Short-Term Physiological and Transcriptomic Analysis of Saxitoxin-Producing and Nonproducing Raphidiopsis raciborskii (Cyanobacteria) under an Extremely High CO2 Environment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cyanobacterial Strains and Culture Conditions
2.2. Experimental Setup
2.3. Growth and Photosynthesis Measurements
2.4. Transcriptomic Profile of CCM and Photosynthesis-Related Genes
2.5. Sample Processing and RNA Extraction
2.6. Library, Sequencing and Gene Expression Analysis
2.7. Saxitoxin Extraction and HPLC-FLD Analysis
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Paerl, H.W.; Huisman, J. Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environ. Microbiol. Rep. 2009, 11, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.; Visser, P.M. Cyanobacterial blooms. Nat. Rev. Microb. 2018, 16, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Pachauri, R.K.L.; Meyer, G.K.; Plattner, T.; Stocker, T.F. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2015. [Google Scholar]
- Raven, J.A.; Gobler, C.J.; Hansen, P.J. Dynamic CO2 and pH levels in coastal, estuarine, and inland waters: Theoretical and observed effects on harmful algal blooms. Harmful Algae 2020, 91, 101594. [Google Scholar] [CrossRef] [PubMed]
- Kasting, J.F.; Howard, M.T. Atmospheric composition and climate on the early Earth. Philos. Trans. R. Soc. B Biol. Sci. 2006, 3611474, 1733–1742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bergman, N.M.; Lenton, T.M.; Watson, A.J. COPSE: A new model of biogeochemical cycling over Phanerozoic time. Am. J. Sci. 2004, 3045, 397–437. [Google Scholar] [CrossRef]
- Lehmer, O.R.; Catling, D.C.; Buick, R.; Brownlee, D.E.; Newport, S. Atmospheric CO2 levels from 2.7 billion years ago inferred from micrometeorite oxidation. Sci. Adv. 2020, 6, eaay4644. [Google Scholar] [CrossRef] [Green Version]
- Altermann, W. The Early Earth’s Record of Supposed Extremophilic Bacteria and Cyanobacteria, at 3.8 to 2.5 GA. In Algae and Cyanobacteria in Extreme Environments; Springer: Dordrecht, The Netherlands, 2007; pp. 759–778. [Google Scholar]
- Van de Waal, D.B.; Brandenburg, K.M.; Keuskamp, J.; Trimborn, S.; Rokitta, S.; Kranz, S.A.; Rost, B. Highest plasticity of carbon-concentrating mechanisms in earliest evolved phytoplankton. LO Lett. 2019, 42, 37–43. [Google Scholar] [CrossRef] [Green Version]
- Burnap, R.L.; Hagemann, M.; Kaplan, A. Regulation of CO2 concentrating mechanism in cyanobacteria. Life 2015, 5, 348–371. [Google Scholar] [CrossRef] [Green Version]
- Hagemann, M.; Kern, R.; Maurino, V.G.; Hanson, D.T.; Weber, A.P.; Sage, R.F.; Bauwe, H. Evolution of photorespiration from cyanobacteria to land plants, considering protein phylogenies and acquisition of carbon concentrating mechanisms. J. Exp. Bot. 2016, 6710, 2963–2976. [Google Scholar] [CrossRef] [Green Version]
