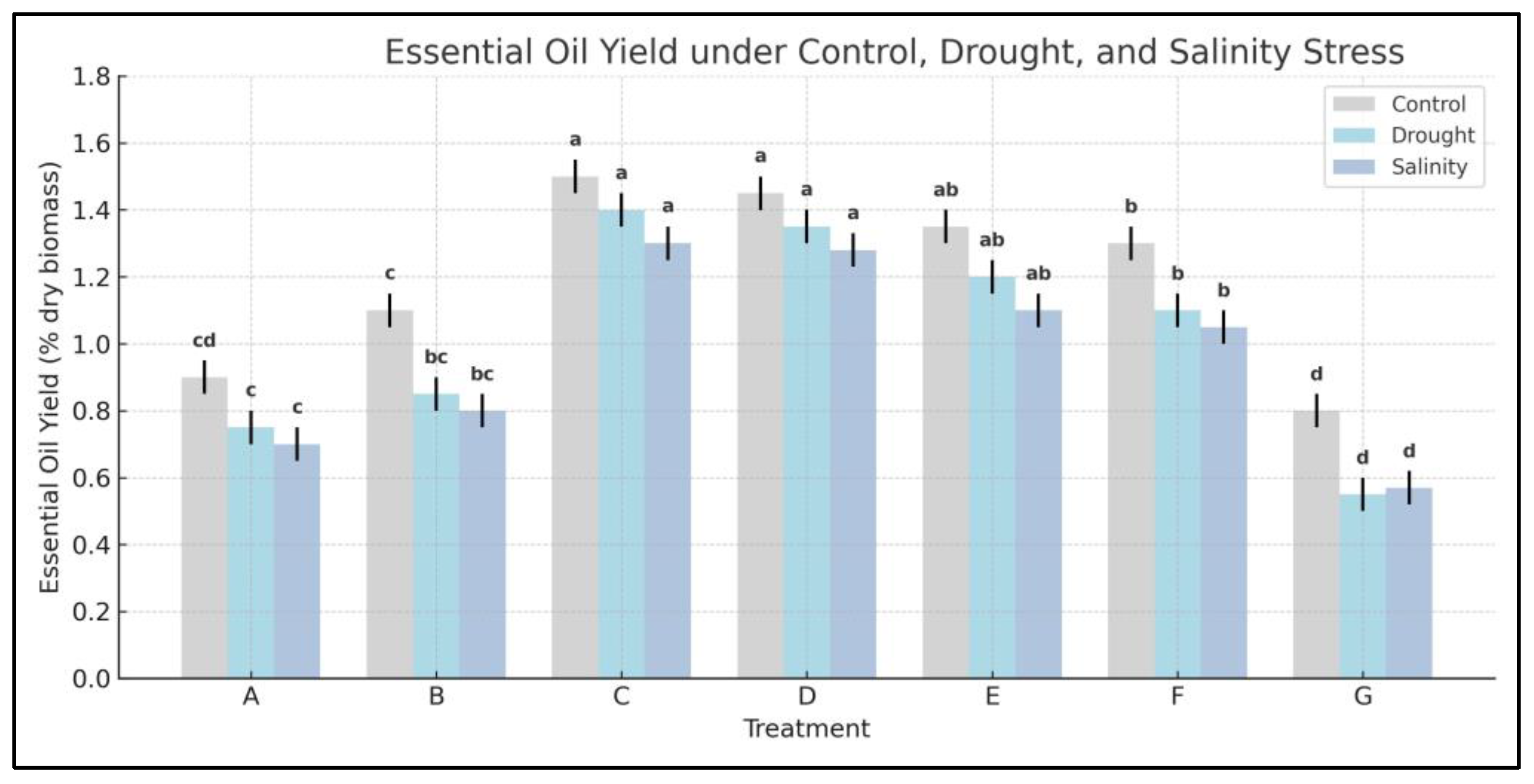
3.11. Improvement Plant Responses to Abiotic Stress Conditions
3.11.1. Effect of Drought and Salinity on Agronomic Characters of Lavender
3.11.2. Essential Oil Yield Preservation
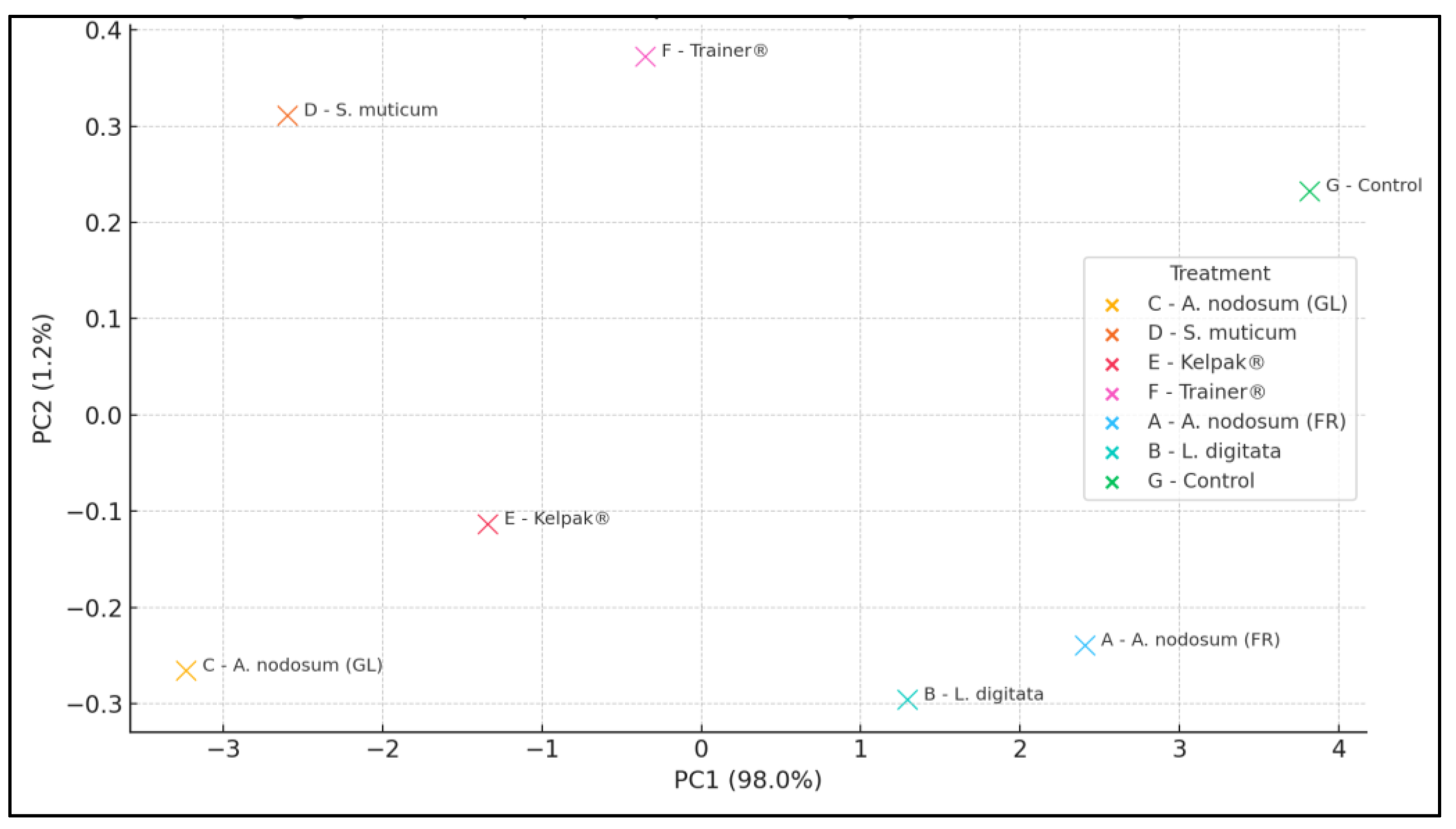
3.12. Principal Component Analysis (PCA)
4. Discussion
4.1. Growth and Morphological Improvements
4.2. Chlorophyll Content and Photosynthetic Capacity
4.3. Biomass Accumulation and Nutrient Efficiency
4.4. Essential Oil Production
4.5. Rhizosphere Effects and Microbial Stimulation
- Improved nutrient cycling;
- Increased enzyme activity;
- Suppression of pathogenic organisms.
4.6. Abiotic Stress Tolerance
4.7. Comparison with Commercial Products
4.8. Origin and Composition of Algae Matter
4.9. Antioxidant Defense and Osmotic Adjustment Under Drought Stress
4.9.1. Enhanced Antioxidant Enzyme Activity
4.9.2. Osmoprotectant Accumulation and Osmotic Regulation
4.10. Plant Responses to Abiotic Stress Conditions
4.11. Principal Component Analysis (PCA)—Interpretation
4.12. Agronomic Relevance and Applications
4.13. Future Directions
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pirani, H.; Ebadi, M.-T.; Rezaei, A. Effect of seaweed fertiliser foliar application on growth parameters, yield, essential oil content and composition of Hyssopus officinalis. Iran. J. Med. Aromat. Plants Res. 2020, 36, 3. [Google Scholar]
- Caccialupi, G.; Caradonia, F.; Ronga, D.; Ben Hassine, M.; Truzzi, E.; Benvenuti, S.; Francia, E. Protein hydrolysate and brown algal extracts enhance lavandin biomass under field conditions. Agronomy 2022, 12, 2189. [Google Scholar] [CrossRef]
- Gavrić, T.; Gadžo, D.; Erhatić, R.; Hafner-Vuk, K. The effect of the use of biostimulant on the yield and quality of lavandin and lavender. Not. Bot. Horti Agrobot. Cluj-Napoca 2024, 52, 13967. [Google Scholar] [CrossRef]
- Di Fan, A.T.; Critchley, A.T. Seaweed extracts-treated food and their benefits for shelf life and animal/human consumption. In Applications of Seaweeds in Food and Nutrition; Elsevier: Amsterdam, The Netherlands, 2024; pp. 129–174. [Google Scholar] [CrossRef]
- Albertos, I.; Martin-Diana, A.B.; Burón, M.; Rico, D. Development of functional bio-based seaweed (Himanthalia elongata and Palmaria palmata) edible films for extending the shelf life of fresh fish burgers. Food Packag. Shelf Life 2019, 22, 100382. [Google Scholar] [CrossRef]
- Hussain, H.I.; Kasinadhuni, N.; Arioli, T. The effect of seaweed extract on tomato plant growth, productivity and soil. J. Appl. Phycol. 2021, 33, 1305–1314. [Google Scholar] [CrossRef]
- Wally, O.S.D.; Critchley, A.T.; Hiltz, D.; Craigie, J.S.; Han, X.; Zaharia, L.I.; Abrams, S.R.; Prithiviraj, B. Regulation of phytohormone biosynthesis and accumulation in Arabidopsis following treatment with commercial extract from the marine macroalga Ascophyllum nodosum. J. Plant Growth Regul. 2013, 32, 324–339. [Google Scholar] [CrossRef]
- Trejo Valencia, R.; Sánchez Acosta, L.; Fortis Hernández, M.; Preciado Rangel, P.; Gallegos Robles, M.Á.; Antonio Cruz, R.d.C.; Vázquez Vázquez, C. Effect of seaweed aqueous extracts and compost on vegetative growth, yield, and nutraceutical quality of cucumber (Cucumis sativus L.) fruit. Agronomy 2018, 8, 264. [Google Scholar] [CrossRef]
- Kumari, S.; Sehrawat, K.D.; Phogat, D.; Sehrawat, A.R.; Chaudhary, R.; Sushkova, S.N.; Voloshina, M.S.; Rajput, V.D.; Shmaraeva, A.N.; Marc, R.A. Ascophyllum nodosum (L.) Le Jolis, a pivotal biostimulant toward sustainable agriculture: A comprehensive review. Agriculture 2023, 13, 1179. [Google Scholar] [CrossRef]
