50/60 Hz Power Grid Noise as a Skin Contact Measure of Textile ECG Electrodes
Abstract
:1. Introduction
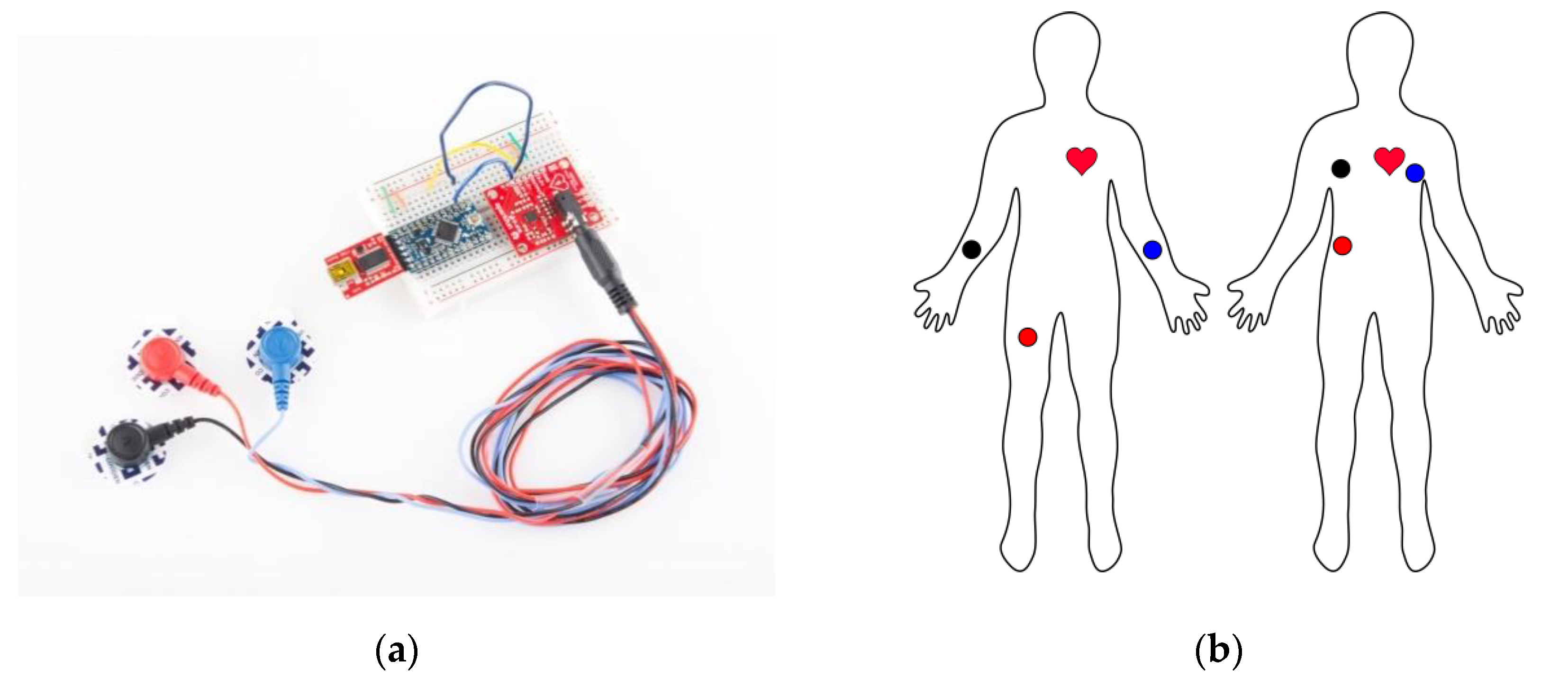
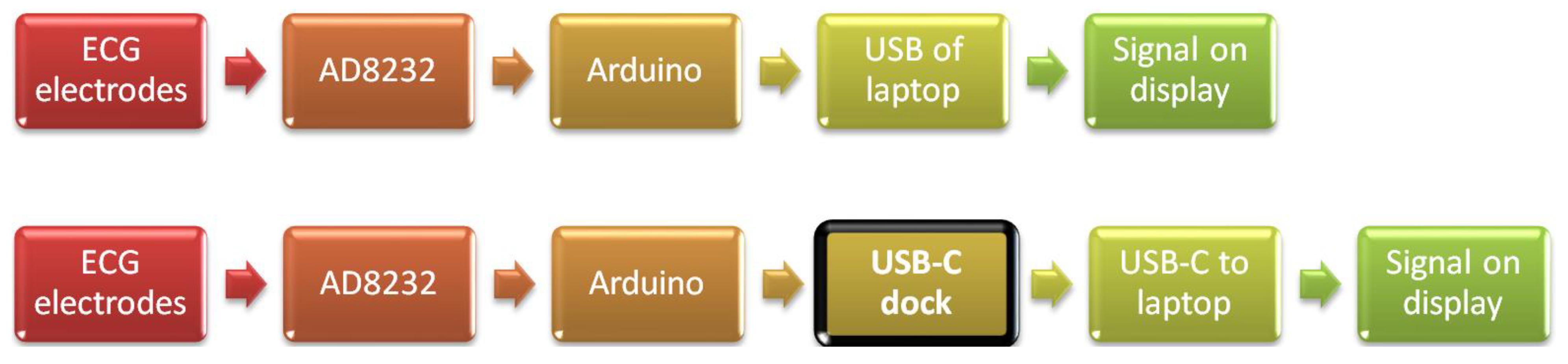
2. Materials and Methods
- -
- Commercial glued gel electrodes;
- -
- -
- -
- -
- Powersil coating on cotton without conductive yarn (Figure 2d);
- -
- Ripstop Silver Fabric (Less EMF, NY, USA; Figure 2e);
- -
- Shieldit Super (Less EMF; Figure 2f);
- -
- Hand-sewn Shieldex yarn on Hansaplast Sensitive Fixation plaster (band-aid, Figure 2g);
- -
- Poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS) double-coated woven fabric (Figure 2h);
- -
- -
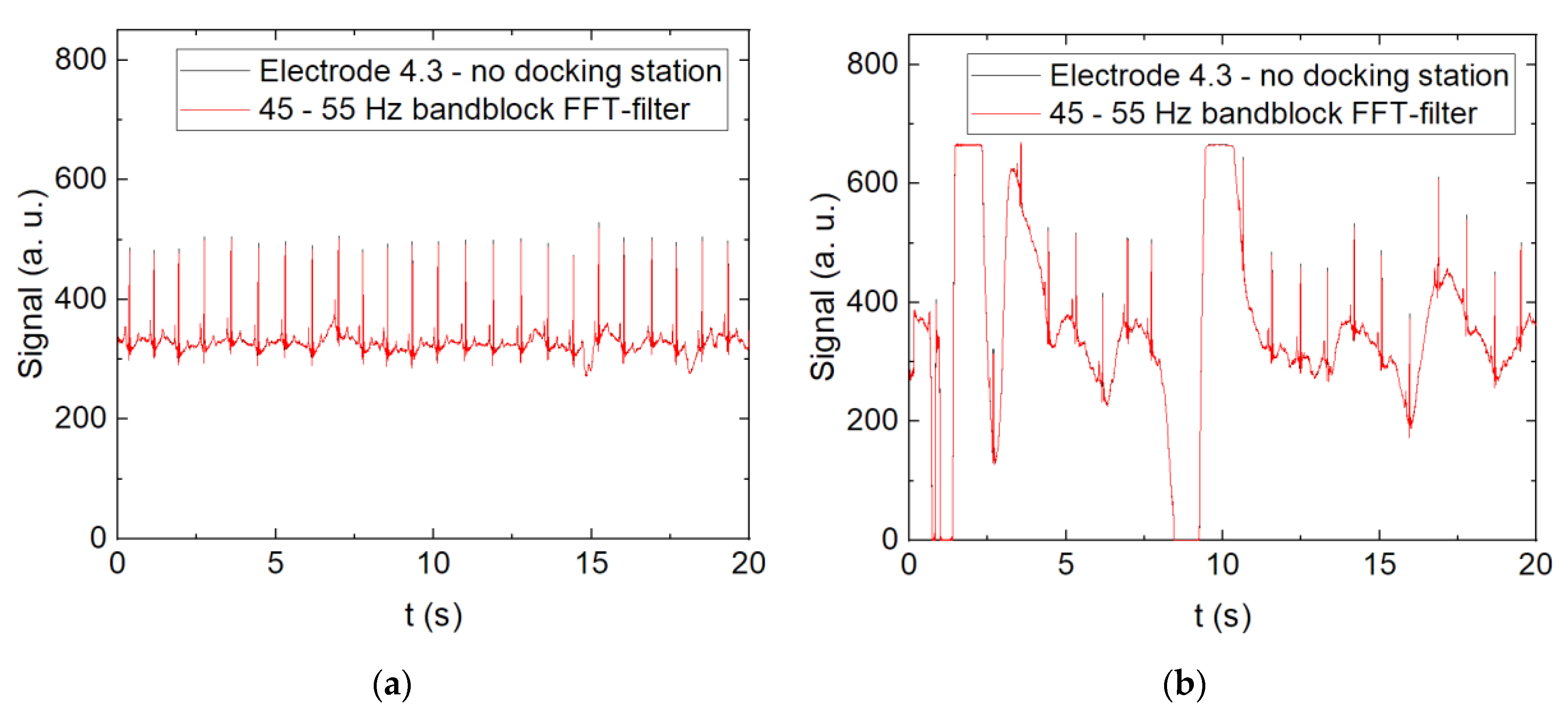
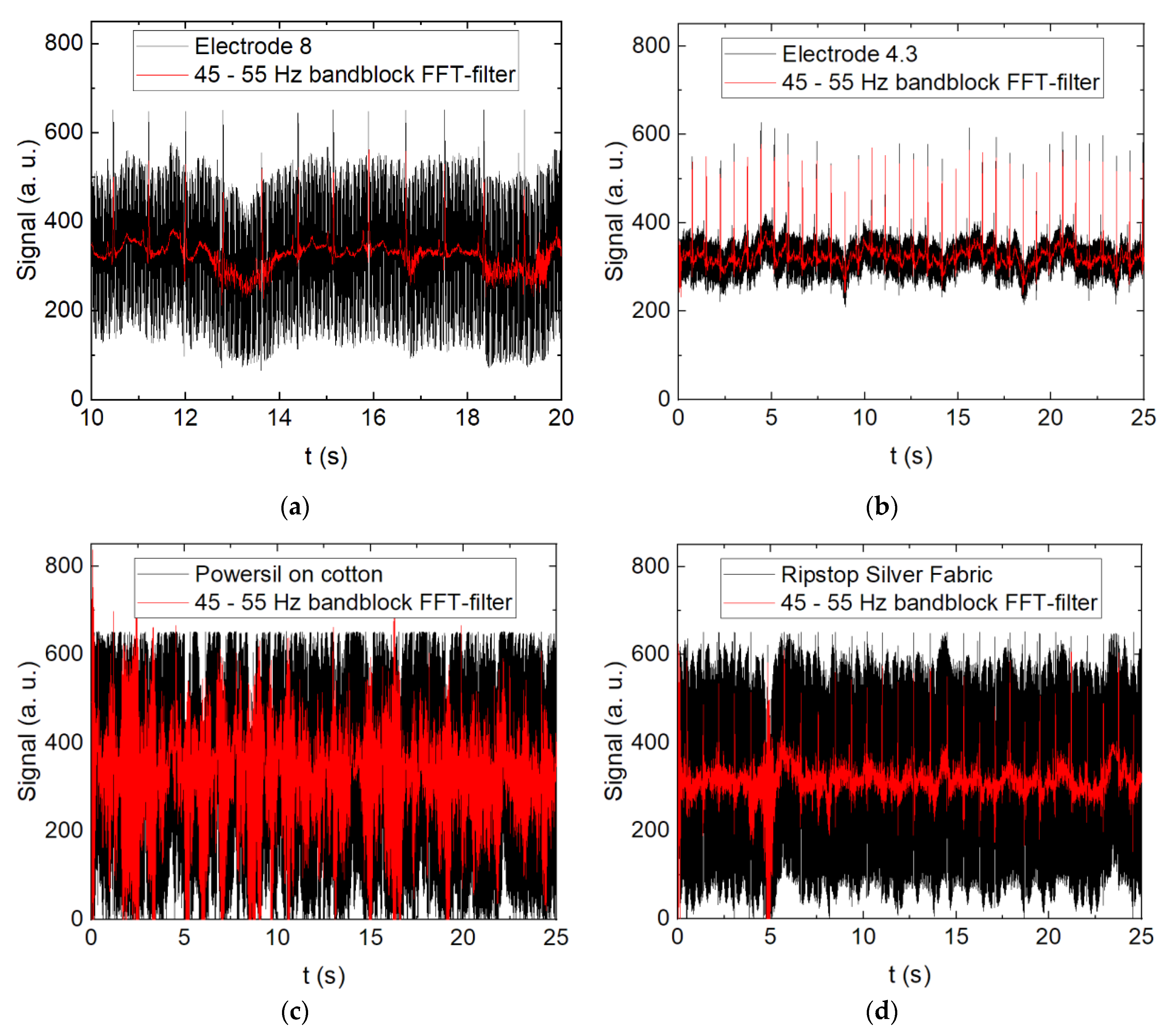
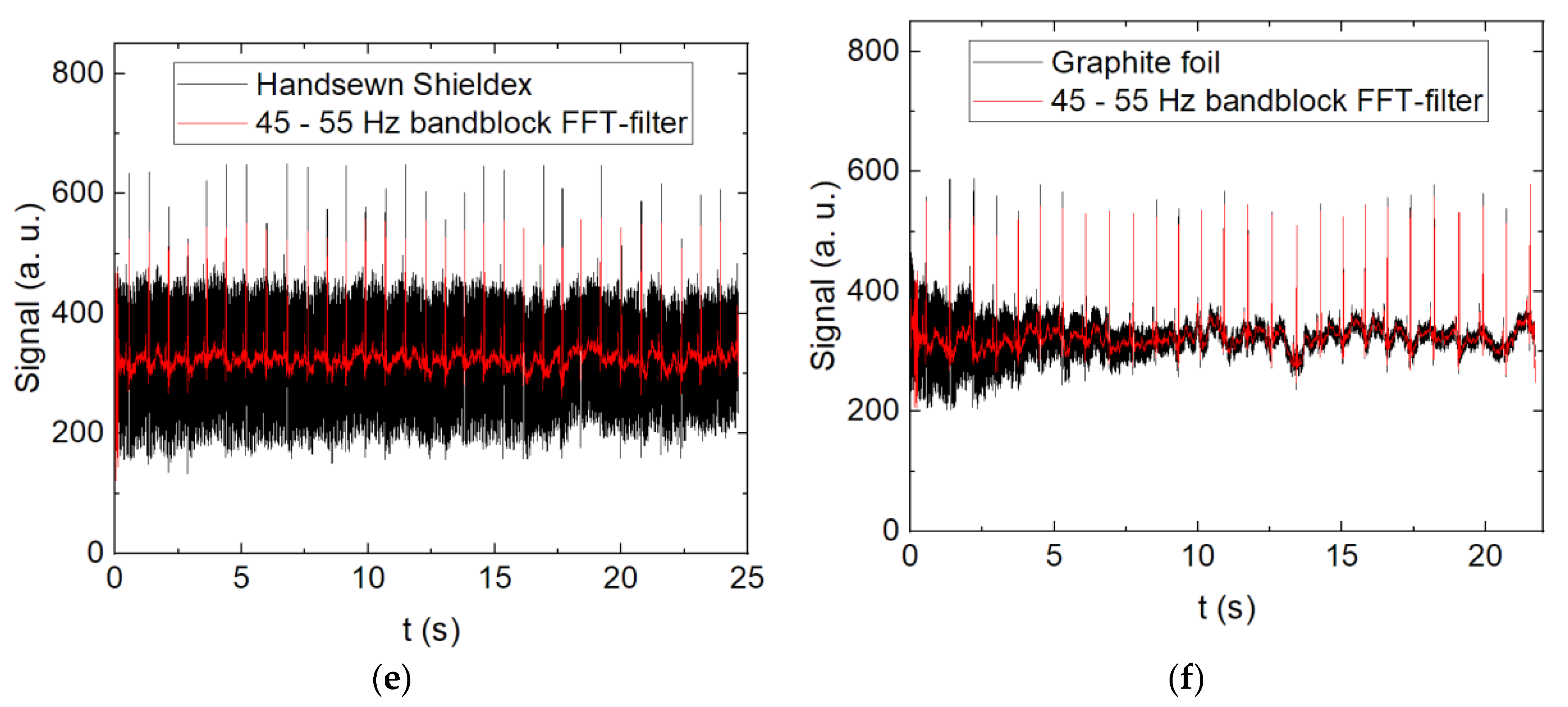
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ahmad, F.B.; Anderson, R.N. The leading causes of death in the US for 2020. JAMA 2021, 325, 1829–1830. [Google Scholar] [CrossRef]
- Ritchie, H.; Roser, M. Causes of Death. Our World in Data 2018. Available online: https://ourworldindata.org/causes-of-death (accessed on 7 January 2022).
- Armstrong, W.F.; Jordan, J.W. Prevalence and magnitude of ST-segment and T wave abnormalities in men during continuous ambulatory electrocardiography. Am. J. Cardiol. 1981, 49, 249–251. [Google Scholar]
