Abstract
Methylmercury (MeHg) is the most important and the most abundant organic Hg pollutant in the aquatic ecosystem that can affect human health through biomagnification. It is the most toxic organic Hg form, which occurs naturally and by human-induced contamination in water and is further biomagnified in the aquatic food web. MeHg is the only Hg form that accumulates in living organisms and is able to cross the blood–brain barrier, presenting an enormous health risk. Anthropogenic activity increases eutrophication of coastal waters worldwide, which promotes algae blooms. Microalgae, as primary producers, are especially sensitive to MeHg exposure in water and are an important entrance point for MeHg into the aquatic food web. MeHg assimilated by microalgae is further transferred to fish, wildlife and, eventually, humans as final consumers. MeHg biomagnifies and bioaccumulates in living organisms and has serious negative health effects on humans, especially newborns and children. Knowledge of the microalgae–MeHg interaction at the bottom of the food web provides key insights into the control and prevention of MeHg exposure in humans and wildlife. This review aims to summarize recent findings in the literature on the microalgae–MeHg interaction, which can be used to predict MeHg transfer and toxicity in the aquatic food web.
1. Introduction
Mercury is a naturally occurring element that is highly poisonous to humans and wildlife [1]. Exposure to Hg has a number of potential negative effects on human health, including cognitive deficits in children due to fetal exposure, increases in fatal and nonfatal heart attacks and increases in premature death in adults [1]. Anthropogenic activities largely contribute to the mobilization of Hg from long-term geologic storage into the biosphere, where Hg cycles between the air, soil and water. While inorganic Hg does not accumulate in living organisms, microbes convert a portion of Hg to methylmercury (MeHg), which is then subjected to further biomagnification in the aquatic food web [1,2] and causes health risks to humans, especially among populations that frequently consume fish [3]. MeHg is bound to fish proteins that form muscle tissue and cannot be removed by cooking. Even the consumption of farmed fish can lead to MeHg exposure because of the presence of MeHg in feed [3]. To prevent MeHg accumulation, it is important to understand how MeHg enters at the base of the aquatic food web, where the natural feed for fish is located [4]. MeHg is a highly toxic organic Hg form present in natural water ecosystems; it is easily taken up by microalgae and significantly concentrates in the aquatic food web [5]. Once accumulated inside the microalgae cell, it is further transferred to grazers, fish larvae, fish, aquatic birds and, eventually, humans as final consumers.
Eutrophication is another phenomenon that can enhance the transfer of MeHg, not only to microalgae, but also to higher trophic levels [6]. Eutrophication in coastal areas is a globally expanding problem that can effect an increase in MeHg production and its accumulation in food webs [6]. This phenomenon also leads to greater net primary production, which increases organic matter (OM) concentration and decreases light penetration, thereby changing redox conditions in the water column and sediments [6].
Although there are significant data on MeHg uptake and its toxicity in microalgae [2,4,5,7,8,9,10], a detailed understanding of the accumulation processes in microalgae at the molecular level is still partially missing [9]. This review aims to summarize recent findings in the literature that can provide more a detailed understanding of the uptake, distribution, toxicity and complexation of MeHg in cultures of green microalgae. Data presented and discussed here can be used to better predict MeHg transfer and toxicity in the aquatic food web based on MeHg concentration and phytoplankton population size.
2. MeHg Interaction with Organic Matter (OM), S and Se Organic Compounds
A key aspect in MeHg bioaccumulation and toxicity is the degree to which MeHg interacts with complexing agents in solution (OH−, Cl−, organic sulfur compounds—especially thiols (ligands containing sulfhydryl group R–SH), OM, artificial chelating agents such as EDTA salts) [4,7]. OM that is naturally present in aqueous systems promotes Hg (II) methylation mainly because it contains metabolic substrate molecules essential for heterotrophic microbes and strong ligands that bind Hg(II) [11]. In the case of highly enclosed terrestrial water basins, such as the Baltic Sea, hypoxic and anoxic zones are caused by an increased biological oxygen demand (BOD) due to the excess of nutrients and OM runoffs where Hg binds to allochthonous (terrestrially discharged) OM in the surface layers of the water column and sinks to deeper waters, where it can be released in the microbial remineralization process of OM [12,13,14]. In natural waters where OM and reduced S species are absent, MeHg forms inorganic complexes, MeHgCl and MeHgOH, with chloride and hydroxide ions [15]. The chemical speciation of MeHg prior to cell exposure controls its bioavailability and is a key factor determining the uptake rate and accumulation in microalgae [10,16,17]. MeHg forms stronger complexes with organic thiols, which also have higher stability constants than complexes with OH− and Cl− groups [18].
The bioconcentration step of MeHg from water to the base of the food web is crucial for MeHg concentrations found in aquatic organisms higher up in the food web [5,15,19]. Uptake by microalgae is the first and largest step of MeHg bioaccumulation in aquatic food webs, which is influenced by many factors that drive seasonal changes in water MeHg concentrations [11]. OM is an important parameter of MeHg production and uptake by phytoplankton. Measurements from the Bothnian Sea in the Northern Baltic, taken as one of the few studied models of biogeochemical cycling of MeHg in natural aquatic environments, showed that MeHg concentrations can vary by up to an order of magnitude between years (from 103 ± 12 fM in September 2014 to 18 ± 9 fM in August 2016), which underlines the interannual variability in water column MeHg concentrations [11]. Both Hg(II) and MeHg preferentially bind to DOM over inorganic particles, which affects Hg bioavailability [11]. DOM can also decrease light attenuation in water, which decreases MeHg photodegradation. The association between MeHg and dissolved organic carbon (DOC) suggests that labile DOC is the important factor for the remineralization rate and Hg(II) methylation potential [11]. DOM promotes methylation of Hg(II) by (i) stimulating microbial activity and methylation processes, (ii) providing methyl groups for methylation, and (iii) enhancing the solubility of HgS(s) in mineral form [20]. Increased DOM inputs from terrestrial runoffs may lead to higher inputs of Hg and MeHg and increase MeHg formation in the aquatic ecosystem [20].
Hg methylation mainly takes place within 24 h after entering aquatic ecosystems, and to a greater extent under nonequilibrium conditions, before inorganic Hg becomes complexed with substances present in DOM [20]. It is often difficult to characterize DOM, and because ~50% of DOM is organic carbon, it is usually measured and expressed as DOC [16]. Natural levels of DOC vary in different aquatic environments: 4.0 ± 0.02 mg/L in the Baltic Sea; 3.78 ± 1.42 mg/L in Lake Titicaca (Bolivia), with peaks of up to 8 mg/L in some regions; 1.57–17.6 mg/L in the Sacramento River Delta (Sacramento, CA, USA) [16,21,22,23]. Humic matter forms part of allochthonous DOM in seawater [11]. In the case of the Northern Baltic, humic matter concentration decreases from the Bothnian Bay to the Bothnian Sea and further remains constant around 10 µg/L [11]. Humic content reduces microbial MeHg degradation by decreasing its bioavailability and decreases photodegradation of MeHg by increasing light attenuation [11]. Humic substances can also bind Hg (II) and correlate more strongly than DOC with Hg(II) concentration in water streams. Binding to humic substances makes Hg(II) less available to microbial reduction and methylation [24]. Humic substances are not easily remineralized; thus, an increased proportion of humic matter can decrease the activity of microorganisms that act by chemical reduction and methylation of Hg (II), which further decreases the rates of Hg transformation reactions [11]. In the Northern Baltic Sea, most Hg(II) was available for methylation by forming stable complexes with humic substances or by converting Hg(II) to elemental Hg [11]. The stability constants for Hg–ligand complexes drive metal internalization, thus determining rates of further chemical conversion of Hg species inside the cell. In this sense, it was demonstrated that a Hg–ligand (Hg–L) complex in the culture medium reacts with a biotic ligand (R) at the microalgae cell surface, forming a new complex (Hg–R) prior to metal internalization, and the rate for the formation of complexes at the cell surface is determined by the relative thermodynamic stability constants for Hg–L and Hg–R [10].
Similar to thiols (R–SH), organic selenium also has an affinity towards MeHg and can modify its toxicity by complexation [2]. Selenium belongs to the same group in the periodic table as S and exerts similar chemical affinities, so it can act as a S analogue in amino acids (selenomethionine—SeMet and selenocysteine—SeCys) as well as other organic compounds [25]. Some microalgae strains can take up Se(VI) salts from the medium, biotransform it to organic Se compounds, such as SeMet, which is less toxic to the cell than Se(VI), and accumulate it in the biomass [26,27]. A biologically important Se amino acid—SeMet [25]—significantly inhibits the uptake of MeHg by diatoms and mussels [28]. Detoxification of MeHg in seabirds and marine mammals involves its demethylation by reactive oxygen species (ROS) and the subsequent formation of high-molecular-weight Hg–Se–protein compounds, which are then degraded in lysosomes, forming insoluble Hg–Se compounds [29]. It was demonstrated that fish and wildlife living in environments with elevated Se levels exhibit lower MeHg accumulation [29]. There are a few proposed mechanisms for the protective role of Se against Hg toxicity, such as competition for binding sites or the formation of Hg–Se complexes. It was suggested that the increased Hg(II) uptake in the presence of SeMet is due to the formation of Hg–Se complexes that can be transported across the membrane at a faster rate [28].
3. Biogeochemical Cycling of MeHg and Its Presence in Global Aquatic Ecosystem
Human exposure to MeHg comes predominately from diets containing Hg-contaminated fish and seafood [30,31]. Understanding the cycling of Hg in aquatic systems is essential to assess the environmental risks to human health. Hg cycling differs in freshwater and ocean ecosystems. Hg can be released into the atmosphere both by natural (e.g., volcano eruptions) and anthropogenic sources: mining and burning of fossil fuels [32,33]. However, the more toxic organic form, MeHg, is the dominant species found in rice paddies and fish [34,35,36]. Hg speciation in nature has three different forms: elemental Hg(0), Hg(II) bound to particulate OM and cationic inorganic Hg(II), mainly as HgCl2. Elemental Hg(0) travels around the globe, and cationic Hg is transported at intermediate distances, while particulate is not transported very far [37].