- Di Sario, L.; Boeri, P.; Matus, J.T.; Pizzio, G.A. Plant biostimulants to enhance abiotic stress resilience in crops. Int. J. Mol. Sci. 2025, 26, 1129. [Google Scholar] [CrossRef]
- Shukla, P.S.; Borza, T.; Critchley, A.T.; Prithiviraj, B. Seaweed-based compounds and products for sustainable protection against plant pathogens. Mar. Drugs 2021, 19, 59. [Google Scholar] [CrossRef]
- Trejo-Téllez, L.I.; Gómez-Merino, F.C. Phosphite as an inductor of adaptive responses to stress and stimulator of better plant performance. In Biotic and Abiotic Stress Tolerance in Plants; Vats, S., Ed.; Springer: Singapore, 2018. [Google Scholar] [CrossRef]
- Ahmed, M.; Ullah, H.; Piromsri, K.; Tisarum, R.; Cha-um, S.; Datta, A. Effects of an Ascophyllum nodosum seaweed extract application dose and method on growth, fruit yield, quality, and water productivity of tomato under water-deficit stress. S. Afr. J. Bot. 2022, 151 Part A, 95–107. [Google Scholar] [CrossRef]
- Stirk, W.A.; Arthur, G.D.; Lourens, A.F.; Novak, O.; Strnad, M.; van Staden, J. Changes in cytokinin and auxin concentrations in seaweed concentrates when stored at an elevated temperature. J. Appl. Phycol. 2004, 16, 31–39. [Google Scholar] [CrossRef]
- Kocira, A.; Lamorska, J.; Kornas, R.; Nowosad, N.; Tomaszewska, M.; Leszczyńska, D.; Kozłowicz, K.; Tabor, S. Changes in biochemistry and yield in response to biostimulants applied in bean (Phaseolus vulgaris L.). Agronomy 2020, 10, 189. [Google Scholar] [CrossRef]
- Drury, R.E. Interaction of plant hormones. Science 1969, 164, 564–565. [Google Scholar] [CrossRef]
- Abou Baker, D.H.; Amarowicz, R.; Kandeil, A.; Ali, M.A.; Ibrahim, E.A. Antiviral activity of Lavandula angustifolia L. and Salvia officinalis L. essential oils against avian influenza H5N1 virus. J. Agric. Food Res. 2021, 4, 100135. [Google Scholar] [CrossRef]
- Truzzi, E.; Benvenuti, S.; Bertelli, D.; Francia, E.; Ronga, D. Effects of biostimulants on the chemical composition of essential oil and hydrosol of lavandin (Lavandula x intermedia Emeric ex Loisel.) cultivated in Tuscan-Emilian Apennines. Molecules 2021, 26, 6157. [Google Scholar] [CrossRef]
- Bulgari, R.; Franzoni, G.; Ferrante, A. Biostimulants application in horticultural crops under abiotic stress conditions. Agronomy 2019, 9, 306. [Google Scholar] [CrossRef]
- Boutahiri, S.; Benrkia, R.; Tembeni, B.; Idowu, O.E.; Olatunji, O.J. Effect of biostimulants on the chemical profile of food crops under normal and abiotic stress conditions. Curr. Plant Biol. 2024, 40, 100410. [Google Scholar] [CrossRef]
- Matarese, F.; Cuzzola, A.; Scalabrelli, G.; D’Onofrio, C. Expression of terpene synthase genes associated with the formation of volatiles in different organs of Vitis vinifera. Phytochemistry 2014, 105, 12–24. [Google Scholar] [CrossRef]
- Ali, O.; Ramsubhag, A.; Jayaraman, J. Biostimulant properties of seaweed extracts in plants: Implications towards sustainable crop production. Plants 2021, 10, 531. [Google Scholar] [CrossRef]
- De Clercq, P.; Pauwels, E.; Top, S.; Steppe, K.; Van Labeke, M.-C. Effect of seaweed-based biostimulants on growth and development of Hydrangea paniculata under continuous or periodic drought stress. Horticulturae 2023, 9, 509. [Google Scholar] [CrossRef]
- Hernández-Herrera, R.M.; Gómez-Leyva, J.F.; Sánchez-Hernández, C.V.; Ocampo-Álvarez, H.; Ramírez-Romero, R.; Palmeros-Suárez, P.A. Seaweed extract ameliorates salt stress in tomato plants by enhancing the antioxidant system and expression of stress-responsive genes. J. Appl. Phycol. 2024, 36, 2269–2282. [Google Scholar] [CrossRef]
