Vaccine Breakthrough COVID-19 Outbreak in Section of a Hospital with 88% Attack Rate: Lessons to Be Learned
Abstract
1. Introduction
Motivation behind This Study
2. Materials and Methods
2.1. Setting
2.2. Design
2.3. Details of the Event
3. Results
3.1. Index Case
3.2. Identified Risky Exposures
3.3. Epi-Curve
3.4. Pre-Existing Medical Conditions
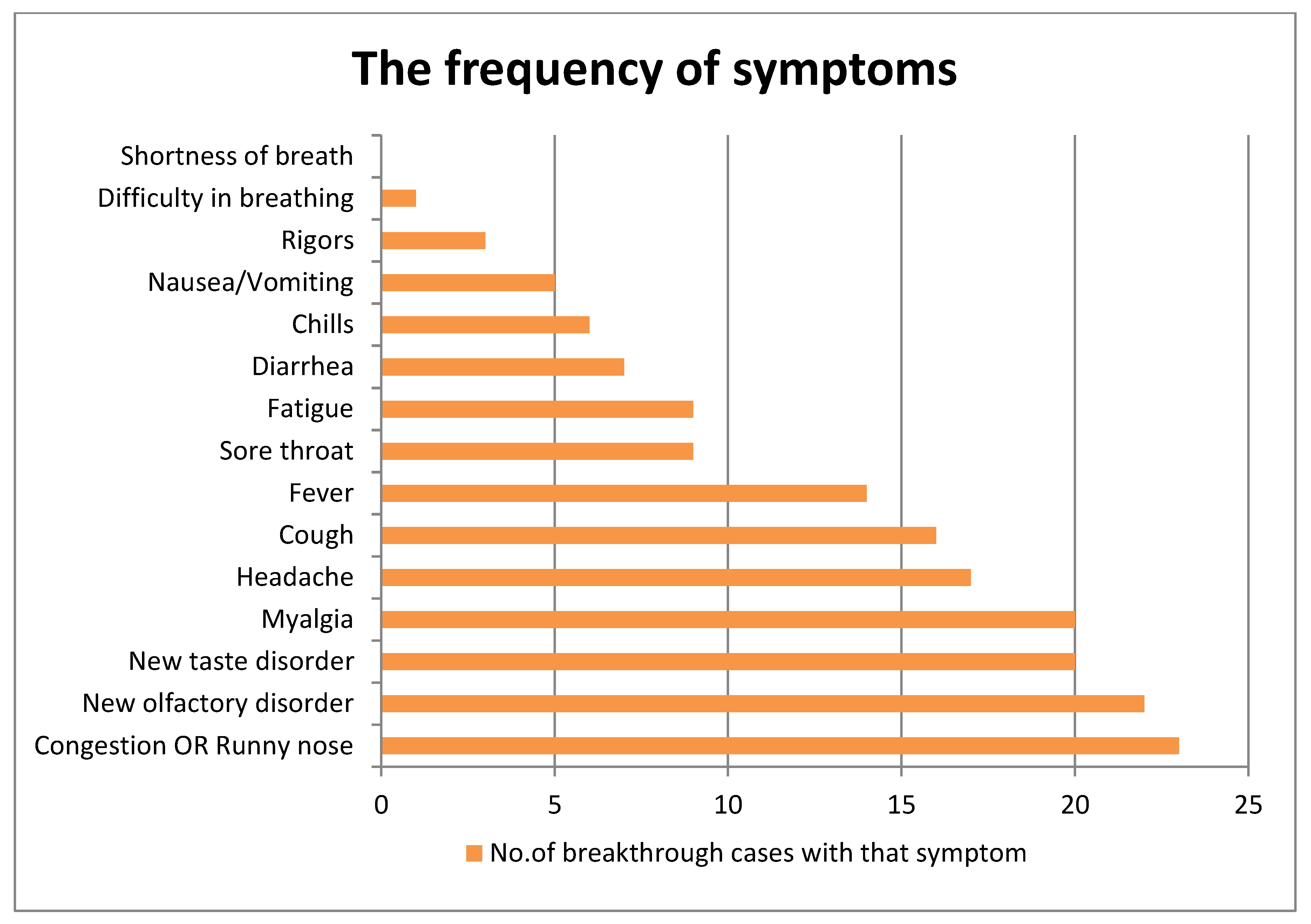
3.5. Description of Symptoms
3.6. Maintenance of the Cold Chain
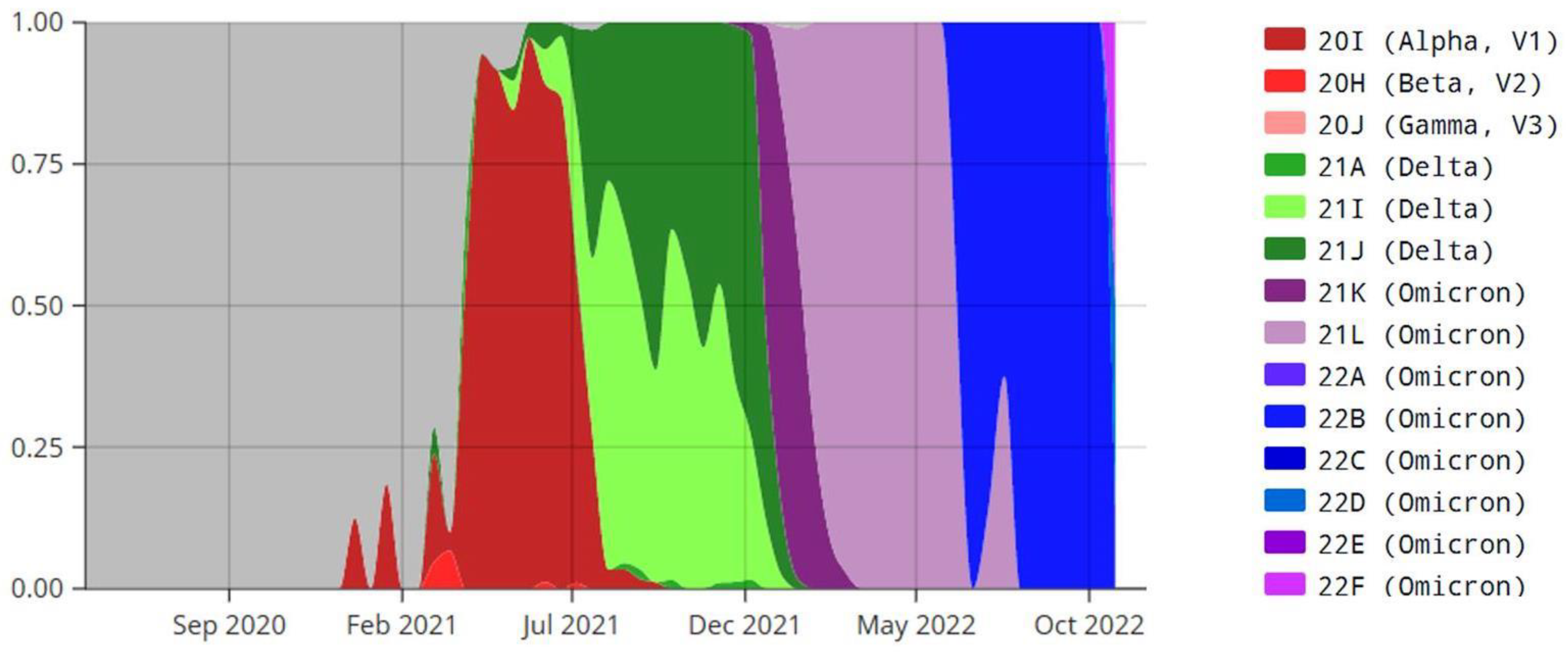
3.7. Identification of the Variant of the Virus
3.8. Further Information about SGs
4. Discussion
4.1. Our Preventive Work among the Hospital Workers
4.2. Comparison of Our Findings with the Recent Literature Related to Our Study
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zheng, C.; Shao, W.; Chen, X.; Zhang, B.; Wang, G.; Zhang, W. Real-world effectiveness of COVID-19 vaccines: A literature review and meta-analysis. Int. J. Infect. Dis. 2022, 114, 252–260. [Google Scholar] [CrossRef]
- Feikin, D.R.; Higdon, M.M.; Abu-Raddad, L.J.; Andrews, N.; Araos, R.; Goldberg, Y.; Groome, M.J.; Huppert, A.; O'Brien, K.L.; Smith, P.G.; et al. Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: Results of a systematic review and meta-regression. Lancet 2022, 399, 924–944. [Google Scholar] [CrossRef] [PubMed]
