Longevity, Aging and Cancer: Thermodynamics and Complexity
Abstract
:1. Introduction
2. Overview of the Thermodynamics of Complex Processes
- for everything in and ;
- The Eulerian derivative, for all in .
3. Longevity and Aging and Their Relationship with the Emergence and Evolution of Cancer
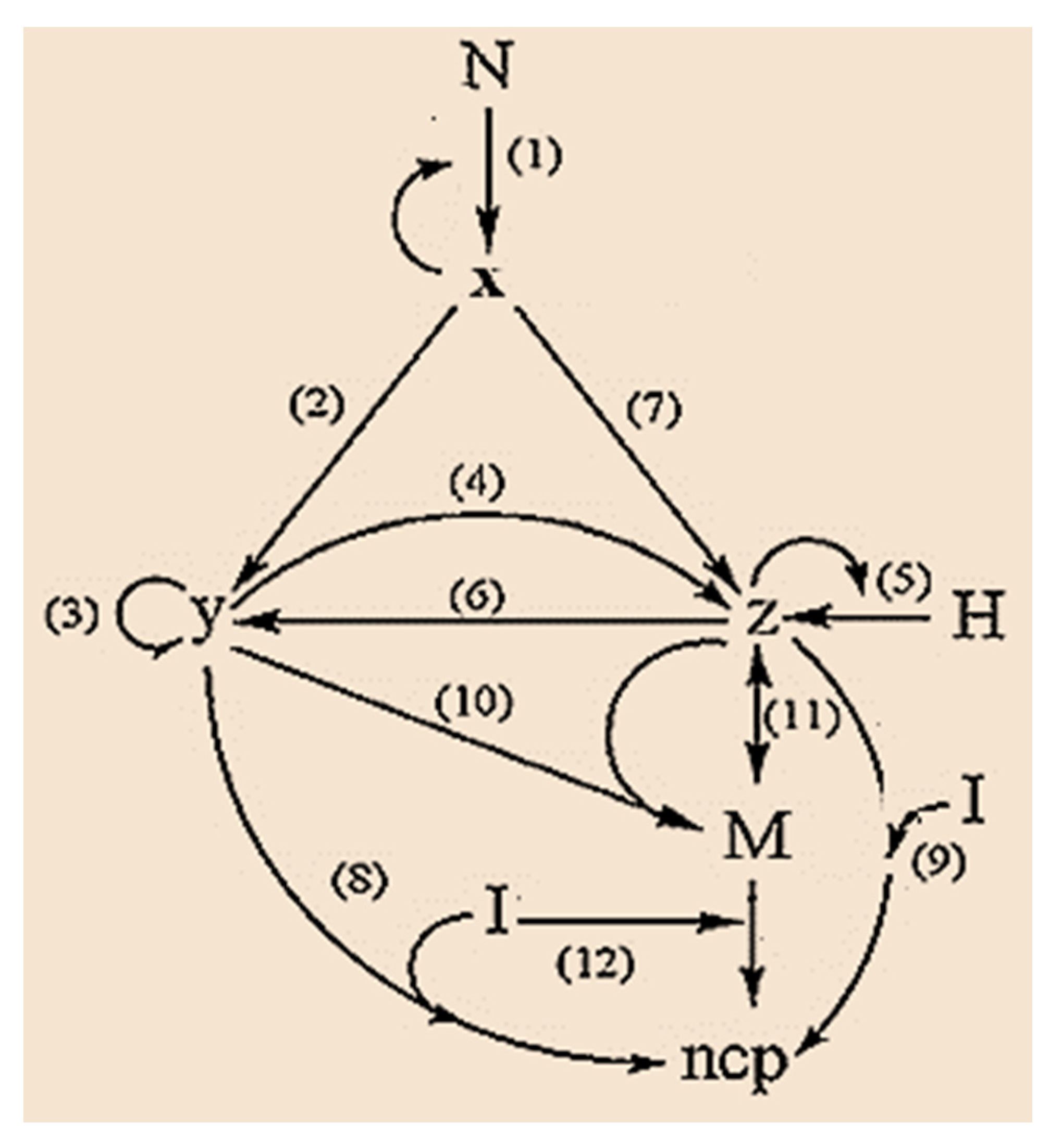
4. Ferroptosis and Cancer
5. Concluding Remarks
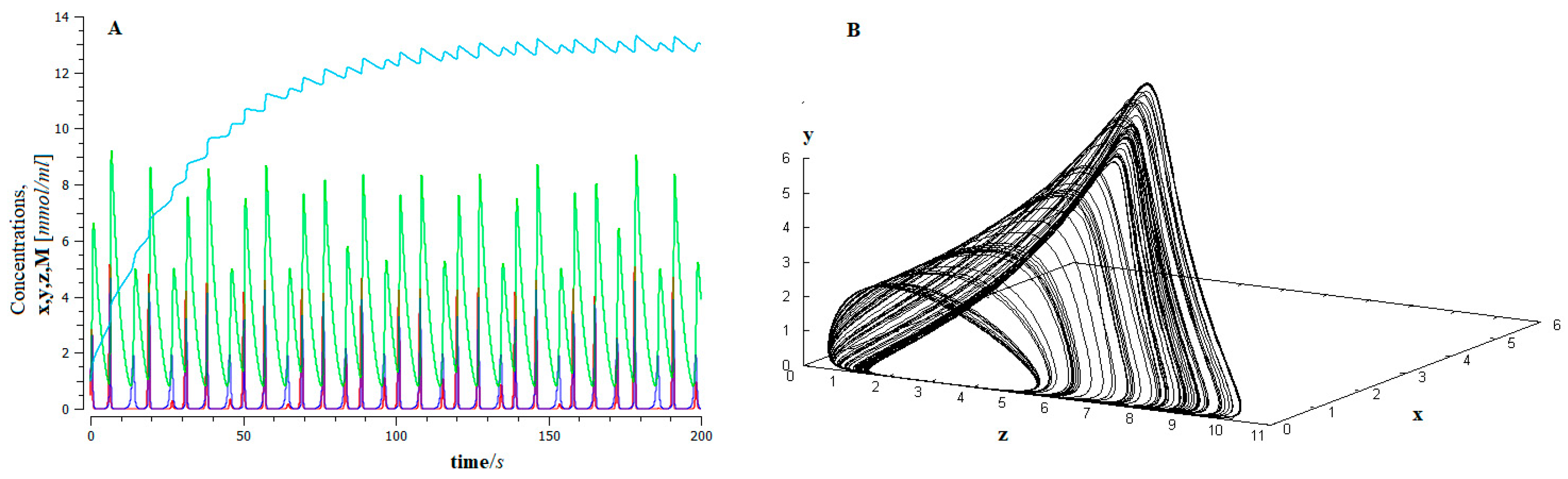
- The process of metastasis occurs through epithelial–mesenchymal transition (EMT), appears as a phase transition away from thermodynamic equilibrium, and exhibits Shilnikov chaos-like dynamic behavior. This dynamic guarantees the robustness of the process and, in turn, its unpredictability.
- The aging process, as well as the evolution of cancer, goes through what we have called a “biological phase transition”.
- The rate of entropy production can be used as an index of robustness, plasticity, and aggressiveness of cancer. It can also be used as a measure of biological age.
- It was shown that the extent to which the ferroptosis process is strengthened decreases the complexity in the dynamics associated with the emergency and evolution of cancer.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hayflick, L. Biological Aging Is No Longer an Unsolved Problem. Ann. N. Y. Acad. Sci. 2007, 1100, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ames, B.N.; Shigenaga, M.K.; Hagen, T.M. Oxidants, antioxidants, and the degenerative diseases of aging. Proc. Natl. Acad. Sci. USA 1993, 90, 7915–7922. [Google Scholar] [CrossRef] [PubMed]
- Harman, D. Free Radical Theory of Aging: An Update: Increasing the Functional Life Span. Ann. N. Y. Acad. Sci. 2006, 1067, 10–21. [Google Scholar] [CrossRef]
