Insulin Resistance Develops Due to an Imbalance in the Synthesis of Cyclic AMP and the Natural Cyclic AMP Antagonist Prostaglandylinositol Cyclic Phosphate (Cyclic PIP)
Abstract
:1. Introduction
2. Cyclic AMP
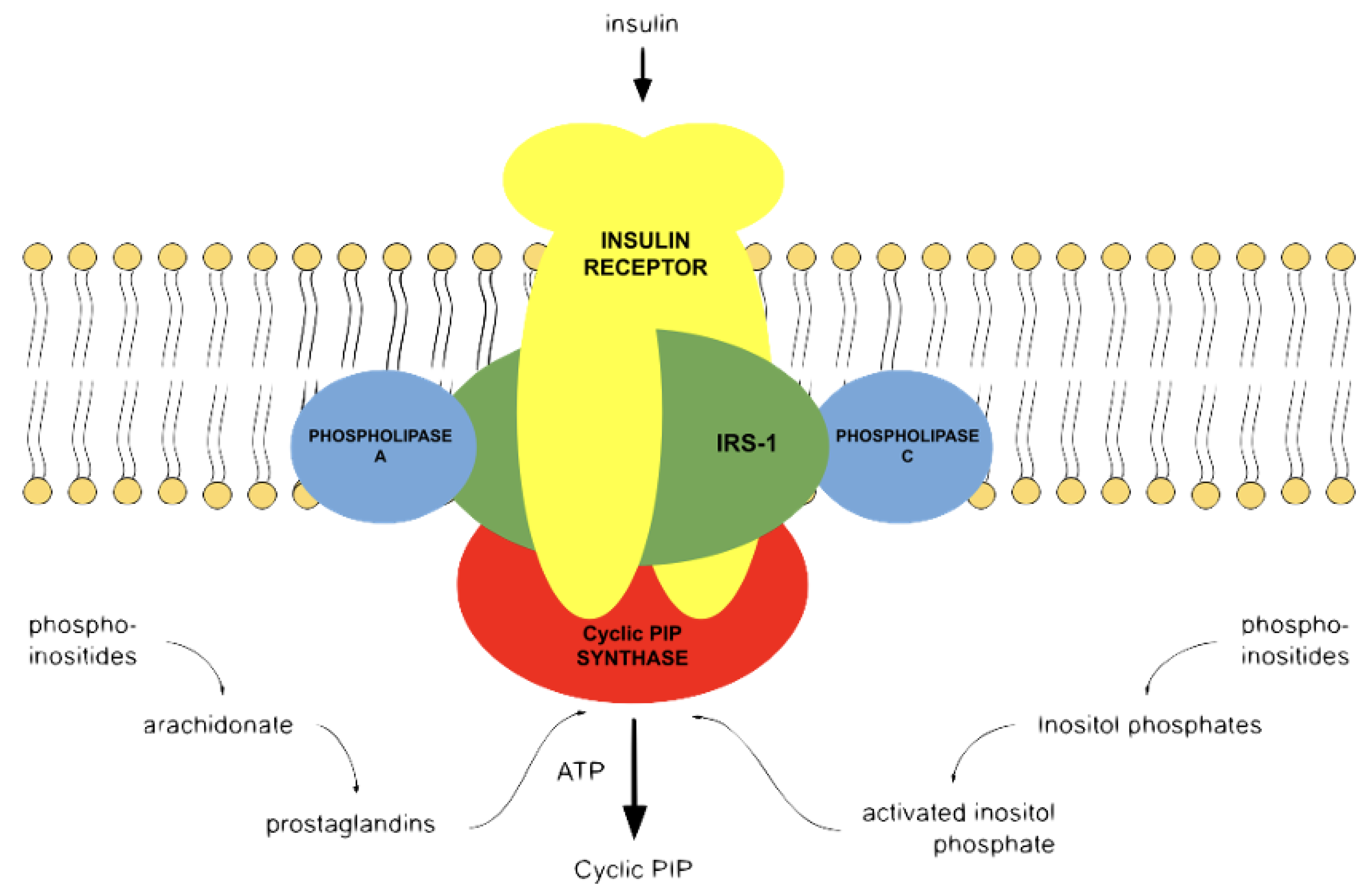
3. The Natural Cyclic AMP Antagonist Cyclic PIP
4. The Interplay of Cyclic AMP and Cyclic PIP
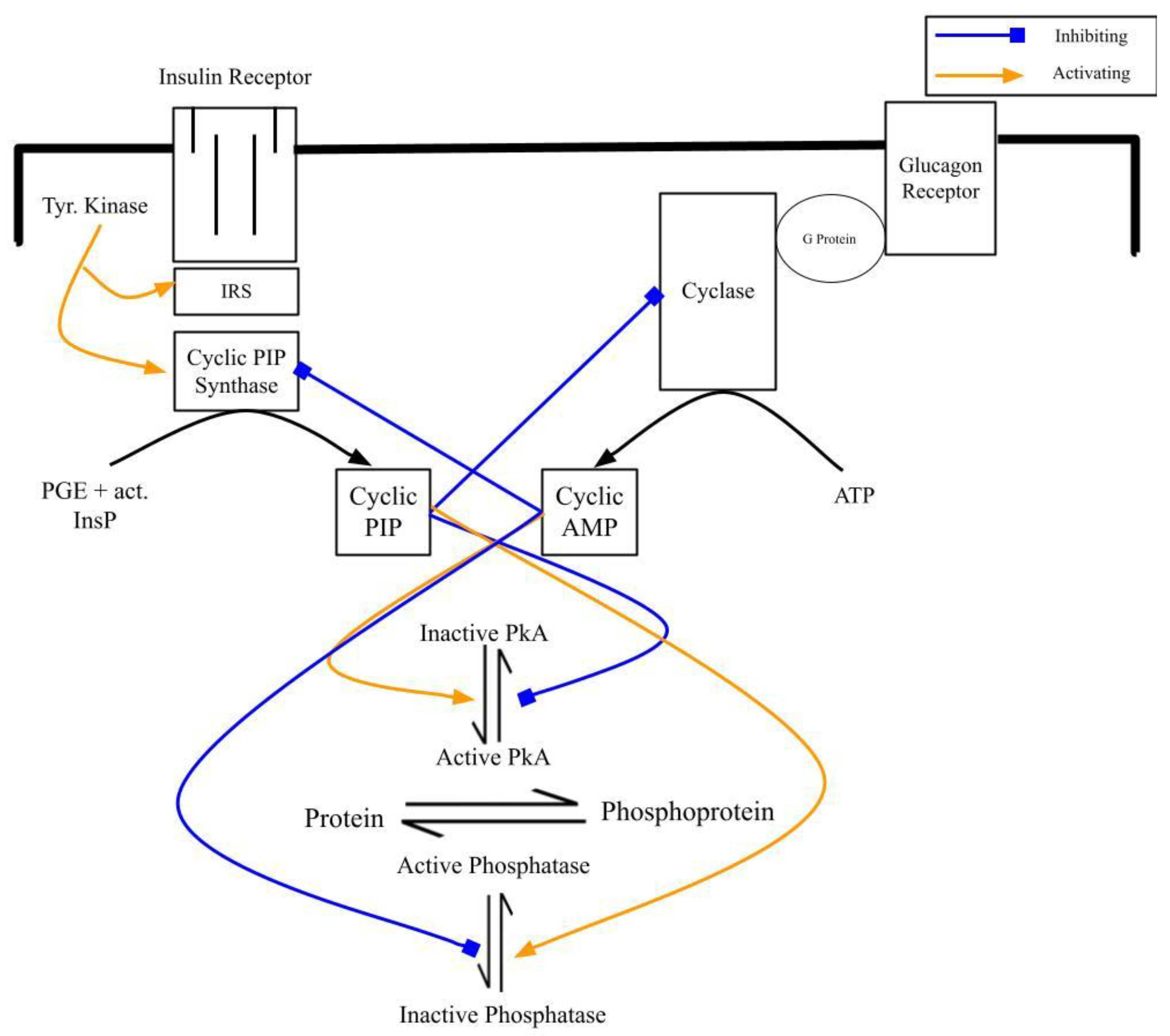
4.1. At the Level of Their Biosynthesis: Cyclic AMP Inhibits Cyclic PIP Synthase and Cyclic PIP Inhibits Adenylate Cyclase
4.2. At the Level of the Regulation of Metabolism: Increasing Cyclic AMP and Decreasing Cyclic PIP Synthesis Lead to Dominance of Catabolism over Anabolism
5. Illnesses Connected with Decreased Synthesis of Cyclic PIP
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nelson, M.E.; Madsen, S.; Cooke, K.C.; Fritzen, A.M.; Thorius, I.H.; Masson, S.W.C.; Carroll, L.; Weiss, F.C.; Seldin, M.M.; Potter, M.; et al. Systems-level analysis of insulin action in mouse strains provides insight into tissue- and pathway-specific interaction that drive insulin resistance. Cell Metab. 2022, 34, 227–239. [Google Scholar] [CrossRef]
- Mejhert, N.; Ryden, M. Understanding the complexity of insulin resistance. Nat. Rev. Endocrinol. 2022, 18, 269–270. [Google Scholar] [CrossRef]
- Petersen, M.C.; Shulman, G.I. Mechanisms of insulin action and insulin resistance. Physiol. Rev. 2018, 98, 2133–2223. [Google Scholar] [CrossRef]
- Galicia-Garcia, U.; Benito-Vicente, A.; Jebari, S.; Larrea-Sebal, A.; Siddiqi, H.; Uribe, K.B.; Ostolaza, H.; Martin, C. Pathophysiology of type 2 diabetes mellitus. Int. J. Mol. Sci. 2020, 21, 6275. [Google Scholar] [CrossRef]
- White, M.F.; Kahn, C.R. Insulin action at a molecular level—100 years of progress. Mol. Metab. 2021, 52, 101304. [Google Scholar] [CrossRef] [PubMed]
- Nagao, H.; Cai, W.; Brandao, B.B.; Wever Albrechtsen, N.J.; Steger, M.; Gattu, A.K.; Pan, H.; Dreyfuss, J.M.; Wunderlich, T.; Mann, M.; et al. Leucine-973 is a crucial residue differentiating insulin and IGF-1 receptor signaling. J. Clin. Invest. 2023, 133, E161472. [Google Scholar] [CrossRef]
- Robison, G.A.; Butcher, R.W.; Sutherland, E.W. Cyclic AMP; Academic Press: New York, NY, USA, 1971. [Google Scholar]
