Trefoil Factor Family (TFF) Peptides
Definition
1. Introduction or History
2. Structure and Expression
2.1. Genomic Organization, Structure and Natural Forms of TFF Peptides
2.2. Mosaic Proteins Containing TFF Domains
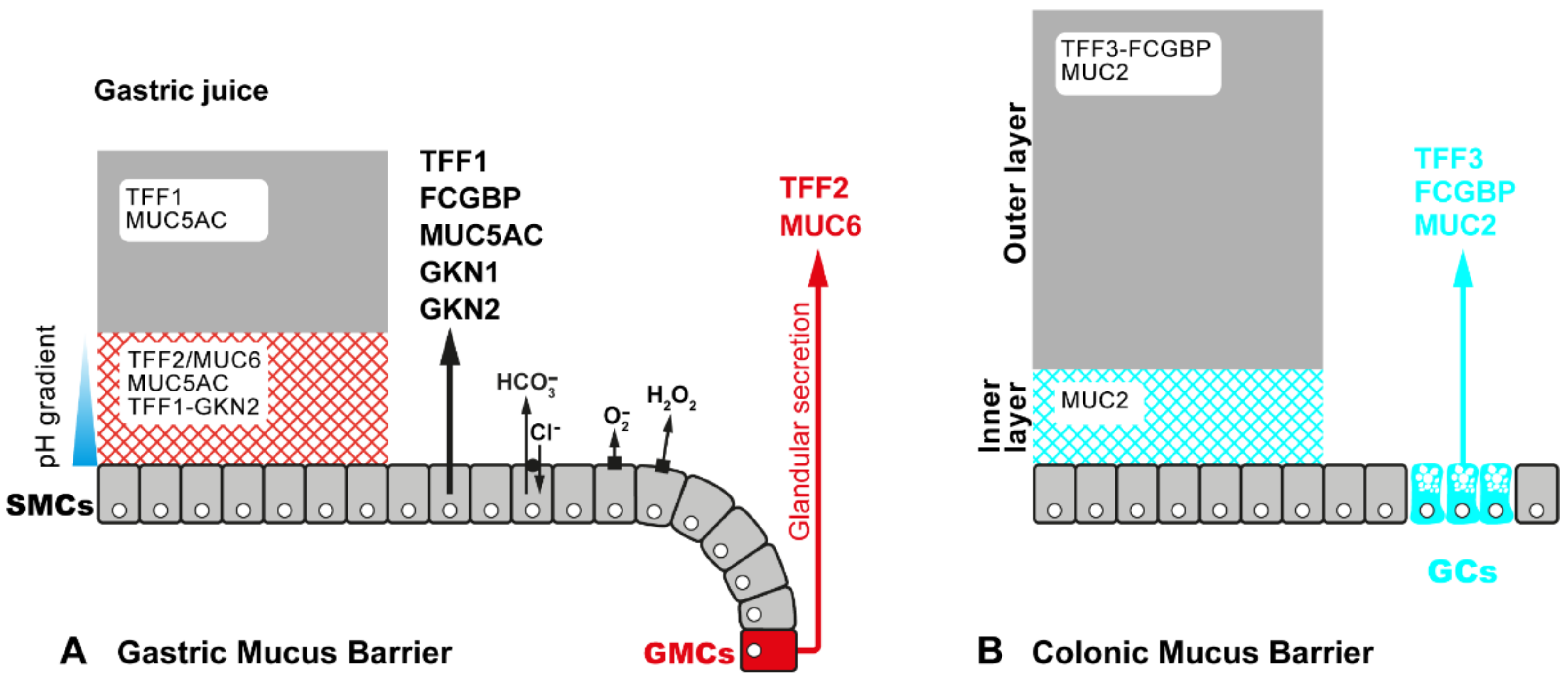
2.3. Exocrine Secretion of TFF Peptides in Mucous Epithelia
2.4. Endocrine Secretion of TFF Peptides
2.5. Pathological Expression of TFF Peptides: Links to Inflammation and Cancer
3. Functional Aspects
3.1. TFF Domains Have Different Lectin Activities
3.2. Exocrine TFF Peptides Occur in Different Molecular Forms and Have Diverse Molecular Functions
3.3. Tff-Deficient (TffKO) Mice Have Different Phenotypes: Functional Implications
3.3.1. TFF1 Is a Gastric Tumor Suppressor
3.3.2. TFF2: Component of the Gastric Mucus Barrier, Anti-Inflammatory Peptide, and Induction of IL-33 (Promotion of Th2 Immunity)
3.3.3. TFF3: Component of the Intestinal Mucus Barrier
3.4. TFF Peptides Weakly Enhance Cell Migration: TFF Binding Sites and Hypothetical Lectin-Triggered Activation of Transmembrane Glycoproteins
3.5. Lectin-Triggered Receptor Blocking Hypothesis (Anti-Inflammatory Action)
4. Medical Perspectives
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Entry Link on the Encyclopedia Platform
Abbreviations
| A4GNT | α1,4-N-acetylglucosaminyltransferase |
| DSS | Dextran sulfate sodium |
| ER | Endoplasmic reticulum |
| FCGBP | IgG Fc binding protein |
| IL | Interleukin |
| ROS | Reactive oxygen species |
| TFF | Trefoil factor family |
| TLR | Toll-like receptor |
References
- Thim, L. A new family of growth factor-like peptides. ‘Trefoil’ disulphide loop structures as a common feature in breast cancer associated peptide (pS2), pancreatic spasmolytic polypeptide (PSP), and frog skin peptides (spasmolysins). FEBS Lett. 1989, 250, 85–90. [Google Scholar] [CrossRef]
- Poulsom, R.; Wright, N.A. Trefoil peptides: A newly recognized family of epithelial mucin-associated molecules. Am. J. Physiol. Liver Physiol. 1993, 265, G205–G213. [Google Scholar] [CrossRef]
- Hoffmann, W.; Hauser, F. The P-domain or trefoil motif: A role in renewal and pathology of mucous epithelia? Trends Biochem. Sci. 1993, 18, 239–243. [Google Scholar] [CrossRef]
- Sands, B.E.; Podolsky, D.K. The trefoil peptide family. Annu. Rev. Physiol. 1996, 58, 253–273. [Google Scholar] [CrossRef] [PubMed]
- Thim, L. Trefoil peptides: From structure to function. Cell. Mol. Life Sci. 1997, 53, 888. [Google Scholar] [CrossRef]
- Ribieras, S.; Tomasetto, C.; Rio, M.C. The pS2/TFF1 trefoil factor, from basic research to clinical applications. Biochim. Biophys. Acta 1998, 1378, F61–F77. [Google Scholar] [CrossRef]
- Wong, W.M.; Poulsom, R.; Wright, N.A. Trefoil peptides. Gut 1999, 44, 890–895. [Google Scholar] [CrossRef]
- Taupin, D.; Podolsky, D.K. Trefoil factors: Initiators of mucosal healing. Nat. Rev. Mol. Cell Biol. 2003, 4, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Thim, L.; May, F.E.B. Structure of mammalian trefoil factors and functional insights. Cell. Mol. Life Sci. 2005, 62, 2956–2973. [Google Scholar] [CrossRef] [PubMed]
