Abstract
Emerging research has shown that pharmacist-led comprehensive medication management (CMM) can be an effective strategy for controlling hypertension. A synthesis of the evidence on the overall effects of CMM on clinical, quality, and economic outcomes could help inform and contribute to improvements in programming and practice. Presently, such a synthesis is limited in the literature. To address this gap, we conducted a scoping review of CMM effects on these outcomes, organized by 4 domains: therapeutic, humanistic, safety and economic. Using predefined search terms for articles on studies published between 2010 and 2024, we performed a literature search utilizing these terms to search the MEDLINE, Cochrane Library and CINAHL databases. For each of the identified studies, we applied a multi-stage screening process to extract data, chart results, and synthesize findings. The process took into account methodology of study design, patient population involved, CMM implementation, relevance of outcomes to clinical improvement, and factors that were deemed relevant to study selection. In total, 49 experimental, observational, and simulation-based studies were included in the scoping review. The synthesis focused on outcomes most frequently reported and those rigorously evaluated by the studies in the review. They included clinical measures of blood pressure reduction and control, frequency and duration of healthcare visits, and changes in medication therapy regimen and medication adherence. Overall, CMM interventions were found to have significantly favorable effects on systolic blood pressure reduction, hypertension control, and medication changes. Other outcomes, which showed positive effects, included self-reported patient experience and behaviors, emergency department visits, hospitalizations, mortality, and program costs and related savings from implementing a CMM program. Some results, however, were mixed. For example, a number of studies reported outcomes data without significance testing and many generally lacked consistent characterization of their programming and implementation processes. Future research and practice evaluations should include these elements in their documentation. Furthermore, a more consistent approach to implementing CMM in the field may lead to better support of program delivery fidelity, helping to optimize CMM, moving it from demonstrated efficacy to intervention effectiveness in the real world.
1. Introduction
Cardiovascular disease is the leading cause of death in the United States (U.S.), accounting for approximately 1 in 3 deaths annually [1]. Hypertension, which affects more than 48% of the U.S. population, is considered the most modifiable and preventable risk factor for cardiovascular disease [2]. This condition was the primary or contributing cause of nearly 700,000 deaths nationwide in 2022 alone [2]. Despite this, effective control of hypertension remains elusive across the country. Only 1 in 4 U.S. adults with diagnosed hypertension have their condition under control [2].
Antihypertensive medications are an essential tool for controlling hypertension and preventing cardiovascular disease [2,3]. However, both patients and providers need additional support to ensure that antihypertensive medication therapy is accessible and appropriately prescribed. Misuse, underuse and/or overuse can diminish medication effects or cause harm, leading to other health problems. Pharmacists are in a unique position to assist individuals with hypertension and reduce medication harm [3]. They can provide medication therapy expertise in collaboration with patients’ provider care teams. The American College of Clinical Pharmacy (ACCP) has promoted a collaborative drug therapy management framework since the late 1990s. This framework describes how pharmacists and physicians can work together to optimize medication therapy for patients [3]. Pharmacist-led, team-based care interventions are growing in popularity, driven by the profession’s nationwide doctoral-level training requirements, extensive clinical education, and opportunities for post-graduate residency training and board certifications.
The passage of the Medicare Modernization Act in 2003, Medicare Part D, required plans to offer Medication Therapy Management (MTM) to eligible beneficiaries [3]. MTM is a reimbursable drug benefit program in which a pharmacist conducts an annual one-time review of a patient’s medication regimen to identify medication problems and advise providers and patients on how best to correct them [4]. While MTM activities do not fully address the principles outlined in the ACCP collaborative framework, the reimbursement of MTM served to legitimize pharmacist-provided services under Medicare and build the case to expand pharmacist roles in team-based care and related models of practice, including comprehensive medication management (CMM).
CMM is a patient-centered approach, in which pharmacists review not only the patient’s medications but also their health history and behaviors to develop a personalized care plan and provide ongoing support to ensure adherence over time. According to ACCP, a CMM intervention includes four essential steps: (1) a thorough review of the patient’s medical and medication history; (2) identification of medication related problems based on the patient’s history and each medication’s appropriateness, effectiveness and safety; (3) development and initiation of a care plan designed to optimize medication therapy for the patient; and (4) adjustment of the care plan as needed, informed by results from clinical follow-up and collaboration with healthcare providers [5].
While CMM is longitudinal, more comprehensive, and potentially a better fit than MTM for integrating within a team-based care model, the healthcare community is more familiar with Medicare-defined MTM and its narrower scope of practice [6]. The variability in state legislative provisions to support (or to not support) collaborative drug therapy reimbursements further complicates this landscape. For instance, clinicians and researchers alike often confuse MTM nomenclature and terminology with those of CMM. In addition, the dynamic nature of CMM services lends itself to frequent variation in clinical practice settings, where the four essential steps of the model are not always operationalized in the same way as described in established frameworks or the literature.
Nonetheless, a growing body of literature points favorably to CMM’s effects on chronic disease control. Studies have shown that participation in CMM could lead to improvements in health (e.g., blood pressure [BP], glycemic control [hemoglobin A1c], lipids), functional (e.g., medication safety, medication adherence), and economic (e.g., healthcare cost savings) outcomes [7,8,9]. Presently, there is limited synthesis of this evidence in the literature. Such a synthesis could offer invaluable insights into better ways to conduct hypertension management and prevention for healthcare providers, program implementers, and researchers.
This scoping review sought to address this gap by describing the scope and nature of studies that have examined the effects of CMM on a broad spectrum of outcomes, extending beyond traditional measures of BP control. Pharmacist-led, pharmaceutical interventions, such as CMM, have the potential to affect not only medication and disease management, but also healthcare utilization and expenditures, and—most importantly—patient behaviors and experience. Summarizing the effects of CMM on hypertension from this multidimensional perspective could help enhance our understanding and advance the application of this team-based care model in real-world practice. Furthermore, because CMM has not been defined consistently in the literature and is often used interchangeably with MTM, we believe a scoping review could be used to help clarify the distinction between CMM and other pharmacist-led interventions, as well as capture trends in outcomes and effects evaluated in CMM studies. This review conducted its synthesis of the evidence with this in mind, focusing on four domains of outcomes: therapeutic, humanistic, safety and economic.
2. Materials and Methods
We conducted a scoping review following the framework designed by Arksey and O’Malley, and updated by Levac et al. [10,11]. The Arksey and O’Malley framework proposes a 5-stage methodology for investigators to carry out in scoping reviews and to report results with consistency and clarity. For transparency and reproducibility, we also followed the Preferred Reporting Items for Systematic Reviews and Meta-analysis extension for scoping reviews (PRISMA-ScR) [12]. The review protocol was registered in Open Science Framework Registries (https://doi.org/10.17605/OSF.IO/4XV7G, accessed on 27 August 2025).
The processes followed at each of the review stages are described below.
2.1. Identifying the Research Questions
Our review focused on CMM interventions provided to a population with or at risk for hypertension. The following research questions guided the search strategy and analysis plan:
- What is the scope of outcomes that are evaluated and reported in studies assessing the effects of CMM services delivered to a population of individuals with or at risk for hypertension?
- What is the overall effect of CMM interventions on these outcomes within this population?
- To what degree does the delivery of CMM services to this population vary in practice and characterization?
