Comparison of the Therapeutic Effects of Native and Anionic Nanofibrillar Cellulose Hydrogels for Full-Thickness Skin Wound Healing
Abstract
:1. Introduction
2. Materials and Methods
2.1. Nanofibrillar Cellulose
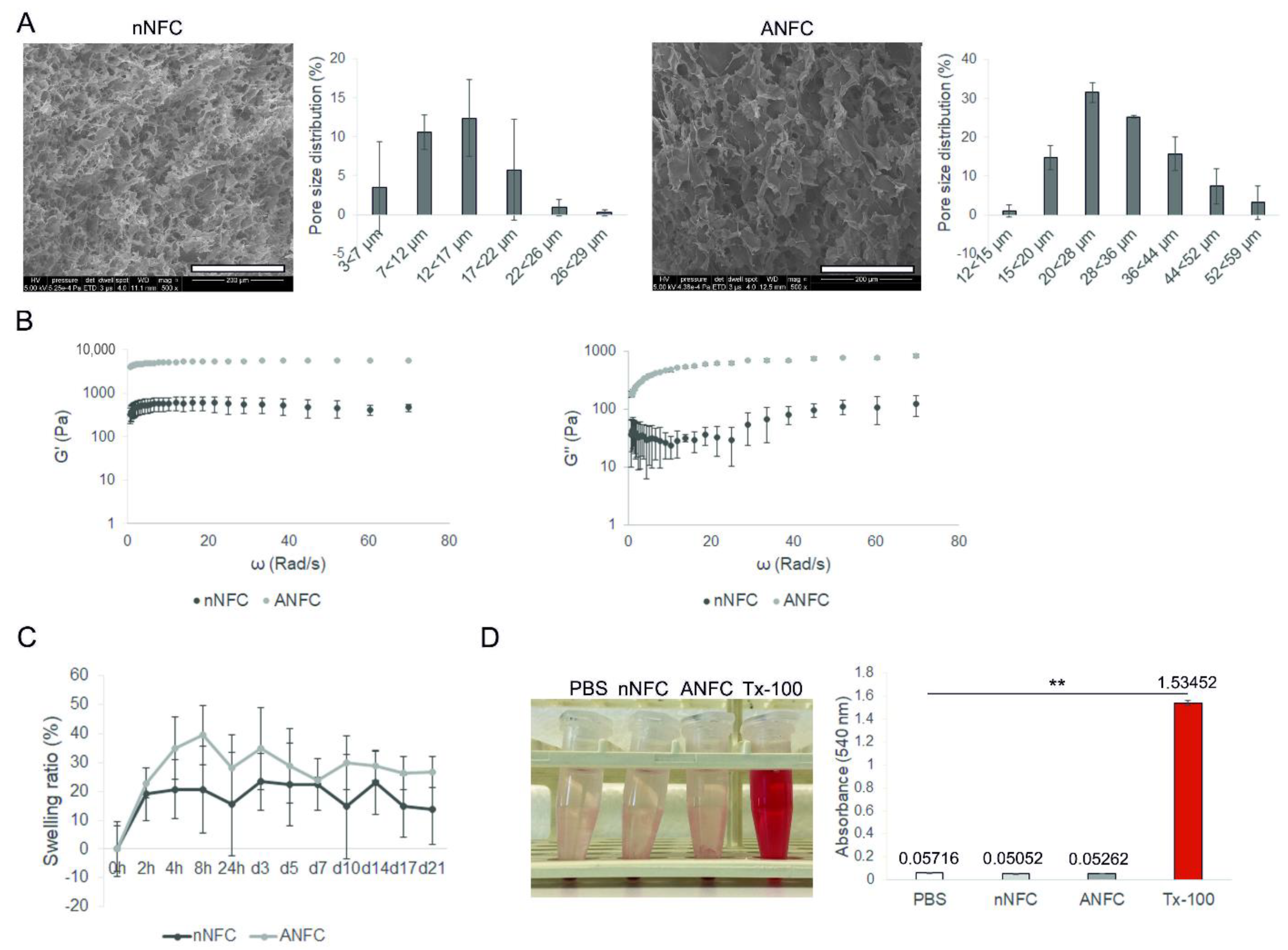
2.2. Scanning Electron Microscopy (SEM)
2.3. Rheological Measurements
2.4. Swelling and Water Retention Profile
2.5. Hemolytic Activity
2.6. Animals
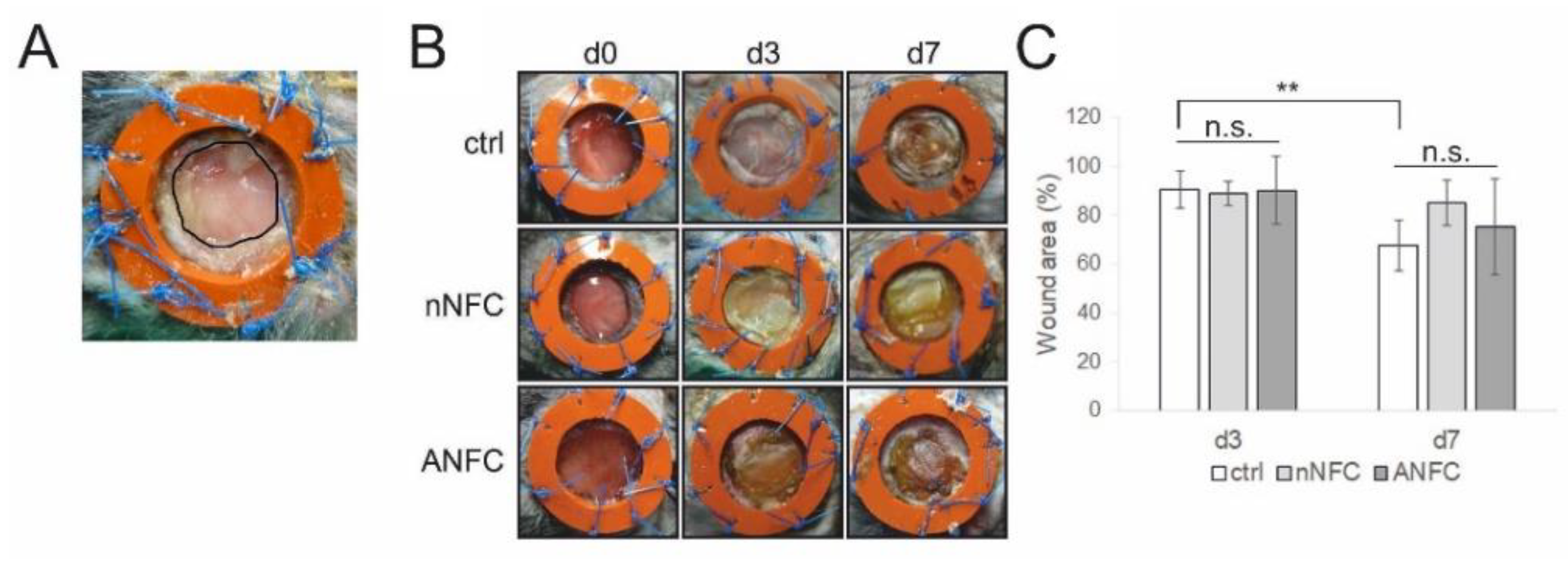
2.7. Surgical Procedure
2.8. Measurement of Wound Closure Rate
2.9. Exclusion Criteria
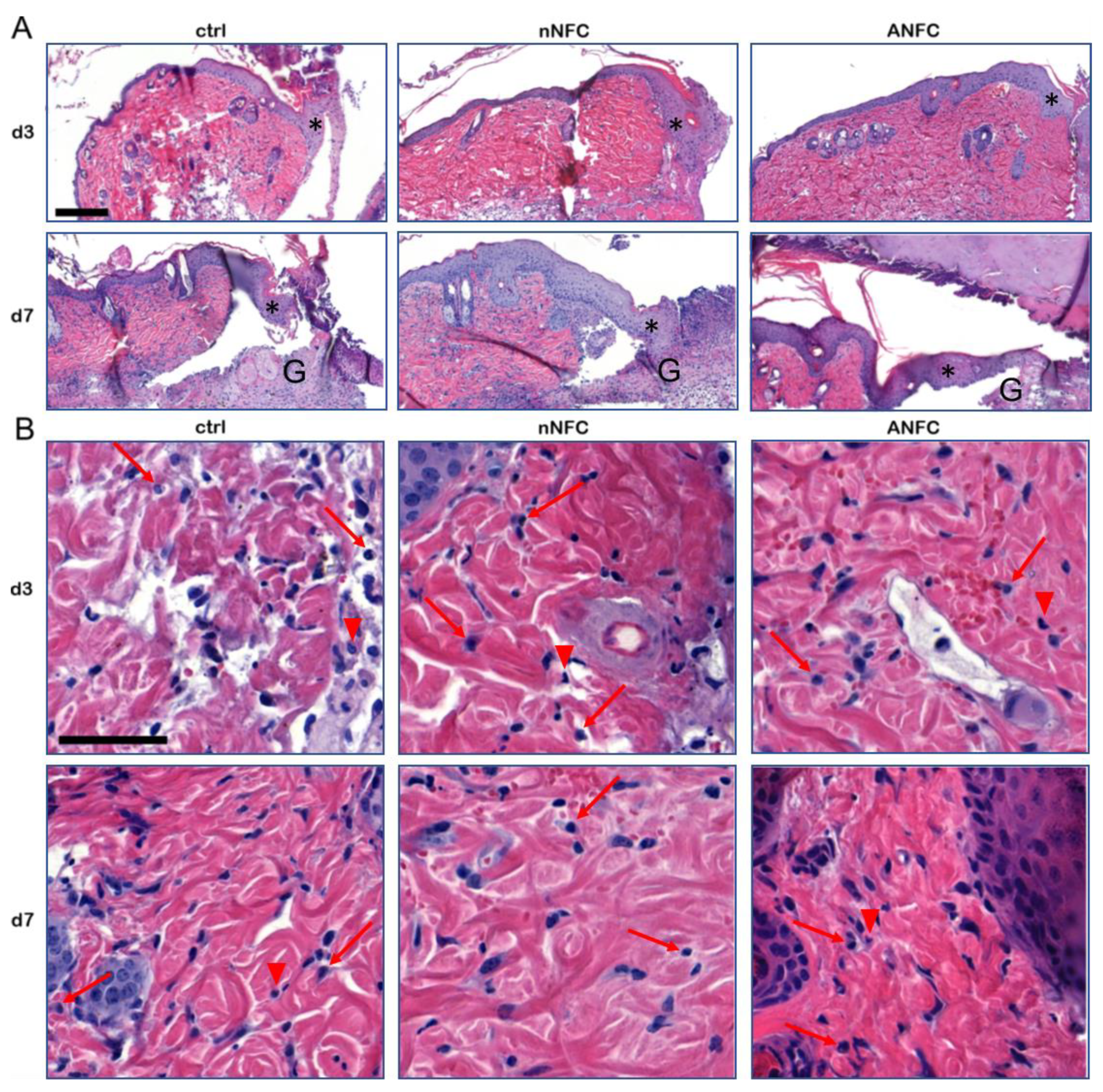
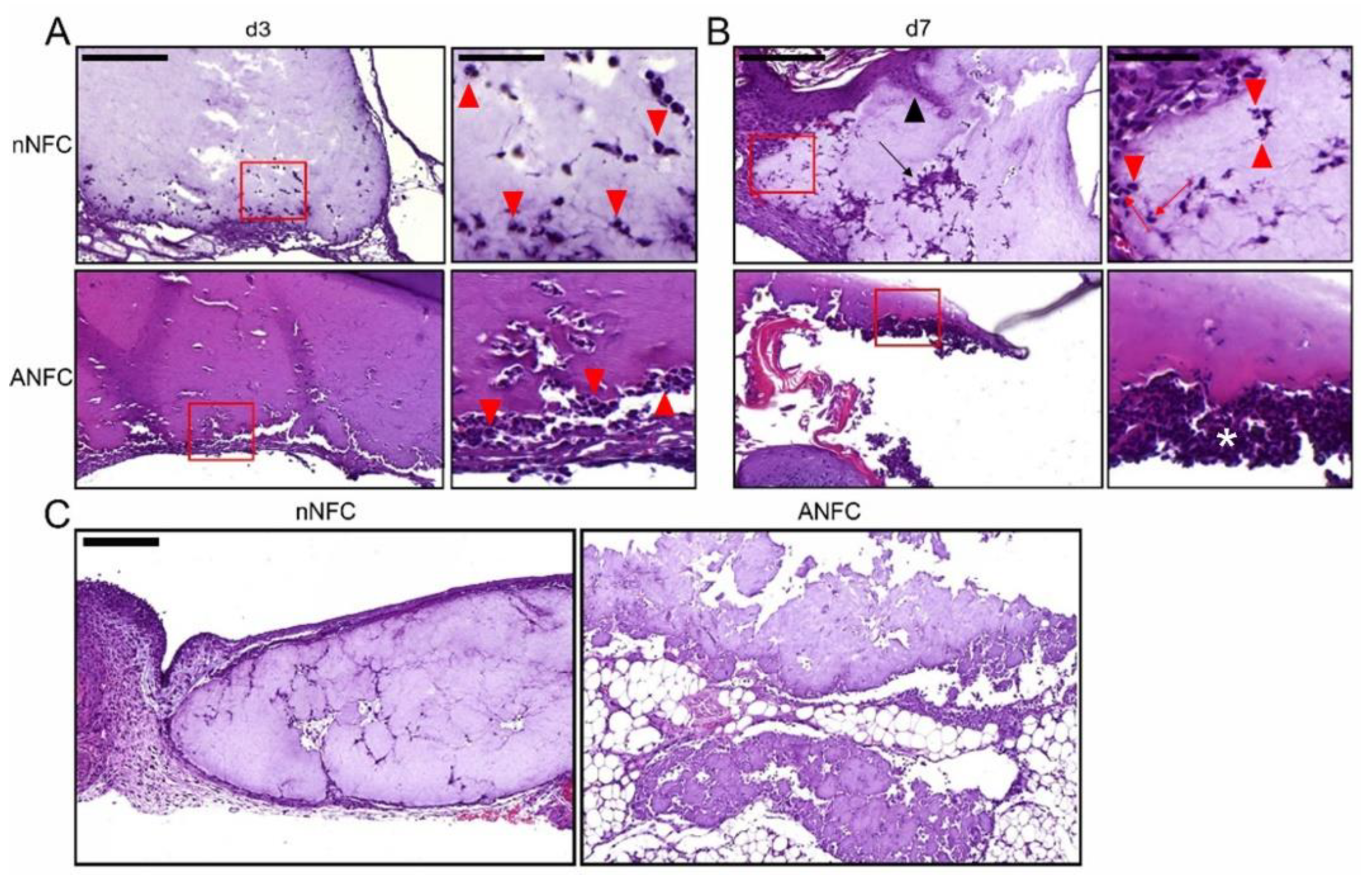
2.10. Histopathology and Immunohistochemistry
2.11. Immunofluorescence
2.12. Western Blotting
2.13. Statistical Analysis
3. Results
3.1. Characterization of NFC Hydrogels
3.2. The Effect of Nanofibrillar Cellulose Hydrogels on Wound Closure and Inflammation
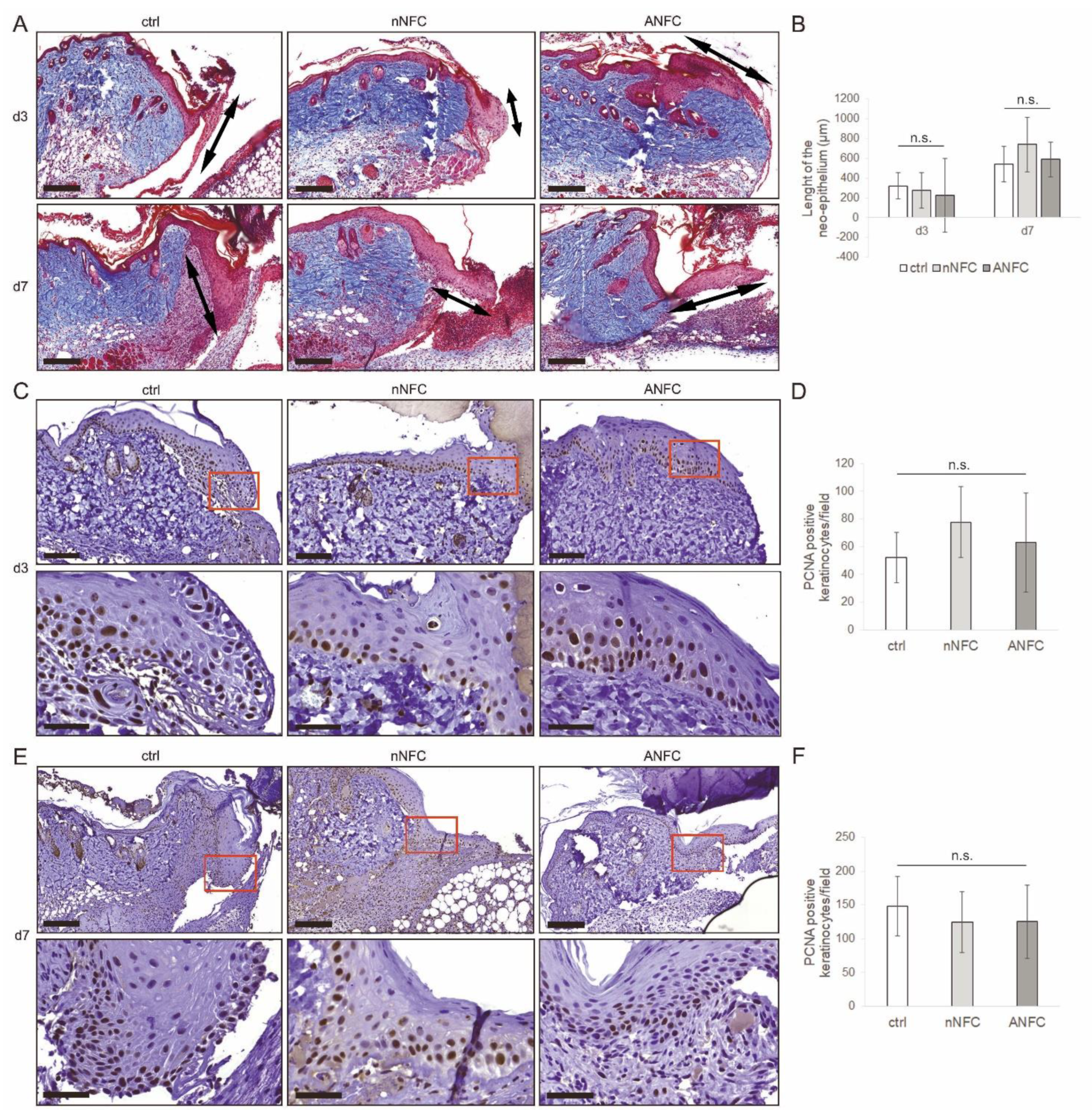
3.3. Nanofibrillar Cellulose Hydrogels Provide Efficient Wound Re-Epithelialization
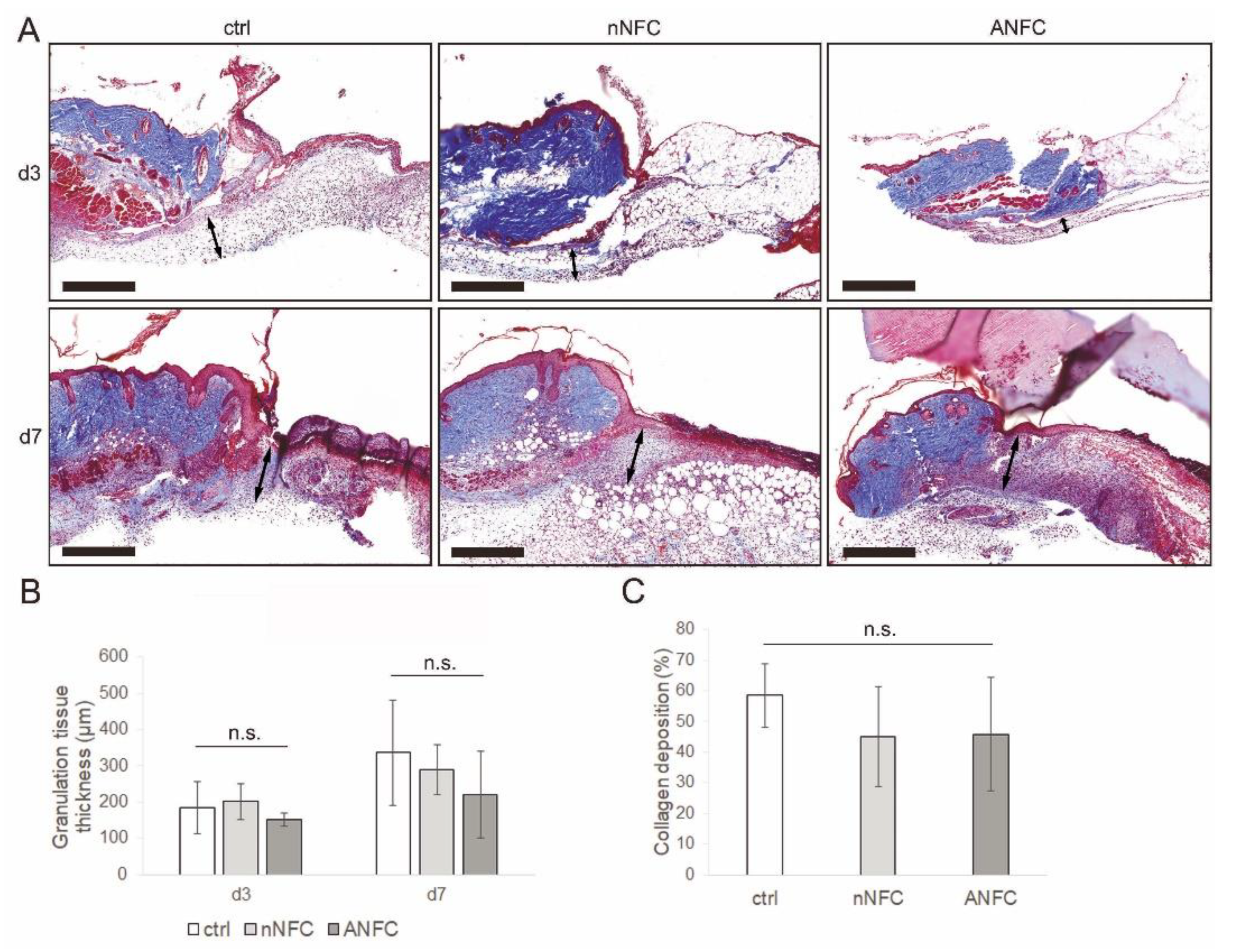
3.4. Granulation Tissue Formation Appears Normal in NFC Hydrogel-Treated Wounds
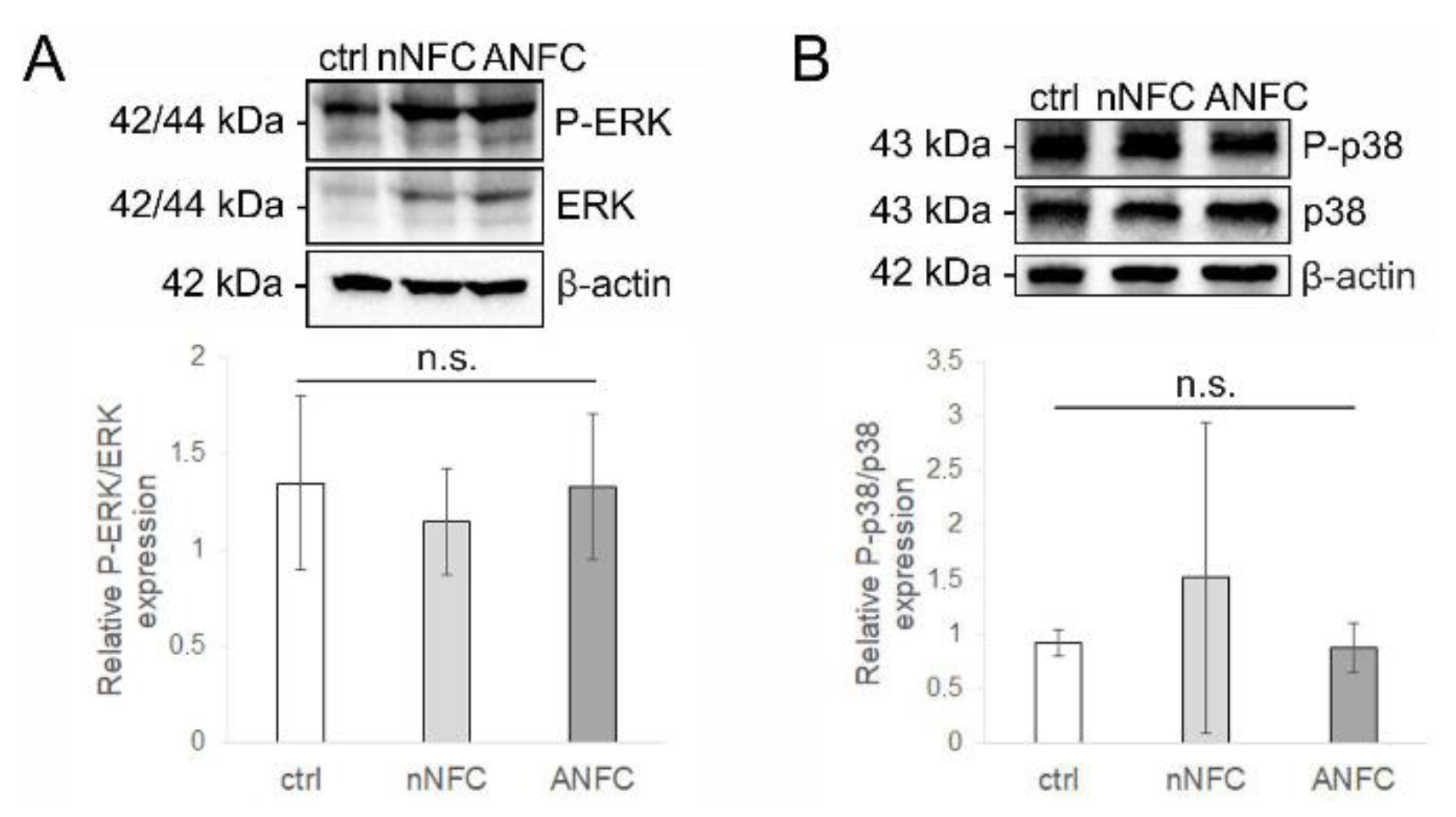
3.5. Nanofibrillar Cellulose Hydrogels Provide Normal Angiogenesis and Tissue Maturation in a Wound
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| α-SMA | Alpha-smooth muscle actin |
| ANFC | Anionic nanofibrillar cellulose |
| BSA | Bovine serum albumin |
| CD31 | Platelet endothelial cell adhesion molecule (PECAM-1) |
| Ctrl | Control |
| ECM | Extracellular matrix |
| ERK | Extracellular signal-regulated kinase |
| G’ | Storage modulus |
| G’’ | Loss modulus |
| H&E | Hematoxylin and eosin |
| IHC | Immunohistochemical |
| MAPK | Mitogen-activated protein kinase |
| NFC | Nanofibrillar cellulose |
| nNFC | Native nanofibrillar cellulose |
| PBS | Phosphate-buffered saline |
| PCNA | Proliferating cell nuclear antigen |
| RBC | Red blood cell |
| RT | Room temperature |
| SD | Standard deviation |
| SEM | Scanning electron microscopy |
| TBS-T | Tris buffered saline with Tween 20 |
| TEMPO | 2,2,6,6-tetramethylpiperidine-1-oxyl |
| TGF-β1 | Transforming growth factor beta 1 |
References