- Kjellev, S. The trefoil factor family—Small peptides with multiple functionalities. Cell. Mol. Life Sci. 2008, 66, 1350–1369. [Google Scholar] [CrossRef]
- Aihara, E.; Engevik, K.A.; Montrose, M.H. Trefoil factor peptides and gastrointestinal function. Annu. Rev. Physiol. 2017, 79, 357–380. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, W. Trefoil factor family (TFF) peptides and their diverse molecular functions in mucus barrier protection and more: Changing the paradigm. Int. J. Mol. Sci. 2020, 21, 4535. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, W. Trefoil factor family (TFF) peptides and their links to inflammation: A re-evaluation and new medical perspectives. Int. J. Mol. Sci. 2021, 22, 4909. [Google Scholar] [CrossRef]
- Hoffmann, W. Trefoil factor family (TFF) peptides and their different roles in the mucosal innate immune defense and more: An update. Curr. Med. Chem. 2021, 28. [Google Scholar] [CrossRef] [PubMed]
- Masiakowski, P.; Breathnach, R.; Bloch, J.; Gannon, F.; Krust, A.; Chambon, P. Cloning of cDNA sequences of hor-mone-regulated genes from the MCF-7 human breast cancer cell line. Nucleic Acids Res. 1982, 10, 7895–7903. [Google Scholar] [CrossRef]
- Jørgensen, K.H.; Thim, L.; Jacobsen, H.E. Pancreatic spasmolytic polypeptide (PSP): I. Preparation and initial chemical char-acterization of a new polypeptide from porcine pancreas. Regul. Pept. 1982, 3, 207–219. [Google Scholar] [CrossRef]
- Hoffmann, W. A new repetitive protein from Xenopus laevis skin highly homologous to pancreatic spasmolytic polypeptide. J. Biol. Chem. 1988, 263, 7686–7690. [Google Scholar] [CrossRef]
- Suemori, S.; Lynch-Devaney, K.; Podolsky, D.K. Identification and characterization of rat intestinal trefoil factor: Tissue and cell-specific member of the trefoil protein family. Proc. Natl. Acad. Sci. USA 1991, 88, 11017–11021. [Google Scholar] [CrossRef]
- Hauser, F.; Poulsom, R.; Chinery, R.; Rogers, L.A.; Hanby, A.M.; Wright, N.; Hoffmann, W. hP1.B, a human P-domain peptide homologous with rat intestinal trefoil factor, is expressed also in the ulcer-associated cell lineage and the uterus. Proc. Natl. Acad. Sci. USA 1993, 90, 6961–6965. [Google Scholar] [CrossRef]
- Podolsky, D.; Lynch-Devaney, K.; Stow, J.; Oates, P.; Murgue, B.; DeBeaumont, M.; Sands, B.; Mahida, Y. Identification of human intestinal trefoil factor. Goblet cell-specific expression of a peptide targeted for apical secretion. J. Biol. Chem. 1993, 268, 6694–6702. [Google Scholar] [CrossRef]
- Hoffmann, W.; Jagla, W. Cell type specific expression of secretory TFF peptides: Colocalization with mucins and synthesis in the brain. Int. Rev. Cytol. 2002, 213, 147–181, 182e–188e. [Google Scholar] [CrossRef]
- Wright, N.; Hoffmann, W.; Otto, W.; Rio, M.-C.; Thim, L. Rolling in the clover: Trefoil factor family (TFF)-domain peptides, cell migration and cancer. FEBS Lett. 1997, 408, 121–123. [Google Scholar] [CrossRef]
- Sommer, P.; Blin, N.; Gött, P. Tracing the evolutionary origin of the TFF-domain, an ancient motif at mucous surfaces. Gene 1999, 236, 133–136. [Google Scholar] [CrossRef]