- Wasner, H.K. Prostaglandylinositol cyclic phosphate, the natural antagonist of cyclic AMP. IUBMB Life 2020, 72, 2282–2289. [Google Scholar] [CrossRef] [PubMed]
- Wasner, H.K. Metformin’s mechanism of action is stimulation of the biosynthesis of the natural cyclic AMP antagonist prostaglandylinositol cyclic phosphate (cyclic PIP). Int. J. Mol. Sci. 2022, 23, 2200. [Google Scholar] [CrossRef] [PubMed]
- Yarwood, S.J. Special issue on “new advances in cyclic AMP signaling”—An editorial overview. Cells 2020, 9, 2274. [Google Scholar] [CrossRef] [PubMed]
- von Keulen, S.C.; Rothlisberger, U. Exploring the inhibition mechanism of adenylyl cyclase type 5 by n-terminal myristoylated Gai1. PLoS. Comput. Biol. 2017, 13, e1005673. [Google Scholar] [CrossRef]
- Sabol, S.L.; Nirenberg, M. Regulation of adenylate cyclase of neuroblastoma x glioma hybrid cells by a-adrenergic receptors. J. Biol. Chem. 1979, 254, 1913–1920. [Google Scholar] [CrossRef] [PubMed]
- Wasner, H.K.; Gebel, M.; Hucken, S.; Schaefer, M.; Kincses, M. Two different mechanisms for activation of cyclic PIP synthase: By a G protein or by protein tyrosine phosphorylation. Biol. Chem. 2000, 382, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Wasner, H.K.; Salge, U.; Gebel, M. The endogenous cyclic AMP antagonist, cyclic PIP: Its ubiquity, hormone-stimulated synthesis and identification as prostaglandylinositol cyclic phosphate. Acta Diabetol. 1993, 30, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Wasner, H.K.; Lessmann, M.; Conrad, M.; Amini, H.; Psarakis, E.; Mir-Mohammad-Sadegh, A. Biosynthesis of the endogenous cyclic adenosine monophosphate (AMP) antagonist, prostaglandylinositol cyclic phosphate (cyclic PIP), from prostaglandin E and activated inositol polyphosphate in rat liver plasma membranes. Acta Diabetol. 1996, 33, 126–138. [Google Scholar] [CrossRef]
- Wasner, H.K.; Salge, U. Prostaglandylinositol cyclic phosphate, a second messenger for insulin. Excerpta Medica Int. Congr. Ser. 1987, 726, 226–231. [Google Scholar]
- Brautigan, D.L. Protein ser/thr phosphatases—The ugly ducklings of cell signaling. FEBS J. 2013, 280, 324–345. [Google Scholar] [CrossRef]