- Mansori, M.; Chernane, H.; Latique, S.; Benaliat, A.; Hsissou, D.; El Kaoua, M. Seaweed extract effect on water deficit and antioxidative mechanisms in bean plants (Phaseolus vulgaris L.). J. Appl. Phycol. 2015, 27, 1689–1698. [Google Scholar] [CrossRef]
- Kumar, G.; Nanda, S.; Singh, S.K.; Kumar, S.; Singh, D.; Singh, B.N.; Mukherjee, A. Seaweed extracts: Enhancing plant resilience to biotic and abiotic stresses. Front. Mar. Sci. 2024, 11, 1457500. [Google Scholar] [CrossRef]
- Ali, S.; Akhtar, M.S.; Siraj, M.; Zaman, W. Molecular communication of microbial plant biostimulants in the rhizosphere under abiotic stress conditions. Int. J. Mol. Sci. 2024, 25, 12424. [Google Scholar] [CrossRef]
- Sabir, A.; Yazar, K.; Sabir, F.; Kara, Z.; Yazici, M.A.; Goksu, N. Vine growth, yield, berry quality attributes and leaf nutrient content of grapevines as influenced by seaweed extract (Ascophyllum nodosum) and nanosize fertilizer pulverizations. Sci. Hortic. 2014, 175, 1–8. [Google Scholar] [CrossRef]
- Cataldo, E.; Fucile, M.; Mattii, G.B. Biostimulants in viticulture: A sustainable approach against biotic and abiotic stresses. Plants 2022, 11, 162. [Google Scholar] [CrossRef]
- Sleighter, R.L.; Hanson, T.; Holden, D.; Richards, K.M. Abiotic stress mitigation: A case study from 21 trials using a natural organic matter-based biostimulant across multiple geographies. Agronomy 2023, 13, 728. [Google Scholar] [CrossRef]
- Rejeb, I.B.; Pastor, V.; Mauch-Mani, B. Plant responses to simultaneous biotic and abiotic stress: Molecular mechanisms. Plants 2014, 3, 458–475. [Google Scholar] [CrossRef] [PubMed]
- Poveda, J.; Baptista, P. Filamentous fungi as biocontrol agents in olive (Olea europaea L.) diseases: Mycorrhizal and endophytic fungi. Crop Prot. 2021, 146, 105672. [Google Scholar] [CrossRef]
- Zhao, J.; Davis, L.C.; Verpoorte, R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol. Adv. 2005, 23, 283–333. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Kulkarni, M.G.; White, J.F.; Stirk, W.A.; Papenfus, H.B.; Doležal, K.; Ördög, V.; Norrie, J.; Critchley, A.T.; Van Staden, J. Categories of various plant biostimulants—Mode of application and shelf-life. In Biostimulants for Crops from Seed Germination to Plant Development; Gupta, S., Van Staden, J., Eds.; Academic Press: Cambridge, MA, USA, 2021; pp. 1–60. [Google Scholar] [CrossRef]
- Wang, M.; Chen, L.; Li, Y.; Chen, L.; Liu, Z.; Wang, X.; Yan, P.; Qin, S. Responses of soil microbial communities to a short-term application of seaweed fertilizer revealed by deep amplicon sequencing. Appl. Soil Ecol. 2018, 125, 288–296. [Google Scholar] [CrossRef]
- Raghunandan, B.L.; Vyas, R.V.; Patel, H.K.; Jhala, Y.K. Perspectives of seaweed as organic fertilizer in agriculture. In Soil Fertility Management for Sustainable Development; Panpatte, D., Jhala, Y., Eds.; Springer: Singapore, 2019. [Google Scholar] [CrossRef]
- Mayer, A.M.S.; Mayer, V.A.; Swanson-Mungerson, M.; Pierce, M.L.; Rodríguez, A.D.; Nakamura, F.; Taglialatela-Scafati, O. Marine pharmacology in 2019–2021: Marine compounds with antibacterial, antidiabetic, antifungal, anti-inflammatory, antiprotozoal, antituberculosis and antiviral activities; affecting the immune and nervous systems, and other miscellaneous mechanisms of action. Mar. Drugs 2024, 22, 309. [Google Scholar] [CrossRef]