2.2. Search Strategy
Relevant studies were identified using MEDLINE, Cochrane Library and CINAHL databases. We included original research studies in articles published between 2010 and 2024. Review articles, meta-analyses, book chapters, conference proceedings and study protocols were excluded. Search terms were developed using a combination of controlled vocabulary terms (e.g., MeSH) and free-text key words. Boolean operators (AND/OR) were used to combine terms across the following themes: (1) experimental and observational study designs (e.g., “randomized controlled trials”, “cohort studies”); (2) hypertension and heart disease (e.g., “essential hypertension,” “blood pressure monitoring”); (3) pharmacists and pharmacist-led interventions (e.g., “pharmacist-based,” “pharmacy-managed”); (4) health economics (e.g., “cost benefit analysis,” “cost utility”) and (5) CMM interventions. As many interventions are not always referred to as CMM in the literature, a wide variety of search terms that could apply to CMM delivery were utilized, such as “CMM,” “MTM,” “medication review,” “drug regimen,” “Medicare Part D” and “Medicare Prescription Drug Improvement and Modernization Act of 2003.”
2.3. Study Selection
Studies identified in the database searches were imported into Zotero reference management software and duplicates were removed. Article titles and abstracts from these studies were screened for eligibility using the following criteria:
- They were conducted in the U.S. and published in English.
- They treated a patient population of individuals with or at risk for hypertension.
- They reported on patient-related therapeutic, humanistic, safety or economic outcomes related to BP management.
- They administered a CMM intervention.
To determine whether each study described an intervention that is CMM or could be classified as such, reviewers verified that the description of the intervention being studied indicated that: (1) the intervention was pharmacist-led with provider support; (2) contained a comprehensive medication review; (3) incorporated a patient care plan; and (4) was monitored over time through clinical follow-up.
Titles and abstracts were independently screened by two reviewers. Any resultant discrepancies were assessed and reconciled by the project leads (D.R.-S. and N.C.B.). A full text review of the remaining studies was conducted after the initial screening. This was followed by a review of citations in each study. Through the citation review, additional studies were identified and included.
2.4. Charting the Data
Once the full text screening was completed, a reviewer extracted and charted the data using an extraction form that captured study and intervention characteristics, as well as outcomes data. Study characteristics included publication year, study design, follow-up duration and sample size. Intervention characteristics included CMM methodology and delivery. And outcomes data included measured outcomes organized by four domains: therapeutic, humanistic, safety and economic. Therapeutic outcomes were defined as clinical events related to the intervention, such as change in BP, ambulatory care visitation or medication changes. Humanistic outcomes were defined as measures of patient functional status or quality of life, such as medication adherence, satisfaction with care or behavior change. Safety outcomes were defined as adverse events related to the intervention, such as adverse drug reactions, medication errors, emergency department (ED) visits or hospitalizations. Economic outcomes were defined as costs or savings related to the intervention, such as cumulative costs of healthcare utilization for intervention participants or cost-effectiveness demonstrated via economic modeling. To examine trends in the way outcomes were measured and reported across these domains, each domain was further broken down into specific categories. For example, within the therapeutic domain, categories of BP control included change in systolic blood pressure (SBP), change in diastolic blood pressure (DBP), percent of patients meeting BP control and change in hypertension disease stage. Additionally, as there is significant variation in how CMM is implemented in the field and described in the literature, we assessed variation in how these interventions were presented by examining differences in both the way they were labeled and in the way they discussed patient follow-up. Extracted data from each study were reviewed by a second member of the team to ensure consistency and clarity.
2.5. Collating, Summarizing and Reporting Results
Once the extracted data were tabulated, a descriptive analysis was conducted. Study characteristics; intervention characteristics; and outcome domains, including categories and effects, were synthesized and summarized. To determine the overall effect of CMM by outcome category, the intervention effects were classified as follows:
- Favorable—study outcomes with statistically significant p values or confidence intervals (CIs) indicating positive changes due to the implementation of CMM.
- Not favorable—study outcomes with statistically significant p values or CIs indicating negative changes due to the implementation of CMM.
- No effect—study outcomes with no statistically significant changes in either direction.
- Mixed—study outcomes that were stratified with mixed results (e.g., favorable, not favorable or no effect).
- Unclear—study outcomes that were reported without significance testing to validate the results.
Frequencies and percentages of intervention characteristics and outcome effects were generated to help summarize trends in intervention implementation and related results. Characteristics related to CMM delivery were also analyzed to look for trends that reflect how these interventions were adopted and implemented.
3. Results
3.1. Characteristics of the Studies That Were Included in the Scoping Review
The database search identified 264 studies for potential inclusion (Figure 1). After removing duplicates, 221 were retained for title and abstract screening, 40 of which were included in this review. A citation review of studies included from database searches identified an additional 51 studies, 9 of which were included in this review, resulting in a body of literature comprising 49 studies (see Table 1 and Supplemental Table S1) [13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61]. Of the 49, 25 (51%) were randomized controlled trials (RCTs), 17 (37%) were longitudinal evaluations, 4 (8%) were cohort studies, and 3 (6%) were economic modeling studies. The majority were published between 2010–2014 (41%) and 2015–2019 (47%). Sample sizes varied extensively, with the majority assessing less than 500 patients (73%). Follow-up duration ranged from 1 to 66 months. Forty-six studies reported therapeutic outcomes, 29 reported humanistic outcomes, 21 reported safety outcomes, and 10 reported economic outcomes. Of the 18 outcome categories identified, 6 were evaluated with significance testing to validate results in more than 10 studies (Figure 2).
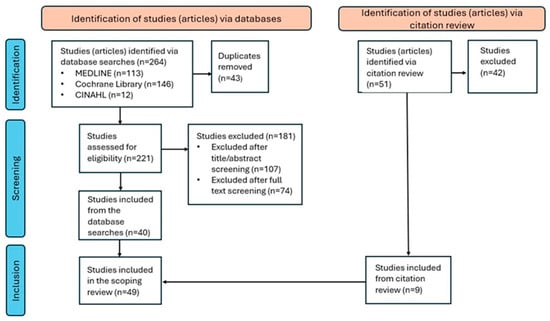
Figure 1.
PRISMA-ScR flowchart of study identification, screening and selection—articles included in the scoping review.

Table 1.
Characteristics of the studies that were included in the scoping review, 2010–2024.
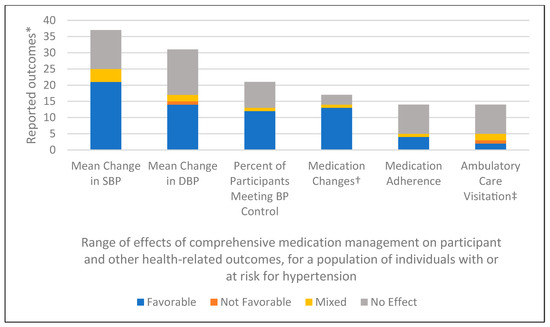
Figure 2.
Comprehensive medication management outcomes reported in 10 or more studies included in the scoping review, with statistically validated results. * Results for the range of effects of comprehensive medication management (CMM) on participant and other health-related outcomes were classified as: (1) Favorable: outcomes with statistically significant p values or confidence intervals (CIs) indicating positive changes due to the implementation of CMM; (2) Not favorable: outcomes with statistically significant p values or CIs indicating negative changes due to the implementation of CMM; (3) No effect: outcomes with no statistically significant changes in either direction; or (4) Mixed: outcomes that have a mixed or a combination of results (e.g., favorable, not favorable, or no effect). † Medication changes over time or differences in endpoint total or mean number of medications between intervention and control. ‡ More visitations for CMM patients were characterized as favorable and less visitations for CMM patients were characterized as not favorable. SBP = systolic blood pressure; DBP = diastolic blood pressure; BP = blood pressure.