- Wasner, H.K.; Salge, U.; Psarakis, E.; Niktopoulos, A. Insulin resistance, a result of reduced synthesis of prostaglandylinositol cyclic phosphate, a mediator of insulin action? Regulation of cyclic PIP synthase activity by oral antidiabetic and antihypertensive drugs. Acta Diabetol. 1997, 34, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Weber, S.; Lemoine, H.; Wasner, H.K. Prostaglandin deficiency promotes sensitization of adenylyl cyclase. Biol. Chem. 2000, 381, 525–529. [Google Scholar] [CrossRef]
- Kalogriopoulos, N.A.; Lopez-Sanchez, I.; Lin, C.; Ngo, T.; Midde, K.K.; Roy, S.; Aznar, N.; Murray, F.; Garcia-Marcos, M.; Kufareva, I.; et al. Receptor tyrosine kinases activate heterotrimeric G proteins via phosphorylation within the interdomain cleft of Gai. Proc. Natl. Acad. Sci. USA 2020, 117, 28763–28774. [Google Scholar] [CrossRef]
- Wasner, H.K.; Weber, S.; Partke, H.J.; Amini-Hadi-Kiashar, H. Indomethacin treatment causes loss of insulin action in rats: Involvement of prostaglandins in the mechanism of insulin action. Acta Diabetol. 1994, 31, 175–182. [Google Scholar] [CrossRef]
- Wasner, H.; Lemoine, H.; Junger, E.; Lessmann, M.; Kaufmann, R. Prostaglandyl-inositol cyclic phosphate, a new second messenger. In Prostaglandins, Leukotrienes, Lipoxins and PAF; Bailey, J.M., Ed.; Plenum Press: New York, NY, USA, 1991; pp. 153–168. [Google Scholar]
- Salge, U. Untersuchungen zur Biologischen Synthese des Cyclo AMP-Antagonisten. Ph.D. Thesis, Heinrich-Heine-University Duesseldorf, Duesseldorf, Germany, 1987. [Google Scholar]
- Gundula, N.K.V.; Naidu, V.G.M.; Das, U.N. Amelioration of streptozotocin-induced type 2 diabetes mellitus in Wistar rats by arachidonic acid. Biochem. Biophys. Res. Commun. 2018, 496, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Hobday, A.L.; Parmar, M.S. The link between diabetes mellitus and tau hyperphosphorylation: Implications for risk of Alzheimer’s disease. Cureus 2021, 13, e18362. [Google Scholar] [CrossRef]
- Gypakis, A.; Wasner, H.K. Phosphatidyl 3-kinase and prostaglandylinositol cyclic phosphate (cyclic PIP), a mediator of insulin action, in the signal transduction of insulin. Biol. Chem. 2000, 381, 1139–1141. [Google Scholar] [CrossRef] [PubMed]
- Gratuze, M.; Julien, J.; Petry, F.R.; Morin, F.; Planel, E. Insulin deprivation induces PP2A inhibition and tau hyperphosphorylation in htau mice, a model of Alzheimer’s disease-like tau pathology. Sci. Rep. 2017, 7, 46359. [Google Scholar] [CrossRef]