- Trummer, S.; Ehrmann, A.; Büsgen, A. Development of underwear with integrated 12 channel ECG for men and women. AUTEX Res. J. 2017, 17, 344–349. [Google Scholar] [CrossRef]
- Coosemans, J.; Hermans, B.; Puers, R. Integrating wireless ECG monitoring in textiles. Sens. Actuators A Phys. 2006, 130–131, 48–53. [Google Scholar] [CrossRef]
- Aumann, S.; Trummer, S.; Brücken, A.; Ehrmann, A.; Büsgen, A. Conceptual design of a sensory shirt for fire-fighters. Text. Res. J. 2014, 84, 1661–1665. [Google Scholar] [CrossRef]
- Lee, E.; Kim, H.; Liu, H.; Cho, G. Exploration of AgNW/PU nanoweb as ECG textile electrodes and comparison with Ag/AgCl electrodes. Fibers Polym. 2017, 18, 1749–1753. [Google Scholar] [CrossRef]
- Matsouka, D.; Vassiliadis, S.; Tao, X.; Koncar, V.; Bahadir, S.K.; Kalaoglu, F.; Jevsnik, S. Electrical connection issues on wearable electronics. IOP Conf. Ser. Mater. Sci. Eng. 2018, 459, 012017. [Google Scholar] [CrossRef]
- An, X.; Stylios, G.K. A hybrid textile electrode for electrocardiogram (ECG) measurement and motion tracking. Materials 2018, 11, 1887. [Google Scholar] [CrossRef] [Green Version]
- Ehrmann, G.; Ehrmann, A. Electronic textiles. Encyclopedia 2021, 1, 115–130. [Google Scholar] [CrossRef]
