Association between Malnutrition Risk Factors and Physical Function in Community-Dwelling Adults ≥80 Years
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
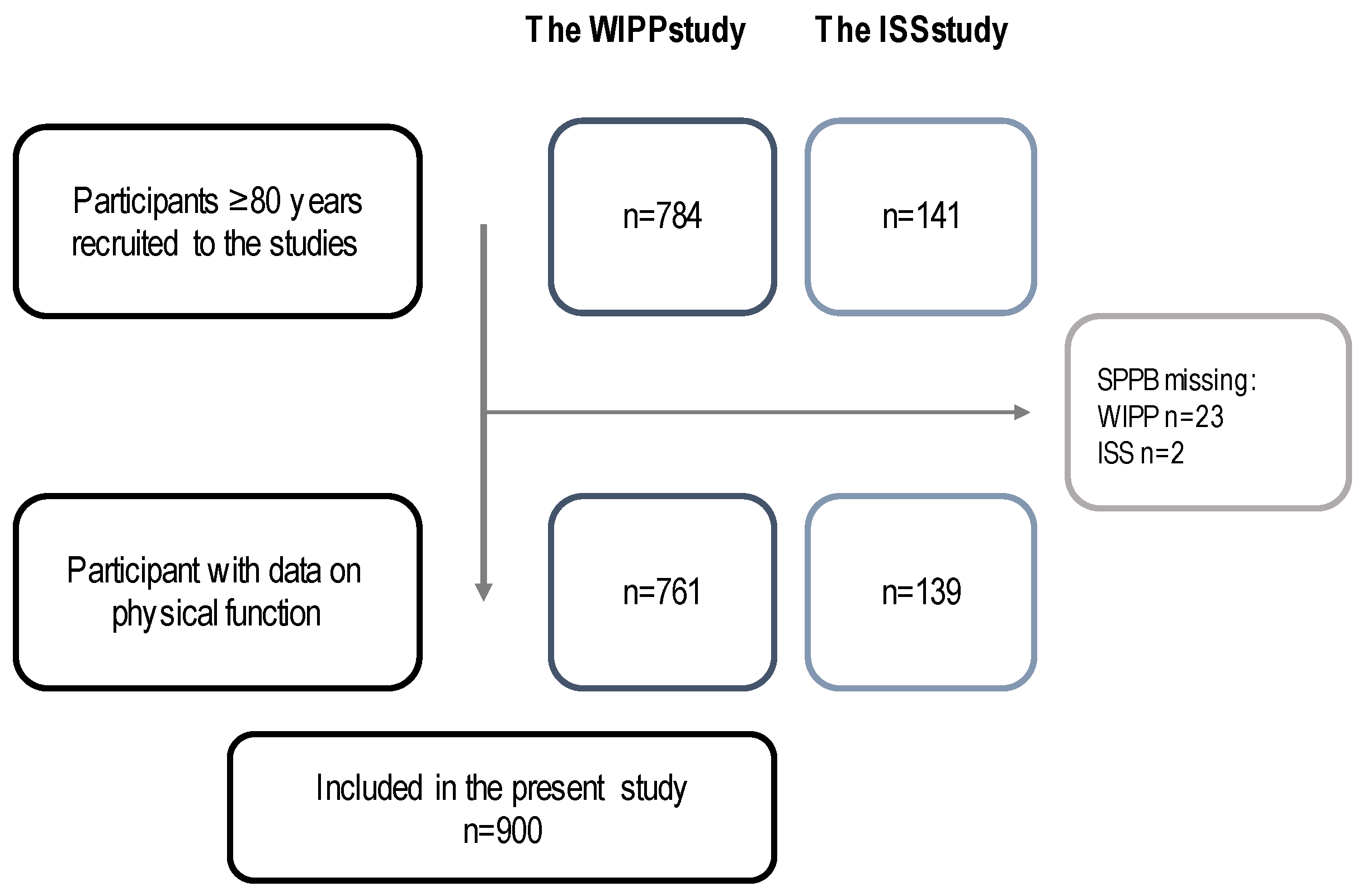
2.2. Study Population
2.3. Data Collection
2.3.1. Reduced Physical Function
2.3.2. Poor Appetite
2.3.3. Dysphagia
2.3.4. Poor Dental State
2.3.5. Descriptive Characteristics
2.3.6. Statistical Methods
3. Results
3.1. Prevalence of Malnutrition Risk Factors
3.2. Associations between Low Physical Function and Malnutrition Risk Factors
3.3. Sensitivity Analysis
4. Discussion
4.1. Strengths
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eurostat. Population Structure and Ageing. Statistics Explained. 2022. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php?title=Population_structure_and_ageing#The_share_of_elderly_people_continues_to_increase (accessed on 7 October 2022).