- Price, G.D.; Badger, M.R.; Woodger, F.J.; Long, B.M. Advances in understanding the cyanobacterial CO2-concentrating-mechanism (CCM): Functional components, Ci transporters, diversity, genetic regulation and prospects for engineering into plants. J. Exp. Bot. 2008, 59, 1441–1461. [Google Scholar] [CrossRef]
- Sandrini, G.; Ji, X.; Verspagen, J.M.; Tann, R.P.; Slot, P.C.; Luimstra, V.M.; Schuurmans, J.M.; Matthijs, H.C.P.; Huisman, J. Rapid adaptation of harmful cyanobacteria to rising CO2. Proc. Natl. Acad. Sci. USA 2016, 113, 9315–9320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raven, J.A.; Giordano, M.; Beardall, J.; Maberly, S.C. Algal and aquatic plant carbon concentrating mechanisms in relation to environmental change. Photosynth. Res. 2011, 109, 281–296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aguilera, A.; Gómez, E.B.; Kaštovský, J.; Echenique, R.O.; Salerno, G.L. The polyphasic analysis of two native Raphidiopsis isolates supports the unification of the genera Raphidiopsis and Cylindrospermopsis (Nostocales, Cyanobacteria). Phycologia 2018, 57, 130–146. [Google Scholar] [CrossRef]
- Burford, M.A.; Mcneale, K.L.; Mckenzi-Smith, F.J. The role of nitrogen in promoting the toxic cyanophyte Cylindrospermopsis raciborskii in a subtropical water reservoir. Freshw. Biol. 2006, 5111, 2143–2153. [Google Scholar] [CrossRef] [Green Version]
- Chonudomkul, D.; Yongmanitchai, W.; Theeragool, G.; Kawachi, M.; Kasai, F.; Kaya, K.; Watanabe, M.M. Morphology, genetic diversity, temperature tolerance and toxicity of Cylindrospermopsis raciborskii Nostocales, Cyanobacteria strains from Thailand and Japan. FEMS Microbiol. Ecol. 2004, 483, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Xu, H.; McCarthy, M.J.; Zhu, G.; Qin, B.; Li, Y.; Gardner, W.S. Controlling harmful cyanobacterial blooms in a hyper-eutrophic lake (Lake Taihu, China): The need for a dual nutrient (N & P) management strategy. Water Res. 2011, 45, 1973–1983. [Google Scholar] [PubMed]
- Antunes, J.; Leão, P.; Vasconcelos, V. Cylindrospermopsis raciborskii: Review of the distribution, phylogeography, and ecophysiology of a global invasive species. Front. Microbiol. 2015, 6, 473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holland, D.P.; Pantorno, A.; Orr, P.T.; Stojkovic, S.; Beardall, J. The impacts of a high CO2 environment on a bicarbonate user: The cyanobacterium Cylindrospermopsis raciborskii. Water Res. 2012, 465, 1430–1437. [Google Scholar] [CrossRef] [PubMed]
- Vilar, M.C.P.; Molica, R.J.R. Changes in pH and dissolved inorganic carbon in water affect the growth, saxitoxins production and toxicity of the cyanobacterium Raphidiopsis raciborskii ITEP-A1. Harmful Algae 2020, 97, 101870. [Google Scholar] [CrossRef]