- Jacomassi, L.M.; Viveiros, J.O.; Oliveira, M.P.; Momesso, L.; de Siqueira, G.F.; Crusciol, C.A.C. A seaweed extract-based biostimulant mitigates drought stress in sugarcane. Front. Plant Sci. 2022, 13, 865291. [Google Scholar] [CrossRef]
- Garcia-Caparros, P.; Ciriello, M.; Rouphael, Y.; Giordano, M. The role of organic extracts and inorganic compounds as alleviators of drought stress in plants. Horticulturae 2025, 11, 91. [Google Scholar] [CrossRef]
- Chen, J.M.; Black, T.A. Defining leaf area index for non-flat leaves. Plant Cell Environ. 1992, 15, 421–429. [Google Scholar] [CrossRef]
- Shibaeva, T.G.; Mamaev, A.V.; Sherudilo, E.G. Evaluation of a SPAD-502 Plus chlorophyll meter to estimate chlorophyll content in leaves with interveinal chlorosis. Russ. J. Plant Physiol. 2020, 67, 690–696. [Google Scholar] [CrossRef]
- Larkin, R.P.; Honeycutt, C.W. Effects of different 3-year cropping systems on soil microbial communities and Rhizoctonia diseases of potato. Phytopathology 2006, 96, 68–79. [Google Scholar] [CrossRef]
- Kaya, D.A.; Ghica, M.V.; Dănilă, E.; Öztürk, Ş.; Türkmen, M.; Albu Kaya, M.G.; Dinu-Pîrvu, C.-E. Selection of optimal operating conditions for extraction of Myrtus communis L. essential oil by the steam distillation method. Molecules 2020, 25, 2399. [Google Scholar] [CrossRef]
- Ghaderi, N.; Hatami, M.R.; Mozafari, A.; Siosehmardeh, A. Change in antioxidant enzymes activity and some morpho-physiological characteristics of strawberry under long-term salt stress. Physiol. Mol. Biol. Plants 2018, 24, 833–843. [Google Scholar] [CrossRef]
- Birhanie, Z.M.; Yang, D.; Luan, M.; Xiao, A.; Liu, L.; Zhang, C.; Biswas, A.; Dey, S.; Deng, Y.; Li, D. Salt stress induces changes in physiological characteristics, bioactive constituents, and antioxidants in kenaf (Hibiscus cannabinus L.). Antioxidants 2022, 11, 2005. [Google Scholar] [CrossRef]
- Greenacre, M.; Groenen, P.J.F.; Hastie, T.; d’Enza, A.I.; Markos, A.; Tuzhilina, E. Principal component analysis. Nat. Rev. Methods Primers 2022, 2, 100. [Google Scholar] [CrossRef]
- Consentino, B.B.; Ciriello, M.; Sabatino, L.; Vultaggio, L.; Baldassano, S.; Vasto, S.; Rouphael, Y.; La Bella, S.; De Pascale, S. Current acquaintance on agronomic biofortification to modulate the yield and functional value of vegetable crops: A review. Horticulturae 2023, 9, 219. [Google Scholar] [CrossRef]
- Yakhin, O.I.; Lubyanov, A.A.; Yakhin, I.A.; Brown, P.H. Biostimulants in plant science: A global perspective. Front. Plant Sci. 2017, 7, 2049. [Google Scholar] [CrossRef]
- Ericksen, P.J.; Ingram, J.S.I.; Liverman, D.M. Food security and global environmental change: Emerging challenges. Environ. Sci. Policy 2009, 12, 373–377. [Google Scholar] [CrossRef]
- Zuma, M.; Arthur, G.; Coopoosamy, R.; Naidoo, K. Incorporating cropping systems with eco-friendly strategies and solutions to mitigate the effects of climate change on crop production. J. Agric. Food Res. 2023, 14, 100722. [Google Scholar] [CrossRef]
- Knapp, S.; Peralta, I.E. The tomato (Solanum lycopersicum L., Solanaceae) and its botanical relatives. In The Tomato Genome; Causse, M., Giovannoni, J., Bouzayen, M., Zouine, M., Eds.; Compendium of Plant Genomes; Springer: Berlin/Heidelberg, Germany, 2016; pp. 7–21. ISBN 978-3-662-53387-1. [Google Scholar]
- Keatinge, J.D.H.; Yang, R.-Y.; Hughes, J.d.A.; Easdown, W.J.; Holmer, R. The importance of vegetables in ensuring both food and nutritional security in attainment of the Millennium Development Goals. Food Secur. 2011, 3, 491–501. [Google Scholar] [CrossRef]