MeHg contamination is also associated with long-term emissions from fossil fuel combustion throughout the industrialized world [38]. The burning of fossil fuels, coal, the extraction of gold, smelting and chemical production increase atmospheric Hg input in terrestrial and aquatic ecosystems [39]. The burning of coal is the most abundant anthropogenic source of Hg emissions in the USA [38]. Hg leaching from fossil fuel combustion can contribute to a significant increase in Hg concentration in rivers that enter the sea [30]. Subsequently, Hg(II) reduces to elemental Hg(0), which is usually volatilized to the atmosphere, while a small portion of Hg(II) is converted to MeHg, which is the most toxic form of Hg and acts primarily as a neurotoxin in humans and wildlife [31]. It is known that Hg emissions are ubiquitous, and MeHg as a contaminant bioaccumulates to a great extent in the aquatic food web [37]. It was estimated that bioconcentration factors of MeHg are 106–108 fold from initial water MeHg concentrations, resulting in MeHg levels that are of global toxicological concern [32,37].
Biogeochemical cycling of Hg includes emissions of anthropogenic Hg, transport through the atmosphere, deposition into ocean, subsequent transformation into MeHg [40], and its incorporation into aquatic food webs, which depends on its absorption or modification by microorganisms (Figure 1a). Microorganisms perform four types of Hg transformation: reduction of Hg(II) to Hg(0), degradation of organic Hg compounds, methylation of Hg(II) and the oxidation of Hg(0) to Hg(II) [31]. Methylation of Hg occurs in wetlands and lakes as a biological process performed by a range of known heterotrophic microbes (iron- and sulfur-reducing bacteria, methanogens and nitrite oxidizers) [11]. The bulk of MeHg in natural ecosystems originates from methylation of atmospherically deposited Hg by sulfur-reducing bacteria within aquatic sediments [32,38]. Reducing anaerobic conditions within aquatic sediments that support the growth and Hg methylation activity of sulfur-reducing bacteria are in redox potential (Eh) and pH ranges of 0.4 ≤ Eh ≤ 0.0 V and 5 ≤ pH ≤ 10 [38]. MeHg is predominately formed in hypoxic and anoxic environments from its inorganic form, Hg(II), via biological reactions, and the methylation process has been mostly linked to the presence of sulfate- and iron-reducing bacteria [41]. However, more recently, methanogens and other microbes have been found to play important roles in the MeHg formation process [42,43,44,45,46].
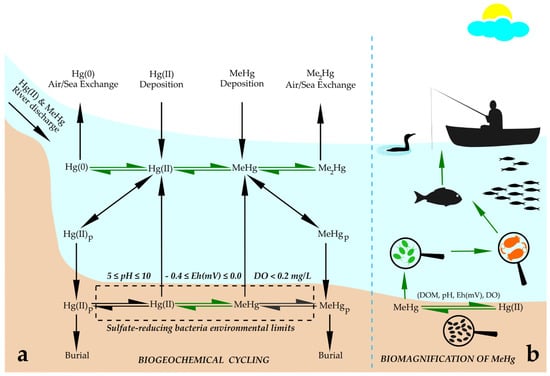
Figure 1.
Schematic representation of (a) basic processes in biogeochemical cycling of Hg in coastal areas. Abbreviations: Hg(0), elemental mercury; Hg(II), divalent mercury; MeHg, methylmercury; Me2Hg, dimethylmercury; MeHgP, methylmercury bound to particulate organic matter; Hg(II)P, divalent mercury bound to particulate organic matter. Green arrows indicate biologically mediated processes; environmental conditions for sulfate-reducing bacteria, where Hg methylation occurs, are: 5 ≤ pH ≤ 10; −0.4 ≤ Eh(mV) ≤ 0.0; DO < 0.2 mg/L. (b) Biomagnification of MeHg through the aquatic food web: MeHg enters the food web at the very bottom via bacterial methylation activity, then it is partially taken up by microalgae; from that point on, it biomagnifies through rotifers that graze on microalgae, and further via smaller fish that prey on rotifers, then predator fish that prey on smaller fish, and finally transfers to aquatic birds and humans that consume contaminated fish. Abbreviations: Hg(II), divalent mercury; MeHg, methylmercury; DOM, dissolved organic matter; Eh(mV), redox potential; DO, dissolved oxygen. Adapted from [6,32,37,38,62].
Microalgae play a direct seasonal role in Hg chemical species circulation in aquatic environments [11]. As already explained above, MeHg enters the food web via microalgae and further bioaccumulates in aquatic organisms, which can finally result in biomagnification to concentrations with adverse effects on reproduction and neural development in fish and mammals [47]. The biggest MeHg increase in water and microalgae biomass in the Baltic Sea was found in summer and fall, with 60–70% increase in the intracellular MeHg content of algae biomass compared to a yearly average [6]. It was suggested that this occurs due to the remineralization of dead algae biomass from the spring and summer blooms, which results in higher direct methylation and uptake by microalgae [6]. It was found that the maximal impact of eutrophication on the intracellular MeHg content coincides with the highest zooplankton grazing on algae biomass [6]. Extreme MeHg levels in natural water bodies were reported for the Olt River (Romania) and its three reservoirs, which are heavily contaminated with Hg from a nearby chlor-alkali plant [48]. Total Hg concentrations in the Olt River were up to 88 ng/L but decreased by 10 times inside the reservoir (to 8 ng/L) [48]. MeHg concentrations increase from the entrance to the reservoir to the central part to 0.7 ng/L, confirming high methylation within the eutrophic reservoir [48]. Based on the vertical water column profiles of total Hg and MeHg, it was suggested that Hg methylation mainly occurs deep in the water column, close to the bottom, at the sediment–water interface. MeHg concentrations and bioconcentration factors of phytoplankton confirm that the highest biomagnification of MeHg takes place in the primary producers [48]. High MeHg concentrations (>15 ng/L) in the surface water of some artificial wetlands and its accumulation in saturated sediments can affect the invertebrate community to levels that are potentially hazardous for insectivorous biota such as birds [49].
Similar to freshwater systems, Hg(II) can be deposited to the ocean by dry and wet deposition, and elemental Hg(0) by dry deposition, where wet deposition is defined as removal from the atmosphere through precipitation, while dry deposition is considered to be Hg settling or uptake without precipitation [31,32]. The total Hg deposition (wet and dry) to oceans was estimated to be 10 Mmol/year [31]. A significant load of Hg is introduced into the sea by rivers, with a high amount of Hg contributed by river sediment [30]. The load of Hg introduced with suspended and trailing river debris leads to Hg enrichment of the marine coastal zone [30]. The Hg(II) deposited to the ocean can be reduced to Hg(0), adsorbed onto particles or methylated [32]. The general trend is that oxygen-depleted areas are spreading in coastal and offshore waters worldwide [12,50]. Seawater can be considered as normoxic, with a normal dissolved oxygen (DO) concentration of 5–8 mg/L; hypoxic (DO < 0.2 mg/L); or anoxic, with the presence of hydrogen sulfide and a DO < 0.1 mg/L [6,51]. One of the main processes responsible for a DO decrease is O2 consumption by bacteria during the decomposition and mineralization of OM [51]. Excessive eutrophication negatively affects the oxygenation of seawater as the enrichment of water with nutrients such as N, P and DOC in the surface layer results in increased phytoplankton growth and biomass production, which causes algae blooms and reduces DO and water transparency [51]. Total Hg content in marine ecosystems is in the picomolar (pM) range and depends on the position, size and exposure to anthropogenic activity of the aquatic ecosystem (Table 1). MeHg accounts for 3–35% of total Hg levels in the oceans, and its concentration in water is usually in femtomolar range (fM) [11,32,47].

Table 1.
Concentrations of total Hg and MeHg in different aquatic ecosystems according to the literature. Data are presented in the original units (ng/L, pM and fM) that were used by the respective authors.
As explained above, methylation of Hg mainly occurs in hypoxic and anoxic water conditions [32]. Nevertheless, new evidence has led to the hypothesis that MeHg can also be formed in oxic brackish coastal waters. It was demonstrated that the concentration of MeHg in experimental microcosms containing oxic surface waters from the pelagic zone of the Bothnian Sea significantly increased within 27 h after the addition of 250 pM Hg(II) and under normal DOC concentration (4.0 mg/L), where bacterial activity played a central role in MeHg production [20]. It was also found that enhanced MeHg production was not reflected by substantial changes in the bacterial community composition or gene pool [20].
It was found that the contribution of MeHg in total Hg in Canadian lakes is strongly and positively correlated with turbidity and total dissolved N and slightly, more weakly correlated with chlorophyll–a and DOC, which suggests that Hg methylation rates are higher in lakes with greater productivity and algae biomass, where increased oxygen use by decomposers in sediments receiving high deposition rates of labile OM and related anoxia in sediments would be expected to drive Hg methylation rates [39]. Although the basic parameters driving the biogeochemical cycling of Hg are known, in-lake influences on these parameters and Hg cycling are complex and highly variable and can mask climate- or industry-related changes over time [39].