- Hoffmann, W. TFF Peptides. In Handbook of Biologically Active Peptides, 2nd ed.; Karstin, A.J., Ed.; Academic Press: Boston, MA, USA, 2013; pp. 1338–1345. [Google Scholar]
- Heuer, F.; Stürmer, R.; Heuer, J.; Kalinski, T.; Lemke, A.; Meyer, F.; Hoffmann, W. Different Forms of TFF2, A Lectin of the Human Gastric Mucus Barrier: In vitro binding studies. Int. J. Mol. Sci. 2019, 20, 5871. [Google Scholar] [CrossRef]
- Znalesniak, E.B.; Salm, F.; Hoffmann, W. Molecular alterations in the stomach of Tff1-deficient mice: Early steps in antral carcinogenesis. Int. J. Mol. Sci. 2020, 21, 644. [Google Scholar] [CrossRef] [PubMed]
- Hanisch, F.-G.; Ragge, H.; Kalinski, T.; Meyer, F.; Kalbacher, H.; Hoffmann, W. Human gastric TFF2 peptide contains an N-linked fucosylated N,N’-diacetyllactosediamine (LacdiNAc) oligosaccharide. Glycobiology 2012, 23, 2–11. [Google Scholar] [CrossRef][Green Version]
- Stürmer, R.; Reising, J.; Hoffmann, W. The TFF peptides xP1 and xP4 appear in distinctive forms in the Xenopus laevis gastric mucosa: Indications for different protective functions. Int. J. Mol. Sci. 2019, 20, 6052. [Google Scholar] [CrossRef]
- Heuer, J.; Heuer, F.; Stürmer, R.; Harder, S.; Schlüter, H.; Emidio, N.B.; Muttenthaler, M.; Jechorek, D.; Meyer, F.; Hoffmann, W. The tumor suppressor TFF1 occurs in different forms and interacts with multiple partners in the human gastric mucus barrier: Indications for diverse protective functions. Int. J. Mol. Sci. 2020, 21, 2508. [Google Scholar] [CrossRef]
- Westley, B.R.; Griffin, S.M.; May, F.E.B. Interaction between TFF1, a gastric tumor suppressor trefoil protein, and TFIZ1, a brichos domain-containing protein with homology to SP-C. Biochemistry 2005, 44, 7967–7975. [Google Scholar] [CrossRef]
- Kouznetsova, I.; Laubinger, W.; Kalbacher, H.; Kalinski, T.; Meyer, F.; Roessner, A.; Hoffmann, W. Biosynthesis of gastrokine-2 in the human gastric mucosa: Restricted spatial expression along the antral gland axis and differential interaction with TFF1, TFF2 and mucins. Cell. Physiol. Biochem. 2007, 20, 899–908. [Google Scholar] [CrossRef]
- Stürmer, R.; Müller, S.; Hanisch, F.-G.; Hoffmann, W. Porcine gastric TFF2 is a mucus constituent and differs from pancreatic TFF2. Cell. Physiol. Biochem. 2014, 33, 895–904. [Google Scholar] [CrossRef]
- Stürmer, R.; Harder, S.; Schlüter, H.; Hoffmann, W. Commercial porcine gastric mucin preparations, also used as artificial saliva, are a rich source for the lectin TFF2: In vitro binding studies. ChemBioChem 2018, 19, 2598–2608. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, W. TFF2, a MUC6-binding lectin stabilizing the gastric mucus barrier and more (Review). Int. J. Oncol. 2015, 47, 806–816. [Google Scholar] [CrossRef]
- Albert, T.K.; Laubinger, W.; Müller, S.; Hanisch, F.-G.; Kalinski, T.; Meyer, F.; Hoffmann, W. Human Intestinal TFF3 Forms disulfide-linked heteromers with the mucus-associated FCGBP protein and is released by hydrogen sulfide. J. Proteome Res. 2010, 9, 3108–3117. [Google Scholar] [CrossRef]
- Houben, T.; Harder, S.; Schlüter, H.; Kalbacher, H.; Hoffmann, W. Different forms of TFF3 in the human saliva: Heterodi-merization with IgG Fc binding protein (FCGBP). Int. J. Mol. Sci. 2019, 20, 5000. [Google Scholar] [CrossRef]
