Examining Evidence of Benefits and Risks for Pasteurizing Donor Breastmilk
Abstract
1. Introduction
2. Methodologic Approach
3. Results
3.1. Raw Breastmilk Ecosystem
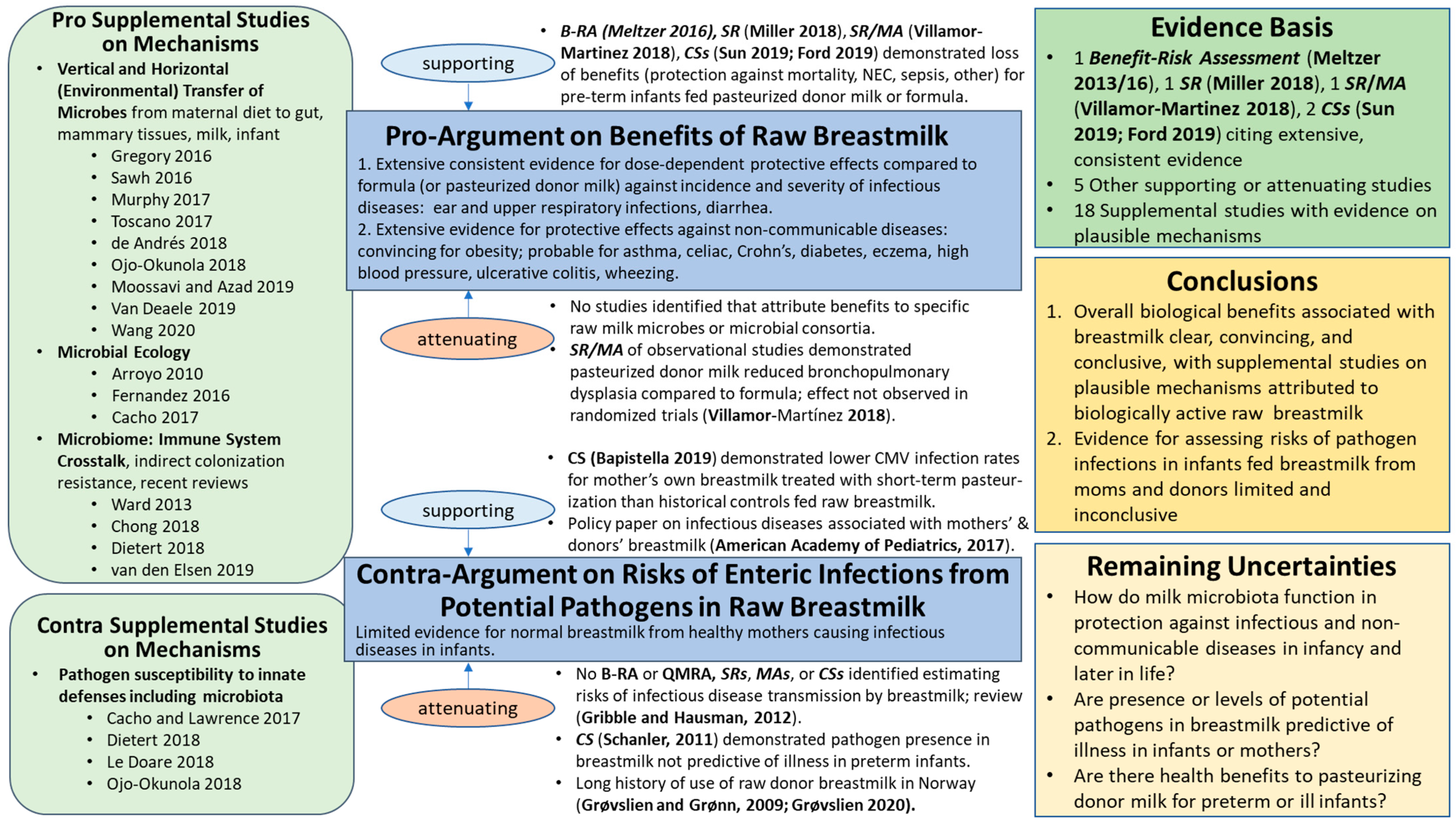
3.1.1. Benefits: Pro-Argument
‘the benefits of breastmilk clearly outweigh the possible risks from contaminants’ for neurodevelopment, defense against infections, and growth abnormalities. Probable beneficial effects later in life were noted for type 1 and type 2 diabetes, as well as high blood pressure. No conclusions were drawn regarding other diseases of the immune system (allergy, asthma and wheezing, atopic dermatitis) due to ‘inconclusive results on the benefit side and few and disperse studies on the risk side’.