- Portela, D.; Almada, M.; Midão, L.; Costa, E. Instrumental Activities of Daily Living (iADL) Limitations in Europe: An Assessment of SHARE Data. Int. J. Environ. Res. Public Health 2020, 17, 7387. [Google Scholar] [CrossRef] [PubMed]
- Wahrendorf, M.; Reinhardt, J.; Siegrist, J. Relationships of disability with age among adults aged 50 to 85: Evidence from the United States, England and continental europe. PLoS ONE 2013, 8, e71893. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eurostat. Disability Statistics-Elderly Needs for Help or Assistance. Statistics Explained. 2022. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php?oldid=456538#Need_for_help_with_personal_care_or_household_activities (accessed on 28 November 2022).
- Commission, E. Economic and budgetary projections for the 28 EU Member States (2013–2060). In European Economy; The 2015 Ageing Report; European Commission: Brussels, Belgium, 2015. [Google Scholar]
- Cederholm, T.; Barazzoni, R.; Austin, P.; Ballmer, P.; Biolo, G.; Bischoff, S.C.; Compher, C.; Correia, I.; Higashiguchi, T.; Holst, M.; et al. ESPEN guidelines on definitions and terminology of clinical nutrition. Clin. Nutr. 2017, 36, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Deutz, N.E.; Bauer, J.M.; Barazzoni, R.; Biolo, G.; Boirie, Y.; Bosy-Westphal, A.; Cederholm, T.; Cruz-Jentoft, A.; Krznariç, Z.; Nair, K.S.; et al. Protein intake and exercise for optimal muscle function with aging: Recommendations from the ESPEN Expert Group. Clin. Nutr. 2014, 33, 929–936. [Google Scholar] [CrossRef] [Green Version]
- Laur, C.V.; McNicholl, T.; Valaitis, R.; Keller, H.H. Malnutrition or frailty? Overlap and evidence gaps in the diagnosis and treatment of frailty and malnutrition. Appl. Physiol. Nutr. Metab. 2017, 42, 449–458. [Google Scholar] [CrossRef] [Green Version]
- Agarwal, E.; Miller, M.; Yaxley, A.; Isenring, E. Malnutrition in the elderly: A narrative review. Maturitas 2013, 76, 296–302. [Google Scholar] [CrossRef] [Green Version]
- Morley, J.E.; Vellas, B.; van Kan, G.A.; Anker, S.D.; Bauer, J.M.; Bernabei, R.; Cesari, M.; Chumlea, W.C.; Doehner, W.; Evans, J.; et al. Frailty consensus: A call to action. J. Am. Med. Dir. Assoc. 2013, 14, 392–397. [Google Scholar] [CrossRef] [Green Version]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef]
- Sieber, C.C. Malnutrition and sarcopenia. Aging Clin. Exp. Res. 2019, 31, 793–798. [Google Scholar] [CrossRef]
- Donini, L.M.; Busetto, L.; Bischoff, S.C.; Cederholm, T.; Ballesteros-Pomar, M.D.; Batsis, J.A.; Bauer, J.M.; Boirie, Y.; Cruz-Jentoft, A.J.; Dicker, D.; et al. Definition and Diagnostic Criteria for Sarcopenic Obesity: ESPEN and EASO Consensus Statement. Obes. Facts 2022, 15, 321–335. [Google Scholar] [CrossRef]
- Leij-Halfwerk, S.; Verwijs, M.H.; van Houdt, S.; Borkent, J.W.; Guaitoli, P.R.; Pelgrim, T.; Heymans, M.W.; Power, L.; Visser, M.; Corish, C.A.; et al. Prevalence of protein-energy malnutrition risk in European older adults in community, residential and hospital settings, according to 22 malnutrition screening tools validated for use in adults ≥65 years: A systematic review and meta-analysis. Maturitas 2019, 126, 80–89. [Google Scholar] [CrossRef] [PubMed]
- O’Keeffe, M.; Kelly, M.; O’Herlihy, E.; O’Toole, P.W.; Kearney, P.M.; Timmons, S.; O’Shea, E.; Stanton, C.; Hickson, M.; Rolland, Y.; et al. Potentially modifiable determinants of malnutrition in older adults: A systematic review. Clin. Nutr. 2019, 38, 2477–2498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Volkert, D.; Kiesswetter, E.; Cederholm, T.; Donini, L.M.; Eglseer, D.; Norman, K.; Schneider, S.M.; Ströbele-Benschop, N.; Torbahn, G.; Wirth, R.; et al. Development of a Model on Determinants of Malnutrition in Aged Persons: A MaNuEL Project. Gerontol. Geriatr. Med. 2019, 5, 2333721419858438. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, T.; Jensen, G.L.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. Clin. Nutr. 2019, 38, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Authority, D.H. Underernæring: Opsporing, Behandling og Opfølgning af Borgere og Patienter i Ernæringsrisiko. Vejledning Til Kommune, Sygehus og Almen Praksis; Danish Health Authority: Copenhagen, Denmark, 2022; pp. 1–152.