- Marra, A.R.; Kobayashi, T.; Suzuki, H.; Alsuhaibani, M.; Schweizer, M.L.; Diekema, D.J.; Tofaneto, B.M.; Bariani, L.M.; Auler, M.d.A.; Salinas, J.L.; et al. The long-term effectiveness of coronavirus disease 2019 (COVID-19) vaccines: A systematic literature review and meta-analysis. Antimicrob. Steward. Health Epidemiol. 2022, 2, e22. [Google Scholar] [CrossRef] [PubMed]
- Shao, W.; Chen, X.; Zheng, C.; Liu, H.; Wang, G.; Zhang, B.; Li, Z.; Zhang, W. Effectiveness of COVID-19 vaccines against SARS-CoV-2 variants of concern in real-world: A literature review and meta-analysis. Emerg. Microbes Infect. 2022, 11, 2383–2392. [Google Scholar] [CrossRef] [PubMed]
- Niyas, V.K.; Arjun, R. Breakthrough COVID-19 infections among health care workers after two doses of ChAdOx1 nCoV-19 vaccine. QJM Int. J. Med. 2021, 114, 757–758. [Google Scholar] [CrossRef]
- The Secretary-Ministry of Health of Sri Lanka. Screening and Management of Healthcare Workers Following Exposure to a Confirmed/Suspected Case of COVID-19 (Updated-20th October 2020); Ministry of Health-Sri Lanka: Colombo, Sri Lanka, 2020; No: EPID/400/2019nCov dated 02/11/2020. [Google Scholar]
- Centers for Disease Control of the USA. Public Health Investigations of COVID-19 Vaccine Breakthrough Cases—Case Investigation Protocol. 2021. Available online: https://www.cdc.gov/vaccines/covid-19/downloads/COVID-vaccine-breakthrough-case-investigations-protocol.pdf (accessed on 25 November 2022).