Supporting
Attenuating
- Vertical transmission of microbes from mother to infant, including in utero microbes, anaerobes typical of the GI tract, strains of oral probiotic supplements during pregnancy and lactation, and microbiota of breast tissue, vaginal tissues, and skin;
- Anti-infective functions, including colonization resistance by commensals for resistance to acute infections and induction of oral tolerance;
- Immunomodulatory activities of T-regulatory cells, peripheral blood mononuclear cell subsets, cytotoxic T-cells (CD8+), natural killer (NK) cells (non-specific), and cytokines for milk microbiota. Decrease exaggerated inflammatory responses to colonizing bacteria (commensals and opportunistic pathogens under certain conditions);
- Anti-allergic properties attributed to LABs and commensals of milk microbiota that decrease the occurrence and severity of allergic responses in animal models and some human studies; and
- Metabolic activities of LABs and commensals essential for digestion of oligosaccharides into short chain fatty acids (SCFAs) that become an energy source for host cells in the colon, thus increasing nutrient availability and absorption for the host. Further, Sozańska [35] cited studies demonstrating that SCFAs in the GI tract enhance the epithelial barrier function of the gut, influence bone marrow dendritic cell maturation, and inhibited Th2-dependent response, interconnecting metabolic functions to functions 2, 3, and 4 above.
3.1.2. Risks: Contra-Argument
Supporting
Attenuating
3.1.3. Benefit–Risk Conclusions
3.1.4. Remaining Uncertainties
- How do milk microbiota function in protection against infectious and non-communicable diseases in infancy and later in life?
- Are presence or levels of potential pathogens in breastmilk predictive of illness in infants or mothers?
- Are there health benefits to alternative pasteurization conditions for feeding donor milk to preterm or sick infants? Particularly, what risk management strategies (e.g., conventional Holder pasteurization and alternative conditions such as short-term pasteurization) provide optimal balance of health benefits and protection against neonatal disease?
4. Discussion
4.1. Might Scientific Revolutions Be Shifting Our Paradigms?
4.2. Updating Earlier Notions from Science, Medicine, and Risk Analysis
4.3. Updating Preconceived Notions on Breastmilk Ecosystem Structure and Function
4.4. Including Benefits of Microbiota-Mediated Colonization Resistance
| Direct Mechanisms of Microbiota-Medicated Colonization Resistance | Indirect Mechanisms of Microbiota-Medicated Colonization Resistance: |
|---|---|
Outcompete enteropathogens for:
|
|
Antagonize or kill enteropathogens by directly producing:
|
4.5. Updating Earlier Notions from Decision Science
5. Future Direction
‘The biggest challenge, generalizing from the phrases cited earlier from the Orange Book [67], is to formulate the right analysis for the problem, and then, working with scientists and other experts who have the relevant information and experience, and the stakeholders who are concerned with the consequences, to do the analysis right and communicate it so the insights from the analysis are understood.’