- Buhl, S.F.; Beck, A.M.; Christensen, B.; Kock, G.; Boyle, E.; Caserotti, P. Prevalence of low protein intake in 80+-year-old community-dwelling adults and association with dietary patterns and modifiable risk factors: A cross-sectional study. Br. J. Nutr. 2022, 127, 266–277. [Google Scholar] [CrossRef]
- Landi, F.; Calvani, R.; Tosato, M.; Martone, A.M.; Ortolani, E.; Savera, G.; Sisto, A.; Marzetti, E. Anorexia of Aging: Risk Factors, Consequences, and Potential Treatments. Nutrients 2016, 8, 69. [Google Scholar] [CrossRef]
- Melgaard, D.; Rodrigo-Domingo, M.; Mørch, M.M. The Prevalence of Oropharyngeal Dysphagia in Acute Geriatric Patients. Geriatrics 2018, 3, 15. [Google Scholar] [CrossRef] [Green Version]
- Baijens, L.W.; Clavé, P.; Cras, P.; Ekberg, O.; Forster, A.; Kolb, G.F.; Leners, J.C.; Masiero, S.; Mateos-Nozal, J.; Ortega, O.; et al. European Society for Swallowing Disorders-European Union Geriatric Medicine Society white paper: Oropharyngeal dysphagia as a geriatric syndrome. Clin. Interv. Aging 2016, 11, 1403–1428. [Google Scholar] [CrossRef] [Green Version]
- Woo, J.; Tong, C.; Yu, R. Chewing Difficulty Should be Included as a Geriatric Syndrome. Nutrients 2018, 10, 1997. [Google Scholar] [CrossRef] [Green Version]
- Iwasaki, M.; Yoshihara, A.; Sato, M.; Minagawa, K.; Shimada, M.; Nishimuta, M.; Ansai, T.; Yoshitake, Y.; Miyazaki, H. Dentition status and frailty in community-dwelling older adults: A 5-year prospective cohort study. Geriatr. Gerontol. Int. 2018, 18, 256–262. [Google Scholar] [CrossRef]
- Buhl, S.F.; Beck, A.M.; Olsen, P.; Kock, G.; Christensen, B.; Wegner, M.; Vaarst, J.; Caserotti, P. Relationship between physical frailty, nutritional risk factors and protein intake in community-dwelling older adults. Clin. Nutr. ESPEN 2022, 49, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Vandenbroucke, J.P.; von Elm, E.; Altman, D.G.; Gøtzsche, P.C.; Mulrow, C.D.; Pocock, S.J.; Poole, C.; Schlesselman, J.J.; Egger, M. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and elaboration. PLoS Med. 2007, 4, e297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef] [PubMed]
- Freiberger, E.; de Vreede, P.; Schoene, D.; Rydwik, E.; Mueller, V.; Frändin, K.; Hopman-Rock, M. Performance-based physical function in older community-dwelling persons: A systematic review of instruments. Age Ageing 2012, 41, 712–721. [Google Scholar] [CrossRef] [Green Version]
- Guralnik, J.M.; Ferrucci, L.; Pieper, C.F.; Leveille, S.G.; Markides, K.S.; Ostir, G.V.; Studenski, S.; Berkman, L.F.; Wallace, R.B. Lower extremity function and subsequent disability: Consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J. Gerontol. A Biol. Sci. Med. Sci. 2000, 55, M221–M231. [Google Scholar] [CrossRef] [Green Version]
- Vasunilashorn, S.; Coppin, A.K.; Patel, K.V.; Lauretani, F.; Ferrucci, L.; Bandinelli, S.; Guralnik, J.M. Use of the Short Physical Performance Battery Score to predict loss of ability to walk 400 meters: Analysis from the InCHIANTI study. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 223–229. [Google Scholar] [CrossRef]
