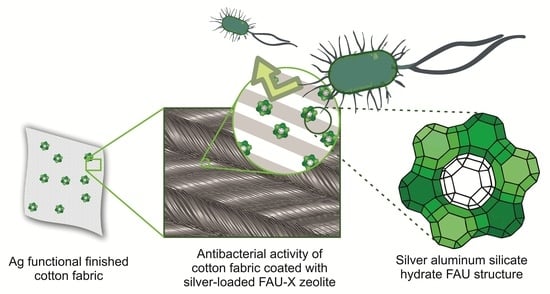
Antimicrobial Textile Finishing Based on Silver Nanostructures
Abstract
1. Introduction
2. Materials and Methods
2.1. Fabric Pretreatment
2.2. Silver Finishing Process
2.3. Finishing Application on Textile
2.4. Characterization
2.5. Bacterial Study on Textiles
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rajendran, R. Superbug Infection. J. Drug Metab. Toxicol. 2018, 9, 238. [Google Scholar] [CrossRef]
- Naghavi, M.; Vollset, S.E.; Ikuta, K.S.; Swetschinski, L.R.; Gray, A.P.; Wool, E.E.; Aguilar, G.R.; Mestrovic, T.; Smith, G.; Han, C.; et al. Global burden of bacterial antimicrobial resistance 1990–2021: A systematic analysis with forecasts to 2050. Lancet 2024, 404, 1199–1226. [Google Scholar] [CrossRef]
- Dube, E.; Okuthe, G.E. Silver Nanoparticle-Based Antimicrobial Coatings: Sustainable Strategies for Microbial Contamination Control. Microbiol. Res. 2025, 16, 110. [Google Scholar] [CrossRef]
- Michels, H.T.; Keevil, C.W.; Salgado, C.D.; Schmidt, M.G. From Laboratory Research to a Clinical Trial. HERD Health Environ. Res. Des. J. 2015, 9, 64–79. [Google Scholar] [CrossRef]
- Widyastuti, E.; Chiu, C.-T.; Hsu, J.-L.; Lee, Y.C. Photocatalytic Antimicrobial and Photostability Studies of TiO2/ZnO Thin Films. Arab. J. Chem. 2023, 16, 105010. [Google Scholar] [CrossRef]
- Ong, W.T.J.; Nyam, K.L. Evaluation of Silver Nanoparticles in Cosmeceutical and Potential Biosafety Complications. Saudi J. Biol. Sci. 2022, 29, 2085–2094. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.M.V.; Mohammad, N.; Banerjee, S.; Khanna, P.K. Synthesis and Food Packaging Application of Silver Nanoparticles: A Review. Hybrid Adv. 2024, 6, 100230. [Google Scholar] [CrossRef]
- Mondal, M.S.; Paul, A.; Rhaman, M. Recycling of Silver Nanoparticles from Electronic Waste via Green Synthesis and Application of AgNPs–Chitosan Based Nanocomposite on Textile Material. Sci. Rep. 2023, 13, 13798. [Google Scholar] [CrossRef]
- Abdelhamid, S.A.; Mohamed, S.S.; Abdelghaffar, F. Ecofriendly Silver Nanoparticles Biosynthesis from Penicillium Commune NRC 2016-3 as Antimicrobial Agents for Textile Materials. Sci. Rep. 2025, 15, 25522. [Google Scholar] [CrossRef]
- Rashid, S.; Ali, M.; Islam, S.; Iqbal, M.O.; Al-Rawi, M.B.A.; Naseem, M. Enhancing the Antibacterial Properties of Silver-Particles-Coated Cotton Bandages Followed by Natural Extracted Dye. J. Ind. Text. 2025, 55, 1320571. [Google Scholar] [CrossRef]
- Khan, M.; Saeed, M.A.; Ullah, S.; Repon, R.; Pranta, A.D.; Yunusov, N.; Hossain, M. Development of Self-Cleaning and Antibacterial Properties on Cotton Fabric Using Silver Nanoparticles and PFOTS. SPE Polym. 2024, 5, 568–575. [Google Scholar] [CrossRef]
- dos Santos, L.M.G.; Medeiros, R.J.; Maciel-Magalhães, M.; Guedes, N.C.; Brito, T.M.; de Souza, G.F.; Oliveira, M.L.; Pereira, R.A.; Neto, S.A.; Jacob, S.C.; et al. Unravelling the Effects of Silver Nanoparticles on Textiles: A Comprehensive Toxicological and Quantitative Analysis. Health Nanotechnol. 2025, 1, 6. [Google Scholar] [CrossRef]
- Hossain, M.T.; Shahid, M.A.; Limon, M.G.M.; Hossain, I.; Mahmud, N. Techniques, Applications, and Challenges in Textiles for a Sustainable Future. J. Open Innov. Technol. Mark. Complex. 2024, 10, 100230. [Google Scholar] [CrossRef]