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fitzstevens, J.L.; Smith, K.C.; Hagadorn, J.I.; Caimano, M.J.; Matson, A.P.; Brownell, E.A. Systematic Review of the Human Milk Microbiota. Nutr. Clin. Pract. 2016, 32, 354–364. [Google Scholar] [CrossRef]
- Lyons, K.E.; Ryan, C.A.; Dempsey, E.M.; Ross, R.P.; Stanton, C. Breast Milk, a Source of Beneficial Microbes and Associated Benefits for Infant Health. Nutrients 2020, 12, 1039. [Google Scholar] [CrossRef]
- Oikonomou, G.; Addis, M.F.; Chassard, C.; Nader-Macias, M.E.F.; Grant, I.; Delbès, C.; Even, S. Milk Microbiota: What Are We Exactly Talking About? Front. Microbiol. 2020, 11, 60. [Google Scholar]
- Zimmermann, P.; Curtis, N. Breast milk microbiota: A review of the factors that influence composition. J. Infect. 2020, 81, 17–47. [Google Scholar] [CrossRef]
- Carr, L.E.; Virmani, M.D.; Rosa, F.; Munblit, D.; Matazel, K.S.; Elolimy, A.A.; Yeruva, L. Role of Human Milk Bioactives on Infants’ Gut and Immune Health. Front. Immunol. 2021, 12, 290. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.; Tonkin, E.; Damarell, R.A.; McPhee, A.J.; Suganuma, M.; Suganuma, H.; Middleton, P.F.; Makrides, M.; Collins, C.T. A Systematic Review and Meta-Analysis of Human Milk Feeding and Morbidity in Very Low Birth Weight Infants. Nutrients 2018, 10, 707. [Google Scholar] [CrossRef]
- Villamor-Martínez, E.; Pierro, M.; Cavallaro, G.; Mosca, F.; Kramer, B.W.; Villamor, E. Donor Human Milk Protects against Bronchopulmonary Dysplasia: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 238. [Google Scholar] [CrossRef]
- Ford, S.L.; Lohmann, P.; Preidis, G.A.; Gordon, P.S.; O’Donnell, A.; Hagan, J.; Venkatachalam, A.; Balderas, M.; Luna, R.A.; Hair, A.B. Improved feeding tolerance and growth are linked to increased gut microbial community diversity in very-low-birth-weight infants fed mother’s own milk compared with donor breast milk. Am. J. Clin. Nutr. 2019, 109, 1088–1097. [Google Scholar] [CrossRef]
- Dietert, R.R. A Focus on Microbiome Completeness and Optimized Colonization Resistance in Neonatology. NeoReviews 2018, 19, e78–e88. [Google Scholar] [CrossRef]
- Zhang, X.; Mushajiang, S.; Luo, B.; Tian, F.; Ni, Y.; Yan, W. The Composition and Concordance of Lactobacillus Populations of Infant Gut and the Corresponding Breast milk and Maternal Gut. Front. Microbiol. 2020, 11, 597911. [Google Scholar] [CrossRef]
- Boudry, G.; Charton, E.; Le Huerou-Luron, I.; Ferret-Bernard, S.; Le Gall, S.; Even, S.; Blat, S. The Relationship Between Breast Milk Components and the Infant Gut Microbiota. Front. Nutr. 2021, 8, 629740. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Luo, B.; Zhang, X.; Ni, Y.; Tian, F. Association and Occurrence of Bifidobacterial Phylotypes Between Breast Milk and Fecal Microbiomes in Mother–Infant Dyads During the First 2 Years of Life. Front. Microbiol. 2021, 12, 669442. [Google Scholar] [CrossRef] [PubMed]
- Nolan, L.S.; Rimer, J.M.; Good, M. The Role of Human Milk Oligosaccharides and Probiotics on the Neonatal Microbiome and Risk of Necrotizing Enterocolitis: A Narrative Review. Nutrients 2020, 12, 3052. [Google Scholar] [CrossRef]
- Seghesio, E.; De Geyter, C.; Vandenplas, Y. Probiotics in the Prevention and Treatment of Necrotizing Enterocolitis. Pediatr. Gastroenterol. Hepatol. Nutr. 2021, 24, 245–255. [Google Scholar] [CrossRef]
- Grøvslien, A.H.; Grønn, M. Donor Milk Banking and Breastfeeding in Norway. J. Hum. Lact. 2009, 25, 206–210. [Google Scholar] [CrossRef]
- Mizuno, K.; Sakurai, M.; Itabashi, K. Necessity of Human Milk Banking in Japan: Questionnaire Survey of Neonatologists. Pediatr. Int. 2015, 57, 639–644. [Google Scholar] [CrossRef]
- American Academy of Pediatrics (AAP) Committee Donor Human Milk for the High-Risk Infant: Preparation, Safety, and Usage Options in the United States. Pediatrics 2017, 139, e20163440.