4. MeHg Implementation into the Aquatic Food Web and Its Consequences to Living Organisms
Naturally occurring phytoplankton (which includes the microalgae community) take up part of the dissolved MeHg, and this is the entrance point for this toxin into the food web. Its concentration is subsequently biomagnified (Figure 1b) as zooplankton and other predators, such as fish larvae and fish, feed on phytoplankton contaminated with MeHg and concentrate it in their tissues [32]. The process of bioaccumulation can increase MeHg concentrations in predatory fish by six orders of magnitude [32,37,62]. The transfer of intracellular MeHg from microalgae up the food web depends on the seasonal variability of their MeHg content, the quality of the algae community as a food source and the timespan of the peak zooplankton grazing on microalgae [6]. MeHg concentrations generally increase with trophic level and with increased size and age of the animal [38]. It was reported that MeHg in fish is deposited in skeletal muscle, reducing exposure to the central nervous system [63]. Experiments with small mammals demonstrated that ingestion of 1 µg/g MeHg via diet resulted in visual, cognitive and neurobehavioral deficits and finally led to death in less than a year [38,63]. The reproductive toxicity of MeHg has been confirmed in animal studies [64]. MeHg concentrations in fish increase with trophic level, size and age, as it is the result of bioaccumulation during the entire lifespan of the animal [65]. The European Commission (EC Regulation 1881/2006) limits the total Hg level allowed in seafood to 0.5–1.0 mg/kg, while no restriction exists for MeHg. Analysis of MeHg requires specific analytical equipment, making its detection difficult. For that reason, guidelines concerning the protection of aquatic life regarding % MeHg of total Hg in natural waters are missing [39,66]. The U.S. Environmental Protection Agency (USEPA) limits the Hg content in the tissues of freshwater and estuarine fish at 0.0175 mg/kg [38,67]. These regulations are based on the assumption that human populations traditionally consuming seafood are at an increased risk of MeHg exposure and bioaccumulation through their diet [65].
The foremost toxicological concern about Hg biogeochemical cycling is its methylation in aquatic environments, as the predominant Hg form in consumable fish tissues and vertebrate systems is MeHg [37]. This is the only form of Hg that bioaccumulates in humans and animals, and the only form of Hg that crosses the blood–brain barrier and further accumulates in human neurological tissues [37,68]. Hg intoxication in humans is mainly caused by a low-level, chronic exposure from dietary consumption of fish and seafood that contain MeHg (due to its bioaccumulation in the aquatic food web), although it can also be caused by an acute exposure during an industrial accident or accidental ingestion [64,69,70]. After ingestion, MeHg is absorbed in the gastrointestinal tract, distributed throughout the body, and after crossing the blood–brain barrier, causes damage in the central nervous system. The neurodevelopment of children is especially affected by Hg toxicity [68]. In adults, the central nervous system and cardiovascular system are the most affected, with symptoms throughout the body, including in the liver, kidneys, skin and lungs. Adverse neurological disorders in childhood due to prenatal exposure to MeHg have been confirmed by epidemiological studies showing the relationship between reduced cognitive functioning, including reduced IQ in newborn children and the presence of Hg in maternal bodies [71]. Chronic exposure to MeHg: (i) affects the nervous system, producing alterations in vision and hearing, paresthesia, ataxia and brain function and developmental delay; (ii) causes alterations in the cardiovascular system; and (iii) increases blood pressure and heart rate, with a higher risk of heart attack [72].
MeHg enhances the formation of free radicals, and the antioxidant effect of reduced glutathione (GSH) and catalase is altered due to its high affinity with the thiol group [73]. Free radicals mediate lipid peroxidation and platelet aggregation, and blood coagulation is then promoted, causing sclerosis of the arteries and an increase in blood pressure [64]. Symptoms of human acute poisoning by ingestion of a sublethal Hg(II) dose (2.0–4.0 g of HgCl2) include: nausea, abdominal pain, vomiting and diarrhea, followed by mild hemodynamic instability, acidosis, hypokalemia, leukocytosis and fever [70]. Blood Hg concentration was observed to be 17.9 μM after 3 h, but it decreased to 8.8 μM after 72 h after ingestion, and the patient’s symptoms began to improve 48 h after admission and resolved fully within a week [70]. Clinical treatment included: (i) chelator dimercaprol at 200 mg intramuscularly: first six, then four and finally two times a day; (ii) potassium replacement; and (iii) broad-spectrum antibiotic to treat respiration difficulties and fever [70]. MeHg is excreted primarily via feces and urine and can also be excreted through breastmilk during lactation, but there is no antidote to its exposure [74]. Cessation of exposure and chelation therapy are the only treatments available.
5. Biochemical Response of Microalgae to Heavy Metals
Microalgae require metal ion species that are essential to sustain cell metabolism through a variety of biological functions, including their roles as redox-active metals and/or as cofactors in enzymes. It has been widely reported that essential metal excess or the presence of nonessential metals in microalgae cultures induce biochemical responses for which these microorganisms are genetically adapted. These response mechanisms, which are addressed below in this section, allow microalgae cells to minimize the toxic effects of metals, thus remaining biologically active for longer time periods. The microalgae cells pay a cost in return to the expression of response mechanisms, mostly in terms of metabolic energy expenses that are used to overcome the stress imposed and, therefore, are not used for growth.
Many metal species, including Hg species, induce or boost oxidative stress in microalgae, which is mediated by radical oxygen species [75]. From the chemical point of view, the redox-active character is not a requirement for a given metal species to induce oxidative stress. Besides the known redox-active character of Fe(II) and Cu(II), which are involved in the redox process of oxidative stress induction, other metal ions that do not display redox activity in cells have been reported to induce oxidative stress. For instance, Zn has been reported to induce oxidative stress in microalgae cells [76]. Pb and Ni have been found to induce oxidative stress causing membrane lipid peroxidation although Ni does not seem to react directly with ROS [77,78]. Exposure of microalgae cells to Cd(II) in liquid cultures also results in membrane lipid peroxidation and has also been reported to decrease the content of reduced glutathione in cells, probably because of the interaction with Cd(II) [79]. Hg is also involved in the induction of oxidative stress, as specifically addressed below in this section. All the above-mentioned metal ions, not being directly involved in redox cellular processes but originating oxidative activity in algal cells, are examples of the direct relation that exists between metal internalization into cells and increased oxidative cell status. In this sense, the involvement of ROS as mediators for inducing the associated antioxidant response has been reported [75].
Direct involvement in oxidative stress induction through oxidative reactions with biomolecules has been described for Fe(II) and Mn(II) [76]. These are representative examples of the direct implication of metal ion species in the induction of oxidative stress in microalgae. As a paradox, the greater the microalgae antioxidant resistance to metals, the longer the metal transformation and presence lasts in aquatic environments, which applies to Hg uptake and accumulation by microalgae in nature. Nevertheless, knowledge about the specific biochemical resistance mechanisms of microalgae to metals can also be taken as useful information to develop microalgae-based metal-capturing and removal processes.
The antioxidant response of microalgae consists of a group of biochemical mechanisms that provide the microalgal cells with tolerance or resistance to the chemical effects of oxidative stress. The toxicity produced by metals in microalgae cells depends on the action mechanisms of the metal and, obviously, on its concentration in relation to the biomass concentration. According to [75], the metal ion’s toxicity depends on: (1) their affinity for thiol groups (for instance, those present in GSH and phytochelatins); (2) competition with essential metal cations which can be replaced in enzyme active sites; and (3) induction of oxidative stress, which results in the microalgae cells displaying biochemical responses, some of which are briefly described below. Interestingly, an increased oxidative stress scenario does not always imply an increased, measurable antioxidant response [75]. Namely, under severe oxidative stress, a decrease in the cell content of antioxidant molecules can be detected, which is due to their oxidative degradation; the biosynthesis of such antioxidants (for instance, GSH, tocopherol, carotenoids) can be greater under intense stress; however, the balance between their oxidative degradation and biosynthesis increases due to the implication of these molecules in the antioxidant response chemical reactions of the cell [75]. This is a key issue when interpreting and discussing results derived from the effects of oxidative stress on the antioxidant biochemical profile of microalgae.
The following antioxidant responses can be expressed in microalgae cultures exposed to metal species, thus being useful to select biomarkers of oxidative stress in experiments with microalgae cultures exposed to metals: increased synthesis of polyphenols, carotenoids and organic acids that can bind metal ions; expression of phytochelatins that bind metal ions, particularly Cu(II), Cd(II) and Zn(II), and store them in vacuoles; cation binding to negatively charged residues of the cell wall, including sulfated polysaccharides; chemical linkage of metal ions to polyphosphate inclusions; metal ion sequestration by chelating compounds, which includes GSH, short-chain peptides such as Arg–Arg–Glu and polyphenols; and limited uptake or metal exclusion from cells [80,81,82,83].
In particular, Hg(II) added at 104.2 mg/L to Chlorella vulgaris culture was found to induce oxidative stress through significant increases in intracellular ROS and superoxide dismutase (SOD) activity [84]. However, no physiological roles have been described for Hg [85]. This is probably related to its extreme affinity for inorganic phosphate, by which Hg can be expected to interfere with biochemical processes that are key to life, for instance, metabolic energy production in the form of ATP and all those metabolic processes involving nucleotides [75,86]. Hg(II) inhibits photosynthesis. It was suggested that Hg-mediated photosystem II (PSII) inhibition may occur at the electron donor side. The donor side of PSII harbors the catalytic site of water cleavage, which is an inorganic Mn cluster. Hg interference with the water cleavage catalytic site should result in decreased growth and reduced chlorophyll content in the photosystems, among other well-described negative effects, resulting in increased oxidative cell status [87]. Microalgae cells express antioxidant response mechanisms to face oxidative stress produced by the presence of Hg. In fact, Hg-mediated induction of antioxidant response mechanisms has been reported, and these mechanisms are in line with common antioxidant mechanisms of algae in the presence of other metal ions. It was suggested that the relationship between Hg-dependent ROS generation and increased antioxidant enzymes activity levels—determined for superoxide dismutase (SOD) and catalase (CAT)—has been recently proven in C. vulgaris, a study model microalga [84]. Interestingly, besides showing direct evidence for Hg involvement in ROS generation, the levels of SOD and CAT vary differently with Hg concentration, suggesting a possible differential Hg-dependent expression or activation pattern for both enzymes. In fact, SOD shows increased activity levels at low Hg concentrations, conversely to CAT, which shows the highest activity levels when Chlorella cells are exposed to high Hg levels under high ROS intracellular levels. In addition to SOD and CAT, the other enzymes of the antioxidant biochemical machinery, including glutathione reductase and peroxidase, are also activated and/or expressed in the presence of Hg, evidencing its role in the induction of oxidative stress [88]. The former is in coherence with the findings of [85], who described the Hg-mediated induction of glutathione biosynthesis and phytochelatins in plants, in good agreement with earlier evidence found for direct chemical binding between Hg and phytochelatins in Chlorella and Isochrysis strains [89]. The above-described biochemical responses of microalgae antioxidant metabolism to Hg prove a role for Hg in three key features of the Hg-dependent oxidative stress and antioxidant machinery response in microalgae: (i) the induction of ROS generation; (ii) the induction of the antioxidant response based on the expression or activation of enzyme components of the antioxidant machinery; and (iii) direct interaction with nonenzymatic molecules, such as glutathione and phytochelatins.