- Liochev, S.I. Reactive oxygen species and the free radical theory of aging. Free Radic. Biol. Med. 2013, 60, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Medvedev, Z.A. An attempt at a rational classification of theories of ageing. Biol. Rev. 1990, 65, 375–398. [Google Scholar] [CrossRef]
- Ghosh, C.; De, A. Basics of aging theories and disease-related aging-an overview. Pharma Tutor 2017, 5, 16–23. [Google Scholar]
- Harman, D. Aging: A Theory Based on Free Radical and Radiation Chemistry. J. Gerontol. 1956, 11, 298–300. [Google Scholar] [CrossRef]
- Cutler, R.G. Aging and oxygen radicals. Physiol. Oxyg. Radic. 1986, 18, 251–285. [Google Scholar]
- Sohal, R.S. Metabolic Rate, Free Radicals and Aging, in Free Radicals. In Molecular Biology, Aging and Disease, Free Radicals in Molecular Biology, Aging, and Disease; Armstrong, D., Ed.; Raven Production: Nashville, TN, USA, 1984; Volume 27. [Google Scholar]
- Miquel, J.; Economos, A.C.; Johnson, J.E., Jr. A Systems Analysis—Thermodynamic View of Cellular and Organismic Aging, Aging and Cell Function; Springer: New York, NY, USA, 1984. [Google Scholar]
- Balmer, R.T. Entropy and Aging in Biological Systems. Chem. Eng. Commun. 1982, 17, 171–181. [Google Scholar] [CrossRef]
- Gladyshev, G.P. The thermodynamic theory of evolution and aging. Adv. Gerontol. 2014, 4, 109–118. [Google Scholar] [CrossRef]
- Aoki, I. Entropy principle for human development, growth and aging. J. Theor. Biol. 1991, 150, 215–223. [Google Scholar] [CrossRef]
- Nieto-Villar, J.M.; Rieumont, J.; Quintana, R.; Miquel, J. Thermodynamic approach to the aging process of biological systems. Rev. CENIC Cienc. Químicas 2003, 34, 149–157. [Google Scholar]
- WHO. Available online: https://www.who.int/health-topics/cancer (accessed on 15 May 2020).