- Bonilla, S.; Aubriot, L.; Soares, M.C.S.; Gonzalez-Piana, M.; Fabre, A.; Huszar, V.L.; Lürling, M.; Antoniades, D.; Padisák, J.; Kruk, C. What drives the distribution of the bloom-forming cyanobacteria Planktothrix agardhii and Cylindrospermopsis raciborskii? FEMS Microbiol. Ecol. 2012, 793, 594–607. [Google Scholar] [CrossRef]
- Wiese, M.; D’agostino, P.M.; Mihali, T.K.; Moffitt, M.C.; Neilan, B.A. Neurotoxic alkaloids: Saxitoxin and its analogs. Mar. Drug 2010, 8, 2185–2211. [Google Scholar] [CrossRef] [Green Version]
- Scarlett, K.R.; Kim, S.; Lovin, L.M.; Chatterjee, S.; Scott, J.T.; Brooks, B.W. Global scanning of cylindrospermopsin: Critical review and analysis of aquatic occurrence, bioaccumulation, toxicity and health hazards. Sci. Total Environ. 2020, 738, 139807. [Google Scholar] [CrossRef]
- Lagos, N.; Onodera, H.; Zagatto, P.A.; Andrinolo, D.; Azevedo, S.M.; Oshima, Y. The first evidence of paralytic shellfish toxins in the freshwater cyanobacterium Cylindrospermopsis raciborskii, isolated from Brazil. Toxicon 1999, 3710, 1359–1373. [Google Scholar] [CrossRef]
- Molica, R.; Onodera, H.; García, C.; Rivas, M.; Andrinolo, D.; Nascimento, S.; Meguro, H.; Oshima, Y.; Azevedo, S.M.F.O.; Lagos, N. Toxins in the freshwater cyanobacterium Cylindrospermopsis raciborskii Cyanophyceae isolated from Tabocas reservoir in Caruaru, Brazil, including demonstration of a new saxitoxin analogue. Phycologia 2002, 41, 606–611. [Google Scholar] [CrossRef]
- Lorenzi, A.S.; Cordeiro-Araújo, M.K.; Chia, M.A.; Bittencourt-Oliveira, M.C. Cyanotoxin contamination of semiarid drinking water supply reservoirs. Environ. Earth Sci. 2018, 77, 1–8. [Google Scholar] [CrossRef]
- Abreu, V.; Popin, R.V.; Alvarenga, D.O.; Schaker, P.D.C.; Hoff-Risseti, C.; Varani, A.M.; Fiore, M.F. Corrigendum: Genomic and genotypic characterization of Cylindrospermopsis raciborskii: Toward an intraspecific phylogenetic evaluation by comparative genomics. Front. Microbiol. 2018, 9, 979. [Google Scholar] [CrossRef] [PubMed]
- Pierangelini, M.; Stojkovic, S.; Orr, P.T.; Beardall, J. Elevated CO2 causes changes in the photosynthetic apparatus of a toxic cyanobacterium, Cylindrospermopsis raciborskii. J. Plant Physiol. 2014, 17112, 1091–1098. [Google Scholar] [CrossRef] [PubMed]
- Pierangelini, M.; Sinha, R.; Willis, A.; Buford, M.A.; Orr, P.T.; Beardall, J.; Neilan, B.A. Constitutive cylindrospermopsin pool size in Cylindrospermopsis raciborskii under different light and CO2 partial pressure conditions. J. Appl. Environ. Microbiol. 2015, 819, 3069–3076. [Google Scholar] [CrossRef] [Green Version]
- Willis, A.; Chuang, A.W.; Orr, P.T.; Beardall, J.; Burford, M.A. Subtropical freshwater phytoplankton show a greater response to increased temperature than to increased pCO2. Harmful Algae 2019, 90, 101705. [Google Scholar] [CrossRef] [PubMed]
- Rangel, L.M.; Ger, K.A.; Silva, L.H.; Soares, M.C.S.; Faassen, E.J.; Lürling, M. Toxicity overrides morphology on Cylindrospermopsis raciborskii grazing resistance to the calanoid copepod Eudiaptomus gracilis. Microb. Ecol. 2016, 71, 835–844. [Google Scholar] [CrossRef] [Green Version]