- Velnar, T.; Bailey, T.; Smrkolj, V. The Wound Healing Process: An Overview of the Cellular and Molecular Mechanisms. J. Int. Med. Res. 2009, 37, 1528–1542. [Google Scholar] [CrossRef]
- Demidova-Rice, T.N.; Hamblin, M.R.; Herman, I.M. Acute and Impaired Wound Healing: Pathophysiology and current methods for drug delivery, part 1: Normal and chronic wounds: Biology, causes, and approaches to care. Adv. Ski. Wound Care 2012, 25, 304–314. [Google Scholar] [CrossRef] [Green Version]
- Hoque, J.; Prakash, R.G.; Paramanandham, K.; Shome, B.R.; Haldar, J. Biocompatible Injectable Hydrogel with Potent Wound Healing and Antibacterial Properties. Mol. Pharm. 2017, 14, 1218–1230. [Google Scholar] [CrossRef]
- Zhao, X.; Sun, X.; Yildirimer, L.; Lang, Q.; Lin, Z.Y.W.; Zheng, R.; Zhang, Y.; Cui, W.; Annabi, N.; Khademhosseini, A. Cell infiltrative hydrogel fibrous scaffolds for accelerated wound healing. Acta Biomater. 2017, 49, 66–77. [Google Scholar] [CrossRef] [Green Version]
- Kong, X.; Fu, J.; Shao, K.; Wang, L.; Lan, X.; Shi, J. Biomimetic hydrogel for rapid and scar-free healing of skin wounds inspired by the healing process of oral mucosa. Acta Biomater. 2019, 100, 255–269. [Google Scholar] [CrossRef]
- Kathawala, M.H.; Ng, W.L.; Liu, D.; Naing, M.W.; Yeong, W.Y.; Spiller, K.L.; Van Dyke, M.; Ng, K.W. Healing of Chronic Wounds: An Update of Recent Developments and Future Possibilities. Tissue Eng. Part B Rev. 2019, 25, 429–444. [Google Scholar] [CrossRef] [PubMed]
- Chouhan, D.; Dey, N.; Bhardwaj, N.; Mandal, B.B. Emerging and innovative approaches for wound healing and skin regeneration: Current status and advances. Biomaterials 2019, 216, 119267. [Google Scholar] [CrossRef]
- Soriano-Ruiz, J.L.; Gálvez-Martín, P.; Ruiz, E.L.; Suñer-Carbó, J.; Calpena-Campmany, A.C.; Marchal, J.A.; Clares-Naveros, B. Design and evaluation of mesenchymal stem cells seeded chitosan/glycosaminoglycans quaternary hydrogel scaffolds for wound healing applications. Int. J. Pharm. 2019, 570, 118632. [Google Scholar] [CrossRef] [PubMed]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Wang, C.; Varshney, R.R.; Wang, N.-A. Therapeutic cell delivery and fate control in hydrogels and hydrogel hybrids. Adv. Drug Deliv. Rev. 2010, 62, 699–710. [Google Scholar] [CrossRef]