Notably, the 49 studies included in this review reported on results from 41 unique interventions. In some cases, the same intervention was the subject of 2, 3 or even 4 studies, all of which were identified in the search strategy and met the inclusion criteria (see Supplemental Table S1). The outcomes reported in each individual published study, however, were unique. Those that published on the same intervention employed distinct methodologies, such as analyzing data from a different follow-up period or focusing on a subset of patients from a single treatment arm or with a specific comorbidity. In addition, the three economic modeling studies built their models based on data from several RCTs and cohort studies that were already part of the scoping review.
3.2. Effects of CMM on Therapeutic Outcomes
Therapeutic outcomes assessing BP control included mean change in SBP and DBP, percent of patient population meeting BP control, and change in hypertension disease stage (Figure 3). Fifty-four percent (21/39) of studies showed a favorable change in SBP [13,14,21,25,29,31,34,35,36,37,38,41,43,44,46,48,54,55,56,57,58], 31% (12/39) showed no effect [13,20,23,24,26,27,28,30,39,47,59,60], 10% (4/39) had mixed results [16,40,50,51] and 5% (2/39) showed reductions in SBP, but were not validated with significance testing [15,18]. Forty two percent (14/33) of studies showed a favorable change in DBP [13,21,25,30,31,37,38,43,44,46,50,55,56,58], 3% (1/33) showed a greater decrease in DBP in the control group than in the intervention, resulting in a change that was not favorable [47], 42% (14/33) showed no effect [14,23,24,27,28,29,34,35,39,41,48,54,57,59], 6% (2/33) had mixed results [16,40] and 6% (2/33) showed reductions in DBP, but were not validated with significance testing [15,18]. Fifty percent (12/24) of studies showed a favorable change in the percent of patients meeting BP control [13,22,27,29,35,41,46,50,52,55,56,57], 33% (8/24) showed no effect [20,21,25,28,30,34,48,50], 4% (1/24) had mixed results [16] and 13% (3/24) showed improvements in BP control, but were not validated with significance testing [15,42,59]. Three studies measured change in hypertension disease stage, all of which reported favorable results [22,44,59].
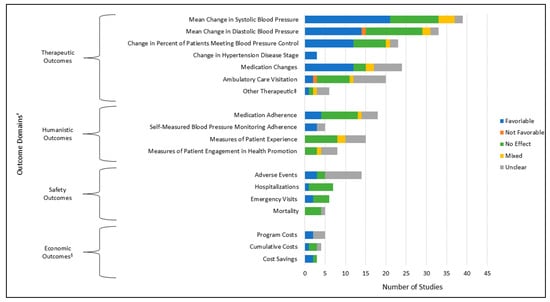
Figure 3.
Results from the scoping review, 2010–2024: Range of effects of comprehensive medication management on participant and other health-related outcomes for a population of individuals with or at risk for hypertension. Results for the range of effects of comprehensive medication management (CMM) on participant and other health-related outcomes were classified as: (1) Favorable: outcomes with statistically significant p values or confidence intervals (CIs) indicating positive changes due to the implementation of CMM; (2) Not favorable: outcomes with statistically significant p values or CIs indicating negative changes due to the implementation of CMM; (3) No effect: outcomes with no statistically significant changes in either direction; (4) Mixed: outcomes that have a mixed or a combination of results (e.g., favorable, not favorable, or no effect); or (5) Unclear: outcomes reported without statistical testing to validate the results. † Outcome domains were defined as: (i) Therapeutic: health outcomes from treatment or clinical intervention; (ii) Humanistic: outcomes that are related to patient symptom status, functional status, and/or quality of life; (iii) Safety: any occurrence of adverse clinical or drug events; and (iv) Economic: direct or indirect costs of implementing the intervention or the intervention’s cost-effectiveness. ‡ Other therapeutic outcomes from randomized controlled trials, cohort studies, and longitudinal evaluations included: Charlson Comorbidity Index scores, patient 10-year cardiovascular disease risk, and the mediated effect of change in weight on SBP reduction. Outcomes from economic modeling studies included: increased life years, increased Quality Adjusted Life Years (QALYs), and lowered cardiovascular disease risk. § Results of outcomes from economic modeling studies were not included in this figure as they were not measured with p values or confidence intervals.
Other therapeutic outcomes that were frequently reported included medication changes and measures related to ambulatory care visitation (e.g., mean number of pharmacy or primary care visits, median duration of visit or interval between visits, etc.). Fifty percent (12/24) of studies measuring medication changes reported significantly more changes among intervention participants, as compared to control [13,20,25,30,34,35,36,39,51,52,55,56], 13% (3/24) showed no effect on this measure [16,47,57] and 8% (2/24) had mixed results [48,50]. Twenty nine percent (7/24) reported on medication changes without providing results of significance testing [14,15,26,27,31,46,58]; of these, the majority were longitudinal evaluations that reported an increase in the number of medications prescribed to CMM patients from baseline to endpoint. Ten percent (2/20) of studies measuring ambulatory care visitation reported significantly more visitation in the intervention group [48,56], 5% (1/20) reported significantly less visitation in the intervention group [21] and 5% (1/20) had mixed results [39]. Forty percent (8/20) reported on visitation outcomes, but without results from significance testing [14,16,18,26,32,34,46,51]; among these, outcomes varied substantially, with 3 separate studies reporting the majority of CMM participants attended all visits.
Six studies reported on other therapeutic outcomes that were not frequently found in the literature. One study reported Charlson Comorbidity Index scores with significant results favoring the intervention group [27], a second reported on patient 10-year cardiovascular disease risk with mixed results [29], and a third reported on the mediated path of change in weight on SBP reduction, which resulted in no significant effect [36]. The 3 economic modeling studies reported projected life years, Quality Adjusted Life Years (QALYs), a combined measure of both health and quality of life, and cardiovascular disease risk [33,45,61]. In these studies, CMM interventions were associated with increased life years and QALYs and lowered cardiovascular disease risk for intervention patients, as compared to control.
3.3. Effects of CMM on Humanistic Outcomes
The most frequently reported humanistic outcome was adherence, either to medications or to self-measured blood pressure monitoring (SMBP), which was measured in 21 studies (Figure 3). Twenty-two percent (4/18) reported favorable results related to medication adherence [17,19,28,49], while 50% (9/18) reported no effect on this outcome [20,21,22,23,25,35,36,47,48]. Twenty two percent (4/18) reported on medication adherence without significance testing, all of which demonstrated that the majority of intervention participants self-reported high levels of medication adherence at study end [14,15,18,31]. Five studies reported on adherence to SMBP, 60% (3/5) of which demonstrated favorable results [36,50,53] and 40% (2/5) that were promising, but not validated with significance testing [32,35].
Other reported humanistic outcomes included patient experience and patient engagement in health promotion activities (Figure 3). Measures of patient experience included measures of patient self-reported functional status and/or quality of life (QOL). Of the studies that reported on patient experience, 2 reporting on patient self-rated health and engagement in care, had mixed results [55,56]. Seven reporting on QOL, self-rated health, perceived social support, depression and hypertension knowledge, had no effect [20,23,24,40,47,51,60]. Additionally, 5 did not validate their results with significance testing [14,18,27,35,53]. Those that did not report on significance testing were longitudinal evaluations with no comparison group, in which most intervention participants reported high levels of satisfaction with care or comfort with managing chronic disease at study end. Measures of participation in health promotion activities included being referred to and/or accessing additional services or demonstrating changes in behavior. Of the studies reporting health promotion outcomes, 1 reported on changes in diet and physical activity with mixed results [50], 3 reported on changes in physical activity and tobacco, alcohol and caffeine consumption with no effect [36,40,51] and 4 did not validate with statistical testing [14,31,35,46]. Those that did not report on significance testing were longitudinal evaluations reporting that pharmacists were referring a large volume of CMM patients to screenings, vaccines, specialty care, lifestyle change programs, social support and community health workers.