- Vaarst, J.; Boyle, E.; Vestergaard, S.; Hvid, L.G.; Strotmeyer, E.S.; Glynn, N.W.; Caserotti, P. Does physical performance and muscle strength predict future personal and nursing care services in community-dwelling older adults aged 75+? Scand. J. Public Health 2021, 49, 441–448. [Google Scholar] [CrossRef]
- Romero-Ortuno, R.; Soraghan, C. A Frailty Instrument for primary care for those aged 75 years or more: Findings from the Survey of Health, Ageing and Retirement in Europe, a longitudinal population-based cohort study (SHARE-FI75+). BMJ Open 2014, 4, e006645. [Google Scholar] [CrossRef] [Green Version]
- Wijnhoven, H.A.H.; Elstgeest, L.E.M.; de Vet, H.C.W.; Nicolaou, M.; Snijder, M.B.; Visser, M. Development and validation of a short food questionnaire to screen for low protein intake in community-dwelling older adults: The Protein Screener 55+ (Pro55+). PLoS ONE 2018, 13, e0196406. [Google Scholar] [CrossRef] [Green Version]
- Berner, L.A.; Becker, G.; Wise, M.; Doi, J. Characterization of dietary protein among older adults in the United States: Amount, animal sources, and meal patterns. J. Acad. Nutr. Diet. 2013, 113, 809–815. [Google Scholar] [CrossRef]
- Beck, A.M.; Beermann, T.; Kjær, S.; Rasmussen, H.H. Ability of different screening tools to predict positive effect on nutritional intervention among the elderly in primary health care. Nutrition 2013, 29, 993–999. [Google Scholar] [CrossRef] [PubMed]
- Vellas, B.; Guigoz, Y.; Garry, P.J.; Nourhashemi, F.; Bennahum, D.; Lauque, S.; Albarede, J.L. The Mini Nutritional Assessment (MNA) and its use in grading the nutritional state of elderly patients. Nutrition 1999, 15, 116–122. [Google Scholar] [CrossRef] [PubMed]
- EuroQol--a new facility for the measurement of health-related quality of life. Health Policy 1990, 16, 199–208. [CrossRef]
- Rabin, R.; Gudex, C.; Selai, C.; Herdman, M. From translation to version management: A history and review of methods for the cultural adaptation of the EuroQol five-dimensional questionnaire. Value Health 2014, 17, 70–76. [Google Scholar] [CrossRef] [Green Version]
- Launer, L.J.; Harris, T.; Rumpel, C.; Madans, J. Body mass index, weight change, and risk of mobility disability in middle-aged and older women. The epidemiologic follow-up study of NHANES I. JAMA 1994, 271, 1093–1098. [Google Scholar] [CrossRef]
- Imeraj, A.; Olesen, T.B.; Laursen, D.H.; Søndergaard, J.; Brandt, C.J. Agreement Between Clinically Measured Weight and Self-reported Weight Among Patients With Type 2 Diabetes Through an mHealth Lifestyle Coaching Program in Denmark: Secondary Analysis of a Randomized Controlled Trial. JMIR Form. Res. 2022, 6, e40739. [Google Scholar] [CrossRef] [PubMed]
- Dahl, A.K.; Hassing, L.B.; Fransson, E.I.; Pedersen, N.L. Agreement between self-reported and measured height, weight and body mass index in old age--a longitudinal study with 20 years of follow-up. Age Ageing 2010, 39, 445–451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alibhai, S.M.; Greenwood, C.; Payette, H. An approach to the management of unintentional weight loss in elderly people. CMAJ 2005, 172, 773–780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jadczak, A.D.; Visvanathan, R. Anorexia of Aging—An Updated Short Review. J. Nutr. Health Aging 2019, 23, 306–309. [Google Scholar] [CrossRef]