- Montero, S.; Martin, R.; Mansilla, R.; Cocho, G.; Nieto-Villar, J.M. Parameters Estimation in Phase-Space Landscape Reconstruction of Cell Fate: A Systems Biology Approach. In Systems Biology; Humana Press: New York, NY, USA, 2018; pp. 125–170. [Google Scholar]
- Izquierdo-Kulich, E.; Nieto-Villar, J.M. Morphogenesis and complexity of the tumor patterns. In Without Bounds: A Scientific Canvas of Nonlinearity and Complex Dynamics; Springer: Berlin/Heidelberg, 2013; pp. 657–691. [Google Scholar]
- Llanos-Pérez, J.; Betancourt-Mar, J.; Cocho, G.; Mansilla, R.; Nieto-Villar, J.M. Phase transitions in tumor growth: III vascular and metastasis behavior. Phys. A Stat. Mech. Appl. 2016, 462, 560–568. [Google Scholar] [CrossRef]
- Bizzarri, M.; Giuliani, A.; Cucina, A.; D’Anselmi, F.; Soto, A.; Sonnenschein, C. Fractal analysis in a systems biology approach to cancer. Semin. Cancer Biol. 2011, 21, 175–182. [Google Scholar] [CrossRef]
- Gottesman, M.M.; Lavi, O.; Hall, M.D.; Gillet, J.P. Toward a better understanding of the complexity of cancer drug resistance. Annu. Rev. Pharmacol. Toxicol. 2016, 56, 85–102. [Google Scholar] [CrossRef]
- Deisboeck, T.S.; Berens, M.E.; Kansal, A.R.; Torquato, S.; Stemmer-Rachamimov, A.O.; Chiocca, E.A. Pattern of self-organization in tumour systems: Complex growth dynamics in a novel brain tumour spheroid model. Cell Prolif. 2001, 34, 115–134. [Google Scholar] [CrossRef]
- Mansilla, R.; Nieto-Villar, J.M. (Coordinadores). La Termodinámica de los Sistemas Complejos; Centro de Investigaciones Interdisciplinarias en Ciencias y Humanidades: Madrid, Spain, 2017. [Google Scholar]
- Nieto-Villar, J.M.; Betancourt-Mar, J.A.; Izquierdo-Kulich, E.; Tejera, E. Complejidad y Auto-organización en Patrones Naturales; Editorial UH: Havana, Cuba, 2013. [Google Scholar]
- Nieto-Villar, J.M.; Quintana, R.; Rieumont, J. Entropy Production Rate as a Lyapunov Function in Chemical Systems: Proof. Phys. Scr. 2003, 68, 163–165. [Google Scholar] [CrossRef]
- Izquierdo-Kulich, E.; Alonso-Becerra, E.; Nieto-Villar, J.M. Entropy Production Rate for Avascular Tumor Growth. J. Mod. Phys. 2011, 2, 615–620. [Google Scholar] [CrossRef]
- Prigogine, I. Introduction to Thermodynamics of Irreversible Processes, 2nd ed.; Wiley: New York, NY, USA, 1961. [Google Scholar]
- Onsager, L. Reciprocal Relations in Irreversible Processes I. Phys. Rev. 1931, 37, 405–426. [Google Scholar] [CrossRef]
- Onsager, L. Reciprocal Relations in Irre-versible Processes II. Phys. Rev. 1931, 38, 2265–2279. [Google Scholar] [CrossRef]
- De Groot, S.R.; Mazur, P. Non-Equilibrium Thermodynamics; North-Holland Publishing Company: Amsterdam, The Netherlands, 1962. [Google Scholar]
- Prigogine, I. Etude Thermodynamique des Phenomenes irreversibles, Theses d´agregation de l´Enseignement Superieur de l´Universite Libre de Bruxelles, Dunod; Editeurs Paris y Editions Desoer Liege: Paris, France, 1947. [Google Scholar]
- Nieto-Villar, J.M. Una mirada a los sistemas complejos desde la termodinámica. Rev. Mex. Física 2020, 1, 17–24. [Google Scholar] [CrossRef]
- Nicolis, G.; Prigogine, I. Self-Organization in Nonequilibrium Systems; Wiley: New York, NY, USA, 1977. [Google Scholar]
- Bizzarri, M.; Palombo, A.; Cucina, A. Theoretical aspects of systems biology. Prog. Biophys. Mol. Biol. 2013, 112, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Nicolis, G.; Nicolis, C. Foundations of Complex Systems: Emergence, Information and Predicition; World Scientific: Singapore, 2012. [Google Scholar]