- Pavlova, E.L.; Nenova, E.P.; Yocheva, L.D.; Ivanova, I.A.; Georgiev, P.A. Antimicrobial and Oxidative Activities of Different Levels of Silver-Exchanged Zeolites X and ZSM-5 and Their Ecotoxicity. Pharmaceuticals 2024, 17, 1586. [Google Scholar] [CrossRef]
- Verboekend, D.; Nuttens, N.; Locus, R.; Van Aelst, J.; Verlome, P.; Groen, J.C.; Perez-Ramirez, J.; Sels, B.F. Synthesis, Characterisation, and Catalytic Evaluation of Hierarchical Faujasite Zeolites: Milestones, Challenges, and Future Directions. Chem. Soc. Rev. 2016, 45, 3331–3352. [Google Scholar] [CrossRef]
- Mauer, V.; Bläker, C.; Pasel, C.; Bathen, D. Energetic Characterization of Faujasite Zeolites Using a Sensor Gas Calorimeter. Catalysts 2021, 11, 98. [Google Scholar] [CrossRef]
- Baerlocher, C.; Brouwer, D.; Marler, B.; McCusker, L.B. Database of Zeolite Structures. Available online: https://www.iza-structure.org/databases/ (accessed on 11 November 2025).
- Cruz-Leal, M.; Goiz, O.; Niño, T.I.M.; Coutino-Gonzalez, E.; Albarado-Ibañez, A.; Pérez-Sánchez, G.F.; Alcantara C, J.A. Silver Cluster Supported in Zeolite as Antimicrobial Agent to Textiles. MRS Adv. 2023, 9, 168–171. [Google Scholar] [CrossRef]
- Zhou, C.-E.; Kan, C.; Yuen, C.M.; Matinlinna, J.P.; Tsoi, J.K.; Zhang, Q. Plasma Treatment Applied in the Pad-Dry-Cure Process for Making Rechargeable Antimicrobial Cotton Fabric That Inhibits S. aureus. Text. Res. J. 2016, 86, 2202–2215. [Google Scholar] [CrossRef]
- Venegas, B.; Tello-Hernández, M.A.; Cepeda-Cornejo, V.; Molina-Romero, D. Calidad Microbiológica: Detección de Aeromonas sp y Pseudomonas sp en Garrafones Provenientes de Pequeñas Plantas Purificadoras de Agua. CienciaUAT 2022, 17, 146–164. [Google Scholar] [CrossRef]
- Sousa, P.B.F.; Bieseki, L.; Pergher, S.B.C. Seed-Assisted Crystallization in the Hydrothermal Synthesis of FAU Zeolite from Acid-Treated Residue Glass Powder. Materials 2025, 18, 1393. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.; Huang, R.; Zhang, H.; Ye, S.; Wang, D. Synthesis of Silver Nanocluster-Loaded FAU Zeolites and the Application in Light Emitting Diode. Chemistry 2025, 7, 90. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, H.; He, F.; Liu, Q.; Xu, N.; Fan, L.; Wang, C.; Zhang, L.; Zhou, R. High-Performance FAU Zeolite Membranes Derived from Nano-Seeds for Gas Separation. Membranes 2023, 13, 858. [Google Scholar] [CrossRef]
- Frising, T.; Leflaive, P. Extraframework Cation Distributions in X and Y Faujasite Zeolites: A Review. Microporous Mesoporous Mater. 2008, 114, 27–63. [Google Scholar] [CrossRef]
- Cerrillo, J.L.; Palomares, A.E.; Rey, F.; Valencia, S.; Palou, L.; Pérez-Gago, M.B. Ag-Zeolites as Fungicidal Material: Control of Citrus Green Mold Caused by Penicillium digitatum. Microporous Mesoporous Mater. 2017, 254, 69–76. [Google Scholar] [CrossRef]
- Romolini, G.; Steele, J.A.; Hofkens, J.; Roeffaers, M.B.J.; Coutino-Gonzalez, E. Tunable Luminescence from Stable Silver Nanoclusters Confined in Microporous Zeolites. Adv. Opt. Mater. 2021, 9, 210094. [Google Scholar] [CrossRef]
- Gicheva, G.; Panayotova, M.; Gemishev, O.; Kulinich, S.A.; Mintcheva, N. Silver Nanoparticles@Zeolite Composites: Preparation, Characterization and Antibacterial Properties. Materials 2025, 18, 3964. [Google Scholar] [CrossRef]