- Vilar, M.C.; Rodrigues, T.F.; Silva, L.O.; Pacheco, A.B.F.; Ferrão-Filho, A.S.; Azevedo, S.M. Ecophysiological aspects and sxt genes expression underlying induced chemical defense in STX-producing Raphidiopsis raciborskii (cyanobacteria) against the zooplankter Daphnia gessneri. Toxins 2021, 13, 406. [Google Scholar] [CrossRef]
- Marinho, M.M.; Souza, M.B.G.; Lürling, M. Light and phosphate competition between Cylindrospermopsis raciborskii and Microcystis aeruginosa is strain dependent. Microb. Ecol. 2013, 66, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Gorham, P.; McLachlan, J.; Hammer, U.T.; Ki, W.K. Isolation and culture of toxic strains of Anabaena flos-aquae lyngb. Bréb SIL Proc. 1964, 15, 796. [Google Scholar]
- Hillebrand, H.; Dürselen, C.D.; Kirschtel, D.; Pollingher, U.; Zohary, T. Biovolume calculation for pelagic and benthic microalgae. J. Phycol. 1999, 35, 403–424. [Google Scholar] [CrossRef]
- Sun, J.; Liu, D. Geometric models for calculating cell biovolume and surface area for phytoplankton. J. Plankton Res. 2003, 25, 1331–1346. [Google Scholar] [CrossRef] [Green Version]
- Reynolds, C.S. The Ecology of Phytoplankton; Cambridge University Press: Cambridge, UK, 2006; 435p. [Google Scholar]
- Robinson, M.D.; Smyth, G.K. Small sample estimation of negative binomial dispersion, with applications to SAGE data. Biostatistics 2008, 9, 321–332. [Google Scholar] [CrossRef] [Green Version]
- Baggerly, K.A.; Deng, L.; Morris, J.S.; Aldaz, C.M. Differential expression in SAGE: Accounting for normal between-library variation. Bioinformatics 2003, 19, 1477–1483. [Google Scholar] [CrossRef]
- Oshima, Y. Postcolumn derivatization liquid chromatographic method for paralytic shellfish toxins. J. AOAC Int. 1995, 782, 528–532. [Google Scholar] [CrossRef]
- Willis, A.; Chuang, A.W.; Woodhouse, J.N.; Neilan, B.A.; Burford, M.A. Intraspecific variation in growth, morphology and toxin quotas for the cyanobacterium, Cylindrospermopsis raciborskii. Toxicon 2016, 119, 307–310. [Google Scholar] [CrossRef]
- Saker, M.L.; Neilan, B.A. Varied diazotrophies, morphologies, and toxicities of genetically similar isolates of Cylindrospermopsis raciborskii (Nostocales, Cyanophyceae) from Northern Australia. Appl. Environ. Microbiol. 2001, 67, 1839–1845. [Google Scholar] [CrossRef] [Green Version]
- Moore, L.; Rocap, G.; Chisholm, S.W. Physiology and molecular phylogeny of coexisting Prochlorococcus ecotypes. Nature 1998, 393, 464–467. [Google Scholar] [CrossRef]