- Kuznetsov, Y.A. Elements of Applied Bifurcation Theory; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013; Volume 112. [Google Scholar]
- Quintana González, R.L.; Giraldo Gutiérrez, L.; Moreno Piraján, J.C.; Nieto-Villar, J.M. Termodinámica, Ediciones Uniandes; Universidad de Los Andes: Bogotá, Columbia, 2005. [Google Scholar]
- Nicolis, G.; Daems, D. Probabilistic and thermodynamic aspects of dynamical systems. Chaos Interdiscip. J. Nonlinear Sci. 1998, 8, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Prigogine, I. Introduction to Thermodynamics of Irreversible Processes, 3rd ed.; Interscience: New York, NY, USA, 1968. [Google Scholar]
- Glansdorff, P.; Prigogine, I. On a general evolution criterion in macroscopic physics. Physica 1964, 30, 351–374. [Google Scholar] [CrossRef]
- Andronov, A.A.; Khaikin, S.Ė. Theory of Oscillations; Princeton University Press: Princeton, NJ, USA, 1949. [Google Scholar]
- Betancourt-Mar, J.A.; Mansilla, R.; Cocho, G.; Nieto-Villar, J.M. On the relationship between aging & cancer. MOJ Gerontol. Ger. 2018, 3, 163–168. [Google Scholar]
- Betancourt-Mar, J.A.; Cocho, G.; Mansilla, R.; Nieto-Villar, J.M. What Can Be Learned from A Phase Transitions in Tumor Growth? Insights Biomed. 2017, 2, 1. [Google Scholar]
- Zotin, A.I. Thermodynamic Principles and Reaction of Organisms; Nauka: Moscow, Nauka, 1988. (In Russian) [Google Scholar]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Boothby, W.M.; Berkson, J.; Dunn, H.L. Studies of the energy of metabolism of normal individuals: A standard for basal metabolism, with a nomogram for clinical application. Am. J. Physiol. Content 1936, 116, 468–484. [Google Scholar] [CrossRef]
- Van der Waals, J.D. The equation of state for gases and liquids. Nobel Lect. Phys. 1920, 1, 254–265. [Google Scholar]
- De la Fuente, M. The immune system, a marker and modulator of the rate of aging. In Immunology of Aging; Springer: Berlin/Heidelberg, Germany, 2014; pp. 3–23. [Google Scholar]
- Miquel, J. An integrated theory of aging as the result of mitochondrial-DNA mutation in differentiated cells. Arch. Gerontol. Geriatr. 1991, 12, 99–117. [Google Scholar] [CrossRef]
- Lipsitz, L.A.; Goldberger, A.L. Loss of complexity and aging: Potential applications of fractals and chaos theory to senescence. JAMA 1992, 267, 1806–1809. [Google Scholar] [CrossRef] [PubMed]
- Alvarado, P.; Mansilla, R.; Alvarado, P.E.; Avila, F.M.; Gonzalez, A.; Gonzalez, C. A43. evolving technologies in critical care: The Bioelectric Signal of The Electrocardiogram (EKG), Analyzed In Critically Ill Patients, Using Immersion Takens Theorem. Am. J. Respir. Crit. Care Med. 2015, 191, 1. [Google Scholar]
- Betancourt-Mar, J.A.; Rodríguez-Ricard, M.; Mansilla, R.; Cocho, G.; Nieto-Villar, J.M. Entropy production: Evolution criteria, robustness and fractal dimension. Rev. Mex. Física 2016, 62, 164–167. [Google Scholar]
- Kyriazis, M. Practical applications of chaos theory to the modulation of human aging: Nature prefers chaos to regularity. Biogerontology 2003, 4, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Cutler, R.G. Dysdifferentiative hypothesis of aging: A review. In Molecular Biology of Aging: Gene Stability and Gene Expression; Raven Press: New York, NY, USA, 1985; pp. 307–340. [Google Scholar]
- Kirkham, F.; Mills, C.; Nambier, K.; Timeyin, J.; Davies, K.; Kern, F.; Cruickshank, J.; Rajkumar, C. Are you really as old as your arteries? predicting biological age using cardio-ankle vascular index as a marker of vascular stiffness. J. Hypertens. 2017, 35, e19–e20. [Google Scholar] [CrossRef]