- van der Pols-Vijlbrief, R.; Wijnhoven, H.A.; Schaap, L.A.; Terwee, C.B.; Visser, M. Determinants of protein-energy malnutrition in community-dwelling older adults: A systematic review of observational studies. Ageing Res. Rev. 2014, 18, 112–131. [Google Scholar] [CrossRef]
- İlhan, B.; Bahat, G.; Erdoğan, T.; Kılıç, C.; Karan, M.A. Anorexia Is Independently Associated with Decreased Muscle Mass and Strength in Community Dwelling Older Adults. J. Nutr. Health Aging 2019, 23, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Martone, A.M.; Onder, G.; Vetrano, D.L.; Ortolani, E.; Tosato, M.; Marzetti, E.; Landi, F. Anorexia of aging: A modifiable risk factor for frailty. Nutrients 2013, 5, 4126–4133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landi, F.; Liperoti, R.; Russo, A.; Giovannini, S.; Tosato, M.; Barillaro, C.; Capoluongo, E.; Bernabei, R.; Onder, G. Association of anorexia with sarcopenia in a community-dwelling elderly population: Results from the ilSIRENTE study. Eur. J. Nutr. 2013, 52, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
- Clegg, M.E.; Godfrey, A. The relationship between physical activity, appetite and energy intake in older adults: A systematic review. Appetite 2018, 128, 145–151. [Google Scholar] [CrossRef]
- Giné-Garriga, M.; Sansano-Nadal, O.; Tully, M.A.; Caserotti, P.; Coll-Planas, L.; Rothenbacher, D.; Dallmeier, D.; Denkinger, M.; Wilson, J.J.; Martin-Borràs, C.; et al. Accelerometer-Measured Sedentary and Physical Activity Time and Their Correlates in European Older Adults: The SITLESS Study. J. Gerontol. A Biol. Sci. Med. Sci. 2020, 75, 1754–1762. [Google Scholar] [CrossRef] [Green Version]
- Honnens de Lichtenberg Broge, E.; Wendin, K.; Rasmussen, M.A.; Bredie, W.L.P. Changes in perception and liking for everyday food odors among older adults. Food Qual. Prefer. 2021, 93, 104254. [Google Scholar] [CrossRef]
- Fluitman, K.S.; Hesp, A.C.; Kaihatu, R.F.; Nieuwdorp, M.; Keijser, B.J.F.; RG, I.J.; Visser, M. Poor Taste and Smell Are Associated with Poor Appetite, Macronutrient Intake, and Dietary Quality but Not with Undernutrition in Older Adults. J. Nutr. 2021, 151, 605–614. [Google Scholar] [CrossRef]
- van der Meij, B.S.; Wijnhoven, H.A.H.; Lee, J.S.; Houston, D.K.; Hue, T.; Harris, T.B.; Kritchevsky, S.B.; Newman, A.B.; Visser, M. Poor Appetite and Dietary Intake in Community-Dwelling Older Adults. J. Am. Geriatr. Soc 2017, 65, 2190–2197. [Google Scholar] [CrossRef] [Green Version]
- Yang, R.Y.; Yang, A.Y.; Chen, Y.C.; Lee, S.D.; Lee, S.H.; Chen, J.W. Association between Dysphagia and Frailty in Older Adults: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 1812. [Google Scholar] [CrossRef]
- Madhavan, A.; LaGorio, L.A.; Crary, M.A.; Dahl, W.J.; Carnaby, G.D. Prevalence of and Risk Factors for Dysphagia in the Community Dwelling Elderly: A Systematic Review. J. Nutr. Health Aging 2016, 20, 806–815. [Google Scholar] [CrossRef]