3.4. Effects of CMM on Safety Outcomes
Safety outcomes included adverse events (AEs), which included medication-related problems (MRPs), such as drug duplications, drug–drug interactions or high-risk medications prescribed, as well as hospitalizations, ED visits, and mortality (Figure 3). Of the 14 studies that reported on AEs, 3 looked at them generally [16,28,35], while the remaining 11 reported solely on MRPs [14,21,27,31,37,39,42,48,49,53,58]. Three of these 14 studies reported favorable results on AEs [21,39,49], 2 reported no effect [28,48] and 9 did not report on significance testing [14,16,27,31,35,37,42,53,58]. Among these 9, the majority were longitudinal evaluations that reported the total number of MRPs or CMM patients experiencing MRPs, without looking at change over time. Seven RCT and cohort studies compared the number of hospitalizations between intervention and control. Of these, 1 had favorable results [39], while 6 had no effect [17,21,51,52,54,60]. Similarly, six studies compared the number of ED visits between intervention and control, 2 of which reported favorable results [17,21] and 4 reported no effect [51,52,54,60]. Four studies compared differences in mortality with no significant differences between groups [17,20,28,60]. One study reported comparable mortality rates between groups, but without significance testing [55].
3.5. Effects of CMM on Economic Outcomes
The economic outcomes assessed in experimental and observational studies from this scoping review included program costs, cumulative costs and healthcare cost savings (Figure 3). Program costs were reported in 5 studies and measured by looking at annual cost per CMM patient. Of these, 2 demonstrated a favorable decrease in cost per patient [59,60], while 3 reported the cost per CMM patient without comparing to cost of control patient or assessing change over time [21,28,35]. Four studies reported cumulative costs by measuring either total prescription costs or costs of all cause medical care per patient. Of these, 1 reported favorable results [43], 2 reported no effect [28,60] and 1 did not report on significance testing to validate the results [61]. Three studies reported on healthcare cost savings. One study compared average reduction in healthcare costs between intervention and control with significant results favoring intervention patients [43]. Another estimated average net benefit per patient with significant results, also favoring the intervention [59]. A third calculated average savings per patient based on change in health care costs over time and found no significant effect [60].
Three studies utilized economic modeling to conduct multivariable and probabilistic sensitivity analyses to estimate the degree to which CMM interventions may have been cost-effective [33,45,61]. These studies reported on the proportion of participants for which CMM interventions were cost-effective at distinct willingness-to-pay thresholds (WTP), as well as the monetary values associated with the incremental cost effectiveness ratios (ICER) per QALY gained for CMM interventions. At the standard WTP threshold of $50,000, the percentage of time that CMM was determined to be cost effective ranged from 49–58% [33,45], however, cost-effectiveness increased at higher willingness to pay thresholds [45]. Additionally, in a ten-year time horizon, the ICER per QALY gained for CMM interventions was approximately $39,000 [33,45]; however, with a lifetime time horizon, cost-effectiveness increased [61], suggesting that a lengthy follow-up period is important for effectively evaluating the economic impact of CMM on a population that has or is at risk for hypertension. Additionally, one study compared cost effectiveness across cohorts of patients with varying degrees of cardiovascular disease risk and demonstrated that CMM was most cost-effective for patients most at risk for cardiovascular disease [33].
3.6. Variation in CMM Implementation
Although all of the published studies in the scoping review were assessed to ensure the interventions described contained the essential elements of CMM, none explicitly labeled their intervention as “CMM” in their written narrative. Ten referred to their intervention as MTM, with the remainder showing substantial variation in their intervention title. The majority (n = 27) called out “pharmacist” or “pharmacy” services in their intervention title (e.g., “pharmacist outreach,” “pharmacist consultation,” “pharmacist management”), while others referred to the intervention in more general language, such as “multifaceted intervention,” “multidisciplinary program,” or “multicomponent model.” To establish that the interventions being described were, in fact, pharmacist-led, we conducted closer readings of these published studies.
We also observed that, in each of the published studies, the activities that followed the initial CMM visit were described in varying degrees of detail. Descriptions of the “patient care plan” component of the CMM model were particularly inconsistent. While it was stated in 12 of the published studies that, after the medication review, the pharmacist shared a personalized “action” or “care” plan with the patient, the others did not explicitly state that a patient “care plan” was developed, but instead described specific follow-up activities typically included in a care plan, such as medication changes, health education, or SMBP goal setting. In reviewing the Supplementary Materials that accompanied each of these published studies, it was apparent that some of them did incorporate pharmacist provision of a personalized patient care plan in their program, but did not explicitly explain this in their written narratives. To this end, it was difficult to fully determine the exact proportion of studies that adopted a formal approach to the patient care plan component of CMM.
4. Discussion
This scoping review provides an overview of the scope and nature of the CMM interventions found in the literature that affect a population of individuals with or at risk for hypertension. We compared the intervention effects across four domains of outcomes: therapeutic, humanistic, safety and economic. In the review, a majority of the studies reported on therapeutic and humanistic outcomes; safety or economic outcomes were less commonly reported. Given the range of outcome results, we were mindful of trends for each of them, paying close attention to how the outcomes were measured and reported, looking at their overall effect(s) or contribution(s) to health impact from a lens of program effectiveness and cost.
Of the therapeutic outcomes, BP control was the most frequently reported and rigorously evaluated with significance testing. The majority demonstrated an improvement in SBP and in the percentage of patients meeting BP control; this is consistent with prior research [9,62,63]. Although the results on DBP were more varied, a large proportion of studies also showed clinical improvements on this outcome, which is also consistent with findings from studies of other pharmacist-led interventions [62]. Notably, 6 studies solely examined SBP without reporting on DBP [17,20,28,36,51,60]—this may have been due to emerging evidence suggesting that SBP has a greater impact on cardiovascular disease outcomes than DBP [64,65]. Those studies that stratified results of BP outcomes by hypertension disease stage indicated that CMM interventions had a greater effect on patients with more severe hypertension, with greater improvements shown in patients with more severe stages.
Of the humanistic outcomes, medication adherence was the most frequently measured and reported. A majority of the studies included in the scoping review reported on this outcome, using significance testing to validate the results, showing that CMM generally had no significant effect on medication adherence when compared with usual care. This result, however, was somewhat surprising, as in a prior meta-analysis examining 22 pharmacist-led interventions, 61.5% of the studies reviewed showed a favorable effect on medication adherence [66]. Similarly, in another systematic review that looked at 12 studies, 43% of them reported a favorable effect on adherence [67].