- Klotz, D.; Jansen, S.; Glanzmann, R.; Haiden, N.; Fuchs, H.; Gebauer, C. Donor Human Milk Programs in German, Austrian and Swiss Neonatal Units-Findings from an International Survey. BMC Pediatr. 2020, 20, 235. [Google Scholar] [CrossRef]
- Picaud, J.C.; Buffin, R.; Gremmo-Feger, G.; Rigo, J.; Putet, G.; Casper, C.; Working group of the French Neonatal Society on fresh human milk use in preterm infants. Review Concludes That Specific Recommendations Are Needed to Harmonise the Provision of Fresh Mother’s Milk to Their Preterm Infants. Acta Paediatr. 2018, 107, 1145–1155. [Google Scholar] [CrossRef]
- Moro, G.E.; Billeaud, C.; Rachel, B.; Calvo, J.; Cavallarin, L.; Christen, L.; Escuder-Vieco, D.; Gaya, A.; Lembo, D.; Wesolowska, A.; et al. Processing of Donor Human Milk: Update and Recommendations From the European Milk Bank Association (EMBA). Front. Pediatr. 2019, 7, 49. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Donor Human Milk for Low-Birth-Weight Infants. Available online: https://www.who.int/elena/titles/donormilk_infants/en/ (accessed on 9 July 2019).
- World Health Organization (WHO). WHO/UNICEF Statement on the 40th Anniversary of the International Code of Marketing Breastmilk Substitutes. Available online: https://www.who.int/news/item/21-05-2021-WHO-UNICEF-statement-on-the-40th-anniversary-of-the-international-code-of-marketing-breastmilk-substitutes (accessed on 28 June 2021).
- Coleman, M.E.; Dietert, R.R.; North, D.W.; Stephenson, M.M. Enhancing Human Superorganism Ecosystem Resilience by Holistically Managing Our Microbes. Appl. Microbiol. 2021. Under review. [Google Scholar]
- Fischhoff, B.; Brewer, N.T.; Downs, J.S. Communicating Risks and Benefits: An Evidence-Based User’s Guide; Food and Drug Administration (FDA): Silver Spring, MD, USA; US Department of Health and Human Services: Washington, DC, USA, 2011. [Google Scholar]
- Wiedemann, P.; Schütz, H.; Spangenberg, A.; Krug, H.F. Evidence Maps: Communicating Risk Assessments in Societal Controversies: The Case of Engineered Nanoparticles. Risk Anal. 2011, 31, 1770–1783. [Google Scholar] [CrossRef]
- Nauta, M.J.; Andersen, R.; Pilegaard, K.; Pires, S.M.; Ravn-Haren, G.; Tetens, I.; Poulsen, M. Meeting the challenges in the development of risk-benefit assessment of foods. Trends Food Sci. Technol. 2018, 76, 90–100. [Google Scholar] [CrossRef]
- Meltzer, H.M.; Knutsen, H.K.; Løland, B.F.; Odland, J.Ø.; Skåre, J.U.; Torheim, L.E.; Brandtzæg, P. Benefit and Risk Assessment of Breastmilk for Infant Health in Norway. Eur. J. Nutr. Food Saf. 2016, 6, 101–110. [Google Scholar]
- Meltzer, H.M.; Brandtzæg, P.; Knutsen, H.K.; Løland, B.F.; Odland, J.Ø.; Skåre, J.U.; Torheim, L.E. Benefit and Risk Assessment of Breastmilk for Infant Health in Norway; Scientific Opinion by the Scientific Steering Committee of VKM: Nydalen, Norway, 2013. [Google Scholar]
- Sun, H.; Han, S.; Cheng, R.; Hei, M.; Kakulas, F.; Lee, S.K. Testing the feasibility and safety of feeding preterm infants fresh mother’s own milk in the NICU: A pilot study. Sci. Rep. 2019, 9, 941. [Google Scholar] [CrossRef]
- Ojo-Okunola, A.; Nicol, M.; Du Toit, E. Human Breast Milk Bacteriome in Health and Disease. Nutrients 2018, 10, 1643. [Google Scholar] [CrossRef]
- Sozańska, B. Raw Cow’s Milk and Its Protective Effect on Allergies and Asthma. Nutrients 2019, 11, 469. [Google Scholar] [CrossRef]