- Naahidi, S.; Jafari, M.; Logan, M.; Wang, Y.; Yuan, Y.; Bae, H.; Dixon, B.; Chen, P. Biocompatibility of hydrogel-based scaffolds for tissue engineering applications. Biotechnol. Adv. 2017, 35, 530–544. [Google Scholar] [CrossRef]
- Kamoun, E.A.; Kenawy, E.-R.S.; Chen, X. A review on polymeric hydrogel membranes for wound dressing applications: PVA-based hydrogel dressings. J. Adv. Res. 2017, 8, 217–233. [Google Scholar] [CrossRef] [PubMed]
- Bai, Q.; Han, K.; Dong, K.; Zheng, C.; Zhang, Y.; Long, Q.; Lu, T. Potential Applications of Nanomaterials and Technology for Diabetic Wound Healing. Int. J. Nanomed. 2020, 15, 9717–9743. [Google Scholar] [CrossRef]
- Tavakoli, S.; Klar, A.S. Advanced Hydrogels as Wound Dressings. Biomolecules 2020, 10, 1169. [Google Scholar] [CrossRef] [PubMed]
- Mantha, S.; Pillai, S.; Khayambashi, P.; Upadhyay, A.; Zhang, Y.; Tao, O.; Pham, H.M.; Tran, S.D. Smart Hydrogels in Tissue Engineering and Regenerative Medicine. Materials 2019, 12, 3323. [Google Scholar] [CrossRef] [Green Version]
- Pourshahrestani, S.; Zeimaran, E.; Kadri, N.A.; Mutlu, N.; Boccaccini, A.R. Polymeric Hydrogel Systems as Emerging Biomaterial Platforms to Enable Hemostasis and Wound Healing. Adv. Healtch Mater. 2020, 9, 2000905. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration (FDA). Medical Devices Containing Materials Derived from Animal Sources (Except for In Vitro Diagnostics Devices) Guidance for Industry and Food and Drug Administration Staff; U.S. Food and Drug Administration: Rockville, MD, USA, 25 March 2019; pp. 1–10.
- Sahana, T.G.; Rekha, P.D. Biopolymers: Applications in wound healing and skin tissue engineering. Mol. Biol. Rep. 2018, 45, 2857–2867. [Google Scholar] [CrossRef]
- Bhattacharya, M.; Malinen, M.M.; Laurén, P.; Lou, Y.-R.; Kuisma, S.W.; Kanninen, L.; Lille, M.; Corlu, A.; GuGuen-Guillouzo, C.; Ikkala, O.; et al. Nanofibrillar cellulose hydrogel promotes three-dimensional liver cell culture. J. Control. Release 2012, 164, 291–298. [Google Scholar] [CrossRef]
- Junker, J.P.; Kamel, R.A.; Caterson, E.; Eriksson, E. Clinical Impact Upon Wound Healing and Inflammation in Moist, Wet, and Dry Environments. Adv. Wound Care 2013, 2, 348–356. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Nypelö, T.; Salas, C.; Arboleda, J.; Hoeger, I.C.; Rojas, O. Cellulose Nanofibrils. J. Renew. Mater. 2013, 1, 195–211. [Google Scholar] [CrossRef] [Green Version]