A potential explanation for this lower-than-expected impact on medication adherence could be due to the higher variability in the measurement methods used by the studies included in the present scoping review (see Supplementary Table S1). For example, 8 studies administered the Morisky Medication Adherence Scale (MMAS) [20,21,22,23,25,35,36,48], while one study administered the Pharmacist Drug Adherence Work-up (DRAW) Tool [49]—both tools are validated survey instruments that document self-reported medication taking behaviors. Additionally, 3 studies in the review utilized the pharmacy refill data formula for average proportion of days covered [17,19,28], 1 study utilized the formula for the average patient medication possession ratio [34] and 5 studies ascertained medication adherence through patient self-report, without the use of pharmacy refill data or a validated tool. Interestingly, compared to the largely favorable results from the studies that utilized pharmacy refill data to indirectly assess medication consumption, the vast majority of those that utilized validated survey instruments that directly assess medication consumption (e.g., MMAS, DRAW) showed limited to no intervention effect on medication adherence. Self-report biases associated with these latter instruments may have contributed to this pattern of non-favorable findings.
Of all safety outcomes included in the scoping review, MRPs were the most frequently reported. Most studies reported a wide range of MRPs, including drug duplications, drug-disease interactions, drug–drug interactions, adverse drug reactions, high risk medications prescribed, sub-therapeutic drug dosing, and medication nonadherence. While some studies provided composite scores on the number of MRPs identified and addressed, others presented data stratified by MRP type. Many that presented data stratified by MRP type reported mixed results, indicating that CMM had a favorable effect on certain MRPs, but less of an effect on others. A more granular examination of the effect(s) of CMM on specific types of MRPs in future research may reveal additional trends and nuances regarding how CMM is mitigating these different types of medication-related problems.
In accordance with prior research, analyses of costs and savings associated with CMM interventions in this scoping review yielded mixed results [8,63,68]. Studies of return on investment of pharmacist-led medication management services across numerous settings have shown CMM as a viable, promising strategy for hypertension management and prevention, but its cost–benefit profile varied substantially [5]. That is, even though the aim of CMM is to reduce healthcare utilization and correspondingly health care expenditures, their more personalized services approach (more frequent visits with patients and tailoring of medication management) may come at a cost and may, in fact, increase healthcare utilization among those who need more complex coordination of services [68]. For instance, the experiences of the three economic modeling studies suggest that, to realize a fuller picture of the intervention’s cost impact, cost–benefit analyses may need to assess longer durations of patient follow-up (e.g., >10 years). Present analyses primarily covered relatively shorter durations, limiting their ability to make inferences about the complexity and time it takes to effectively implement CMM.
The extensive variation observed in the descriptions of CMM delivery across these studies suggest that CMM is not operationalized consistently in the field. Discrepancies in implementation may have contributed to the wide range of reported outcomes and intervention effects. Moreover, the outcome results observed across studies may not be directly attributable to the CMM model, as the fidelity, intensity and consistency of its implementation were often unclear or inadequately described in the literature. Without a clear understanding of the “dose” or adherence to core CMM components, it is difficult to determine whether the observed effects reflect the true potential impact of the model. Greater clarity around the concrete steps, core elements, and consistent implementation of the CMM model is needed to ensure fidelity across settings and to strengthen the scientific understanding of pharmacy practice, particularly its impact on therapeutic, humanistic, safety and economic outcomes, as well as the role of the pharmacist in medication-related care.
4.1. Limitations
This scoping review has several limitations. While our search strategy was comprehensive and drew from three separate library databases, it is possible that some studies were missed by our search terms. Additionally, the CMM approach or methodology was not always thoroughly nor consistently described in the studies we reviewed; this limitation may have led to certain CMM studies being mischaracterized and/or misclassified by our review protocol.
Because the objective of the review was to determine the overall effect of CMM across a broad spectrum of outcomes, our analyses did not fully account for variations in study design and outcome measurement. For example, experimental and observational studies were grouped together and the outcomes of each were considered with equal weight. Additionally, among longitudinal evaluations, which were typically retrospective evaluations of CMM delivery in health systems, sample size varied substantially, ranging from 15 to 5942 patients (see Supplementary Table S1). Furthermore, the majority of reported humanistic outcomes that evaluated patient experience were measured using self-reported survey instruments, many of which were not statistically validated. Results of these outcomes may have been subject to social desirability bias, as well as weakened validity and low reliability.
An additional limitation of this review is that we did not assess whether CMM was delivered in community pharmacy settings versus clinic-based or integrated health system environments. This distinction may influence both the effectiveness of CMM interventions and feasibility of conducting high-quality studies evaluating their impact. For example, integrated health systems may offer more streamlined access to clinical data and care coordination processes, which can support both the delivery of CMM and rigorous evaluation of outcomes. In contrast, delivering CMM through community pharmacies can involve unique challenges that could negatively affect both implementation and impact. These challenges include limited or delayed access to clinical information such as lab results and vital signs, delays in executing care plans due to the need for physician approval, and greater difficulties in conducting timely quality improvement activities. Future research should more explicitly consider and report the care setting, as the structural differences may influence the quality and interpretation of CMM outcomes data.
Nonetheless, a strength of this scoping review is its purposeful synthesis and description of the evidence. Our review highlights the scope and nature of CMM interventions, showing that this team-based care approach or model has salutary effects on a range of outcomes in the clinical environment, as well as in support of a better experience for the patient.
4.2. Future Directions
CMM is a comprehensive, longitudinal, patient-centered approach to medication optimization that leverages pharmacists as critical members of the care team and has the potential to impact health care delivery and population health. To better understand the full effects of CMM, more emphasis should be placed on evaluating outcomes that go beyond disease control and medication management and strive to measure healthcare quality and cost. In order to do this, CMM researchers should determine which economic and humanistic outcomes are priority metrics for understanding CMM’s cost-effectiveness and impact on patient experience and behaviors. Furthermore, healthcare providers, program implementers, and researchers should aim to implement CMM according to the tenets of ACCP, describe this methodology more explicitly in the literature and evaluate the various components of CMM to identify which features of CMM interventions (e.g., follow-up frequency, team integration, training, etc.) are most associated with favorable results. These efforts would facilitate further synthesis and comparisons of these model interventions to better support program delivery, assess for fidelity, and optimize implementation, moving this team-based care approach from demonstrated efficacy to intervention effectiveness in the real world.
5. Conclusions
Hypertension is one of the most prevalent risk factors for cardiovascular disease, as well as one of the most preventable. CMM is a well-defined, patient-centered, team-based process for medication optimization that can help individuals manage their hypertension. CMM has the potential to favorably influence a wide variety of outcomes. Results from this scoping review provide a meaningful synthesis of the current evidence in support of CMM interventions’ use in the clinical environment. The review offers valuable insights into ways CMM is currently being applied to improve hypertension management and cardiovascular disease reduction. Its results on the four domains of outcomes could and should be used to help guide and inform present and future health policies and planning efforts related to this team-based care approach.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/encyclopedia5030133/s1, Supplemental Table S1: Summary of study characteristics, outcomes and results from the scoping review, 2010–2024; n = 49.
Author Contributions
Conceptualization, D.R.-S. and N.C.B.; Data Curation, D.R.-S., N.C.B., S.W., A.C., D.L., L.L. and E.P.III; Formal Analysis, D.R.-S. and N.C.B.; Writing—Original Draft Preparation, D.R.-S. and N.C.B.; Writing—Review & Editing, D.R.-S., N.C.B., S.W., A.C., D.L., L.L., E.P.III, S.C. and T.K.; Fund Acquisition—T.K. and N.C.B. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported, in part, by funding from the Centers for Disease Control and Prevention through cooperative agreements with the local health department in Los Angeles County (NU58DP006619 and NU58DP007576).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Acknowledgments
The conclusions presented in this article are those of the authors and do not necessarily represent the views of the funder, grantee, or any of the other organizations referenced in the text. The authors thank Perla Hernandez, MLIS, from the Los Angeles County Department of Public Health, for her support with the search strategy development.