6. MeHg Interaction with Microalgae in Aquatic Environments on Molecular and Physiological Levels
Microalgae are unicellular autotrophic microorganisms that use photosynthesis as the main mechanism for producing energy, basic cell constituents and organic compounds, as well as for cell division. Photosynthesis in green algae is a light-driven process that passes reductive equivalents (electrons) from water to CO2 through the chain of oxidoreduction reactions resulting in O2 evolution, ATP production and, finally, CO2 fixation to monosaccharide molecules in its dark phase [90]. As the reaction evolves against the electrochemical gradient, from more positive Eh(O2) = +0.82 V to more negative Eh(CO2) = −0.42 V, it needs light energy to proceed. It was estimated that global annual photosynthetic O2 production is 3.57·1011 tons, of which phytoplankton in oceans produces 1.96·1011 tons, which is 22% more O2 produced than that by terrestrial plants occupying land surfaces [91]. Microalgae along with photosynthetic cyanobacteria form phytoplankton in Earth’s aquatic ecosystem and are responsible for the greater part of global oxygen production. Light is not only essential for the process of photosynthesis in algae; it also photodegrades MeHg. Twenty-four hours of light exposure in algae cultures led to the removal of one-third of added MeHg from the solution due to abiotic loss not related to algae, which includes photodegradation [22].
Negatively charged functional groups in the cell wall constituents can capture and remove metal ions from the growth medium via counter-ion interactions, making the cell wall the main participator in heavy metal removal [92]. Negatively charged functional groups that act as ligands in metal binding are: carboxyl, hydroxyl, sulphate, thiol, phosphate, amino, amide, imine, thioether, phenol, carbonyl, imidazole, phosphonate and phosphodiester [92,93]. The uptake of metal cations from water involves diffusion from the culture medium to the algae cell surface, followed by passive or active transport to the cytoplasm. Passive transport is a simple diffusion through the cell wall, while active transport involves binding to the reactive center at the cell wall and active transport into the cell that requires energy from ATP [2,94,95]. MeHg uptake in algae includes active transport, which was demonstrated by addition of phosphorylation-inhibiting chemicals that suppress ATP synthesis and decrease uptake of MeHg in Selenastrum capricornutum cultures [5]. Similarly, dead diatom cells take up 10 times less MeHg than healthy cultures, indicating a biochemically active Hg transport process [4]. Microalgae cell walls are composed of cellulose-like carbohydrates, interconnected with proteins and lipids, which results in a negatively charged cell surface [92,96,97,98]. The cellular uptake rate and accumulation of MeHg by phytoplankton or bacteria is limited by MeHg bioavailability, which is in turn dictated by its chemical speciation [16,17]. In microalgae, inorganic Hg(II) binds to cell membranes while MeHg is imported intracellularly and accumulates in the cytoplasm [7]. Levels of MeHg in microalgae are strongly associated with water MeHg concentrations [11]. Complexation of MeHg with EDTA in artificial culture medium can also limit the bioavailability of MeHg to algae, which must be avoided by changing the recipe for the culture media that will be used, as it readily contains EDTA sodium salts [8,10]. It was reported that at least 70–78% of MeHg added to algae culture is further associated with the cell walls of the cell [10,22], which underlines the importance of algae cell walls in Hg removal. It was suggested that the internalization of MeHg in algae cells is mainly associated with active transport involving transporters in the membrane that are saturable, and so MeHg internalization may fluctuate during exposure [5,22].
MeHg uptake by microalgae affects several physiological processes within the cell population, including photosynthesis and growth rate [2,99]. Various exposure experiments confirmed that MeHg primarily affects algae cultures via elevated toxicity and growth inhibition [2,99]. To diminish these negative effects, algae have developed several detoxification strategies, such as the reduction of MeHg to elemental Hg or its complexation with intracellular ligands [2]. Culture temperature and population size also affect MeHg uptake and its toxicity in microalgae. There is a linear relationship between temperature and MeHg uptake rate, while increased temperature has a positive impact on enzyme-like transporters of MeHg involved in active transport [5]. The population density of microalgae has a direct impact on MeHg accumulation in grazers and subsequent consumers [2]. The transfer of MeHg from microalgae to zooplankton decreases at higher population densities due to the biodilution. An enhanced algae growth rate results in a lower concentration of MeHg per single cell and, subsequently, a lower rate of transfer to grazers [2]. High variability of Hg uptake in algae is attributed to the complexity of the process resulting from the balance between influx and efflux rates [10,22,100].
The green microalga Chlamydomonas reinhardtii exposed to 5–50 nM of Hg(II) and MeHg accumulated metabolites involved in amino acid and nucleotide synthesis and degradation, fatty acid, carbohydrate, tricarboxylic acid (TCA) and antioxidant metabolism, and photorespiration [101]. Hg(II) toxicity induced more perturbations in fatty acid and TCA metabolism than exposure to MeHg [101]. This physiological and transcriptomic study further revealed its effect on the biosynthesis of various amino acids, glutathione metabolism and isoquinoline–alkaloid biosynthesis [101]. The addition of Hg(II) significantly affected α–linolenic acid metabolism, which was not the case for MeHg addition [101]. It was suggested that the accumulation of amino acids facilitates chelation of Hg(II) and MeHg cations inside the cells [101]. Glutamine is part of the glutamine–glutamate cycle responsible for ammonia assimilation, while enzyme glutamine synthetase plays an important role in nitrate assimilation in C. reinhardtii, which suggests that one response to Hg exposure is accelerated assimilation of nitrogen from the medium [101]. The addition of 5–50 nM of Hg(II) and MeHg triggered an antioxidative response in C. reinhardtii by increasing the GSH concentration, which is central to redox control in the cell [101]. GSH has various modes of action as the response to Hg exposure: it acts as protection against the ROS; it acts as an important metal chelator in plant cells and contributes to Hg detoxification; it acts as the precursor for the synthesis of phytochelatins, which are important intracellular heavy metal chelators [2,101,102]. He-binding phytochelatins have been found in C. sorokiniana, which suggests that a broader cation spectrum can be potentially bound by microalgae phytochelatins and, in addition, confirms the involvement of thiol groups in Hg internalization within microalgae cells [103].
A natural microalgae consortium isolated from Lake Titicaca (Bolivia), comprised of Oedogonium spp., Chlorella spp. and Scenedesmus spp., was exposed to a range of MeHg concentrations, from 5–200 ng/L, which resulted in the maximum MeHg removal (85%) with an uptake capacity of 2863 ng/gDW for an initial MeHg concentration of 200 ng/L applied to 0.02 g/L of algae culture [22]. MeHg uptake experiments indicated a bimodal kinetics with two phases: a rapid increase in MeHg uptake in the first 1.5 h of exposure followed by an uptake plateau until the end of the experiment [22]. It was also demonstrated that the degree of MeHg association to the algae cell surface is higher when cells are exposed to MeHg complexes with lower thermodynamic stability (MeHgOH) compared to more stable MeHg complexes with thiols [10]. The association rate and extent are higher for MeHg complexes with small thiol ligands with a relatively simple chemical structure compared to complexes with thiols of larger size and/or a more branched chemical structure [10].
From Table 2, it can be concluded that the most frequently used microalgae species in MeHg experiments are green algae: C. reinhardtii, S. capricornutum and Chlorella sp. Exposure concentrations of MeHg vary from 10 pM ([104]) to 50 nM ([101]), while the exposure period varies from 0.083 to 72 h. The total uptake increases with time and exposure concentration, although most MeHg is taken up at the fastest rate during the first few hours of exposure [10,22,99]. Similar uptake kinetics were reported in aquatic bacterial communities, where freshly deposited Hg (in the presence of which DOM facilitates the cell uptake of Hg by altering the bacterial membrane properties) is taken up by bacteria within a few hours (~24 h) after entering the aquatic system, after which the bioavailability of Hg for bacterial uptake is reduced [20]. Toxicity of MeHg on the growth of marine diatoms T. pseudonana and T. weissflogii was reported at concentrations of 0.57 nM and 0.10 nM, respectively [19,28]. On the other hand, a toxic effect on green algae C. reinhardtii was confirmed at the metabolic level with much higher MeHg concentrations (5–50 nM MeHg) [101], suggesting more resistance to Hg toxicity in green algae. C. reinhardtii exposed to 10 pM–10 nM of MeHg or Hg(II) was very resistant to Hg toxicity, and despite the strong gene response for both treatments, the respective physiological responses were low, and only one treatment (100 pM MeHg) resulted in a significant increase in cellular ROS and oxidative stress [105]. T. pseudonana, C. autotrophica and I. galbana growth rates decreased significantly at higher MeHg concentrations (3–9 nM), in the following order: T. pseudonana > I. galbana > C. autotrophica [99]. A naturally occurring algal consortium from Lake Titicaca comprised of three species, Oedogonium spp., Chlorella spp. and Scenedesmus spp., with relative abundances of 63%, 18% and 19%, respectively, exposed to 0.997 nM of MeHg had no reported toxic effect on its composition and abundances [22]. The cellular uptake rate also depends on the phytoplankton species used in the study. This effect can be attributed to the substantial differences in the cell wall structure of green algae and diatoms [10]. Moreover, variations in MeHg uptake by marine diatoms in different studies can also be explained by factors not directly related to cellular uptake or experimental conditions, such as MeHg concentration or exposure time. Namely, it was observed that MeHg uptake in T. pseudonana decreased in the presence of 100 µM DOC, which was not added to the culture medium but originated from the decomposing dead diatom cells [106].

Table 2.
Experimental concentrations of MeHg and its internalization by various microalgae species according to the literature.