- Hakkarainen, T.; Koivuniemi, R.; Kosonen, M.; Escobedo-Lucea, C.; Sanz-García, A.; Vuola, J.; Valtonen, J.; Tammela, P.; Mäkitie, A.; Luukko, K.; et al. Nanofibrillar cellulose wound dressing in skin graft donor site treatment. J. Control. Release 2016, 244, 292–301. [Google Scholar] [CrossRef]
- Koivuniemi, R.; Hakkarainen, T.; Kiiskinen, J.; Kosonen, M.; Vuola, J.; Valtonen, J.; Luukko, K.; Kavola, H.; Yliperttula, M. Clinical Study of Nanofibrillar Cellulose Hydrogel Dressing for Skin Graft Donor Site Treatment. Adv. Wound Care 2020, 9, 199–210. [Google Scholar] [CrossRef]
- Nuutila, K.; Laukkanen, A.; Lindford, A.; Juteau, S.; Nuopponen, M.; Vuola, J.; Kankuri, E. Inhibition of Skin Wound Contraction by Nanofibrillar Cellulose Hydrogel. Plast. Reconstr. Surg. 2018, 141, 357e–366e. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Kimura, S.; Nishiyama, Y.; Isogai, A. Cellulose Nanofibers Prepared by TEMPO-Mediated Oxidation of Native Cellulose. Biomacromolecules 2007, 8, 2485–2491. [Google Scholar] [CrossRef]
- Alexandrescu, L.; Syverud, K.; Gatti, A.; Chinga-Carrasco, G. Cytotoxicity tests of cellulose nanofibril-based structures. Cellullose 2013, 20, 1765–1775. [Google Scholar] [CrossRef]
- Azoidis, I.; Metcalfe, J.; Reynolds, J.; Keeton, S.; Hakki, S.S.; Sheard, J.; Widera, D. Three-dimensional cell culture of human mesenchymal stem cells in nanofibrillar cellulose hydrogels. MRS Commun. 2017, 7, 458–465. [Google Scholar] [CrossRef] [Green Version]
- Sheard, J.J.; Bicer, M.; Meng, Y.; Frigo, A.; Aguilar, R.M.; Vallance, T.M.; Iandolo, D.; Widera, D. Optically Transparent Anionic Nanofibrillar Cellulose Is Cytocompatible with Human Adipose Tissue-Derived Stem Cells and Allows Simple Imaging in 3D. Stem Cells Int. 2019, 2019, 3106929. [Google Scholar] [CrossRef]
- Paukkonen, H.; Kunnari, M.; Laurén, P.; Hakkarainen, T.; Auvinen, V.-V.; Oksanen, T.; Koivuniemi, R.; Yliperttula, M.; Laaksonen, T. Nanofibrillar cellulose hydrogels and reconstructed hydrogels as matrices for controlled drug release. Int. J. Pharm. 2017, 532, 269–280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galiano, R.D.; Michaels, V.J.; Dobryansky, M.; Levine, J.P.; Gurtner, G.C. Quantitative and reproducible murine model of excisional wound healing. Wound Repair Regen. 2004, 12, 485–492. [Google Scholar] [CrossRef]
