Effective Energy Harvesting in Polymer Solar Cells Using NiS/Co as Nanocomposite Doping
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Nanocomposites
2.3. Device Fabrication
3. Results and Discussion
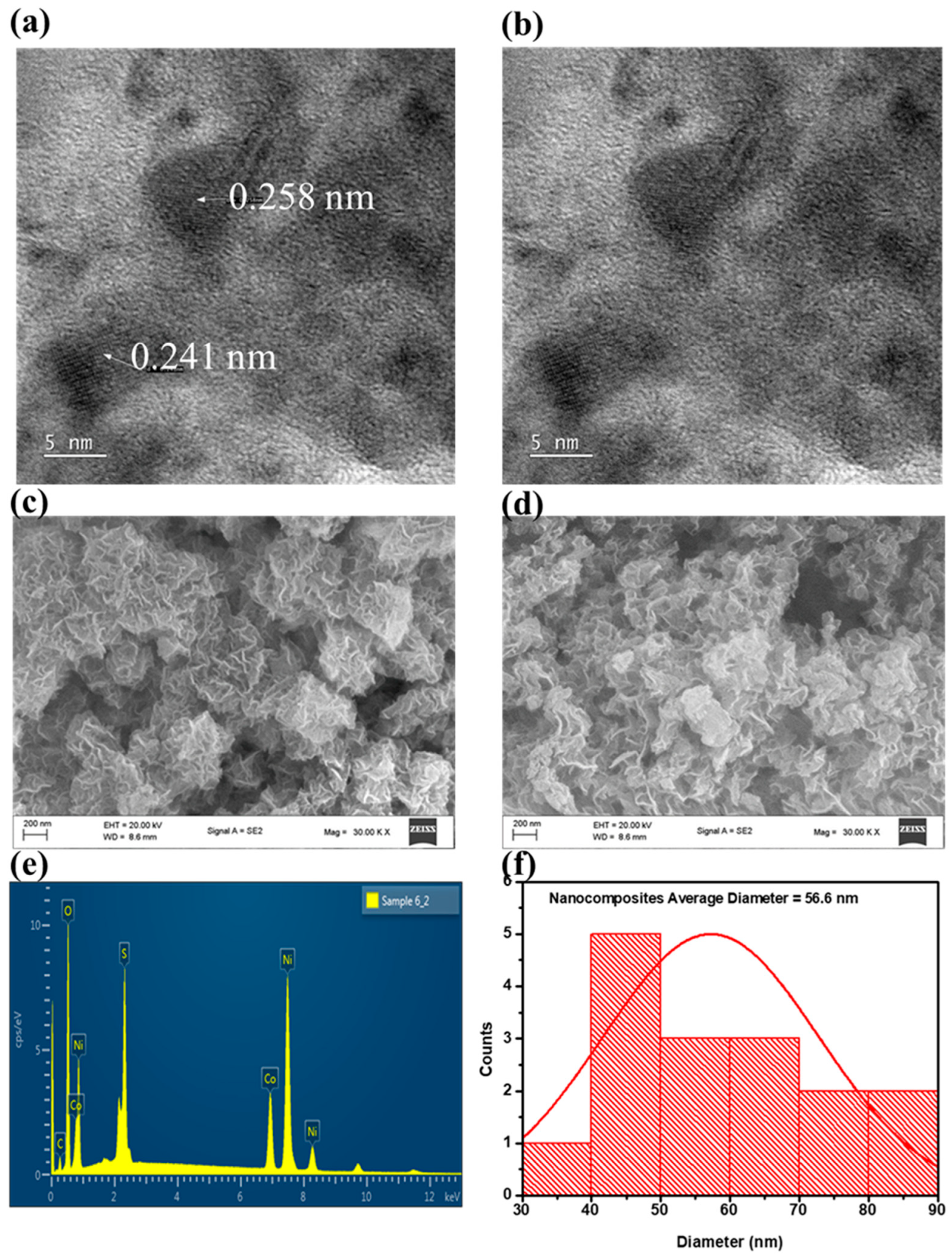
3.1. Morphological and Structural Analysis via Electron Microscopy of Nanocomposites
3.2. Fourier Transform Infrared Spectroscopy of Nanocomposites
3.3. Optical Properties of Nanocomposite-Doped Active Layer
3.4. Photovoltaic Performance of -Doped Solar Cell Devices
3.5. Charge-Transport Characteristics of Nanocomposites
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, C.; Zhang, S.; Ren, J.; Gao, M.; Bi, P.; Ye, L.; Hou, J. Molecular design of a non-fullerene acceptor enables a P3HT-based organic solar cell with 9.46% efficiency. Energy Environ. Sci. 2020, 13, 2864–2869. [Google Scholar] [CrossRef]
- Inganäs, O. Organic photovoltaics over three decades. Adv. Mater. 2018, 30, 1800388. [Google Scholar] [CrossRef]
- Wadsworth, A.; Moser, M.; Marks, A.; Little, M.S.; Gasparini, N.; Brabec, C.J.; Baran, D.; McCulloch, I. Critical review of the molecular design progress in non-fullerene electron acceptors towards commercially viable organic solar cells. Chem. Soc. Rev. 2019, 48, 1596–1625. [Google Scholar] [CrossRef]
- Hu, Z.; Wang, J.; Ma, X.; Gao, J.; Xu, C.; Yang, K.; Wang, Z.; Zhang, J.; Zhang, F. A critical review on semitransparent organic solar cells. Nano Energy 2020, 78, 105376. [Google Scholar] [CrossRef]
- Cui, Y.; Yao, H.; Zhang, J.; Xian, K.; Zhang, T.; Hong, L.; Wang, Y.; Xu, Y.; Ma, K.; An, C.; et al. Single-junction organic photovoltaic cells with approaching 18% efficiency. Adv. Mater. 2020, 32, 1908205. [Google Scholar] [CrossRef]
- Ali, I.; Islam, M.R.; Yin, J.; Eichhorn, S.J.; Chen, J.; Karim, N.; Afroi, S. Advances in smart photovoltaic textiles. ACS Nano 2024, 18, 3871–3915. [Google Scholar] [CrossRef]
- Du, J.; Tong, B.; Yuan, S.; Dai, N.; Liu, R.; Zhang, D.; Cheng, H.M.; Ren, W. Advances in flexible optoelectronics based on chemical vapor deposition-grown graphene. Adv. Funct. Mater. 2022, 32, 2203115. [Google Scholar] [CrossRef]
- Hamed, M.S.; Ike, J.N.; Wang, Y.; Zhou, K.; Ma, W.; Mola, G.T. Reducing energy loss in polymer solar cell through optimization of novel metal nanocomposite. Energy Fuels 2023, 37, 6129–6137. [Google Scholar] [CrossRef]