- Hecht, F.; Pessoa, C.F.; Gentile, L.B.; Rosenthal, D.; Carvalho, D.P.; Fortunato, R.S. The role of oxidative stress on breast cancer development and therapy. Tumor Biol. 2016, 37, 4281–4291. [Google Scholar] [CrossRef]
- Seyfried, T.N.; Flores, R.; Poff, A.M.; D’Agostino, D.P. Cancer as a metabolic disease: Implications for novel therapeutics. Carcinogenesis 2014, 35, 515–527. [Google Scholar] [CrossRef]
- Fenninger, L.D.; Mider, G. Energy and Nitrogen Metabolism in Cancer. Adv. Cancer Res. 1954, 2, 229–253. [Google Scholar] [CrossRef]
- Tidwell, T.R.; Søreide, K.; Hagland, H.R. Aging, metabolism, and cancer development: From Peto’s paradox to the War-burg effect. Aging Dis. 2017, 8, 662. [Google Scholar] [CrossRef]
- Finkel, T.; Serrano, M.; Blasco, M.A. The common biology of cancer and ageing. Nature 2007, 448, 767–774. [Google Scholar] [CrossRef]
- Holroyde, C.P.; Gabuzda, T.G.; Putnam, R.C.; Paul, P.; Reichard, G.A. Altered glucose metabolism in metastatic carcinoma. Cancer Res. 1975, 35, 3710–3714. [Google Scholar] [PubMed]
- Izquierdo-Kulich, E.; Rebelo, I.; Tejera, E.; Nieto-Villar, J.M. Phase transition in tumor growth: I avascular development. Phys. A Stat. Mech. Appl. 2013 392, 6616–6623. [CrossRef]
- Llanos-Pérez, J.; Betancourt-Mar, A.; De Miguel, M.; Izquierdo-Kulich, E.; Royuela-García, M.; Tejera, E.; Nieto-Villar, J. Phase transitions in tumor growth: II prostate cancer cell lines. Phys. A Stat. Mech. Appl. 2015, 426, 88–92. [Google Scholar] [CrossRef]
- Warburg, O. On Respiratory Impairment in Cancer Cells. Science 1956, 124, 269–270. [Google Scholar] [CrossRef]
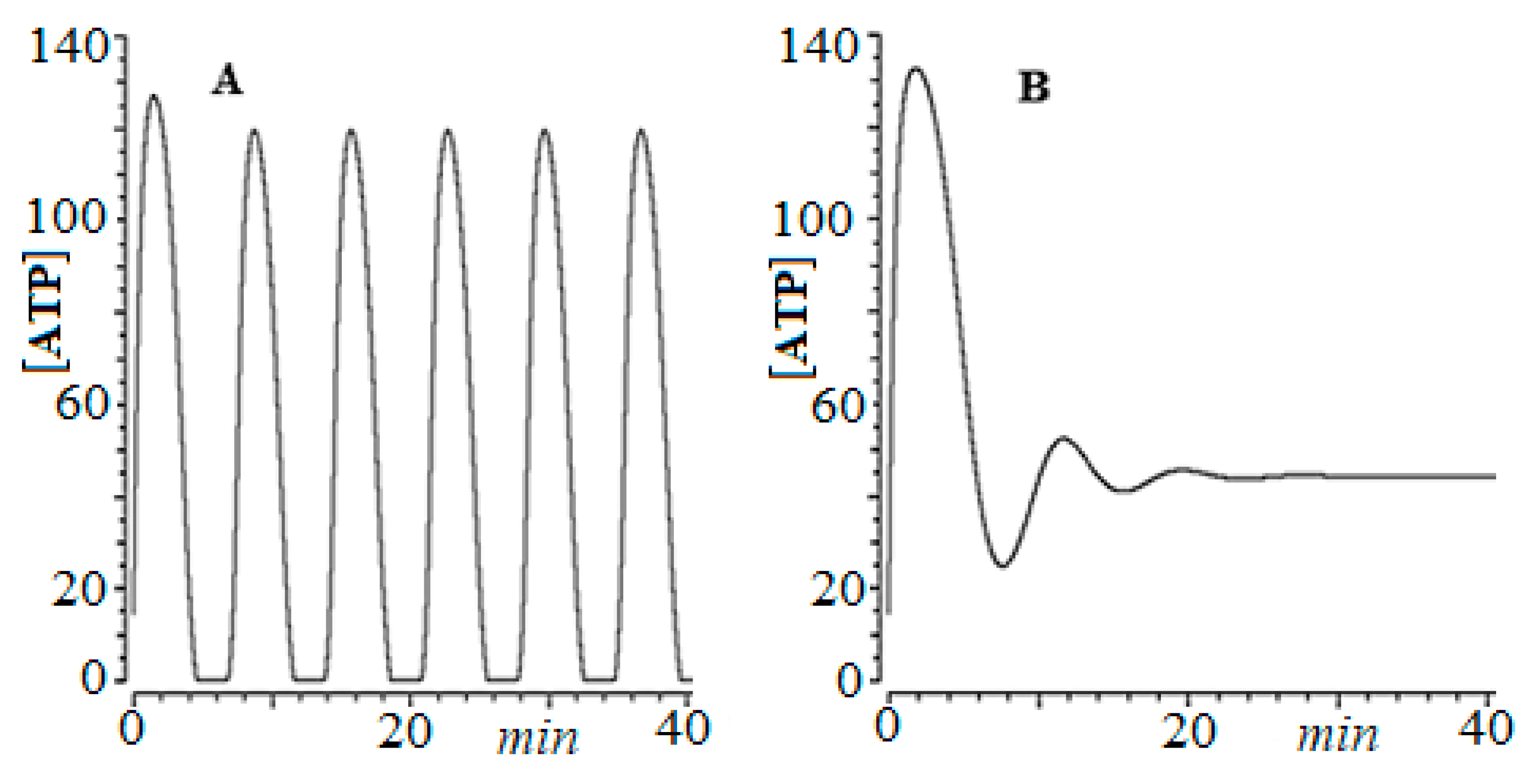
- Martin, R.R.; Montero, S.; Silva, E.; Bizzarri, M.; Cocho, G.; Mansilla, R.; Nieto-Villar, J.M. Phase transitions in tumor growth: V what can be expected from cancer glycolytic oscillations? Phys. A Stat. Mech. Appl. 2017, 486, 762–771. [Google Scholar] [CrossRef]
- Amemiya, T.; Shibata, K.; Itoh, Y.; Itoh, K.; Watanabe, M.; Yamaguchi, T. Primordial oscillations in life: Direct observation of glycolytic oscillations in individual HeLa cervical cancer cells. Chaos Interdiscip. J. Nonlinear Sci. 2017, 27, 104602. [Google Scholar] [CrossRef] [PubMed]
- Soto, A.M.; Maffini, M.V.; Sonnenschein, C. Neoplasia as development gone awry: The role of endocrine disruptors. Int. J. Androl. 2007, 31, 288–293. [Google Scholar] [CrossRef]
- Betancourt-Mar, J.A.; Llanos-Pérez, J.A.; Cocho, G.; Mansilla, R.; Martin, R.; Montero, S.; Nieto-Villar, J.M. Phase transi-tions in tumor growth: IV relationship between metabolic rate and fractal dimension of human tumor cells. Phys. A Stat. Mech. Appl. 2017, 473, 344–351. [Google Scholar] [CrossRef]
- Roose, T.; Chapman, S.; Maini, P.K. Mathematical Models of Avascular Tumor Growth. SIAM Rev. 2007, 49, 179–208. [Google Scholar] [CrossRef]
- Cao, H.; Xu, E.; Liu, H.; Wan, L.; Lai, M. Epithelial–mesenchymal transition in colorectal cancer metastasis: A system review. Pathol. Res. Pract. 2015, 211, 557–569. [Google Scholar] [CrossRef]
- Guerra-González, A.; Silva, E.; Montero, S.; Rodríguez, D.J.; Mansilla, R.; Nieto-Villar, J.M. Metástasis: Un hito para el conocimiento, un reto para la ciencia. Rev. Cuba. Med. 2020, 59, 1–20. [Google Scholar]
- MacLean, A.L.; Harrington, H.A.; Stumpf, M.P.H.; Hansen, M.D.H. Epithelial-Mesenchymal Transition in Metastatic Cancer Cell Populations Affects Tumor Dormancy in a Simple Mathematical Model. Biomedicines 2014, 2, 384–402. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.; Nieto, M.A. Epithelial-mesenchymal transitions in development and disease. Cell 2009, 139, 871–890. [Google Scholar] [CrossRef]