- Ivars-Martinez, E.; Martin-Cuadrado, A.B.; D’Auria, G.; Mira, A.; Ferriera, S.; Johnson, J.; Friedman, R.; Rodriguez-Valera, F. Comparative genomics of two ecotypes of the marine planktonic copiotroph Alteromonas macleodii suggests alternative lifestyles associated with different kinds of particulate organic matter. ISME J. 2008, 2, 1194–1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carlson, C.A.; Morris, R.; Parsons, R.; Treusch, A.H.; Giovannoni, S.J.; Vergin, K. Seasonal dynamics of SAR11 populations in the euphotic and mesopelagic zones of the northwestern Sargasso Sea. ISME J. 2009, 3, 83–295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lima, D.V.N.; Pacheco, A.B.F.; Goulart, C.L.; Azevedo, S.M.F.O. Physiological responses of Raphidiopsis raciborskii (Cyanobacteria) strains to water conductivity: Effect of sodium and magnesium ions. Hydrobiologia 2020, 847, 2449–2464. [Google Scholar] [CrossRef]
- Sandrini, G.; Cunsolo, S.; Schuurmans, J.M.; Matthijs, H.C.; Huisman, J. Changes in gene expression, cell physiology and toxicity of the harmful cyanobacterium Microcystis aeruginosa at elevated CO2. Front. Microbiol. 2015, 6, 401. [Google Scholar] [CrossRef]
- Sinha, R.; Pearson, L.A.; Davis, T.W.; Buford, M.A.; Orr, P.T.; Neilan, B.A. Increased incidence of Cylindrospermopsis raciborskii in temperate zones–is climate change responsible? Water Res. 2012, 465, 1408–1419. [Google Scholar] [CrossRef]
- Mantzouki, E.; Lürling, M.; Fastner, J.; de Domis, L.S.; Wilk-Woźniak, E.; Koreivienė, J.; Seelen, L.; Teurlincx, S.; Verstijnen, Y.; Krztoń, W.; et al. Temperature effects explain continental scale distribution of cyanobacterial toxins. Toxins 2018, 10, 156. [Google Scholar] [CrossRef] [Green Version]
- Paerl, H.W.; Paul, V.J. Climate change: Links to global expansion of harmful cyanobacteria. Water Res. 2012, 465, 1349–1363. [Google Scholar] [CrossRef]
- Paul, V.J.; Ritson-Williams, R.; Campbell, J.; Craft, J.D.; Langdon, C. Algal chemical ecology in a changing ocean. Planta Med. 2014, 80, IL11. [Google Scholar] [CrossRef]
- Ma, J.; Wang, P. Effects of rising atmospheric CO2 levels on physiological response of cyanobacteria and cyanobacterial bloom development: A review. Sci. Total Environ. 2021, 754, 141889. [Google Scholar] [CrossRef]

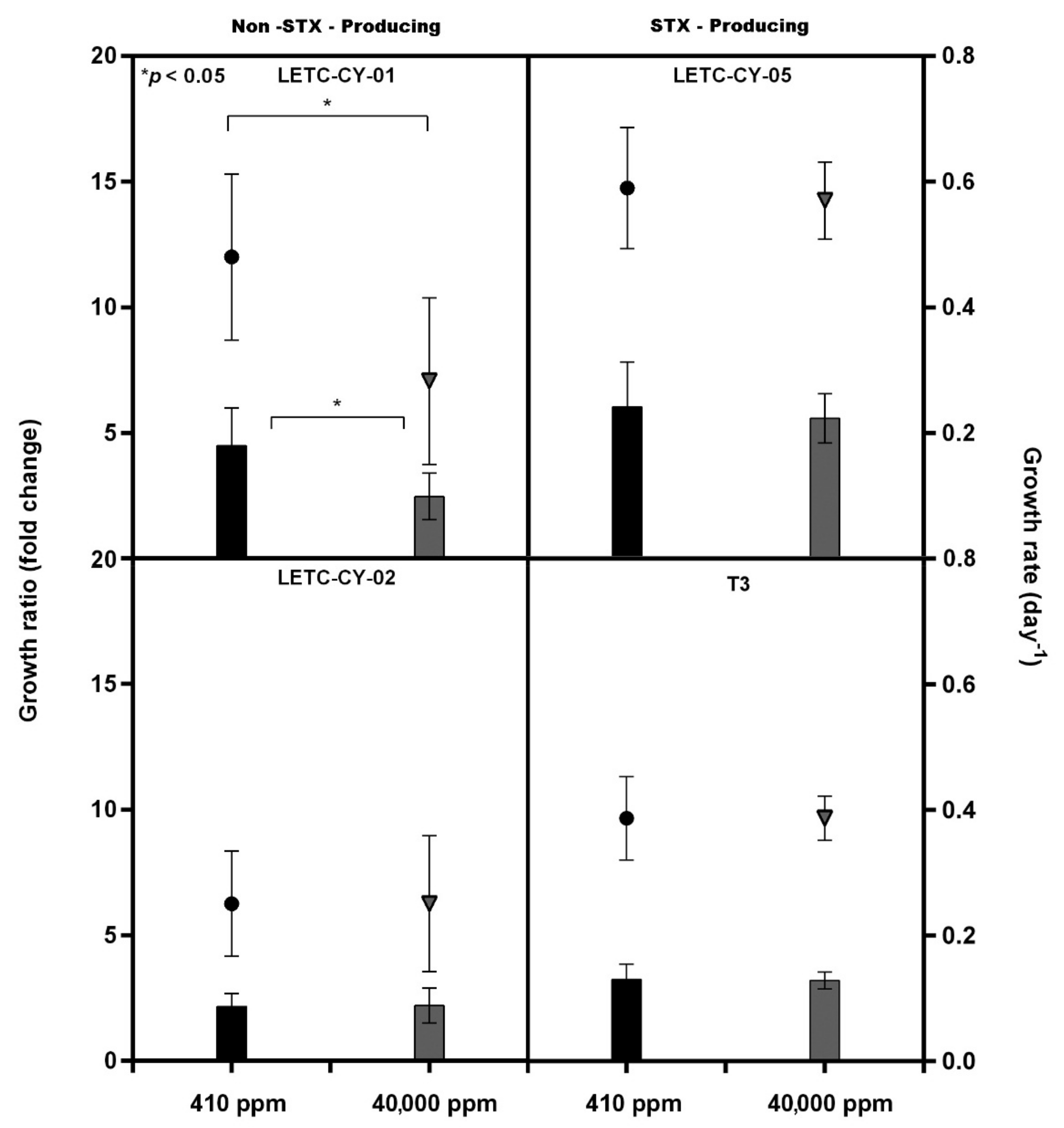

| Non-STX Producing | STX Producing | |||||||
|---|---|---|---|---|---|---|---|---|
| LETC-CY-01 | LETC-CY-02 | LETC-CY-05 | T3 | |||||
| Time | 410 ppm | 40,000 ppm | 410 ppm | 40,000 ppm | 410 ppm | 40,000 ppm | 410 ppm | 40,000 ppm |
| 0 h | 0.53 ± 0.00 a | 0.53 ± 0.00 a | 0.46 ± 0.00 | 0.46 ± 0.00 | 0.63 ± 0.00 | 0.63 ± 0.00 | 0.53 ± 0.00 | 0.53 ± 0.00 |
| 24 h | 0.45 ± 0.05 a | 0.33 ± 0.01 b | 0.52 ± 0.02 | 0.49 ± 0.04 | 0.59 ± 0.02 | 0.60 ± 0.01 | 0.48 ± 0.01 | 0.49 ± 0.01 |
| 48 h | 0.48 ± 0.03 a | 0.36 ± 0.02 b | 0.54 ± 0.01 | 0.51 ± 0.01 | 0.57 ± 0.01 | 0.54 ± 0.02 | 0.53 ± 0.01 a | 0.46 ± 0.03 b |
| 72 h | 0.53 ± 0.02 a | 0.45 ± 0.02 b | 0.47 ± 0.03 | 0.48 ± 0.01 | 0.57 ± 0.02 | 0.54 ± 0.03 | 0.55 ± 0.01 | 0.53 ± 0.02 |
| LETC-CY-01 | ||||