- Kumari, R.; Kaur, I.; Bhatnagar, A.K. Effect of aqueous extract of Sargassum johnstonii Setchell & Gardner on growth, yield and quality of Lycopersicon esculentum Mill. J. Appl. Phycol. 2011, 23, 623–633. [Google Scholar] [CrossRef]
- Perveen, R.; Suleria, H.A.; Anjum, F.M.; Butt, M.S.; Pasha, I.; Ahmad, S. Tomato (Solanum lycopersicum) carotenoids and lycopenes chemistry; metabolism, absorption, nutrition, and allied health claims—A comprehensive review. Crit. Rev. Food Sci. Nutr. 2015, 55, 919–929. [Google Scholar] [CrossRef] [PubMed]
- Igile, G.O.; Ekpe, O.O.; Essien, N.M.; Bassey, S.C.; Agiang, M.A. Quality characteristics of tomato juice produced and preserved with and without its seeds. Dannish J. Food Sci. Technol. 2016, 2, 1–9. [Google Scholar]
- Aldrich, H.T.; Salandanan, K.; Kendall, P.; Bunning, M.; Stonaker, F.; Külen, O.; Stushnoff, C. Cultivar choice provides options for local production of organic and conventionally produced tomatoes with higher quality and antioxidant content. J. Sci. Food Agric. 2010, 90, 46–55. [Google Scholar] [CrossRef]
- Felföldi, Z.; Ranga, F.; Roman, I.A.; Sestras, A.F.; Vodnar, D.C.; Prohens, J.; Sestras, R.E. Analysis of physico-chemical and organoleptic fruit parameters relevant for tomato quality. Agronomy 2022, 12, 1232. [Google Scholar] [CrossRef]
- Sardaro, M.L.S.; Marmiroli, M.; Maestri, E.; Marmiroli, N. Genetic characterization of Italian tomato varieties and their traceability in tomato food products. Food Sci. Nutr. 2013, 1, 54–62. [Google Scholar] [CrossRef]
- Petro-Turza, M. Flavor of tomato and tomato products. Food Rev. Int. 1986, 2, 309–351. [Google Scholar] [CrossRef]
- Gautier, H.; Diakou-Verdin, V.; Bénard, C.; Reich, M.; Buret, M.; Bourgaud, F.; Poëssel, J.L.; Caris-Veyrat, C.; Génard, M. How does tomato quality (sugar, acid, and nutritional quality) vary with ripening stage, temperature, and irradiance? J. Agric. Food Chem. 2008, 56, 1241–1250. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Jiang, P.; Liu, Y.; Miao, X.; Liu, A. Distinct changes of taste quality and metabolite profile in different tomato varieties revealed by LC-MS metabolomics. Food Chem. 2024, 442, 138456. [Google Scholar] [CrossRef]
- Pahalvi, H.N.; Rafiya, L.; Rashid, S.; Nisar, B.; Kamili, A.N. Chemical fertilizers and their impact on soil health. In Microbiota and Biofertilizers; Dar, G.H., Bhat, R.A., Mehmood, M.A., Hakeem, K.R., Eds.; Springer International Publishing: Cham, Switzerland, 2021; Volume 2, pp. 1–20. ISBN 978-3-030-61009-8. [Google Scholar]
- Priya, A.K.; Alagumalai, A.; Balaji, D.; Song, H. Bio-based agricultural products: A sustainable alternative to agrochemicals for promoting a circular economy. RSC Sustain. 2023, 1, 746–762. [Google Scholar] [CrossRef]
- Osorio-Reyes, J.G.; Valenzuela-Amaro, H.M.; Pizaña-Aranda, J.J.P.; Ramírez-Gamboa, D.; Meléndez-Sánchez, E.R.; López-Arellanes, M.E.; Castañeda-Antonio, M.D.; Coronado-Apodaca, K.G.; Gomes Araújo, R.; Sosa-Hernández, J.E.; et al. Microalgae-based biotechnology as alternative biofertilizers for soil enhancement and carbon footprint reduction: Advantages and implications. Mar. Drugs 2023, 21, 93. [Google Scholar] [CrossRef] [PubMed]
- Rayirath, P.; Benkel, B.; Hodges, D.M.; Allan-Wojtas, P.; Mackinnon, S.; Critchley, A.T.; Prithiviraj, B. Lipophilic components of the brown seaweed, Ascophyllum nodosum, enhance freezing tolerance in Arabidopsis thaliana. Planta 2009, 230, 135–147. [Google Scholar] [CrossRef]