- Zhu, W.; Cai, M.Y.; Tong, Z.T.; Dong, S.S.; Mai, S.J.; Liao, Y.J.; Bian, X.W.; Lin, M.C.; Kung, H.F.; Zeng, Y.X.; et al. Overexpression of EIF5A2 promotes colorectal carcinoma cell aggressiveness by upregulating MTA1 through C-myc to induce epithelial-mesenchymal transition. Gut 2012, 61, 562–575. [Google Scholar] [CrossRef] [PubMed]
- Turner, C.; Kohandel, M. Investigating the link between epithelial-mesenchymal transition and the cancer stem cell phenotype: A mathematical approach. J. Theoret. Biol. 2010, 265, 329–335. [Google Scholar] [CrossRef]
- Magi, S.; Iwamoto, K.; Okada-Hatakeyama, M. Current status of mathematical modeling of cancer—From the viewpoint of cancer hallmarks. Curr. Opin. Syst. Biol. 2017, 2, 39–48. [Google Scholar] [CrossRef]
- Guerra, A.; Rodriguez, D.J.; Montero, S.; Betancourt-Mar, J.A.; Martin, R.R.; Silva, E.; Bizzarri, M.; Cocho, G.; Mansilla, R.; Nieto-Villar, J.M. Phase transitions in tumor growth VI: Epithelial–Mesenchymalmal transition. Phys. A Stat. Mechanics Its Appl. 2018, 499, 208–215. [Google Scholar] [CrossRef]
- Brú, A.; Albertos, S.; Subiza, J.L.; García-Asenjo, J.L.; Brú, I. The Universal Dynamics of Tumor Growth. Biophys. J. 2003, 85, 2948–2961. [Google Scholar] [CrossRef]
- Nasir, N.A. Selected Aspects of Cancer Progression: Metastasis. In Apoptosis and Immune Response; Kaiser, H.E., Nasir, A., Eds.; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2008; Volume 11. [Google Scholar]
- Thiery, J.P.; Sleeman, J.P. Complex networks orchestrate epithelial–mesenchymal transitions. Nat. Rev. Mol. Cell Biol. 2006, 7, 131–142. [Google Scholar] [CrossRef]
- Shilnikov, L. Mathematical Problems of Nonlinear Dynamics: A Tutorial. Int. J. Bifurc. Chaos 1997, 7, 1953–2001. [Google Scholar] [CrossRef]
- Shilnikov, A.L.; Turaev, D.V.; Chua, L.O. Methods of Qualitative Theory in Nonlinear Dynamics; World Scientific: Singapore, 2001. [Google Scholar]
- Tripathi, S.C.; Peters, H.L.; Taguchi, A.; Katayama, H.; Wang, H.; Momin, A.; Jolly, M.K.; Celiktas, M.; Rodriguez-Canales, J.; Liu, H.; et al. Immunoproteasome deficiency is a feature of non-small cell lung cancer with a mesenchymal phenotype and is associated with a poor outcome. Proc. Natl. Acad. Sci. USA 2016, 113, E1555–E1564. [Google Scholar] [CrossRef] [PubMed]
- Betancourt-Mar, J.A.; Nieto-Villar, J.M. Theoretical models for chronotherapy: Periodic perturbations in funnel chaos type. Math. Biosci. Eng. 2007, 4, 177–186. [Google Scholar] [PubMed]
- Kitano, H. Cancer robustness: Tumor tactics. Nature 2003, 426, 125. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lahmy, R.; Riha, C.; Yang, C.; Jakubison, B.L.; van Niekerk, J.; Staub, C.; Wu, Y.; Gates, K.; Dong, D.S.; et al. The Basic Helix-Loop-Helix Transcription Factor E47 Reprograms Human Pancreatic Cancer Cells to a Quies-cent Acinar State with Reduced Tumorigenic Potential. Pancreas 2015, 44, 718–727. [Google Scholar] [CrossRef]
- Volkenstein, M.V. Entropy and Information; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2009; Volume 57. [Google Scholar]
- Enderling, H.; Almog, N.; Hlatky, L. Systems Biology of Tumor Dormancy; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Pantel, K.; Alix-Panabiéres, C.; Riethdorf, S. Cancer micrometastasis. Nat. Rev. Clin. Oncol. 2009, 6, 339–351. [Google Scholar] [CrossRef] [PubMed]