- Pani, D.; Achilli, A.; Bonfiglio, A. Survey on Textile Electrode Technologies for Electrocardiographic (ECG) Monitoring, from Metal Wires to Polymers. Adv. Mater. Technol. 2018, 3, 1800008. [Google Scholar] [CrossRef]
- An, X.; Tangsirinaruenart, O.; Stylios, G.K. Investigating the performance of dry textile electrodes for wearable end-uses. J. Text. Inst. 2019, 110, 151–158. [Google Scholar] [CrossRef]
- Takeshita, T.; Yoshida, M.; Takei, Y.; Ouchi, A.; Hinoki, A.; Uchida, H.; Kobayashi, T. Relationship between Contact Pressure and Motion Artifacts in ECG Measurement with Electrostatic Flocked Electrodes Fabricated on Textile. Sci. Rep. 2019, 9, 5897. [Google Scholar] [CrossRef] [PubMed]
- Nigusse, A.B.; Malengier, B.; Mengistie, D.A.; Tseghai, G.B.; van Langenhove, L. Development of Washable Silver Printed Textile Electrodes for Long-Term ECG Monitoring. Sensors 2020, 20, 6233. [Google Scholar] [CrossRef]
- Euler, L.; Guo, L.; Persson, N.-K. Textile Electrodes: Influence of Knitting Construction and Pressure on the Contact Impedance. Sensors 2021, 21, 1578. [Google Scholar] [CrossRef]