- Dunn, L.; Prosser, H.C.G.; Tan, M.-J.; Vanags, L.Z.; Ng, M.K.C.; Bursill, C. Murine Model of Wound Healing. J. Vis. Exp. 2013, 75, e50265. [Google Scholar] [CrossRef] [Green Version]
- Wright-Williams, S.; Flecknell, P.A.; Roughan, J.V. Comparative Effects of Vasectomy Surgery and Buprenorphine Treatment on Faecal Corticosterone Concentrations and Behaviour Assessed by Manual and Automated Analysis Methods in C57 and C3H Mice. PLoS ONE 2013, 8, e75948. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign Body Reaction to Biomaterials. In Seminars in Immunology; Elsevier: Amsterdam, The Netherlands, 2008; Volume 20, pp. 86–100. [Google Scholar]
- Hinz, B.; Celetta, G.; Tomasek, J.J.; Gabbiani, G.; Chaponnier, C. Alpha-Smooth Muscle Actin Expression Upregulates Fibroblast Contractile Activity. Mol. Biol. Cell 2001, 12, 2730–2741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Z.; Shou, K.; Li, Z.; Jian, C.; Qi, B.; Yu, A. Negative pressure wound therapy promotes vessel destabilization and maturation at various stages of wound healing and thus influences wound prognosis. Exp. Ther. Med. 2016, 11, 1307–1317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albelda, S.M.; A Muller, W.; A Buck, C.; Newman, P.J. Molecular and cellular properties of PECAM-1 (endoCAM/CD31): A novel vascular cell-cell adhesion molecule. J. Cell Biol. 1991, 114, 1059–1068. [Google Scholar] [CrossRef]
- Horak, E.; Klenk, N.; Leek, R.; LeJeune, S.; Smith, K.; Stuart, N.; Harris, A.; Greenall, M.; Stepniewska, K. Angiogenesis, assessed by platelet/endothelial cell adhesion molecule antibodies, as indicator of node metastases and survival in breast cancer. Lancet 1992, 340, 1120–1124. [Google Scholar] [CrossRef]
- Roberts, A.B.; Sporn, M.B.; Assoian, R.K.; Smith, J.M.; Roche, N.S.; Wakefield, L.; Heine, U.I.; Liotta, L.A.; Falanga, V.; Kehrl, J.H. Transforming growth factor type beta: Rapid induction of fibrosis and angiogenesis in vivo and stimulation of collagen formation in vitro. Proc. Natl. Acad. Sci. USA 1986, 83, 4167–4171. [Google Scholar] [CrossRef] [Green Version]