- Li, C.; Song, J.; Lai, H.; Zhang, H.; Zhou, R.; Xu, J.; Huang, H.; Liu, L.; Gao, J.; Li, Y.; et al. Non-fullerene acceptors with high crystallinity and photoluminescence quantum yield enable > 20% efficiency organic solar cells. Nat. Mater. 2025, 24, 433–443. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, L.; Guo, C.; Xiao, J.; Liu, C.; Chen, C.; Xia, W.; Gan, Z.; Cheng, J.; Zhou, J.; et al. π-extended nonfullerene acceptor for compressed molecular packing in organic solar cells to achieve over 20% efficiency. J. Am. Chem. Soc. 2024, 146, 12011–12019. [Google Scholar] [CrossRef]
- Jiang, Y.; Sun, S.; Xu, R.; Liu, F.; Miao, X.; Ran, G.; Liu, K.; Yi, Y.; Zhang, W.; Zhu, X. Non-fullerene acceptor with asymmetric structure and phenyl-substituted alkyl side chain for 20.2% efficiency organic solar cells. Nat. Energy 2024, 9, 975–986. [Google Scholar] [CrossRef]
- Cheng, P.; Zhan, X. Versatile third components for efficient and stable organic solar cells. Mater. Horiz. 2015, 2, 462–485. [Google Scholar] [CrossRef]
- Waketola, A.G.; Hone, F.G.; Geldasa, F.T.; Genene, Z.; Mammo, W.; Tegegne, N.A. Enhancing the performance of wide-bandgap polymer-based organic solar cells through silver nanorod integration. ACS Omega 2024, 9, 8082–8091. [Google Scholar] [CrossRef]
- Lai, Y.-Y.; Cheng, Y.-J.; Hsu, C.-S. Applications of functional fullerene materials in polymer solar cells. Energy Environ. Sci. 2014, 7, 1866–1883. [Google Scholar] [CrossRef]
- Laquai, F.; Andrienko, D.; Mauer, R.; Blom, P.W. Charge carrier transport and photogeneration in P3HT: PCBM photovoltaic blends. Macromol. Rapid Commun. 2015, 36, 1001–1025. [Google Scholar] [CrossRef]
- Alam, S.; Anand, A.; Islam, M.M.; Meitzner, R.; Djoumessi, A.S.; Slowik, J.; Teklu, Z.; Fischer, P.; Kästner, C.; Khan, J.I.; et al. P3HT: PCBM polymer solar cells from a didactic perspective. J. Photonics Energy 2022, 12, 035501. [Google Scholar] [CrossRef]
- Liu, F.; Chen, D.; Wang, C.; Luo, K.; Gu, W.; Briseno, A.L.; Hsu, J.W.; Russell, T.P. Molecular weight dependence of the morphology in P3HT: PCBM solar cells. ACS Appl. Mater. Interfaces 2014, 6, 19876–19887. [Google Scholar] [CrossRef]
- Liu, C.; Wang, K.; Gong, X.; Heeger, A.J. Low bandgap semiconducting polymers for polymeric photovoltaics. Chem. Soc. Rev. 2016, 45, 4825–4846. [Google Scholar] [CrossRef]
- Holliday, S.; Ashraf, R.S.; Wadsworth, A.; Baran, D.; Yousaf, S.A.; Nielsen, C.B.; Tan, C.H.; Dimitrov, S.D.; Shang, Z.; Gasparini, N.; et al. High-efficiency and air-stable P3HT-based polymer solar cells with a new non-fullerene acceptor. Nat. Commun. 2016, 7, 11585. [Google Scholar] [CrossRef]
- Alkhalayfeh, M.A.; Aziz, A.A.; Pakhuruddin, M.Z. An overview of enhanced polymer solar cells with embedded plasmonic nanoparticles. Renew. Sustain. Energy Rev. 2021, 141, 110726. [Google Scholar] [CrossRef]