Scientific information on the effect of MeHg at the molecular level is still scarce. Nevertheless, efforts are being made by the scientific community in the field that will allow us to understand what genes are mainly involved in the microalgae response when exposed to MeHg. To identify relevant genes, transcriptomics (RNA Sequencing; RNA-Seq) is a highly powerful tool that allows the early detection of adverse outcome pathways as well as tolerance response [105]. As mentioned, MeHg mostly binds to the cell wall in microalgae cells, showing a high affinity for thiol groups. Hg uptake and internalization through its interaction with a variety of metabolites and metabolic pathways address global changes in gene expression. Coming back to C. reinhardtii, a well-known microalgae study model, it has been demonstrated in recent years that roughly 47% of the total C. reinhardtii genes become dysregulated under treatment with Hg, the number of downregulated genes in treatment with MeHg being larger than upregulated genes, though this is MeHg concentration-dependent [105]. The impact of MeHg affects gene expression of a wide range of metabolic pathways with rather different functionalities. At least, the following pathways have been described as being altered in their expression at a transcriptional level: the regulation of gene expression in nucleotide-to-protein synthesis; cell processes such as motility, cell division and development; energy metabolism genes, including photosynthesis and carbohydrates; and genes involved in lipid and amino acid metabolism, responses to stress and transport functions. Among the many genes dysregulated by the presence of MeHg, some that are involved in key cell functions can be mentioned. For instance, LCIB, a gene involved in inorganic carbon accumulation in the chloroplast, is upregulated [108], and a number of genes in TCA cycle reactions are also dysregulated. These results support that energy metabolism becomes affected by MeHg. Moreover, the expression of genes involved in maintaining redox homeostasis are affected, as expected, by toxicity from MeHg; for instance, genes coding thioredoxin and glutathione reductase are upregulated by MeHg [105]. An approach to the molecular level is crucial for an accurate and integrated evaluation of MeHg and other toxic compounds in microalgae, helping us to understand further impacts and consequences of MeHg toxicity in nature, and also for designing biotechnological approaches to use microalgae in MeHg detoxification processes.
7. Future Prospective and Challenges
Anthropogenic Hg emitted in the past represents approx. one-third of present-day Hg in global cycling [32], which underlines the importance of managing Hg already circulating globally. It was already suggested that a reduction in Hg(II) will have local and regional benefits in communities that consume local fish and seafood, while a reduction in elemental Hg will have global benefits via reductions in wet and dry deposition and decreasing Hg global cycling [32]. Scientific knowledge of complex Hg interactions with naturally occurring bacteria and phytoplankton in the aquatic food web of global water ecosystems still remains uncertain. One of the critical challenges for society is to reduce Hg exposure to wildlife and humans through diet. MeHg production in aquatic environments is a complex process depending on a range of factors: DO level, OM concentration, redox potential, bacterial activity, terrestrial runoffs and wet and dry Hg depositions. Natural microalgae communities are the MeHg entrance point in the food web, which emphasizes the importance of future research on the MeHg interaction with microalgae.
Recommended areas for future research that could contribute to efficient MeHg control in aquatic wildlife include:
- (1)
- Physiological effects of realistic levels of MeHg on natural algal consortiums in contaminated areas;
- (2)
- Predictions of whether OM will have an overall positive or negative impact on future MeHg concentrations [11];
- (3)
- Effects of S and Se compounds on Hg methylation rates under various OM conditions;
- (4)
- Relationship between realistic levels of MeHg and composition shifts in natural microalgae consortiums, as well as seasonal changes;
- (5)
- MeHg and Hg(II) internalization capacities in the Hg-resistant strains with possible applications in Hg-contaminated water remediation.
Modern experimental procedures, such as shotgun metagenomics and targeted metabolomics, give us the possibility to assess MeHg effects in algae on molecular and genetic levels. Shotgun metagenomics offers a wide perspective of the taxonomic composition of microorganisms involved in Hg cycling, which can give better insight into the metabolic response of bacterial communities and phytoplankton under increased levels of MeHg and DOM [20]. Targeted metabolomics provides information on the metabolic changes in microalgae exposed to MeHg and can improve biological understanding of the molecular basis of Hg’s effect on various metabolic pathways [101].
8. Conclusions
Hg is a global pollutant naturally present in the environment that causes harmful effects in humans and wildlife. The more toxic organic form, MeHg, has the ability to bioaccumulate and biomagnify in the aquatic food web; therefore, it is important to improve our understanding of these processes considering its harmful effects in humans and wildlife. MeHg is the only Hg form that accumulates in living organisms and is able to cross the blood–brain barrier and presents an enormous health risk. The bioavailability of Hg(II) in the aquatic environment limits the formation of MeHg, governed by concentrations of inorganic sulfide, pH, redox conditions and characteristics of OM.
Exposure of laboratory cultures of green microalgae to nanomolar concentrations of Hg(II) and MeHg resulted in alterations in amino acid and nucleotide metabolism, photorespiration, the tricarboxylic acid (TCA) cycle, metabolism of fatty acids and carbohydrates, and antioxidant formation. Some naturally occurring algae consortiums are able to grow in MeHg concentrations three orders of magnitude higher than in their natural habitats and accumulate MeHg in high doses, which makes them excellent candidates for future applications in treatment of Hg-contaminated wastewater. The exact mechanism of MeHg uptake in green algae, as well as factors controlling the process in aquatic ecosystems, remains to be identified in future research.
At the molecular level, some efforts have been made to identify genes dysregulated by MeHg in microalgae; this is a first step to understand the basis of physiological alterations and how this information can be used to assess the impact of MeHg in nature on a global scale. Nevertheless, this scientific knowledge remains to be unveiled and further used for the benefit of understanding changes on a global scale produced by MeHg.
Although there are important findings on MeHg uptake and effect in algae, a detailed understanding of its metabolism in cells and its further transfer up the food web is partially missing. Understanding how different factors affect MeHg uptake in microalgae is crucial to control MeHg transfer up the aquatic food web and the eventual exposure risk for humans. In order to control MeHg exposure in aquatic wildlife, it is critical to understand the uptake of MeHg by microalgae and to identify the natural defense strategies of algae against MeHg uptake. Knowledge of the microalgae–MeHg interaction at the bottom of the food web provides a key insight into the control and prevention of MeHg exposure and poisoning in humans and wildlife. This review aims to summarize recent findings in the literature that can provide more a detailed understanding of the uptake, distribution, toxicity and complexation of MeHg in cultures of green microalgae. This information can be used to better predict MeHg transfer and toxicity in the aquatic food web and may also be valuable to define MeHg control and removal strategies. It also may help to give insight into this complex process and open the path for further research on the prevention of human exposure to MeHg contamination.
Author Contributions
Conceptualization, Z.G. and C.V.; writing—original draft preparation, Z.G.; writing—review and editing, A.S., I.G., C.V., C.F. and Z.G.; supervision, C.V.; funding acquisition, C.V. and I.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Spanish Ministry of Economic Transformation, Industry, Knowledge and Universities; by the European Regional Development Fund (FEDER) within the framework of the FEDER program of Andalusia (Spain) 2014–2020, grant number UHU–202065; and by Grant P20-00930 from the Andalusian Plan for Research, Development and Innovation, within the frame of the operational program “FEDER Andalucía 2014–2020”.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Acknowledgments