- González-Fernández, M.; Humbert, I.; Winegrad, H.; Cappola, A.R.; Fried, L.P. Dysphagia in old-old women: Prevalence as determined according to self-report and the 3-ounce water swallowing test. J. Am. Geriatr. Soc. 2014, 62, 716–720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strömberg, E.; Hagman-Gustafsson, M.L.; Holmén, A.; Wårdh, I.; Gabre, P. Oral status, oral hygiene habits and caries risk factors in home-dwelling elderly dependent on moderate or substantial supportive care for daily living. Community Dent. Oral Epidemiol. 2012, 40, 221–229. [Google Scholar] [CrossRef] [PubMed]
- BERKHOUT, A.M.M.; COOLS, H.J.M.; VAN HOUWELINGEN, H.C. The relationship between difficulties in feeding oneself and loss of weight in nursing-home patients with dementia. Age Ageing 1998, 27, 637–641. [Google Scholar] [CrossRef] [Green Version]
- Sullivan, D.H.; Martin, W.; Flaxman, N.; Hagen, J.E. Oral health problems and involuntary weight loss in a population of frail elderly. J. Am. Geriatr. Soc. 1993, 41, 725–731. [Google Scholar] [CrossRef]
- Ástvaldsdóttir, Á.; Boström, A.M.; Davidson, T.; Gabre, P.; Gahnberg, L.; Sandborgh Englund, G.; Skott, P.; Ståhlnacke, K.; Tranaeus, S.; Wilhelmsson, H.; et al. Oral health and dental care of older persons-A systematic map of systematic reviews. Gerodontology 2018, 35, 290–304. [Google Scholar] [CrossRef]
- Yamaga, T.; Yoshihara, A.; Ando, Y.; Yoshitake, Y.; Kimura, Y.; Shimada, M.; Nishimuta, M.; Miyazaki, H. Relationship Between Dental Occlusion and Physical Fitness in an Elderly Population. J. Gerontol. Ser. A 2002, 57, M616–M620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andersen, A.L.; Nielsen, R.L.; Houlind, M.B.; Tavenier, J.; Rasmussen, L.J.H.; Jørgensen, L.M.; Treldal, C.; Beck, A.M.; Pedersen, M.M.; Andersen, O.; et al. Risk of Malnutrition upon Admission and after Discharge in Acutely Admitted Older Medical Patients: A Prospective Observational Study. Nutrients 2021, 13, 2757. [Google Scholar] [CrossRef]
- Gawel, J.; Vengrow, D.; Collins, J.; Brown, S.; Buchanan, A.; Cook, C. The short physical performance battery as a predictor for long term disability or institutionalization in the community dwelling population aged 65 years old or older. Phys. Ther. Rev. 2012, 17, 37–44. [Google Scholar] [CrossRef]
| Variables | All | n | Normal Physical Function * | n | Reduced Physical Function ** | n | p-Value |
|---|---|---|---|---|---|---|---|
| Age, years | 85.1 ± 3.7 | 900 | 84.4 ± 3.3 | 489 | 85.9 ± 4.0 | 411 | p < 0.001 |
| Females, n (%) | 548 (60.9%) | 900 | 290 (59.3%) | 489 | 258 (62.8%) | 411 | p = 0.288 |
| Physical function | 9.1 ± 2.7 | 900 | 11.1 ± 0.8 | 489 | 6.7 ± 2.3 | 411 | p < 0.001 |
| BMI, kg/m2 | 25.9 ± 4.1 | 850 | 25.5 ± 3.8 | 470 | 26.3 ± 4.4 | 380 | p = 0.006 |
| BMI categories, n (%) | 850 | 470 | 380 | p = 0.002 | |||
| Low | 151 (17.8%) | 93 (19.8%) | 58 (15.3%) | ||||
| Normal | 401 (47.2%) | 236 (50.2%) | 165 (43.4%) | ||||
| High | 298 (35.1%) | 141 (30%) | 157 (41.3%) | ||||
| Unintentional weight loss, n (%) | 56 (6.2%) | 897 | 27 (5.5%) | 489 | 29 (7.1%) | 408 | p = 0.328 |
| Chronic conditions, n (%) | 875 | 480 | 395 | p = 0.295 | |||
| None | 403 (46.1%) | 229 (47.7%) | 174 (44%) | ||||
| One | 210 (24%) | 120 (25%) | 90 (22.8%) | ||||
| Two | 123 (14.1%) | 63 (13.1%) | 60 (15.2%) | ||||
| Three or more | 139 (15.9%) | 68 (14.2%) | 71 (18%) | ||||
| Recent illness | 68 (7.6%) | 896 | 36 (7.4%) | 488 | 32 (7.8%) | 408 | p = 0.529 |