- Acar, G.; Ozturk, O.; Golparvar, A.J.; Elboshra, T.A.; Böhringer, K.; Yapici, M.K. Wearable and Flexible Textile Electrodes for Biopotential Signal Monitoring: A review. Electronics 2019, 8, 479. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Zhong, Y.Q. Development of woven textile electrodes with enhanced moisture-wicking properties. J. Text. Inst. 2021, 112, 983–995. [Google Scholar] [CrossRef]
- Terada, T.; Toyoura, M.; Sato, T.; Mao, X.Y. Noise-Reducing Fabric Electrode for ECG Measurement. Sensors 2021, 21, 4305. [Google Scholar] [CrossRef]
- Uz Zaman, S.; Tao, X.Y.; Cochrane, C.; Koncar, V. Smart E-Textile Systems: A Review for Healthcare Applications. Electronics 2022, 11, 99. [Google Scholar] [CrossRef]
- Schäl, P.; Juhász Junger, I.; Grimmelsmann, N.; Ehrmann, A. Development of graphite-based conductive textile coatings. J. Coat. Technol. Res. 2018, 15, 875–883. [Google Scholar] [CrossRef]
- Ankhili, A.; Tao, X.Y.; Cochrane, C.; Koncar, V.; Coulon, D.; Tarlet, J.-M. Ambulatory Evaluation of ECG Signals Obtained Using Washable Textile-Based Electrodes Made with Chemically Modified PEDOT:PSS. Sensors 2019, 19, 416. [Google Scholar] [CrossRef] [Green Version]
- Shathi, M.A.; Chen, M.Z.; Khoso, N.A.; Rahman, M.T.; Thattacharjee, B. Graphene coated textile based highly flexible and washable sports bra for human health monitoring. Mater. Des. 2020, 193, 108792. [Google Scholar] [CrossRef]