- Desmoulière, A.; Geinoz, A.; Gabbiani, F. Transforming growth factor-beta 1 induces alpha-smooth muscle actin expression in granulation tissue myofibroblasts and in quiescent and growing cultured fibroblasts. J. Cell Biol. 1993, 122, 103–111. [Google Scholar] [CrossRef] [Green Version]
- Border, W.A.; Noble, N.A. Transforming Growth Factor β in Tissue Fibrosis. N. Engl. J. Med. 1994, 331, 1286–1292. [Google Scholar] [CrossRef]
- Loughlin, D.T.; Artlett, C.M. Modification of Collagen by 3-Deoxyglucosone Alters Wound Healing through Differential Regulation of p38 MAP Kinase. PLoS ONE 2011, 6, e18676. [Google Scholar] [CrossRef] [Green Version]
- Escuin-Ordinas, H.; Li, S.; Xie, M.W.; Sun, L.; Hugo, W.; Huang, R.R.; Jiao, J.; De-Faria, F.M.; Realegeno, S.; Krystofinski, P.; et al. Cutaneous wound healing through paradoxical MAPK activation by BRAF inhibitors. Nat. Commun. 2016, 7, 12348. [Google Scholar] [CrossRef]
- Beserra, F.P.; Xue, M.; Maia, G.L.D.A.; Rozza, A.L.; Pellizzon, C.H.; Jackson, C.J. Lupeol, a Pentacyclic Triterpene, Promotes Migration, Wound Closure, and Contractile Effect In Vitro: Possible Involvement of PI3K/Akt and p38/ERK/MAPK Pathways. Molecules 2018, 23, 2819. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Brien, F.J. Biomaterials & scaffolds for tissue engineering. Mater. Today 2011, 14, 88–95. [Google Scholar] [CrossRef]
- Rittié, L. Cellular mechanisms of skin repair in humans and other mammals. J. Cell Commun. Signal. 2016, 10, 103–120. [Google Scholar] [CrossRef] [Green Version]
- Sorg, H.; Tilkorn, D.J.; Hager, S.; Hauser, J.; Mirastschijski, U. Skin Wound Healing: An Update on the Current Knowledge and Concepts. Eur. Surg. Res. 2017, 58, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Volk, S.W.; Bohling, M.W. Comparative wound healing: Are the small animal veterinarian’s clinical patients an improved translational model for human wound healing research? Wound Repair Regen. 2013, 21, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Desmoulière, A.; Chaponnier, C.; Gabbiani, G. Tissue repair, contraction, and the myofibroblast. Wound Repair Regen. 2005, 13, 7–12. [Google Scholar] [CrossRef]