- Tabassum, M.R.; Xia, A.; Murphy, J.D. Seasonal variation of chemical composition and biomethane production from the brown seaweed Ascophyllum nodosum. Bioresour. Technol. 2016, 216, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Arias, A.; Feijoo, G.; Moreira, M.T. Macroalgae as a sustainable biostimulant for crop production according to techno-economic and environmental criteria. Sustain. Prod. Consum. 2024, 48, 169–180. [Google Scholar] [CrossRef]
- Kholssi, R.; Lougraimzi, H.; Grina, F.; Lorentz, J.F.; Silva, I.; Castaño-Sánchez, O.; Marks, E.A.N. Green agriculture: A review of the application of micro- and macroalgae and their impact on crop production on soil quality. J. Soil Sci. Plant Nutr. 2022, 22, 4627–4641. [Google Scholar] [CrossRef]
- Wan, A.H.L.; Davies, S.J.; Soler-Vila, A.; Fitzgerald, R.; Johnson, M.P. Macroalgae as a sustainable aquafeed ingredient. Rev. Aquac. 2019, 11, 458–492. [Google Scholar] [CrossRef]
- Bayat, F.; Afshar, A.; Baghban, N. Algal cells-derived extracellular vesicles: A review with special emphasis on their antimicrobial effects. Front. Microbiol. 2021, 12, 785716. [Google Scholar] [CrossRef]
- Spagnuolo, D.; Russo, V.; Manghisi, A.; Di Martino, A.; Morabito, M.; Genovese, G.; Trifilò, P. Screening on the presence of plant growth regulators in high biomass forming seaweeds from the Ionian Sea (Mediterranean Sea). Sustainability 2022, 14, 3914. [Google Scholar] [CrossRef]
- Rathod, S.G.; Bhushan, S.; Mantri, V.A. Phytohormones and pheromones in the phycology literature: Benchmarking of dataset and developing critical tools of biotechnological implications for commercial aquaculture industry. Phycology 2023, 4, 1–36. [Google Scholar] [CrossRef]
- Khan, W.; Rayirath, U.P.; Subramanian, S.; Jithesh, M.N.; Rayorath, P.; Hodges, D.M.; Critchley, A.T.; Craigie, J.S.; Norrie, J.; Prithiviraj, B. Seaweed extracts as biostimulants of plant growth and development. J. Plant Growth Regul. 2009, 28, 386–399. [Google Scholar] [CrossRef]
- Cozzolino, E.; Di Mola, I.; Ottaiano, L.; El-Nakhel, C.; Rouphael, Y.; Mori, M. Foliar application of plant-based biostimulants improve yield and upgrade qualitative characteristics of processing tomato. Ital. J. Agron. 2021, 16, 1825. [Google Scholar] [CrossRef]
- Song, C.; Lorz, L.R.; Lee, J.; Cho, J.Y. In vitro photoprotective, anti-inflammatory, moisturizing, and antimelanogenic effects of a methanolic extract of Chrysophyllum lucentifolium Cronquist. Plants 2021, 11, 94. [Google Scholar] [CrossRef] [PubMed]
- Battacharyya, D.; Babgohari, M.Z.; Rathor, P.; Prithiviraj, B. Seaweed extracts as biostimulants in horticulture. Sci. Hortic. 2015, 196, 39–48. [Google Scholar] [CrossRef]
- Gharib, F.A.E.L.; Zeid, I.M.; Salem, O.M.A.H.; Zakaria, E. Effects of Sargassum latifolium extract on growth, oil content and enzymatic activities of rosemary plants under salinity stress. Life Sci. J. 2014, 11, 933–945. [Google Scholar]
- Alestra, T.; Schiel, D.R. Impacts of local and global stressors in intertidal habitats: Influence of altered nutrient, sediment and temperature levels on the early life history of three habitat-forming macroalgae. J. Exp. Mar. Biol. Ecol. 2015, 468, 29–36. [Google Scholar] [CrossRef]
- Cruz de Carvalho, M.H. Drought stress and reactive oxygen species: Production, scavenging and signaling. Plant Signal Behav. 2008, 3, 156–165. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Zulfiqar, F.; Raza, A.; Mohsin, S.M.; Mahmud, J.A.; Fujita, M.; Fotopoulos, V. Reactive oxygen species and antioxidant defense in plants under abiotic stress: Revisiting the crucial role of a universal defense regulator. Antioxidants 2020, 9, 681. [Google Scholar] [CrossRef] [PubMed]