The authors wish to thank Erik Björn from Department of Chemistry, Umeå University, Sweden, for his constructive comments on the paper’s content. We wish to thank personnel from LICAH (Laboratorio de Investigación y Control Agroalimentario), University of Huelva, for their collaboration and cooperation under FEDER 2014–2020 UHU–202065 project. We also want to thank colleagues from BITAL (Algae Biotechnology Group), University of Huelva, for their kind assistance in the lab and for creating a productive working environment.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Swain, E.B.; Jakus, P.M.; Rice, G.; Lupi, F.; Maxson, P.A.; Pacyna, J.M.; Penn, A.; Spiegel, S.J.; Veiga, M.M. Socioeconomic Consequences of Mercury Use and Pollution. Ambio 2007, 36, 45–61. [Google Scholar] [CrossRef]
- Le Faucheur, S.; Campbell, P.G.C.; Fortin, C.; Slaveykova, V.I. Interactions between Mercury and Phytoplankton: Speciation, Bioavailability, and Internal Handling. Environ. Toxicol. Chem. 2014, 33, 1211–1224. [Google Scholar] [CrossRef] [PubMed]
- Mergler, D.; Anderson, H.A.; Chan, L.H.M.; Mahaffey, K.R.; Murray, M.; Sakamoto, M.; Stern, A.H. Methylmercury Exposure and Health Effects in Humans: A Worldwide Concern. Ambio 2007, 36, 3–11. [Google Scholar] [CrossRef]
- Pickhardt, P.C.; Fisher, N.S. Accumulation of Inorganic and Methylmercury by Freshwater Phytoplankton in Two Contrasting Water Bodies. Environ. Sci. Technol. 2007, 41, 125–131. [Google Scholar] [CrossRef]
- Moye, H.A.; Miles, C.J.; Phlips, E.J.; Sargent, B.; Merritt, K.K. Kinetics and Uptake Mechanisms for Monomethylmercury between Freshwater Algae and Water. Environ. Sci. Technol. 2002, 36, 3550–3555. [Google Scholar] [CrossRef] [PubMed]
- Soerensen, A.L.; Schartup, A.T.; Gustafsson, E.; Gustafsson, B.G.; Undeman, E.; Björn, E. Eutrophication Increases Phytoplankton Methylmercury Concentrations in a Coastal Sea—A Baltic Sea Case Study. Environ. Sci. Technol. 2016, 50, 11787–11796. [Google Scholar] [CrossRef]
- Gorski, P.R.; Armstrong, D.E.; Hurley, J.P.; Krabbenhoft, D.P. Influence of Natural Dissolved Organic Carbon on the Bioavailability of Mercury to a Freshwater Alga. Environ. Pollut. 2008, 154, 116–123. [Google Scholar] [CrossRef]
- Gorski, P.R.; Armstrong, D.E.; Hurley, J.P.; Shafer, M.M. Speciation of Aqueous Methylmercury Influences Uptake by a Freshwater Alga (Selenastrum capricornutum). Environ. Toxicol. Chem. 2006, 25, 534–540. [Google Scholar] [CrossRef]
- Bravo, A.G.; Faucheur, S.L.; Monperrus, M.; Amouroux, D.; Slaveykova, V.I. Species-Specific Isotope Tracers to Study the Accumulation and Biotransformation of Mixtures of Inorganic and Methyl Mercury by the Microalga Chlamydomonas reinhardtii. Environ. Pollut. 2014, 192, 212–215. [Google Scholar] [CrossRef]
- Skrobonja, A.; Gojkovic, Z.; Soerensen, A.L.; Westlund, P.-O.; Funk, C.; Björn, E. Uptake Kinetics of Methylmercury in a Freshwater Alga Exposed to Methylmercury Complexes with Environmentally Relevant Thiols. Environ. Sci. Technol. 2019, 53, 13757–13766. [Google Scholar] [CrossRef]
- Soerensen, A.L.; Schartup, A.T.; Skrobonja, A.; Björn, E. Organic Matter Drives High Interannual Variability in Methylmercury Concentrations in a Subarctic Coastal Sea. Environ. Pollut. 2017, 229, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Soerensen, A.L.; Schartup, A.T.; Skrobonja, A.; Bouchet, S.; Amouroux, D.; Liem-Nguyen, V.; Björn, E. Deciphering the Role of Water Column Redoxclines on Methylmercury Cycling Using Speciation Modeling and Observations from the Baltic Sea. Glob. Biogeochem. Cycles 2018, 32, 1498–1513. [Google Scholar] [CrossRef]
- Deutsch, B.; Alling, V.; Humborg, C.; Korth, F.; Mörth, C.M. Tracing Inputs of Terrestrial High Molecular Weight Dissolved Organic Matter within the Baltic Sea Ecosystem. Biogeosciences 2012, 9, 4465–4475. [Google Scholar] [CrossRef]
- Ripszam, M.; Paczkowska, J.; Figueira, J.; Veenaas, C.; Haglund, P. Dissolved Organic Carbon Quality and Sorption of Organic Pollutants in the Baltic Sea in Light of Future Climate Change. Environ. Sci. Technol. 2015, 49, 1445–1452. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.J.; Moye, H.A.; Phlips, E.J.; Sargent, B. Partitioning of Monomethylmercury between Freshwater Algae and Water. Environ. Sci. Technol. 2001, 35, 4277–4282. [Google Scholar] [CrossRef]
- Luengen, A.C.; Fisher, N.S.; Bergamaschi, B.A. Dissolved Organic Matter Reduces Algal Accumulation of Methylmercury. Environ. Toxicol. Chem. 2012, 31, 1712–1719. [Google Scholar] [CrossRef]
- Ndu, U.; Mason, R.P.; Zhang, H.; Lin, S.; Visscher, P.T. Effect of Inorganic and Organic Ligands on the Bioavailability of Methylmercury as Determined by Using a Mer-Lux Bioreporter. Appl. Environ. Microbiol. 2012, 78, 7276–7282. [Google Scholar] [CrossRef]
- Berthon, G. Critical Evaluation of the Stability Constants of Metal Complexes of Amino Acids with Polar Side Chains (Technical Report). Pure Appl. Chem. 1995, 67, 1117–1240. [Google Scholar] [CrossRef]
- Mason, R.P.; Reinfelder, J.R.; Morel, F.M.M. Uptake, Toxicity, and Trophic Transfer of Mercury in a Coastal Diatom. Environ. Sci. Technol. 1996, 30, 1835–1845. [Google Scholar] [CrossRef]
- Rodríguez, J.; Andersson, A.; Björn, E.; Timonen, S.; Brugel, S.; Skrobonja, A.; Rowe, O. Inputs of Terrestrial Dissolved Organic Matter Enhance Bacterial Production and Methylmercury Formation in Oxic Coastal Water. Front. Microbiol. 2022, 13, 809166. [Google Scholar] [CrossRef]
- Andersson, A.; Brugel, S.; Paczkowska, J.; Rowe, O.F.; Figueroa, D.; Kratzer, S.; Legrand, C. Influence of Allochthonous Dissolved Organic Matter on Pelagic Basal Production in a Northerly Estuary. Estuar. Coast. Shelf Sci. 2018, 204, 225–235. [Google Scholar] [CrossRef]
- Quiroga-Flores, R.; Guédron, S.; Achá, D. High Methylmercury Uptake by Green Algae in Lake Titicaca: Potential Implications for Remediation. Ecotoxicol. Environ. Saf. 2021, 207, 111256. [Google Scholar] [CrossRef] [PubMed]
- Achá, D.; Guédron, S.; Amouroux, D.; Point, D.; Lazzaro, X.; Fernandez, P.E.; Sarret, G. Algal Bloom Exacerbates Hydrogen Sulfide and Methylmercury Contamination in the Emblematic High-Altitude Lake Titicaca. Geosciences 2018, 8, 438. [Google Scholar] [CrossRef]
- Schartup, A.T.; Ndu, U.; Balcom, P.H.; Mason, R.P.; Sunderland, E.M. Contrasting Effects of Marine and Terrestrially Derived Dissolved Organic Matter on Mercury Speciation and Bioavailability in Seawater. Environ. Sci. Technol. 2015, 49, 5965–5972. [Google Scholar] [CrossRef] [PubMed]
- Gojkovic, Ž.; Garbayo, I.; Ariza, J.L.G.; Márová, I.; Vílchez, C. Selenium Bioaccumulation and Toxicity in Cultures of Green Microalgae. Algal Res. 2015, 7, 106–116. [Google Scholar] [CrossRef]
- Umysová, D.; Vítová, M.; Doušková, I.; Bišová, K.; Hlavová, M.; Čížková, M.; Machát, J.; Doucha, J.; Zachleder, V. Bioaccumulation and Toxicity of Selenium Compounds in the Green Alga Scenedesmus Quadricauda. BMC Plant Biol. 2009, 9, 58. [Google Scholar] [CrossRef] [PubMed]
- Gojkovic, Ž.; Vílchez, C.; Torronteras, R.; Vigara, J.; Gómez-Jacinto, V.; Janzer, N.; Garbayo, I. Effect of Selenate on Viability and Selenomethionine Accumulation of Chlorella sorokiniana Grown in Batch Culture. Sci. World J. 2014, 2014, 13. [Google Scholar] [CrossRef]
- Wang, W.-X.; Wong, R.S.K.; Wang, J.; Yen, Y. Influences of Different Selenium Species on the Uptake and Assimilation of Hg(II) and Methylmercury by Diatoms and Green Mussels. Aquat. Toxicol. 2004, 68, 39–50. [Google Scholar] [CrossRef]
- Scheuhammer, A.M.; Meyer, M.W.; Sandheinrich, M.B.; Murray, M.W. Effects of Environmental Methylmercury on the Health of Wild Birds, Mammals, and Fish. Ambio 2007, 36, 12–18. [Google Scholar] [CrossRef]
- Gębka, K.; Bełdowska, M. Variation in the Content of Different Forms of Mercury in River Catchments of the Southern Baltic Sea—Case Study. Pol. Hyperb. Res. 2020, 72, 63–72. [Google Scholar] [CrossRef]
- Gworek, B.; Bemowska-Kałabun, O.; Kijeńska, M.; Wrzosek-Jakubowska, J. Mercury in Marine and Oceanic Waters—A Review. Water Air Soil Pollut. 2016, 227, 371. [Google Scholar] [CrossRef] [PubMed]
- Selin, N.E. Global Biogeochemical Cycling of Mercury: A Review. Annu. Rev. Environ. Resour. 2009, 34, 43–63. [Google Scholar] [CrossRef]
- Malm, O.; Pfeiffer, W.C.; Souza, C.M.M.; Reuther, R. Mercury Pollution Due to Gold Mining in the Madeira River Basin, Brazil. AMBIO A J. Hum. Environ. 1990, 19, 11–15. [Google Scholar]
- Zhang, H.; Feng, X.; Larssen, T.; Qiu, G.; Vogt, R.D. In Inland China, Rice, Rather than Fish, Is the Major Pathway for Methylmercury Exposure. Environ. Health Perspect. 2010, 118, 1183–1188. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency, US EPA. Biennial National Listing of Fish Advisories Fact Sheet EPA-823-F-09-007 September 2009. Available online: https://www.epa.gov/sites/default/files/2019-05/documents/2008-biennial-nlfa.pdf (accessed on 13 August 2022).