- Pääkkö, M.; Ankerfors, M.; Kosonen, H.; Nykänen, A.; Ahola, S.; Österberg, M.; Ruokolainen, J.; Laine, J.; Larsson, P.T.; Ikkala, O.; et al. Enzymatic Hydrolysis Combined with Mechanical Shearing and High-Pressure Homogenization for Nanoscale Cellulose Fibrils and Strong Gels. Biomacromolecules 2007, 8, 1934–1941. [Google Scholar] [CrossRef]
- Anderson, J.M. Inflammatory response to implants. ASAIO Trans. 1988, 34, 101–107. [Google Scholar] [CrossRef]
- A DiPietro, L. Wound healing: The role of the macrophage and other immune cells. Shock 1995, 4, 233–240. [Google Scholar] [CrossRef]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nat. Cell Biol. 2008, 453, 314–321. [Google Scholar] [CrossRef]
- Sheikh, Z.; Brooks, P.J.; Barzilay, O.; Fine, N.; Glogauer, M. Macrophages, Foreign Body Giant Cells and Their Response to Implantable Biomaterials. Materials 2015, 8, 5671–5701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rashad, A.; Suliman, S.; Mustafa, M.; Pedersen, T.Ø.; Campodoni, E.; Sandri, M.; Syverud, K.; Mustafa, K. Inflammatory responses and tissue reactions to wood-Based nanocellulose scaffolds. Mater. Sci. Eng. C 2019, 97, 208–221. [Google Scholar] [CrossRef] [PubMed]
- Sadtler, K.; Allen, B.W.; Estrellas, K.; Housseau, F.; Pardoll, D.M.; Elisseeff, J.H. The scaffold immune microenvironment: Biomateri-al-mediated immune polarization in traumatic and nontraumatic applications. Tissue Eng. Part A 2017, 23, 1044–1053. [Google Scholar] [CrossRef] [PubMed]
- Lopes, V.R.; Sanchez-Martinez, C.; Strømme, M.; Ferraz, N. In vitro biological responses to nanofibrillated cellulose by human dermal, lung and immune cells: Surface chemistry aspect. Part. Fibre Toxicol. 2017, 14, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lou, Y.-R.; Kanninen, L.; Kuisma, T.; Niklander, J.; Noon, L.A.; Burks, D.; Urtti, A.; Yliperttula, M. The Use of Nanofibrillar Cellulose Hydrogel As a Flexible Three-Dimensional Model to Culture Human Pluripotent Stem Cells. Stem Cells Dev. 2014, 23, 380–392. [Google Scholar] [CrossRef] [Green Version]
- Malinen, M.M.; Kanninen, L.; Corlu, A.; Isoniemi, H.M.; Lou, Y.-R.; Yliperttula, M.L.; Urtti, A.O. Differentiation of liver progenitor cell line to functional organotypic cultures in 3D nanofibrillar cellulose and hyaluronan-gelatin hydrogels. Biomaterials 2014, 35, 5110–5121. [Google Scholar] [CrossRef]