- Li, Y.-F.; Kou, Z.L.; Feng, J.; Sun, H.B. Plasmon-enhanced organic and perovskite solar cells with metal nanoparticles. Nanophotonics 2020, 9, 3111–3133. [Google Scholar] [CrossRef]
- Fahimi-Kashani, N.; Orouji, A.; Ghamsari, M.; Sahoo, S.K.; Hormozi-Nezhad, M.R. Plasmonic noble metal (Ag and Au) nanoparticles: From basics to colorimetric sensing applications. In Gold and Silver Nanoparticles; Elsevier: Amsterdam, The Netherlands, 2023; pp. 1–58. [Google Scholar] [CrossRef]
- Craciun, A.M.; Focsan, M.; Magyari, K.; Vulpoi, A.; Pap, Z. Surface plasmon resonance or biocompatibility—Key properties for determining the applicability of noble metal nanoparticles. Materials 2017, 10, 836. [Google Scholar] [CrossRef]
- Ng, A.; Yiu, W.K.; Foo, Y.; Shen, Q.; Bejaoui, A.; Zhao, Y.; Gokkaya, H.C.; Djurišić, A.B.; Zapien, J.A.; Chan, W.K.; et al. Enhanced performance of PTB7: PC71BM solar cells via different morphologies of gold nanoparticles. ACS Appl. Mater. Interfaces 2014, 6, 20676–20684. [Google Scholar] [CrossRef]
- Dlamini, M.W.; Mbuyise, X.G.; Mola, G.T. ZnO: Ag nano-particles decorated hole transport layer for improved photon harvesting. Appl. Phys. A 2022, 128, 125. [Google Scholar] [CrossRef]
- Liu, S.; Jiang, R.; You, P.; Zhu, X.; Wang, J.; Yan, F. Au/Ag core–shell nanocuboids for high-efficiency organic solar cells with broadband plasmonic enhancement. Energy Environ. Sci. 2016, 9, 898–905. [Google Scholar] [CrossRef]
- Nair, A.T.; Anoop, C.S.; Vinod, G.A.; Reddy, V.S. Efficiency enhancement in polymer solar cells using combined plasmonic effects of multi-positional silver nanostructures. Org. Electron. 2020, 86, 105872. [Google Scholar] [CrossRef]
- Ashagre, S.; Ogundele, A.K.; Ike, J.N.; Gebremichael, B.; Bekele, M.; Sharma, G.D.; Mola, G.T. Synergistic contribution of potassium sulfide doped with silver nanoparticles on the performance of thin film organic solar cells. J. Phys. Chem. Solids 2023, 177, 111290. [Google Scholar] [CrossRef]
- Mahajan, P.; Singh, A.; Datt, R.; Gupta, V.; Arya, S. Realization of inverted organic solar cells by using sol-gel synthesized ZnO/Y2O3 core/shell nanoparticles as electron transport layer. IEEE J. Photovolt. 2020, 10, 1744–1749. [Google Scholar] [CrossRef]
- Ike, J.N.; Taziwa, R.T. Optimizing Organic Photovoltaic Efficiency Through Controlled Doping of ZnS/Co Nanoparticles. Solids 2025, 6, 69. [Google Scholar] [CrossRef]
- Kristl, M.; Dojer, B.; Gyergyek, S.; Kristl, J. Synthesis of nickel and cobalt sulfide nanoparticles using a low cost sonochemical method. Heliyon 2017, 3, e00273. [Google Scholar] [CrossRef]