- Sugumaran, R.; Padam, B.S.; Yong, W.T.L.; Saallah, S.; Ahmed, K.; Yusof, N.A. A retrospective review of global commercial seaweed production—Current challenges, biosecurity and mitigation measures and prospects. Int. J. Environ. Res. Public Health 2022, 19, 7087. [Google Scholar] [CrossRef]
- Desoky, E.M.S.; Elrys, A.S.; Mansour, E.; Eid, R.S.M.; Selem, E.; Rady, M.M.; Ali, E.F.; Mersal, G.A.M.; Semida, W.M. Application of biostimulants promotes growth and productivity by fortifying the antioxidant machinery and suppressing oxidative stress in faba bean under various abiotic stresses. Sci. Hortic. 2021, 288, 110340. [Google Scholar] [CrossRef]
- Renzetti, M.; Funck, D.; Trovato, M. Proline and ROS: A unified mechanism in plant development and stress response? Plants 2025, 14, 2. [Google Scholar] [CrossRef]
- Paulert, R.; Ascrizzi, R.; Malatesta, S.; Berni, P.; Noseda, M.D.; de Carvalho, M.M.; Marchioni, I.; Pistelli, L.; Duarte, M.E.R.; Mariotti, L. Ulva intestinalis extract acts as biostimulant and modulates metabolites and hormone balance in basil (Ocimum basilicum L.) and parsley (Petroselinum crispum L.). Plants 2021, 10, 1391. [Google Scholar] [CrossRef]
- Kohli, S.K.; Khanna, K.; Bhardwaj, R.; Abd Allah, E.F.; Ahmad, P.; Corpas, F.J. Assessment of subcellular ROS and NO metabolism in higher plants: Multifunctional signaling molecules. Antioxidants 2019, 8, 641. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, P.; Torrecillas, A.; Morales, M.A.; Ortuño, M.F.; Sánchez-Blanco, M.J. Effects of NaCl salinity and water stress on growth and leaf water relations of Asteriscus maritimus plants. Environ. Exp. Bot. 2005, 53, 113–123. [Google Scholar] [CrossRef]
- Carillo, P. Can biostimulants enhance plant resilience to heat and water stress in the Mediterranean hotspot? Plant Stress 2025, 16, 100802. [Google Scholar] [CrossRef]
- Yetik, A.K.; Şen, B. Optimizing lavender (Lavandula angustifolia Mill.) yield and water productivity with deficit irrigation in semi-arid climates. Agronomy 2025, 15, 1009. [Google Scholar] [CrossRef]
- Rouphael, Y.; Carillo, P.; Garcia-Perez, P.; Cardarelli, M.; Senizza, B.; Miras-Moreno, B.; Colla, G.; Lucini, L. Plant biostimulants from seaweeds or vegetal proteins enhance the salinity tolerance in greenhouse lettuce by modulating plant metabolism in a distinctive manner. Sci. Hortic. 2022, 305, 111368. [Google Scholar] [CrossRef]
- Cerruti, P.; Campobenedetto, C.; Montrucchio, E.; Agliassa, C.; Contartese, V.; Acquadro, A.; Bertea, C.M. Antioxidant activity and comparative RNA-seq analysis support mitigating effects of an algae-based biostimulant on drought stress in tomato plants. Physiol. Plant. 2024, 176, e70007. [Google Scholar] [CrossRef]
- Priolo, D.; Tolisano, C.; Brienza, M.; Del Buono, D. Insight into the biostimulant effect of an aqueous duckweed extract on tomato plants. Agriculture 2024, 14, 808. [Google Scholar] [CrossRef]
- Regni, L.; Tolisano, C.; Del Buono, D.; Priolo, D.; Proietti, P. Role of an aqueous extract of duckweed (Lemna minor L.) in increasing salt tolerance in Olea europaea L. Agriculture 2024, 14, 375. [Google Scholar] [CrossRef]
- Prisa, D.; Spagnuolo, D. Evaluation of the bio-stimulating activity of lake algae extracts on edible cacti Mammillaria prolifera and Mammillaria glassii. Plants 2022, 11, 3586. [Google Scholar] [CrossRef]