- Giesecke, J. Primary and index cases. Lancet 2014, 384, 2024. Available online: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(14)62331-X/fulltext (accessed on 26 January 2023). [CrossRef]
- Gao, W.; Lv, J.; Pang, Y.; Li, L.-M. Role of asymptomatic and pre-symptomatic infections in COVID-19 pandemic. BMJ 2021, 375, n2342. [Google Scholar] [CrossRef]
- Ehelepola, N.D.B.; Wijewardana, B.A.S. An episode of transmission of COVID-19 from a vaccinated healthcare worker to co-workers. Infect. Dis. 2021, 54, 297–302. [Google Scholar] [CrossRef]
- Jeewandara, C.; Jayathilaka, D.; Ranasinghe, D.; Hsu, N.S.; Ariyaratne, D.; Jayadas, T.T.; Arachchige, D.M.P.; Lindsey, B.B.; Gomes, L.; Parker, M.D.; et al. Genomic and Epidemiological Analysis of SARS-CoV-2 Viruses in Sri Lanka. Front. Microbiol. 2021, 12, 722838. [Google Scholar] [CrossRef]
- Allergy, Immunology and Cell Biology Unit of the Department of Immunology and Molecular Medicine of the University of Sri Jayewardenepura, Sri Lanka. USJ Researchers Found Four Mutant Covid Delta Variants with Two New Sri Lankan Mutations (A701S, R24C). University of Sri Jayewardenepura. 30 August 2021. Available online: https://www.sjp.ac.lk/news/usj-researchers-found-four-mutant-covid-delta-variants-with-two-new-sri-lankan-mutations-a701s-r24c/ (accessed on 26 January 2023).