- Hauser, F.; Hoffmann, W. xP1 and xPP-domain peptides expressed in Xenopus laevis stomach mucosa. J. Biol. Chem. 1991, 266, 21306–21309. [Google Scholar] [CrossRef]
- Jagla, W.; Wiede, A.; Hoffmann, W.; Kölle, S. Differential expression of the TFF-peptides xP1 and xP4 in the gastrointestinal tract of Xenopus laevis. Cell Tissue Res. 1997, 291, 13–18. [Google Scholar] [CrossRef]
- Botzler, C.; Oertel, M.; Hinz, M.; Hoffmann, W. Structure of the Xenopus laevis TFF-gene xP4.1, differentially expressed to its duplicated homolog xP4. Biochim. Biophys. Acta (BBA)-Gene Struct. Expr. 1999, 1489, 345–353. [Google Scholar] [CrossRef]
- Hoffmann, W.; Jagla, W.; Wiede, A. Molecular medicine of TFF-peptides: From gut to brain. Histol. Histopathol. 2001, 16, 319–334. [Google Scholar] [PubMed]
- Madsen, J.; Nielsen, O.; Tornøe, I.; Thim, L.; Holmskov, U. Tissue localization of human trefoil factors 1, 2, and 3. J. Histochem. Cytochem. 2007, 55, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Cook, G.; Familari, M.; Thim, L.; Giraud, A. The trefoil peptides TFF2 and TFF3 are expressed in rat lymphoid tissues and participate in the immune response. FEBS Lett. 1999, 456, 155–159. [Google Scholar] [CrossRef]
- Baus-Loncar, M.; Kayademir, T.; Takaishi, S.; Wang, T. Trefoil factor family 2 deficiency and immune response. Cell. Mol. Life Sci. 2005, 62, 2947–2955. [Google Scholar] [CrossRef] [PubMed]
- Kurt-Jones, E.A.; Cao, L.; Sandor, F.; Rogers, A.B.; Whary, M.T.; Nambiar, P.R.; Cerny, A.; Bowen, G.; Yan, J.; Takaishi, S.; et al. Trefoil family factor 2 is expressed in murine gastric and immune cells and controls both gastrointestinal inflammation and systemic immune responses. Infect. Immun. 2007, 75, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Probst, J.C.; Skutella, T.; Müller-Schmid, A.; Jirikowski, G.F.; Hoffmann, W. Molecular and cellular analysis of rP1.B in the rat hypothalamus: In situ hybridization and immunohistochemistry of a new P-domain neuropeptide. Mol. Brain Res. 1995, 33, 269–276. [Google Scholar] [CrossRef]
- Wright, N.A. Trefoil peptides and the gut. Gut 1993, 34, 577–579. [Google Scholar] [CrossRef][Green Version]
- Wong, W.M.; Playford, R.J.; Wright, N.A. Peptide gene expression in gastrointestinal mucosal ulceration: Ordered sequence or redundancy? Gut 2000, 46, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, W. TFF (trefoil factor family) peptides and their potential roles for differentiation processes during airway remod-eling. Curr. Med. Chem. 2007, 14, 2716–2719. [Google Scholar] [CrossRef]
- Goldenring, J.R.; Nam, K.T.; Wang, T.C.; Mills, J.C.; Wright, N. Spasmolytic polypeptide-expressing metaplasia and intestinal metaplasia: Time for reevaluation of metaplasias and the origins of gastric cancer. Gastroenterology 2010, 138, 2207–2210.e1. [Google Scholar] [CrossRef]
- May, F.E.; Westley, B.R. Trefoil proteins: Their role in normal and malignant cells. J. Pathol. 1997, 183, 4–7. [Google Scholar] [CrossRef]
- Katoh, M. Trefoil factors and human gastric cancer (Review). Int. J. Mol. Med. 2003, 12, 3–9. [Google Scholar] [CrossRef]