- Tseghai, G.B.; Melengier, B.; Fante, K.A.; Nigusse, A.B.; Etana, B.B.; van Langenhove, L. PEDOT:PSS/PDMS-Coated Cotton Fabric for ECG Electrode. In Proceedings of the 2020 IEEE International Conference on Flexible and Printable Sensors and Systems (FLEPS), Manchester, UK, 16–19 August 2020; pp. 1–4. [Google Scholar]
- Xu, X.W.; Liang, Y.; He, P.; Yang, J.L. Adaptive Motion Artifact Reduction Based on Empirical Wavelet Transform and Wavelet Thresholding for the Non-Contact ECG Monitoring Systems. Sensors 2019, 19, 2916. [Google Scholar] [CrossRef] [Green Version]
- Xiong, F.; Chen, D.Y.; Huang, M. A Wavelet Adaptive Cancellation Algorithm Based on Multi-Inertial Sensors for the Reduction of Motion Artifacts in Ambulatory ECGs. Sensors 2020, 20, 970. [Google Scholar] [CrossRef] [Green Version]
- An, X.; Stylios, G.K. Comparison of Motion Artefact Reduction Methods and the Implementation of Adaptive Motion Artefact Reduction in Wearable Electrocardiogram Monitoring. Sensors 2020, 20, 1468. [Google Scholar] [CrossRef] [Green Version]
- Xiong, F.; Chen, D.Y.; Chen, Z.H.; Dai, S.M. Cancellation of motion artifacts in ambulatory ECG signals using TD-LMS adaptive filtering techniques. J. Vis. Commun. Image Represent. 2019, 58, 606–618. [Google Scholar] [CrossRef]