- Wang, S.; Egan, J.; Salmon, S. Preparation and Characterization of Cotton Fiber Fragments from Model Textile Waste via Mechanical Milling and Enzyme Degradation. Cellulose 2023, 30, 10879–10904. [Google Scholar] [CrossRef]
- Schindler, W.D.; Hauser, P.J. Chemical Finishing Processes. In Chemical Finishing of Textiles; Elsevier: Cambridge, UK, 2004; pp. 7–28. [Google Scholar] [CrossRef]
- Gao, Y.; Cranston, R. Recent Advances in Antimicrobial Treatments of Textiles. Text. Res. J. 2008, 78, 60–72. [Google Scholar] [CrossRef]
- Pojnar, K.; Pilch-Pitera, B.; Ataei, S.; Gazdowicz, P.; Mossety-Leszczak, B.; Grabowska, B.; Bobrowski, A. Self-Healing Thermal-Reversible Low-Temperature Polyurethane Powder Coating Based on Diels–Alder Reaction. Materials 2024, 17, 3555. [Google Scholar] [CrossRef]
- Patti, A. Green Advances in Wet Finishing Methods and Nanoparticles for Daily Textiles. Macromol. Rapid Commun. 2025, 46, 2400636. [Google Scholar] [CrossRef]
- Tian, Y.; Huang, X.; Cheng, Y.; Niu, Y.; Ma, J.; Zhao, Y.; Kou, X.; Ke, Q. Applications of Adhesives in Textiles: A Review. Eur. Polym. J. 2022, 167, 111089. [Google Scholar] [CrossRef]
- Ghetas, H.A.; Abdel-Razek, N.; Shakweer, M.S.; Abotaleb, M.M.; Paray, B.A.; Ali, S.; Eldessouki, E.A.; Dawood, M.A.; Khalil, R.H. Antimicrobial Activity of Chemically and Biologically Synthesized Silver Nanoparticles against Some Fish Pathogens. Saudi J. Biol. Sci. 2022, 29, 1298–1305. [Google Scholar] [CrossRef]
- Samani, P.A.; Ghavam, M. Synthesis of Silver Nanoparticles from Pure and Combined Extracts of Satureja bachtiarica Bung. and Satureja hortensis L. effective on Some Microbial Strains Causing Digestive Diseases. Discov. Nano 2025, 20, 90. [Google Scholar] [CrossRef] [PubMed]
- Mikhailova, E.O. Silver Nanoparticles: Mechanism of Action and Probable Bio-Application. J. Funct. Biomater. 2020, 11, 84. [Google Scholar] [CrossRef]
- More, P.R.; Pandit, S.; De Filippis, A.; Franci, G.; Mijakovic, I.; Galdiero, M. Silver Nanoparticles: Bactericidal and Mechanistic Approach against Drug-Resistant Pathogens. Microorganisms 2023, 11, 369. [Google Scholar] [CrossRef]
- Alzahrani, S.O.; Al-Ghamdi, S.A.; Alsahag, M.; Alatawi, K.; Alaysuy, O.; Al-Ahmed, Z.A.; El-Metwaly, N.M. Nanosilver-Doped Carbon Quantum Dots for Industrialization of Efficacious Textiles with Superior UV-Resistance and Antimicrobial Performance. Diam. Relat. Mater. 2023, 138, 110249. [Google Scholar] [CrossRef]
- Vaishampayan, A.; Grohmann, E. Antimicrobials Functioning through ROS-Mediated Mechanisms: Current Insights. Microorganisms 2022, 10, 61. [Google Scholar] [CrossRef]
- Mai-Prochnow, A.; Clauson, M.; Hong, J.; Murphy, A.B. Gram-Positive and Gram-Negative Bacteria Differ in Their Sensitivity to Cold Plasma. Sci. Rep. 2016, 6, 38610. [Google Scholar] [CrossRef]
- Dong, X.; Liang, W.; Meziani, M.J.; Sun, Y.-P.; Yang, L. Carbon Dots as Potent Antimicrobial Agents. Theranostics 2020, 10, 671–686. [Google Scholar] [CrossRef]
- Li Petri, G.; Facchiano, S.; Trovato, V.; Vineis, C.; Rosace, G.; Pagliaro, M.; Ciriminna, R. Antibacterial Activity of Textiles Functionalized with SilverSil. ChemNanoMat 2025, 11, e202500132. [Google Scholar] [CrossRef]