- Keim, S.A.; Hogan, J.S.; McNamara, K.A.; Gudimetla, V.; Dillon, C.E.; Kwiek, J.J.; Geraghty, S.R. Microbial Contamination of Human Milk Purchased Via the Internet. Pediatrics 2013, 132, e1227–e1235. [Google Scholar] [CrossRef]
- Lindemann, P.C.; Foshaugen, I.; Lindemann, R. Characteristics of breast milk and serology of women donating breast milk to a milk bank. Arch. Dis. Child.-Fetal Neonatal Ed. 2004, 89, F440–F441. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bapistella, S.; Hamprecht, K.; Thomas, W.; Speer, C.P.; Dietz, K.; Maschmann, J.; Poets, C.F.; Goelz, R. Short-term Pasteurization of Breast Milk to Prevent Postnatal Cytomegalovirus Transmission in Very Preterm Infants. Clin. Infect. Dis. 2018, 69, 438–444. [Google Scholar] [CrossRef]
- Gribble, K.D.; Hausman, B.L. Milk Sharing and Formula Feeding: Infant Feeding Risks in Comparative Perspective? Austr. Med. J. 2012, 5, 275–283. [Google Scholar] [CrossRef]
- Schanler, R.J.; Fraley, J.K.; Lau, C.; Hurst, N.M.; Horvath, L.; Rossmann, S.N. Breastmilk cultures and infection in extremely premature infants. J. Perinatol. 2011, 31, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Grøvslien, A.H. Personal Communication; Updated Guidelines for Human Milk Banks in Norway; Oslo University Hospital: Oslo, Norway, 2020. [Google Scholar]
- Robertson, R.C.; Manges, A.R.; Finlay, B.B.; Prendergast, A. The Human Microbiome and Child Growth—First 1000 Days and Beyond. Trends Microbiol. 2018, 27, 131–147. [Google Scholar] [CrossRef]
- North, D.W. Uncertainties, precaution, and science: Focus on the state of knowledge and how it may change. Risk Anal. 2011, 31, 1526–1529. [Google Scholar] [CrossRef] [PubMed]
- Gayà, A.; Rittà, M.; Lembo, D.; Tonetto, P.; Cresi, F.; Sottemano, S.; Bertino, E.; Moro, G.E.; Calvo, J.; Donalisio, M. Analysis of Thermal Sensitivity of Human Cytomegalovirus Assayed in the Conventional Conditions of a Human Milk Bank. Front. Pediatr. 2021, 9, 640638. [Google Scholar] [CrossRef] [PubMed]
- Henrick, B.M.; Rodriguez, L.; Lakshmikanth, T.; Pou, C.; Henckel, E.; Arzoomand, A.; Olin, A.; Wang, J.; Mikes, J.; Tan, Z.; et al. Bifidobacteria-mediated immune system imprinting early in life. Cell 2021, 184, 3884–3898.e11. [Google Scholar] [CrossRef]
- Hu, Y.; Chen, Y.; Liu, S.; Jiang, F.; Wu, M.; Yan, C.; Tan, J.; Yu, G.; Hu, Y.; Yin, Y.; et al. Breastfeeding duration modified the effects of neonatal and familial risk factors on childhood asthma and allergy: A population-based study. Respir. Res. 2021, 22, 41. [Google Scholar] [CrossRef]
- Steele, C. Best Practices for Handling and Administration of Expressed Human Milk and Donor Human Milk for Hospitalized Preterm Infants. Front. Nutr. 2018, 5, 76. [Google Scholar] [CrossRef]
- Fernández, L.; Ruiz, L.; Jara, J.; Orgaz, B.; Rodríguez, J. Strategies for the Preservation, Restoration and Modulation of the Human Milk Microbiota. Implications for Human Milk Banks and Neonatal Intensive Care Units. Front. Microbiol. 2018, 9, 2676. [Google Scholar] [CrossRef]
- Dietert, R.R.; Silbergeld, E.K. Biomarkers for the 21st Century: Listening to the Microbiome. Toxicol. Sci. 2015, 144, 208–216. [Google Scholar] [CrossRef]
- Dietert, R.R. The Human Superorganism: How the Microbiome is Revolutionizing the Pursuit of a Healthy Life; Dutton: New York, NY, USA, 2016. [Google Scholar]