- Huang, M.; Chen, D.Y.; Xiong, F. An Effective Adaptive Filter to Reduce Motion Artifacts from ECG Signals Using Accelerometer. In Proceedings of the 2019 9th International Conference on Biomedical Engineering and Technology, Tokyo, Japan, 28–30 March 2019; pp. 83–88. [Google Scholar]
- Suchetha, M.; Kumaravel, N.; Jagannath, M.; Jaganathan, S.K. A comparative analysis of EMD based filtering methods for 50 Hz noise cancellation in ECG signal. Inform. Med. Unlocked 2017, 8, 54–59. [Google Scholar] [CrossRef]
- Babusiak, B.; Borik, S.; Balogova, L. Textile electrodes in capacitive signal sensing applications. Measurement 2018, 114, 69–77. [Google Scholar] [CrossRef]
- Fink, P.L.; Sayem, A.S.M.; Teay, S.H.; Ahmad, F.; Shahariar, H.; Albarbar, A. Development and wearer trial of ECG-garment with textile-based dry electrodes. Sens. Actuators A Phys. 2021, 328, 112784. [Google Scholar] [CrossRef]
- Nikolova, E.; Ganev, B.; Gieva, E. Wearable Intelligent Textile Suits for Telemetry Monitoring in Pediatrics. In Proceedings of the 2021 XXX International Scientific Conference Electronics (ET), Sozopol, Bulgaria, 15–17 September 2021; pp. 1–6. [Google Scholar]
- Ehrmann, G.; Ehrmann, A. Suitability of common single circuit boards for sensing and actuating in smart textiles. CDATP 2020, 1, 170–179. [Google Scholar] [CrossRef]