Conflicts of Interest
Scott Weber is a Clinical Pharmacy Resident at Navitus Health Solutions, LLC. Employment at Navitus began after the completion and submission of this article. The other authors declare no conflicts of interest.
References
- Centers for Disease Control and Prevention. Heart Disease Facts. Available online: https://www.cdc.gov/heart-disease/data-research/facts-stats/index.html (accessed on 16 May 2025).
- Centers for Disease Control and Prevention. High Blood Pressure Facts. Available online: https://www.cdc.gov/high-blood-pressure/data-research/facts-stats/index.html (accessed on 16 May 2025).
- McBane, S.E.; Dopp, A.L.; Abe, A.; Benavides, S.; Chester, E.A.; Dixon, D.L.; Dunn, M.; Johnson, M.D.; Nigro, S.J.; Rothrock-Christian, T.; et al. Collaborative drug therapy management and comprehensive medication management. Pharmacotherapy 2015, 35, e39–e50. [Google Scholar] [CrossRef]
- American College of Clinical Pharmacy. What Is Medication Therapy Management (MTM)? Available online: https://www.accp.com/docs/govt/advocacy/Leadership%20for%20Medication%20Management%20-%20MTM%20101.pdf (accessed on 16 May 2025).
- American College of Clinical Pharmacy. Comprehensive Medication Management in Team-Based Care. Available online: https://insurance.maryland.gov/Consumer/Documents/agencyhearings/CMM-Brief.pdf (accessed on 16 May 2025).
- Rojas, E.I.; Dupotey, N.M.; De Loof, H. Comprehensive medication management services with a holistic point of view, a scoping review. Pharmacy 2023, 11, 37. [Google Scholar] [CrossRef] [PubMed]
- Brummel, A.; Carlson, A.M. Comprehensive medication management and medication adherence for chronic conditions. J. Manag. Care Spec. Pharm. 2016, 22, 56–62. [Google Scholar] [CrossRef]
- Chung, T.H.; Hernandez, R.J.; Libaud-Moal, A.; Nguyen, L.K.; Lal, L.S.; Swint, J.M.; Lansangan, P.J.; Le, Y.L. The evaluation of comprehensive medication management for chronic diseases in primary care clinics, a Texas delivery system reform incentive payment program. BMC Health Serv. Res. 2020, 20, 671. [Google Scholar] [CrossRef]
- Prudencio, J.; Cutler, T.; Roberts, S.; Marin, S.; Wilson, M. The effect of clinical pharmacist-led comprehensive medication management on chronic disease state goal attainment in a patient-centered medical home. J. Manag. Care Spec. Pharm. 2018, 24, 423–429. [Google Scholar] [CrossRef]
- Arksey, H.; O’Malley, L. Scoping studies: Towards a methodological framework. Int. J. Soc. Res. Methodol. 2005, 8, 19–32. [Google Scholar] [CrossRef]
- Levac, D.; Colquhoun, H.; O’Brien, K.K. Scoping studies: Advancing the methodology. Implement. Sci. 2010, 5, 69. [Google Scholar] [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Anderegg, M.D.; Gums, T.H.; Uribe, L.; Coffey, C.S.; James, P.A.; Carter, B.L. Physician–pharmacist collaborative management. Hypertens. 2016, 68, 1314–1320. [Google Scholar] [CrossRef] [PubMed]
- Axon, D.R.; Johnson, M.; Abeln, B.; Forbes, S.; Anderson, E.J.; Taylor, A.M.; Aseret-Manygoats, T.; Warholak, T.; Hall-Lipsy, E. An academic-community collaboration to deliver medication therapy management (MTM) services to patients living in rural counties of a southwestern state in the United States. J. Pharm. Pract. 2022, 35, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Beran, M.; Asche, S.E.; Bergdall, A.R.; Crabtree, B.; Green, B.B.; Groen, S.E.; Klotzle, K.J.; Michels, R.D.; Nyboer, R.A.; O’Connor, P.J.; et al. Key components of success in a randomized trial of blood pressure telemonitoring with medication therapy management pharmacists. J. Am. Pharm. Assoc. 2003, 58, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Carter, B.L.; Coffey, C.S.; Ardery, G.; Uribe, L.; Ecklund, D.; James, P.; Egan, B.; Vander Weg, M.; Chrischilles, E.; Vaughn, T.A. Cluster-randomized trial of a physician\pharmacist collaborative model to improve blood pressure control. Circ. Cardiovasc. Qual. Outcomes 2015, 8, 235–243. [Google Scholar] [CrossRef]
- Choudhry, N.K.; Isaac, T.; Lauffenburger, J.C.; Gopalakrishnan, C.; Lee, M.; Vachon, A.; Iliadis, T.L.; Hollands, W.; Elman, S.; Kraft, J.M.; et al. Effect of a remotely delivered tailored multicomponent approach to enhance medication taking for patients with hyperlipidemia, hypertension, and diabetes: The STIC2IT Cluster Randomized Clinical Trial. JAMA Intern. Med. 2018, 178, 1182–1189. [Google Scholar] [CrossRef]
- Coe, A.B.; Choe, H.M.; Diez, H.L.; Rockey, N.G.; Ashjian, E.J.; Dorsch, M.P.; Kim, H.M.; Farris, K.B. Pharmacists providing care in statewide physician organizations: Findings from the Michigan Pharmacists Transforming Care and Quality Collaborative. J. Manag. Care Spec. Pharm. 2020, 26, 1558–1566. [Google Scholar] [CrossRef]
- Colvin, N.N.; Mospan, C.M.; Buxton, J.A.; Waggett, J.D.; Gillette, C. Using Indian Health Service (IHS) counseling techniques in an independent community pharmacy to improve adherence rates among patients with diabetes, hypertension, or hyperlipidemia. J. Am. Pharm. Assoc. 2018, 58, S59–S63. [Google Scholar] [CrossRef]
- Cooney, D.; Moon, H.; Liu, Y.; Miller, R.T.; Perzynski, A.; Watts, B.; Drawz, P.E. A pharmacist-based intervention to improve the care of patients with CKD: A pragmatic, randomized, controlled trial. BMC Nephrol. 2015, 16, 56. [Google Scholar] [CrossRef] [PubMed]
- Edelman, D.; Fredrickson, S.K.; Melnyk, S.D.; Coffman, C.J.; Jeffreys, A.S.; Datta, S.; Jackson, G.L.; Harris, A.C.; Hamilton, N.S.; Stewart, H.; et al. Medical clinics versus usual care for patients with both diabetes and hypertension: A randomized trial. Ann. Intern. Med. 2010, 152, 689–696. [Google Scholar] [CrossRef]
- Fortuna, R.J.; Nagel, A.K.; Rose, E.; McCann, R.; Teeters, J.C.; Quigley, D.D.; Bisognano, J.D.; Legette-Sobers, S.; Liu, C.; Rocco, T.A. Effectiveness of a multidisciplinary intervention to improve hypertension control in an urban underserved practice. J. Am. Soc. Hypertens. 2015, 9, 966–974. [Google Scholar] [CrossRef]
- Gatwood, J.; Hanley, R.; Moore, J.S.; Hohmeier, K. Community pharmacist led, employer-based wellness services: A pilot study. Res. Social. Adm. Pharm. 2019, 15, 615–618. [Google Scholar] [CrossRef]