- Cevik, M.; Tate, M.; Lloyd, O.; Maraolo, A.E.; Schafers, J.; Ho, A. SARS-CoV-2, SARS-CoV, and MERS-CoV viral load dynamics, duration of viral shedding, and infectiousness: A systematic review and meta-analysis. Lancet Microbe 2021, 2, e13–e22. [Google Scholar] [CrossRef]
- Alishaq, M.; Nafady-Hego, H.; Jeremijenko, A.; Al Ajmi, J.A.; Elgendy, M.; Vinoy, S.; Fareh, S.B.; Plaatjies, J.V.; Nooh, M.; Alanzi, N.; et al. Risk factors for breakthrough SARS-CoV-2 infection in vaccinated healthcare workers. PLoS ONE 2021, 16, e0258820. [Google Scholar] [CrossRef] [PubMed]
- Marcus, J.E.; Frankel, D.N.; Pawlak, M.T.; Casey, T.M.; Cybulski, R.J.; Enriquez, E.; Okulicz, J.F.; Yun, H.C. Risk Factors Associated With COVID-19 Transmission Among US Air Force Trainees in a Congregate Setting. JAMA Netw. Open 2021, 4, e210202. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.-Y.; Jian, S.-W.; Liu, D.-P.; Ng, T.-C.; Huang, W.-T.; Lin, H.-H.; The Taiwan COVID-19 Outbreak Investigation Team. Contact Tracing Assessment of COVID-19 Transmission Dynamics in Taiwan and Risk at Different Exposure Periods Before and After Symptom Onset. JAMA Intern. Med. 2020, 180, 1156–1163. [Google Scholar] [CrossRef]
- Rosser, J.I.; Tayyar, R.; Giardina, R.; Kolonoski, P.; Kenski, D.; Shen, P.; Steinmetz, L.M.; Hung, L.-Y.; Xiao, W.; Bains, K.; et al. Case-control study evaluating risk factors for SARS-CoV-2 outbreak amongst healthcare personnel at a tertiary care center. Am. J. Infect. Control. 2021, 49, 1457–1463. [Google Scholar] [CrossRef] [PubMed]
- Hatmi, Z.N. A Systematic Review of Systematic Reviews on the COVID-19 Pandemic. SN Compr. Clin. Med. 2021, 3, 419–436. [Google Scholar] [CrossRef]
- Byambasuren, O.; Stehlik, P.; Clark, J.; Alcorn, K.; Glasziou, P. Impact of COVID-19 vaccination on long COVID: A systematic review and meta-analysis. medRxiv 2022. [Google Scholar] [CrossRef]
- Lee, C.J.; Woo, W.; Kim, A.Y.; Yon, D.K.; Lee, S.W.; Koyanagi, A.; Kim, M.S.; Tizaoui, K.; Dragioti, E.; Radua, J.; et al. Clinical manifestations of COVID-19 breakthrough infections: A systematic review and meta-analysis. J. Med. Virol. 2022, 94, 4234–4245. [Google Scholar] [CrossRef]
- De Maria, L.; Sponselli, S.; Caputi, A.; Stefanizzi, P.; Pipoli, A.; Giannelli, G.; Delvecchio, G.; Tafuri, S.; Inchingolo, F.; Migliore, G.; et al. SARS-CoV-2 Breakthrough Infections in Health Care Workers: An Italian Retrospective Cohort Study on Characteristics, Clinical Course and Outcomes. J. Clin. Med. 2023, 12, 628. [Google Scholar] [CrossRef]
- Porru, S.; Monaco, M.G.L.; Spiteri, G.; Carta, A.; Pezzani, M.D.; Lippi, G.; Gibellini, D.; Tacconelli, E.; Vecchia, I.D.; Sala, E.; et al. SARS-CoV-2 Breakthrough Infections: Incidence and Risk Factors in a Large European Multicentric Cohort of Health Workers. Vaccines 2022, 10, 1193. [Google Scholar] [CrossRef]
- Maltezou, H.C.; Gamaletsou, M.N.; Giannouchos, T.V.; Koukou, D.-M.; Karapanou, A.; Sourri, F.; Syrimi, N.; Lemonakis, N.; Peskelidou, E.; Papanastasiou, K.; et al. Timing of last COVID-19 vaccine dose and SARS-CoV-2 breakthrough infections in fully (boosted) vaccinated healthcare personnel. J. Hosp. Infect. 2022, 132, 46–51. [Google Scholar] [CrossRef]
- Santi, T.; Kamarga, L.; De Samakto, B.; Jo, J. SARS-CoV-2 Breakthrough Infection after mRNA-1273 Booster among CoronaVac-Vaccinated Healthcare Workers. Infect. Chemother. 2022, 54, 774–780. [Google Scholar] [CrossRef]
- Offit, P.A. Bivalent COVID-19 Vaccines–A Cautionary Tale. N. Engl. J. Med. 2023. [Google Scholar] [CrossRef]
- The Open SAFELY Collaborative; Green, A.; Curtis, H.; Hulme, W.; Williamson, E.; McDonald, H.; Bhaskaran, K.; Rentsch, C.; Schultze, A.; MacKenna, B.; et al. Describing the population experiencing COVID-19 vaccine breakthrough following second vaccination in England: A cohort study from OpenSAFELY. BMC Med. 2022, 20, 1–14. [Google Scholar] [CrossRef]
