Deactivation of SARS-CoV-2 via Shielding of Spike Glycoprotein Using Carbon Quantum Dots: Bioinformatic Perspective
Abstract
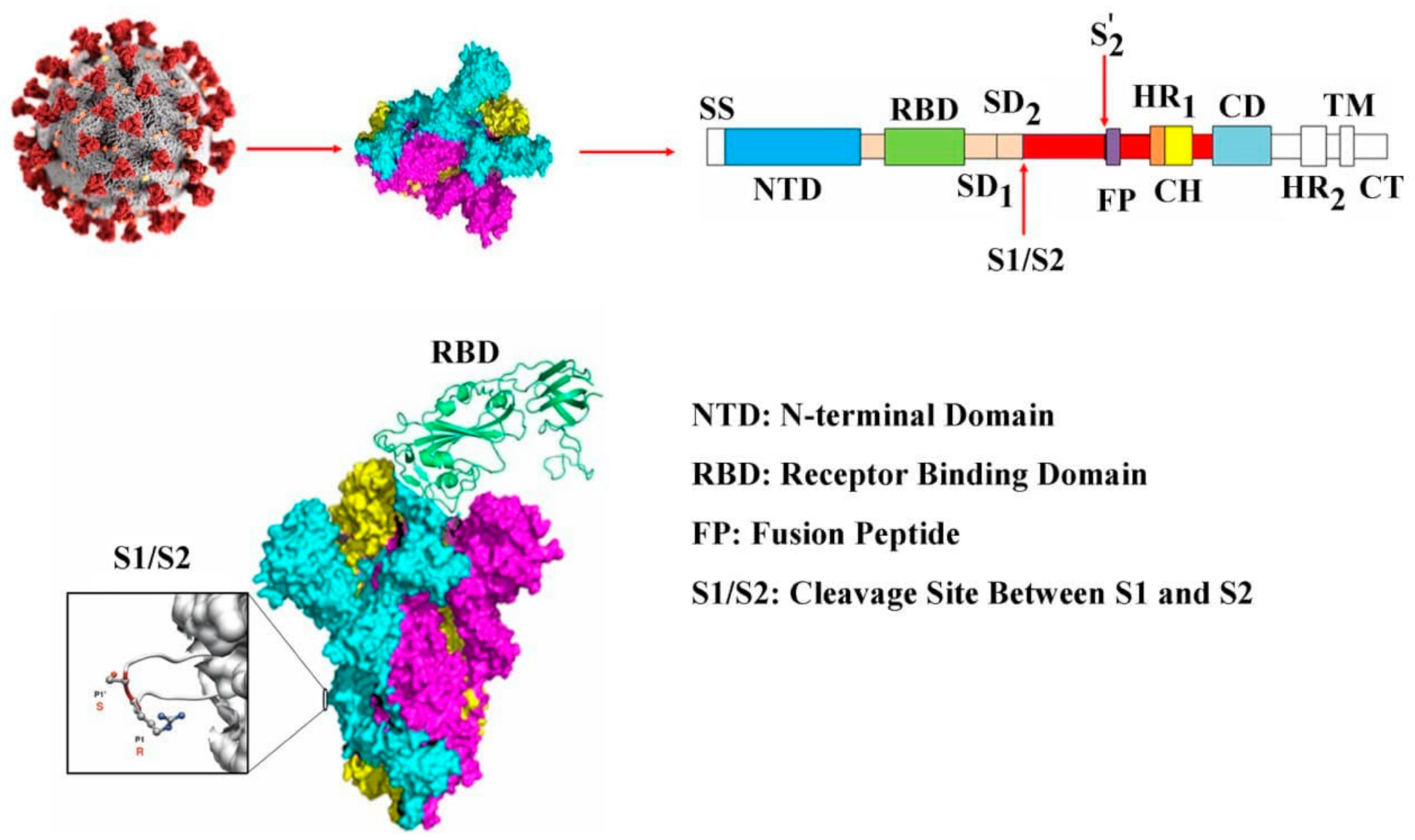
:1. Introduction
2. Methods
2.1. Coordinate Structure Retrieval and Preparation
2.2. Molecular Docking Simulations
2.3. Molecular Dynamic (MD) Simulation
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vankadari, N.; Jacqueline, A.; Wilce, J.A. Emerging WuHan (COVID-19) coronavirus: Glycan shield and structure prediction of spike glycoprotein and its interaction with human CD26. Emerg. Microbes Infect. 2020, 9, 601–604. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Allen, J.D.; Wrapp, D.; McLellan, J.S.; Crispin, M. Site-specific glycan analysis of the SARS-CoV-2 spike. Science 2020, 369, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.K.; Cao, Y.; Frank, M.; Woo, H.; Park, S.-J.; Yeom, M.S.; Croll, T.I.; Seok, C.; Im, W. Structure, dynamics, receptor binding, and antibody binding of the fully glycosylated full-length SARS-CoV-2 spike protein in a viral membrane. J. Chem. Theory Comput. 2021, 17, 2479–2487. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Petitjean, S.J.L.; Koehler, M.; Zhang, Q.; Dumitru, A.C.; Chen, W.; Derclaye, S.; Vincent, S.P.; Soumillion, P.; Alsteens, D. Molecular interaction and inhibition of SARS-CoV-2 binding to the ACE2 receptor. Nat. Commun. 2020, 11, 1–10. [Google Scholar]
- Walls, A.C.; Park, Y.J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell 2020, 181, 281–292.e6. [Google Scholar] [CrossRef]
- Schoeman, D.; Fielding, B.C. Coronavirus envelope protein: Current knowledge. Virol. J. 2019, 6, 69. [Google Scholar] [CrossRef] [Green Version]
- Xia, S.; Liu, M.; Wa, C.; Xu, W.; Lan, Q.; Feng, S.; Feifei, Q.; Bao, L.; Du, L.; Liu, S.; et al. Inhibition of SARS-CoV-2 (previously 2019-nCoV) infection by a highly potent pan-coronavirus fusion inhibitor targeting its spike protein that harbors a high capacity to mediate membrane fusion. Cell Res. 2020, 30, 343–355. [Google Scholar] [CrossRef] [Green Version]
- Acquavia, M.A.; Foti, L.; Pascale, R.; Nicolo., A.; Brancaleone, V.; Cataldi, T.R.I.; Martelli, G.; Scrano, L.; Bianco, G. Detection and quantification of Covid-19 antiviral drugs in biological fluids and tissues. Talanta 2021, 224, 121862. [Google Scholar] [CrossRef]
- Kheiri, S.; Liu, X.; Thompson, M. Nanoparticles at biointerfaces: Antibacterial activity and nanotoxicology. Colloids Surf. B Biointerfaces 2019, 184, 110550. [Google Scholar] [CrossRef]