- Emami, S.; Rodrigues, S.; Rodrigue, C.M.; Le Floch, N.; Rivat, C.; Attoub, S.; Bruyneel, E.; Gespach, C. Trefoil factor family (TFF) peptides and cancer progression. Peptides 2004, 25, 885–898. [Google Scholar] [CrossRef] [PubMed]
- Regalo, G.; Wright, N.A.; Machado, J.C. Trefoil factors: From ulceration to neoplasia. Cell. Mol. Life Sci. 2005, 62, 2910–2915. [Google Scholar] [CrossRef] [PubMed]
- Perry, J.K.; Kannan, N.; Grandison, P.M.; Mitchell, M.D.; Lobie, P.E. Are trefoil factors oncogenic? Trends Endocrinol. Metab. 2008, 19, 74–81. [Google Scholar] [CrossRef]
- Beck, S.; Sommer, P.; Blin, N.; Gött, P. 5′-flanking motifs control cell-specific expression of trefoil factor genes (TFF). Int. J. Mol. Med. 1998, 2, 353–414. [Google Scholar] [CrossRef] [PubMed]
- Baus-Loncar, M.; Giraud, A.S. Multiple regulatory pathways for trefoil factor (TFF) genes. Cell. Mol. Life Sci. 2005, 62, 2921–2931. [Google Scholar] [CrossRef]
- Giraud, A.S.; Jackson, C.; Menheniott, T.R.; Judd, L.M. Differentiation of the gastric mucosa IV. Role of trefoil peptides and IL-6 cytokine family signaling in gastric homeostasis. Am. J. Physiol. Liver Physiol. 2007, 292, G1–G5. [Google Scholar] [CrossRef]
- Järvå, M.A.; Lingford, J.; John, A.; Soler, N.M.; Scott, N.E.; Goddard-Borger, E.D. Trefoil factors share a lectin activity that defines their role in mucus. Nat. Commun. 2020, 11, 2265. [Google Scholar] [CrossRef] [PubMed]
- Reeves, E.P.; Ali, T.; Leonard, P.; Hearty, S.; O’Kennedy, R.; May, F.E.B.; Westley, B.R.; Josenhans, C.; Rust, M.; Suerbaum, S.; et al. Helicobacter pylori lipopolysaccharide interacts with TFF1 in a pH-dependent manner. Gastroenterology 2008, 135, 2043–2054. [Google Scholar] [CrossRef]
- Clyne, M.; May, F.E.B. The interaction of helicobacter pylori with TFF1 and its role in mediating the tropism of the bacteria within the stomach. Int. J. Mol. Sci. 2019, 20, 4400. [Google Scholar] [CrossRef] [PubMed]
- Emidio, N.B.; Baik, H.; Lee, D.; Stuermer, R.; Heuer, J.; Elliott, A.G.; Blaskovich, M.A.T.; Haupenthal, K.; Tegtmeyer, N.; Hoffmann, W.; et al. Chemical synthesis of human trefoil factor 1 (TFF1) and its homodimer provides novel insights into their mechanisms of action. Chem. Commun. 2020, 56, 6420–6423. [Google Scholar] [CrossRef] [PubMed]
- Hanisch, F.-G.; Bonar, D.; Schloerer, N.; Schroten, H. Human trefoil factor 2 is a lectin that binds α-GlcNAc-capped mucin glycans with antibiotic activity against Helicobacter pylori. Cell. Physiol. Biochem. 2014, 289, 27363–27375. [Google Scholar]
- Stürmer, R.; Reising, J.; Hoffmann, W. Trefoil factor family (TFF) modules are characteristic constituents of separate mucin complexes in the xenopus laevis integumentary mucus: In vitro binding studies with FIM-A. 1. Int. J. Mol. Sci. 2020, 21, 2400. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, H.; Hoffmann, W. Subcellular localization of the TFF peptides xP1 and xP4 in the Xenopus laevis gastric/esophageal mucosa: Different secretion modes reflecting diverse protective functions. Int. J. Mol. Sci. 2020, 21, 761. [Google Scholar] [CrossRef] [PubMed]