- Ehrmann, G.; Blachowicz, T.; Homburg, S.V.; Ehrmann, A. Measuring biosignals with single circuit boards. Bioengineering 2020, 9, 84. [Google Scholar] [CrossRef]
- Searson, P.C.; Dawson, J.L. Analysis of electrochemical noise generated by corroding electrodes under open-circuit conditions. J. Electrochem. Soc. 1988, 135, 1908. [Google Scholar] [CrossRef]
- Rothenhagen, K.; Fuchs, F.W. Performance of Diagnosis Methods for IGBT Open Circuit Faults in Voltage Source Active Rectifiers. In Proceedings of the 2004 IEEE 35th Annual Power Electronics Specialists Conference, Aachen, Germany, 20–25 June 2004; pp. 4348–4354. [Google Scholar]
- Wan, S.W.S.; Nguyen, H.T. 50 Hz interference and noise in ECG recordings—A review. Australas. Phys. Eng. Sci. Med. 1994, 17, 108–115. [Google Scholar]
- Fernandez, M.; Pallas-Areny, R. Electrode Contact Noise in Surface Biopotential Measurements. In Proceedings of the 14th Annual International Conference of the IEEE in Medicine and Biology Society, Paris, France, 29 October–1 November 1992; pp. 123–124. [Google Scholar]
- Sparkfun. AD8232 Heart Rate Monitor Hookup Guide. Available online: https://learn.sparkfun.com/tutorials/ad8232-heart-rate-monitor-hookup-guide/all (accessed on 7 January 2022).