|---|---|---|---|---|
| Group | Annotation | Fold Change | p-Value | Feature ID |
| Membrane transport | Possible carbon dioxide concentrating mechanism protein CCM Activity K | −1.8 | 0.01 | CRC_00971 |
| Phosphoribulokinase (EC 2.7.1.19) | −1.79 | 0.01 | CRC_00573 | |
| TRAP dicarboxylate transporter, DctQ subunit, unknown substrate 6 | −1.78 | 0.03 | CRC_02038 | |
| Carbon dioxide concentrating mechanism protein CCM Activity O | −1.76 | 0.001 | CRC_00967 | |
| Carboxysome protein CCM Activity N | −1.58 | 0.01 | CRC_00968 | |
| Bicarbonate transporter, bicarbonate binding protein | 1.79 | 0.04 | CRC_00146 | |
| DevC protein | 1.56 | 0.05 | CRC_01107 | |
| ABC-transporter DevC-like protein | 2.1 | 0.01 | CRC_00437 | |
| Phospholipid-lipopolysaccharide ABC transporter | 3.5 | 0.002 | CRC_01570 | |
| RuBisCO regulation | Ribulose-1,5-bisphosphate carboxylase/oxygenase | −1.61 | 0.03 | rbcS |
| Ribulose-phosphate 3-epimerase (EC 5.1.3.1) | −1.54 | 0.03 | CRC_01082 | |
| Protein CP12, regulation of Calvin cycle via association/dissociation of PRK/CP12/GAPDH complex | −1.52 | 0.04 | CRC_01224 | |
| LETC-CY-05 | ||||
| Group | Annotation | Fold Change | p-Value | Feature ID |
| Membrane transport | Bicarbonate transport system permease protein | 3.36 | 0.04 | CRC_00145 |
| Bicarbonate transporter, bicarbonate binding protein | 5.02 | 0.01 | CRC_00146 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paranhos, R.R.; Vilar, M.; Hoffmann, L.; Bottaro, T.; Silva, R.; Pereira, R.C.; Azevedo, S.M.F.O. Short-Term Physiological and Transcriptomic Analysis of Saxitoxin-Producing and Nonproducing Raphidiopsis raciborskii (Cyanobacteria) under an Extremely High CO2 Environment. Phycology 2022, 2, 319-331. https://doi.org/10.3390/phycology2030017
Paranhos RR, Vilar M, Hoffmann L, Bottaro T, Silva R, Pereira RC, Azevedo SMFO. Short-Term Physiological and Transcriptomic Analysis of Saxitoxin-Producing and Nonproducing Raphidiopsis raciborskii (Cyanobacteria) under an Extremely High CO2 Environment. Phycology. 2022; 2(3):319-331. https://doi.org/10.3390/phycology2030017
Chicago/Turabian StyleParanhos, Ricardo Rogers, Mauro Vilar, Luísa Hoffmann, Thayane Bottaro, Rosane Silva, Renato C. Pereira, and Sandra M. F. O. Azevedo. 2022. "Short-Term Physiological and Transcriptomic Analysis of Saxitoxin-Producing and Nonproducing Raphidiopsis raciborskii (Cyanobacteria) under an Extremely High CO2 Environment" Phycology 2, no. 3: 319-331. https://doi.org/10.3390/phycology2030017
APA StyleParanhos, R. R., Vilar, M., Hoffmann, L., Bottaro, T., Silva, R., Pereira, R. C., & Azevedo, S. M. F. O. (2022). Short-Term Physiological and Transcriptomic Analysis of Saxitoxin-Producing and Nonproducing Raphidiopsis raciborskii (Cyanobacteria) under an Extremely High CO2 Environment. Phycology, 2(3), 319-331. https://doi.org/10.3390/phycology2030017