- Thim, L.; Madsen, F.; Poulsen, S.S. Effect of trefoil factors on the viscoelastic properties of mucus gels. Eur. J. Clin. Investig. 2002, 32, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Lang, T.; Klasson, S.; Larsson, E.; Johansson, M.E.; Hansson, G.C.; Samuelsson, T. Searching the evolutionary origin of epi-thelial mucus protein components-mucins and FCGBP. Mol. Biol. Evol. 2016, 33, 1921–1936. [Google Scholar] [CrossRef]
- Lencer, W.I.; Blumberg, R.S. A passionate kiss, then run: Exocytosis and recycling of IgG by FcRn. Trends Cell Biol. 2005, 15, 5–9. [Google Scholar] [CrossRef]
- Schwartz, J.L. Fcgbp—A potential viral trap in RV144. Open AIDS J. 2014, 8, 21–24. [Google Scholar] [CrossRef]
- Madsen, J.; Sørensen, G.L.; Nielsen, O.S.; Tornøe, I.; Thim, L.; Fenger, C.; Mollenhauer, J.; Holmskov, U. A variant form of the human deleted in malignant brain tumor 1 (DMBT1) gene shows increased expression in inflammatory bowel diseases and interacts with dimeric trefoil factor 3 (TFF3). PLoS ONE 2013, 8, e64441. [Google Scholar] [CrossRef]
- Madsen, J.; Mollenhauer, J.; Holmskov, U. Review: Gp-340/DMBT1 in mucosal innate immunity. Innate Immun. 2010, 16, 160–167. [Google Scholar] [CrossRef]
- Tomasetto, C.; Rio, M.C. Pleiotropic effects of Trefoil Factor 1 deficiency. Cell. Mol. Life Sci. 2005, 62, 2916–2920. [Google Scholar] [CrossRef]
- Lefebvre, O.; Chenard, M.-P.; Masson, R.; Linares, J.; Dierich, A.; LeMeur, M.; Wendling, C.; Tomasetto, C.; Chambon, P.; Rio, M.-C. Gastric mucosa abnormalities and tumorigenesis in mice lacking the pS2 trefoil protein. Science 1996, 274, 259–262. [Google Scholar] [CrossRef] [PubMed]
- Soutto, M.; Belkhiri, A.; Piazuelo, M.B.; Schneider, B.G.; Peng, D.; Jiang, A.; Washington, M.K.; Kokoye, Y.; Crowe, S.E.; Zaika, A.; et al. Loss of TFF1 is associated with activation of NF-kappaB-mediated inflammation and gastric neoplasia in mice and humans. J. Clin. Investig. 2011, 121, 1753–1767. [Google Scholar] [CrossRef]
- Saukkonen, K.; Tomasetto, C.; Narko, K.; Rio, M.-C.; Ristimäki, A. Cyclooxygenase-2 expression and effect of celecoxib in gastric adenomas of trefoil factor 1-deficient mice. Cancer Res. 2003, 63, 3032–3036. [Google Scholar]
- Thiem, S.; Eissmann, M.F.; Elzer, J.; Jonas, A.; Putoczki, T.L.; Poh, A.; Nguyen, P.; Preaudet, A.; Flanagan, D.; Vincan, E.; et al. Stomach-specific activation of oncogenic KRAS and STAT3-dependent in-flammation cooperatively promote gastric tumorigenesis in a preclinical model. Cancer Res. 2016, 76, 2277–2287. [Google Scholar] [CrossRef] [PubMed]
- Torres, L.-F.; Karam, S.M.; Wendling, C.; Chenard, M.-P.; Kershenobich, D.; Tomasetto, C.; Rio, M.-C. TreFoil factor 1 (TFF1/pS2) deficiency activates the unfolded protein response. Mol. Med. 2002, 8, 273–282. [Google Scholar] [CrossRef]
- Farrell, J.J.; Taupin, D.; Koh, T.J.; Chen, D.; Zhao, C.M.; Podolsky, D.K.; Wang, T.C. TFF2/SP-deficient mice show decreased gastric proliferation, increased acid secretion, and increased susceptibility to NSAID injury. J. Clin. Investig. 2002, 109, 193–204. [Google Scholar] [CrossRef]