- Gerber, B.S.; Biggers, A.; Tilton, J.J.; Marsh, D.E.S.; Lane, R.; Mihailescu, D.; Lee, J.; Sharp, L.K. Mobile health intervention in patients with Type 2 diabetes: A randomized clinical trial. JAMA Netw. Open. 2023, 6, e2333629. [Google Scholar] [CrossRef] [PubMed]
- Gums, T.; James, P.; Carter, B.; Weg, M.V.; Uribe, L.; Coffey, C. Pharmacist intervention for blood pressure control: Medication intensification and adherence. J. Am. Soc. Hypertens. 2015, 9, 569–578. [Google Scholar] [CrossRef]
- Heisler, M.; Hofer, T.P.; Schmittdiel, J.A.; Selby, J.V.; Klamerus, M.L.; Bosworth, H.B.; Bermann, M.; Kerr, E.A. Improving blood pressure control through a clinical pharmacist outreach program in patients with diabetes mellitus in 2 high-performing health systems: The adherence and intensification of medications cluster randomized, controlled pragmatic trial. Circulation 2012, 125, 2863–2872. [Google Scholar] [CrossRef]
- Hirsch, J.D.; Steers, N.; Adler, D.S.; Kuo, G.M.; Morello, C.M.; Lang, M.; Singh, R.F.; Wood, Y.; Kaplan, R.M.; Mangione, C.M. Primary care-based, pharmacist-physician collaborative medication-therapy management of hypertension: A randomized, pragmatic trial. Clin. Ther. 2014, 36, 1244–1254. [Google Scholar] [CrossRef] [PubMed]
- Ho, P.M.; Lambert-Kerzner, A.; Carey, E.P.; Fahdi, I.E.; Bryson, C.L.; Melnyk, S.D.; Bosworth, H.B.; Radcliff, T.; Davis, R.; Mum, H.; et al. Multifaceted intervention to improve medication adherence and secondary prevention measures after acute coronary syndrome hospital discharge: A randomized clinical trial. JAMA Intern. Med. 2014, 174, 186–193. [Google Scholar] [CrossRef]
- Ip, E.J.; Shah, B.M.; Yu, J.; Chan, J.; Nguyen, L.T.; Bhatt, D.C. Enhancing diabetes care by adding a pharmacist to the primary care team. Am. J. Health Syst. Pharm. 2013, 70, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, M.; Sherry, P.S.; Taylor, L.M.; Amato, M.; Tataronis, G.R.; Cushing, G. Pharmacist assisted medication program enhancing the regulation of diabetes (PAMPERED) study. J. Am. Pharm. Assoc. 2012, 52, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.; Jastrzab, R.; Tate, J.; Johnson, K.; Hall-Lipsy, E.; Martin, R.; Taylor, A.M.; Warholak, T. Evaluation of an academic-community partnership to implement MTM services in rural communities to improve pharmaceutical care for patients with diabetes and/or hypertension. J. Manag. Care Spec. Pharm. 2018, 24, 132–141. [Google Scholar] [CrossRef]
- Kerby, T.J.; Asche, S.E.; Maciosek, M.V.; O’Connor, P.J.; Sperl-Hillen, J.M.; Margolis, K.L. Adherence to blood pressure telemonitoring in a cluster-randomized clinical trial. J. Clin. Hypertens. 2012, 14, 668–674. [Google Scholar] [CrossRef]
- Kulchaitanarouj, P.; Brooks, J.M.; Chaiyakunapruk, N.; Goedken, A.M.; Chrischilles, E.A.; Carter, B.L. Cost-utility analysis of physician-pharmacist collaborative intervention for treating hypertension compared with usual care. J. Hypertens. 2017, 35, 178–187. [Google Scholar] [CrossRef]
- Magid, D.J.; Ho, M.; Olson, K.L.; Brand, D.W.; Welch, L.K.; Snow, K.E.; Lambert-Kerzner, A.C.; Plomondon, M.E.; Havranek, E.P. A multimodal blood pressure control intervention in 3 healthcare systems. Am. J. Manag. Care 2011, 17, e96–e103. [Google Scholar]
- Margolis, K.L.; Asche, S.E.; Bergdall, A.R.; Dehmer, S.P.; Groen, S.E.; Kadrmas, H.M.; Kerby, T.J.; Klotzle, K.J.; Maciosek, M.V.; Michels, R.D.; et al. Effect of home blood pressure telemonitoring and pharmacist management on blood pressure control: A cluster randomized clinical trial. JAMA 2013, 310, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Margolis, K.L.; Asche, S.E.; Bergdall, A.R.; Dehmer, S.P.; Maciosek, M.V.; Nyboer, R.A.; O’Connor, P.J.; Pawloski, P.A.; Sperl-Hillen, J.M.; Trower, N.K.; et al. A successful multifaceted trial to improve hypertension control in primary care: Why did it work. J. Gen. Intern. Med. 2015, 30, 1665–1672. [Google Scholar] [CrossRef]
- Migliozzi, D.R.; Zullo, A.R.; Collins, C.; Elsaid, K.A. Achieving blood pressure control among renal transplant recipients by integrating electronic health technology and clinical pharmacy services. Am. J. Health Syst. Pharm. 2015, 72, 1987–1992. [Google Scholar] [CrossRef]
- Nuffer, W.; McCollum, M.; Ellis, S.L.; Turner, C.J. Further development of pharmacy student-facilitated diabetes management clinics. Am. J. Pharm. Educ. 2012, 76, 50. [Google Scholar] [CrossRef]
- Pepper, M.J.; Mallory, N.; Coker, T.N.; Chaki, A.; Sando, K.R. Pharmacists’ impact on improving outcomes in patients with type 2 diabetes mellitus. Diabetes Educ. 2012, 38, 409–416. [Google Scholar] [CrossRef]
- Pinto, S.L.; Kumar, J.; Partha, G.; Bechtol, R.A. Pharmacist-provided medication therapy management (MTM) program impacts outcomes for employees with diabetes. Popul. Health Manag. 2014, 17, 21–27. [Google Scholar] [CrossRef]
- Planas, L.G.; Crosby, K.M.; Farmer, K.C.; Harrison, D.L. Evaluation of a diabetes management program using selected HEDIS measures. J. Am. Pharm. Assoc. 2012, 52, e130–e138. [Google Scholar] [CrossRef]
- Rodis, J.L.; Sevin, A.; Awad, M.H.; Porter, B.; Glasgow, K.; Fox, C.H.; Pryor, B. Improving chronic disease outcomes through medication therapy management in Federally Qualified Health Centers. J. Prim. Care Community Health 2017, 8, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez de Bittner, M.; Chirikov, V.V.; Breunig, I.M.; Zaghab, R.W.; Shaya, F.T. Clinical effectiveness and cost savings in diabetes care, supported by pharmacist counselling. J. Am. Pharm. Assoc. 2017, 57, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Ross, L.A.; Bloodworth, L.S.; Brown, M.A.; Malinowski, S.S.; Crane, R.; Sutton, V.; Karimi, M.; Brown, A.C.D.; Dobbs, T.; Hites, L. The Mississippi Delta Health Collaborative Medication Therapy Management Model: Public health and pharmacy working together to improve population health in the Mississippi Delta. Prev. Chronic Dis. 2020, 17, e108. [Google Scholar] [CrossRef] [PubMed]
- Schultz, B.G.; Tilton, J.; Jun, J.; Scott-Horton, T.; Quach, D.; Touchette, D.R. Cost-effectiveness analysis of a pharmacist-led medication therapy management program: Hypertension management. Value Health 2021, 24, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Sease, J.M.; Franklin, M.A.; Gerrald, K.R. Pharmacist management of patients with diabetes mellitus enrolled in a rural free clinic. Am. J. Health Syst. Pharm. 2012, 70, 43–47. [Google Scholar] [CrossRef]