- Bizzarri, M.; Cucina, A.; Proietti, S. Tumor reversion: Mesenchymal-epithelial transition as a critical step in managing the tumor microenvironment cross-talk. Curr. Pharm. Des. 2017, 23, 4705–4715. [Google Scholar] [CrossRef] [PubMed]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An Iron-Dependent Form of Nonapoptotic Cell Death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef]
- Schulz, W. Molecular Biology of Human Cancers; Springer Science: Berlin/Heidelberg, Germany, 2005. [Google Scholar]
- Lane, D.; Levine, A. p53 Research: The past thirty years and the next thirty years. Cold Spring Harb. Perspect Biol. 2010, 2, a000893. [Google Scholar] [CrossRef]
- Dixon, S.J.; Stockwell, B.R. The Hallmarks of Ferroptosis. Annu. Rev. Cancer Biol. 2019, 3, 35–54. [Google Scholar] [CrossRef]
- Feng, H.; Stockwell, B.R. Unsolved mysteries: How does lipid peroxidation cause ferroptosis? PLoS Biol. 2018, 16, e2006203. [Google Scholar] [CrossRef]
- Yan, H.F. Ferroptosis: Mechanisms and links with diseases. Signal Transduct. Target. Ther. 2021, 6, 49. [Google Scholar] [CrossRef]
- Shen, Z.; Song, J.; Yung, B.C. Emerging strategies of cancer therapy based on ferroptosis. Adv. Mater. 2018, 30, 1704007. [Google Scholar] [CrossRef] [PubMed]
- Shimada, K.; Skouta, R.; Kaplan, A.; Yang, W.S.; Hayano, M.; Dixon, S.; Brown, L.M.; Valenzuela, C.A.; Wolpaw, A.; Stockwell, B.R. Global survey of cell death mechanisms reveals metabolic regulation of ferroptosis. Nat. Chem. Biol. 2016, 12, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.E.; Zhang, L.; Ma, K.; Riegman, M.; Chen, F.; Ingold, I.; Conrad, M.; Turker, M.Z.; Gao, M.; Jiang, X.; et al. Ultrasmall nanoparticles induce ferroptosis in nutrient-deprived cancer cells and suppress tumor growth. Nat. Nanotechnol. 2016, 11, 977–985. [Google Scholar] [CrossRef]
- Kagan, V.E.; Mao, G.; Qu, F.; Angeli, J.P.F.; Doll, S.; Croix, C.S.; Dar, H.H.; Liu, B.; Tyurin, V.A.; Ritov, V.B.; et al. Oxidized Arachidonic and Adrenic PEs Navigate Cells to Ferroptosis. Nat. Chem. Biol. 2017, 13, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Agmon, E.; Solon, J.; Bassereau, P.; Stockwell, B.R. Modeling the effects of lipid peroxidation during ferroptosis on mem-brane properties. Sci. Rep. 2018, 8, e5155. [Google Scholar] [CrossRef]
- Konstorum, A.; Tesfay, L.; Paul, B.T.; Torti, F.M.; Laubenbacher, R.C.; Torti, S.V. Systems biology of ferroptosis: A modeling approach. J. Theor. Biol. 2020, 493, 110222. [Google Scholar] [CrossRef]
- Dierge, E.; Debock, E.; Guilbaud, C.; Corbet, C.; Mignolet, E.; Mignard, L.; Bastien, E.; Dessy, C.; Larondelle, Y.; Feron, O. Peroxidation of n-3 and n-6 polyunsaturated fatty acids in the acidic tumor environment leads to ferropto-sis-mediated anticancer effects. Cell Metab. 2021, 33, 1–15. [Google Scholar] [CrossRef]
- Hassannia, B.; Vandenabeele, P.; Berghe, T.V. Targeting Ferroptosis to Iron Out Cancer. Cancer Cell 2019, 35, 830–849. [Google Scholar] [CrossRef]
- Nieto-Villar, J.M.; Mansilla, R. Ferroptosis as a Biological Phase Transition I: Avascular and vascular tumor growth, European. J. Biomed. Pharm. Sci. 2021, 8, 63–70. [Google Scholar]
- Wang, S.; Liao, H.; Li, F.; Ling, D. A mini-review and perspective on ferroptosis-inducing strategies in cancer therapy. Chin. Chem. Lett. 2019, 30, 847–852. [Google Scholar] [CrossRef]
- Lei, G.; Zhang, Y.; Koppula, P.; Liu, X.; Zhang, J.; Lin, S.H.; Ajani, J.A.; Xiao, Q.; Liao, Z.; Wang, H.; et al. The role of ferroptosis in ionizing radiation-induced cell death and tumor suppression. Cell Res. 2020, 30, 146–162. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Li, Y. What Is Responsible for the Initiating Chemistry of Iron-Mediated Lipid Peroxidation: An Update. Chem. Rev. 2007, 107, 748–766. [Google Scholar] [CrossRef] [PubMed]