- Fox, J.G.; Rogers, A.B.; Whary, M.T.; Ge, Z.; Ohtani, M.; Jones, E.K.; Wang, T.C. Accelerated progression of gastritis to dysplasia in the pyloric antrum of TFF2-/-C57BL6 x Sv129 Helicobacter pylori-infected mice. Am. J. Pathol. 2007, 171, 1520–1528. [Google Scholar] [CrossRef] [PubMed]
- McBerry, C.; Egan, C.E.; Rani, R.; Yang, Y.; Wu, D.; Boespflug, N.; Boon, L.; Butcher, B.; Mirpuri, J.; Hogan, S.P.; et al. Trefoil Factor 2 negatively regulates Type 1 immunity against toxoplasma gondii. J. Immunol. 2012, 189, 3078–3084. [Google Scholar] [CrossRef] [PubMed]
- Wills-Karp, M.; Rani, R.; Dienger, K.; Lewkowich, I.; Fox, J.G.; Perkins, C.; Lewis, L.; Finkelman, F.D.; Smith, D.E.; Bryce, P.J.; et al. Trefoil factor 2 rapidly induces interleukin 33 to promote type 2 immunity during allergic asthma and hookworm infection. J. Exp. Med. 2012, 209, 607–622. [Google Scholar] [CrossRef]
- Buzzelli, J.N.; Chalinor, H.V.; Pavlic, D.I.; Sutton, P.; Menheniott, T.R.; Giraud, A.S.; Judd, L.M. IL33 is a stomach alarmin that initiates a skewed Th2 response to injury and infection. Cell. Mol. Gastroenterol. Hepatol. 2015, 1, 203–221.e3. [Google Scholar] [CrossRef]
- Mashimo, H.; Wu, D.-C.; Podolsky, D.K.; Fishman, M.C. Impaired defense of intestinal mucosa in mice lacking intestinal trefoil factor. Science 1996, 274, 262–265. [Google Scholar] [CrossRef]
- Beck, P.L.; Wong, J.F.; Li, Y.; Swaminathan, S.; Xavier, R.J.; Devaney, K.L.; Podolsky, D.K. Chemotherapy-and radiothera-py-induced intestinal damage is regulated by intestinal trefoil factor. Gastroenterology 2004, 126, 796–808. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.E.V.; Gustafsson, J.K.; Sjöberg, K.E.; Petersson, J.; Holm, L.; Sjövall, H.; Hansson, G.C. Bacteria penetrate the inner mucus layer before inflammation in the dextran sulfate colitis model. PLoS ONE 2010, 5, e12238. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, W. TFF (trefoil factor family) peptide-triggered signals promoting mucosal restitution. Cell. Mol. Life Sci. 2005, 62, 2932–2938. [Google Scholar] [CrossRef]
- Cho, S.Y.; Klemke, R.L. Extracellular-regulated kinase activation and Cas/Crk coupling regulate cell migration and suppress apoptosis during invasion of the extracellular matrix. J. Cell Biol. 2000, 149, 223–236. [Google Scholar] [CrossRef]
- Otto, W.R.; Thim, L. Trefoil factor family-interacting proteins. Cell. Mol. Life Sci. 2005, 62, 2939–2946. [Google Scholar] [CrossRef]
- Tajadura-Ortega, V.; Gambardella, G.; Skinner, A.; Halim, A.; Coillie, J.V.; Schjoldager, K.T.-B.G.; Beatson, R.; Graham, R.; Achkova, D.; Taylor-Papadimitriou, J.; et al. O-linked mucin-type glycosylation regulates the tran-scriptional programme downstream of EGFR. Glycobiology 2021, 31, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, S.S.; Thulesen, J.; Nexø, E.; Thim, L. Distribution and metabolism of intravenously administered trefoil factor 2/porcine spasmolytic polypeptide in the rat. Gut 1998, 43, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, S.S.; Thulesen, J.; Hartmann, B.; Kissow, H.; Nexø, E.; Thim, L. Injected TFF1 and TFF3 bind to TFF2-immunoreactive cells in the gastrointestinal tract in rats. Regul. Pept. 2003, 115, 91–99. [Google Scholar] [CrossRef]