- Hamed, M.S.; Oseni, S.O.; Kumar, A.; Sharma, G.; Mola, G.T. Nickel sulphide nano-composite assisted hole transport in thin film polymer solar cells. Sol. Energy 2020, 195, 310–317. [Google Scholar] [CrossRef]
- Reddy, P.L.; Deshmukh, K.; Kovářík, T.; Reiger, D.; Nambiraj, N.A.; Lakshmipathy, R.; SK, K.P. Enhanced dielectric properties of green synthesized Nickel Sulphide (NiS) nanoparticles integrated polyvinylalcohol nanocomposites. Mater. Res. Express 2020, 7, 064007. [Google Scholar] [CrossRef]
- Buchmaier, C.; Glänzer, M.; Torvisco, A.; Poelt, P.; Wewerka, K.; Kunert, B.; Gatterer, K.; Trimmel, G.; Rath, T. Nickel sulfide thin films and nanocrystals synthesized from nickel xanthate precursors. J. Mater. Sci. 2017, 52, 10898–10914. [Google Scholar] [CrossRef]
- Ike, J.N.; Jili, N.; Kumar, A.; Sharma, G.; Mola, G.T. The impact of Ag/Co nanocomposite on organic charge transport medium for improved photocurrent in polymer solar cell. J. Polym. Sci. 2023, 61, 2721–2732. [Google Scholar] [CrossRef]
- Roushdy, N.; Elnouby, M.S.; Farag, A.A.M.; Ramadan, M.; El-Shazly, O.; El-Wahidy, E.F. Structural and electrical characterization of nickel sulfide nanoparticles. Opt. Quantum Electron. 2024, 56, 1794. [Google Scholar] [CrossRef]
- Seemab, M.; Nabi, G. Structural transformations and enhanced electrochemical performance of Co doped NiS2 nanosheets for supercapacitor applications. Ceram. Int. 2024, 50, 27856–27866. [Google Scholar] [CrossRef]
- Aiswarya, K.; Raguram, T.; Rajni, K. Synthesis and characterisation of nickel cobalt sulfide nanoparticles by the solvothermal method for dye-sensitized solar cell applications. Polyhedron 2020, 176, 114267. [Google Scholar] [CrossRef]
- Mohamed, W.S.; Ezzeldien, M.; Alshammari, A.H.; Alshammari, K.; Alhassan, S.; Hadia, N.M.A. Facile hydrothermal synthesis and characterization of novel Co-doped ZnS nanoparticles with superior physical properties. Opt. Mater. 2024, 157, 116345. [Google Scholar] [CrossRef]
- Makuła, P.; Pacia, M.; Macyk, W. How to correctly determine the band gap energy of modified semiconductor photocatalysts based on UV–Vis spectra. J. Phys. Chem. Lett. 2018, 9, 6814–6817. [Google Scholar] [CrossRef]
- Wang, C.; MacKenzie, R.C.; Würfel, U.; Neher, D.; Kirchartz, T.; Deibel, C.; Saladina, M. Transport resistance dominates the fill factor losses in record organic solar cells. Adv. Energy Mater. 2025, 16, 2405889. [Google Scholar] [CrossRef]
- Mousavifar, S.M.; Ghasemi, M.; Haidari, G. Near and far-field plasmonic enhancement from thermally evaporated Ag nanostructures in polymer photovoltaic cells: Simulation and experimental study. IEEE J. Photovolt. 2020, 10, 1735–1743. [Google Scholar] [CrossRef]