- Sharma, S.; Saini, S.; Khangembam, M.; Singh, V. Nanomaterials-based biosensors for COVID-19 detection-A Review. IEEE Sens. J. 2021, 21, 5598–5611. [Google Scholar] [CrossRef]
- Farzin, L.; Sadjadi, S.; Sheini, A.; Mohagheghpour, E. A nanoscale genosensor for early detection of COVID-19 by voltammetric determination of RNA-dependent RNA polymerase (RdRP) sequence of SARS-CoV-2 virus. Microchim. Acta 2021, 188, 121. [Google Scholar] [CrossRef]
- Singh, S.; Shauloff, N.; Sharma, C.P.; Shimoni, R.; Arnusch, C.J.; Jelinek, R. Carbon dot-polymernanoporousmembraneforrecyclablesunlight-sterilizedfacemasks. J. Colloid Interface Sci. 2021, 592, 342–348. [Google Scholar] [CrossRef]
- Loczechin, A.; Seron, K.; Barras, A.; Giovanelli, E.; Belouzard, S.; Chen, Y.T.; Metzler-Nolte, N.; Boukherroub, R.; Dubuisson, J.; Szunerits, S. Functional carbon quantum dots as medical counter measures to human coronavirus. ACS Appl. Mater. Interfaces 2019, 11, 42964–42974. [Google Scholar] [CrossRef]
- Saraf, M.; Prateek, M.T.Y.; Tan, Y.N.; Gupta, R.K. Insights and perspectives regarding nanostructure dfluorescent materials toward tackling COVID-19 and future pandemics. ACS Appl. Nano Mater. 2021, 4, 911–948. [Google Scholar] [CrossRef]
- Mallik, A.K.; Mukherjee, S.; Panchagnula, M.V. An experimental study of respiratory aerosol transport in phantom lung bronchioles. Phys. Fluids 2020, 32, 111903. [Google Scholar] [CrossRef]
- Jena, A.B.; Kanungo, N.; Nayak, V.; Chainy, G.B.N.; Dandapat, J. Catechin and curcumin interact with S protein of SARS-CoV2 and ACE2 of human cell membrane: Insights from computational studies. Sci. Rep. 2021, 11, 2043. [Google Scholar] [CrossRef]
- Sheybani, Z.; Dokoohaki, H.M.; Negahdaripour, M.; Dehdashti, M.; Zolghadr, H.; Moghadami, M.; Masoompour, S.M.; Zolghadr, A.R. The Role of Folic Acid in the Management of Respiratory Disease Caused by COVID-19. ChemRxiv 2020. [Google Scholar] [CrossRef]
- Liu, H.; Li, Z.; Sun, Y.; Geng, X.; Hu, Y.; Meng, H.; Ge, J.; Qu, L. Synthesis of luminescent carbon dots with ultrahigh quantum yield and inherent folate receptor-positive cancer cell targetability. Sci. Rep. 2018, 8, 1086. [Google Scholar] [CrossRef] [Green Version]
- Kotta, S.; Aldawsari, H.M.; Badr-Eldin, S.M.; Alhakamy, N.A.; Md, S.; Nair, A.B.; Deb, P.K. Exploring the potential of carbon dots to combat COVID-19. Front. Mol. Biosci. 2020, 7. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09; Gaussian, Inc.: Wallingford, CT, USA, 2009; Available online: https://www.cwu.edu/chemistry/sites/cts.cwu.edu.chemistry/files/documents/Gaussian_09_ReferenceManual.pdf (accessed on 15 December 2020).