- Batista, T.M.; Jayavelu, A.K.; Wewer Albrechtsen, N.J.; Iovino, S.; Lebastchi, J.; Pan, H.; Dreyfuss, J.M.; Krook, A.; Zierath, J.R.; Mann, M.; et al. A cell-autonomous signature of dysregulated protein phosphorylation underlies muscle insulin resistance in type 2 diabetes. Cell Metab. 2020, 32, 844–859. [Google Scholar] [CrossRef]
- James, D.E.; Stöckli, J.; Birnbaum, M.J. The aetiology and molecular landscape of insulin resistance. Nat. Rev. Mol. Cell Biol. 2018, 22, 751–771. [Google Scholar] [CrossRef]
- Suiso, A.; Glaser, B.; Dor, Y. Metabolic stress and compromised identity of pancreatic beta cells. Front. Genet. 2017, 8, 21. [Google Scholar] [CrossRef]
- Michell, R.B. Do inositol supplements enhance phosphatidylinositol supply and thus support endoplasmic reticulum function? Br. J. Nutr. 2018, 120, 301–316. [Google Scholar] [CrossRef] [PubMed]
- Lepore, E.; Lauretta, R.; Bianchini, M.; Mormando, M.; Di Lorenzo, C.; Unfer, V. Inositols depletion and resistance: Principal mechanisms and therapeutic strategies. Int. J. Mol. Sci. 2021, 22, 6796. [Google Scholar] [CrossRef] [PubMed]
- Wong, H.; Singh, J.; Go, R.M.; Ahluwalia, N.; Guerrero-Go, M.A. The effect of mental stress on non-insulin-dependent diabetes: Determining the relationship between catecholamine and adrenergic signals from stress, anxiety, and depression on the physiological changes in the pancreatic hormone secretion. Cureus 2019, 11, e5474. [Google Scholar] [CrossRef]
- Reich, K.M.; Gay, C.V.; Frangos, J.A. Fluid shear stress as a mediator of osteoblast cyclic adenosine monophosphate production. J. Cell. Physiol. 1990, 143, 100–104. [Google Scholar] [CrossRef]
- Stone, E.A.; John, S.M. Stress-induced increase of extracellular levels of cyclic AMP in rat cortex. Brain Res. 1992, 597, 144–147. [Google Scholar] [CrossRef]
- Pataky, M.W.; Young, W.F.; Nair, K.S. Hormonal and metabolic changes of aging and the influence of lifestyle modifications. Mayo Clin. Proc. 2021, 96, 788–814. [Google Scholar] [CrossRef]
- Ortmeyer, H.K. Insulin decreases skeletal muscle cAMP-dependent protein kinase (PKA) activity in normal monkeys and increases PKA activity in insulin-resistant Rhesus monkeys. J. Basic Clin. Physiol. Pharmacol. 1997, 8, 223–236. [Google Scholar] [CrossRef]