| Histopathologic Scale to Evaluate Inflammation | |||||||
|---|---|---|---|---|---|---|---|
| Day | Group | Total | Grade 1 | Grade 2 | Grade 3 | Grade 4 | p Value |
| 3 | ctrl | 5 | 0 | 2 | 3 | 0 | |
| nNFC | 4 | 0 | 1 | 3 | 0 | ||
| ANFC | 4 | 2 | 1 | 1 | 0 | 0.129 | |
| 7 | ctrl | 4 | 0 | 1 | 3 | 0 | |
| nNFC | 4 | 1 | 1 | 2 | 0 | ||
| ANFC | 3 | 2 | 0 | 1 | 0 | 0.323 | |
| Histopathologic Scale to Evaluate Inflammation | |||||||
|---|---|---|---|---|---|---|---|
| Day | Group | Total | Grade 1 | Grade 2 | Grade 3 | Grade 4 | p Value |
| 3 | ctrl | 6 | 2 | 3 | 1 | 0 | |
| nNFC | 6 | 2 | 3 | 1 | 0 | ||
| ANFC | 6 | 2 | 2 | 2 | 0 | 0.918 | |
| 7 | ctrl | 4 | 0 | 0 | 4 | 0 | |
| nNFC | 5 | 0 | 1 | 4 | 0 | ||
| ANFC | 2 | 0 | 0 | 2 | 0 | 0.6 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koivuniemi, R.; Xu, Q.; Snirvi, J.; Lara-Sáez, I.; Merivaara, A.; Luukko, K.; Nuopponen, M.; Wang, W.; Yliperttula, M. Comparison of the Therapeutic Effects of Native and Anionic Nanofibrillar Cellulose Hydrogels for Full-Thickness Skin Wound Healing. Micro 2021, 1, 194-214. https://doi.org/10.3390/micro1020015
Koivuniemi R, Xu Q, Snirvi J, Lara-Sáez I, Merivaara A, Luukko K, Nuopponen M, Wang W, Yliperttula M. Comparison of the Therapeutic Effects of Native and Anionic Nanofibrillar Cellulose Hydrogels for Full-Thickness Skin Wound Healing. Micro. 2021; 1(2):194-214. https://doi.org/10.3390/micro1020015
Chicago/Turabian StyleKoivuniemi, Raili, Qian Xu, Jasmi Snirvi, Irene Lara-Sáez, Arto Merivaara, Kari Luukko, Markus Nuopponen, Wenxin Wang, and Marjo Yliperttula. 2021. "Comparison of the Therapeutic Effects of Native and Anionic Nanofibrillar Cellulose Hydrogels for Full-Thickness Skin Wound Healing" Micro 1, no. 2: 194-214. https://doi.org/10.3390/micro1020015
APA StyleKoivuniemi, R., Xu, Q., Snirvi, J., Lara-Sáez, I., Merivaara, A., Luukko, K., Nuopponen, M., Wang, W., & Yliperttula, M. (2021). Comparison of the Therapeutic Effects of Native and Anionic Nanofibrillar Cellulose Hydrogels for Full-Thickness Skin Wound Healing. Micro, 1(2), 194-214. https://doi.org/10.3390/micro1020015