- Nakayama, J. Dual roles of gastric gland mucin-specific o-glycans in prevention of gastric cancer. Acta Histochem. Cytochem. 2014, 47, 1–9. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Emidio, N.B.; Brierley, S.M.; Schroeder, C.I.; Muttenthaler, M. Structure, function, and therapeutic potential of the trefoil factor family in the gastrointestinal tract. ACS Pharmacol. Transl. Sci. 2020, 3, 583–597. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, W. Trefoil factor family (TFF) peptides and chemokine receptors: A promising relationship. J. Med. Chem. 2009, 52, 6505–6510. [Google Scholar] [CrossRef]
- Dubeykovskaya, Z.; Si, Y.; Chen, X.; Worthley, D.L.; Renz, B.W.; Urbanska, A.M.; Hayakawa, Y.; Xu, T.; Westphalen, C.B.; Dubeykovskiy, A.; et al. Neural innervation stimulates splenic TFF2 to arrest myeloid cell expansion and cancer. Nat. Commun. 2016, 7, 10517. [Google Scholar] [CrossRef] [PubMed]
- Porębska, N.; Poźniak, M.; Matynia, A.; Żukowska, D.; Zakrzewska, M.; Otlewski, J.; Opaliński, Ł. Galectins as modulators of receptor tyrosine kinases signaling in health and disease. Cytokine Growth Factor Rev. 2021, 60, 89–106. [Google Scholar] [CrossRef] [PubMed]
- Soutto, M.; Chen, Z.; Bhat, A.A.; Wang, L.; Zhu, S.; Gomaa, A.; Bates, A.; Bhat, N.S.; Peng, D.; Belkhiri, A.; et al. Activation of STAT3 signaling is mediated by TFF1 silencing in gastric neoplasia. Nat. Commun. 2019, 10, 3039. [Google Scholar] [CrossRef] [PubMed]
- Meanwatthana, J.; Majam, T. Interleukin-6 antagonists: Lessons from cytokine release syndrome to the therapeutic application in severe COVID-19 infection. J. Pharm. Pract. 2021. [Google Scholar] [CrossRef]
- Emidio, N.B.; Hoffmann, W.; Brierley, S.; Muttenthaler, M. Trefoil factor family: Unresolved questions and clinical perspectives. Trends Biochem. Sci. 2019, 44, 387–390. [Google Scholar] [CrossRef]
- Chen, R.-M.; Chiou, Y.-S.; Chong, Q.-Y.; Poh, H.-M.; Tan, T.-Z.; Zhang, M.-Y.; Ma, L.; Zhu, T.; Pandey, V.; Kumar, A.P.; et al. Pharmacological inhibition of TFF3 enhances sensitivity of CMS4 colorectal carcinoma to 5-fluorouracil through inhibition of p44/42 MAPK. Int. J. Mol. Sci. 2019, 20, 6215. [Google Scholar] [CrossRef]
- Bolle, T.; Meyer, F.; Walcher, F.; Lohmann, C.; Jockenhövel, S.; Gries, T.; Hoffmann, W. Materials/biomaterials in clinical practice—A short review and current trends. Zentralbl. Chir. 2017, 142, 216–225. [Google Scholar] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoffmann, W. Trefoil Factor Family (TFF) Peptides. Encyclopedia 2021, 1, 974-987. https://doi.org/10.3390/encyclopedia1030074
Hoffmann W. Trefoil Factor Family (TFF) Peptides. Encyclopedia. 2021; 1(3):974-987. https://doi.org/10.3390/encyclopedia1030074
Chicago/Turabian StyleHoffmann, Werner. 2021. "Trefoil Factor Family (TFF) Peptides" Encyclopedia 1, no. 3: 974-987. https://doi.org/10.3390/encyclopedia1030074
APA StyleHoffmann, W. (2021). Trefoil Factor Family (TFF) Peptides. Encyclopedia, 1(3), 974-987. https://doi.org/10.3390/encyclopedia1030074