- Pádua, A.S.; Rosa, R.M.; Vieira, T.; Henriques, C.; Silva, J.C. Electrospun Poly(N-Vinylpyrrolidone) Membranes with Ag Nanoparticles for Wound Dressings: Production and Characterization. J. Drug Deliv. Sci. Technol. 2024, 101, 106303. [Google Scholar] [CrossRef]
- Delgado-Beleño, Y.; Martinez-Nuñez, C.E.; Cortez-Valadez, M.; Flores-López, N.S.; Flores-Acosta, M. Optical Properties of Silver, Silver Sulfide and Silver Selenide Nanoparticles and Antibacterial Applications. Mater. Res. Bull. 2018, 99, 385–392. [Google Scholar] [CrossRef]
- Taheri, P.; Khajeh-Amiri, A. Antibacterial Cotton Fabrics via Immobilizing Silver Phosphate Nanoparticles onto the Chitosan Nanofiber Coating. Int. J. Biol. Macromol. 2020, 158, 282–289. [Google Scholar] [CrossRef] [PubMed]





| Types of Minerals (wt%) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Sample | C | O | Si | Al | Na | Ag | Ca | Other |
| Pristine cotton | 48.7 | 50.57 | 0.50 | - | - | - | 0.23 | traces |
| FAU-X 10%/cotton | - | 53.63 | 20.13 | 15.36 | 4.36 | 4.61 | 1.46 | traces |
| FAU-X 100%/cotton | - | 54.10 | 16.42 | 12.33 | 3.43 | 10.81 | 1.56 | traces |
| Inhibition Zone (mm) * | |||
|---|---|---|---|
| Gram Negatives | FAU-X | FAU-X 10% | FAU-X 100% |
| Shigella sp. | - | 8 | 10 |
| Pseudomonas sp. M13 | - | 9 | 11 |
| Pseudomonas sp. M14 | 7 | 9 | 10 |
| Pseudomonas putida KT2440 | - | 8 | 11 |
| Escherichia coli | - | - | 11 |
| Gram Positives | |||
| Staphylococcus aureus ATCC 29213 | - | 7 | 11 |
| Streptococcus agalactiae ATCC 12386 | - | 8 | 14 |
| Material | Main Finding | Limitation | Reference |
|---|---|---|---|
| Ag, Ag2S, and Ag2Se nanoparticles | AgNPs showed the strongest effect against E. coli | Low stability in aqueous media | [38] |
| Electrospun poly(N-vinylpyrrolidone) membranes with AgNPs | Gram-positive bacteria revealed a higher susceptibility to silver | Difficult to scale up for industrial use | [37] |
| Ag/ZnO NPs in cotton fibers | Developing antimicrobial cotton fibers | Experiments were limited to two bacterial strains. | [40] |
| Silver-loaded chitosan nanoparticles applied on cotton fabric using a layer-by-layer self-assembly coating method. | Demonstrated effective inhibition against both E. coli and S. aureus. | Antibacterial tests were only performed on two bacterial strains. | [41] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres, J.Á.J.; Cabrera, M.L.C.; Romero, D.M.; Torres Avila, I.P.; Goiz, O.; Cruz-Leal, M. Antimicrobial Textile Finishing Based on Silver Nanostructures. Textiles 2025, 5, 61. https://doi.org/10.3390/textiles5040061
Torres JÁJ, Cabrera MLC, Romero DM, Torres Avila IP, Goiz O, Cruz-Leal M. Antimicrobial Textile Finishing Based on Silver Nanostructures. Textiles. 2025; 5(4):61. https://doi.org/10.3390/textiles5040061
Chicago/Turabian StyleTorres, José Ángel Juárez, María Luisa Castellanos Cabrera, Dalia Molina Romero, Itzel Pamela Torres Avila, Oscar Goiz, and Miriam Cruz-Leal. 2025. "Antimicrobial Textile Finishing Based on Silver Nanostructures" Textiles 5, no. 4: 61. https://doi.org/10.3390/textiles5040061
APA StyleTorres, J. Á. J., Cabrera, M. L. C., Romero, D. M., Torres Avila, I. P., Goiz, O., & Cruz-Leal, M. (2025). Antimicrobial Textile Finishing Based on Silver Nanostructures. Textiles, 5(4), 61. https://doi.org/10.3390/textiles5040061