- Stouten, V.; Hubin, P.; Haarhuis, F.; van Loenhout, J.A.F.; Billuart, M.; Brondeel, R.; Braeye, T.; Van Oyen, H.; Wyndham-Thomas, C.; Catteau, L. Incidence and Risk Factors of COVID-19 Vaccine Breakthrough Infections: A Prospective Cohort Study in Belgium. Viruses 2022, 14, 802. [Google Scholar] [CrossRef]
- Jeewandara, C.; Aberathna, I.S.; Gomes, L.; Pushpakumara, P.D.; Danasekara, S.; Guruge, D.; Ranasinghe, T.; Gunasekera, B.; Kamaladasa, A.; Kuruppu, H.; et al. Kinetics of immune responses to the AZD1222/Covishield vaccine with varying dose intervals in Sri Lankan individuals. Immun. Inflamm. Dis. 2022, 10, e592. [Google Scholar] [CrossRef] [PubMed]
- Ibekwe, T.S.; Fasunla, A.J.; Orimadegun, A.E. Systematic Review and Meta-analysis of Smell and Taste Disorders in COVID-19. OTO Open 2020, 4, 2473974X20957975. [Google Scholar] [CrossRef]
- Purja, S.; Shin, H.; Lee, J.-Y.; Kim, E. Is loss of smell an early predictor of COVID-19 severity: A systematic review and meta-analysis. Arch. Pharmacal Res. 2021, 44, 725–740. [Google Scholar] [CrossRef] [PubMed]
- Emary, K.R.W.; Golubchik, T.; Aley, P.K.; Ariani, C.V.; Angus, B.; Bibi, S.; Blane, B.; Bonsall, D.; Cicconi, P.; Charlton, S.; et al. Efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine against SARS-CoV-2 variant of concern 202012/01 (B.1.1.7): An exploratory analysis of a randomised controlled trial. Lancet 2021, 397, 1351–1362. [Google Scholar] [CrossRef]
- Lim, W.-Y.; Tan, G.; Htun, H.; Phua, H.; Kyaw, W.; Guo, H.; Cui, L.; Mak, T.; Poh, B.; Wong, J.; et al. First nosocomial cluster of COVID-19 due to the Delta variant in a major acute care hospital in Singapore: Investigations and outbreak response. J. Hosp. Infect. 2022, 122, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Vignier, N.; Bérot, V.; Bonnave, N.; Peugny, S.; Ballet, M.; Jacoud, E.; Michaud, C.; Gaillet, M.; Djossou, F.; Blanchet, D.; et al. Breakthrough Infections of SARS-CoV-2 Gamma Variant in Fully Vaccinated Gold Miners, French Guiana, 2021. Emerg. Infect. Dis. 2021, 27, 2673–2676. [Google Scholar] [CrossRef]
- Dunbar, R.I.M. Breaking Bread: The Functions of Social Eating. Adapt. Hum. Behav. Physiol. 2017, 3, 198–211. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Contact Tracing in the Context of COVID-19—Interim Guidance. 1 February 2021. Available online: https://apps.who.int/iris/bitstream/handle/10665/339128/WHO-2019-nCoV-Contact_Tracing-2021.1-eng.pdf?sequence=24&isAllowed=y (accessed on 28 January 2023).


| The Number of Symptoms | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 |
| The Count of SGs with that number of symptoms | 2 | 3 | 4 | 2 | 5 | 4 | 3 | 3 | 2 | 2 | 3 | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ehelepola, N.D.B.; Wijewardana, B.A.S. Vaccine Breakthrough COVID-19 Outbreak in Section of a Hospital with 88% Attack Rate: Lessons to Be Learned. COVID 2023, 3, 226-237. https://doi.org/10.3390/covid3020017
Ehelepola NDB, Wijewardana BAS. Vaccine Breakthrough COVID-19 Outbreak in Section of a Hospital with 88% Attack Rate: Lessons to Be Learned. COVID. 2023; 3(2):226-237. https://doi.org/10.3390/covid3020017
Chicago/Turabian StyleEhelepola, N. D. B., and B. A. S. Wijewardana. 2023. "Vaccine Breakthrough COVID-19 Outbreak in Section of a Hospital with 88% Attack Rate: Lessons to Be Learned" COVID 3, no. 2: 226-237. https://doi.org/10.3390/covid3020017
APA StyleEhelepola, N. D. B., & Wijewardana, B. A. S. (2023). Vaccine Breakthrough COVID-19 Outbreak in Section of a Hospital with 88% Attack Rate: Lessons to Be Learned. COVID, 3(2), 226-237. https://doi.org/10.3390/covid3020017