- Eyster, C.A.; Matsuzaki, S.; Newhardt, M.F.; Giorgione, J.R.; Humphries, K.M. Diabetes induced decreases in PKA signaling in cardiomyocytes: The role of insulin. PLoS ONE 2020, 15, e0231806. [Google Scholar] [CrossRef] [PubMed]
- Bockus, L.B.; Humphries, K.M. cAMP-dependent protein kinase (PKA) signaling is impaired in diabetic heart. J. Biol. Chem. 2015, 290, 29250–29258. [Google Scholar] [CrossRef] [PubMed]
- London, E.; Bloyd, M.; Stratakis, C.A. PKA functions in metabolism and resistance to obesity: Lessons from mouse and human studies. J. Endocrinol. 2020, 246, R51–R64. [Google Scholar] [CrossRef] [PubMed]
| Cyclic PIP Synthesis (Units/g Wet Weight) | ||||
|---|---|---|---|---|
| Control Rats | Diabetic Rats | |||
| Organs | Basal | Insulin stim. | Basal | Insulin stim. |
| Brain | 1.7 | 9.3 | 3.6 | 5 |
| Heart | 4.7 | 15.1 | 3.2 | 4.9 |
| Intestine | 5.2 | 15.7 | n.d. | n.d. |
| Kidney | 4.6 | 12.8 | 7.7 | 9 |
| Liver | 4.1 | 8.7 | 7 | 5.5 |
| Lung | 4.6 | 7 | 5.7 | 6.7 |
| Muscle | n.d. | n.d. | 7 | 8.2 |
| Spleen | 1.5 | 19.7 | 5 | 6.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wasner, H.K. Insulin Resistance Develops Due to an Imbalance in the Synthesis of Cyclic AMP and the Natural Cyclic AMP Antagonist Prostaglandylinositol Cyclic Phosphate (Cyclic PIP). Stresses 2023, 3, 762-772. https://doi.org/10.3390/stresses3040052
Wasner HK. Insulin Resistance Develops Due to an Imbalance in the Synthesis of Cyclic AMP and the Natural Cyclic AMP Antagonist Prostaglandylinositol Cyclic Phosphate (Cyclic PIP). Stresses. 2023; 3(4):762-772. https://doi.org/10.3390/stresses3040052
Chicago/Turabian StyleWasner, Heinrich K. 2023. "Insulin Resistance Develops Due to an Imbalance in the Synthesis of Cyclic AMP and the Natural Cyclic AMP Antagonist Prostaglandylinositol Cyclic Phosphate (Cyclic PIP)" Stresses 3, no. 4: 762-772. https://doi.org/10.3390/stresses3040052
APA StyleWasner, H. K. (2023). Insulin Resistance Develops Due to an Imbalance in the Synthesis of Cyclic AMP and the Natural Cyclic AMP Antagonist Prostaglandylinositol Cyclic Phosphate (Cyclic PIP). Stresses, 3(4), 762-772. https://doi.org/10.3390/stresses3040052