- Prasad, A.S.; Kavanashree, N. ECG Monitoring System Using AD8232 Sensor. In Proceedings of the 2019 International Conference on Communication and Electronics Systems (ICCES), Coimbatore, India, 17–19 July 2019; pp. 976–980. [Google Scholar]
- Wu, T.Y.; Redouté, J.-M.; Yuce, M. AWearable, Low-Power, Real-Time ECG Monitor for Smart T-shirt and IoT Healthcare Applications. In Advances in Body Area Networks I; Fortino, H., Wang, Z., Eds.; Internet of Things; Springer: Cham, Switzerland, 2019; pp. 165–173. [Google Scholar]
- Simanjuntak, J.E.S.; Khodra, M.L.; Manullang, M.C.T. Design Methods of Detecting Atrial Fibrillation Using the Recurrent Neural Network Algorithm on the Arduino AD8232 ECG Module. IOP Conf. Ser. Earth Environ. Sci. 2019, 537, 012022. [Google Scholar] [CrossRef]
- Malek, A.S.; Elnahrawy, A.; Anwar, H.; Naeem, M. From fabric to smart T-shirt: Fine tuning an improved robust system to detect arrhythmia. Textile Res. J. 2021. online first. [Google Scholar] [CrossRef]
- Analog Devices. Single-Lead, Heart Rate Monitor Front End AD8232 Data Sheet. Available online: https://www.analog.com/media/en/technical-documentation/data-sheets/ad8232.pdf (accessed on 7 January 2022).
- Sparkfun/AD8232_Heart_Rate_Monitor. Available online: https://github.com/sparkfun/AD8232_Heart_Rate_Monitor (accessed on 7 January 2022).
- Meding, J.T.; Tuvshinbayar, K.; Döpke, C.; Tamoue, F. Textile electrodes for bioimpedance measuring. Commun. Dev. Assem. Text. Prod. 2021, 2, 49–60. [Google Scholar] [CrossRef]
- Graßmann, C.; Mann, M.; van Langenhove, L.; Schwarz-Pfeiffer, A. Textile Based Electrochromic Cells Prepared with PEDOT: PSS and Gelled Electrolyte. Sensors 2020, 20, 5691. [Google Scholar] [CrossRef]
- Hellert, C.; Wortmann, M.; Frese, N.; Grötsch, G.; Cornelißen, C.; Ehrmann, A. Adhesion of Electrospun Poly(acrylonitrile) Nanofibers on Conductive and Isolating Foil Substrates. Coatings 2021, 11, 249. [Google Scholar] [CrossRef]
- Popovic-Maneski, L.; Ivanovic, M.D.; Atanasoski, V.; Miletic, M.; Zdolsek, S.; Bojovic, B.; Hadzievski, L. Properties of different types of dry electrodes for wearable smart monitoring devices. Biomed. Eng. 2020, 65, 405–415. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tuvshinbayar, K.; Ehrmann, G.; Ehrmann, A. 50/60 Hz Power Grid Noise as a Skin Contact Measure of Textile ECG Electrodes. Textiles 2022, 2, 265-274. https://doi.org/10.3390/textiles2020014
Tuvshinbayar K, Ehrmann G, Ehrmann A. 50/60 Hz Power Grid Noise as a Skin Contact Measure of Textile ECG Electrodes. Textiles. 2022; 2(2):265-274. https://doi.org/10.3390/textiles2020014
Chicago/Turabian StyleTuvshinbayar, Khorolsuren, Guido Ehrmann, and Andrea Ehrmann. 2022. "50/60 Hz Power Grid Noise as a Skin Contact Measure of Textile ECG Electrodes" Textiles 2, no. 2: 265-274. https://doi.org/10.3390/textiles2020014
APA StyleTuvshinbayar, K., Ehrmann, G., & Ehrmann, A. (2022). 50/60 Hz Power Grid Noise as a Skin Contact Measure of Textile ECG Electrodes. Textiles, 2(2), 265-274. https://doi.org/10.3390/textiles2020014