- Spagnuolo, D.; Bressi, V.; Chiofalo, M.T.; Morabito, M.; Espro, C.; Genovese, G.; Iannazzo, D.; Trifilò, P. Using the aqueous phase produced from hydrothermal carbonization process of brown seaweed to improve the growth of Phaseolus vulgaris. Plants 2023, 12, 2745. [Google Scholar] [CrossRef] [PubMed]
- Fatimah, S.; Daud, N. The effect of seaweed extract (Sargassum sp.) used as fertilizer on plant growth of Capsicum annum (chilli) and Lycopersicon esculentum (tomato). Indones. J. Sci. Technol. 2018, 3, 115–123. [Google Scholar] [CrossRef]
- Espinosa-Antón, A.A.; Zamora-Natera, J.F.; Zarazúa-Villaseñor, P.; Santacruz-Ruvalcaba, F.; Sánchez-Hernández, C.V.; Águila Alcántara, E.; Torres-Morán, M.I.; Velasco-Ramírez, A.P.; Hernández-Herrera, R.M. Application of seaweed generates changes in the substrate and stimulates the growth of tomato plants. Plants 2023, 12, 1520. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Wang, X.; Chen, B.; Zhang, M.; Ma, J. Seaweed extract improved yields, leaf photosynthesis, ripening time, and net returns of tomato (Solanum lycopersicum Mill.). ACS Omega 2020, 5, 4242–4249. [Google Scholar] [CrossRef] [PubMed]
- González-González, M.F.; Ocampo-Álvarez, H.; Santacruz-Ruvalcaba, F.; Sánchez-Hernández, C.V.; Casarrubias-Castillo, K.; Becerril-Espinosa, A.; Castañeda-Nava, J.J.; Hernández-Herrera, R.M. Physiological, ecological, and biochemical implications in tomato plants of two plant biostimulants: Arbuscular mycorrhizal fungi and seaweed extract. Front. Plant Sci. 2020, 11, 999. [Google Scholar] [CrossRef] [PubMed]
- Yao, G.-F.; Li, C.; Sun, K.-K.; Tang, J.; Huang, Z.-Q.; Yang, F.; Huang, G.-G.; Hu, L.-Y.; Jin, P.; Hu, K.-D. Hydrogen sulfide maintained the good appearance and nutrition in post-harvest tomato fruits by antagonizing the effect of ethylene. Front. Plant Sci. 2020, 11, 584. [Google Scholar] [CrossRef]
- Jalali, P.; Roosta, H.R.; Khodadadi, M.; Torkashvand, A.M.; Jahromi, M.G. Effects of brown seaweed extract, silicon, and selenium on fruit quality and yield of tomato under different substrates. PLoS ONE 2022, 17, e0277923. [Google Scholar] [CrossRef]
- Rama Rao, K. Effect of aqueous seaweed extract on Zizyphus mauritiana Lam. J. Indian Bot. Soc. 1991, 71, 19–21. [Google Scholar]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spagnuolo, D.; Jamal, A.; Prisa, D. Comparative Evaluation of Marine Algae-Based Biostimulants for Enhancing Growth, Physiological Performance, and Essential Oil Yield in Lavender (Lavandula angustifolia) Under Greenhouse Conditions. Phycology 2025, 5, 41. https://doi.org/10.3390/phycology5030041
Spagnuolo D, Jamal A, Prisa D. Comparative Evaluation of Marine Algae-Based Biostimulants for Enhancing Growth, Physiological Performance, and Essential Oil Yield in Lavender (Lavandula angustifolia) Under Greenhouse Conditions. Phycology. 2025; 5(3):41. https://doi.org/10.3390/phycology5030041
Chicago/Turabian StyleSpagnuolo, Damiano, Aftab Jamal, and Domenico Prisa. 2025. "Comparative Evaluation of Marine Algae-Based Biostimulants for Enhancing Growth, Physiological Performance, and Essential Oil Yield in Lavender (Lavandula angustifolia) Under Greenhouse Conditions" Phycology 5, no. 3: 41. https://doi.org/10.3390/phycology5030041
APA StyleSpagnuolo, D., Jamal, A., & Prisa, D. (2025). Comparative Evaluation of Marine Algae-Based Biostimulants for Enhancing Growth, Physiological Performance, and Essential Oil Yield in Lavender (Lavandula angustifolia) Under Greenhouse Conditions. Phycology, 5(3), 41. https://doi.org/10.3390/phycology5030041