- Jiang, G.-B.; Shi, J.-B.; Feng, X.-B. Mercury Pollution in China. An Overview of the Past and Current Sources of the Toxic Metal. Environ. Sci. Technol. 2006, 40, 3673–3678. [Google Scholar] [CrossRef] [PubMed]
- Krabbenhoft, D. Methylmercury Contamination of Aquatic Ecosystems: A Widespread Problem with Many Challenges for the Chemical Sciences; Norling, P., Wood-Black, F., Masciangioli, T., Eds.; Water and Sustainable Development: Opportunities for the Chemical Sciences—A Workshop Report to the Chemical Sciences Roundtable; National Academy of Sciences: Washington, DC, USA, 2004; ISBN 0-309-53173-X. [Google Scholar]
- Sams, C.E. Methylmercury Contamination: Impacts on Aquatic Systems and Terrestrial Species, and Insights for Abatement. In Advancing the Fundamental Sciences: Proceedings of the Forest Service National Earth Sciences Conference, San Diego, CA, USA, 18–22 October 2004; Furniss, M., Clifton, C., Ronnenberg, K., Eds.; U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station: San Diego, CA, USA, 2007. [Google Scholar]
- Emmerton, C.A.; Cooke, C.A.; Wentworth, G.R.; Graydon, J.A.; Ryjkov, A.; Dastoor, A. Total Mercury and Methylmercury in Lake Water of Canada’s Oil Sands Region. Environ. Sci. Technol. 2018, 52, 10946–10955. [Google Scholar] [CrossRef] [PubMed]
- Cinnirella, S.; Bruno, D.E.; Pirrone, N.; Horvat, M.; Živković, I.; Evers, D.C.; Johnson, S.; Sunderland, E.M. Mercury Concentrations in Biota in the Mediterranean Sea, a Compilation of 40 Years of Surveys. Sci. Data 2019, 6, 205. [Google Scholar] [CrossRef]
- Bravo, A.G.; Peura, S.; Buck, M.; Ahmed, O.; Mateos-Rivera, A.; Ortega, S.H.; Schaefer, J.K.; Bouchet, S.; Tolu, J.; Björn, E.; et al. Methanogens and Iron-Reducing Bacteria: The Overlooked Members of Mercury-Methylating Microbial Communities in Boreal Lakes. Appl. Environ. Microbiol. 2018, 84, e01774-18. [Google Scholar] [CrossRef]
- Compeau, G.C.; Bartha, R. Sulfate-Reducing Bacteria: Principal Methylators of Mercury in Anoxic Estuarine Sediment. Appl. Environ. Microbiol 1985, 50, 498–502. [Google Scholar] [CrossRef]
- Kerin, E.J.; Gilmour, C.C.; Roden, E.; Suzuki, M.T.; Coates, J.D.; Mason, R.P. Mercury Methylation by Dissimilatory Iron-Reducing Bacteria. Appl. Environ. Microbiol. 2006, 72, 7919–7921. [Google Scholar] [CrossRef]
- Gilmour, C.C.; Podar, M.; Bullock, A.L.; Graham, A.M.; Brown, S.D.; Somenahally, A.C.; Johs, A.; Hurt, R.A.; Bailey, K.L.; Elias, D.A. Mercury Methylation by Novel Microorganisms from New Environments. Environ. Sci. Technol. 2013, 47, 11810–11820. [Google Scholar] [CrossRef]
- Parks, J.M.; Johs, A.; Podar, M.; Bridou, R.; Hurt, R.A.J.; Smith, S.D.; Tomanicek, S.J.; Qian, Y.; Brown, S.D.; Brandt, C.C.; et al. The Genetic Basis for Bacterial Mercury Methylation. Science 2013, 339, 1332–1335. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.-Q.; Reinfelder, J.R.; Hines, M.E.; Barkay, T. Mercury Methylation by the Methanogen Methanospirillum hungatei. Appl. Environ. Microbiol. 2013, 79, 6325–6330. [Google Scholar] [CrossRef] [PubMed]
- Canário, J.; Vale, C.; Poissant, L.; Nogueira, M.; Pilote, M.; Branco, V. Mercury in Sediments and Vegetation in a Moderately Contaminated Salt Marsh (Tagus Estuary, Portugal). J. Environ. Sci. 2010, 22, 1151–1157. [Google Scholar] [CrossRef]
- Bravo, A.G.; Cosio, C.; Amouroux, D.; Zopfi, J.; Chevalley, P.-A.; Spangenberg, J.E.; Ungureanu, V.-G.; Dominik, J. Extremely Elevated Methyl Mercury Levels in Water, Sediment and Organisms in a Romanian Reservoir Affected by Release of Mercury from a Chlor-Alkali Plant. Water Res. 2014, 49, 391–405. [Google Scholar] [CrossRef]
- Sinclair, K.A.; Xie, Q.; Mitchell, C.P.J. Methylmercury in Water, Sediment, and Invertebrates in Created Wetlands of Rouge Park, Toronto, Canada. Environ. Pollut. 2012, 171, 207–215. [Google Scholar] [CrossRef]
- Breitburg, D.; Levin, L.A.; Oschlies, A.; Grégoire, M.; Chavez, F.P.; Conley, D.J.; Garçon, V.; Gilbert, D.; Gutiérrez, D.; Isensee, K. Declining Oxygen in the Global Ocean and Coastal Waters. Science 2018, 359, eaam7240. [Google Scholar] [CrossRef]
- Rak, D.; Walczowski, W.; Dzierzbicka-Głowacka, L.; Shchuka, S. Dissolved Oxygen Variability in the Southern Baltic Sea in 2013–2018. Oceanologia 2020, 62, 525–537. [Google Scholar] [CrossRef]
- Canário, J.; Santos-Echeandia, J.; Padeiro, A.; Amaro, E.; Strass, V.; Klaas, C.; Hoppema, M.; Ossebaar, S.; Koch, B.P.; Laglera, L.M. Mercury and Methylmercury in the Atlantic Sector of the Southern Ocean. Deep Sea Res. Part II Top. Stud. Oceanogr. 2017, 138, 52–62. [Google Scholar] [CrossRef]
- Mao, L.; Liu, X.; Wang, B.; Lin, C.; Xin, M.; Zhang, B.-T.; Wu, T.; He, M.; Ouyang, W. Occurrence and Risk Assessment of Total Mercury and Methylmercury in Surface Seawater and Sediments from the Jiaozhou Bay, Yellow Sea. Sci. Total Environ. 2020, 714, 136539. [Google Scholar] [CrossRef]
- Ci, Z.; Zhang, X.; Wang, Z.; Niu, Z. Phase Speciation of Mercury (Hg) in Coastal Water of the Yellow Sea, China. Mar. Chem. 2011, 126, 250–255. [Google Scholar] [CrossRef]
- Fu, X.; Feng, X.; Zhang, G.; Xu, W.; Li, X.; Yao, H.; Liang, P.; Li, J.; Sommar, J.; Yin, R.; et al. Mercury in the Marine Boundary Layer and Seawater of the South China Sea: Concentrations, Sea/Air Flux, and Implication for Land Outflow. J. Geophys. Res. Atmos. 2010, 115, 1–11. [Google Scholar] [CrossRef]
- Mason, R.P.; Rolfhus, K.R.; Fitzgerald, W.F. Mercury in the North Atlantic. Mar. Chem. 1998, 61, 37–53. [Google Scholar] [CrossRef]
- Laurier, F.J.G.; Mason, R.P.; Gill, G.A.; Whalin, L. Mercury Distributions in the North Pacific Ocean—20 Years of Observations. Mar. Chem. 2004, 90, 3–19. [Google Scholar] [CrossRef]
- Cossa, D.; Martin, J.-M.; Takayanagi, K.; Sanjuan, J. The Distribution and Cycling of Mercury Species in the Western Mediterranean. Deep Sea Res. Part II Top. Stud. Oceanogr. 1997, 44, 721–740. [Google Scholar] [CrossRef]
- Lamborg, C.H.; Fitzgerald, W.F.; Damman, A.W.H.; Benoit, J.M.; Balcom, P.H.; Engstrom, D.R. Modern and Historic Atmospheric Mercury Fluxes in Both Hemispheres: Global and Regional Mercury Cycling Implications. Glob. Biogeochem. Cycles 2002, 16, 51-1–51-11. [Google Scholar] [CrossRef]
- Ramlal, P.S.; Bugenyi, F.W.B.; Kling, G.W.; Nriagu, J.O.; Rudd, J.W.M.; Campbell, L.M. Mercury Concentrations in Water, Sediment, and Biota from Lake Victoria, East Africa. J. Great Lakes Res. 2003, 29, 283–291. [Google Scholar] [CrossRef]
- Morel, F.M.M.; Kraepiel, A.M.L.; Amyot, M. The chemical cycle and bioaccumulation of mercury. Annu. Rev. Ecol. Syst. 1998, 29, 543–566. [Google Scholar] [CrossRef]
- Engstrom, D.R. Fish Respond When the Mercury Rises. Proc. Natl. Acad. Sci. USA 2007, 104, 16394–16395. [Google Scholar] [CrossRef]
- Wiener, J.G.; Krabbenhoft, D.P.; Heinz, G.H.; Scheuhammer, A.M. Ecotoxicology of Mercury. In Handbook of Ecotoxicology, 2nd ed.; Hoffman, D.J., Rattner, B.A., Burton, G.A., Jr., Cairns, J., Jr., Eds.; Lewis Publishers: Boca Raton, FL, USA, 2003; pp. 409–463. [Google Scholar]
- Hong, Y.-S.; Kim, Y.-M.; Lee, K.-E. Methylmercury Exposure and Health Effects. J. Prev. Med. Public Health 2012, 45, 353–363. [Google Scholar] [CrossRef]
- De Giovanni, A.; Giuliani, C.; Marini, M.; Luiselli, D. Methylmercury and Polycyclic Aromatic Hydrocarbons in Mediterranean Seafood: A Molecular Anthropological Perspective. Appl. Sci. 2021, 11, 1179. [Google Scholar] [CrossRef]
- EC Regulation No 1881/2006 Commission Regulation (EC) No 1881/2006 of 19 December 2006 Setting Maximum Levels for Certain Contaminants in Foodstuffs (Text with EEA Relevance). 2006. Available online: https://eur-lex.eu/eli/reg/2006/1881/oj (accessed on 13 August 2022).
- United States Environmental Protection Agency, US EPA. EPA-823-F-01-001 Water Quality Criterion for the Protection of Human Health: Methylmercury (Executive Summary). 2001. Available online: https://www.epa.gov/sites/default/files/2020-01/documents/methylmercury-criterion-2001.pdf (accessed on 13 August 2022).