- Blaser, M.J. The Microbiome Revolution. J. Clin. Investig. 2014, 124, 4162–4165. [Google Scholar] [CrossRef]
- Kuhn, T.S. The Structure of Scientific Revolutions; Fiftieth Anniversary; University of Chicago Press: Chicago, IL, USA, 2012. [Google Scholar]
- Coleman, M.; Elkins, C.; Gutting, B.; Mongodin, E.; Solano-Aguilar, G.; Walls, I. Microbiota and Dose Response: Evolving Paradigm of Health Triangle. Risk Anal. 2018, 38, 2013–2028. [Google Scholar] [CrossRef]
- O’Connor, G.T.; Lynch, S.V.; Bloomberg, G.R.; Kattan, M.; Wood, R.A.; Gergen, P.J.; Jaffee, K.F.; Calatroni, A.; Bacharier, L.B.; Beigelman, A.; et al. Early-life home environment and risk of asthma among inner-city children. J. Allergy Clin. Immunol. 2017, 141, 1468–1475. [Google Scholar] [CrossRef] [PubMed]
- Parajuli, A.; Grönroos, M.; Siter, N.; Puhakka, R.; Vari, H.K.; Roslund, M.I.; Jumpponen, A.; Nurminen, N.; Laitinen, O.; Hyoty, H.; et al. Urbanization Reduces Transfer of Diverse Environmental Microbiota Indoors. Front. Microbiol. 2018, 9, 84. [Google Scholar] [CrossRef] [PubMed]
- Haahtela, T. A biodiversity hypothesis. Allergy 2019, 74, 1445–1456. [Google Scholar] [CrossRef] [PubMed]
- Dietert, R.R.; Dietert, J.M. Twentieth Century Dogmas Prevent Sustainable Healthcare. Am. J. Biomed. Sci. Res. 2021, 13, 409–417. [Google Scholar]
- Naito, Y.; Kashiwagi, K.; Takagi, T.; Andoh, A.; Inoue, R. Intestinal Dysbiosis Secondary to Proton-Pump Inhibitor Use. Digestion 2018, 97, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Collineau, L.; Boerlin, P.; Carson, C.A.; Chapman, B.; Fazil, A.; Hetman, B.; McEwen, S.A.; Parmley, E.J.; Reid-Smith, R.J.; Taboada, E.; et al. Integrating Whole-Genome Sequencing Data into Quantitative Risk Assessment of Foodborne Antimicrobial Resistance: A Review of Opportunities and Challenges. Front. Microbiol. 2019, 10, 1107. [Google Scholar] [CrossRef]
- EFSA Panel on Biological Hazards (EFSA BIOHAZ Panel); Koutsoumanis, K.; Allende, A.; Alvarez-Ordóñez, A.; Bolton, D.; Bover-Cid, S.; Lindqvist, R. Whole Genome Sequencing and Metagenomics for Outbreak Investigation, Source Attribution and Risk Assessment of Food-Borne Microorganisms. EFSA J. 2019, 17, 05898. [Google Scholar]
- Toscano, M.; De Grandi, R.; Peroni, D.G.; Grossi, E.; Facchin, V.; Comberiati, P.; Drago, L. Impact of delivery mode on the colostrum microbiota composition. BMC Microbiol. 2017, 17, 205. [Google Scholar] [CrossRef]
- Ruiz, L.; García-Carral, C.; Rodríguez, J. Unfolding the Human Milk Microbiome Landscape in the Omics Era. Front. Microbiol. 2019, 10, 1378. [Google Scholar] [CrossRef]
- Wang, S.; Ryan, C.A.; Boyaval, P.; Dempsey, E.M.; Ross, R.; Stanton, C. Maternal Vertical Transmission Affecting Early-life Microbiota Development. Trends Microbiol. 2019, 28, 28–45. [Google Scholar] [CrossRef] [PubMed]
- Van Daele, E.; Knol, J.; Belzer, C. Microbial transmission from mother to child: Improving infant intestinal microbiota development by identifying the obstacles. Crit. Rev. Microbiol. 2019, 45, 613–648. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Kalyan, S.; Steck, N.; Turner, L.; Harr, B.; Künzel, S.; Vallier, M.; Häsler, R.; Franke, A.; Oberg, H.-H.; et al. Analysis of intestinal microbiota in hybrid house mice reveals evolutionary divergence in a vertebrate hologenome. Nat. Commun. 2015, 6, 6440. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Covington, A.; Pamer, E.G. The intestinal microbiota: Antibiotics, colonization resistance, and enteric pathogens. Immunol. Rev. 2017, 279, 90–105. [Google Scholar] [CrossRef]