| Polypharmacy | 323 (36%) | 897 | 160 (32.8%) | 488 | 163 (39.9%) | 409 | p = 0.028 |
| Quality of life, VAS | 76 (60–90) | 873 | 80 (70–90) | 481 | 70 (50–80) | 392 | p < 0.001 |
| Fall during the last year, n (%) | 262 (29.3%) | 895 | 122 (25%) | 488 | 140 (34.4%) | 407 | p = 0.002 |
| All | n | Normal Physical Function * | n | Reduced Physical Function ** | n | p-Value | |
|---|---|---|---|---|---|---|---|
| Poor appetite | 97 (10.8%) | 899 | 36 (7.4%) | 489 | 61 (14.9%) | 410 | p < 0.001 |
| Dysphagia | 53 (6.3%) | 843 | 28 (6.1%) | 457 | 25 (6.5%) | 386 | p = 0.835 |
| Poor dental state | 27 (3.2%) | 846 | 13 (2.8%) | 459 | 14 (3.6%) | 387 | p = 0.517 |
| No risk factors | 694 (82.3%) | 843 | 388 (84.9%) | 457 | 306 (79.3%) | 386 | p = 0.034 |
| Any risk factor | 132 (15.7%) | 64 (14.0%) | 68 (17.6%) | ||||
| Two or more risk factors | 17 (2.0%) | 5 (1%) | 12 (3.1%) |
| Logistic Regression Analyses | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Reduced Physical Function # (Ref. Normal Physical Function ##) | Model I * | Model II ** | Model III *** | ||||||
| n = 843 | OR (95%CI) | p-Value | n = 843 | OR (95%CI) | p-Value | n = 789 | OR (95%CI) | p-Value | |
| Poor appetite | 2.01 (1.27–3.16) | 0.003 | 1.80 (1.13–2.86) | 0.014 | 1.93 (1.18–3.15) | 0.008 | |||
| Dysphagia | 1.01 (0.58–1.78) | 0.964 | 1.01 (0.57–1.80) | 0.971 | 0.96 (0.53–1.75) | 0.900 | |||
| Poor dental state | 1.15 (0.53–2.52) | 0.723 | 1.05 (0.47–2.37) | 0.898 | 0.99 (0.41–2.35) | 0.978 | |||
| Logistic Regression Analyses | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Reduced Physical Function # (ref. Normal physical function ##) | Model I * | Model II ** | Model III *** | ||||||
| n = 792 | OR (95%CI) | p-Value | n = 792 | OR (95%CI) | p-Value | n = 744 | OR (95%CI) | p-Value | |
| Poor appetite | 2.36 (1.39–4.02) | 0.002 | 2.24 (1.30–3.86) | 0.004 | 2.21 (1.26–3.87) | 0.006 | |||
| Dysphagia | 0.76 (0.40–1.42) | 0.383 | 0.78 (0.41–1.47) | 0.439 | 0.73 (0.38–1.41) | 0.370 | |||
| Poor dental state | 0.98 (0.42–2.31) | 0.964 | 0.90 (0.37–2.16) | 0.809 | 0.84 (0.33–2.13) | 0.719 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buhl, S.F.; Olsen, P.Ø.; Thilsing, T.; Caserotti, P. Association between Malnutrition Risk Factors and Physical Function in Community-Dwelling Adults ≥80 Years. J. Ageing Longev. 2023, 3, 33-45. https://doi.org/10.3390/jal3010003
Buhl SF, Olsen PØ, Thilsing T, Caserotti P. Association between Malnutrition Risk Factors and Physical Function in Community-Dwelling Adults ≥80 Years. Journal of Ageing and Longevity. 2023; 3(1):33-45. https://doi.org/10.3390/jal3010003
Chicago/Turabian StyleBuhl, Sussi F., Pia Ø. Olsen, Trine Thilsing, and Paolo Caserotti. 2023. "Association between Malnutrition Risk Factors and Physical Function in Community-Dwelling Adults ≥80 Years" Journal of Ageing and Longevity 3, no. 1: 33-45. https://doi.org/10.3390/jal3010003
APA StyleBuhl, S. F., Olsen, P. Ø., Thilsing, T., & Caserotti, P. (2023). Association between Malnutrition Risk Factors and Physical Function in Community-Dwelling Adults ≥80 Years. Journal of Ageing and Longevity, 3(1), 33-45. https://doi.org/10.3390/jal3010003