- Harada, M. Minamata Disease: Methylmercury Poisoning in Japan Caused by Environmental Pollution. Crit. Rev. Toxicol. 1995, 25, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Berlin, M.; Zalups, R.; Fowler, B. Hg. In Handbook on the Toxicology of Metals, 4th ed.; Nordberg, G., Fowler, B., Nordberg, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; ISBN 978-0-12-397339-9. [Google Scholar]
- Beasley, D.M.G.; Schep, L.J.; Slaughter, R.J.; Temple, W.A.; Michel, J.M. Full Recovery from a Potentially Lethal Dose of Mercuric Chloride. J. Med. Toxicol. 2014, 10, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Murata, K.; Weihe, P.; Budtz-Jørgensen, E.; Jørgensen, P.J.; Grandjean, P. Delayed Brainstem Auditory Evoked Potential Latencies in 14-Year-Old Children Exposed to Methylmercury. J. Pediatr. 2004, 144, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; Murata, K.; Budtz-Jørgensen, E.; Weihe, P. Cardiac Autonomic Activity in Methylmercury Neurotoxicity: 14-Year Follow-up of a Faroese Birth Cohort. J. Pediatr. 2004, 144, 169–176. [Google Scholar] [CrossRef]
- Clarkson, T.W.; Magos, L. The Toxicology of Mercury and Its Chemical Compounds. Crit. Rev. Toxicol. 2006, 36, 609–662. [Google Scholar] [CrossRef]
- Dorea, J.G. Mercury and Lead during Breast-Feeding. Br. J. Nutr. 2004, 92, 21–40. [Google Scholar] [CrossRef]
- Nowicka, B. Heavy Metal-Induced Stress in Eukaryotic Algae-Mechanisms of Heavy Metal Toxicity and Tolerance with Particular Emphasis on Oxidative Stress in Exposed Cells and the Role of Antioxidant Response. Environ. Sci. Pollut. Res. Int. 2022, 29, 16860–16911. [Google Scholar] [CrossRef]
- DalCorso, G. Heavy Metal Toxicity in Plants. In Plants and Heavy Metals; Furini, A., Ed.; Springer: Dordrecht, The Netherlands, 2012; pp. 1–25. ISBN 978-94-007-4441-7. [Google Scholar]
- Kaur, R.; Nayyar, H. Ascorbic Acid: A Potent Defender Against Environmental Stresses. In Oxidative Damage to Plants: Antioxidant Networks and Signaling, 1st ed.; Parvaiz, A., Ed.; Elsevier Academic Press: Cambridge, MA, USA; pp. 235–287. ISBN 978-0-12-799963-0.
- Shahzad, B.; Tanveer, M.; Rehman, A.; Cheema, S.A.; Fahad, S.; Rehman, S.; Sharma, A. Nickel; Whether Toxic or Essential for Plants and Environment—A Review. Plant Physiol. Biochem. 2018, 132, 641–651. [Google Scholar] [CrossRef]
- Benavides, M.; Gallego, S.; Tomaro, M. Cadmium Toxicity in Plants. Braz. J. Plant Physiol. 2005, 17, 21–34. [Google Scholar] [CrossRef]
- Chekroun, K.B.; Baghour, M. The Role of Algae in Phytoremediation of Heavy Metals: A Review. J. Mater. Environ. Sci. 2013, 4, 873–880. [Google Scholar]
- Nowicka, B.; Kruk, J. Powered by Light: Phototrophy and Photosynthesis in Prokaryotes and Its Evolution. Microbiol. Res. 2016, 186–187, 99–118. [Google Scholar] [CrossRef] [PubMed]
- Balzano, S.; Sardo, A.; Blasio, M.; Chahine, T.B.; Dell’Anno, F.; Sansone, C.; Brunet, C. Microalgal Metallothioneins and Phytochelatins and Their Potential Use in Bioremediation. Front. Microbiol. 2020, 11, 517. [Google Scholar] [CrossRef] [PubMed]
- Zolotareva, E.; Mokrosnop, V.; Stepanov, S. Polyphenol Compounds of Macroscopic and Microscopic Algae. Int. J. Algae 2019, 21, 5–24. [Google Scholar] [CrossRef]
- Ajitha, V.; Sreevidya, C.P.; Sarasan, M.; Park, J.C.; Mohandas, A.; Singh, I.S.B.; Puthumana, J.; Lee, J.-S. Effects of Zinc and Mercury on ROS-Mediated Oxidative Stress-Induced Physiological Impairments and Antioxidant Responses in the Microalga Chlorella vulgaris. Environ. Sci. Pollut. Res. 2021, 28, 32475–32492. [Google Scholar] [CrossRef]
- Küpper, H.; Andresen, E. Mechanisms of Metal Toxicity in Plants. Metallomics 2016, 8, 269–285. [Google Scholar] [CrossRef]
- Manomita Patra; Archana Sharma Mercury Toxicity in Plants. Bot. Rev. 2000, 66, 379–422. [CrossRef]
- Shrivastava, S.; Shrivastav, A.; Sharma, J. Detoxification Mechanisms of Mercury Toxicity in Plants—A Review. Recent Adv. Biol. Med. 2015, 1, 60–68. [Google Scholar] [CrossRef]
- Sharaf, A.; De Michele, R.; Sharma, A.; Fakhari, S.; Oborník, M. Transcriptomic Analysis Reveals the Roles of Detoxification Systems in Response to Mercury in Chromera velia. Biomolecules 2019, 9, 647. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, W.-X. Intracellular Speciation and Transformation of Inorganic Mercury in Marine Phytoplankton. Aquat. Toxicol. 2014, 148, 122–129. [Google Scholar] [CrossRef]
- Tredici, M.R. Photobiology of Microalgae Mass Cultures: Understanding the Tools for the next Green Revolution. Biofuels 2010, 1, 143–162. [Google Scholar] [CrossRef]
- Borisov, A.Y.; Björn, L.O. On Oxygen Production by Photosynthesis: A Viewpoint. Photosynthetica 2018, 56, 44–47. [Google Scholar] [CrossRef]
- Spain, O.; Plöhn, M.; Funk, C. The Cell Wall of Green Microalgae and Its Role in Heavy Metal Removal. Physiol. Plant 2021, 173, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Javanbakht, V.; Alavi, S.A.; Zilouei, H. Mechanisms of Heavy Metal Removal Using Microorganisms as Biosorbent. Water Sci. Techno.l 2014, 69, 1775–1787. [Google Scholar] [CrossRef] [PubMed]
- Campbell, P.G.C.; Errécalde, O.; Fortin, C.; Hiriart-Baer, V.P.; Vigneault, B. Metal Bioavailability to Phytoplankton—Applicability of the Biotic Ligand Model. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2002, 133, 189–206. [Google Scholar] [CrossRef]
- Dranguet, P.; Flück, R.; Regier, N.; Cosio, C.; Le Faucheur, S.; Slaveykova, V.I. Towards Mechanistic Understanding of Mercury Availability and Toxicity to Aquatic Primary Producers. CHIMIA Int. J. Chem. 2014, 68, 799–805. [Google Scholar] [CrossRef]
- Shchukarev, A.; Gojkovic, Z.; Funk, C.; Ramstedt, M. Cryo-XPS Analysis Reveals Surface Composition of Microalgae. Appl. Surf. Sci. 2020, 526, 146538. [Google Scholar] [CrossRef]
- Gojkovic, Z.; Shchukarev, A.; Ramstedt, M.; Funk, C. Cryogenic X-ray Photoelectron Spectroscopy Determines Surface Composition of Algal Cells and Gives Insights into Their Spontaneous Sedimentation. Algal Res. 2020, 47, 101836. [Google Scholar] [CrossRef]
- Hadjoudja, S.; Deluchat, V.; Baudu, M. Cell Surface Characterisation of Microcystis Aeruginosa and Chlorella vulgaris. J. Colloid Interface Sci. 2010, 342, 293–299. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, W.-X. Accumulation, Subcellular Distribution and Toxicity of Inorganic Mercury and Methylmercury in Marine Phytoplankton. Environ. Pollut. 2011, 159, 3097–3105. [Google Scholar] [CrossRef]
- Luoma, S.N.; Rainbow, P.S. Why Is Metal Bioaccumulation So Variable? Biodynamics as a Unifying Concept. Environ. Sci. Technol. 2005, 39, 1921–1931. [Google Scholar] [CrossRef] [PubMed]
- Slaveykova, V.I.; Majumdar, S.; Regier, N.; Li, W.; Keller, A.A. Metabolomic Responses of Green Alga Chlamydomonas reinhardtii Exposed to Sublethal Concentrations of Inorganic and Methylmercury. Environ. Sci. Technol. 2021, 55, 3876–3887. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Noctor, G. Ascorbate and Glutathione: The Heart of the Redox Hub. Plant Physiol. 2011, 155, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Jacinto, V.; García-Barrera, T.; Gómez-Ariza, J.L.; Garbayo-Nores, I.; Vílchez-Lobato, C. Elucidation of the Defence Mechanism in Microalgae Chlorella sorokiniana under Mercury Exposure. Identification of Hg–Phytochelatins. Chem.-Biol. Interact. 2015, 238, 82–90. [Google Scholar] [CrossRef]
- Duboc, P.; Marison, I.; von Stockar, U. Quantitative Calorimetry and Biochemical Engineering. In Handbook of Thermal Analysis and Calorimetry; Kemp, R., Ed.; Elsevier: Amsterdam, The Netherlands, 1999; Volume 4, pp. 267–365. ISBN 978-0-08-053569-2. [Google Scholar]
- Beauvais-Flück, R.; Slaveykova, V.I.; Cosio, C. Cellular Toxicity Pathways of Inorganic and Methyl Mercury in the Green Microalga Chlamydomonas reinhardtii. Sci. Rep. 2017, 7, 8034. [Google Scholar] [CrossRef]
- Zhong, H.; Wang, W.-X. Controls of Dissolved Organic Matter and Chloride on Mercury Uptake by a Marine Diatom. Environ. Sci. Technol. 2009, 43, 8998–9003. [Google Scholar] [CrossRef]
- Bouchet, S.; Björn, E. Analytical Developments for the Determination of Monomethylmercury Complexes with Low Molecular Mass Thiols by Reverse Phase Liquid Chromatography Hyphenated to Inductively Coupled Plasma Mass Spectrometry. J. Chromatogr. A 2014, 1339, 50–58. [Google Scholar] [CrossRef]
- Duanmu, D.; Miller, A.R.; Horken, K.M.; Weeks, D.P.; Spalding, M.H. Knockdown of Limiting-CO2–Induced Gene HLA3 Decreases HCO3− Transport and Photosynthetic Ci Affinity in Chlamydomonas reinhardtii. Proc. Natl. Acad. Sci. USA 2009, 106, 5990–5995. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).