- Stein, R.R.; Bucci, V.; Toussaint, N.; Buffie, C.G.; Rätsch, G.; Pamer, E.G.; Sander, C.; Xavier, J.B. Ecological Modeling from Time-Series Inference: Insight into Dynamics and Stability of Intestinal Microbiota. PLoS Comput. Biol. 2013, 9, e1003388. [Google Scholar] [CrossRef]
- Buffie, C.G.; Bucci, V.; Stein, R.R.; McKenney, P.T.; Ling, L.; Gobourne, A.; No, D.; Liu, H.; Kinnebrew, M.; Viale, A.; et al. Precision microbiome reconstitution restores bile acid mediated resistance to Clostridium difficile. Nat. Cell Biol. 2014, 517, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Becattini, S.; Pamer, E.G. Multifaceted Defense against Listeria monocytogenes in the Gastro-Intestinal Lumen. Pathogens 2017, 7, 1. [Google Scholar] [CrossRef]
- Baumgartner, M.; Cardozo, K.P.; Hall, A. Microbial community composition interacts with local abiotic conditions to drive colonization resistance in human gut microbiome samples. Proc. R. Soc. B 2021, 288, 20203106. [Google Scholar] [CrossRef]
- National Research Council (NRC). Understanding Risk: Informing Decisions in a Democratic Society; National Academies Press: Washington, DC, USA, 1996. [Google Scholar]
- North, D.W. Risk Analysis, Decision analysis, causal analysis, and economics: A personal perspective from more than 40 years experience. Risk Anal. 2020, 40, 2178–2190. [Google Scholar] [CrossRef]
- North, D.W.; Cox, L.A.; Popken, D.A.; Sun, R.X. Mega-Review: Causality Books. Causal Analytics for Applied Risk Analysis. Risk Anal. Int. J. 2019, 39, 1647–1654. [Google Scholar]
- Pearl, J.; Mackenzie, D. The Book of Why: The New Science of Cause and Effect; Basic Books: New York, NY, USA, 2018. [Google Scholar]
- North, D.W. Commentary on “Should Health Risks of Air Pollution Be Studied Scientifically?” by Louis Anthony Cox, Jr. Glob. Epidemiol. 2020, 2, 100021. [Google Scholar]
| Predominant Bacteria Isolated from Breastmilk | Number of Isolates | Percent of Isolates |
|---|---|---|
| Staphylococcus epidermis | 548 | 42.4 |
| Enterococcus faecalis | 152 | 11.8 |
| Acintobacter spp. | 127 | 9.8 |
| Streptococcus spp. (coagulase-negative) | 89 | 6.9 |
| Stenotrophomonas maltophila | 79 | 6.1 |
| Enterobacter spp. | 69 | 5.3 |
| Staphylococcus aureus | 65 | 5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coleman, M.E.; North, D.W.; Dietert, R.R.; Stephenson, M.M. Examining Evidence of Benefits and Risks for Pasteurizing Donor Breastmilk. Appl. Microbiol. 2021, 1, 408-425. https://doi.org/10.3390/applmicrobiol1030027
Coleman ME, North DW, Dietert RR, Stephenson MM. Examining Evidence of Benefits and Risks for Pasteurizing Donor Breastmilk. Applied Microbiology. 2021; 1(3):408-425. https://doi.org/10.3390/applmicrobiol1030027
Chicago/Turabian StyleColeman, Margaret E., D. Warner North, Rodney R. Dietert, and Michele M. Stephenson. 2021. "Examining Evidence of Benefits and Risks for Pasteurizing Donor Breastmilk" Applied Microbiology 1, no. 3: 408-425. https://doi.org/10.3390/applmicrobiol1030027
APA StyleColeman, M. E., North, D. W., Dietert, R. R., & Stephenson, M. M. (2021). Examining Evidence of Benefits and Risks for Pasteurizing Donor Breastmilk. Applied Microbiology, 1(3), 408-425. https://doi.org/10.3390/applmicrobiol1030027