- Röhr, J.A.; Moia, D.; Haque, S.A.; Kirchartz, T.; Nelson, J. Exploring the validity and limitations of the Mott–Gurney law for charge-carrier mobility determination of semiconducting thin-films. J. Phys. Condens. Matter 2018, 30, 105901. [Google Scholar] [CrossRef]
- Seimela, T.E.; Hamed, M.S.; Diale, M. Enhancing organic solar cell performance via Cu nanorods-doped PEDOT: PSS: A pathway to efficient charge transport and plasmonic enhancement. RSC Adv. 2025, 15, 25929–25939. [Google Scholar] [CrossRef]






| Device Architecture | NPs | NPs Location | PCE (%) Without NPs | PCE (%) with NPs | Ref |
|---|---|---|---|---|---|
| ITO/PEDOT:PSS:Au/PTB7:PC71BM/Ca | Au | PEDOT:PSS | 7.50 | 8.10 | [24] |
| ITO/PEDOT:PSS-ZnO:Ag/P3HT:PCBM/LiF/Al | ZnO/Ag | P3HT:PCBM | 2.56 | 4.88 | [25] |
| ITO/PEDOT:PSS/PTB7:PC71BM/Ca/Al | Au@Ag@SiO2 | PEDOT:PSS | 7.72 | 9.04 | [26] |
| ITO/ZnO/PTB7:PCBM/MoO3/Al | Ag | ZnO | 6.53 | 7.25 | [27] |
| ITO/PEDOT:PSS/P3HT:PCBM:NPs/LiF/Al | K2S/Ag | P3HT:PCBM | 2.30 | 5.12 | [28] |
| ITO/ZnS/Y2O3/PTB7:PC71BM/MoO3/Ag | ZnO/Y2O3 | ZnO | 5.77 | 6.22 | [29] |
| ITO/PEDOT:PSS/PTB7:PC71BM/Ca/Al | Au@Ag@SiO2 | PTB7:PC71BM | 7.72 | 9.56 | [26] |
| ITO/PEDOT:PSS/P3HT:PCBM:NPs/LiF/Al | ZnS/Co | P3HT:PCBM | 2.35 | 4.76 | [30] |
(wt%) | (eV) | (eV) | (V) | (mAcm−2) | (%) | (%) | (Ωcm2) |
|---|---|---|---|---|---|---|---|
| 0% (Pristine) | 1.76 | 1.21 | 0.55 | 10.21 | 45.33 | 2.48 | 770 |
| 1% | 1.57 | 1.01 | 0.56 | 16.01 | 56.71 | 6.11 | 282 |
| 2% | 1.63 | 1.07 | 0.56 | 14.79 | 52.53 | 5.03 | 463 |
| 3% | 1.70 | 1.14 | 0.56 | 12.64 | 50.68 | 3.85 | 549 |
(wt%) | () | () |
|---|---|---|
| 0% (Pristine) | ||
| 1% | ||
| 2% | ||
| 3% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ike, J.N.; Taziwa, R.T. Effective Energy Harvesting in Polymer Solar Cells Using NiS/Co as Nanocomposite Doping. Micro 2026, 6, 22. https://doi.org/10.3390/micro6010022
Ike JN, Taziwa RT. Effective Energy Harvesting in Polymer Solar Cells Using NiS/Co as Nanocomposite Doping. Micro. 2026; 6(1):22. https://doi.org/10.3390/micro6010022
Chicago/Turabian StyleIke, Jude N., and Raymond Tichaona Taziwa. 2026. "Effective Energy Harvesting in Polymer Solar Cells Using NiS/Co as Nanocomposite Doping" Micro 6, no. 1: 22. https://doi.org/10.3390/micro6010022
APA StyleIke, J. N., & Taziwa, R. T. (2026). Effective Energy Harvesting in Polymer Solar Cells Using NiS/Co as Nanocomposite Doping. Micro, 6(1), 22. https://doi.org/10.3390/micro6010022