- Sharp, L.K.; Tilton, J.J.; Touchette, D.R.; Xia, Y.; Mihailescu, D.; Berbaum, M.L.; Gerber, B.S. Community health workers supporting clinical pharmacists in diabetes management: A randomized controlled trial. Pharmacotherapy 2018, 38, 58–68. [Google Scholar] [CrossRef]
- Smith, S.M.; Carris, N.W.; Dietrich, E.; Gums, J.G.; Uribe, L.; Coffey, C.S.; Gums, T.H.; Carter, B.L. Physician-pharmacist collaboration versus usual care for treatment-resistant hypertension. J. Am. Soc. Hypertens. 2016, 10, 307–317. [Google Scholar] [CrossRef]
- Steele, K.M.; Ruisinger, J.F.; Bates, J.; Prohaska, E.S.; Melton, B.L.; Hipp, S. Home-based comprehensive medication reviews: Pharmacist’s Impact on drug therapy problems in geriatric patients. Consult. Pharm. 2016, 31, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Taveira, T.H.; Friedmann, P.D.; Cohen, L.B.; Dooley, A.G.; Khatana, S.A.; Pirraglia, P.A.; Wu, W.C. Pharmacist-led group medical appointment model in type 2 diabetes. Diabetes Educ. 2010, 36, 109–117. [Google Scholar] [CrossRef]
- Taveira, T.H.; Dooley, A.G.; Cohen, L.B.; Khatana, S.A.M.; Wu, W.-C. Pharmacist-led group medical appointments for the management of type 2 diabetes with comorbid depression in older adults. Ann. Pharmacother. 2011, 45, 1346–1355. [Google Scholar] [CrossRef]
- Taveira, T.H.; Wu, W.C. Interventions to maintain cardiac risk control after discharge from a cardiovascular risk reduction clinic: A randomized controlled trial. Diabetes Res. Clin. Pract. 2014, 105, 327–335. [Google Scholar] [CrossRef]
- Thompson, H.; Swander, L.; Cohen, R.; Lukazewski, A.; Bartholow, T.; Pesik, M.; Trapskin, K. Hypertension-focused medication therapy management: A collaborative pilot program uniting pharmacists, public health, and health insurers in Wisconsin. Prev. Chronic Dis. 2020, 17, e105. [Google Scholar] [CrossRef] [PubMed]
- Tilton, J.J.; Edakkunnathu, M.G.; Moran, K.M.; Markel Vaysman, A.; DaPisa, J.L.; Goen, B.M.; Touchette, D.R. Impact of a medication therapy management clinic on glycosylated hemoglobin, blood pressure, and resource utilization. Ann. Pharmacother. 2019, 53, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Victor, R.G.; Lynch, K.; Li, N.; Blyler, C.; Muhammad, E.; Handler, J.; Brettler, J.; Rashid, M.; Hsu, B.; Foxx-Drew, D.; et al. A cluster-randomized trial of blood-pressure reduction in black barbershops. N. Engl. J. Med. 2018, 378, 1291–1301. [Google Scholar] [CrossRef]
- Victor, R.G.; Blyler, C.A.; Li, N.; Lynch, K.; Moy, N.B.; Rashid, M.; Chang, L.C.; Handler, J.; Brettler, J.; Rader, F.; et al. Sustainability of blood pressure reduction in black barbershops. Circulation 2019, 139, 10–19. [Google Scholar] [CrossRef]
- Wentzlaff, D.M.; Carter, B.L.; Ardery, G.; Franciscus, C.L.; Doucette, W.R.; Chrischilles, E.A.; Rosenkrans, K.A.; Buys, L.M. Sustained blood pressure control following discontinuation of a pharmacist intervention. J. Clin. Hypertens. 2011, 13, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Wheat, L.; Roane, T.E.; Connelly, A.; Zeigler, M.; Wallace, J.; Kim, J.H.; Segal, R. Using a pharmacist-community health worker collaboration to address medication adherence barriers. J. Am. Pharm. Assoc. 2020, 60, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Wittayanukorn, S.; Westrick, S.C.; Hansen, R.A.; Billor, N.; Braxton-Lloyd, K.; Fox, B.I.; Garza, K.B. Evaluation of medication therapy management services for patients with cardiovascular disease in a self-insured employer health plan. J. Manag. Care Pharm. 2013, 19, 385–395. [Google Scholar] [CrossRef]
- Wu, W.C.; Taveira, T.H.; Jeffery, S.; Jiang, L.; Tokuda, L.; Musial, J.; Cohen, L.B.; Uhrle, F. Costs and effectiveness of pharmacist-led group medical visits for type-2 diabetes: A multi-center randomized controlled trial. PLoS ONE 2018, 13, e0195898. [Google Scholar] [CrossRef]
- Yu, J.; Shah, B.M.; Ip, E.J.; Chan, J. A Markov model of the cost-effectiveness of pharmacist care for diabetes in prevention of cardiovascular diseases: Evidence from Kaiser Permanente Northern California. J. Manag. Care Pharm. 2013, 19, 102–114. [Google Scholar] [CrossRef] [PubMed]
- Santschi, V.; Chiolero, A.; Colosimo, A.L.; Platt, R.W.; Taffé, P.; Burnier, M.; Burnand, B.; Paradis, G. Improving blood pressure control through pharmacist interventions: A meta-analysis of randomized controlled trials. J. Am. Heart Assoc. 2014, 3, e000718. [Google Scholar] [CrossRef]
- McFarland, M.S.; Buck, M.L.; Crannage, E.; Armistead, L.T.; Ourth, H.; Finks, S.W.; McClurg, M.R. Assessing the impact of comprehensive medication management on achievement of the quadruple aim. Am. J. Med. 2021, 134, 456–461. [Google Scholar] [CrossRef]
- Flint, A.C.; Conell, C.; Ren, X.; Banki, N.M.; Chan, S.L.; Rao, V.A.; Melles, R.B.; Bhatt, D.L. Effect of systolic and diastolic blood pressure on cardiovascular outcomes. N. Engl. J. Med. 2019, 381, 243–251. [Google Scholar] [CrossRef]
- Levene, J.; Voigt, A.; Thoma, F.; Mulukutla, S.; Bhonsale, A.; Kancharla, K.; Shalaby, A.; Estes, N.A.M., 3rd; Jain, S.; Saba, S. Patient outcomes by ventricular systolic and diastolic function. J. Am. Heart Assoc. 2024, 13, e033211. [Google Scholar] [CrossRef] [PubMed]
- Milosavljevic, A.; Aspden, T.; Harrison, J. Community pharmacist-led interventions and their impact on patients’ medication adherence and other health outcomes: A systematic review. Int. J. Pharm. Pract. 2018, 5, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Blalock, S.J.; Roberts, A.W.; Lauffenburger, J.C.; Thompson, T.; O’Connor, S.K. The effect of community pharmacy-based interventions on patient health outcomes: A systematic review. Med. Care Res. Rev. 2013, 70, 235–266. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, M.; Kahwati, L.C.; Golin, C.E.; Blalock, S.J.; Coker-Schwimmer, E.; Posey, R.; Lohr, K.N. Medication therapy management interventions in outpatient settings: A systematic review and meta-analysis. JAMA Intern. Med. 2015, 175, 76–87. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).