- Letko, M.; Marzi, A.; Munster, V. Functional assessment of cell entry and receptorusage for lineage Bβ corona viruses, including 2019-nCoV. Nat. Microbiol. 2020, 5, 562–569. [Google Scholar] [CrossRef] [Green Version]
- Agrawal, U.; Raju, R.; Udwadi, Z.F. Favipiravir: Anew and emerging antiviral optionin COVID-19. Med. J. 2020, 76, 370–376. [Google Scholar]
- Zhao, X.; Zhang, J.; Shi, L.; Xian, M.; Dong, C.; Shuang, S. Folic acid-conjugated carbon dots as green fluorescent probes based on cellular targeting imaging for recognizing cancer cells. RSC Adv. 2017, 7, 42159–42167. [Google Scholar] [CrossRef] [Green Version]




| Parameter | Value | Description |
|---|---|---|
| Grid Dimension | 0.6 | With 100 solutions |
| Receptor range | 60 | With step size 7.5 |
| Ligand (CQDS) range | 60 | With step size 7.5 |
| Twist range | 90 | With step size 5.5 |
| Distance range | 40 | With box size 10 |
| Scan step | 0.8 | |
| Final Scan | 25 |
| Carbon Species | kJ·mole −1 | 319–541 a | 788–806 b |
|---|---|---|---|
| OH-CQDs | −699.3 | 80 | 14 |
| Amide-CQDs | −689.7 | 64 | 0 |
| COOH-CQDs | −652.1 | 67 | 3 |
| NH2-OH-CQDs | −592.2 | 85 | 14 |
| THG1 | −369.8 | 0 | 0 |
| THG2 | −422.9 | 0 | 0 |
| Favipirvir | −487.2 | 44 | 0 |
| Folic Acid | −425.3 | 10 | 46 |
| EtOH | −148.5 | 84 | 5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramezani, Z.; Dayer, M.R.; Noorizadeh, S.; Thompson, M. Deactivation of SARS-CoV-2 via Shielding of Spike Glycoprotein Using Carbon Quantum Dots: Bioinformatic Perspective. COVID 2021, 1, 120-129. https://doi.org/10.3390/covid1010011
Ramezani Z, Dayer MR, Noorizadeh S, Thompson M. Deactivation of SARS-CoV-2 via Shielding of Spike Glycoprotein Using Carbon Quantum Dots: Bioinformatic Perspective. COVID. 2021; 1(1):120-129. https://doi.org/10.3390/covid1010011
Chicago/Turabian StyleRamezani, Zahra, Mohammad Reza Dayer, Siamak Noorizadeh, and Michael Thompson. 2021. "Deactivation of SARS-CoV-2 via Shielding of Spike Glycoprotein Using Carbon Quantum Dots: Bioinformatic Perspective" COVID 1, no. 1: 120-129. https://doi.org/10.3390/covid1010011
APA StyleRamezani, Z., Dayer, M. R., Noorizadeh, S., & Thompson, M. (2021). Deactivation of SARS-CoV-2 via Shielding of Spike Glycoprotein Using Carbon Quantum Dots: Bioinformatic Perspective. COVID, 1(1), 120-129. https://doi.org/10.3390/covid1010011