- Bebber, C.M.; Müller, F.; Prieto Clemente, L.; Weber, J.; Von Karstedt, S. Ferroptosis in Cancer Cell Biology. Cancers 2020, 12, 164. [Google Scholar] [CrossRef] [PubMed]
- Spiteller, G.; Afzal, M. The Action of Peroxyl Radicals, Powerful Deleterious Reagents, Explains Why Neither Cholesterol Nor Saturated Fatty Acids Cause Atherogenesis and Age-Related Diseases. Chem.-A Eur. J. 2014, 20, 14928–14945. [Google Scholar] [CrossRef]
- Dreger, H.; Westphal, K.; Weller, A.; Baumann, G.; Stangl, V.; Meiners, S.; Stangl, K. Nrf2-dependent upregulation of antioxidative enzymes: A novel pathway for proteasome inhibitor-mediated cardioprotection. Cardiovasc. Res. 2003, 83, 354–361. [Google Scholar] [CrossRef]
- Molnar, J.; Thornton, B.; Molnar, A.; Gaal, D.; Luo, L.; Bergmann-Leitner, E. Thermodynamic Aspects of Cancer: Possible Role of Negative Entropy in Tumor Growth, its Relation to Kinetic and Genetic Resistance. Lett. Drug Des. Discov. 2005, 2, 429–438. [Google Scholar] [CrossRef]
- Lucia, U.; Ponzetto, A.; Deisboeck, T.S. A thermodynamic approach to the ‘mitosis/apoptosis’ ratio in cancer. Phys. A Stat. Mech. Its Appl. 2015, 436, 246–255. [Google Scholar] [CrossRef]
- Lucia, U.; Grisolia, G. Second law efficiency for living cells. Front. Biosci. 2017, 9, 270–275. [Google Scholar] [CrossRef]
- Marin, D.; Sabater, B. The cancer Warburg effect may be a testable example of the minimum entropy production rate principle. Phys. Biol. 2017, 14, 024001. [Google Scholar] [CrossRef]
- Montemayor-Aldrete, J.A.; Márquez-Caballé, R.F.; del Castillo-Mussot, M.; Cruz-Peregrino, F. General Thermodynamic Efficiency Loss and Scaling Behavior of Eukaryotic Organisms. Biophys. Rev. Lett. 2020, 15, 143–169. [Google Scholar] [CrossRef]
- Luo, L.-F. Entropy production in a cell and reversal of entropy flow as an anticancer therapy. Front. Phys. China 2009, 4, 122–136. [Google Scholar] [CrossRef]
- Lucia, U. Entropy generation and cell growth with comments for a thermodynamic anticancer approach. Phys. A Stat. Mech. Appl. 2014, 406, 107–118. [Google Scholar] [CrossRef]
- Villar, J.N.; Guerra, A.; Rodriguez, D.; Silva, E.; Mar, J.B.; Cocho, G.; Mansilla, R. Chronotherapy of cancer: Epithelial-mesenchymal transition. MOJ Gerontol. Geriatr. 2019, 4, 124–127. [Google Scholar] [CrossRef]
- Sourailidis, D.; Volos, C.; Moysis, L.; Meletlidou, E.; Stouboulos, I. The Study of Square Periodic Perturbations as an Immunotherapy Process on a Tumor Growth Chaotic Model. Dynamics 2022, 2, 8. [Google Scholar] [CrossRef]
- Jaime, J.C.; Mesa-Álvarez, M.D.; Martin, R.R.; Betancourt-Mar, J.A.; Cocho, G.; Mansilla, R.; Nieto-Villar, J.M. Chronothera-py of cancer: Periodic perturbations in vascular growth and metastasis. Biol. Rhythm Res. 2018, 50, 495–504. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nieto-Villar, J.M.; Mansilla, R. Longevity, Aging and Cancer: Thermodynamics and Complexity. Foundations 2022, 2, 664-680. https://doi.org/10.3390/foundations2030045
Nieto-Villar JM, Mansilla R. Longevity, Aging and Cancer: Thermodynamics and Complexity. Foundations. 2022; 2(3):664-680. https://doi.org/10.3390/foundations2030045
Chicago/Turabian StyleNieto-Villar, J. M., and R. Mansilla. 2022. "Longevity, Aging and Cancer: Thermodynamics and Complexity" Foundations 2, no. 3: 664-680. https://doi.org/10.3390/foundations2030045
APA StyleNieto-Villar, J. M., & Mansilla, R. (2022). Longevity, Aging and Cancer: Thermodynamics and Complexity. Foundations, 2(3), 664-680. https://doi.org/10.3390/foundations2030045






